Health Risk Assessment of Cyanobacterial (Blue-green Algal) Toxins in Drinking Water
Abstract
:Introduction
How Abundant are Cyanobacterial Toxins?
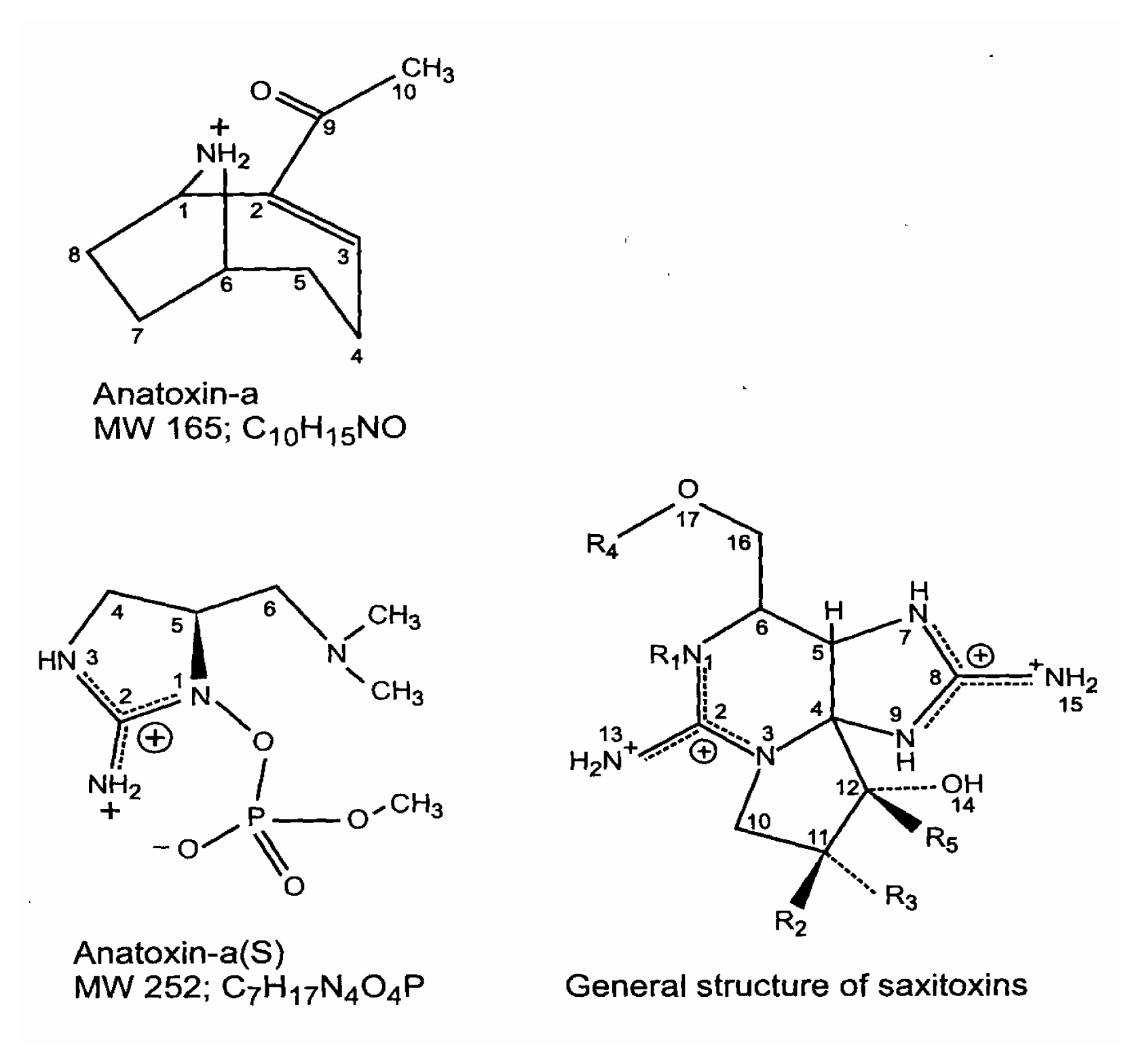
Cyanobacterial Toxins
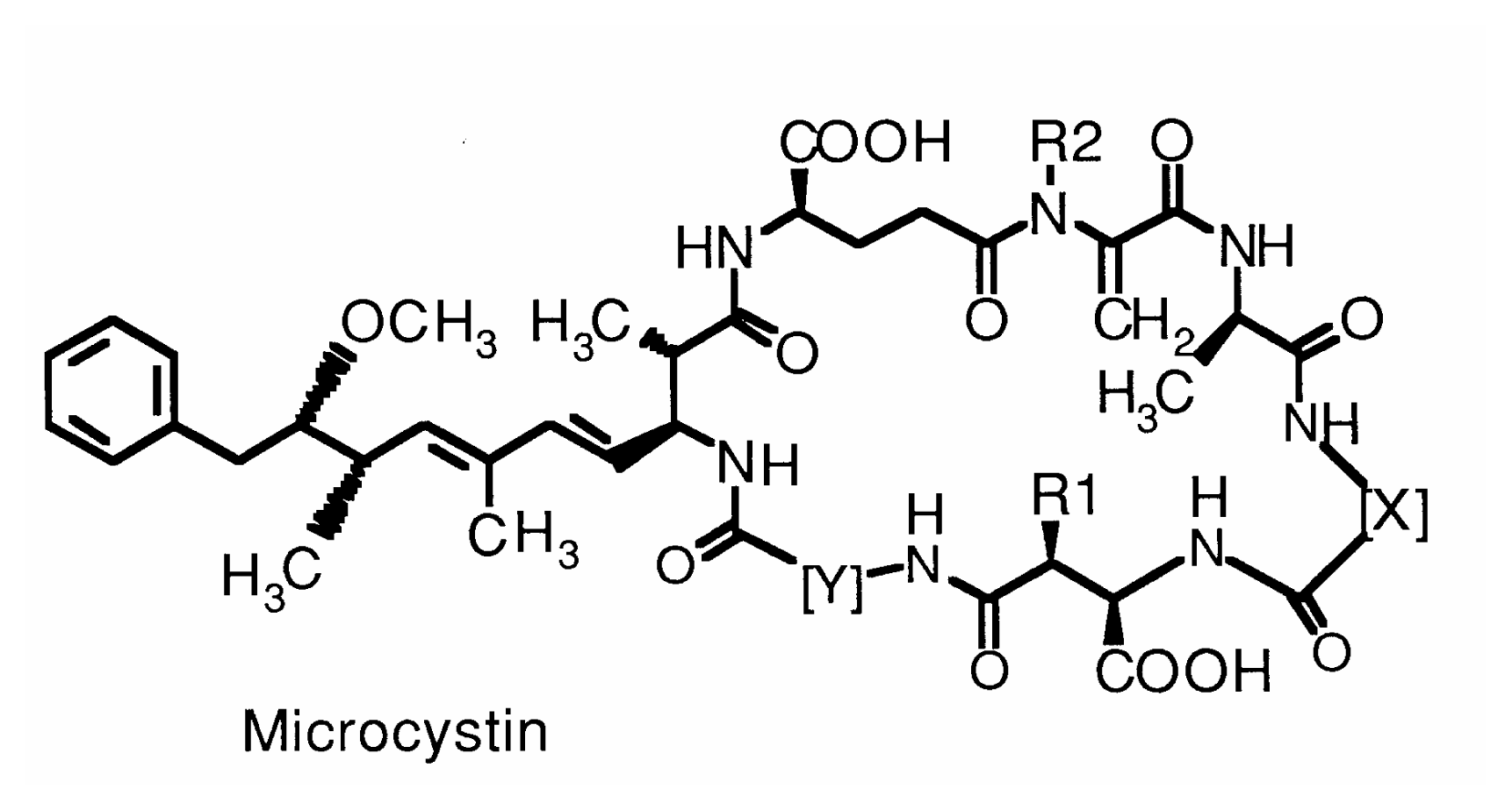
Hepatotoxins
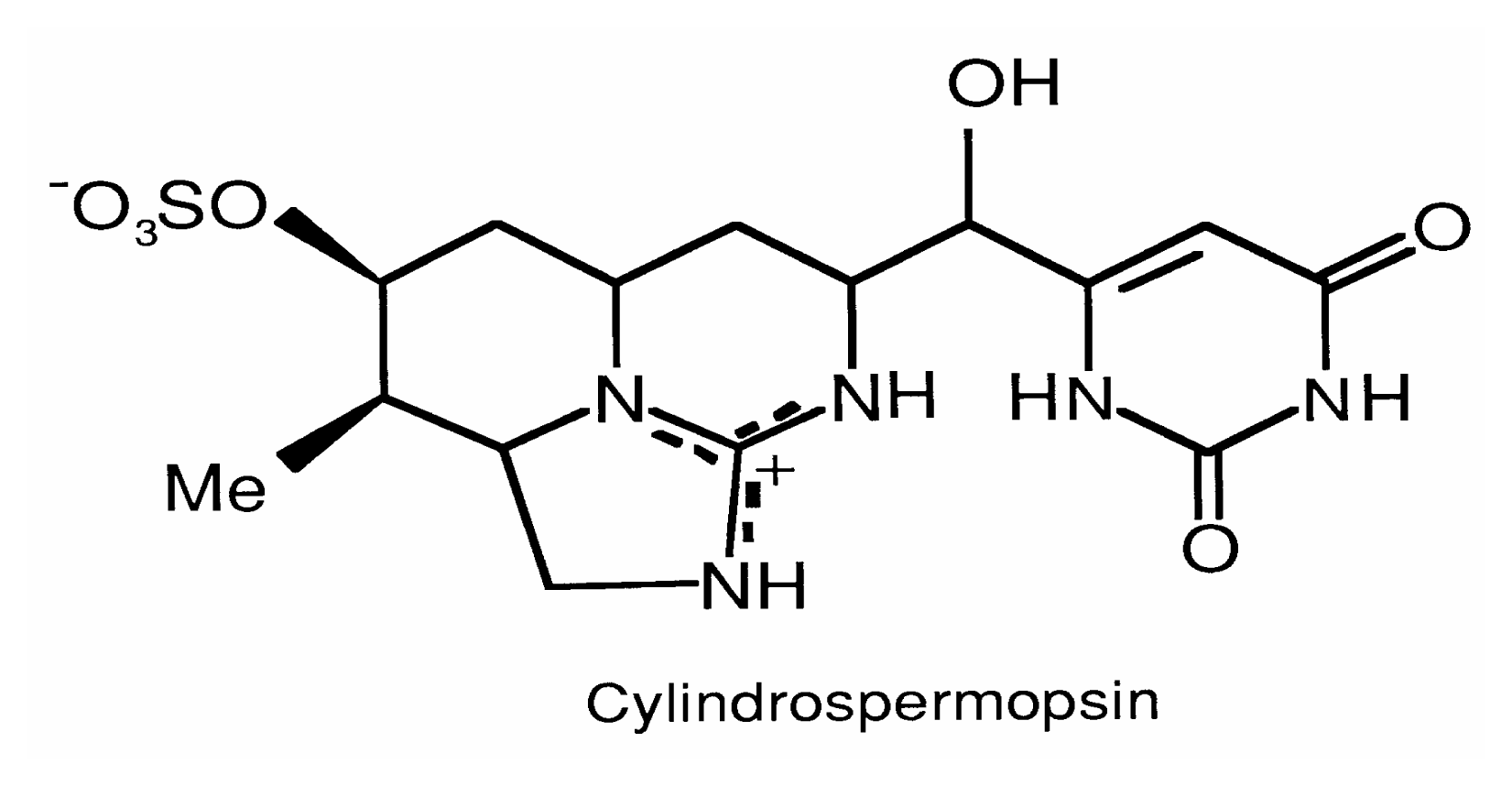
Cylindrospermopsins
Evaluation of the Risks to the Population from Cyanobacterial Toxins
Conclusion
References
- Falconer, I. R. Cyanobacterial Toxins of Drinking Water Supplies: Cylindrospermopsins and Microcystins. Chapter 5 Cyanobacterial poisoning of livestock and people; CRC Press: Boca Raton, Fl., 2005. [Google Scholar]
- Bowling, L. The cyanobacterial (blue-green algal) bloom in the Darling/Barwon River system, November-December, 1991; Technical Services Division, New South Wales Department of Water Resources: Australia, Sydney, 1992. [Google Scholar]
- Steyn, D. G. Poisoning of animals and human beings by algae. South African Journal of Science 1945, 41, 243–244. [Google Scholar]
- Teixera, M. G. L. C.; Costa, M. C. N.; Carvalho, V. L. P.; Pereira, M. S.; Hage, E. Gastroenteritis epidemic in the area of the Itaparica Dam, Bahia, Brazil. Bulletin of the Pan-American Health Organisation 1993, 27, 244–253. [Google Scholar]
- Jochimsen, E. M.; Carmichael, W. W.; An, J. S.; Cardo, D. M.; Cookson, S. T.; Holmes, C. E. M.; Antunes, M. B. D.; Demelo, D. A.; Lyra, T. M.; Barreto, V. S. T.; Azevedo, S. M. F. O.; Jarvis, W. R. Liver failure and death after exposure to microcystins at a hemodialysis center in Brazil. The New England Journal of Medicine 1998, 338, 873–878. [Google Scholar]
- Carmichael, W. W.; Azevedo, S. M. F. O.; An, J. S.; Molica, R. J. R.; Jochimsen, E. M.; Lau, S.; Rinehardt, K. L.; Shaw, G. R.; Eaglesham, G. K. Human Fatalities from Cyanobacteria: Chemical and Biological Evidence for Cyanotoxins. Environmental Health Perspectives 2001, 109, 663–668. [Google Scholar]
- Azevedo, S. M. F. O.; Carmichael, W. W.; Jochimsen, E. M.; Rinehart, K. L.; Lau, S.; Shaw, G. R.; Eaglesham, G. K. Human intoxication by microcystins during renal dialysis treatment in Caruaru - Brazil. Toxicology 2002, 181, 441–446. [Google Scholar]
