One-Year Follow-Up of Seroprevalence of SARS-CoV-2 Infection and Anxiety among Health Workers of a French Cancer Center: The PRO-SERO-COV Study
Abstract
1. Introduction
2. Materials and Methods
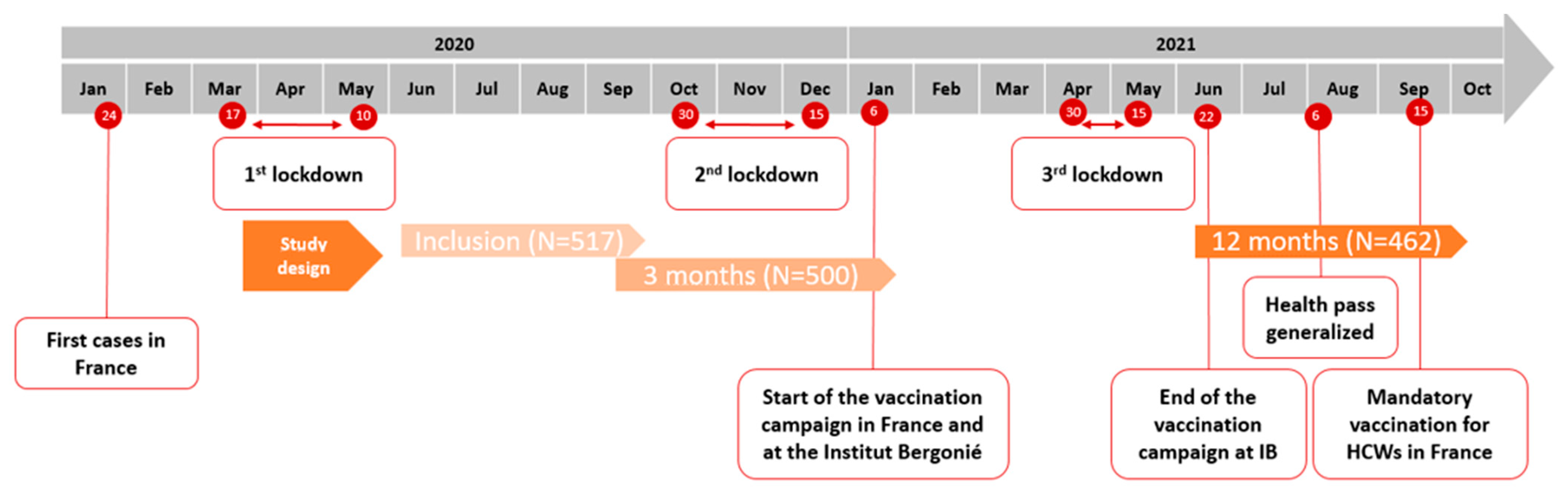
2.1. Study Design and Participants
2.2. Study Questionnaires
2.3. Serological Assay
2.4. Statistical Analysis
3. Results
3.1. Characteristics and Exposition to SARS-CoV-2
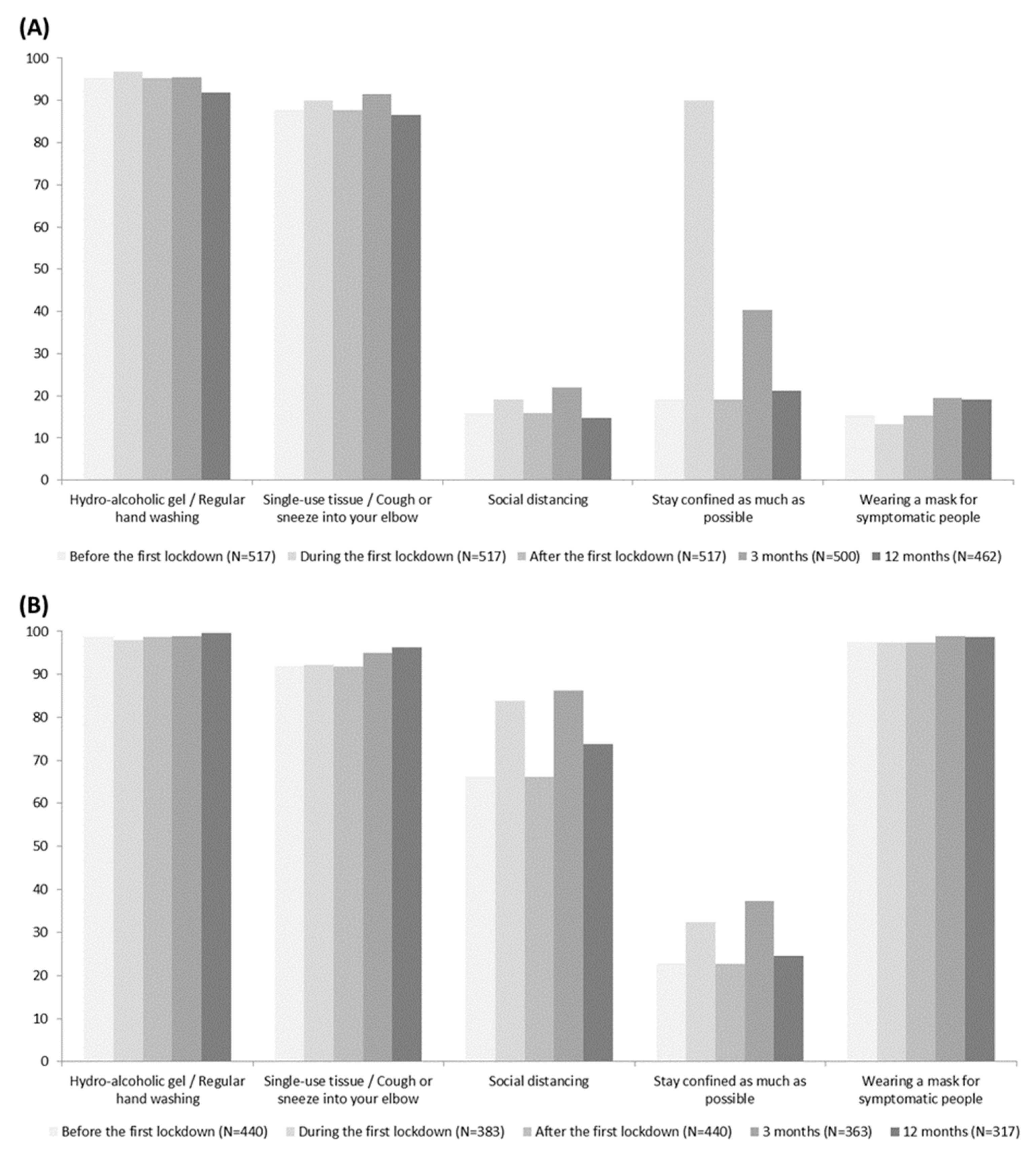
3.2. Barrier Gestures and Vaccine Hesitancy
3.3. Seroprevalence against SARS-CoV-2
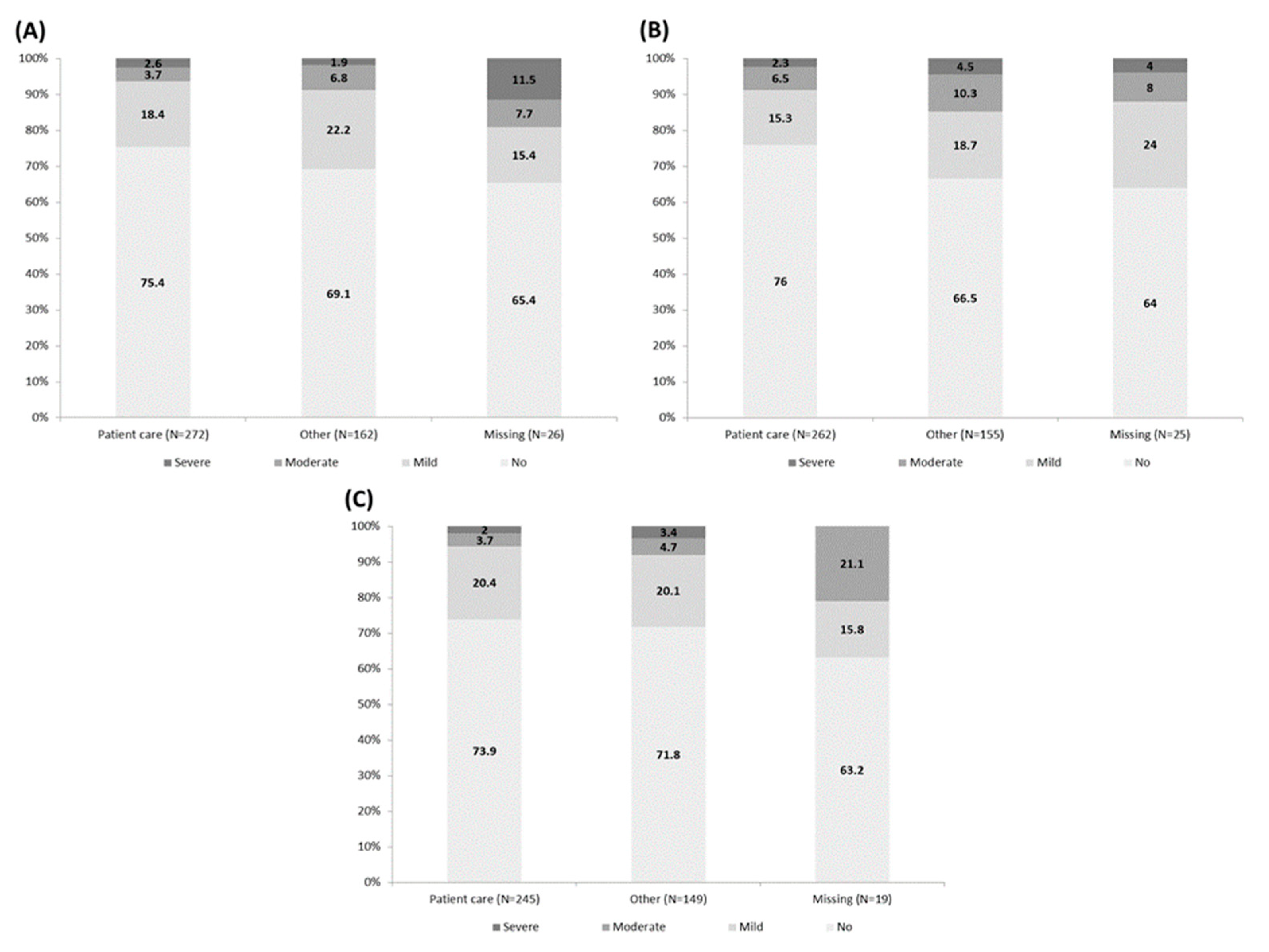
3.4. Anxiety
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lu, R.; Zhao, X.; Li, J.; Niu, P.; Yang, B.; Wu, H.; Wang, W.; Song, H.; Huang, B.; Zhu, N.; et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding. Lancet 2020, 395, 565–574. [Google Scholar] [CrossRef]
- WHO Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int (accessed on 3 May 2023).
- Ma, Q.; Liu, J.; Liu, Q.; Kang, L.; Liu, R.; Jing, W.; Wu, Y.; Liu, M. Global Percentage of Asymptomatic SARS-CoV-2 Infections Among the Tested Population and Individuals With Confirmed COVID-19 Diagnosis: A Systematic Review and Meta-analysis. JAMA Netw. Open 2021, 4, e2137257. [Google Scholar] [CrossRef]
- Guo, L.; Ren, L.; Yang, S.; Xiao, M.; Chang, D.; Yang, F.; Dela Cruz, C.S.; Wang, Y.; Wu, C.; Xiao, Y.; et al. Profiling Early Humoral Response to Diagnose Novel Coronavirus Disease (COVID-19). Clin. Infect. Dis. 2020, 71, 778–785. [Google Scholar] [CrossRef]
- Jiang, H.; Li, Y.; Zhang, H.; Wang, W.; Yang, X.; Qi, H.; Li, H.; Men, D.; Zhou, J.; Tao, S. SARS-CoV-2 proteome microarray for global profiling of COVID-19 specific IgG and IgM responses. Nat. Commun. 2020, 11, 3581. [Google Scholar] [CrossRef]
- Kayı, İ.; Madran, B.; Keske, Ş.; Karanfil, Ö.; Arribas, J.R.; Pshenichnaya, N.; Petrosillo, N.; Gönen, M.; Ergönül, Ö. The seroprevalence of SARS-CoV-2 antibodies among health care workers before the era of vaccination: A systematic review and meta-analysis. Clin. Microbiol. Infect. 2021, 27, 1242–1249. [Google Scholar] [CrossRef]
