Adherence to Mediterranean Diet and Risk of Pancreatic Cancer: Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy and Data Source
2.2. Inclusion/Exclusion Criteria
2.3. Selection Process and Data Extraction
2.4. Strategy for Data Synthesis
2.5. Critical Appraisal
2.6. Statistical Analysis
2.7. Sensitivity and Subgroup Analysis
3. Results
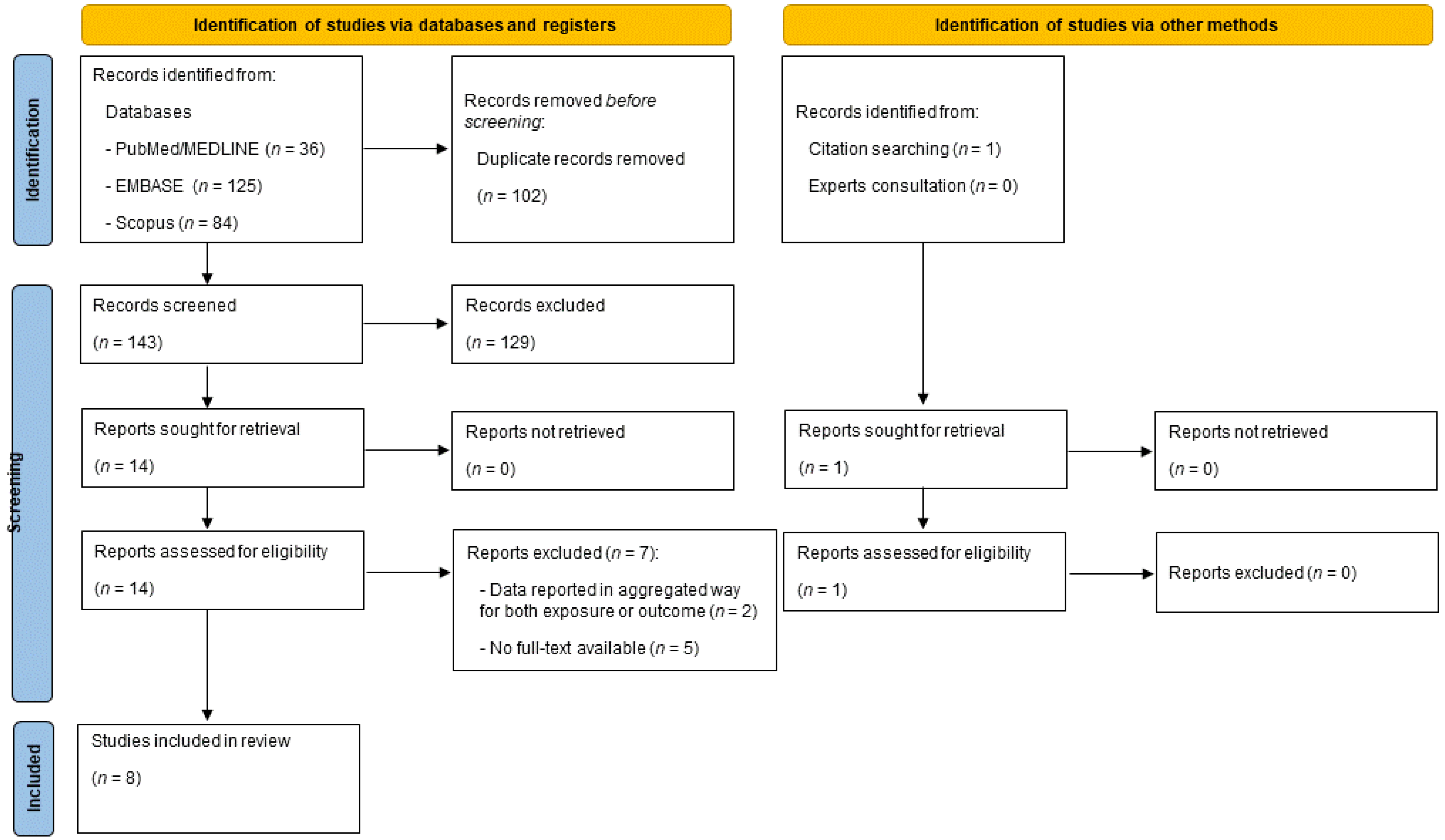
3.1. Literature Search
3.2. Main Characteristics of Included Articles
3.3. Characteristics of the Studied Populations
3.4. Mediterranean Diet and Scores Used to Assess Adherence Thereto
3.5. Critical Appraisal Results
3.6. Results of Meta-Analysis
3.7. Sensitivity Analyses
3.8. Subgroup Analyses
4. Discussion
4.1. Data Interpretation
4.2. Potential Biological Mechanisms
4.3. Strengths and Limitations
4.4. Implications for Public Health Policies and Practice
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lippi, G.; Mattiuzzi, C. The global burden of pancreatic cancer. Arch Med. Sci. 2020, 16, 820–824. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Said Abasse, K.; Essien, E.E.; Abbas, M.; Yu, X.; Xie, W.; Sun, J.; Akter, L.; Cote, A. Association between Dietary Nitrate, Nitrite Intake, and Site-Specific Cancer Risk: A Systematic Review and Meta-Analysis. Nutrients 2022, 14, 666. [Google Scholar] [CrossRef] [PubMed]
- Nucci, D.; Santangelo, O.E.; Provenzano, S.; Fatigoni, C.; Nardi, M.; Ferrara, P.; Gianfredi, V. Dietary Fiber Intake and Risk of Pancreatic Cancer: Systematic Review and Meta-Analysis of Observational Studies. Int. J. Environ. Res. Public Health 2021, 18, 11556. [Google Scholar] [CrossRef] [PubMed]
- Casari, I.; Falasca, M. Diet and Pancreatic Cancer Prevention. Cancers 2015, 7, 2309–2317. [Google Scholar] [CrossRef]
- Wilson, L.F.; Antonsson, A.; Green, A.C.; Jordan, S.J.; Kendall, B.J.; Nagle, C.M.; Neale, R.E.; Olsen, C.M.; Webb, P.M.; Whiteman, D.C. How many cancer cases and deaths are potentially preventable? Estimates for Australia in 2013. Int. J. Cancer 2018, 142, 691–701. [Google Scholar] [CrossRef]
- Debras, C.; Chazelas, E.; Srour, B.; Druesne-Pecollo, N.; Esseddik, Y.; Szabo de Edelenyi, F.; Agaesse, C.; De Sa, A.; Lutchia, R.; Gigandet, S.; et al. Artificial sweeteners and cancer risk: Results from the NutriNet-Sante population-based cohort study. PLoS Med. 2022, 19, e1003950. [Google Scholar] [CrossRef]
- Bae, J.M.; Shim, S.R. Coffee Consumption and Pancreatic Cancer Risk: A Meta-Epidemiological Study of Population-based Cohort Studies. Asian Pac. J. Cancer Prev. 2020, 21, 2793–2798. [Google Scholar] [CrossRef]
- Bao, Y.; Michaud, D.S.; Spiegelman, D.; Albanes, D.; Anderson, K.E.; Bernstein, L.; van den Brandt, P.A.; English, D.R.; Freudenheim, J.L.; Fuchs, C.S.; et al. Folate intake and risk of pancreatic cancer: Pooled analysis of prospective cohort studies. J. Natl. Cancer Inst. 2011, 103, 1840–1850. [Google Scholar] [CrossRef]
