Assessment of Arsenic Contamination of Groundwater and Health Problems in Bangladesh
Abstract
:Introduction
Geology of Bangladesh in Relation to Arsenic in Groundwater
Origin and Mechanism of Arsenic Mobilization
- (i)
- Oxidation of arsenical pyrite;
- (ii)
- Reductive dissolution of FeOOH (hydrous ferric oxides or HFO) resulting in the release of sediment-bound arsenic;
- (iii)
- Anion (competitive) exchange of sediment-bound arsenic with phosphate from fertilizers;
- (iv)
- Release of arsenic from the degradation of pesticides* and fertilizers. We used published data as well as the knowledge of socio-environmental and geologic conditions in Bangladesh in critically reviewing these mechanisms to explain arsenic release to groundwater.
Oxidation of Arsenical
- (i)
- Why is arsenic found in areas that are not affected by upstream diversion of surface water, such as in the Meghna basin (NE Bangladesh)?
- (ii)
- Why are arsenic concentrations not highest in the unsaturated zone beneath the surface where the groundwater table fluctuates between dry and wet seasons?
- (iii)
- Does arsenic pollution increase during the wet season?
- (iv)
- Are the Eh-pH field changes in the aquifer due to drawdown?
- (v)
- Why is the arsenic pollution so severe in West Bengal and Bihar where upstream diversion of surface water did not occur?
- (vi)
- Why does the arsenic pollution occur in areas where drawdown is not a likely cause for oxidation, such as in Wisconsin, USA?
Reductive Dissolution of Hydrous Ferric Oxides (HFO)
- (i)
- Is there a peat layer above the aquifer in the entire area that is polluted with arsenic?
- (ii)
- Even if peat layers and/or organic rich sediments occur in aquifer materials, at what depth do they occur?
- (iii)
- Why is there arsenic pollution along the Brahmaputra riverbanks, where sediments are predominantly sandy in nature?
- (iv)
- The peat layer associated with the low sea level position in the Bengal Basin is found at depths ranging from 2 to 7 m below mean sea level and about 80 to 120 km landward of the present shoreline location. Why then is the aquifer located at depths ranging from 15 to 75 m polluted with arsenic? How does the arsenic produced in the peat layer located at 2 to 7 m move to a greater depth?
- (v)
- (vi)
- The Madhupur Tract and Barind Tract are also very similar in their geologic origin, i.e. they represent ancient delta plain and floodplains. Why are there no organic-rich sediments and high concentrations of arsenic in the aquifers in those areas?
- (vii)
- Can the hypothesis account for the variability in arsenic concentrations in terms of time-dependent field data? [20].
Competitive ion Exchange
- (i)
- Why do not the phosphate anions from fertilizers force the release of arsenic in all geologic settings equally?
- (ii)
- Why are arsenic concentrations not high in surface water where phosphate anions are supposed to be in high concentration?
- (iii)
- Is there any relationship between fertilizer application and elevated arsenic concentrations in Bangladesh or elsewhere?
Release of Arsenic from Degradation of Pesticides and Fertilizers
- (i)
- What are the pathways and sinks of pesticides and fertilizers as they relate to groundwater movement?
- (ii)
- How do the pesticides and phosphate fertilizers move through the fine grained sediments into the deep aquifers?
- (iii)
- Why is the amount of arsenic in groundwater not proportional to the amount of pesticides and fertilizers used Bangladesh?
- (iv)
- Are there any seasonal variations in the amount of arsenic observed in groundwater that reflects the usage of pesticides and fertilizers?