- Zhou, K.; Blanc-Lapierre, A.; Seegers, V.; Boisdron-Celle, M.; Bigot, F.; Bourdon, M.; Mahammedi, H.; Lambert, A.; Campone, M.; Conroy, T.; et al. Anosmia but Not Ageusia as a COVID-19-Related Symptom among Cancer Patients-First Results from the PAPESCO-19 Cohort Study. Cancers 2021, 13, 3389. [Google Scholar] [CrossRef]
- Fuereder, T.; Berghoff, A.S.; Heller, G.; Haslacher, H.; Perkmann, T.; Strassl, R.; Berger, J.M.; Puhr, H.C.; Kreminger, J.; Moik, F.; et al. SARS-CoV-2 seroprevalence in oncology healthcare professionals and patients with cancer at a tertiary care centre during the COVID-19 pandemic. ESMO Open 2020, 5, e000889. [Google Scholar] [CrossRef]
- Fong, D.; San Nicolò, K.O.; Alber, M.; Mitterer, M. Evaluating the longitudinal effectiveness of preventive measures against COVID-19 and seroprevalence of IgG antibodies to SARS-CoV-2 in cancer outpatients and healthcare workers. Wien. Klin. Wochenschr. 2021, 133, 359–363. [Google Scholar] [CrossRef]
- Silva, D.F.; Silva, P.D.; Torgal, A.; Braga, S.; Rocha, D.; Ochoa, C.; Oliveira, Á.; Rocha, L.; Dias, J.M.; Baldaque, I. Seroprevalence of Anti-SARS-CoV-2 Antibodies Three Months Post Infection in Healthcare Professionals at an Oncology Hospital in Northern Portugal. Acta Med. Port. 2021, 34, 484–485. [Google Scholar] [CrossRef]
- Favara, D.M.; McAdam, K.; Cooke, A.; Bordessa-Kelly, A.; Budriunaite, I.; Bossingham, S.; Houghton, S.; Doffinger, R.; Ainsworth, N.; Corrie, P.G. SARS-CoV-2 Infection and Antibody Seroprevalence among UK Healthcare Professionals Working with Cancer Patients during the First Wave of the COVID-19 Pandemic. Clin. Oncol. 2021, 33, 667–675. [Google Scholar] [CrossRef]
- Anna, F.; Goyard, S.; Lalanne, A.I.; Nevo, F.; Gransagne, M.; Souque, P.; Louis, D.; Gillon, V.; Turbiez, I.; Bidard, F.-C.; et al. High seroprevalence but short-lived immune response to SARS-CoV-2 infection in Paris. Eur. J. Immunol. 2021, 51, 180–190. [Google Scholar] [CrossRef]
- Ladoire, S.; Goussot, V.; Redersdorff, E.; Cueff, A.; Ballot, E.; Truntzer, C.; Ayati, S.; Bengrine-Lefevre, L.; Bremaud, N.; Coudert, B.; et al. Seroprevalence of SARS-CoV-2 among the staff and patients of a French cancer centre after first lockdown: The canSEROcov study. Eur. J. Cancer 2021, 148, 359–370. [Google Scholar] [CrossRef]