- Huang, X.; Gao, Y.; Zhi, X.; Ta, N.; Jiang, H.; Zheng, J. Association between vitamin A, retinol and carotenoid intake and pancreatic cancer risk: Evidence from epidemiologic studies. Sci. Rep. 2016, 6, 38936. [Google Scholar] [CrossRef]
- Gianfredi, V.; Ferrara, P.; Dinu, M.; Nardi, M.; Nucci, D. Diets, Dietary Patterns, Single Foods and Pancreatic Cancer Risk: An Umbrella Review of Meta-Analyses. Int. J. Environ. Res. Public Health 2022, 19, 14787. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Martinez-Gonzalez, M.A.; Tong, T.Y.; Forouhi, N.G.; Khandelwal, S.; Prabhakaran, D.; Mozaffarian, D.; de Lorgeril, M. Definitions and potential health benefits of the Mediterranean diet: Views from experts around the world. BMC Med. 2014, 12, 112. [Google Scholar] [CrossRef]
- Dinu, M.; Pagliai, G.; Angelino, D.; Rosi, A.; Dall’Asta, M.; Bresciani, L.; Ferraris, C.; Guglielmetti, M.; Godos, J.; Del Bo, C.; et al. Effects of Popular Diets on Anthropometric and Cardiometabolic Parameters: An Umbrella Review of Meta-Analyses of Randomized Controlled Trials. Adv. Nutr. 2020, 11, 815–833. [Google Scholar] [CrossRef]
- Dinu, M.; Pagliai, G.; Casini, A.; Sofi, F. Mediterranean diet and multiple health outcomes: An umbrella review of meta-analyses of observational studies and randomised trials. Eur. J. Clin. Nutr. 2018, 72, 30–43. [Google Scholar] [CrossRef]
- Higgins, J.P.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Gianfredi, V.; Nucci, D.; Vannini, S.; Villarini, M.; Moretti, M. In vitro Biological Effects of Sulforaphane (SFN), Epigallocatechin-3-gallate (EGCG), and Curcumin on Breast Cancer Cells: A Systematic Review of the Literature. Nutr. Cancer 2017, 69, 969–978. [Google Scholar] [CrossRef]
- Provenzano, S.; Gentile, L.; Cuda, A.; Santangelo, O.E.; Dallagiacoma, G.; Gianfredi, V. Is there an association between self-reported vaccination knowledge and the immunization behaviour of Health Sciences students. Ann Ig. 2020, 32, 223–233. [Google Scholar] [CrossRef]
- Nucci, D.; Fatigoni, C.; Amerio, A.; Odone, A.; Gianfredi, V. Red and Processed Meat Consumption and Risk of Depression: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2020, 17, 6686. [Google Scholar] [CrossRef]
- Firenze, A.; Santangelo, O.E.; Gianfredi, V.; Alagna, E.; Cedrone, F.; Provenzano, S.; La Torre, G. Violence on doctors. An observational study in Northern Italy. Med. Lav. 2020, 111, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.A.; Shea, B.; O’Connell, D.; Paterson, J.; Welch, V.; Losos, M.P.T. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 18 November 2022).
