A Compositional Analysis of Physical Activity, Sedentary Time, and Sleep and Associated Health Outcomes in Children and Adults with Cystic Fibrosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Measurements
2.3. Statistical Analysis
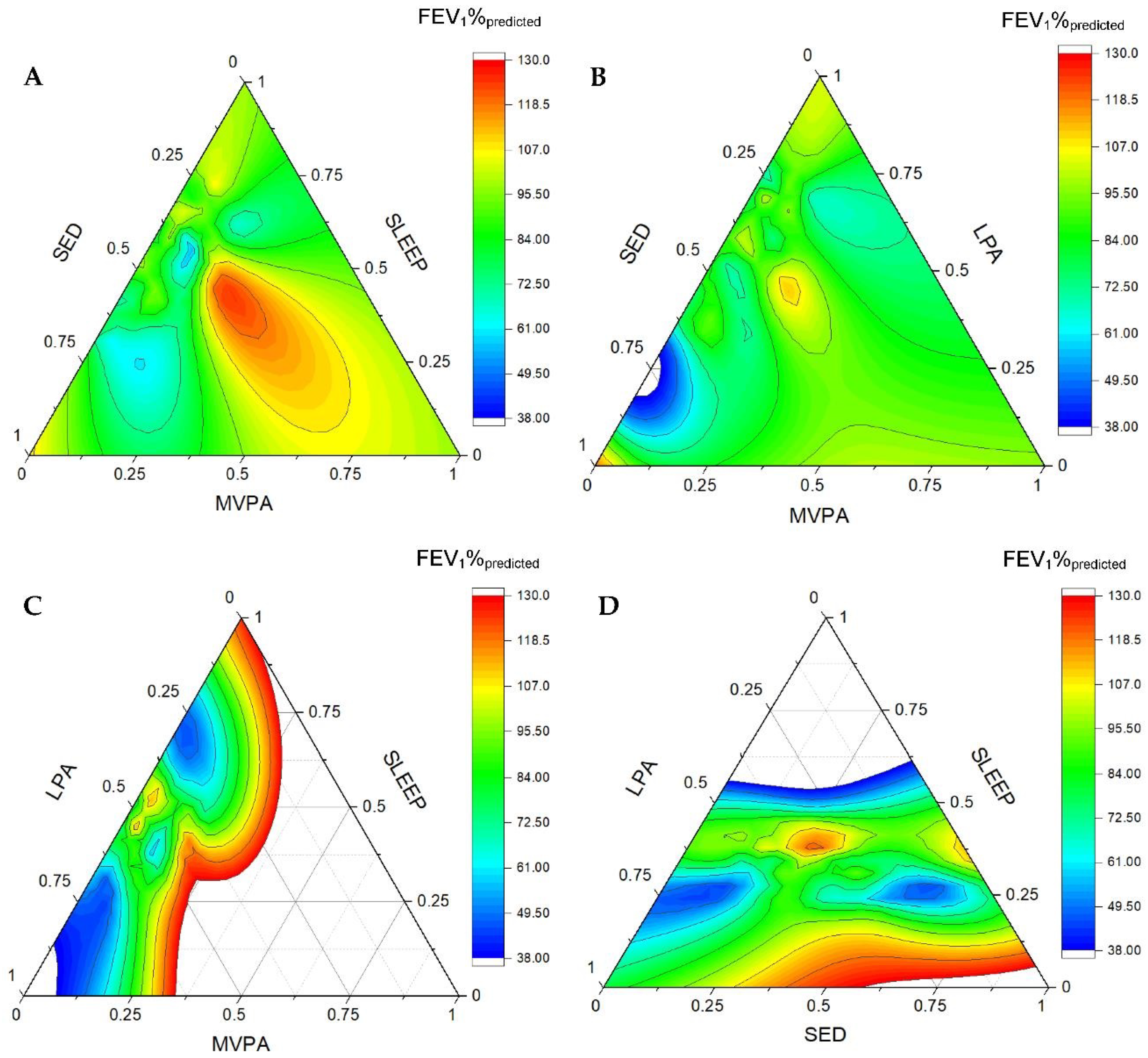
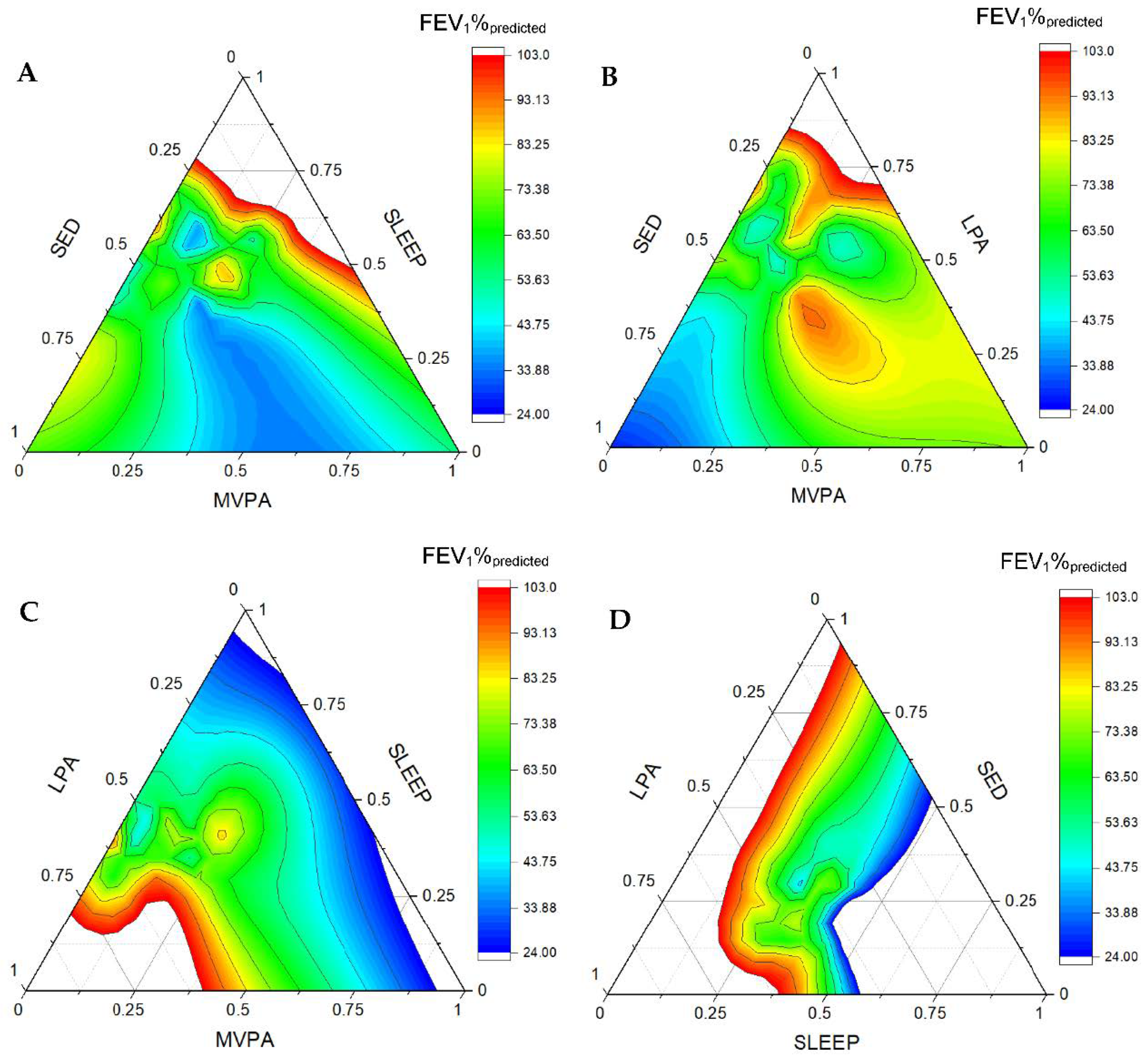
3. Results
Sequential Binary Partitioning
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Trust, C.F. UK Cystic Fibrosis Registry 2018 Annual Data Report; CFT: London, UK, 2018. [Google Scholar]
- Hebestreit, H.; Schmid, K.; Kieser, S.; Junge, S.; Ballmann, M.; Roth, K.; Hebestreit, A.; Schenk, T.; Schindler, C.; Posselt, H.G.; et al. Quality of life is associated with physical activity and fitness in cystic fibrosis. BMC Pulm. Med. 2014, 14, 26. [Google Scholar] [CrossRef] [PubMed]
- Hebestreit, H.; Kieser, S.; Rudiger, S.; Schenk, T.; Junge, S.; Hebestreit, A.; Ballmann, M.; Posselt, H.G.; Kriemler, S. Physical activity is independently related to aerobic capacity in cystic fibrosis. Eur. Respir. J. 2006, 28, 734–739. [Google Scholar] [CrossRef]
- Savi, D.; Simmonds, N.; Di Paolo, M.; Quattrucci, S.; Palange, P.; Banya, W.; Hopkinson, N.S.; Bilton, D. Relationship between pulmonary exacerbations and daily physical activity in adults with cystic fibrosis. BMC Pulm. Med. 2015, 15, 151. [Google Scholar] [CrossRef] [PubMed]
- Cox, N.S.; Alison, J.A.; Button, B.M.; Wilson, J.W.; Morton, J.M.; Holland, A.E. Physical Activity Participation by Adults With Cystic Fibrosis: An Observational Study. Respirology 2016, 21, 511–518. [Google Scholar] [CrossRef]
- Mackintosh, K.A.; Ridgers, N.D.; Evans, R.E.; McNarry, M.A. Physical Activity and Sedentary Time Patterns in Children and Adolescents With Cystic Fibrosis and Age- And Sex-Matched Healthy Controls. J. Phys. Act. Health 2018, 15, 82–88. [Google Scholar] [CrossRef]
- Collaco, J.M.; Blackman, S.M.; Raraigh, K.S.; Morrow, C.B.; Cutting, G.R.; Paranjape, S.M. Self-reported exercise and longitudinal outcomes in cystic fibrosis: A retrospective cohort study. BMC Pulm. Med. 2014, 14, 159. [Google Scholar] [CrossRef]
- Savi, D.; Di Paolo, M.; Simmonds, N.; Onorati, P.; Internullo, M.; Quattrucci, S.; Winston, B.; Laveneziana, P.; Palange, P. Relationship between daily physical activity and aerobic fitness in adults with cystic fibrosis. BMC Pulm. Med. 2015, 15, 59. [Google Scholar] [CrossRef] [PubMed]
- Schneiderman, J.E.; Wilkes, D.L.; Atenafu, E.G.; Nguyen, T.; Wells, G.D.; Alarie, N.; Tullis, E.; Lands, L.C.; Coates, A.L.; Corey, M.; et al. Longitudinal relationship between physical activity and lung health in patients with cystic fibrosis. Eur. Respir. J. 2013, 43, 817–823. [Google Scholar] [CrossRef]
- Cox, N.S.; Alison, J.A.; Button, B.M.; Wilson, J.W.; Morton, J.M.; Holland, A. Accumulating Physical Activity in at Least 10-minute Bouts Predicts Better Lung Function After 3-years in Adults With Cystic Fibrosis. ERJ Open Res. 2018, 4, 00095–2017. [Google Scholar] [CrossRef]
- Radtke, T.; Nevitt, S.J.; Hebestreit, H.; Kriemler, S. Physical Exercise Training for Cystic Fibrosis. Cochrane Database Syst. Rev. 2017, 11, CD002768. [Google Scholar] [CrossRef]
- Carson, V.; Hunter, S.; Kuzik, N.; Gray, C.E.; Poitras, V.J.; Chaput, J.P.; Saunders, T.J.; Katzmarzyk, P.T.; Okely, A.D.; Connor Gorber, S.; et al. Systematic review of sedentary behaviour and health indicators in school-aged children and youth: An update. Appl. Physiol. Nutr. Metab. 2016, 41, S240–S265. [Google Scholar] [CrossRef] [PubMed]
