Self-Reported Medication Use among Pregnant and Postpartum Women during the Third Wave of the COVID-19 Pandemic: A European Multinational Cross-Sectional Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Design and Study Population
2.2. Data Collection
2.3. Use of Medication
2.4. Covariates
2.5. Data Analyses
3. Results
3.1. Characteristics of the Population
3.2. Overall Medication Use
3.3. Self-Medication
3.4. Changes in Chronic Medication Use Related to COVID-19
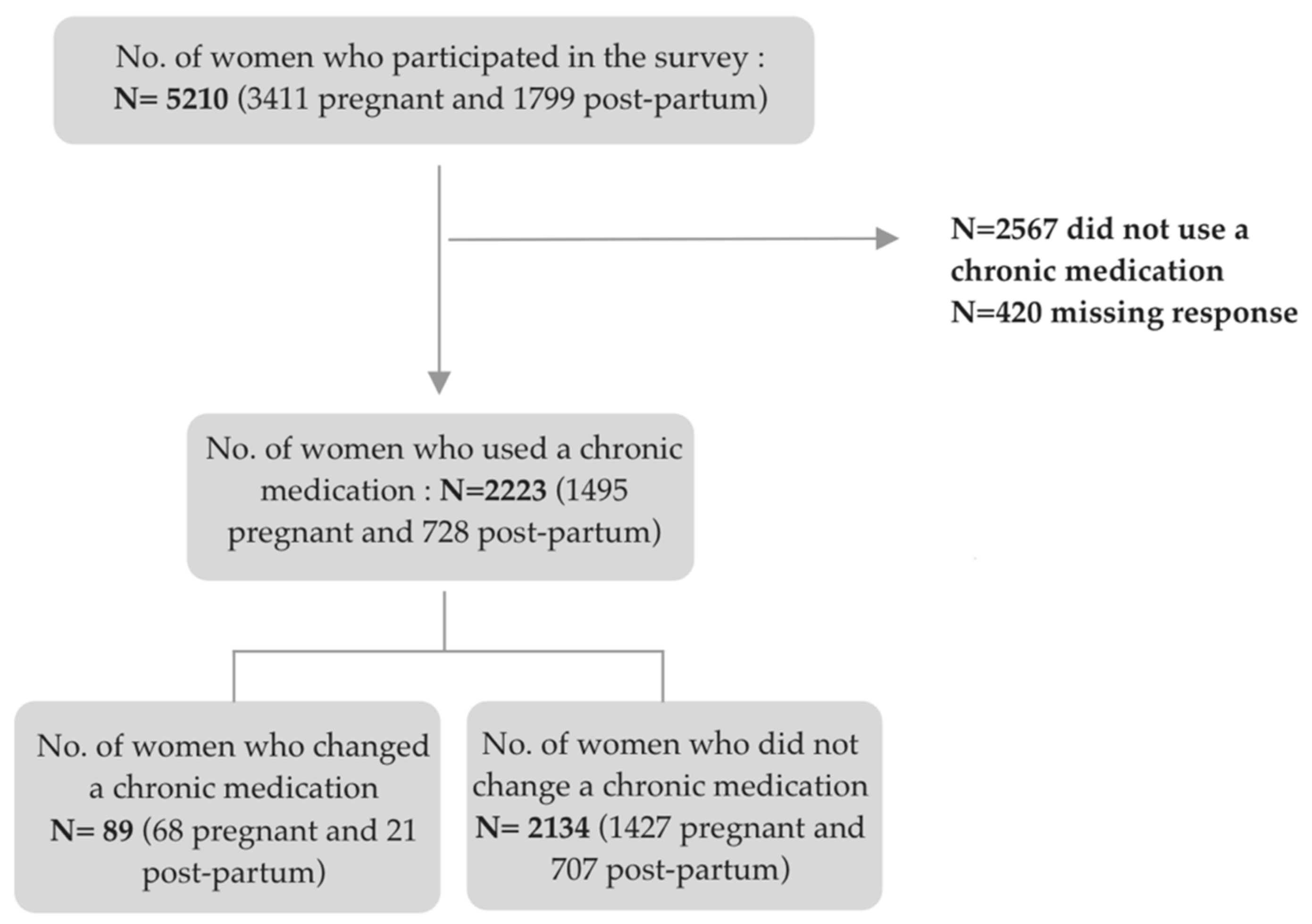
3.5. Factors Associated with Changes of Chronic Medication Due to COVID-19
4. Discussion
4.1. Main Findings
4.2. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wu, T.; Jia, X.; Shi, H.; Niu, J.; Yin, X.; Xie, J.; Wang, X. Prevalence of mental health problems during the COVID-19 pandemic: A systematic review and meta-analysis. J. Affect. Disord. 2021, 281, 91–98. [Google Scholar] [CrossRef]
- Ceulemans, M.; Foulon, V.; Ngo, E.; Panchaud, A.; Winterfeld, U.; Pomar, L.; Lambelet, V.; Cleary, B.; O’Shaughnessy, F.; Passier, A.; et al. Mental health status of pregnant and breastfeeding women during the COVID-19 pandemic-A multinational cross-sectional study. Acta Obstet. Gynecol. Scand. 2021, 100, 1219–1229. [Google Scholar] [CrossRef] [PubMed]
- WHO Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int (accessed on 22 December 2021).