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Duval, S.; Tweedie, R. A nonparametric “Trim and Fill” method of accounting for Publication Bias in Meta-Analysis. J. Am. Stat. Assoc. 2000, 95, 89–98. [Google Scholar] [CrossRef]
- Nucci, D.; Fatigoni, C.; Salvatori, T.; Nardi, M.; Realdon, S.; Gianfredi, V. Association between Dietary Fibre Intake and Colorectal Adenoma: A Systematic Review and Meta-Analysis. Int J. Environ. Res. Public Health 2021, 18, 4168. [Google Scholar] [CrossRef]
- Steel, H.; Park, S.Y.; Lim, T.; Stram, D.; Marchand, L.L.; Wu, A.H.; Setiawan, V.W. Diet quality and pancreatic cancer incidence in the Multiethnic Cohort. Cancer Res. 2022, 82, 5870. [Google Scholar] [CrossRef]
- Julián-Serrano, S.; Reedy, J.; Robien, K.; Stolzenberg-Solomon, R. Index-based dietary patterns and risk of pancreatic ductal adenocarcinoma: Results from the NIH-AARP Diet and Health Study. Cancer Res. 2021, 81, 852. [Google Scholar] [CrossRef]
- Luu, H.N.; Paragomi, P.; Jin, A.; Wang, R.; Neelakantan, N.; Van Dam, R.M.; Brand, R.E.; Koh, W.P.; Yuan, J.M. Dietary quality indexes and risk of pancreatic cancer: Findings from a prospective cohort study. Cancer Res. 2021, 81, 854. [Google Scholar] [CrossRef]
- Luu, H.N.; Paragomi, P.; Wang, R.; Jin, A.; Brand, R.; Koh, W.P.; Yuan, J.M. Composite Score of Healthy Lifestyle Factors And Risk Of Pancreatic Cancer In A Prospective Cohort Study. Gastroenterology 2021, 160, S474. [Google Scholar] [CrossRef]
- Schulpen, M.; Peeters, P.H.; Van Den Brandt, P.A. Mediterranean diet adherence and risk of pancreatic cancer: A pooled analysis of two Dutch cohorts (conference abstract). Br. J. Cancer 2018, 119, 33–34. [Google Scholar] [CrossRef]
- Jiao, L.; Mitrou, P.N.; Reedy, J.; Graubard, B.I.; Hollenbeck, A.R.; Schatzkin, A.; Stolzenberg-Solomon, R. A combined healthy lifestyle score and risk of pancreatic cancer in a large cohort study. Arch. Intern. Med. 2009, 169, 764–770. [Google Scholar] [CrossRef]
- Barrea, L.; Altieri, B.; Muscogiuri, G.; Laudisio, D.; Annunziata, G.; Colao, A.; Faggiano, A.; Savastano, S. Impact of Nutritional Status on Gastroenteropancreatic Neuroendocrine Tumors (GEP-NET) Aggressiveness. Nutrients 2018, 10, 1854. [Google Scholar] [CrossRef]
- Bodén, S.; Myte, R.; Wennberg, M.; Harlid, S.; Johansson, I.; Shivappa, N.; Hébert, J.R.; Van Guelpen, B.; Nilsson, L.M. The inflammatory potential of diet in determining cancer risk; A prospective investigation of two dietary pattern scores. PLoS ONE 2019, 14, e0214551. [Google Scholar] [CrossRef]
- Bosetti, C.; Turati, F.; Dal Pont, A.; Ferraroni, M.; Polesel, J.; Negri, E.; Serraino, D.; Talamini, R.; La Vecchia, C.; Zeegers, M.P. The role of Mediterranean diet on the risk of pancreatic cancer. Br. J. Cancer 2013, 109, 1360–1366. [Google Scholar] [CrossRef]
- Julián-Serrano, S.; Reedy, J.; Robien, K.; Stolzenberg-Solomon, R. Adherence to 5 Diet Quality Indices and Pancreatic Cancer Risk in a Large US Prospective Cohort. Am. J. Epidemiol. 2022, 191, 1584–1600. [Google Scholar] [CrossRef]
- Luu, H.N.; Paragomi, P.; Jin, A.; Wang, R.; Neelakantan, N.; van Dam, R.M.; Brand, R.E.; Koh, W.P.; Yuan, J.M. Quality Diet Index and Risk of Pancreatic Cancer: Findings from the Singapore Chinese Health Study. Cancer Epidemiol. Biomark. Prev. 2021, 30, 2068–2078. [Google Scholar] [CrossRef]
- Molina-Montes, E.; Sánchez, M.J.; Buckland, G.; Bueno-de-Mesquita, H.B.; Weiderpass, E.; Amiano, P.; Wark, P.A.; Kühn, T.; Katzke, V.; Huerta, J.M.; et al. Mediterranean diet and risk of pancreatic cancer in the European Prospective Investigation into Cancer and Nutrition cohort. Br. J. Cancer 2017, 116, 811–820. [Google Scholar] [CrossRef]
- Rosato, V.; Polesel, J.; Bosetti, C.; Serraino, D.; Negri, E.; La Vecchia, C. Population attributable risk for pancreatic cancer in Northern Italy. Pancreas 2015, 44, 216–220. [Google Scholar] [CrossRef]
- Schulpen, M.; Peeters, P.H.; van den Brandt, P.A. Mediterranean diet adherence and risk of pancreatic cancer: A pooled analysis of two Dutch cohorts. Int. J. Cancer 2019, 144, 1550–1560. [Google Scholar] [CrossRef]
- Tognon, G.; Nilsson, L.M.; Lissner, L.; Johansson, I.; Hallmans, G.; Lindahl, B.; Winkvist, A. The Mediterranean diet score and mortality are inversely associated in adults living in the subarctic region. J. Nutr. 2012, 142, 1547–1553. [Google Scholar] [CrossRef]