- Falconer, I. R.; Beresford, A. M.; Runnegar, M. T. Evidence of liver damage by toxin from a bloom of the blue-green alga Microcystis aeruginosam. Med. J. Aust 1983, 1, 511–4. [Google Scholar]
- Byth, S. Palm Island Mystery Disease. Medical Journal of Australia 1980, 2, 40–42. [Google Scholar]
- Hawkins, P. R.; Runnegar, M. T. C.; Jackson, A. R. B.; Falconer, I. R. Severe hepatotoxicity caused by the tropical cyanobacterium (blue-green alga) Cylindrospermopsis raciborskii (Woloszynska) Seenaya and Subba Raju isolated form a domestic supply reservoir. Applied and Environmental Microbiology 1985, 50, 1292–1295. [Google Scholar]
- Ohtani, L.; Moore, R. E.; Runnegar, M. T. C. Cylindrospermopsin: a potent hepatotoxin from the blue-green alga Cylindrospermopsis raciborskii. J. of the American Chem. Soc 1992, 114, 7941–7942. [Google Scholar]
- Falconer, I. R. Cyanobacterial Toxins of Drinking Water Supplies: Cylindrospermopsins and Microcystins; CRC Press: Boca Raton, 2005. [Google Scholar]
- Mur, L. R.; Skulberg, O. M.; Utkilen, H. Cyanobacteria in the Environment. In Toxic Cyanobacteria in Water. A Guide to their Public Health Consequences, Monitoring and Management; Chorus, I., Bartram, J., Eds.; E & FN Spon on behalf of WHO: London, 1999; pp. 15–40. [Google Scholar]
- Carmichael, W. W.; Mahmood, N. A. Toxins from freshwater cyanobacteria. In Seafood toxins; American Chemical Society: Washington, DC, 1984; pp. 377–389. [Google Scholar]
- Behm, D. Coroner cites algae in teen’s death; Milwaukee Journal Sentinel: Milwaukee, 2003. [Google Scholar]
- Edwards, C.; Beattie, K. A.; Scrimgeour, C. M.; Codd, G. A. Identification of anatoxin-a in benthic cyanobacteria (blue-green algae) and in associated dog poisonings at Loch Insh, Scotland. Toxicon 1992, 30, 1165–1175. [Google Scholar]
- Chorus, I.; Falconer, I. R.; Salas, H. J.; Bartram, J. Health Risks Caused by Freshwater Cyanobacteria in Recreational Waters. J. of Toxicol. and Environ. Health, Part B 2000, 3, 323–347. [Google Scholar]
