Phase One of a Global Evaluation of Suction-Based Airway Clearance Devices in Foreign Body Airway Obstructions: A Retrospective Descriptive Analysis
Abstract
1. Introduction
2. Methods
3. Data Collection
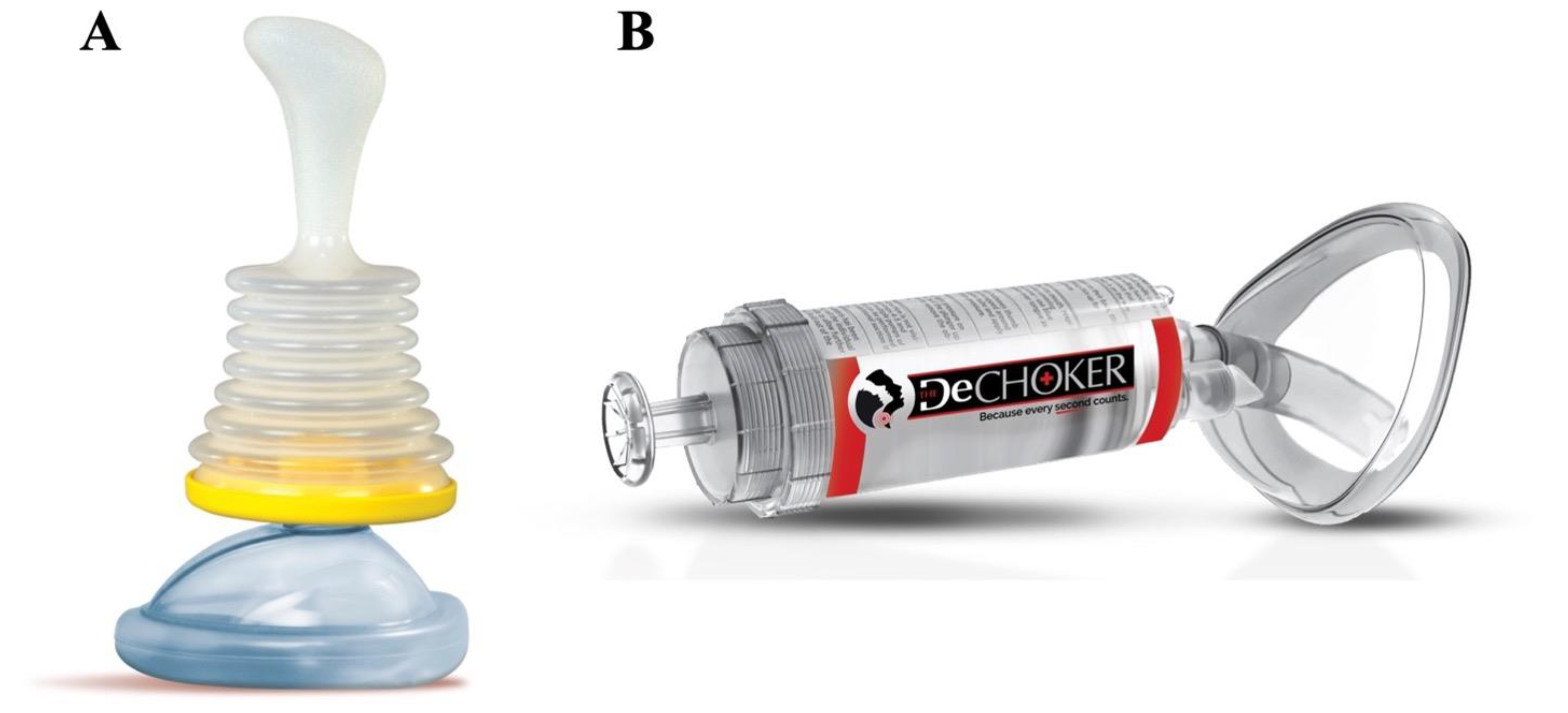
3.1. Non-Invasive ACD
3.2. Minimally Invasive ACD
3.3. Variables
3.4. Outcomes
3.5. Data Analysis
4. Results
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Disclaimer
References
- Health Canada. Canadian Injury Data: Mortality 2005 and Hospitalizations, 2001–2005; Government of Canada: Ottawa, ON, Canada, 2011. [Google Scholar]
- Injury Facts. Preventable Death and Death Rates per 100,000 Population in the Home and Community by Cause and Age Group, United States; National Safety Council: Itasca, IL, USA, 2017; Available online: https://injuryfacts.nsc.org/home-and-community/home-and-community-overview/deaths-in-the-home-and-community-by-age-group-and-cause/ (accessed on 5 August 2021).
