The Gram-Negative Bacilli Isolated from Caves—Sphingomonas paucimobilis and Hafnia alvei and a Review of Their Involvement in Human Infections
Abstract
1. Introduction
2. Materials and Methods
2.1. Cave Samples Collection and Bacterial Identification
2.1.1. Cave Samples Collection
2.1.2. Bacteriological Identification and Antibiotic Phenotype of the Cave Isolates
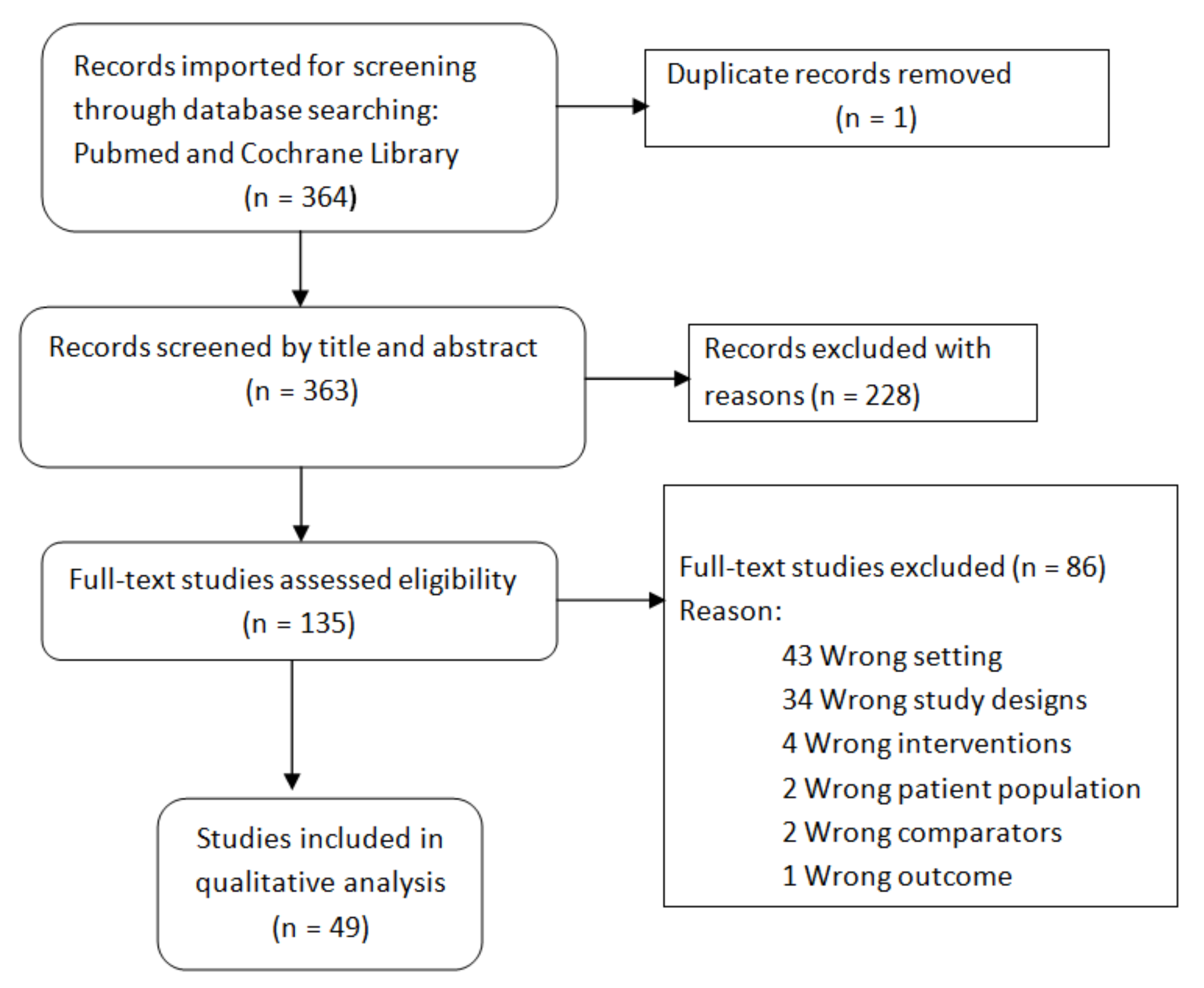
2.2. Search Strategy for Literature Review
References Collection, Screening, and Selection
2.3. Statistical Analysis
3. Results
3.1. The Cave Isolates
3.2. The Results of the Literature Review