- Fromme, H.; Kohler, A.; Krause, R.; Fuhrling, D. Occurrence of cyanobacterial toxins – microcystins and anatoxin-a - in Berlin water bodies with implications to human health and regulations. Environ. Toxicol 2000, 75, 120–130. [Google Scholar]
- Mahmood, N. A.; Carmichael, W. W. Anatoxin-a(s), an anticholinesterase from the cyanobacterium Anabaena flos-aquae NRC-525-17. Toxicon 1987, 25, 1221–1227. [Google Scholar]
- Falconer, I. Algal Toxins in Seafood and Drinking Water; Academic Press: London, 1993. [Google Scholar]
- Velzeboer, R. M. A.; Baker, P. D.; Rositano, J.; Heresztyn, T.; Codd, G. A.; Raggett, S. L. Geographical patterns of occurrence and compositions of saxitoxins in the cyanobacterial genus Anabaena (Nostocales Cyanophyta) in Australia. Phycologia 2000, 39, 395–407. [Google Scholar]
- Ferreira, F. M. B.; Soler, J. M. F.; Fidalgo, M. L.; Fernandez-vila, P. PSP toxins from Aphanizomenon flos-aquae (cyanobacteria) collected in the Crestuma-Lever reservoir (Douro River, Northern Portugal). Toxicon 2001, 39, 757–761. [Google Scholar]
- Carmichael, W. W.; Evans, W. R.; Yin, Q. Q.; Bell, P.; Moczydlowski, E. Evidence for paralytic shellfish poisons in the freshwater cyanobacterium Lyngbya wollei (Farlow ex Gomont) comb. nov. Applied and Environmental Microbiology 1997, 65, 3104–3110. [Google Scholar]
- Bartram, J.; Burch, M.; Falconer, I. R.; Jones, G.; Kuiper - Goodman, T. Situation Assessment Planning and Management. In Toxic Cyanobacteria in Water. A Guide to their Public Health Consequences, Monitoring and Management; Chorus, I., Bartram, J., Eds.; E & FN Spon on behalf of WHO: London, 1999; pp. 179–09. [Google Scholar]
- Falconer, I. R.; Runnegar, M. T. C.; Buckley, T.; Huyn, V. L.; Bradshaw, P. Use of Powdered and Granular Activated Carbon to Remove Toxicity from Drinking Water Containing Cyanobacterial Toxins. J. American Water Works Association 1989, 18, 102–105. [Google Scholar]
- Kuiper-Goodman, T.; Falconer, I.; Fitzgerald, J. Human Health Aspects. In Toxic Cyanobacteria in Water. A Guide to their Public Health Consequences, Monitoring and Management; Chorus, I., Bartram, J., Eds.; E & FN Spon on behalf of WHO: London, 1999; pp. 113–153. [Google Scholar]