- Andersson, G.; Borgquist, S.; Jirstrom, K. Hormonal factors and pancreatic cancer risk in women: The Malmo Diet and Cancer Study. Int. J. Cancer 2018, 143, 52–62. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G. Adherence to Mediterranean diet and risk of cancer: A systematic review and meta-analysis of observational studies. Int. J. Cancer 2014, 135, 1884–1897. [Google Scholar] [CrossRef] [PubMed]
- Morze, J.; Danielewicz, A.; Przybylowicz, K.; Zeng, H.; Hoffmann, G.; Schwingshackl, L. An updated systematic review and meta-analysis on adherence to mediterranean diet and risk of cancer. Eur. J. Nutr. 2021, 60, 1561–1586. [Google Scholar] [CrossRef] [PubMed]
- La Vecchia, C. Association between Mediterranean dietary patterns and cancer risk. Nutr Rev. 2009, 67 (Suppl. S1), S126–S129. [Google Scholar] [CrossRef] [PubMed]
- Gianfredi, V.; Nucci, D.; Abalsamo, A.; Acito, M.; Villarini, M.; Moretti, M.; Realdon, S. Green Tea Consumption and Risk of Breast Cancer and Recurrence-A Systematic Review and Meta-Analysis of Observational Studies. Nutrients 2018, 10, 1886. [Google Scholar] [CrossRef] [PubMed]
- Kris-Etherton, P.M.; Hecker, K.D.; Bonanome, A.; Coval, S.M.; Binkoski, A.E.; Hilpert, K.F.; Griel, A.E.; Etherton, T.D. Bioactive compounds in foods: Their role in the prevention of cardiovascular disease and cancer. Am. J. Med. 2002, 113 (Suppl. S9B), 71S–88S. [Google Scholar] [CrossRef]
- Grosso, G.; Buscemi, S.; Galvano, F.; Mistretta, A.; Marventano, S.; La Vela, V.; Drago, F.; Gangi, S.; Basile, F.; Biondi, A. Mediterranean diet and cancer: Epidemiological evidence and mechanism of selected aspects. BMC Surg. 2013, 13 (Suppl. S2), S14. [Google Scholar] [CrossRef]
- Kaaks, R.; Bellati, C.; Venturelli, E.; Rinaldi, S.; Secreto, G.; Biessy, C.; Pala, V.; Sieri, S.; Berrino, F. Effects of dietary intervention on IGF-I and IGF-binding proteins, and related alterations in sex steroid metabolism: The Diet and Androgens (DIANA) Randomised Trial. Eur. J. Clin. Nutr. 2003, 57, 1079–1088. [Google Scholar] [CrossRef]
- D’Eliseo, D.; Velotti, F. Omega-3 Fatty Acids and Can.ncer Cell Cytotoxicity: Implications for Multi-Targeted Cancer Therapy. J. Clin. Med. 2016, 5, 15. [Google Scholar] [CrossRef]
- Gianfredi, V.; Nucci, D.; Salvatori, T.; Dallagiacoma, G.; Fatigoni, C.; Moretti, M.; Realdon, S. Rectal Cancer: 20% Risk Reduction Thanks to Dietary Fibre Intake. Systematic Review and Meta-Analysis. Nutrients 2019, 11, 1579. [Google Scholar] [CrossRef]
- Gianfredi, V.; Salvatori, T.; Villarini, M.; Moretti, M.; Nucci, D.; Realdon, S. Is dietary fibre truly protective against colon cancer? A systematic review and meta-analysis. Int. J. Food Sci. Nutr. 2018, 69, 904–915. [Google Scholar] [CrossRef]
- Diet, Nutrition, Physical Activity and Colorectal Cancer; Continuos Update Project Expert Report 2018; World Cancer Research Fund: London, UK; American Institute for Cancer Research: Washington, DC, USA, 2018.
- Bultman, S.J. The microbiome and its potential as a cancer preventive intervention. Semin Oncol 2016, 43, 97–106. [Google Scholar] [CrossRef]
- Williams, M.T.; Hord, N.G. The role of dietary factors in cancer prevention: Beyond fruits and vegetables. Nutr. Clin. Pract. 2005, 20, 451–459. [Google Scholar] [CrossRef]
- Almiron-Roig, E.; Solis-Trapala, I.; Dodd, J.; Jebb, S.A. Estimating food portions. Influence of unit number, meal type and energy density. Appetite 2013, 71, 95–103. [Google Scholar] [CrossRef]
- Gianfredi, V.; Koster, A.; Odone, A.; Amerio, A.; Signorelli, C.; Schaper, N.C.; Bosma, H.; Kohler, S.; Dagnelie, P.C.; Stehouwer, C.D.A.; et al. Associations of Dietary Patterns with Incident Depression: The Maastricht Study. Nutrients 2021, 13, 1034. [Google Scholar] [CrossRef]
- World Health Organization. Gender Mainstreaming for Health Managers: A Practical Approach; World Health organization: Geneve, Switzerland, 2011. [Google Scholar]
- Jaeschke, L.; Steinbrecher, A.; Luzak, A.; Puggina, A.; Aleksovska, K.; Buck, C.; Burns, C.; Cardon, G.; Carlin, A.; Chantal, S.; et al. Socio-cultural determinants of physical activity across the life course: A Determinants of Diet and Physical Activity (DEDIPAC) umbrella systematic literature review. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 173. [Google Scholar] [CrossRef]