- Chastin, S.F.; Palarea-Albaladejo, J.; Dontje, M.L.; Skelton, D. Combined Effects of Time Spent in Physical Activity, Sedentary Behaviors and Sleep on Obesity and Cardio-Metabolic Health Markers: A Novel Compositional Data Analysis Approach. PLoS ONE 2015, 10, e0139984. [Google Scholar] [CrossRef] [PubMed]
- Dumuid, D.; Stanford, T.E.; Martin-Fernandez, J.A.; Pedisic, Z.; Maher, C.A.; Lewis, L.K.; Hron, K.; Katzmarzyk, P.T.; Chaput, J.P.; Fogelholm, M.; et al. Compositional data analysis for physical activity, sedentary time and sleep research. Stat. Methods Med. Res. 2018, 27, 3726–3738. [Google Scholar] [CrossRef] [PubMed]
- McGregor, D.E.; Palarea-Albaladejo, J.; Dall, P.M.; Del Pozo Cruz, B.; Chastin, S.F. Compositional Analysis of the Association Between Mortality and 24-hour Movement Behaviour From NHANES. Eur. J. Prev. Cardiol. 2019, 28, 791–798. [Google Scholar] [CrossRef]
- McGregor, D.E.; Palarea-Albaladejo, J.; Dall, P.; Stamatakis, E.; Chastin, S. Differences in physical activity time-use composition associated with cardiometabolic risks. Prev. Med. Rep. 2019, 13, 23–29. [Google Scholar] [CrossRef]
- Swindell, N.; Paul, R.; Mikael, F.; Mathijs, D.; Ian, M.; Martinez, J.A.; Santiago, N.-C.; Teodora, H.-D.; Nadka, B.; Georgi, B.; et al. Compositional analysis of the associations between 24-h movement behaviours and cardio-metabolic risk factors in overweight and obese adults with pre-diabetes from the PREVIEW study: Cross-sectional baseline analysis. Int. J. Behav. Nutr. Phys. Act. 2020, 17, 29. [Google Scholar] [CrossRef]
- Moore, V.C. Spirometry: Step by step. Breathe 2012, 8, 232–240. [Google Scholar] [CrossRef]
- Graham, B.L.; Steenbruggen, I.; Miller, M.R.; Barjaktarevic, I.Z.; Cooper, B.G.; Hall, G.L.; Hallstrand, T.S.; Kaminsky, D.A.; McCarthy, K.; McCormack, M.C.; et al. Standardization of Spirometry 2019 Update. An Official American Thoracic Society and European Respiratory Society Technical Statement. Am. J. Respir. Critial Care 2019, 2019, 200. [Google Scholar] [CrossRef]
- Quanjer, P.H.; Stanojevic, S.; Cole, T.J.; Baur, X.; Hall, G.L.; Culver, B.H.; Enright, P.L.; Hankinson, J.L.; Ip, M.S.; Zheng, J.; et al. Multi-ethnic reference values for spirometry for the 3-95-yr age range: The global lung function 2012 equations. Eur. Respir. J. 2012, 40, 1324–1343. [Google Scholar] [CrossRef]
- Davies, J.C.; Alton, E.W. Monitoring Respiratory Disease Severity in Cystic Fibrosis. Respir. Care 2009, 54, 606–617. [Google Scholar] [CrossRef]
- Migueles, J.H.; Rowlands, A.V.; Huber, F.; Sabia, S.; van Hees, V.T. GGIR: A Research Community–Driven Open Source R Package for Generating Physical Activity and Sleep Outcomes From Multi-Day Raw Accelerometer Data in: Journal for the Measurement of Physical Behaviour. J. Meas. Phys. Behav. 2019, 2, 188–196. [Google Scholar] [CrossRef]
- Haszard, J.J.; Meredith-Jones, K.; Farmer, V.; Williams, S.; Galland, B.; Taylor, R. Non-Wear Time and Presentation of Compositional 24-Hour Time-Use Analyses Influence Conclusions About Sleep and Body Mass Index in Children. Human Kinet. 2020, 3, 204–210. [Google Scholar] [CrossRef]