- New Zealand Ministry of Health, Guidelines for Drinking Water Quality Management for New Zealand; Ministry of Health: Wellington, New Zealand.
- Fitzgerald, D. J.; Cunliffe, D. A.; Burch, M. D. Development of health alerts for cyanobacteria and related toxins in drinking-water in South Australia. Environmental Toxicology 1999, 14, 203–209. [Google Scholar]
- Bartram, J.; Vapnek, J. C.; Jones, G.; Bowling, L.; Falconer, I.; Codd, G. A. Implementation of Management Plans. In Toxic Cyanobacteria in Water. A Guide to their Public Health Consequences, Monitoring and Management; Chorus, J Bartram, Ed.; E & FN Spon: London, 1999; pp. 211–234. [Google Scholar]
- Chorus, I.; Bartram, J. Toxic cyanobacteria in water. In A Guide to their Public Health Consequences, Monitoring and Management; E & FN Spon (on behalf of World Health Organisation): London, 1999. [Google Scholar]
- Runnegar, M.; Berndt, N.; Kaplowitz, N. Microcystin uptake and inhibition of protein phosphatases: effects of chemoprotectants and self-inhibition in relation to known hepatic transporters. Toxicol. App. Pharmacol 1995, 134, 264–72. [Google Scholar]
- Falconer, I. R.; Jackson, A. R. B.; Langley, J.; Runnegar, M. T. C. Liver pathology in mice in poisoning by the blue-green alga in Microcystis aeruginosa. Australian J. of Biol. Sci 1981, 34, 179–187. [Google Scholar]
- Jackson, A. R. B.; McInnes, A.; Falconer, I. R.; Runnegar, M. T. C. Clinical and pathological changes in sheep experimentally poisoned by the blue-green alga Microcystis aeruginosa. Veterinary Pathology 1984, 21, 102–113. [Google Scholar]
- Falconer, I. R.; Smith, J. V.; Jackson, A. R.; Jones, A.; Runnegar, M. T. Oral toxicity of a bloom of the cyanobacterium Microcystis aeruginosa administered to mice over periods up to 1 year. J. Toxicol Environ Health 1988, 24, 291–305. [Google Scholar]
- Ito, E.; Kondo, F.; Terao, K.; Harada, K. L. Neoplastic nodular formation in mouse liver induced by repeated intraperitoneal injections of microcystin-LR. Toxicon 1997, 55, 1453–1457. [Google Scholar]
