Assessment of Cellular Responses to Oxidative Stress using MCF-7 Breast Cancer Cells, Black Seed (N. Sativa L.) Extracts and H2O2
Abstract
:Introduction
Materials and Methods
Chemicals
Tissue Culture
Experimental Procedure
Results
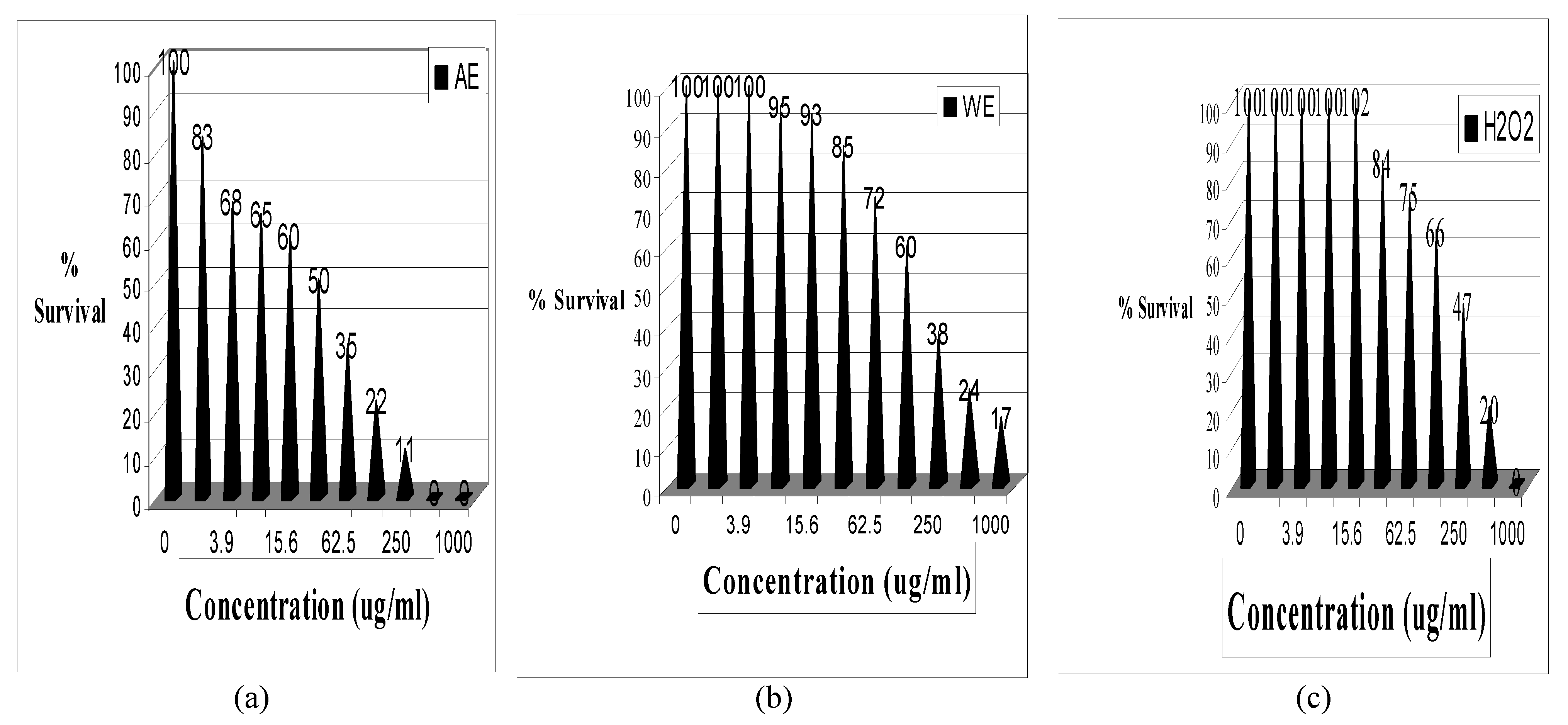
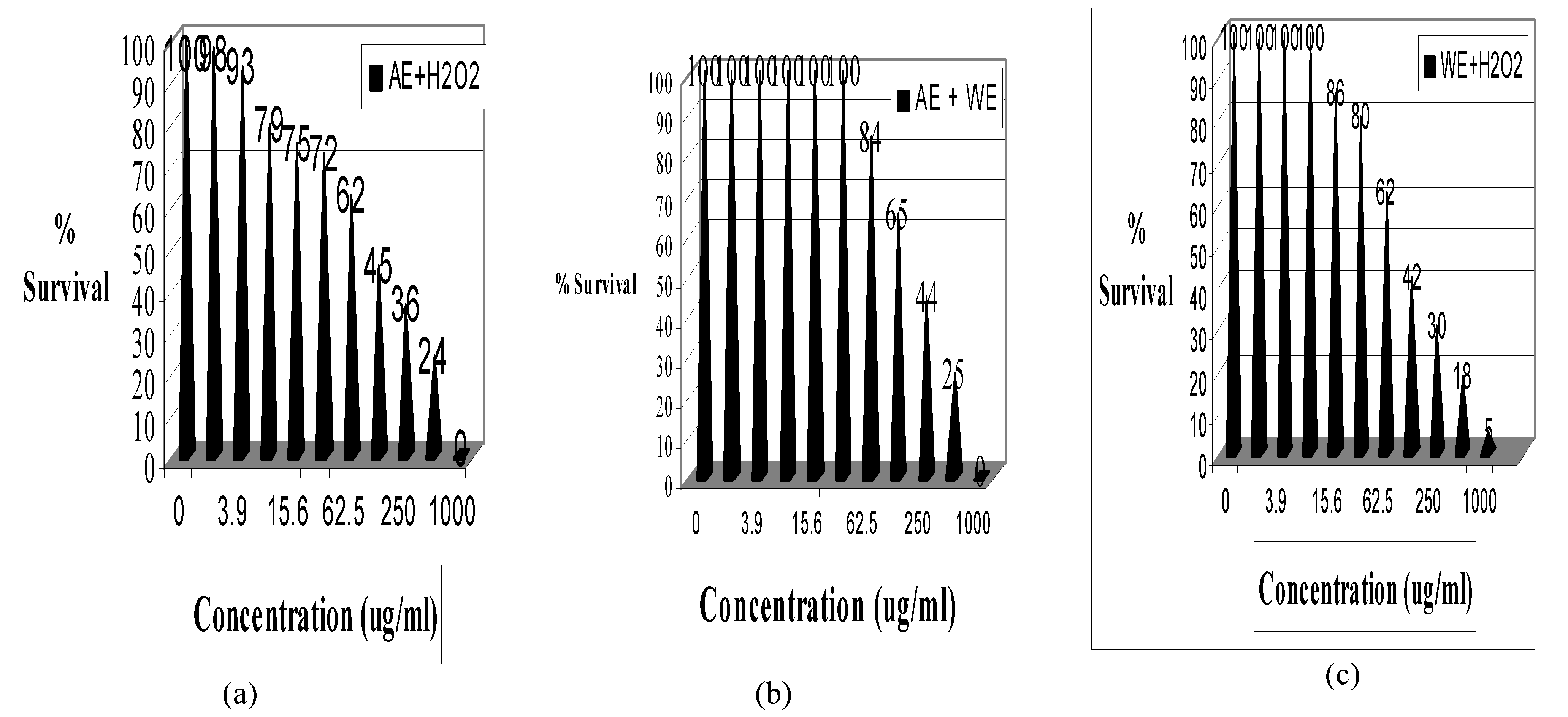
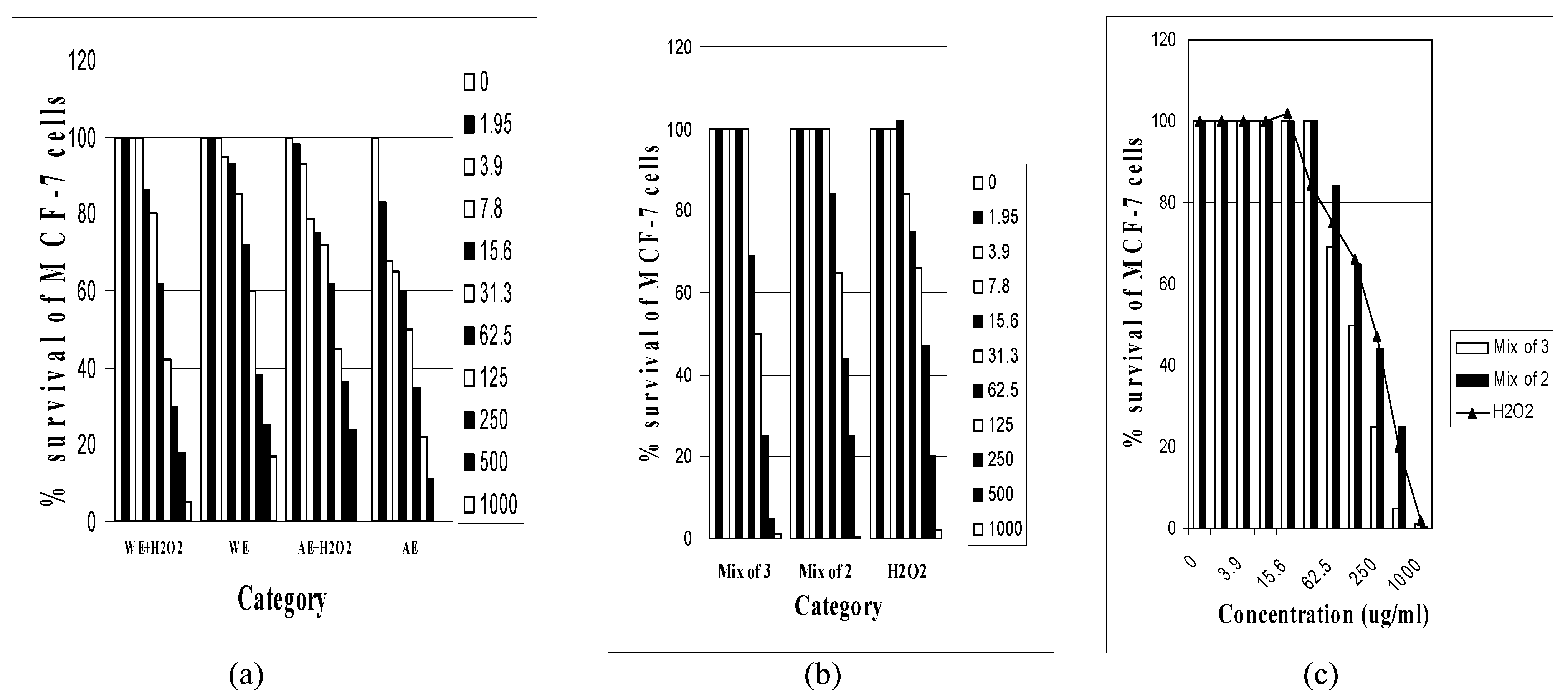
| Treatment | R2 | LC50 | SD | 95% CI** |
| AE+H2O2 | 0.9499 | 357.15a | 20.46 | 313.98 – 400.32 |
| AE | 0.9835 | 418.25 a | 25.3 | 306.42 – 530.08 |
| H2O2 | 0.9682 | 636.71 b | 40.84 | 550.54 – 722.88 |
| WE+H2O2 | 0.9811 | 652.00 b | 52.28 | 541.69 – 762.31 |
| AE + WE | 0.996 | 747.49 b | 35.02 | 676.60 – 821.38 |
| WE | 0.9598 | 796.70 b | 62.8 | 664.19 – 929.21 |
| Mix of 3 | 0.9692 | 809.50 b | 87.49 | 624.90 – 994.10 |
| Parameters | AE | WE | H2O2 | AE+ H2O2 | WE+H2O2 | AE +WE | MIX of 3 |
| Maximum resisted concentration | 0 | 3.9 | 62.5 | 0 | 7.8 | 31.3 | 31.3 |
| First effective concentration | 1.95 | 7.8 | 125 | 1.95 | 15.6 | 62.5 | 62.5 |
| Population size | 1 | 3 | 2 | 1 | 2 | 3 | 3 |
| LC50 | 418.25 | 796.70 | 636.71 | 357.15 | 652.00 | 747.49 | 809.50 |
| Slope | -10.51 | -18.30 | -16.50 | -9.53 | -15.32 | -19.94 | -20.49 |
| Y-intercept | 107.20 | 118.30 | 188.29 | 119.35 | 114.00 | 222.51 | 215.63 |
| Endpoint concentration | 250 - 500 | >1000 | >1000 | 500 - 1000 | >1000 | 500 - 1000 | >1000 |
Discussion
Conclusions
Acknowledgments
References
- Miquel, J.; Quintanilha, A.; Weber, H. Handbook of free radicals and antioxidants in biomedicine. Vol. II, III, and I. CRC Press, CRC Inc.: Bocca Raton, Florida, 1989; Volume 55-56, pp. 7-16,98-101. [Google Scholar]
- Agarwal, M.L.; Taylor, W.R.; Chernov, M.V.; Chernova, O.V.; Stark, G.R. The p53 network. Journal of biological chemistry on line 1998, 273, 1–4, mini-review, issue 1. [Google Scholar] [CrossRef]