- Van Hees, V.T.; Gorzelniak, L.; Dean León, E.C.; Eder, M.; Pias, M.; Taherian, S.; Ekelund, U.; Renström, F.; Franks, P.W.; Horsch, A.; et al. Separating Movement and Gravity Components in an Acceleration Signal and Implications for the Assessment of Human Daily Physical Activity. PLoS ONE 2013, 8, e61691. [Google Scholar] [CrossRef]
- Hildebrand, M.; van Hees, V.T.; Hansen, B.H.; Ekelund, U. Age group comparability of raw accelerometer output from wrist- and hip-worn monitors. Med. Sci. Sports Exerc. 2014, 46, 3. [Google Scholar] [CrossRef]
- van Hees, V.T.; Sabia, S.; Anderson, K.N.; Denton, S.J.; Oliver, J.; Catt, M.; Abell, J.G.; Kivimäki, M.; Trenell, M.I.; Singh, M. A Novel, Open Access Method to Assess Sleep Duration Using a Wrist-Worn Accelerometer. PLoS ONE 2015, 10, e142533. [Google Scholar] [CrossRef]
- Van Hees, V.T.; Sabia, S.; Jones, S.E.; Wood, A.R.; Anderson, K.N.; Kivimäki, M.; Frayling, T.M.; Pack, A.I.; Bucan, M.; Trenell, M.I.; et al. Estimating sleep parameters using an accelerometer without sleep diary. Sci. Rep. 2018, 8, 12975. [Google Scholar] [CrossRef]
- Aitchison, J. The Statistical Analysis of Compositional Data. J. R. Stat. Soc. Ser. B 1982, 44, 139–177. [Google Scholar] [CrossRef]
- Van de Boogaart, K.G.; Tolosana-Delgado, R. “compositions”: A unified R package to analyze compositional data. Comput. Geosci. 2008, 34, 320–338. [Google Scholar] [CrossRef]
- Stevens, M.L.; Crowley, P.; Rasmussen, C.L.; Hallman, D.M.; Mortensen, O.S.; Nygård, C.H.; Holtermann, A. Accelerometer-Measured Physical Activity at Work and Need for Recovery: A Compositional Analysis of Cross-sectional Data. Ann. Work Expo. Health 2020, 64, 138–151. [Google Scholar] [CrossRef] [PubMed]
- Pawlowsky-Glahn, V.; Egozcue, J.J.; Tolosana-Delgado, R. Modeling and Analysis of Compositional Data; John Wiley & Sons: Hoboken, NJ, USA, 2015. [Google Scholar]
- Carson, V.; Tremblay, M.S.; Chaput, J.P.; Chastin, S. Associations Between Sleep Duration, Sedentary Time, Physical Activity, and Health Indicators Among Canadian Children and Youth Using Compositional Analyses. Appl. Physiol. Nutr. Metab. 2016, 41, S294–S302. [Google Scholar] [CrossRef]
- Štefelová, N.; Dygrýn, J.; Hron, K.; Gába, A.; Rubín, L.; Palarea-Albaladejo, J. Robust Compositional Analysis of Physical Activity and Sedentary Behaviour Data. Int. J. Environ. Res. Public Health 2018, 15, 2248. [Google Scholar] [CrossRef] [PubMed]
- Liou, T.G.; Elkin, E.P.; Pasta, D.J.; Jacobs, J.R.; Konstan, M.W.; Morgan, W.J.; Wagener, J.S. Year-to-year changes in lung function in individuals with cystic fibrosis. J. Cyst. Fibros. 2010, 9, 250–256. [Google Scholar] [CrossRef] [PubMed]
- De Boeck, K.; Zolin, A. Year to year change in FEV1 in patients with cystic fibrosis and different mutation classes. J. Cyst. Fibros. 2017, 16, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Mogayzel, P.J.; Naureckas, E.T.; Robinson, K.A.; Mueller, G.; Hadjiliadis, D.; Hoag, J.B.; Lubsch, L.; Hazle, L.; Sabadosa, K.; Marshall, B. Cystic fibrosis pulmonary guidelines. Chronic medications for maintenance of lung health. Am. J. Respir. Crit. Care Med. 2013, 187, 680–689. [Google Scholar] [CrossRef] [PubMed]
- Wainwright, C.E.; Elborn, J.S.; Ramsey, B.W.; Marigowda, G.; Huang, X.; Cipolli, M.; Colombo, C.; Davies, J.C.; De Boeck, K.; Flume, P.A.; et al. Lumacaftor-Ivacaftor in Patients with Cystic Fibrosis Homozygous for Phe508del CFTR. N. Engl. J. Med. 2015, 373, 1–3. [Google Scholar] [CrossRef]