- Ladoire, S.; Rederstorff, E.; Goussot, V.; Parnalland, S.; Briot, N.; Ballot, E.; Truntzer, C.; Ayati, S.; Bengrine-Lefevre, L.; Bremaud, N.; et al. Parallel evolution and differences in seroprevalence of SARS-CoV-2 antibody between patients with cancer and health care workers in a tertiary cancer centre during the first and second wave of COVID-19 pandemic: canSEROcov-II cross-sectional study. Eur. J. Cancer 2022, 165, 13–24. [Google Scholar] [CrossRef]
- Van Dam, P.; Huizing, M.; Roelant, E.; Hotterbeekx, A.; De Winter, F.H.R.; Kumar-Singh, S.; Moons, P.; Amajoud, Z.; Vulsteke, C.; Croes, L.; et al. Immunoglobin G/total antibody testing for SARS-CoV-2: A prospective cohort study of ambulatory patients and health care workers in two Belgian oncology units comparing three commercial tests. Eur. J. Cancer 2021, 148, 328–339. [Google Scholar] [CrossRef]
- Panteli, D.; Reichebner, C.; Rombey, T.; Berger, E.; Winkelmann, J.; Eckhardt, H.; Nimptsch, U.; Busse, R. Health care patterns and policies in 18 European countries during the first wave of the COVID-19 pandemic: An observational study. Eur. J. Public Health 2022, 32, 557–564. [Google Scholar] [CrossRef]
- Brooks, S.K.; Webster, R.K.; Smith, L.E.; Woodland, L.; Wessely, S.; Greenberg, N.; Rubin, G.J. The psychological impact of quarantine and how to reduce it: Rapid review of the evidence. Lancet 2020, 395, 912–920. [Google Scholar] [CrossRef]
- Saeed, H.; Eslami, A.; Nassif, N.T.; Simpson, A.M.; Lal, S. Anxiety Linked to COVID-19: A Systematic Review Comparing Anxiety Rates in Different Populations. Int. J. Environ. Res. Public Health 2022, 19, 2189. [Google Scholar] [CrossRef]
- Al Maqbali, M.; Al Sinani, M.; Al-Lenjawi, B. Prevalence of stress, depression, anxiety and sleep disturbance among nurses during the COVID-19 pandemic: A systematic review and meta-analysis. J. Psychosom. Res. 2021, 141, 110343. [Google Scholar] [CrossRef]
- Marvaldi, M.; Mallet, J.; Dubertret, C.; Moro, M.R.; Guessoum, S.B. Anxiety, depression, trauma-related, and sleep disorders among healthcare workers during the COVID-19 pandemic: A systematic review and meta-analysis. Neurosci. Biobehav. Rev. 2021, 126, 252–264. [Google Scholar] [CrossRef]