- Nasserie, T.; Hittle, M.; Goodman, S.N. Assessment of the Frequency and Variety of Persistent Symptoms among Patients with COVID-19: A Systematic Review. JAMA Netw. Open 2021, 4, e2111417. [Google Scholar] [CrossRef]
- Bernal, A.J.; da Silva, M.M.G.; Musungaie, D.B.; Kovalchuk, E.; Gonzalez, A.; Reyes, V.D.; Martín-Quirós, A.; Caraco, Y.; Williams-Diaz, A.; Brown, M.L.; et al. Molnupiravir for Oral Treatment of COVID-19 in Nonhospitalized Patients. N. Engl. J. Med. 2022, 386, 509–520. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. EMA Receives Application for Conditional Marketing Authorisation Paxlovid (PF-07321332 and Ritonavir) Treating Patients with COVID-19. 2022. Available online: https://www.ema.europa.eu/en/news/ema-receives-application-conditional-marketing-authorisation-paxlovid-pf-07321332-ritonavir-treating (accessed on 13 January 2022).
- European Medicines Agency. EMA Endorses use of Dexamethasone in COVID-19 Patients on Oxygen or Mechanical Ventilation. 2020. Available online: https://www.ema.europa.eu/en/news/ema-endorses-use-dexamethasone-covid-19-patients-oxygen-mechanical-ventilation (accessed on 13 January 2022).
- Asselah, T.; Durantel, D.; Pasmant, E.; Lau, G.; Schinazi, R.F. COVID-19: Discovery, diagnostics and drug development. J. Hepatol. 2021, 74, 168–184. [Google Scholar] [CrossRef]
- WHO Solidarity Trial Consortium. Repurposed Antiviral Drugs for COVID-19—Interim WHO Solidarity Trial Results. N. Engl. J. Med. 2021, 384, 497–511. [Google Scholar] [CrossRef] [PubMed]
- The Recovery Collaborative Group. Effect of Hydroxychloroquine in Hospitalized Patients with COVID-19. N. Engl. J. Med. 2020, 383, 2030–2040. [Google Scholar] [CrossRef] [PubMed]
- Solidarity Therapeutics Trial Produces Conclusive Evidence on the Effectiveness of Repurposed Drugs for COVID-19 in Record Time. Available online: https://www.who.int/news/item/15-10-2020-solidarity-therapeutics-trial-produces-conclusive-evidence-on-the-effectiveness-of-repurposed-drugs-for-covid-19-in-record-time (accessed on 23 December 2021).
- European Medicines Agency. COVID-19: Reminder of Risk Serious Side Effects with Chloroquine and Hydroxychloroquine. 2020. Available online: https://www.ema.europa.eu/en/news/covid-19-reminder-risk-serious-side-effects-chloroquine-hydroxychloroquine (accessed on 23 December 2021).