- Elkins, M.R.; Robinson, M.; Rose, B.R.; Harbour, C.; Moriarty, C.P.; Marks, G.B.; Belousova, E.G.; Xuan, W.; Bye, P.T. A controlled trial of long-term inhaled hypertonic saline in patients with cystic fibrosis. N. Engl. J. Med. 2006, 354, 229–240. [Google Scholar] [CrossRef]
- Carson, V.; Tremblay, M.S.; Chaput, J.P.; McGregor, D.; Chastin, S. Compositional Analyses of the Associations Between Sedentary Time, Different Intensities of Physical Activity, and Cardiometabolic Biomarkers Among Children and Youth From the United States. PLoS ONE 2019, 14, e220009. [Google Scholar] [CrossRef]
- Shakkottai, A.; O’Brien, L.M.; Nasr, S.Z.; Chervin, R.D. Sleep Disturbances and their Impact in Pediatric Cystic Fibrosis. Sleep Med. Rev. 2018, 42, 100–110. [Google Scholar] [CrossRef]
- Barbosa, R.R.B.; Coelho, P.F.; Liberato, F.M.G.; Vidal, P.R.; Carvalho, R.B.C.O.; Melotti, R.C.N.C.; Donadio, M.V.F. Sleep disorders are distinctively associated with exercise intolerance and sedentary behavior in children with cystic fibrosis. Sleep Med. 2020, 74, 145–151. [Google Scholar] [CrossRef]
- Cox, N.S.; Eldridge, B.; Rawlings, S.; Dreger, J.; Corda, J.; Hauser, J.; Button, B.M.; Bishop, J.; Nichols, A.; Middleton, A.; et al. A web-based intervention to promote physical activity in adolescents and young adults with cystic fibrosis: Protocol for a randomized controlled trial. BMC Pulm. Med. 2019, 19, 253. [Google Scholar] [CrossRef]
- Dietz-Terjung, S.; Gruber, W.; Sutharsan, S.; Taube, T.; Olivier, M.; Mellies, U.; Koerner-Rettberg, C.; Dillenhöfer, S.; Stehling, F.; Matthias, W. Association between habitual physical activity (HPA) and sleep quality in patients with cystic fibrosis. Sleep Breath. 2020, 25, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, M.S.; Carson, V.; Chaput, J.P.; Connor Gorber, S.; Dinh, T.; Duggan, M.; Faulkner, G.; Gray, C.E.; Gruber, R.; Janson, K.; et al. Canadian 24-Hour Movement Guidelines for Children and Youth: An Integration of Physical Activity, Sedentary Behaviour, and Sleep. Appl. Physiol. Nutr. Metab. 2016, 41, S311–S327. [Google Scholar] [CrossRef] [PubMed]
- Cox, N.S.; Holland, A. Current Perspectives of Physical Activity in Cystic Fibrosis. Expert Rev. Respir. Med. 2019, 13, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Shelley, J.; Boddy, L.M.; Knowles, Z.R.; Stewart, C.E.; Dawson, E.A. Physical Activity and Associations With Clinical Outcome Measures in Adults With Cystic Fibrosis; A Systematic Review. J. Cyst. Fibros. 2019, 18, 590–601. [Google Scholar] [CrossRef]
- Pelclová, J.; Štefelová, N.; Dumuid, D.; Pedišić, Ž.; Hron, K.; Gába, A.; Olds, T.; Pechová, J.; Zając-Gawlak, I.; Tlučáková, L. Are longitudinal reallocations of time between movement behaviours associated with adiposity among elderly women? A compositional isotemporal substitution analysis. Int. J. Obes. 2020, 44, 857–864. [Google Scholar] [CrossRef]
- Biddle, G.J.H.; Edwardson, C.L.; Henson, J.; Davies, M.J.; Khunti, K.; Rowlands, A.V.; Yates, T. Associations of Physical Behaviours and Behavioural Reallocations With Markers of Metabolic Health: A Compositional Data Analysis. Int. J. Environ. Res. Public Health 2018, 15, 2280. [Google Scholar] [CrossRef]
- Taylor-Robinson, D.; Whitehead, M.; Diderichsen, F.; Olesen, H.V.; Pressler, T.; Smyth, R.L.; Diggle, P. Understanding the natural progresion in %FEV1 decline in patients with cystic fibrosis: A longitudinal study. Thorax 2012, 67, 860–866. [Google Scholar] [CrossRef]