- (v)
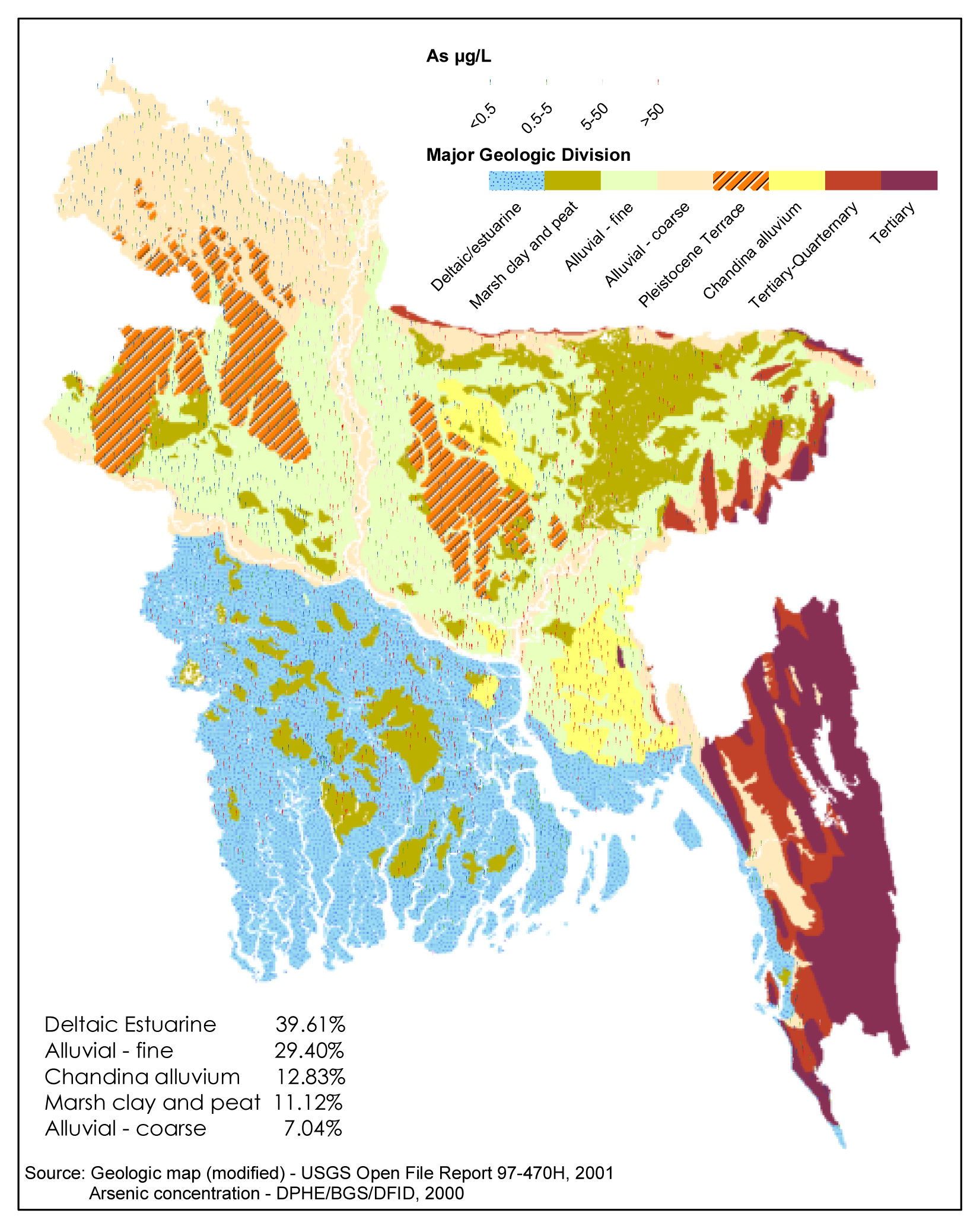
- Why are the high concentrations of arsenic predominantly associated with the low-lying coastal/deltaic environments that are characterized by fine-grained sediments (Fig. 2)?
Unsolved Geologic Problem and Its Consequences on Human Health
Effect of Arsenic Poisoning on Health
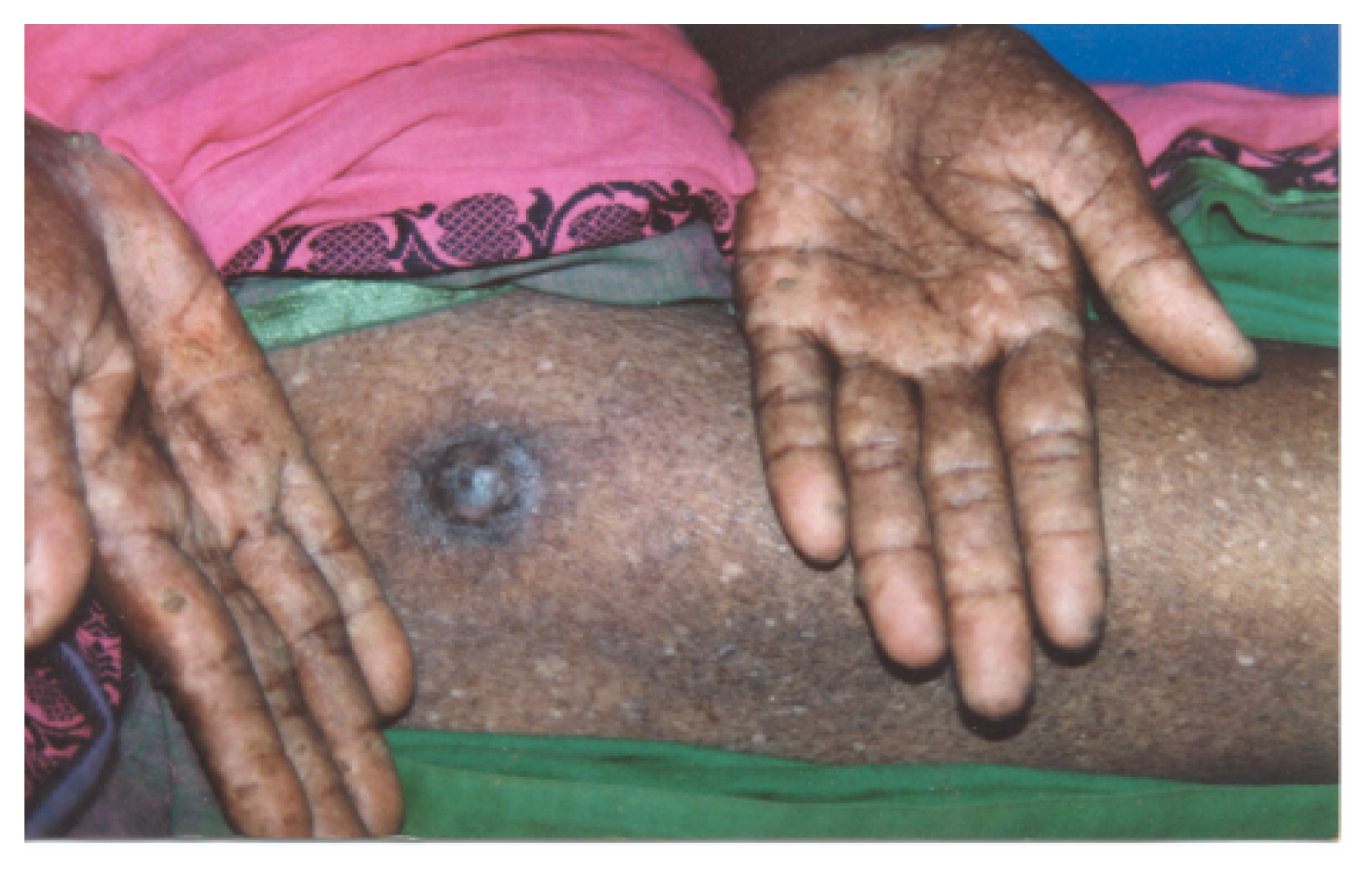
Skin Lesions
Skin Cancer
Clinical Symptoms and Findings
Nutritional Status and Disease
Arsenic Contaminating Vegetables and Crops
Conclusions
- (i)
- A comprehensive study of the geologic history including the compilation of paleogeographic interpretations for various time intervals.
- (ii)
- The identification of the biogeochemical processes active in different geographic and geologic settings.
- (iii)
- The investigation of regional land-use practices.
- (iv)
- The design of appropriate conceptual models for determining groundwater flowpaths.
- (v)
- The design of appropriate geochemical models of reaction mechanisms.
- (vi)
- A comprehensive study of the epidemiological aspects of the problem. These studies need to be undertaken in order to establish relationships between biogeochemical factors responsible for the release of arsenic and the observed concentrations of arsenic in groundwater in Bangladesh.
Future Research and Policy Implications
- (i)
- Detailed lithologic descriptions of well logs (types and characteristics of units) to establish a 3-D framework of the geologic strata to identify and characterize those units acting as aquifers (porous and permeable layers that hold and transmit water to tubewells) aquitards (impervious layers that impede water movement through them) in the arsenic-affected areas.
- (ii)
- The geologic history of evolution of the Bengal Basin and the resultant sediments as a function of time and space.
- (iii)
- Reconstruct paleogeographic maps of Bangladesh, which will illustrate the types of sediments deposited at various locations and depths at a given time period.
- (iv)
- Develop conceptual models of groundwater flowpaths, with evidence supported by computer simulations.
- (v)
- Use geochemical data collected from the existing pumping wells, as well as from newly drilled wells, to develop geochemical models, which can be used to determine the types of chemical reactions and products, including possible chemical complexes. The best way to determine complexes is to utilize geochemical computer codes, such as MINTEQ or PHREEQE [47].
- (vi)
- Compile geologic information for sediments and chemicals observed at various depths to decipher spatial and temporal relationships between sediments and arsenic concentrations.
- (vii)
- Multidisciplinary studies to better understand biogeochemical environments and epidemiological relationships to the natural environments.
- (i)
- Enhance public awareness of the health problems from contaminated water.
- (ii)
- Take short- and long-term intervention strategies to curb the exposure.
- (iii)
- Strengthen rapid diagnostic facilities.
- (iv)
- Establish effective treatment facilities in rural areas.