- Yu, S. Z. Primary prevention of hepatocellular carcinoma. J. of Gastroenterology and Hepatology 1995, 10, 674–682. [Google Scholar]
- Ueno, Y.; Nagata, S.; Tsutsumi, T.; Hasegawa, A.; Watanabe, M.; Park, H.; Chen, G. C.; Chen, G.; Yu, S. Detection of microcystins, a blue-green algal hepatotoxin, in drinking water sampled in Haimen and Fusui, endemic areas of primary liver cancer in China, by highly sensitive immunoassay. Carcino genesis 1996, 17, 1317–1321. [Google Scholar]
- Falconer, I.; Bartram, J.; Chorus, I.; Kuiper Goodman, T.; Utkilen, H.; Burch, M.; Codd, G. A. Safe Levels and Safe Practices. In Toxic Cyanobacteria in Water. A Guide to their Public Health Consequences, Monitoring and Management; Chorus, I., Bartram, J., Eds.; E & FN Spon on behalf of WHO: London, 1999; pp. 55–178. [Google Scholar]
- Fawell, J. K.; James, C. P.; James, H. A. Toxins from blue-green algae: Toxicological assessment of microcystin-LR and a method for its determination in water; WRc pic: Medmenham, 1994. [Google Scholar]
- Australian National Health and Medical Research Council, Drinking Water Guidelines; NH & MRC: Canberra, Australia, 2004.
- Zhou, L.; Yu, H.; Chen, K. Relationship between microcystin in drinking water and colorectal cancer. Biomedical and Environmental Science 2002, 75, 166–171. [Google Scholar]
- Falconer, I. R.; Hardy, S. J.; Humpage, A. R.; Froscio, S. M.; Tozer, G. J.; Hawkins, P. R. Hepatic and renal toxicity of the blue-green alga (cyanobacterium) Cylindrospermopsis raciborskii in male Swiss Albino mice. Environmental Toxicology 1999, 14, 143–150. [Google Scholar]
- Seawright, A. A.; Nolan, C. C.; Shaw, G. R.; Chiswell, R. K.; Norris, R. L.; Moore, M. R.; Smith, M. J. The oral toxicity for mice of the tropical cyanobacterium Cylindrospermopsis raciborskii (Woloszynska). Environmental Toxicology 1999, 14, 135–142. [Google Scholar]
- Froscio, S. M. Investigation of the mechanisms involved in cylindrospermopsin toxicity: Hepatocyte culture and reticulocyte lysate studies. In PhD Thesis, Department of Clinical and Experimental Pharmacology; University of Adelaide: Adelaide, 2002; p. 139. [Google Scholar]