- Gavillet, M.; Rolnik, D.L.; Hoffman, M.K.; Panchaud, A.; Baud, D. Should we stop aspirin prophylaxis in pregnant women diagnosed with COVID-19? Ultrasound Obstet. Gynecol. 2020, 55, 843–844. [Google Scholar] [CrossRef]
- Fang, L.; Karakiulakis, G.; Roth, M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir. Med. 2020, 8, e21. [Google Scholar] [CrossRef]
- Kaye, L.; Theye, B.; Smeenk, I. Changes in medication adherence among patients with asthma and COPD during the COVID-19 pandemic. J. Allergy Clin. Immunol. Pract. 2020, 8, 2384–2385. [Google Scholar] [CrossRef]
- Chang, C.; Zhang, L.; Dong, F. Asthma control, self-management, and healthcare access during the COVID-19 epidemic in Beijing. Allergy 2020, 76, 586–588. [Google Scholar] [CrossRef] [PubMed]
- Baptist, A.P.; Lowe, D.; Sarsour, N. Asthma disparities during the COVID-19 pandemic: A survey of patients and physicians. J. Allergy Clin. Immunol. Pract. 2020, 8, 3371–3377. [Google Scholar] [CrossRef] [PubMed]
- Misra, S.; Barron, E.; Vamos, E.; Thomas, S.; Dhatariya, K.; Kar, P.; Young, B.; Khunti, K.; Valabhji, J. Temporal trends in emergency admissions for diabetic ketoacidosis in people with diabetes in England before and during the COVID-19 pandemic: A population-based study. Lancet Diabetes Endocrinol. 2021, 9, 671–680. [Google Scholar] [CrossRef]
- Nune, A.; Iyengar, K.P.; Ahmed, A.; Bilgrami, S.; Sapkota, H.R. Impact of COVID-19 on rheumatology practice in the UK—A pan-regional rheumatology survey. Clin. Rheumatol. 2021, 40, 2499–2504. [Google Scholar] [CrossRef] [PubMed]
- Rabeea, S.A.; Merchant, H.A.; Khan, M.U.; Kow, C.S.; Hasan, S.S. Surging trends in prescriptions and costs of antidepressants in England amid COVID-19. Daru J. Pharm. Sci. 2021, 29, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Estrela, M.; Silva, T.M.; Gomes, E.R.; Piñeiro, M.; Figueiras, A.; Roque, F.; Herdeiro, M.T. Prescription of anxiolytics, sedatives, hypnotics and antidepressants in outpatient, universal care during the COVID-19 pandemic in Portugal: A nationwide, interrupted time-series approach. J. Epidemiol. Community Health 2022, 76, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Campitelli, M.A.; Bronskill, S.E.; Maclagan, L.C.; Harris, D.A.; Cotton, C.A.; Tadrous, M.; Gruneir, A.; Hogan, D.B.; Maxwell, C.J. Comparison of Medication Prescribing Before and After the COVID-19 Pandemic Among Nursing Home Residents in Ontario, Canada. JAMA Netw. Open 2021, 4, e2118441. [Google Scholar] [CrossRef]
- Ceulemans, M.; Foulon, V.; Panchaud, A.; Winterfeld, U.; Pomar, L.; Lambelet, V.; Cleary, B.; O’Shaughnessy, F.; Passier, A.; Richardson, J.L.; et al. Self-Reported Medication Use among Pregnant and Breastfeeding Women during the COVID-19 Pandemic: A Cross-Sectional Study in Five European Countries. Int. J. Environ. Res. Public Health 2022, 19, 1389. [Google Scholar] [CrossRef]
- Ceulemans, M.; Foulon, V.; Panchaud, A.; Winterfeld, U.; Pomar, L.; Lambelet, V.; Cleary, B.; O’Shaughnessy, F.; Passier, A.; Richardson, J.L.; et al. Vaccine Willingness and Impact of the COVID-19 Pandemic on Women’s Perinatal Experiences and Practices—A Multinational, Cross-Sectional Study Covering the First Wave of the Pandemic. Int. J. Environ. Res. Public Health 2021, 18, 3367. [Google Scholar] [CrossRef]
- WHOCC—ATC/DDD Index. Available online: https://www.whocc.no/atc_ddd_index/ (accessed on 17 December 2021).