- Aznar, S.; Gallardo, C.; Fiuza-Luces, C.; Santana-Sosa, E.; López-Mojares, L.M.; Santalla, A.; Rodríguez-Romo, G.; Pérez, M.; Garatachea, N.; Lucia, A. Levels of moderate--vigorous physical activity are low in Spanish children with cystic fibrosis: A comparison with healthy controls. J. Cyst. Fibros 2014, 13, 335–340. [Google Scholar] [CrossRef]
- Troosters, T.; Langer, D.; Vrijsen, B.; Segers, J.; Wouters, K.; Janssens, W.; Gosselink, R.; Decramer, M.; Dupont, L. Skeletal Muscle Weakness, Exercise Tolerance and Physical Activity in Adults With Cystic Fibrosis. Eur. Respir. J. 2009, 33, 99–106. [Google Scholar] [CrossRef]
- Arikan, H.; Yatar, İ.; Calik-Kutukcu, E.; Aribas, Z.; Saglam, M.; Vardar-Yagli, N.; Savci, S.; Inal-Ince, D.; Ozcelik, U.; Kiper, N. A Comparison of Respiratory and Peripheral Muscle Strength, Functional Exercise Capacity, Activities of Daily Living and Physical Fitness in Patients With Cystic Fibrosis and Healthy Subjects. Res. Dev. Disabil. 2015, 45, 147–156. [Google Scholar] [CrossRef]
- Polito, R.; Nigro, E.; Elce, A.; Monaco, M.L.; Iacotucci, P.; Carnovale, V.; Comegna, M.; Gelzo, M.; Zarrilli, F.; Corso, G.; et al. Adiponectin Expression Is Modulated by Long-Term Physical Activity in Adult Patients Affected by Cystic Fibrosis. Mediat. Inflamm. 2019, 2019, 2153934. [Google Scholar] [CrossRef]
- Owen, N.; Sparling, P.B.; Healy, G.N.; Dunstan, D.W.; Matthews, C.E. Sedentary Behavior: Emerging Evidence for a New Health Risk. Mayo Clin. Proc. 2010, 85, 1138–1141. [Google Scholar] [CrossRef] [PubMed]
- González, K.; Fuentes, J.; Márquez, J.L. Physical Inactivity, Sedentary Behavior and Chronic Diseases. Korean J. Fam. Med. 2017, 38, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Ortega, F.B.; Konstabel, K.; Pasquali, E.; Ruiz, J.R.; Hurtig-Wennlöf, A.; Mäestu, J.; Löf, M.; Harro, J.; Bellocco, R.; Labayen, I.; et al. Objectively Measured Physical Activity and Sedentary Time during Childhood, Adolescence and Young Adulthood: A Cohort Study. PLoS ONE 2013, 8, e60871. [Google Scholar] [CrossRef]
- Shei, R.J.; Mackintosh, K.A.; Peabody Lever, J.E.; McNarry, M.A.; Krick, S. Exercise Physiology Across the Lifespan in Cystic Fibrosis. Front. Physiol. 2019, 10, 1382. [Google Scholar] [CrossRef] [PubMed]
- Kühnhausen, J.; Dirk, J.; Schmiedek, F. Individual Classification of Elementary School Children’s Physical Activity: A Time-Efficient, Group-Based Approach to Reference Measurements. Behav. Res. Methods 2017, 49, 685–697. [Google Scholar] [CrossRef]
- Bianchim, M.S.; McNarry, M.A.; Evans, R.; Thia, L.; Barker, A.R.; Williams, C.A.; Denford, S.; Mackintosh, K.A. Calibration and Cross-validation of Accelerometry in Children and Adolescents with Cystic Fibrosis. Meas. Phys. Educ. Exerc. Sci. 2022. [Google Scholar] [CrossRef]
- Hildebrand, M.; Hansen, B.H.; van Hees, V.T.; Ekelund, U. Evaluation of Raw Acceleration Sedentary Thresholds in Children and Adults. Scand. J. Med. Sci. Sports 2017, 27, 1814–1823. [Google Scholar] [CrossRef]
| Children | Adults | |||||
|---|---|---|---|---|---|---|
| Variable (Unit) | Overall (n = 86) | Girls (n = 41) | Boys (n = 45) | Overall (n = 43) | Females (n = 21) | Males (n = 22) |
| Age (years) | 13.6 ± 2.8 | 13.7 ± 2.7 | 13.5 ± 2.8 | 24.6 ± 4.7 | 23.6 ± 3.5 | 25.5 ± 5.5 |