- Gianfredi, V.; Bertarelli, G.; Minelli, L.; Nucci, D. Promoting healthy eating in childhood: Results from the COcONUT (Children PrOmOting Nutrition throUght Theatre) project. Minerva Pediatr. 2021, in press. [Google Scholar] [CrossRef]
- Gianfredi, V.; Buffoli, M.; Rebecchi, A.; Croci, R.; Oradini-Alacreu, A.; Stirparo, G.; Marino, A.; Odone, A.; Capolongo, S.; Signorelli, C. Association between Urban Greenspace and Health: A Systematic Review of Literature. Int. J. Environ. Res. Public Health 2021, 18, 5137. [Google Scholar] [CrossRef]
- Hawkes, C.; Jewell, J.; Allen, K. A food policy package for healthy diets and the prevention of obesity and diet-related non-communicable diseases: The NOURISHING framework. Obes Rev. 2013, 14 (Suppl. S2), 159–168. [Google Scholar] [CrossRef]
- World Cancer Research Fund International and American Institute for Cancer Research. Diet, Nutrition, Physical Activity and Cancer: A Global Perspective; Continuous Update Project Expert, Report; International WCRF, Ed.; World Cancer Research Fund International: London, UK, 2018. [Google Scholar]

| Database | Syntax |
|---|---|
| PubMed/MEDLINE | (((“tumor*” [Title/Abstract] OR “tumour*” [Title/Abstract] OR “cancer*” [Title/Abstract] OR “neoplasm*” [Title/Abstract] OR “malignanc*” [Title/Abstract] OR “neoplastic” [Title/Abstract]) AND (“pancreas” [Title/Abstract] OR “pancreatic” [Title/Abstract])) OR “pancreatic neoplasms” [MeSH Terms]) AND (“diet, mediterranean” [MeSH Terms] OR (“Mediterranean” [Title/Abstract] AND (“diet” [Title/Abstract] OR “diets” [Title/Abstract] OR “dietary pattern” [Title/Abstract] OR “dietary patterns” [Title/Abstract] OR “dietary” [Title/Abstract] OR “dietary adherence” [Title/Abstract] OR “dietary score” [Title/Abstract] OR “dietary scores” [Title/Abstract] OR “dietary index” [Title/Abstract] OR “dietary intervention” [Title/Abstract] OR “dietary behaviour” [Title/Abstract] OR “dietary behavior” [Title/Abstract]))) |
| Scopus | ((TITLE-ABS-KEY (pancreatic OR pancreas) AND TITLE-ABS-KEY (neoplasm OR cancer OR tumor OR tumour OR malignanc*))) AND ((TITLE-ABS-KEY (mediterranean) AND TITLE-ABS-KEY (diet OR dietary OR pattern OR intervention OR behavior OR behaviour OR index OR score OR adherence))) |
| EMBASE | (‘mediterranean diet’/exp OR ((diet* OR dietary OR score* OR intervention* OR ‘behavior’/exp OR behavior OR ‘behaviour’/exp OR behaviour OR ‘index’/exp OR index OR ‘adherence’/exp OR adherence) AND (‘mediterranean’/exp OR mediterranean OR meddiet))) AND (‘pancreas cancer’/exp OR ((‘pancreas’/exp OR pancreas OR pancreatic) AND (neoplasm* OR cancer* OR malignan* OR tumor* OR tumour*))) |
| Search Strategy | Details |
|---|---|
| Inclusion criteria | P: adults ≥ 18 years (both female and male) I/E: higher adherence to Mediterranean diet C: lowest or no adherence to Mediterranean diet or adherence to other dietary patterns O: pancreatic cancer risk S: all original study types (both observational and trial-based) |
| Exclusion criteria | P: people < 18 years old I/E: other diets, dietary patterns, dietary supplementation, single food or food components or micro/macro-nutrients intake C: study assessing the effect of other diets, dietary patterns, dietary supplementation, single food or food components or micro/macro-nutrients intake O: other outcomes or data combined for pancreatic cancer with other gastrointestinal cancers. S: non-original papers, paper without data, articles not published as peer-reviewed in international journals |
| Language | English |
| Time filter | No filter (from inception) |
| Databases | PubMed/Medline; EMBASE, Scopus |
| Author, Year [Ref.] | Country | Study Period | Study Design | Population Characteristics | Tool Used for Dietary Assessment | Mediterranesn Score Used | Diagnostic Assessment | Outcome Definition | Cancer Type | Funds | CoI |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Bodèn, 2019 [33] | Sweden | 1990–2016; 15 y FU | CO | Male and female from the VIP | three versions of validated FFQs (84-items, 64-items and 66-items) | aMDS | Swedish Cancer Registry | Cases were defined based on ICD-10 codes as the first incident of primary malignancy via annual linkage | All types | yes | yes |