- Cox, J.L.; Holden, J.M.; Sagovsky, R. Detection of postnatal depression. Development of the 10-item Edinburgh Postnatal Depression Scale. Br. J. Psychiatry 1987, 150, 782–786. [Google Scholar] [CrossRef] [Green Version]
- Spitzer, R.L.; Kroenke, K.; Williams, J.B.W.; Löwe, B. A brief measure for assessing generalized anxiety disorder: The GAD-7. Arch. Intern. Med. 2006, 166, 1092–1097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iborra, I.; Puig, M.; Marín, L.; Calafat, M.; Cañete, F.; Quiñones, C.; González-González, L.; Cardona, G.; Mañosa, M.; Domènech, E. Treatment Adherence and Clinical Outcomes of Patients with Inflammatory Bowel Disease on Biological Agents During the SARS-CoV-2 Pandemic. Dig. Dis. Sci. 2021, 66, 4191–4196. [Google Scholar] [CrossRef] [PubMed]
- Musoke, P.; Nantaayi, B.; Ndawula, R.K.; Wannyana, B.; Ssewante, N.; Wekha, G.; Olum, R.; Nakyagaba, L.; Nassozi, D.R.; Nabukeera, G.; et al. Fear of COVID-19 and the Media Influence on Herbal Medication Use in Uganda: A Cross-Sectional Study. RMHP 2021, 14, 3965–3975. [Google Scholar] [CrossRef] [PubMed]
- Grenard, J.L.; Munjas, B.A.; Adams, J.L.; Suttorp, M.; Maglione, M.; McGlynn, E.A.; Gellad, W.F. Depression and Medication Adherence in the Treatment of Chronic Diseases in the United States: A Meta-Analysis. J. Gen. Intern. Med. 2011, 26, 1175–1182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bautista, L.E.; Vera-Cala, L.M.; Colombo, C.; Smith, P. Symptoms of depression and anxiety and adherence to antihypertensive medication. Am. J. Hypertens. 2012, 25, 505–511. [Google Scholar] [CrossRef] [Green Version]
- Lupattelli, A.; Spigset, O.; Twigg, M.J.; Zagorodnikova, K.; Mårdby, A.C.; Moretti, M.E.; Drozd, M.; Panchaud, A.; Hämeen-Anttila, K.; Rieutord, A.; et al. Medication use in pregnancy: A cross-sectional, multinational web-based study. BMJ Open 2014, 4, e004365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gerbier, E.; Graber, S.M.; Rauch, M.; Marxer, C.A.; Meier, C.R.; Baud, D.; Winterfeld, U.; Blozik, E.; Surbek, D.; Spoendlin, J.; et al. Use of drugs to treat symptoms and acute conditions during pregnancy in outpatient care in Switzerland between 2014 and 2018: Analysis of Swiss healthcare claims data. Swiss. Med. Wkly. 2021, 151, w30048. [Google Scholar] [CrossRef]
- Blix, H.S.; Høye, S. Use of antibiotics during the COVID-19 pandemic. Tidsskr. Den Nor. Legeforening 2021, 141, 4–10. [Google Scholar] [CrossRef]
- Ceulemans, M.; Van Calsteren, K.; Allegaert, K.; Foulon, V. Health products’ and substance use among pregnant women visiting a tertiary hospital in Belgium: A cross-sectional study. Pharmacoepidemiol. Drug Saf. 2019, 28, 1231–1238. [Google Scholar] [CrossRef]
- Okagbue, H.I.; Adamu, P.I.; Bishop, S.A.; Oguntunde, P.E.; Opanuga, A.A.; Akhmetshin, E.M. Systematic Review of Prevalence of Antepartum Depression during the Trimesters of Pregnancy. Open Access Maced. J. Med. Sci. 2019, 7, 1555–1560. [Google Scholar] [CrossRef] [Green Version]
- Woody, C.A.; Ferrari, A.J.; Siskind, D.J.; Whiteford, H.A.; Harris, M.G. A systematic review and meta-regression of the prevalence and incidence of perinatal depression. J. Affect. Disord. 2017, 219, 86–92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gavin, N.I.; Gaynes, B.N.; Lohr, K.N.; Meltzer-Brody, S.; Gartlehner, G.; Swinson, T. Perinatal Depression: A Systematic Review of Prevalence and Incidence. Obstet. Gynecol. 2005, 106, 1071–1083. [Google Scholar] [CrossRef] [PubMed]
- Underwood, L.; Waldie, K.; D’Souza, S.; Peterson, E.R.; Morton, S. A review of longitudinal studies on antenatal and postnatal depression. Arch. Womens Ment. Health 2016, 19, 711–720. [Google Scholar] [CrossRef] [PubMed]
- Lebel, C.; MacKinnon, A.; Bagshawe, M.; Tomfohr-Madsen, L.; Giesbrecht, G. Elevated depression and anxiety symptoms among pregnant individuals during the COVID-19 pandemic. J. Affect. Disord. 2020, 277, 5–13. [Google Scholar] [CrossRef]
- Ferit, D.; Erson, A. Effects of the COVID-19 Pandemic on Anxiety and Depressive Symptoms in Pregnant Women: A Preliminary Study. Available online: https://www.tandfonline.com/doi/epub/10.1080/14767058.2020.1763946?needAccess=true (accessed on 18 January 2022).