| Height (cm) | 154.2 ± 14.9 1 | 152.5 ± 13.1 | 155. ± 16.13 | 166.0 ± 28.7 | 153.2 ± 35.6 2 | 178.1 ± 10.0 |
| BMI (kg·m−2) | 18.7 ± 3.4 1 | 18.8 ± 2.5 | 18.6 ± 4.1 | 21.2 ± 4.4 | 20.0 ± 5.3 | 22.4 ± 2.9 |
| zBMI | −0.2 ± 0.9 | −0.1 ± 0.8 | −0.3 ± 1.1 | - | - | - |
| FEV1 (l) | 2.3 ± 0.8 | 2.1 ± 0.7 2 | 2.5 ± 0. 7 | 2.4 ± 1.0 | 1.9 ± 0.7 2 | 2.9 ± 1.0 |
| FEV1%predicted (%) | 86 ± 21 1 | 84 ± 24 | 88 ± 18 | 63 ± 21 | 62 ± 1 | 64 ± 21 |
| Sleep (min) | 479.3 ± 70.6 1 | 471.2 ± 63.4 | 487.0 ± 77.5 | 453. 3 ± 64.5 | 452.1 ± 68.0 | 454.5 ± 62.6 |
| SED (min) | 344.8 ± 165.7 | 382.1 ± 196.0 | 313.7 ± 123.1 | 341.5 ± 116.0 | 381.3 ± 122.9 2 | 303 ± 97.1 |
| LPA (min) | 562.0 ± 140.5 | 543.7 ± 167.9 | 574.6 ± 105.5 | 529.6 ± 121.1 | 491.3 ± 125.7 2 | 566.3 ± 106.8 |
| MVPA (min) | 53.7 ± 115.3 1 | 42.8 ± 32.0 2 | 64.5 ± 53.0 | 115.3 ± 83.6 | 115.1 ± 68.6 | 115.5 ± 97.5 |
| Sleep | SED | LPA | MVPA | |
|---|---|---|---|---|
| Children | ||||
| Unadjusted (min) | 477.2 ± 74.2 | 345.0 ± 166.5 | 561.7 ± 136.3 | 56.0 ± 50.3 |
| Compositional | 0.34 | 0.22 | 0.39 | 0.02 |
| Sleep | __ | 0.05 | 0.02 | −0.15 |
| SED | 0.05 | __ | −0.05 | −0.34 |
| LPA | 0.02 | −0.05 | __ | −0.11 |
| MVPA | −0.15 | −0.34 | −0.11 | __ |
| Adults | ||||
| Unadjusted (min) | 457.9 ± 61.8 | 331.1 ± 98.9 | 528.8 ± 114.3 | 122.0 ± 84.2 |
| Compositional | 0.33 | 0.23 | 0.37 | 0.06 |
| Sleep | __ | 0.06 | 0.03 | −0.18 |
| SED | 0.06 | __ | 0.02 | −0.25 |
| LPA | 0.03 | 0.02 | __ | −0.16 |
| MVPA | −1.18 | −0.25 | −0.16 | __ |
| Children | Adults | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Non-Adjusted | Adjusted | Non-Adjusted | Adjusted | ||||||||||
| Retained ilr Coordinate | Clinical Covariates | Regression Coefficient | Standard Error | p-Value | Regression Coefficient | Standard Error | p-Value | Regression Coefficient | Standard Error | p-Value | Regression Coefficient | Standard Error | p-Value |
| Sleep | 13.22 | 4.46 | 0.004 * | 12.77 | 4.80 | 0.009 * | −7.68 | 9.61 | 0.43 | −16.66 | 10.64 | 0.13 | |
| SED | −2.85 | 1.65 | 0.08 | −2.97 | 1.77 | 0.09 | −4.27 | 4.93 | 0.39 | 2.04 | 6.63 | 0.76 | |
| MVPA | LPA | −3.56 | 2.18 | 0.10 | −3.04 | 2.26 | 0.18 | 4.40 | 4.39 | 0.32 | 3.19 | 4.85 | 0.51 |
| Sex | N/A | N/A | N/A | 6.86 | 5.08 | 0.18 | N/A | N/A | N/A | 10.27 | 11.01 | 0.35 | |
| Age | N/A | N/A | N/A | −1.35 | 0.77 | 0.08 | N/A | N/A | N/A | −1.52 | 0.99 | 0.13 | |
| Genotype | N/A | N/A | N/A | 2.46 | 5.15 | 0.63 | N/A | N/A | N/A | 14.49 | 8.83 | 0.11 | |
| SED | −7.09 | 2.30 | 0.002 * | −7.06 | 2.49 | 0.006 * | −1.46 | 6.50 | 0.82 | 7.48 | 8.59 | 0.39 | |
| LPA | −8.84 | 3.57 | 0.01 * | −8.15 | 3.75 | 0.03 * | 8.15 | 7.33 | 0.27 | 10.28 | 7.55 | 0.18 | |
| Sleep | MVPA | −8.19 | 3.05 | 0.009 * | −8.05 | 3.32 | 0.01 * | 5.30 | 6.32 | 0.40 | 11.41 | 7.07 | 0.11 |
| Sex | N/A | N/A | N/A | 6.86 | 5.08 | 0.18 | N/A | N/A | N/A | 10.27 | 11.01 | 0.35 | |
| Age | N/A | N/A | N/A | −1.35 | 0.77 | 0.08 | N/A | N/A | N/A | −1.52 | 0.99 | 0.13 | |
| Genotype | N/A | N/A | N/A | 2.46 | 5.15 | 0.63 | N/A | N/A | N/A | 14.49 | 8.83 | 0.11 | |
| LPA | −5.97 | 3.12 | 0.05 | −5.33 | 3.26 | 0.10 | 8.17 | 6.49 | 0.21 | 7.19 | 6.99 | 0.31 | |
| MVPA | −2.27 | 1.59 | 0.15 | −2.28 | 1.73 | 0.19 | 3.92 | 2.92 | 0.19 | 4.64 | 2.93 | 0.12 | |
| SED | Sleep | 12.44 | 3.89 | 0.002 * | 12.15 | 4.20 | 0.005 * | −3.80 | 9.23 | 0.68 | −14.78 | 11.10 | 0.19 |