- Cénat, J.M.; Blais-Rochette, C.; Kokou-Kpolou, C.K.; Noorishad, P.-G.; Mukunzi, J.N.; McIntee, S.-E.; Dalexis, R.D.; Goulet, M.-A.; Labelle, P.R. Prevalence of symptoms of depression, anxiety, insomnia, posttraumatic stress disorder, and psychological distress among populations affected by the COVID-19 pandemic: A systematic review and meta-analysis. Psychiatry Res. 2021, 295, 113599. [Google Scholar] [CrossRef]
- Preti, E.; Di Mattei, V.; Perego, G.; Ferrari, F.; Mazzetti, M.; Taranto, P.; Di Pierro, R.; Madeddu, F.; Calati, R. The Psychological Impact of Epidemic and Pandemic Outbreaks on Healthcare Workers: Rapid Review of the Evidence. Curr. Psychiatry Rep. 2020, 22, 43. [Google Scholar] [CrossRef] [PubMed]
- Hilmi, M.; Boilève, A.; Ducousso, A.; Michalet, M.; Turpin, A.; Neuzillet, C.; Naoun, N. Professional and Psychological Impacts of the COVID-19 Pandemic on Oncology Residents: A National Survey. JCO Glob. Oncol. 2020, 6, 1674–1683. [Google Scholar] [CrossRef]
- Spitzer, R.L.; Kroenke, K.; Williams, J.B.W.; Löwe, B. A brief measure for assessing generalized anxiety disorder: The GAD-7. Arch. Intern. Med. 2006, 166, 1092–1097. [Google Scholar] [CrossRef]
- Micoulaud-Franchi, J.-A.; Lagarde, S.; Barkate, G.; Dufournet, B.; Besancon, C.; Trébuchon-Da Fonseca, A.; Gavaret, M.; Bartolomei, F.; Bonini, F.; McGonigal, A. Rapid detection of generalized anxiety disorder and major depression in epilepsy: Validation of the GAD-7 as a complementary tool to the NDDI-E in a French sample. Epilepsy Behav. 2016, 57, 211–216. [Google Scholar] [CrossRef]
- Plateforme COVID-19. Available online: https://covid-19.sante.gouv.fr/tests (accessed on 4 July 2022).
- Martínez-Flores, D.; Zepeda-Cervantes, J.; Cruz-Reséndiz, A.; Aguirre-Sampieri, S.; Sampieri, A.; Vaca, L. SARS-CoV-2 Vaccines Based on the Spike Glycoprotein and Implications of New Viral Variants. Front. Immunol. 2021, 12, 701501. [Google Scholar] [CrossRef]
- Carrat, F.; de Lamballerie, X.; Rahib, D.; Blanché, H.; Lapidus, N.; Artaud, F.; Kab, S.; Renuy, A.; Szabo de Edelenyi, F.; Meyer, L.; et al. Antibody status and cumulative incidence of SARS-CoV-2 infection among adults in three regions of France following the first lockdown and associated risk factors: A multicohort study. Int. J. Epidemiol. 2021, 50, 1458–1472. [Google Scholar] [CrossRef]