- Dennis, C.L.; Falah-Hassani, K.; Shiri, R. Prevalence of antenatal and postnatal anxiety: Systematic review and meta-analysis. Br. J. Psychiatry 2017, 210, 315–323. [Google Scholar] [CrossRef]
- Davies, G.A.; Alsallakh, M.A.; Sivakumaran, S.; Vasileiou, E.; Lyons, R.A.; Robertson, C.; Sheikh, A. Impact of COVID-19 lockdown on emergency asthma admissions and deaths: National interrupted time series analyses for Scotland and Wales. Thorax 2021, 76, 867–873. [Google Scholar] [CrossRef] [PubMed]
- Sousa-Pinto, B.; Heffler, E.; Antó, A.; Czarlewski, W.; Bedbrook, A.; Gemicioglu, B.; Canonica, G.W.; Antó, J.M.; Fonseca, J.A.; Bousquet, J. Anomalous asthma and chronic obstructive pulmonary disease Google Trends patterns during the COVID-19 pandemic. Clin. Transl. Allergy 2020, 10, 47. [Google Scholar] [CrossRef]
- CDC. Public Health Matters Blog. Managing Asthma during COVID-19. Available online: https://blogs.cdc.gov/publichealthmatters/2020/04/asthma/ (accessed on 2 February 2022).
- Schlegl, S.; Maier, J.; Meule, A.; Voderholzer, U. Eating disorders in times of the COVID-19 pandemic-Results from an online survey of patients with anorexia nervosa. Int. J. Eat. Disord. 2020, 53, 1791–1800. [Google Scholar] [CrossRef]
- Ayaz, R.; Hocaoğlu, M.; Günay, T.; Yardımcı, O.D.; Turgut, A.; Karateke, A. Anxiety and depression symptoms in the same pregnant women before and during the COVID-19 pandemic. J. Perinat. Med. 2020, 48, 965–970. [Google Scholar] [CrossRef]
- Basu, A.; Kim, H.H.; Basaldua, R.; Choi, K.W.; Charron, L.; Kelsall, N.; Hernandez-Diaz, S.; Wyszynski, D.F.; Koenen, K.C. A cross-national study of factors associated with women’s perinatal mental health and wellbeing during the COVID-19 pandemic. PLoS ONE 2021, 16, e0249780. [Google Scholar] [CrossRef]
- Mitchell, A.A.; Gilboa, S.M.; Werler, M.M.; Kelley, K.E.; Louik, C.; Hernández-Díaz, S. Medication Use During Pregnancy, With Particular Focus on Prescription Drugs: 1976–2008. Am. J. Obstet. Gynecol. 2011, 205, e1–e51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| All Pregnant Women (n = 3411) | Pregnant Women Who Used at Least One Medication during the Three Previous Months (n = 997) | All Post-Partum Women (n = 1799) | Postpartum Women Who Used at Least One Medication during the Three Previous Months (n = 1161) | |
|---|---|---|---|---|
| N (%) | N (%) | N (%) | N (%) | |
| Baseline characteristics | ||||
| Country | ||||
| Norway | 2376 (69.7) | 408 (40.9) | 1113 (61.9) | 726 (62.5) |
| Belgium | 360 (10.6) | 229 (23.0) | 235 (13.1) | 161 (13.9) |
| United Kingdom | 290 (8.5) | 166 (16.7) | 120 (6.7) | 70 (6.0) |
| Switzerland | 210 (6.2) | 113 (11.3) | 176 (9.8) | 124 (10.7) |
| The Netherlands | 175 (5.1) | 81 (8.1) | 155 (8.6) | 80 (6.9) |
| Maternal age (years) | ||||
| 18–30 | 1374 (40.3) | 387 (38.8) | 675 (37.5) | 473 (40.7) |
| 31–40 | 1707 (50.0) | 544 (54.6) | 897 (49.9) | 631 (54.4) |
| >40 | 68 (2.0) | 35 (3.5) | 44 (2.4) | 31 (2.7) |
| Relationship status | ||||
| Married/cohabiting/partner | 3092 (90.1) | 941 (94.4) | 1595 (88.7) | 1120 (96.5) |
| Single | 57 (1.7) | 25 (2.5) | 21 (1.2) | 15 (1.3) |
| Professional status * | ||||
| Professionally active | 2799 (82.1) | 869 (87.2) | 1430 (79.5) | 1005 (86.6) |
| Not professionally active | 348 (10.2) | 96 (9.6) | 176 (9.8) | 122 (10.5) |
| Working in health care | ||||