- Knowles, H.J.; Harris, A.L. Hypoxia and Oxidative stress in breast cancer, Hypoxia and tumorigensis. Breast cancer Research 2001, 3(5), 318–322. [Google Scholar] [CrossRef]
- Brown, N.S.; Ricknel, R. Hypoxia and oxidative stress in breast cancer: Oxidative stress; its effects on growth, metastasis potential in response to therapy to breast cancer. Breast cancer research 2001, 3(5), 323–327. [Google Scholar] [CrossRef]
- ACS, NCHS_CDC. Cancer Statistics. Cancer Facts and Figures 2004, 4. [Google Scholar]
- Pugh, C.W.; Gleadle, J.; Maxwell, P.H. Hypoxia and oxidative stress in breast cancer. Hypoxia signaling pathways. Breast Cancer Research 2001, 3(5), 313–317. [Google Scholar]
- Seven, A.; Erbil, Y.; Seven, R.; Inci, F.; Gulyasar, T.; Barutcu, B.; Candan, G. Breast cancer and benign breast disease patients evaluated in relation to oxidative stress. Cancer Biochemistry Biophysics 1998, 16(4), 333–345. [Google Scholar]
- Swamy, S.M.; Huat, B.T. Intracellular glutathione depletion and reactive oxygen species generation are important in alpha-hederin-induced apoptosis of P388 cells. Mol. Cell Biochem. 2003, 127–139. [Google Scholar]
- Sipe, H.R.; Jordan, S.J.; Hanna, P.M.; Mason, R.P. The metabolism of 17 beta-estradiol by Lactoperoxidase: a possible source of oxidative stress in breast cancer. Carcinogenesis 1994, 15(11), 2637–2643. [Google Scholar]
- Iddamaldeniya, S.S.; Wickramassinghe, N.; Thabrew, I.; Ratnatunge, N.; Thammitiyagodage, M.G. Protection against diethylnitrosamine-induced hepatocarcinogenesis by an indigenous medicine comprised of Nigella sativa, Hemidesmus indicus and Smilax glabra: a preliminary study. J. Carcinog. 2003, 2(1), 6–11. [Google Scholar]
- Salim, E.I.; Fukushima, S. Chemopreventive potential of volatile oil from black cumin (Nigella sativa L.) seeds against rat colon carcinogenesis. NUTr. Cancer 2003, 195–202. [Google Scholar]