- (v)
- Improve the nutritional status of the people.
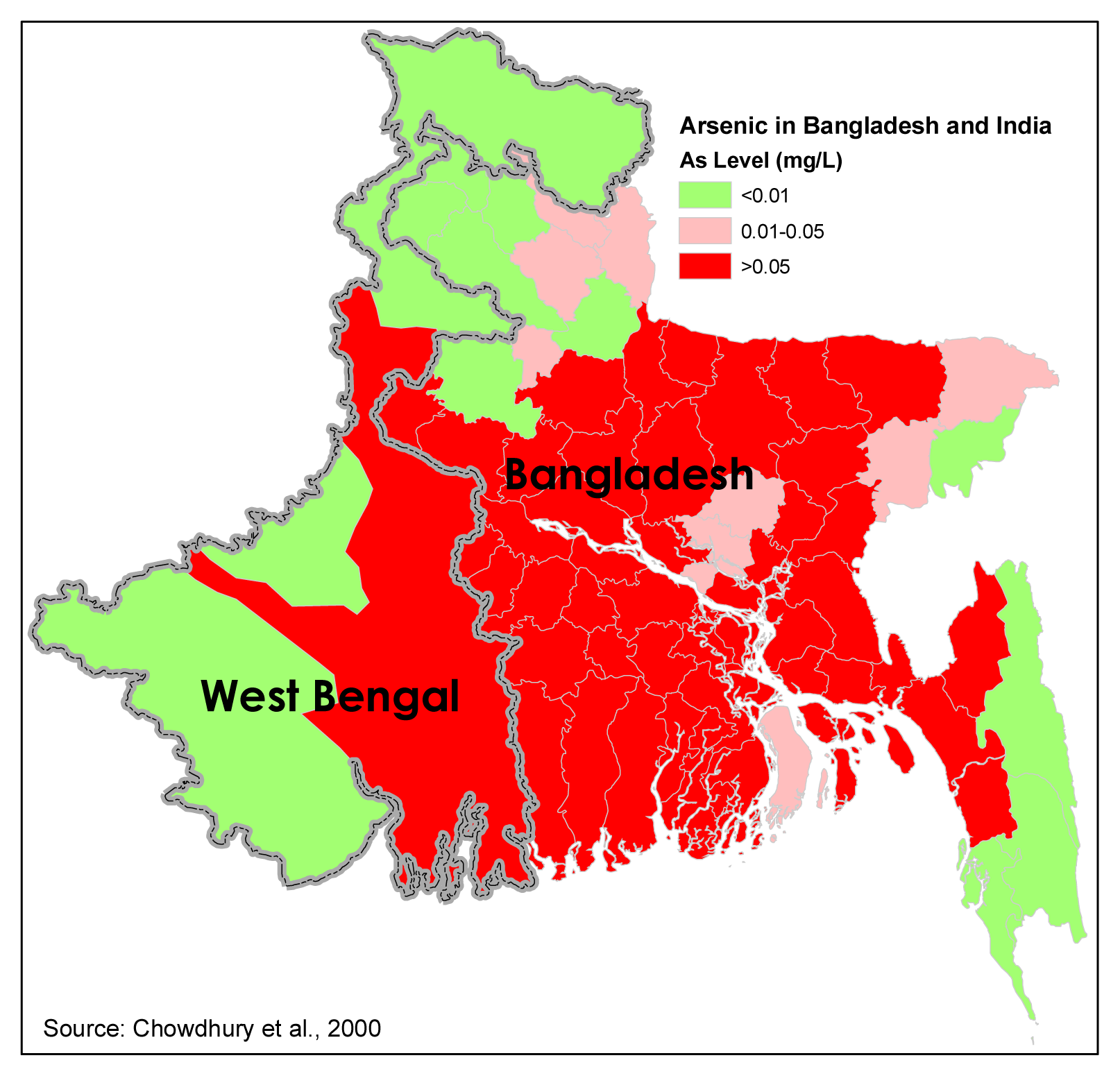
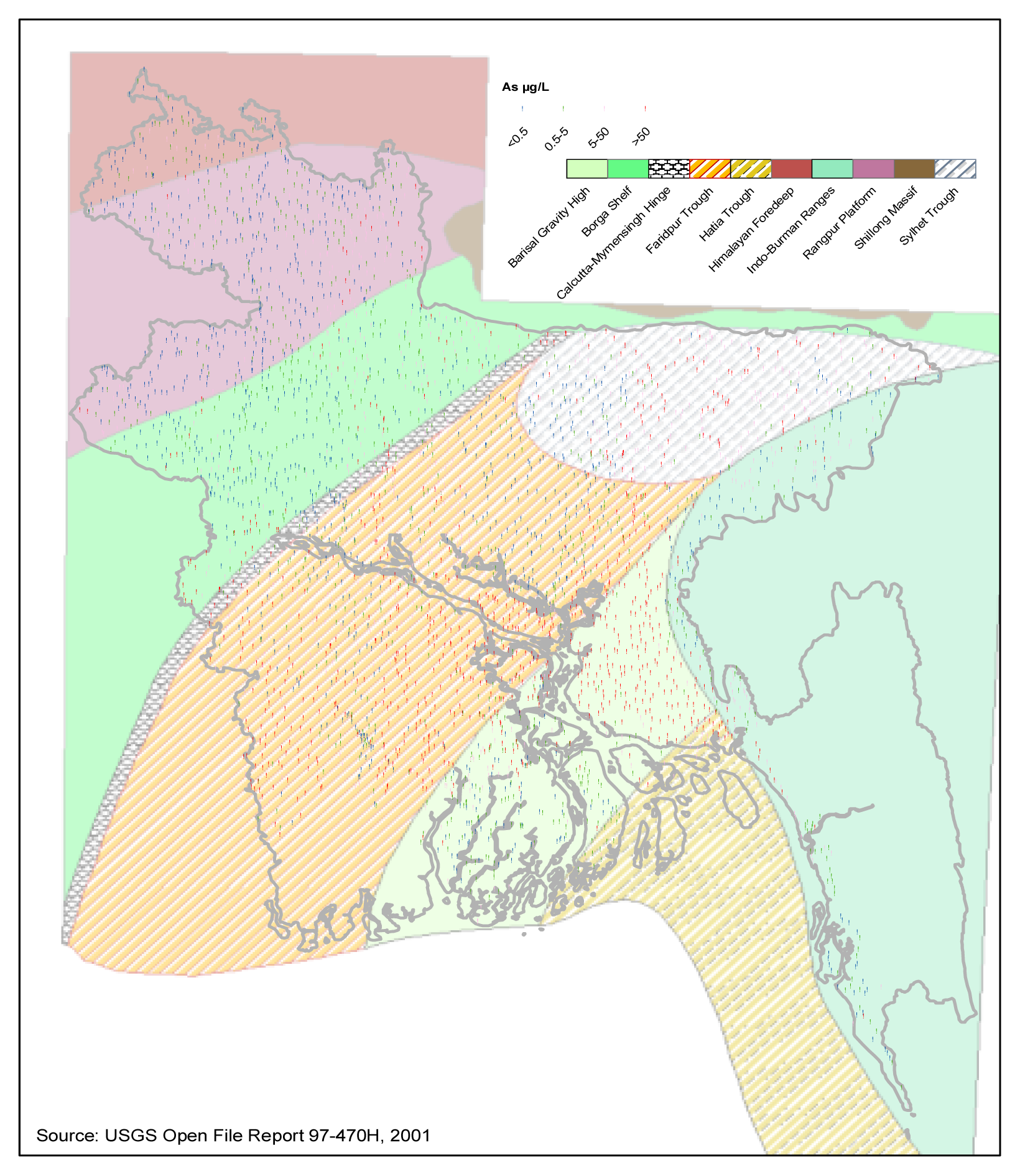
| Hypotheses | Major Determining Factors | References | Major Unexplained Aspects |
|---|---|---|---|
| Oxidation of arsenical pyrite |
| [11, 13, 15] |
|
| Reductive dissolution of hydrous iron oxides (HFO) |
| [6, 19] |
|
| Competitive ion exchange |
| [21] |
|
| Arsenic from pesticides and fertilizers | Requires application of adequate amount of arsenic-bearing pesticides & fertilizers | [22, 23] |
|
| Clinical features | Number (%)* |
|---|---|
| Weakness | 59 (39) |
| Chronic cough | 35 (23) |
| Joint pain or backache | 26 (17) |
| Itching | 26 (17) |
| Abdominal pain | 24 (16) |
| Chest pain | 24 (16) |
| Loss of appetite | 18 (12) |
| Insomnia | 15 (10) |
| Shortness of breath | 15 (10) |
| Frequent urination with burning | 15 (10) |
| Tingling and numbness | 12 (8) |
| Headache | 12 (8) |
| Malaise | 8 (5) |
| Chronic dysentery | 6 (4) |
| Blurred vision | 4 (3) |
| Conjunctivitis | 2 (1) |
| Palpitation | 2 (1) |
| Deceased libido | 1 (1) |
References
- WHO, Guideline for Drinking Water Quality, RecommendationVolume 1, 2nd ed; Geneva; World Health Organization, 1992; p. 41.