| Sex | N/A | N/A | N/A | 6.86 | 5.08 | 0.08 | N/A | N/A | N/A | 10.27 | 11.01 | 0.35 | |
| Age | N/A | N/A | N/A | −1.35 | 0.77 | 0.08 | N/A | N/A | N/A | −1.52 | 0.99 | 0.13 | |
| Genotype | N/A | N/A | N/A | 2.46 | 5.15 | 0.63 | N/A | N/A | N/A | 14.49 | 8.83 | 0.11 | |
| MVPA | −0.15 | 1.27 | 0.90 | −0.37 | 1.36 | 0.78 | 0.97 | 2.12 | 0.64 | 1.97 | 2.26 | 0.39 | |
| Sleep | 13.97 | 4.61 | 0.003 * | 13.41 | 4.94 | 0.008 * | −7.80 | 10.04 | 0.44 | −16.97 | 11.07 | 0.13 | |
| LPA | SED | −0.68 | 2.11 | 0.74 | −1.05 | 2.23 | 0.63 | −5.90 | 5.53 | 0.29 | 0.17 | 7.2 | 0.98 |
| Sex | N/A | N/A | N/A | 6.86 | 5.08 | 0.08 | N/A | N/A | N/A | 10.27 | 11.01 | 0.35 | |
| Age | N/A | N/A | N/A | −1.35 | 0.77 | 0.08 | N/A | N/A | N/A | −1.52 | 0.99 | 0.13 | |
| Genotype | N/A | N/A | N/A | 2.46 | 5.15 | 0.63 | N/A | N/A | N/A | 14.49 | 8.83 | 0.11 | |
| Children | Adults | |||||
|---|---|---|---|---|---|---|
| Reallocation | 10 min | 20 min | 30 min | 10 min | 20 min | 30 min |
| Sleep to SED | −1.14 (−1.61–−0.73) | −2.27 (−3.27–−1.41) | −3.41 (−4.98–−2.05) | 0.38 (−0.28–0.89) | 0.78 (−0.94–2.09) | 1.20 (−1.92–3.55) |
| Sleep to LPA | −0.87 (−1.30–−0.50) | −1.76 (−2.66–−0.97) | −2.65 (−4.07–−1.42) | 0.98 (0.72–1.18) | 1.98 (1.15–2.59) | 2.96 (1.32–4.20) |
| Sleep to MVPA | −0.72 (−1.45–−0.08) | −1.44 (−2.96–−0.12) | −2.18 (−4.51–−0.14) | 0.75 (0.06–1.27) | 1.50 (−0.08–2.69) | 2.24 (−0.39–4.23) |
| SED to Sleep | 1.14 (1.55–0.78) | 2.28 (3.02–1.62) | 3.42 (4.43 –2.54) | −0.37 (−0.15–−0.53) | −0.72 (−0.76–−0.69) | −1.06 (−1.83–−0.48) |
| SED to LPA | 0.28 (0.31–0.25) | 0.57 (0.62–0.53) | 0.89 (0.94–0.84) | 0.60 (0.86–0.41) | 1.20 (1.53–0.95) | 1.80 (2.02–1.12) |
| SED to MVPA | 0.43 (0.17–0.66) | 0.89 (0.32 –1.38) | 1.37 (0.49–2.13) | 0.37 (0.18–0.52) | 0.73 (1.08–0.52) | 1.08 (0.11–1.81) |
| LPA to Sleep | 0.86 (1.24–0.53) | 1.72 (2.42–1.11) | 2.56 (3.52–1.72) | −0.98 (−1.02–−0.95) | −1.96 (−2.34–−1.67) | −2.94 (−3.95–−2.17) |
| LPA to SED | −0.26 (−0.30–−0.22) | −0.50 (−0.60–−0.42) | −0.73 (−0.90–−0.58) | −0.61 (−1.02–−0.29) | −1.22 (−2.19–−0.48) | −1.84 (−3.50–−0.58) |
| LPA to MVPA | 0.16 (−0.16 –0.44) | 0.33 (−0.37–0.94) | 0.51 (−0.59–1.46) | −0.24 (−0.76–0.15) | −0.51 (−1.65–0.36) | −0.80 (−2.66–0.61) |
| MVPA to Sleep | 0.72 (1.30–0.21) | 1.44 (2.27–0.71) | 2.17 (2.60–1.80) | −0.76 (−0.32–−1.10) | −1.53 (−0.94–−1.98) | −2.33 (−1.92–−2.63) |
| MVPA to SED | −0.41 (−0.23–−0.56) | −0.78 (−0.64–−0.91) | −1.12 (−1.46–−0.82) | −0.39 (−0.34–−0.43) | −0.80 (−0.85–−0.76) | −1.23 (−1.55–−0.98) |
| MVPA to LPA | −0.14 (0.05–−0.31) | −0.27 (−0.14–−0.38) | −0.36 (−0.81–0.04) | 0.21 (0.60–−0.08) | 0.39 (1.00–−0.07) | 0.53 (1.16–0.06) |
| Children | Adults | |||||||
|---|---|---|---|---|---|---|---|---|
| Reallocation | Homozygous | Heterozygous | Homozygous | Heterozygous | ||||
| Girls (27) | Boys (22) | Girls (14) | Boys (23) | Females (9) | Males (11) | Females (12) | Males (11) | |
| Sleep to SED | −2.44 (−3.56–−1.76) | −2.46 (−3.38–−1.71) | −3.04 (−3.45–−1.69) | −2.38 (−3.27–−1.65) | −3.58 (−4.71–−2.64) | −3.24 (−4.49–−2.53) | −3.40 (−4.50–−2.50) | −3.24 (−4.35–0.97) |
| Sleep to LPA | −1.59 (−2.47–−1.10) | −1.64 (−2.08–−1.18) | −1.29 (−2.52–−0.95) | −1.59 (−2.26–−1.03) | −2.39 (−3.28–−1.61) | −2.10 (−2.80–−1.69) | −2.27 (−3.33–−1.39) | −2.16 (−3.08–1.21) |