- Mabrouk, G.M.; Moselhy, S.S.; Zohny, S.F.; Ali, E.M.; Helal, T.E.; Amin, A.A.; Khalid, A.A. Inhibition of methylnitrosourea (MNU) induced oxidative stress carcinogenesis by orally administered bee honey and Nigella grains in Sprague Dawely rats. J. Exp. Clin. Cancer Res. 2002, 21(3), 341–346. [Google Scholar]
- Tabata, M.; Honda, G.; Sezik, E.; Yesilada, E. A report on Traditional Medicine and Medical Plants in Turkey (1990, 1991). Faculty of Pharmaceutical Sciences, Koyoto University, 1993; pp. 25–116. [Google Scholar]
- Ghazanfar, A.S.; Mohammed, A. Medical Plants of Northeastern and Central Oman (Arabia)”. Economic Botany 1993, 47(1), 193–199. [Google Scholar]
- Ghazanfar, S.A. Handbook of Arabian Medcinal Plants. CRC Press Inc. 1994, 180. [Google Scholar]
- Haq, A.; Lobo, P.I.; Al-Tufail, M.; Rama, N.R.; Al-Sedairy, S.T. Immunomodulatory effect of Nigella sativa proteins fractionated by ion exchange chromatography. Int. J. Immunopharmacol. 1999, 21(4), 283–295. [Google Scholar]
- Salomi, N.J.; Nair, S.C.; Jayawardhanan, K.K; Varghese, C.D.; Panikkar, K.R. Antitumour principles from Nigella sativa seeds. Cancer Lett. 1992, 63(1), 41–46. [Google Scholar]
- Worthen, D.R.; Ghosheh, O.A.; Crooks, P.A. The in vitro anti-tumor activity of some crude and purified components of blackseed, Nigella sativa L. Anticancer Res. 1998, 18(3A), 1527–1532. [Google Scholar]
- Ali, B.H.; Blunden, G. Pharmacological and toxicological properties of Nigella sativa. Pytother Res. 2003, 299–305. [Google Scholar]
- Sayed, M.D. Traditional Medicine in Health Care. J. Ethnopharmacol. 1980, 2(1), 19–22. [Google Scholar] [CrossRef]
- Enomoto, S.; Asano, R.; Iwahori, Y.; Narui, T.; Okada, Y.; Singab, A.N.; Okuyama, T. Hematological studies on black cumin oil from the seeds of Nigella sativa L. Biol Pharm Bull. 2001, 24(3), 307–310. [Google Scholar]
- Zaoui, A.; Cherrah, Y.; Alaoui, K.; Mahassine, N.; Amarouch, H.; Hassar, M. Effects of Nigells sativa fixed oil on blood homeostasis in rat. J. Ethnopharmacol. 2002, 79(1), 23–26. [Google Scholar]