- U.S. EPA, Drinking Water Regulations and Health Advisories; Washington, DC; U.S. Environmental Protection Agency, Health and Ecological Criteria Division, 1993.
- Saha, K. C. Saha’s grading of arsenicosis progression and treatment. In Arsenic Exposure and Health Effects V; Chappell, W. R., Abernathy, C. O., Calderon, R. I., Thomas, D. J., Eds.; Elsvier: Amsterdam, 2003; pp. 391–414. [Google Scholar]
- Chowdhury, U. K.; Biswas, B. K.; Chowdhury, T. R.; Samantha, G.; Mandal, B.; Basu, G. C.; Chanda, C. R.; Lodh, D.; Saha, K. C.; Mukherjee, S. K.; Roy, S.; Kabir, S.; Quamruzzaman, Q.; Chakrabarti, D. Groundwater arsenic contamination in Bangladesh and West Bengal, India. Environmental Health Perspectives 2000, 108, 393–397. [Google Scholar]
- Frisbie, S. H.; Ortega, R.; Maynard, D. M.; Sarkar, B. The concentration of arsenic and other toxic elements in Bangladesh’s drinking water. Environmental Health Perspectives 2002, 110(11), 1147–1153. [Google Scholar]
- McArthur, J. M.; Ravenscroft, P.; Safiullah, S.; Thirlwall, M. F. Arsenic in groundwater: testing pollution mechanism for sedimentary aquifers in Bangladesh. Water Resources Research 2000, 37(1), 109–117. [Google Scholar]
- DPHE and BGS. Groundwater Studies for Arsenic Contamination in Bangladesh. Draft Final Report Summary 2000.
- U.S. Geological SurveyOpen File Report 97-470H. 2001. http://pubs.usgs.gov/of/1997/ofr-97-470/OF97-470H/.
- Banerjee, M.; Sen, P. K. Paleobiology in understanding the change of sea level and coast line in Bengal Basin during Holocene period. Indian Journal of Earth Sciences 1987, 14(3–4), 307–320. [Google Scholar]
- Bhattachaya, P.; Sracek, A.; Jacks, G. Groundwater arsenic in Bengal Delta Plains – Testing of Hypotheses. 1998. http://wso.net/wei/dch/acic/infobank/bp2.htm.
- Mallick, S.; Rajagopal, N. R. The mischief of oxygen in groundwater. Post conference Report: Experts’ Opinions, Recommendation and Future Planning for Groundwater Problems of West Bengal, School of Environmental Studies, Jadavpur University, Calcutta, India, 1995; pp. 71–73.
- Karim, M.M.; Komori, Y.; Alam, M. Subsurface arsenic occurrence and depth of contamination in Bangladesh. Journal of Environmental Chemistry 1997, 7(4), 783–792. [Google Scholar]
- Mandal, B. K.; Biswas, B. K.; Dhar, R. K.; Roy Chowdhury, T.; Samanta, G.; Basu, G. K.; Chanda, C. R.; Saha, K. C.; Chakrabarti, D.; Kabir, S.; Roy, S. Groundwater arsenic contamination and suffering of people in West Bengal, India and Bangladesh. In Metals and Genetics; Sarkar, B., Ed.; Kluwer Academic/Plenum Publishers: New York, 1999; pp. 41–65. [Google Scholar]
- Nickson, R. T.; McArthur, J. M.; Ravenscroft, P.; Burgess, W. G.; Ahmed, K. M. Mechanism of arsenic release to groundwater, Bangladesh and West Bengal. Applied Geochemistry 1999, 15(4), 1–11. [Google Scholar]
- Ahmed, K. M.; Imam, M. B.; Akhter, S. H.; Hasan, M. A.; Alam, M. M.; Chowdhury, S. Q.; Burgess, W. G.; Nickson, R.; AcArthur, J. J.; Hasan, M. K.; Ravenscroft, P.; Rahman, M. Mechanism of arsenic release to groundwater: geochemical and mineralogical evidence. In Abstracts, International Conference on Arsenic Pollution of Groundwater in Bangladesh: Causes, Effects and Remedies, Dhaka, Bangladesh; 1988; pp. 125–126. [Google Scholar]
- Islam, F. S.; Gault, A. G.; Boothman, C.; Polya, D. A.; Charnock, J. M.; Chatterjee, D.; Llyod, J. R. Role of metal-reducing bacteria in arsenic release from Bengal delta sediments. Nature 2004, 430, 68–70. [Google Scholar]
- Stuben, D.; Berner, Z.; Chandashekaram, D.; Karmakar, J. Arsenic enrichment in groundwater of West Bengal, India: geochemical evidence for mobilization of As under reducing conditions. Applied Geochemistry 2003, 18, 1417–1434. [Google Scholar]