- Hozé, N.; Paireau, J.; Lapidus, N.; Tran Kiem, C.; Salje, H.; Severi, G.; Touvier, M.; Zins, M.; de Lamballerie, X.; Lévy-Bruhl, D.; et al. Monitoring the proportion of the population infected by SARS-CoV-2 using age-stratified hospitalisation and serological data: A modelling study. Lancet Public Health 2021, 6, e408–e415. [Google Scholar] [CrossRef]
- Doernberg, S.B.; Holubar, M.; Jain, V.; Weng, Y.; Lu, D.; Bollyky, J.B.; Sample, H.; Huang, B.; Craik, C.S.; Desai, M.; et al. Incidence and prevalence of COVID-19 within a healthcare worker cohort during the first year of the SARS-CoV-2 pandemic. Clin. Infect. Dis. 2022, 75, 1573–1584. [Google Scholar] [CrossRef]
- Grégoire, C.; Huynen, P.; Gofflot, S.; Seidel, L.; Maes, N.; Vranken, L.; Delcour, S.; Moutschen, M.; Hayette, M.-P.; Kolh, P.; et al. Predictive factors for the presence and long-term persistence of SARS-CoV-2 antibodies in healthcare and university workers. Sci. Rep. 2022, 12, 9790. [Google Scholar] [CrossRef]
- Leister, I.; Ponocny-Seliger, E.; Kollaritsch, H.; Dungel, P.; Holzer, B.; Grillari, J.; Redl, H.; Ponocny, I.; Wilfing, C.; Aigner, L.; et al. Antibody seroprevalence and rate of asymptomatic infections with SARS-CoV-2 in Austrian hospital personnel. BMC Infect. Dis. 2021, 21, 915. [Google Scholar] [CrossRef]
- Lumley, S.F.; O’Donnell, D.; Stoesser, N.E.; Matthews, P.C.; Howarth, A.; Hatch, S.B.; Marsden, B.D.; Cox, S.; James, T.; Warren, F.; et al. Antibody Status and Incidence of SARS-CoV-2 Infection in Health Care Workers. N. Engl. J. Med. 2021, 384, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Sallam, M.; Al-Sanafi, M.; Sallam, M. A Global Map of COVID-19 Vaccine Acceptance Rates per Country: An Updated Concise Narrative Review. J. Multidiscip. Healthc. 2022, 15, 21–45. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Yang, Y.; Liu, Y.; Zheng, D.; Shao, L.; Jin, J.; He, Q. Intention to COVID-19 vaccination and associated factors among health care workers: A systematic review and meta-analysis of cross-sectional studies. Am. J. Infect. Control 2021, 49, 1295–1304. [Google Scholar] [CrossRef] [PubMed]