- El Tahir, K.E.; Ashour, M.M.; Al-Harbi, M.M. The cardiovascular actions of the volatile oil of the black seed (Nigella sativa) in rats: elucidation of the mechanism of action. Gen Pharmacol 1993, 24(5), 1123–1131. [Google Scholar] [CrossRef]
- Gilani, A.H.; Aziz, I.N.; Khurram, M.; KhurramChaudhary, K.S.; Iqbal, A. Bronchodilator, spasmolytic and calcium antagonist activities of Nigella sativa seeds (Kalonji): a traditional herbal product with multiple medicinal uses. J. Pak Med Assoc. 2001, 51(3), 115–120. [Google Scholar]
- Al-Ghamdi, M.S. The anti-inflammatory, analgesic and antipyretic activity of Nigella sativa. J. Ethnopharmacol 2001, 76(1), 45–48. [Google Scholar] [CrossRef]
- Turkdogan, M.K.; Agaoglu, Z.; Yener, Z.; Sekeroglu, R.; Akkan, H.A.; Avci, M.E. The role of antioxidant vitamins (C and E), selenium and Nigella sativa in the prevention of liver fibrosis and cirrhosis in rabbits: new hopes. Dtsch Tierarztl Wochenschr 2001, 108(2), 71–73. [Google Scholar]
- Mahmoud, M.R.; Al-Abhar, H.S.; Saleh, S. The effet of Nigella sativa oil against the liver damage induced by Schistosoma mansoni infection in mice. J. Ethnpharmacol. 2002, 79(1), 1–11. [Google Scholar] [CrossRef]
- El-Dakhakhny, M.; Mady, N.I.; Halim, M.A. Nigella sativa L. oil protects against induced hepatotoxicity and improves serum lipid profile in rats. Arzneimittelforschung 2000, 50(9), 832–836. [Google Scholar]
- El-Dakhakhny, M.; Barakat, M.; El-Halim, M.A; Aly, S.M. Effects of Nigella sativa oil on gastric secretion and ethanol induced ulcer in rats. J. Ethnopharmacol. 2000, 72(1-2), 299–304. [Google Scholar]
- Hanafy, M.S.; Hatem, M.E. Studies on the antimicrobial activity of Nigella sativa seed (black cumin). J. Ethnopharmacol 1991, 34(2-3), 275–278. [Google Scholar] [CrossRef]
- Morsi, N.M. Antimicrobial effect of crude extracts of Nigella sativa on multiple antibiotics-resistant bacteria. Acta Microbiol Pol. 2000, 49(1), 63–74. [Google Scholar]