| Bosetti, 2013 [34] | Italy | 1983–1992 first study and 1992–2008 second study | CC | Cases: subjects with pancreatic cancer (without history of previous cancers) admitted to hospitals in the province of Milan Control: subjects admitted to the same network of hospitals as the cases for a wide spectrum of acute, non-neoplastic conditions Matching ratio 2:1, based on age, sex and study centre | structured questionnaire, simplified dietary section (14 selected indicator foods); and validated and reproducible food frequency questionnaire (78 items) | a priori MDS MDPI MAI | n.s. | Incident cases of pancreatic cancer newly admitted to the hospital | n.s. | yes | n.a. |
| Juliàn-Serrano, 2022 [35] | USA | 1995–2011; 15 y FU | CO | NIH–AARP | validated self-administered semiquantitative 124-item FFQ | aMED | Social Security Administration Death Master File | Cases were defined based on ICD-10 codes as the first incident of primary adenocarcinoma via annual linkage | excluding endocrine tumours, sarcomas and lymphomas | n.a. | no |
| Luu, 2021 [36] | Singapore | 1993–2015; 25 y FU | CO | Singapore Chinese Health Study | validated semiquantitative 165-FFQ | aMED | Singapore Cancer Registry and the Singapore Birth and Death Registry | Incident cases of pancreatic cancer were identified via annual linkage | excluding neuroendocrine pancreatic cancer | yes | no |
| Molina-Montes, 2017 [37] | 23 centres in 10 European countries# | From ‘90 s to 2004–2008 °; 11 y FU | CO | Male and female from the EPIC cohort | country-specific validated dietary questionnaires (FFQ, diet history, and semiquantitative FFQ) | arMED score | cancer registries and national mortality registries | Incident cancer cases identified via annual linkage | only including exocrine adenocarcinomas | n.a. | no |
| Rosato, 2015 [38] | Italy | 1992–2008; | CC | Cases: subjects with pancreatic cancer (without history of previous cancers) admitted to hospitals in the province of Milan and Pordenone Control: subjects admitted to the same network of hospitals as the cases for a wide spectrum of acute, non-neoplastic conditions Matching ratio 2:1, based on age, sex and study centre | 78-item FFQ | a priori MDS | Histological or cytological confirmation (179 patients), ultrasound and/or tomography. | Incident cases of pancreatic cancer newly admitted to the hospital | excluding endocrine pancreatic cancer | yes | no |
| Schulpen, 2018 [39] | The Netherlands | 1986–2006 NLCS and 1993–2014 EPIC-NL; 10 y FU | CO | NLCS and EPIC-NL | validated, self-administered, semiquantitative FFQs (number of items n.s.) | aMED modified MDS (for both scores a non-alcohol score was also estimated) | Netherlands Cancer Registry and nationwide Dutch Pathology Registry | Cases were defined based on ICD-10 codes as the first incident of primary malignancy via annual linkage | excluding endocrine pancreatic cancer | yes | no |
| Tognon, 2012 [40] | Sweden | 1990–2008;18 y FU | CO | VIP | three versions of FFQ (2 × 84-items and 65-items), only one validated | mMDS | Swedish national cause-of-death registry | Deaths were defined based on ICD-10 codes via record linkage | excluding in situ and benign PC | n.a. | no |
| Author, Year [Ref.] | Total Sample | Attrition * | Sex | Age: Mean and/or Range | MDS Categories | Effect Size (95% CI) | Adjustment | QS/9 |
|---|---|---|---|---|---|---|---|---|
| Bodèn, 2019 [33] | Ca: 223 Co: 100,881 | 63% | Ca: F = 107 Co: F = 51,001 | 40–60 y | Tertiles range n.s. | Total sample: HR = 0.90 (0.76–1.07) Male: HR = 1.01 (0.80–1.28) Female: HR = 0.80 (0.63–1.02) | EI, BMI, smoking, PA, and education | 8 |
| Bosetti, 2013 [34] | Ca: 688 Ct: 2204 | 5% | Ca: F = 285 Co: F = 715 | 56 (18–84) y | MDPI high ≥ 65.5 | Total sample: OR 0.44 (0.27–0.73) | centre, age, sex, year of interview, education, BMI, smoking, alcohol, history of T2D | 9 |
| MAI high ≥ 2.48 | Total sample OR 0.68 (0.42–1.11) | |||||||
| a priori MDS high ≥ 6 points | Total sample: OR = 0.48 (0.35–0.67) Male: OR = 0.84 (0.78–0.91) Female: OR = 0.87 (0.79–0.97) | |||||||
| Juliàn-Serrano, 2022 [35] | Ca: 3137 Co: 535,824 | 5.4% | Ca: F = n.a. Co: F = 220,044 | 50–71 y | Quintiles, Q1 = 2.5; Q5 = 7.4 | Total sample: HR = 0.82 (0.73–0.93) Male: HR = 0.97 (0.95–1.00) Female: HR = 0.76 (0.63–0.92) | age at baseline, sex ^, smoking, BMI, T2D, EI | 9 |
| Luu, 2021 [36] | Ca: 311 Co: 61,321 | 3% | Ca: F = 149 Co: F = n.a. | 45–74 y | Quintiles range n.s. | Total sample: HR = 0.57 (0.38–0.85) Male: HR = 0.43 (0.24–0.75) Female: HR = 0.79 (0.45–1.39) | age, sex ^, dialect, year of enrolment, education, smoking, smoking pack-years, coffee drinking, EI, BMI, T2D | 9 |