- Hussam, A.; Habibuddowla, M.; Alauddin, M.; Hossain, Z. A.; Munir, A. K. M.; Khan, A. H. Chemical fate of arsenic and other metals in groundwater of Bangladesh: experimental measurement and chemical equilibrium model. Journal of Environmental Science and Health 2003, A38(1), 71–86. [Google Scholar]
- Mukherjee, A. B.; Bhattachrya, P. Arsenic in groundwater in the Bengal Delta Plain: slow-poisoning in Bangladesh. Environmental Review 2001, 9, 189–220. [Google Scholar]
- Fazal, M. A.; Ichion, E.; Kawachi, T. Validity of the latest research findings on causes of groundwater arsenic contamination in Bangladesh. International Water Resources Association, Water International 2001, 26(2), 380–389. [Google Scholar]
- Acharyya, S. K.; Chakraborty, P.; Lahiri, S.; Raymahashay, B. C.; Guha, S.; Bhowmik, A. Arsenic poisoning in the Ganges delta. Nature 1999, 402, 545. [Google Scholar]
- Chakraborty, A. K.; Saha, K. C. Arsenic dermatosis from tubewell water in West Bengal. Indian Journal of Medical Research 1987, 85, 326–334. [Google Scholar]
- Anwar, J. Arsenic and Uranium in Fertilizer. 2004. http://www.sos-arsenic.net/english/tsp.html.
- Das, D.; Chatterjee, A.; Samanta, G.; Mandal, B.; Roy Chowdhury, T.; Samanta, G.; Chowdhury, P.P.; Chanda, C.; Basu, G.; Lodh, D.; Nandi, S.; Chakraborty, T.; Mandal, S.; Bhattacharya, S.M.; Chakraborti, D. Arsenic contamination in groundwater in six districts in West Bengal, India: the biggest arsenic calamity in the world. Analyst 1994, 119, 168–170. [Google Scholar]
- Tseng, W. P. Effects and dose-response relationships of skin cancer and Blackfoot disease with arsenic. Environ Health Perspectives 1977, 19, 109–19. [Google Scholar]
- Guha Mazumder, D.N.; Haque, R.; Ghose, N.; De, B.K.; Santra, A.; Chakraborty, D. Arsenic levels in drinking water and the prevalence of skin lesions in West Bengal, India. Int J Epidemiol 1998, 27, 871–77. [Google Scholar]
- Guha Mazumder, D. N.; Haque, R.; Ghose, N.; De, B. K.; Santra, A.; Chakraborti, D. Arsenic in drinking water and the prevalence of respiratory effects in West Bengal, India. Int J Epidemiol 2000, 29, 1047–52. [Google Scholar]
- Chen, C. J.; Hsueh, Y. M.; Lai, M. S.; Shyu, M. P.; Chen, S. Y.; Wu, M. M. Increased prevalence of hypertension and long-term arsenic exposure. Hypertension 1995, 25, 53–60. [Google Scholar]
- Huet, P. M.; Guillaume, E.; Cote, J.; Legare, A.; Lavoie, P.; Viallet, A. Noncirrhotic presinusoidal portal hypertension associated with chronic arsenical intoxication. Gastroenterology 1975, 68, 1270–77. [Google Scholar]
- Hsueh, Y. M.; Wu, W. L.; Huang, Y. L.; Chiou, H. Y.; Tseng, C. H.; Chen, C. J. Low serum carotene level and increased risk of ischemic heart disease related to long-term arsenic exposure. Atherosclerosis 1998, 141, 249–57. [Google Scholar]
- Hertz-Picciotto, I.; Arrighi, H.M.; Hu, S.W. Does arsenic exposure increase the risk of circulatory disease? Am J Epidemiol 2000, 151, 174–81. [Google Scholar]
- Curry, A.; Carrin, G.; Bartran, J.; Yamamura, S.; Heeijnen, H.; Sims, J. Towards an assessment of the socioeconomic impact of arsenic poisoning in Bangladesh. Geneva; World Health Organization; In Protection of the human environment: water, sanitation and health series; WHO/SDE/WSH/00.4; 2000; p. 40. [Google Scholar]
- Rahman, M.; Tondel, M.; Ahmad, S. A.; Chowdhury, I. A.; Faruquee, M. H.; Axelson, O. Hypertension and arsenic exposure in Bangladesh. Hypertension 1999, 33, 74–8. [Google Scholar]
- Rahman, M.; Tondel, M.; Ahmad, S. A.; Axelson, O. Diabetes mellitus associated with arsenic exposure in Bangladesh. Am. J. Epidemiol 1998, 148, 198–203. [Google Scholar]