- El-Fatatry, H.M. Isolation and structure assignment of an antimicrobial principle from the volatile oil of Nigella sativa L. seeds. Pharmazie 1975, 30(2), 109–111. [Google Scholar]
- Salem, M.L.; Hossain, M.S. Protective effect of black seed oil from Nigella sativa against murine cytomegalovirus infection. Int. J. Immunopharmacol. 2000, 22(9), 729–740. [Google Scholar] [CrossRef]
- Aqel, M.; Shaheen, R. Effects of the volatile oil of Nigella sativa seeds on the uterine smooth muscle of rat and guinea pig. J. Ethnopharmacol. 1996, 52(1), 23–26. [Google Scholar] [CrossRef]
- Haq, A.; Abdullatif, M.; Lobo, P.I.; Khabar, K.S.; Sheth, K.V.; Al-Sedairy, S.T. Nigella sativa: effect on human lymphocytes and polymorphonuclear leukocyte phagocytic activity. Immunopharmacology 1995, 30(2), 147–155. [Google Scholar]
- Houghton, P.J.; Zarka, R.; De las Heras, B.; Hoult, J.R. Fixed oil of Nigella sativa and derived thymoquinone inhibit eicosanoid generation in leukocytes and membrane lipid peroxidation. Planta Med. 1995, 61(1), 33–36. [Google Scholar]
- Keshri, G.; Singh, M.M.; Lakshmi, V.; Kamboj, V.P. Post-coital contraceptive efficacy of the seeds of Nigella sativa in rats. Indian J. Physiol Pharmacol. 1995, 39(1), 59–62. [Google Scholar]
- Chakravarty, N. Inhibition of histamine release from mast cells by nigellone. Ann Allergy 1993, 70(3), 237–242. [Google Scholar]
- Nair, S.C.; Salomi, M.J.; Panikkar, B.; Panikkar, K.R. Modulatory effects of Crocus sativus and Nigella sativa extracts on cisplatin-induced toxicity in mice. J Ethnopharmacol 1991, 31(1), 75–83. [Google Scholar]
- Salomi, M.J.; Nair, S.C.; Panikkar, K.R. Inhibitory effects of Nigella sativa and saffron (Crocus sativus) on chemical carcinogenesis in mice. Nutr. Cancer 1991, 16(1), 67–72. [Google Scholar] [CrossRef]