- Paris, C.; Bénézit, F.; Geslin, M.; Polard, E.; Baldeyrou, M.; Turmel, V.; Tadié, É.; Garlantezec, R.; Tattevin, P. COVID-19 vaccine hesitancy among healthcare workers. Infect. Dis. Now 2021, 51, 484–487. [Google Scholar] [CrossRef]
- Biswas, N.; Mustapha, T.; Khubchandani, J.; Price, J.H. The Nature and Extent of COVID-19 Vaccination Hesitancy in Healthcare Workers. J. Community Health 2021, 46, 1244–1251. [Google Scholar] [CrossRef]
- Gagneux-Brunon, A.; Detoc, M.; Bruel, S.; Tardy, B.; Rozaire, O.; Frappe, P.; Botelho-Nevers, E. Intention to get vaccinations against COVID-19 in French healthcare workers during the first pandemic wave: A cross-sectional survey. J. Hosp. Infect. 2021, 108, 168–173. [Google Scholar] [CrossRef]
- Verger, P.; Scronias, D.; Fradier, Y.; Meziani, M.; Ventelou, B. Online study of health professionals about their vaccination attitudes and behavior in the COVID-19 era: Addressing participation bias. Hum. Vaccines Immunother. 2021, 17, 2934–2939. [Google Scholar] [CrossRef]
- Verger, P.; Scronias, D.; Dauby, N.; Adedzi, K.A.; Gobert, C.; Bergeat, M.; Gagneur, A.; Dubé, E. Attitudes of healthcare workers towards COVID-19 vaccination: A survey in France and French-speaking parts of Belgium and Canada, 2020. Eurosurveillance 2021, 26, 2002047. [Google Scholar] [CrossRef]
- Halbrook, M.; Gadoth, A.; Martin-Blais, R.; Gray, A.N.; Kashani, S.; Kazan, C.; Kane, B.; Tobin, N.H.; Ferbas, K.G.; Aldrovandi, G.M.; et al. Longitudinal Assessment of Coronavirus Disease 2019 Vaccine Acceptance and Uptake Among Frontline Medical Workers in Los Angeles, California. Clin. Infect. Dis. 2022, 74, 1166–1173. [Google Scholar] [CrossRef]
- Crawshaw, J.; Konnyu, K.; Castillo, G.; van Allen, Z.; Grimshaw, J.M.; Presseau, J. Behavioural determinants of COVID-19 vaccination acceptance among healthcare workers: A rapid review. Public Health 2022, 210, 123–133. [Google Scholar] [CrossRef]
- Bianchi, F.P.; Stefanizzi, P.; Brescia, N.; Lattanzio, S.; Martinelli, A.; Tafuri, S. COVID-19 vaccination hesitancy in Italian healthcare workers: A systematic review and meta-analysis. Expert Rev. Vaccines 2022, 21, 1289–1300. [Google Scholar] [CrossRef]
- Chiffres clés en France—Data Vaccin COVID. Available online: https://datavaccin-covid.ameli.fr/pages/synthese/ (accessed on 3 January 2023).