| Yes | 906 (26.6) | 327 (32.8) | 486 (27.0) | 348 (30.0) |
| No | 1879 (55.1) | 542 (54.4) | 947 (52.6) | 659 (56.8) |
| Level of education | ||||
| Low (primary school) | 73 (2.1) | 16 (1.6) | 34 (1.9) | 22 (1.9) |
| Medium (high school) | 540 (15.8) | 157 (15.7) | 290 (16.1) | 773 (66.6) |
| High (more than high school) | 2516 (73.8) | 781 (78.3) | 1279 (71.1) | 328 (28.3) |
| Smoked during pregnancy ** | ||||
| Yes | 43 (1.3) | 24 (2.4) | 19 (1.1) | 13 (1.1) |
| No | 3106 (91.1) | 942 (94.5) | 1597 (88.8) | 1122 (96.6) |
| At least one chronic disease | ||||
| Yes | 1190 (34.9) | 467 (46.8) | 476 (26.5) | 456 (39.3) |
| No | 2221 (65.1) | 530 (53.2) | 1323 (73.5) | 705 (60.7) |
| Reproductive health | ||||
| Parity | ||||
| Nulliparous | 87 (3.0) | 528 (53.0) | N/A | N/A |
| Multiparous | 1580 (46.3) | 469 (47.0) | N/A | N/A |
| Planned pregnancy | ||||
| Yes | 2673 (78.4) | 831 (83.4) | N/A | N/A |
| No | 208 (4.4) | 57 (5.7) | N/A | N/A |
| No, but it was not unexpected | 465 (10.4) | 109 (10.9) | N/A | N/A |
| Gestational weeks (GW) | ||||
| First trimester (<14 GW) | 393 (11.5) | 121 (12.1) | N/A | N/A |
| Second trimester (>14 and <28 GW) | 1151 (33.7) | 371 (37.2) | N/A | N/A |
| Third trimester (28 GW-end of pregnancy) | 1802 (52.8) | 505 (50.7) | N/A | N/A |
| Infant’s age | ||||
| ≤6 weeks | N/A | N/A | 674 (37.5) | 447 (38.5) |
| 6–12 weeks | N/A | N/A | 1102 (61.3) | 714 (61.5) |
| Currently breastfeeding | ||||
| Yes | N/A | N/A | 1604 (89.2) | 1050 (90.4) |
| No | N/A | N/A | 172 (9.6) | 111 (9.6) |
| Any breastfeeding experience before the pandemic | ||||
| Yes | N/A | N/A | 660 (36.7) | 437 (37.6) |
| No | N/A | N/A | 942 (52.4) | 613 (52.8) |
| COVID-19 related factors | ||||
| Coronavirus infection status *** | ||||
| Positive test | 163 (4.8) | 63 (6.3) | 100 (5.6) | 58 (5.0) |
| Symptomatic but not tested | 224 (6.6) | 85 (8.5) | 159 (8.8) | 102 (8.8) |
| Negative test or no symptoms | 2958 (86.7) | 849 (85.1) | 1501 (83.4) | 259 (22.3) |
| Infection severity | ||||
| No/mild symptoms | 128 (3.8) | 54 (5.4) | 97 (5.4) | 57 (4.9) |
| Moderate symptoms | 185 (5.4) | 68 (6.8) | 72 (4.0) | 45 (3.9) |
| Hospitalization/long term symptoms | 70 (2.1) | 26 (2.6) | 18 (1.0) | 11 (1.0) |
| Family member infection | ||||
| Yes | 233 (6.8) | 124 (12.4) | 153 (8.5) | 125 (10.8) |
| No | 1262 (37.0) | 488 (49.0) | 575 (32.0) | 487 (42.0) |
| Medication Class (ATC) | Pregnant Women (n = 3411) | Postpartum Women (n = 1799) | Medication Class (ATC) | |
|---|---|---|---|---|
| N (%) | N (%) | |||
| 1 | Analgesics (N02) | 470 (13.8) | 791 (44.0) | Analgesics (N02) |
| Paracetamol (N02BE01) | 355 (10.4) | 630 (35.0) | Paracetamol (N02BE01) | |
| 2 | Antihistamines for systemic use (R06) | 248 (7.3) | 243 (12.0) | Antihistamines for systemic use (R06) |
| Cetirizine (R06AE07) | 62 (1.8) | 73 (4.1) | Cetirizine (R06AE07) | |
| 3 | Drugs for acid related disorder (A02) | 243 (7.1) | 214 (11.9) | Drugs for acid related disorder (A02) |
| Omeprazole (A02BC01) | 50 (1.5) | 87 (4.8) | Not specified | |
| 4 | Antithrombotic agents (B01) | 116 (3.4) | 64 (3.6) | Antithrombotic agents (B01) |
| Acetylsalicylic acid (B01AC06) | 75 (2.2) | 18 (1.0) | Enoxaparin (B01AB05) | |
| 5 | Medications for constipation (A06) | 90 (2.6) | 167 (9.3) | Medications for constipation (A06) |