- Al-Gaby, A.M. Amino acid composition and biological effects of supplementing broad bean and corn protein with N. Sativa (black Cumin). Nahrung 1998, 42(5), 290–294. [Google Scholar] [CrossRef]
- Al-Jassir, M.S. Chemical Composition and Microflora of Black Cumin (Nigella sativa) seeds growing in Saudia Arabia. Food Chemistry 1992, 45, 239–242. [Google Scholar] [CrossRef]
- Nergiz, C.; Otler, S. Chemical Composition of Nigella sativa L. seeds. Food Chemistry 1993, 259–261. [Google Scholar] [CrossRef]
- Aboul-Enein, Y.H.; Abou-Basha, I.L. Simple HPLC method for the determination of Thymoquinone in black seed oil (Nigella sative Linn). Liquid Chromatography 1998, 18(5), 895–902. [Google Scholar]
- Haq, A.R.; Al-Sediary, T.S. Fractionation of black seed oil (N. sativa Linn) Proteins by using Rotofor. Liquid Chromatography and Related Technology 1996, 19(4), 593–599. [Google Scholar]
- Ramadan, M.F.; Morsel, J.T. Characterization of Phospholipids composition of black cumin (N. sativa L.) seed oil. Nahrung 2002, 46(4), 240–244. [Google Scholar] [CrossRef]
- Morsel, J.T. Characterization of Phospholipids Composition of black Cumin (Nigella sativa L.) seed oil. Nahrung 2002, 46(4), 240–244. [Google Scholar]
- Badary, O.A.; Gamal El-Din, A.M. Inhibitory effects of thymoquinone against 20-methylcholanthrene-induced fibrosarcoma tumorigenesis. Cancer Detect Prev. 2001, 25(4), 362–368. [Google Scholar]
- Kumara, S.S.; Huat, B.T. Extraction, isolation and characterisation of antitumor principal, alpha-hederin, from the seeds of Nigella sativa. Planta Med. 2001, 67(1), 29–32. [Google Scholar] [CrossRef]
- Shoieb, A.M.; Elgayyar, M.; Dudrick, P.S.; Bell, J.L.; Tithof, P.K. In vitro inhibition of growth and induction of apoptosis in cancer cell lines by thymoquinone. Int J. Oncol. 2003, 22(1), 107–113. [Google Scholar]
- Badary, O.A.; Abdel-Naim, A.B.; Abdel-Wahab, M.H.; Hamada, F.M. The influence of thymoquinone on doxorubicin-induced hyperlipidemic nephropathy in rats. Toxicology 2000, 143(3), 219–226. [Google Scholar]