- Institute for Health Metrics and Evaluation. Disease and Risk Factor Summaries: Pulmonary Aspiration and Foreign Body in Airway; The Lancet: London, UK, 2019; Available online: https://www.thelancet.com/pb-assets/Lancet/gbd/summaries/diseases/pulmonary-aspiration.pdf (accessed on 5 August 2021).
- Fridman, L.; Fraser-Thomas, J.; Pike, I.; Macpherson, A.K. An interprovincial comparison of unintentional childhood injury rates in Canada for the period 2006–2012. Can. J. Public Health 2018, 109, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Norii, T.; Igarashi, Y.; Sung-Ho, K.; Nagata, S.; Tagami, T.; Yoshino, Y.; Hamaguchi, T.; Maejima, R.; Nakao, S.; Albright, D.; et al. Protocol for a nationwide prospective, observational cohort study of foreign-body airway obstruction in Japan: The MOCHI registry. BMJ Open 2020, 10, e039689. [Google Scholar] [CrossRef] [PubMed]
- Injury Facts. Preventable-Injury-Related Deaths by Sex, Age and Cause, United States, 1999–2019; National Safety Council: Itasca, IL, USA, 2019; Available online: https://injuryfacts.nsc.org/all-injuries/deaths-by-demographics/sex-age-and-cause/data-details/ (accessed on 5 August 2021).
- Centers for Disease Control and Prevention. Non-fatal choking-related episodes among children—United States, 2001. MMWR 2002, 51, 945–948. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5142a1.htm (accessed on 4 January 2022).
- Couper, K.; Abu Hassan, A.; Ohri, V.; Patterson, E.; Tang, H.T.; Bingham, R.; Olasveengen, T.; Perkins, G.D. Removal of foreign body airway obstruction: A systematic review of interventions. Resuscitation 2020, 156, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Couper, K.; Abu Hassan, A.; Ohri, V.; Patterson, E.; Tang, H.T.; Bingham, R.; Perkins, G.D.; Avis, S.; Brooks, S.; Castren, M.; et al. Consensus on Science with Treatment Recommendations: Removal of Foreign Body Airway Obstruction; International Liaison Committee on Resuscitation (ILCOR): Niel, Belgium, 2020; Available online: https://costr.ilcor.org/document/removal-of-foreign-body-airway-obstruction-tfsr-costr (accessed on 5 August 2021).
- Soroudi, A.; Shipp, H.E.; Stepanski, B.M. Adult foreign body airway obstruction in the prehospital setting. Prehosp. Emerg. Care 2007, 11, 25–29. [Google Scholar] [CrossRef] [PubMed]
- LifeVac LLC. Lifevac; LifeVac LLC: New York, NY, USA, 2021; Available online: https://www.lifevac.net (accessed on 5 August 2021).
- DeChoker LLC. Dechoker; DeChoker LLC: Wheat Ridge, CO, USA, 2021; Available online: https://www.dechoker.com (accessed on 5 August 2021).
- ExtraLife. Lifewand; ExtraLife: New York, NY, USA, 2021; Available online: https://lifewand.com (accessed on 5 August 2021).
- Dunne, C.; Peden, A.; Queiroga, A.; Gonzalez, C.G.; Valesco, B.; Szpilman, D. A systematic review on the effectiveness of anti-choking suction devices and identification of research gaps. Resuscitation 2020, 153, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Dunne, C.L.; Queiroga, A.C.; Szpilman, D.; Viguers, K.; Osman, S.; Peden, A.E. A protocol for the prospective evaluation of novel suction-based airway clearance devices in the treatment of foreign body airway obstructions. Cureus 2022, 14, e20981. [Google Scholar] [CrossRef]
- Laerdal Medical. V-Vac™ Manual Suction Unit; Laerdal Medical Canada Ltd.: Toronto, ON, Canada, 2021; Available online: https://laerdal.com/ca/doc/123/V-VAC-Manual-Suction-Unit (accessed on 5 August 2021).