| Lactulose (A06AD11) | 36 (1.1) | 52 (2.9) | Lactulose (A06AD11) | |
| 6 | Medications for functional gastrointestinal disorders (A03) | 85 (2.5) | 34 (1.9) | Medications for functional gastrointestinal disorders (A03) |
| Metoclopramide (A03FA01) | 25 (0.7) | 19 (1.1) | Not specified | |
| 7 | Medications for obstructive airway diseases (R03) | 79 (2.3) | 70 (3.9) | Medications for obstructive airway diseases (R03) |
| Salbutamol (R03AC02) | 24 (0.7) | 18 (1.0) | Salbutamol (R03AC02) | |
| 8 | Thyroid therapy (H03) | 74 (2.2) | 68 (3.8) | Thyroid therapy (H03) |
| Levothyroxine sodium (H03AA01) | 73 (2.1) | 68 (3.8) | Levothyroxine sodium (H03AA01) | |
| 9 | Antibacterials for systemic use (J01) | 59 (1.7) | 168 (9.3) | Antibacterials for systemic use (J01) |
| Not specified | 27 (0.8) | 87 (4.8) | Not specified | |
| 10 | Sex hormones and modulators of the genital system (G03) | 55 (1.6) | 32 (1.8) | Sex hormones and modulators of the genital system (G03) |
| Desogestrel (G03AC09) | 15 (0.) | 16 (0.9) | Desogestrel (G03AC09) |
| Chronic Medication Users Who Changed a Chronic Medication (n = 89) | Chronic Medication Users Who Did Not Change a Chronic Medication (n = 2134) | Standardized Differences * | |
|---|---|---|---|
| N (%) | N (%) | ||
| Country of response | 0.39 | ||
| Norway | 67 (75.3) | 1361 (63.8) | |
| Belgium | 10 (11.2) | 275 (12.9) | |
| United Kingdom | 6 (6.7) | 179 (8.4) | |
| Switzerland | <5 | 188 (8.8) | |
| Netherlands | 5 (5.6) | 131 (6.1) | |
| Maternal age | 0.18 | ||
| 18–30 | 32 (36.0) | 888 (41.6) | |
| 31–40 | 50 (56.2) | 1178 (55.2) | |
| >40 | 5 (5.6) | 54 (2.5) | |
| Relationship status | 0.16 | ||
| Married/cohabiting/partner | 3 (3.4) | 40 (1.9) | |
| Single | 84 (94.4) | 2080 (97.5) | |
| Professional status | 0.16 | ||
| Professionally active | 79 (88.8) | 1841 (86.3) | |
| Not professionally active | 8 (9.0) | 273 (12.8) | |
| Working in health care | 0.09 | ||
| Yes | 51 (57.3) | 1210 (56.7) | |
| No | 28 (31.5) | 626 (29.3) | |
| Education level | 0.11 | ||
| Low (primary school) | 3 (3.4) | 44 (2.1) | |
| Medium (high school) | 14 (15.7) | 387 (18.1) | |
| High (more than high school) | 70 (78.7) | 1672 (78.4) | |
| At least one chronic disease | 0.23 | ||
| Yes | 63 (70.8) | 1277 (59.8) | |
| No | 26 (29.2) | 857 (40.2) | |
| Smoked during pregnancy or since delivery | 0.18 | ||
| Yes | 3 (3.4) | 32 (1.5) | |
| No | 84 (94.4) | 2088 (97.8) |
| Changed a Chronic Medication (n = 89) | Did Not Change a Chronic Medication (n = 2134) | cOR (95% CI) | aOR (95% CI) * | |
|---|---|---|---|---|
| N (%) | N (%) | |||
| Depression score (EDS) | ||||
| EDS < 13 | 60 (67.4) | 1175 (55.1) | Ref | Ref |
| EDS ≥13 (possible depressive symptoms) | 29 (32.6) | 959 (44.9) | 0.6 (0.4–0.9) | 0.5 (0.3–0.9) ** |
| GAD-7 | ||||
| 0–4 (no anxiety) | 26 (29.2) | 960 (45.0) | Ref | Ref |
| 5–9 (mild) | 45 (50.6) | 765 (35.9) | 2.2 (1.3–3.6) | 2.3 (1.4–3.8) |
| 10–14 (moderate) | 12 (13.5) | 251 (11.8) | 1.8 (0.9–3.6) | 2.0 (1.0–4.2) |
| 15–21 (severe) | 6 (6.7) | 158 (7.4) | 1.4 (0.6–3.5) | 2.0 (0.8–5.1) |
| Perception of COVID-19 severity during pregnancy/breastfeeding compared to non-pregnant/non-breastfeeding women | ||||
| Not more severe/not very risky/not risky et al | 12 (13.5) | 406 (19.0) | Ref | N/A |