| Molina-Montes, 2017 [37] | Ca: 865 Co: 477,309 | 2% | Ca: F = 469 Co: F = 335,060 | 51.5 y | low (0–5 points), medium (6–9 points) high (10–16 points) | Total sample: HR = 0.99 (0.77–1.26) Male: HR = 1.00 (0.68–1.49) Female: HR = 0.99 (0.72–1.37) | EI, BMI, smoking status and intensity, alcohol, T2D | 9 |
| Rosato, 2015 [38] | Ca: 326 Ct: 652 | 0% | Ca: F = 152 Ct: F = 304 | 63 (34–80) y | low (≤3 points), medium (4–5 points) high (≥6 points) | Total sample: OR = 0.57 (0.34–0.95) | sex, age, study centre, education, BMI, smoking, alcohol, T2D, EI | 9 |
| Schulpen, 2018 [39] | Ca: 311 Co: 61,321 | 14% | Ca: F = 149 Co: F = 28,275 | 45–74 y | aMED: low (0–3 points), medium (4–5 points) high (6–9 points) | Male: HR = 0.70 (0.44–1.12) Female: HR = 1.18 (0.80–1.75) | age, smoking status, smoking frequency, smoking duration, BMI, EI, alcohol, T2D, family history of pancreatic cancer, education, nonoccupational PA | 9 |
| mMDS: low (0–3 points), medium (4–5 points) high (6–8 points) | Male: HR = 0.66 (0.40–1.10) Female: HR = 0.94 (0.63–1.40) | |||||||
| Tognon, 2012 [40] | Ca: 92 Co: 77,151 | Ca: F = 45 Co: F = 39,605 | 30–60 y | high > 4 points | Total sample: HR = 0.82 (0.72–0.94) Male: HR = 0.82 (0.68–0.99) Female: HR = 0.83 (0.69–1.00) | age, obesity, smoking status, education, PA | 9 |
| Scale Type | Food Items Included in the Scale | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vegetables 1 | Legumes | Fruit 2 | Cereals 3 | Fish 4 | Healthy 5 Fats | Alcohol | Meat and Meat Products | Dairy Products | Added Sugars | Score System | Score Range | Ref. | |
| aMDS | x | x | x | x | x | x | x | x | x | - | 1 point if consumption is above */below’ their sex and FFQ-specific median; for alcohol, 1 point if consumption < 50 g/day | 0–8 | [33] |
| A priori MDS | x | x | x | x | x | x | x | x | x | - | 1 point if consumption is above */below’ their sex and FFQ-specific median | 0–9 | [34,38] |
| MDPI | x | x | x | x | x | x | x | x | - | adding up the standardised residuals of the regression of cereals+ fruit+ vegetables+ legumes+ moderate alcohol+ MUFA/SFA ratio on total calories, and subtracting those of milk and meat | 0–100% | [34] | |
| MAI | x | x | x | x | x | x | x | x | x | x | dividing the sum of the intake of bread+ cereals+ fruit+ vegetables+ legumes+ potatoes+ fish+ red wine+ vegetable oils as a percentage of total energy by the sum of milk+ cheese+ meat+ eggs+ animal fats and margarines+ sweet beverages+ cakes+ pies+ cookies+ sugar | 0–44 | [34] |
| aMED | x | x | x | x | x | x | x | x | - | - | 1 point if consumption is above */below’ their study population’s specific median | 0–9 | [35,36] |
| arMED | x | x | x | x | x | x | - | x | x | - | A score from 0 to 2 is assigned according to the first, second or third quartile of consumption, respectively (for vegetables, fruits, legumes, fish and cereals); for olive oil, a maximum score of 2 is assigned if consumption is above the median; a score from 0 to 2 is assigned according to the third, second or first quartile of consumption, respectively (for dairy and meat products) | 0–16 | [37] |
| mMDS | x | - | x | x | x | x | x | x | x | - | 1 point if consumption is above */below’ their sex and FFQ-specific median; for alcohol 1 point if consumption < 50 g/day | 0–8 | [40] |
| non-alcohol mMDS | x | - | x | x | x | x | - | x | x | - | 1 point if consumption is above */below’ their sex and FFQ-specific median | 0–7 | [39] |
| non-alcohol aMED | x | x | x | x | x | x | - | x | - | - | 1 point if consumption is above */below’ their sex and FFQ-specific median | 0–8 | [39] |
| Summary Statistics | Publication Bias | ||||||
|---|---|---|---|---|---|---|---|
| Analysis | Studies Included [Ref.] | No. of Participants | df | HR (95% CI); p-Value | I2; p-Value | Intercept’; p-Value | Estimated a ES; p-Value |
| Overall analysis (both male and female) ^ | Boden (aMDS), Bosetti (a priori), Juliàn-Serrano (aMED), Luu (aMED), Molina-Montes (arMED), Rosato (a priori), Schulpen (aMED), Tognon (mMED) | 1,301,320 | 8 * | FE: 0.82 (0.76–0.88); p < 0.001 | 65.48%; p = 0.003 | −1.24, p = 0.331 | FE: 0.84 (0.79–0.90); p < 0.001 |
| RE: 0.78 (0.68–0.90); p = 0.001 | RE: 0.84 (0.73–0.97); p = 0.017 | ||||||
| Overall analysis (both male and female) $ | Boden (aMDS), Bosetti (a priori), Juliàn-Serrano (aMED), Luu (aMED), Molina-Montes (arMED), Rosato (a priori), Schulpen (mMED), Tognon (mMED) | 1,301,320 | 8 * | FE: 0.81 (0.76–0.87); p < 0.001 | 60.90%; p = 0.009 | −1.57; p = 0.171 | FE: 0.84 (0.78–0.90); p < 0.001 |