- Lai, M. S.; Hsueh, Y. M.; Chen, C. J.; Shyu, M. P.; Chen, S. Y.; Kuo, T. L. Ingested inorganic arsenic and prevalence of diabetes mellitus. Am. J. Epidemiol 1994, 139, 484–92. [Google Scholar]
- Mitra, A. K.; Bose, B. K.; Kabir, H.; Das, B. K.; Hussain, M. Arsenic-related health problems among hospital patients in southern Bangladesh. J. Health Popul Nutr 2002, 20, 198–204. [Google Scholar]
- Ahmad, S. A.; Sayed, M. H.; Barua, S.; Khan, M. H.; Faruquee, M. H.; Jalil, A. Arsenic in drinking water and pregnancy outcomes. Environ Health Perspectives 2001, 109, 629–31. [Google Scholar]
- Smith, A. H.; Lingas, E. O.; Rahman, M. Contamination of drinking-water by arsenic in Bangladesh: a public health emergency. Bull WHO 2000, 78, 1093–1103. [Google Scholar]
- Chiou, H. Y.; Huang, W. I.; Su, C. L.; Chang, S. F.; Hsu, Y. H.; Chen, C. J. Dose-response relationship between prevalence of cardiovascular disease and ingested inorganic arsenic. Stroke 1997, 28, 1717–23. [Google Scholar]
- Buchet, J. P.; Geubel, A.; Pauwels, S.; Mahieu, P.; Lauwerys, R. The influence of liver disease on the methylation of arsenite in humans. Arch Toxicol 1984, 55, 151–4. [Google Scholar]
- Vahter, M.; Marafante, E. Effects of low dietary intake of methionine, choline or proteins on the biotransformation of arsenite in the rabbit. Toxicol Letters 1987, 37, 41–6. [Google Scholar]
- National Research Council, Arsenic in drinking water; National Academy Press: Washington, DC, 1999.
- Guha Mazumder, D. N.; Das Gupta, J.; Santra, A.; Pal, A.; Ghose, A.; Sarkar, S. Non-cancer effects of chronic arsenicosis with special reference to liver damage. In Arsenic: exposure and health effects; Abernathy, C. O., Calderon, R. L., Chappell, W. R., Eds.; Chapman & Hall: London, 1997; pp. 112–23. [Google Scholar]
- Larsen, E. H.; Moseholm, L.; Neilsen, M. M. Atmospheric deposition of trace elements around point sources and human health risk assessment. II. Uptake of arsenic and chromium by vegetables grown near a wood preservation factory. Sci Total Environ 1992, 126, 263–275. [Google Scholar]
- Das, H. K.; Mitra, A. K.; Sengupta, P. K.; Hossain, A.; Islam, F.; Rabbani, G. H. Arsenic concentrations in rice, vegetables, and fish in Bangladesh: a preliminary study. Environ. Int 2004, 30, 283–387. [Google Scholar]
- Abedin, M. J.; Cresser, M. S.; Meharg, A. A..; Feldman, J.; Cotter-Howells, J. Arsenic accumulation and metabolism in rice (Oryza sativa L.). Envron. Sci. Technol 2002, 36, 962–968. [Google Scholar]
- Schwartz, F. W.; Zhang, H. Fundamental of Groundwater; John Wiley & Sons, 2003. [Google Scholar]
© 2005 MDPI. All rights reserved.
Share and Cite
Khalequzzaman, M.; Faruque, F.S.; Mitra, A.K. Assessment of Arsenic Contamination of Groundwater and Health Problems in Bangladesh. Int. J. Environ. Res. Public Health 2005, 2, 204-213. https://doi.org/10.3390/ijerph2005020002
Khalequzzaman M, Faruque FS, Mitra AK. Assessment of Arsenic Contamination of Groundwater and Health Problems in Bangladesh. International Journal of Environmental Research and Public Health. 2005; 2(2):204-213. https://doi.org/10.3390/ijerph2005020002
Chicago/Turabian StyleKhalequzzaman, Md., Fazlay S. Faruque, and Amal K. Mitra. 2005. "Assessment of Arsenic Contamination of Groundwater and Health Problems in Bangladesh" International Journal of Environmental Research and Public Health 2, no. 2: 204-213. https://doi.org/10.3390/ijerph2005020002
APA StyleKhalequzzaman, M., Faruque, F. S., & Mitra, A. K. (2005). Assessment of Arsenic Contamination of Groundwater and Health Problems in Bangladesh. International Journal of Environmental Research and Public Health, 2(2), 204-213. https://doi.org/10.3390/ijerph2005020002