- LifeVac LLC. LifeVac Saved a Life Report; LifeVac LLC: New York, NY, USA, 2021; Available online: https://lifevac.net/lifevac-saved-a-life-report/ (accessed on 30 August 2021).
- American Heart Association. Part 3: Adult basic life support. Circulation 2000, 102, I22–I59. [Google Scholar] [CrossRef]
- Perkins, G.D.; Handley, A.J.; Koster, R.W.; Castrén, M.; Smyth, M.A.; Olasveengen, T.; Monsieurs, K.G. European Resuscitation Council Guidelines for Resuscitation 2015. Section 2. Adult basic life support and automated external defibrillation. Resuscitation 2015, 95, 81–99. [Google Scholar] [CrossRef] [PubMed]
- Safar, P. Resuscitation and brain ischemia. In Brain Failure and Resuscitation; Grenvik, A., Safar, P., Eds.; Churchill Livingstone: New York, NY, USA, 1981; pp. 155–184. [Google Scholar]
- Bhanderi, B.G.; Hill, S.P. Evaluation of DeChoker, an Airway Clearance Device (ACD) Used in Adult Choking Emergencies within the Adult Care Home Sector: A Mixed Methods Case Study. Front. Public Health 2020, 8, e541885. [Google Scholar] [CrossRef] [PubMed]
- Gal, L.L.; Pugleisi, P.; Peterman, D. Resuscitation of choking victims in a pediatric population using a novel portable non-powered suction device: Real world data. Pediatr. Ther. 2020, 10, e371. [Google Scholar] [CrossRef]
- Smith, M. Biased Sample and Extrapolation, Common Mistakes in Using Statistics: Spotting and Avoiding Them. 2012. Available online: https://web.ma.utexas.edu/users/mks/statmistakes/biasedsampling.html (accessed on 5 August 2021).
- Hoopeer, A.J.; Tiballs, J. Comparison of a Trigger Tool and voluntary reporting to identify adverse events in a paediatric intensive care unit. Aneasth. Intensive Care 2014, 42, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Weingart, S.N.; Callanan, L.D.; Ship, A.N.; Aronson, M.D. A physician-based voluntary reporting system for adverse events and medical errors. J. Gen. Intern. Med. 2001, 16, 809–814. [Google Scholar] [CrossRef]
- Ramaswamy, A.T.; Done, A.; Solis, R.; Evangelista, L.; Belafsky, P. The efficacy of two commercially available devices to relieve acute foreign body aspiration. In Proceedings of the Annual Meeting ABEA.