- Hummel, S.; Oetjen, N.; Du, J.; Posenato, E.; Resende de Almeida, R.M.; Losada, R.; Ribeiro, O.; Frisardi, V.; Hopper, L.; Rashid, A.; et al. Mental Health Among Medical Professionals During the COVID-19 Pandemic in Eight European Countries: Cross-sectional Survey Study. J. Med. Internet Res. 2021, 23, e24983. [Google Scholar] [CrossRef] [PubMed]
- Cag, Y.; Erdem, H.; Gormez, A.; Ankarali, H.; Hargreaves, S.; Ferreira-Coimbra, J.; Rubulotta, F.; Belliato, M.; Berger-Estilita, J.; Pelosi, P.; et al. Anxiety among front-line health-care workers supporting patients with COVID-19: A global survey. Gen. Hosp. Psychiatry 2021, 68, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Carmassi, C.; Dell’Oste, V.; Bui, E.; Foghi, C.; Bertelloni, C.A.; Atti, A.R.; Buselli, R.; Di Paolo, M.; Goracci, A.; Malacarne, P.; et al. The interplay between acute post-traumatic stress, depressive and anxiety symptoms on healthcare workers functioning during the COVID-19 emergency: A multicenter study comparing regions with increasing pandemic incidence. J. Affect. Disord. 2022, 298, 209–216. [Google Scholar] [CrossRef] [PubMed]
| Eligible Population N = 517 | |
|---|---|
| Age (years)—mean (SD) | 42 (11) |
| Gender | |
| Male | 86 (16.6%) |
| Female | 431 (83.4%) |
| Last degree obtained | |
| Less than two-year degree | 114 (22.1%) |
| Two-year degree or more | 392 (75.8%) |
| Not available | 11 (2.1%) |
| Activity | |
| Care | 306 (59.2%) |
| Social, educational, psychological, and cultural | 10 (1.9%) |
| Clinical or fundamental research | 48 (9.3%) |
| Engineering and technical maintenance | 2 (0.4%) |
| Purchasing-Logistics | 7 (1.4%) |
| Quality, hygiene, safety, environment | 12 (2.3%) |
| Information systems | 8 (1.5%) |
| Information management | 71 (13.7%) |
| Management, management, and decision support | 23 (4.4%) |
| Not available | 30 (5.8%) |
| Home life | |
| Alone | 96 (18.6%) |
| Alone with one or more children | 53 (10.3%) |
| In couple with or without children | 353 (68.3%) |
| With friends, roommates, family | 14 (2.7%) |
| Not available | 1 (0.2%) |
| Overweight | |
| Yes | 88 (17.8%) |
| No | 411 (79.5%) |
| Don’t know or not available | 18 (3.4%) |
| Smoking | |
| Never smoked | 259 (50.1%) |
| Former smoker | 146 (28.2%) |
| Smoke | 108 (20.9%) |
| Not available | 4 (0.8%) |
| Vaccinated against influenza | |
| No | 220 (42.6%) |
| Yes, 2020 and/or 2019 | 196 (37.9%) |
| Yes, 2015–2018 | 28 (5.4%) |
| Yes before 2015 | 39 (7.5%) |
| Yes, last dose unknown | 29 (5.6%) |
| Not available | 5 (1.0%) |
| Heart disease | 24 (4.6%) |
| Chronic respiratory pathology | 21 (4.1%) |
| Dysthyroidia | 16 (3.1%) |
| Immunosuppression or inflammatory-related pathology | 12 (2.3%) |
| Diabetes | 6 (1.2%) |
| Chronic renal pathology | 3 (0.6%) |
| Cancer | 3 (0.6%) |
| Other pathology | 29 (5.6%) |
| Patient Care | Other | Missing | p * | ||||
|---|---|---|---|---|---|---|---|
| n/N | % (95% CI) | n/N | % (95% CI) | n/N | % (95% CI) | ||
| Inclusion | 9/306 | 2.9 (1.0–4.8) | 9/181 | 5.0 (1.8–8.1) | 0/30 | - | 0.3521 |
| 3 months | 19/295 | 6.4 (3.6–9.2) | 12/176 | 6.8 (3.1–10.5) | 0/29 | - | 0.4340 |
| 12 months | 29/270 | 10.7 (7.0–14.4) | 17/167 | 10.2 (5.6–14.8) | 0/25 | - | 0.2626 |
| Inclusion (N = 460) n (%) | 3-Months (N = 442) n (%) | 12-Months (N = 413) n (%) | |
|---|---|---|---|
| Feeling nervous, anxious, or on edge | |||
| Not at all | 186 (40.4) | 212 (48.0) | 216 (52.3) |
| Several days | 195 (42.4) | 158 (35.7) | 129 (31.2) |
| More than half the days | 47 (10.2) | 44 (10.0) | 50 (12.1) |
| Nearly every day | 29 (6.3) | 27 (6.1) | 17 (4.1) |
| Not available | 3 (0.7) | 1 (0.2) | 1 (0.2) |
| Not being able to stop or control worrying | |||
| Not at all | 346 (75.2) | 336 (76.0) | 321 (77.7) |
| Several days | 88 (19.1) | 76 (17.2) | 66 (16.0) |