- Runnegar, M. T.; Kong, S. M.; Zhong, Y. Z.; Lu, S. C. Inhibition of reduced glutathione synthesis by cyanobacterial alkaloid cylindrospermopsin in cultured rat hepatocytes. Biochemical Pharmacology 1995, 49, 219–225. [Google Scholar]
- Banker, P.; Carmeli, S.; Hadas, O.; Telsch, B.; Porat, R.; Sukenik, A. Identification of cylindrospermopsin in Aphanizomenon ovalisporum (Cyanophyceae) isolated from Lake Kinneret, Israel. J. of Applied Phycology 1997, 33, 613–616. [Google Scholar]
- Harada, K.; Ohtani, I.; Iwamoto, K.; Suzuki, M.; Watanabe, M. F.; Watanabe, M.; Terao, K. Isolation of Cylindrospermopsin from a Cyanobacterium Umezakia Natans and Its Screening Method. Toxicon 1994, 32, 73–84. [Google Scholar]
- Li, R.; Carmichael, W. W.; Brittain, S.; Eaglesham, G. K.; Shaw, G. R.; Mahakhant, A.; Noparatnaraporn, N.; Youngmanichai, W.; Kaya, K.; Watanabe, M. M. Isolation and identification of the cyanotoxin cylindrospermopsin and deoxy-cylindrospermopsin from a Thailand strain of Cylindrospermopsis raciborskii (Cyanobacteria). Toxicon 2001, 39, 973–980. [Google Scholar]
- Stirling, D. J.; Quilliam, M. A. First report of the cyanobacterial toxin cylindrospermopsin in New Zealand. Toxicon 2001, 39, 1219–1222. [Google Scholar]
- Fastner, J.; Heinze, R.; Humpage, A. R.; Mischke, U.; Eaglesham, G. K.; Chorus, I. Cylindrospermopsin occurrence in two German lakes and preliminary assessment of toxicity and toxin production of Cylindrospermopsis raciborskii (Cyanobacteria) isolates. Toxicon 2003, 42, 313–321. [Google Scholar]
- Griffiths, D. J.; Saker, M. L. The Palm Island Mystery Disease 20 Years on: A Review of Research on the Cyanotoxin Cylindrospermopsin. Environmental Toxicology 2003, 18, 78–93. [Google Scholar]
- Flewelling, L.; Pawlowicz, M.; Carmichael, W.; Williams, C. D.; Burns, J.; Chapman, A. Assessment of Cyanotoxins in Florida’s lakes, reservoirs, and rivers. In Cyanobacteria Survey Project; Harmful Algal Bloom Task Force; St. John’s River Water Management District: Palatka, Florida, 2001. [Google Scholar]