- Redding, J.S. The choking controversy: A critique of the Heimlich. Crit. Care Med. 1979, 7, 475–479. [Google Scholar] [CrossRef]
- Heimlich, H.J. A Life-Saving Maneuver to Prevent Food Choking. JAMA 1975, 234, 398–401. [Google Scholar] [CrossRef] [PubMed]
| Non-Invasive ACD (LifeVac©) N = 124 | Minimally Invasive ACD (DeChoker©) N = 61 | ||
|---|---|---|---|
| Patient Gender (n, %) | |||
| M | 56 (45.2) | 24 (39.3) | |
| F | 66 (53.2) | 36 (59.0) | |
| Not recorded | 2 (1.6) | 1 (1.6) | |
| Patient age (median, IQR) | 40 (2–80) | 73 (5–84) | |
| Patient age groups (n, %) | |||
| 0–1 years | 19 (15.3) | 5 (8.2) | |
| 1–5 years | 27 (21.8) | 9 (14.8) | |
| 6–18 years | 9 (7.3) | 8 (13.1) | |
| 18–64 years | 22 (17.7) | 6 (9.8) | |
| 65–80 years | 13 (10.9) | 10 (16.4) | |
| 80+ years | 34 (27.4) | 23 (37.7) | |
| Pre-existing medical conditions (n, %) | |||
| Cardiovascular disease | 4 (3.2) | 0 (0.0) | |
| Neurocognitive disorder | 48 (38.7) | 7 (11.5) | |
| Physical disability | 32 (25.8) | 2 (3.2) | |
| Respiratory disease | 1 (0.8) | 1 (1.6) | |
| Wheelchair use | 18 (14.5) | 2 (3.2) | |
| Other | 16 (12.9) | 1 (1.6) | |
| None | 47 (37.9) | - * | |
| Not recorded | 8 (6.5) | 48 (78.7) | |
| Known history of dysphagia or aspiration (n, %) | |||
| Yes | 17 (13.7) | 3 (4.8) | |
| Not recorded | 107 (84.3) | 58 (95.2) | |
| Non-Invasive ACD LifeVac© (N = 124) | Minimally Invasive ACD Dechoker© (N = 61) | ||
|---|---|---|---|
| Severity of FBAO (n, %) | |||
| Partial | 34 (27.4) | 5 (8.2) | |
| Complete | 52 (41.9) | 8 (13.1) | |
| Unresponsive | 24 (19.4) | 11 (18.0) | |
| Not recorded | 14 (11.3) | 37 (60.7) | |
| Geographical location of FBAO (n, %) | |||
| Home | 28 (22.6) | 21 (34.4) | |
| School/Daycare | 1 (0.8) | 0 (0.0) | |
| Long-term care facility/Nursing home | 45 (36.3) | 24 (39.3) | |
| Other | 11 (8.9) | 2 (3.3) | |
| Not recorded | 39 (31.5) | 14 (23.0) | |
| Foreign body (n, %) | |||
| Food | 105 (84.7) | 56 (91.8) | |
| Toy | 1 (0.8) | 1 (1.6) | |
| Other | 18 (14.5) | 4 (6.6) | |
| Non-Invasive ACD LifeVac© (N = 124) | Minimally Invasive ACD Dechoker© (N = 61) | ||
|---|---|---|---|
| Pre-ACD Intervention | |||
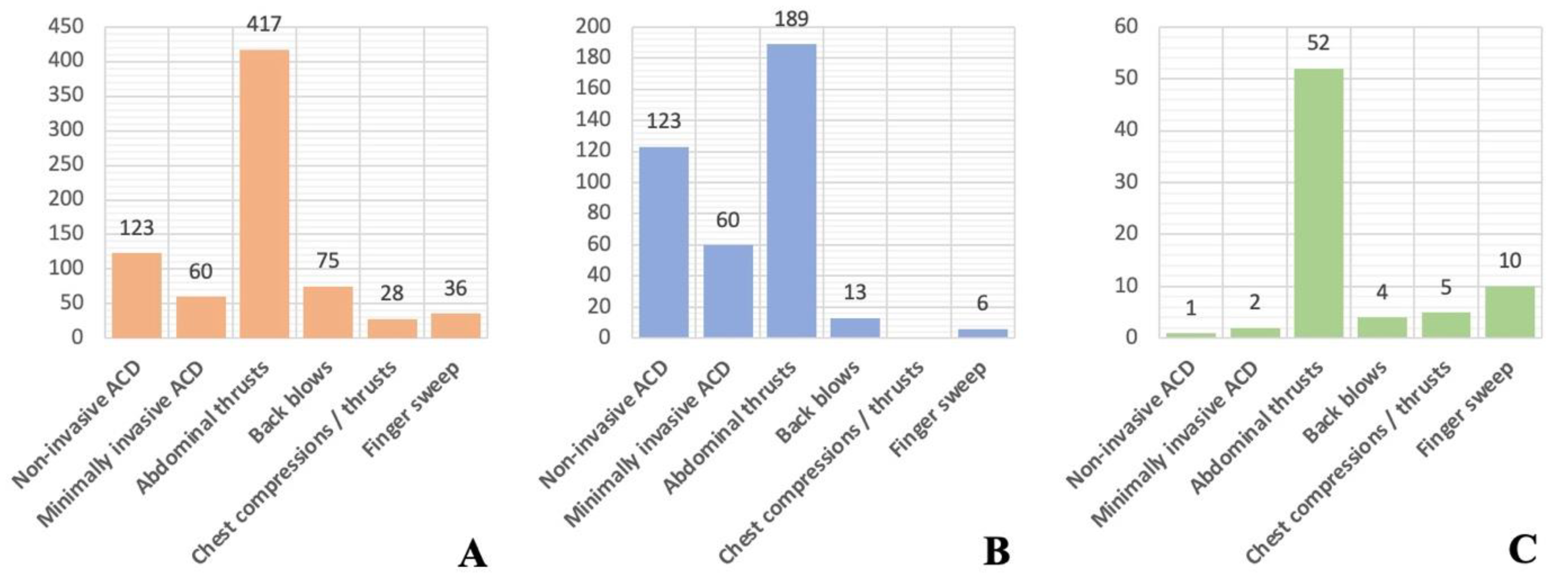
| Abdominal thrusts | 47 (37.9) | 19 (31.1) | |
| Back blows | 49 (39.5) | 25 (41.0) | |