| More than half the days | 19 (4.1) | 16 (3.6) | 20 (4.8) |
| Nearly every day | 3 (0.7) | 9 (2.0) | 3 (0.7) |
| Not available | 4 (0.9) | 5 (1.1) | 3 (0.7) |
| Worrying too much about different things | |||
| Not at all | 357 (77.6) | 336 (76.0) | 324 (78.5) |
| Several days | 73 (15.9) | 68 (15.4) | 67 (16.2) |
| More than half the days | 19 (4.1) | 23 (5.2) | 12 (2.9) |
| Nearly every day | 7 (1.5) | 10 (2.3) | 6 (1.5) |
| Not available | 4 (0.9) | 5(1.1) | 4 (1.0) |
| Trouble relaxing | |||
| Not at all | 231 (50.2) | 235 (53.2) | 227 (55.0) |
| Several days | 166 (36.1) | 1 (31.4) | 137 (33.2) |
| More than half the days | 42 (9.1) | 40 (9.0) | 31 (7.5) |
| Nearly every day | 16 (3.5) | 23 (5.2) | 16 (3.9) |
| Not available | 5 (1.1) | 5 (1.1) | 2 (0.5) |
| Being so restless that it’s hard to sit still | |||
| Not at all | 367 (79.8) | 357(80.8) | 330 (79.9) |
| Several days | 74 (16.1) | 52 (11.8) | 60 (14.5) |
| More than half the days | 13 (2.8) | 20 (4.5) | 15 (3.6) |
| Nearly every day | 3 (0.7) | 7 (1.6) | 3 (0.7) |
| Not available | 3 (0.7) | 6 (1.4) | 5 (1.2) |
| Becoming easily annoyed or irritable | |||
| Not available | 2 (0.4) | 4 (0.9) | 4 (1.0) |
| Not at all | 234 (50.9) | 240 (54.3) | 2 (57.9) |
| Several days | 180 (39.1) | 140 (31.7) | 122 (29.5) |
| More than half the days | 28 (6.1) | 40 (9.0) | 38 (9.2) |
| Nearly every day | 16 (3.5) | 18 (4.1) | 10 (2.4) |
| Feeling afraid as if something awful might happen | |||
| Not at all | 361 (78.5) | 339 (76.7) | 334 (80.9) |
| Several days | 76 (16.5) | 69 (15.6) | 54 (13.1) |
| More than half the days | 12 (2.6) | 19 (4.3) | 15 (3.6) |
| Nearly every day | 9 (2.0) | 10 (2.3) | 7 (1.7) |
| Not available | 2 (0.4) | 5 (1.1) | 3 (0.7) |
| Anxiety disoder | |||
| No | 334 (72.6) | 318 (71.9) | 300 (72.6) |
| Mild | 90 (19.6) | 75 (17.0) | 83 (20.1) |
| Moderate | 23 (5.0) | 35 (7.9) | 20 (4.8) |
| Severe | 13 (2.8) | 14 (3.2) | 10 (2.4) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Richez, B.; Cantarel, C.; Durrieu, F.; Soubeyran, I.; Blanchi, J.; Pernot, S.; Chakiba Brugère, C.; Roubaud, G.; Cousin, S.; Etienne, G.; et al. One-Year Follow-Up of Seroprevalence of SARS-CoV-2 Infection and Anxiety among Health Workers of a French Cancer Center: The PRO-SERO-COV Study. Int. J. Environ. Res. Public Health 2023, 20, 5949. https://doi.org/10.3390/ijerph20115949
Richez B, Cantarel C, Durrieu F, Soubeyran I, Blanchi J, Pernot S, Chakiba Brugère C, Roubaud G, Cousin S, Etienne G, et al. One-Year Follow-Up of Seroprevalence of SARS-CoV-2 Infection and Anxiety among Health Workers of a French Cancer Center: The PRO-SERO-COV Study. International Journal of Environmental Research and Public Health. 2023; 20(11):5949. https://doi.org/10.3390/ijerph20115949
Chicago/Turabian StyleRichez, Brice, Coralie Cantarel, Françoise Durrieu, Isabelle Soubeyran, Julie Blanchi, Simon Pernot, Camille Chakiba Brugère, Guilhem Roubaud, Sophie Cousin, Gabriel Etienne, and et al. 2023. "One-Year Follow-Up of Seroprevalence of SARS-CoV-2 Infection and Anxiety among Health Workers of a French Cancer Center: The PRO-SERO-COV Study" International Journal of Environmental Research and Public Health 20, no. 11: 5949. https://doi.org/10.3390/ijerph20115949
APA StyleRichez, B., Cantarel, C., Durrieu, F., Soubeyran, I., Blanchi, J., Pernot, S., Chakiba Brugère, C., Roubaud, G., Cousin, S., Etienne, G., Floquet, A., Babre, F., Rivalan, J., Lalet, C., Narbonne, M., Belaroussi, Y., Bellera, C., & Mathoulin-Pélissier, S. (2023). One-Year Follow-Up of Seroprevalence of SARS-CoV-2 Infection and Anxiety among Health Workers of a French Cancer Center: The PRO-SERO-COV Study. International Journal of Environmental Research and Public Health, 20(11), 5949. https://doi.org/10.3390/ijerph20115949