- Badary, O.A.; Al-Shabanah, O.A.; Nagi, M.N.; Al-Rikabi, A.C.; Elmazar, M.M. Inhibition of benzo (a) pyrene-induced forestomach carcinogenesis in mice by thymoquinone. Eur J Cancer Prev. 1999, 5, 435–440. [Google Scholar]
- Daba, M.A.; Abdel-Rahman, M.S. Hepatoprotective activity of Thymoquinone in isolated rat hepatocytes. Toxicol Lett. 1998, 95(1), 23–29. [Google Scholar] [CrossRef]
- Nagi, M.N.; Alam, K.; Badary, O.A.; Al-Shabanah, O.A.; Al-Sawaf, H.A.; Al-Bekairi, A.M. Thymoquinone protects against carbon tetrachloride hepatotoxicity in mice via an antioxidant mechanism.Biochem. Biochem. Mol. Biol. Int. 1999, 47(1), 153–159. [Google Scholar]
- Ramadan, M.F.; Kroh, L.W.; Morsel, J.T. Radical scavenging activity of black cumin (Nigella sativa L.) coriander (Corianddrum sativum L), and Niger (Guizotia abyssinica Cass. L.) crude seed oils and oil fractions. J. Agric. Food Chem. 2003, 51(24), 6961–6969. [Google Scholar] [CrossRef]
- Fisher, B.; Constantino, J.; Vickerham, D.; Redmond, C.; Kavanah, M.; Cronin, W.; Vogel, V.; et al. Tamoxifen for the prevention of breast cancer: Report of the national Surgical Adjuvant Breast and Bowel Project P-1 Study. J. Natl. Cancer Inst. 1998, 90, 1371–1398. [Google Scholar] [CrossRef]
- Cummings, S.; Eckert, S.; Kneger, K.; Crady, D.; et al. The effect of Raloxifene on the risk of breast cancer in postmenopausal women-results from the MORE randomized trial. JAMA 1999, 281, 2189–2219. [Google Scholar]
- Singh, D.K.; Lippman, S.M. Cancer chemoprevention part 2: hormones, non-classic antioxidant natural agents, NSAIDs and other agents. Oncology 1998, 12, 1787–1805. [Google Scholar]
- Cauley, J.; Norton, I.; Lippman, M.; et al. Continued breast cancer risk reduction in postmenopausal women treated with raloxifene; 4-year results from the MORE trial. Breast Cancer Res. Treat 2001, 65, 125–134. [Google Scholar] [CrossRef]