| Chest thrusts or compressions | 4 (3.2) | 2 (3.3) | |
| Finger / mouth sweep | 9 (7.3) | 4 (6.6) | |
| Multiple interventions | 25 (20.2) | 15 (24.6) | |
| No intervention | 11 (8.9) | 10 (16.4) | |
| Not recorded | 31 (25.0) | 17 (27.9) | |
| ACD User | |||
| Relative | 42 (33.8) | 22 (36.1) | |
| Healthcare worker | 12 (9.7) | 2 (3.3) | |
| Self | 1 (0.8) | 0 (0.0) | |
| Other | 10 (8.1) | 21 (34.4) | |
| Not recorded | 59 (47.6) | 16 (26.2) | |
| Median number of ACD attempts to FBAO relief (IQR; range) | 2 (1–3; 1–12) | 2 (1–4; 1–12) | |
| Effectiveness Outcomes | |||
| No Further Intervention Required Post-ACD | 123 | 60 | |
| Survival | 123 | 59 * | |
| Safety Outcomes | |||
| EMS called | 33 (42.9) 1 | 13 (35.1) 2 | |
| Hospital admission | 9 (13.6) 3 | 1 (2.8) 4 | |
| Adverse events reported | 1 (1.1) 5 | 2 (5.4) 2 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dunne, C.L.; Osman, S.; Viguers, K.; Queiroga, A.C.; Szpilman, D.; Peden, A.E. Phase One of a Global Evaluation of Suction-Based Airway Clearance Devices in Foreign Body Airway Obstructions: A Retrospective Descriptive Analysis. Int. J. Environ. Res. Public Health 2022, 19, 3846. https://doi.org/10.3390/ijerph19073846
Dunne CL, Osman S, Viguers K, Queiroga AC, Szpilman D, Peden AE. Phase One of a Global Evaluation of Suction-Based Airway Clearance Devices in Foreign Body Airway Obstructions: A Retrospective Descriptive Analysis. International Journal of Environmental Research and Public Health. 2022; 19(7):3846. https://doi.org/10.3390/ijerph19073846
Chicago/Turabian StyleDunne, Cody L., Selena Osman, Kayla Viguers, Ana Catarina Queiroga, David Szpilman, and Amy E. Peden. 2022. "Phase One of a Global Evaluation of Suction-Based Airway Clearance Devices in Foreign Body Airway Obstructions: A Retrospective Descriptive Analysis" International Journal of Environmental Research and Public Health 19, no. 7: 3846. https://doi.org/10.3390/ijerph19073846
APA StyleDunne, C. L., Osman, S., Viguers, K., Queiroga, A. C., Szpilman, D., & Peden, A. E. (2022). Phase One of a Global Evaluation of Suction-Based Airway Clearance Devices in Foreign Body Airway Obstructions: A Retrospective Descriptive Analysis. International Journal of Environmental Research and Public Health, 19(7), 3846. https://doi.org/10.3390/ijerph19073846