- Humpage, A. R.; Falconer, I. R. Oral toxicity of the cyanobacterial toxin cylindrospermopsin in male Swiss albino mice: Determination of No Observed Adverse Effect Level for Deriving a Drinking Water Guideline Value. Environmental Toxicology 2003, 18, 94–103. [Google Scholar]
- Shaw, G. R.; Seawright, A. A.; Moore, M. R.; Lam, P. K. S. Cylindrospermopsin, a cyanobacterial alkaloid: evaluation of its toxicologic activity. Therapeutic Drug Monitoring 2000, 22, 89–92. [Google Scholar]
- Humpage, A. R.; Fenech, M.; Thomas, P.; Falconer, I. R. Micronucleus induction and chromosome loss in WIL2-NS cells exposed to the cyanobacterial toxin, cylindrospermopsin. Mutation Research 2000, 472, 155–161. [Google Scholar]
- Falconer, I. R.; Humpage, A. R. Preliminary Evidence for In-Vivo Tumour Initiation by Oral Administration of Extracts of the Blue-Green Alga Cylindrospermopsis raciborskii containing the toxin Cylindrospermopsin. Environmental Toxicology 2001, 16, 192–195. [Google Scholar]
© 2005 MDPI. All rights reserved.
Share and Cite
Falconer, I.R.; Humpage, A.R. Health Risk Assessment of Cyanobacterial (Blue-green Algal) Toxins in Drinking Water. Int. J. Environ. Res. Public Health 2005, 2, 43-50. https://doi.org/10.3390/ijerph2005010043
Falconer IR, Humpage AR. Health Risk Assessment of Cyanobacterial (Blue-green Algal) Toxins in Drinking Water. International Journal of Environmental Research and Public Health. 2005; 2(1):43-50. https://doi.org/10.3390/ijerph2005010043
Chicago/Turabian StyleFalconer, Ian R., and Andrew R. Humpage. 2005. "Health Risk Assessment of Cyanobacterial (Blue-green Algal) Toxins in Drinking Water" International Journal of Environmental Research and Public Health 2, no. 1: 43-50. https://doi.org/10.3390/ijerph2005010043
APA StyleFalconer, I. R., & Humpage, A. R. (2005). Health Risk Assessment of Cyanobacterial (Blue-green Algal) Toxins in Drinking Water. International Journal of Environmental Research and Public Health, 2(1), 43-50. https://doi.org/10.3390/ijerph2005010043