- Wu, K.; Kim, H.; Rodriquez, S.; et al. Suppression of Mammary Tumorigenesis in Trasgenic Mice by the RXR-selective Retinoid, LGD1069. Cancer Epi, Biomarkers and Prevention 2002, 11, 467–474. [Google Scholar]
- Taffe, B.G.; Suzuki, J. Role of oxidative stress in Tumor promotion. In Handbook of Free radicals and Antoxidants in Biomedicine. J. Miquel, A. T. Quintaanilha and H. Weber (Eds). CRC Press 1989, Vol. III, 35–49. [Google Scholar]
- Borg, D.C.; Schaich, K.M. Pro-oxidant action of Antioxidants. In Handbook of Free radicals and Antoxidants in Biomedicine. J. Miquel, A.T. Quintaanilha and H. Weber (Eds). CRC Press 1989, Vol. I, 63–80. [Google Scholar]
- Burits, M.; Bucar, F. Antioxidant activity of Nigella sativa essential oil. Phytother Res. 2000, 14(5), 323–328. [Google Scholar] [CrossRef]
© 2005 MDPI. All rights reserved.
Share and Cite
Farah, I.O. Assessment of Cellular Responses to Oxidative Stress using MCF-7 Breast Cancer Cells, Black Seed (N. Sativa L.) Extracts and H2O2. Int. J. Environ. Res. Public Health 2005, 2, 411-419. https://doi.org/10.3390/ijerph2005030005
Farah IO. Assessment of Cellular Responses to Oxidative Stress using MCF-7 Breast Cancer Cells, Black Seed (N. Sativa L.) Extracts and H2O2. International Journal of Environmental Research and Public Health. 2005; 2(3):411-419. https://doi.org/10.3390/ijerph2005030005
Chicago/Turabian StyleFarah, Ibrahim O. 2005. "Assessment of Cellular Responses to Oxidative Stress using MCF-7 Breast Cancer Cells, Black Seed (N. Sativa L.) Extracts and H2O2" International Journal of Environmental Research and Public Health 2, no. 3: 411-419. https://doi.org/10.3390/ijerph2005030005
APA StyleFarah, I. O. (2005). Assessment of Cellular Responses to Oxidative Stress using MCF-7 Breast Cancer Cells, Black Seed (N. Sativa L.) Extracts and H2O2. International Journal of Environmental Research and Public Health, 2(3), 411-419. https://doi.org/10.3390/ijerph2005030005