| More severe/risky, very risky, extremely risky | 64 (71.9) | 1514 (70.9) | 1.4 (0.8–2.7) | N/A |
| Changes in schedule of persons providing care (midwife, gynecologist)/cancellation or reduction in appointment | ||||
| No | 56 (62.9) | 1507 (70.6) | Ref | Ref |
| Yes | 33 (24.7) | 627 (29.4) | 1.4 (0.9–2.2) | 1.4 (0.9–2.2) |
| Coronavirus infection status 1 | ||||
| Negative test and absence of symptoms | 78 (87.6) | 1870 (87.6) | Ref | N/A |
| Positive test or symptomatic | 11 (12.4) | 264 (12.4) | 1.0 (0.5–1.9) | N/A |
| Close family member infected | ||||
| No | 74 (83.2) | 1763 (82.6) | Ref | N/A |
| Yes | 15 (16.9) | 371 (17.4) | 1.0 (0.6–1.7) | N/A |
| Vaccine status | ||||
| Unwilling to be vaccinated | 28 (31.5) | 683 (32.0) | Ref | N/A |
| Vaccinated/willing to be vaccinated | 46 (51.7) | 1062 (49.8) | 1.1 (0.7–1.7) | N/A |
| Anti-pandemic measures are an infringement of personal freedom | ||||
| Neutral | 14 (15.7) | 275 (12.9) | Ref | Ref |
| Agree/strongly agree | 11 (12.3) | 473 (22.2) | 0.5 (0.2–1.0) | 0.9 (0.5–1.6) |
| Disagree/strongly disagree | 64 (71.9) | 1386 (64.9) | 0.9 (0.5–1.6) | 0.5 (0.2–1.1) |
| Trust in various sources regarding the COVID-19 pandemic | ||||
| In either the media or the Internet | 18 (20.2) | 293 (13.7) | 1.6 (1.0–2.8) | 1.4 (0.8–2.5) |
| In either the government or health authorities | 76 (85.4) | 1714 (80.3) | 1.2 (1.0–1.4) | 1.4 (0.6–2.5) |
| In at least one health care professional 2 | 79 (88.8) | 1820 (85.3) | 1.4 (0.7–2.7) | N/A |
| In either family or friends | 20 (22.5) | 361 (16.9) | 1.5 (0.9–2.4) | 1.6 (1.0–2.8) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gerbier, E.; Favre, G.; Tauqeer, F.; Winterfeld, U.; Stojanov, M.; Oliver, A.; Passier, A.; Nordeng, H.; Pomar, L.; Baud, D.; et al. Self-Reported Medication Use among Pregnant and Postpartum Women during the Third Wave of the COVID-19 Pandemic: A European Multinational Cross-Sectional Study. Int. J. Environ. Res. Public Health 2022, 19, 5335. https://doi.org/10.3390/ijerph19095335
Gerbier E, Favre G, Tauqeer F, Winterfeld U, Stojanov M, Oliver A, Passier A, Nordeng H, Pomar L, Baud D, et al. Self-Reported Medication Use among Pregnant and Postpartum Women during the Third Wave of the COVID-19 Pandemic: A European Multinational Cross-Sectional Study. International Journal of Environmental Research and Public Health. 2022; 19(9):5335. https://doi.org/10.3390/ijerph19095335
Chicago/Turabian StyleGerbier, Eva, Guillaume Favre, Fatima Tauqeer, Ursula Winterfeld, Milos Stojanov, Alison Oliver, Anneke Passier, Hedvig Nordeng, Léo Pomar, David Baud, and et al. 2022. "Self-Reported Medication Use among Pregnant and Postpartum Women during the Third Wave of the COVID-19 Pandemic: A European Multinational Cross-Sectional Study" International Journal of Environmental Research and Public Health 19, no. 9: 5335. https://doi.org/10.3390/ijerph19095335
APA StyleGerbier, E., Favre, G., Tauqeer, F., Winterfeld, U., Stojanov, M., Oliver, A., Passier, A., Nordeng, H., Pomar, L., Baud, D., Panchaud, A., Meyer-Massetti, C., & Ceulemans, M. (2022). Self-Reported Medication Use among Pregnant and Postpartum Women during the Third Wave of the COVID-19 Pandemic: A European Multinational Cross-Sectional Study. International Journal of Environmental Research and Public Health, 19(9), 5335. https://doi.org/10.3390/ijerph19095335