| RE: 0.77 (0.68–0.88); p < 0.001 | RE: 0.83 (0.71–0.96); p = 0.014 | ||||||
| Excluding mortality data | Boden (aMDS), Bosetti (a priori), Juliàn-Serrano (aMED), Luu (aMED), Molina-Montes (arMED), Rosato (a priori), Schulpen (aMED) | 1,224,007 | 7 * | FE: 0.81 (0.75–0.88); p < 0.001 | 69.79%; p = 0.002 | −1.36; p = 0.351 | FE: 0.81 (0.75–0.88); p < 0.001 |
| RE: 0.77 (0.64–0.92); p = 0.004 | RE: 0.86 (0.79–0.92); p < 0.001 | ||||||
| Excluding potential overlapping cohorts (both male and female) | Boden (aMDS), Bosetti (a priori), Juliàn-Serrano (aMED), Luu (aMED), Molina-Montes (arMED), Schulpen (aMED) | 1,223,009 | 6 * | FE: 0.82 (0.76–0.89); p < 0.001 | 70.94%; p = 0.002 | −1.04; p = 0.550 | FE: 0.87 (0.80–0.94); p < 0.001 |
| RE: 0.79 (0.66–0.95); p = 0.001 | RE: 0.80 (0.68–0.93); p < 0.004 | ||||||
| Only studies using validated FFQ | Juliàn-Serrano (aMED), Luu (aMED), Molina-Montes (arMED), Schulpen (aMED) | 1,119,103 | 4 * | FE: 0.84 (0.76–0.93); p = 0.001 | 55.10%; p = 0.063 | −0.04; p = 0.979 | FE: 0.86 (0.78–0.95); p = 0.002 |
| RE: 0.84 (0.70–1.02); p = 0.084 | RE: 0.90 (0.73–1.12); p = 0.342 | ||||||
| Diagnosis based on record linkage + | Boden (aMDS), Juliàn-Serrano (aMED), Luu (aMED), Molina-Montes (arMED), Schulpen (aMED) | 1,220,207 | 5 * | FE: 0.86 (0.78–0.93); p < 0.001 | 46.64%; p = 0.095 | −0.10; p = 0.947 | FE: 0.86 (0.78–0.93); p < 0.001 |
| RE: 0.86 (0.75–0.99); p = 0.033 | RE: 0.90 (0.77–1.05); p = 0.176 | ||||||
| Only studies conducted in Europe | Boden (aMDS), Bosetti (a priori), Molina-Montes (arMED), Schulpen (aMED) | 622,506 | 4 * | FE: 0.85 (0.76–0.96); p = 0.009 | 76.57%; p = 0.002 | −1.46; p = 0.652 | FE: 0.85 (0.76–0.96); p = 0.009 |
| RE: 0.82 (0.62–1.08); p= 0.152 | RE: 0.82 (0.62–1.08); p = 0.152 | ||||||
| Subgroup analyses | |||||||
| Including only male | Boden (aMDS), Bosetti (a priori), Juliàn-Serrano (aMED), Luu (aMED), Molina-Montes (arMED), Schulpen (aMED) | 585,430 | 5 | FE: 0.95 (0.93–0.98); p < 0.001 | 76.82%; p = 0.001 | −1.45; p = 0.206 | FE: 0.96 (0.93–0.98); p < 0.001 |
| RE: 0.89 (0.78–1.01); p = 0.061 | RE: 0.91 (0.80–1.05); p = 0.197 | ||||||
| Including only female | Boden (aMDS), Bosetti (a priori), Juliàn-Serrano (aMED), Luu (aMED), Molina-Montes (arMED), Schulpen (aMED) | 698,960 | 5 | FE: 0.86 (0.80–0.93); p < 0.001 | 14.94%; p = 0.318 | 0.41; p = 0.698 | FE: 0.86 (0.80–0.93); p < 0.001 |
| RE: 0.86 (0.79–0.95); p = 0.002 | RE: 0.86 (0.79–0.95); p = 0.002 | ||||||
| Cancer type (excluding endocrine PC) | Juliàn-Serrano (aMED), Luu (aMED), Rosato (a priori), Schulpen (aMED) | 641,907 | 4 * | FE: 0.80 (0.72–0.89); p < 0.001 | 56.68%; p = 0.056 | −0.85; p = 0.556 | FE: 0.81 (0.73–0.90); p < 0.001 |
| RE: 0.76 (0.60–0.96); p = 0.021 | RE: 0.80 (0.64–1.00); p = 0.050 | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nucci, D.; Nardi, M.; Cinnirella, A.; Campagnoli, E.; Maffeo, M.; Perrone, P.M.; Shishmintseva, V.; Grosso, F.M.; Castrofino, A.; Castaldi, S.; et al. Adherence to Mediterranean Diet and Risk of Pancreatic Cancer: Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2023, 20, 2403. https://doi.org/10.3390/ijerph20032403
Nucci D, Nardi M, Cinnirella A, Campagnoli E, Maffeo M, Perrone PM, Shishmintseva V, Grosso FM, Castrofino A, Castaldi S, et al. Adherence to Mediterranean Diet and Risk of Pancreatic Cancer: Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health. 2023; 20(3):2403. https://doi.org/10.3390/ijerph20032403
Chicago/Turabian StyleNucci, Daniele, Mariateresa Nardi, Andrea Cinnirella, Emanuela Campagnoli, Manuel Maffeo, Pier Mario Perrone, Viktoriia Shishmintseva, Francesca Maria Grosso, Ambra Castrofino, Silvana Castaldi, and et al. 2023. "Adherence to Mediterranean Diet and Risk of Pancreatic Cancer: Systematic Review and Meta-Analysis" International Journal of Environmental Research and Public Health 20, no. 3: 2403. https://doi.org/10.3390/ijerph20032403
APA StyleNucci, D., Nardi, M., Cinnirella, A., Campagnoli, E., Maffeo, M., Perrone, P. M., Shishmintseva, V., Grosso, F. M., Castrofino, A., Castaldi, S., Romanò, L., & Gianfredi, V. (2023). Adherence to Mediterranean Diet and Risk of Pancreatic Cancer: Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health, 20(3), 2403. https://doi.org/10.3390/ijerph20032403