| Sleep to MVPA | −1.11 (−2.88–0.33) | −1.07 (−2.47–3.32) | −1.66 (−2.65–0.11) | −1.03 (−2.26–−0.10) | −1.56 (−2.46–−0.73) | −1.98 (−1.83–−1.69) | −1.48 (−2.15–−0.83) | −1.41 (−1.63–2.36) |
| SED to Sleep | 2.56 (3.01–2.09) | 2.46 (2.86–2.14) | 1.87 (3.05–2.01) | 2.38 (2.76–1.96) | 3.58 (4.30–3.22) | 3.40 (3.69–3.09) | 3.24 (4.11–3.05) | 3.24 (3.62–6.52) |
| SED to LPA | 0.98 (1.10–0.77) | 0.94 (1.30–0.64) | 0.35 (0.93–0.84) | 0.91 (1.51–0.21) | 1.37 (1.43–1.46) | 1.29 (1.60–1.13) | 1.13 (1.37–1.39) | 1.23 (1.27–4.72) |
| SED to MVPA | 0.73 (−0.41–1.54) | 0.59 (−0.26–1.28) | 0.12 (−0.27–1.27) | 0.57 (0–1.03) | 0.85 (0.82–1.02) | 0.97 (1.03–0.70) | 0.81 (1.17–0.83) | 0.77 (1.27–4.02) |
| LPA to Sleep | 1.71 (1.92–1.43) | 1.53 (1.56–2.50) | 0.94 (1.99–1.16) | 1.47 (2.89–1.24) | 2.22 (2.66–2.05) | 2.10 (2.17–2.11) | 2.11 (2.94–1.66) | 2.00 (2.36–5.41) |
| LPA to SED | −0.73 (−1.23–−0.55) | −0.82 (−1.30–−0.43) | −1.40 (−0.93–−0.74) | −0.79 (−1.13–−0.62) | −1.19 (−1.43–0.88) | −1.13 (−1.64–−0.70) | −1.13 (−0.98–−0.97) | −1.08 (−1.27–2.64) |
| LPA to MVPA | 1.59 (0.68–2.20) | 1.53 (0.78–2.03) | 0.94 (0.80–1.90) | 1.36 (1.01–1.75) | 2.05 (2.25–2.05) | 2.10 (2.55–1.69) | 1.94 (2.54–1.80) | 2.00 (2.54–4.99) |
| MVPA to Sleep | 0.98 (2.33–−0.33) | 0.82 (1.69–0.11) | 0.23 (1.99–−0.11) | 0.79 (1.51–0.21) | 1.19 (1.02–1.61) | 1.29 (0.08–1.97) | 1.13 (0.78–1.66) | 1.08 (0–5.55) |
| MVPA to SED | −1.47 (−0.68–−2.31) | −1.53 (−1.04–−1.82) | −2.11 (−0.93–−2.01) | −1.47 (−1.26–−1.65) | −2.22 (−3.07–−1.46) | −1.94 (−3.54–−98) | −2.11 (−3.13–−1.25) | −2.00 (−3.62–2.77) |
| MVPA to LPA | −0.005 (0.41–−1.54) | −0.006 (0.13–−1.28) | −0.011 (−0.13–−1.16) | −0.006 (−0.38–−0.93) | −0.006 (−1.84–−0.29) | −0.005 (−2.21–0) | −0.006 (−1.96–0) | −0.006 (−2.54–3.74) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bianchim, M.S.; McNarry, M.A.; Holland, A.; Cox, N.S.; Dreger, J.; Barker, A.R.; Williams, C.A.; Denford, S.; Mackintosh, K.A. A Compositional Analysis of Physical Activity, Sedentary Time, and Sleep and Associated Health Outcomes in Children and Adults with Cystic Fibrosis. Int. J. Environ. Res. Public Health 2022, 19, 5155. https://doi.org/10.3390/ijerph19095155
Bianchim MS, McNarry MA, Holland A, Cox NS, Dreger J, Barker AR, Williams CA, Denford S, Mackintosh KA. A Compositional Analysis of Physical Activity, Sedentary Time, and Sleep and Associated Health Outcomes in Children and Adults with Cystic Fibrosis. International Journal of Environmental Research and Public Health. 2022; 19(9):5155. https://doi.org/10.3390/ijerph19095155
Chicago/Turabian StyleBianchim, Mayara S., Melitta A. McNarry, Anne Holland, Narelle S. Cox, Julianna Dreger, Alan R. Barker, Craig A. Williams, Sarah Denford, and Kelly A. Mackintosh. 2022. "A Compositional Analysis of Physical Activity, Sedentary Time, and Sleep and Associated Health Outcomes in Children and Adults with Cystic Fibrosis" International Journal of Environmental Research and Public Health 19, no. 9: 5155. https://doi.org/10.3390/ijerph19095155
APA StyleBianchim, M. S., McNarry, M. A., Holland, A., Cox, N. S., Dreger, J., Barker, A. R., Williams, C. A., Denford, S., & Mackintosh, K. A. (2022). A Compositional Analysis of Physical Activity, Sedentary Time, and Sleep and Associated Health Outcomes in Children and Adults with Cystic Fibrosis. International Journal of Environmental Research and Public Health, 19(9), 5155. https://doi.org/10.3390/ijerph19095155