3.2.1. Type of Studies
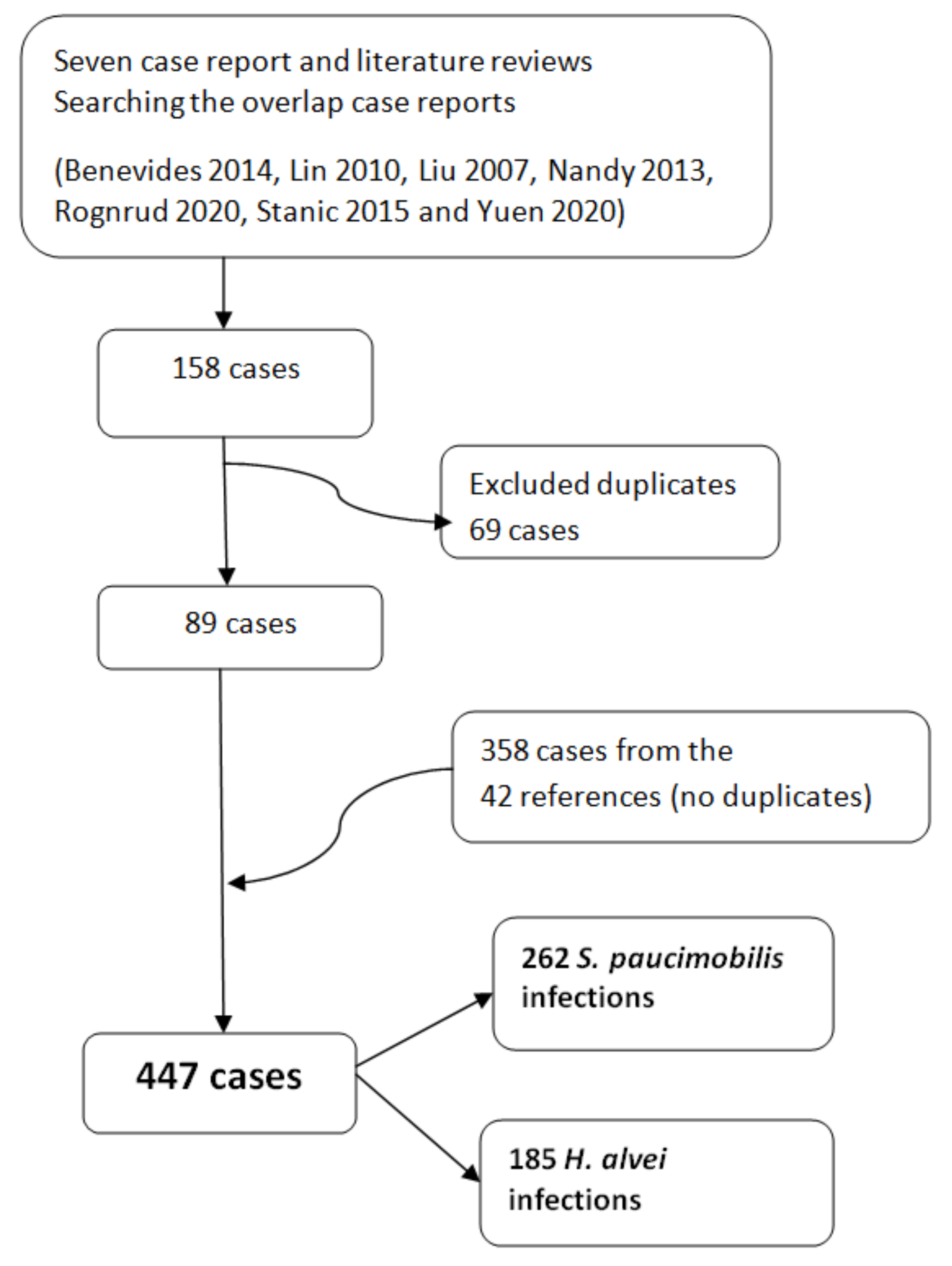
3.2.2. The Case Report and Literature Reviews
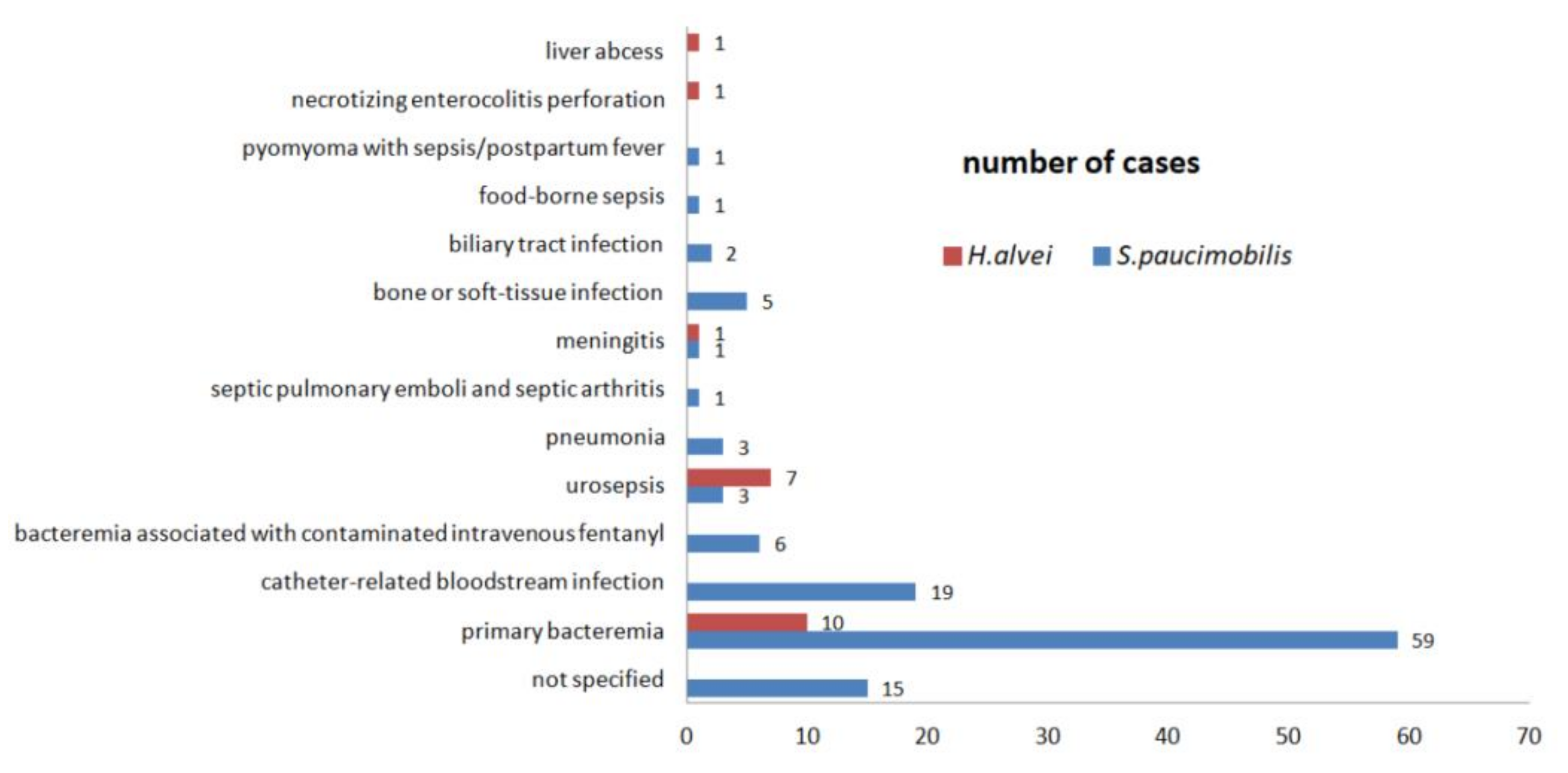
3.2.3. Type of Infections
3.2.4. The Bloodstream Infections
3.2.5. The Urinary Tract Infections
3.2.6. The Respiratory Tract Infections
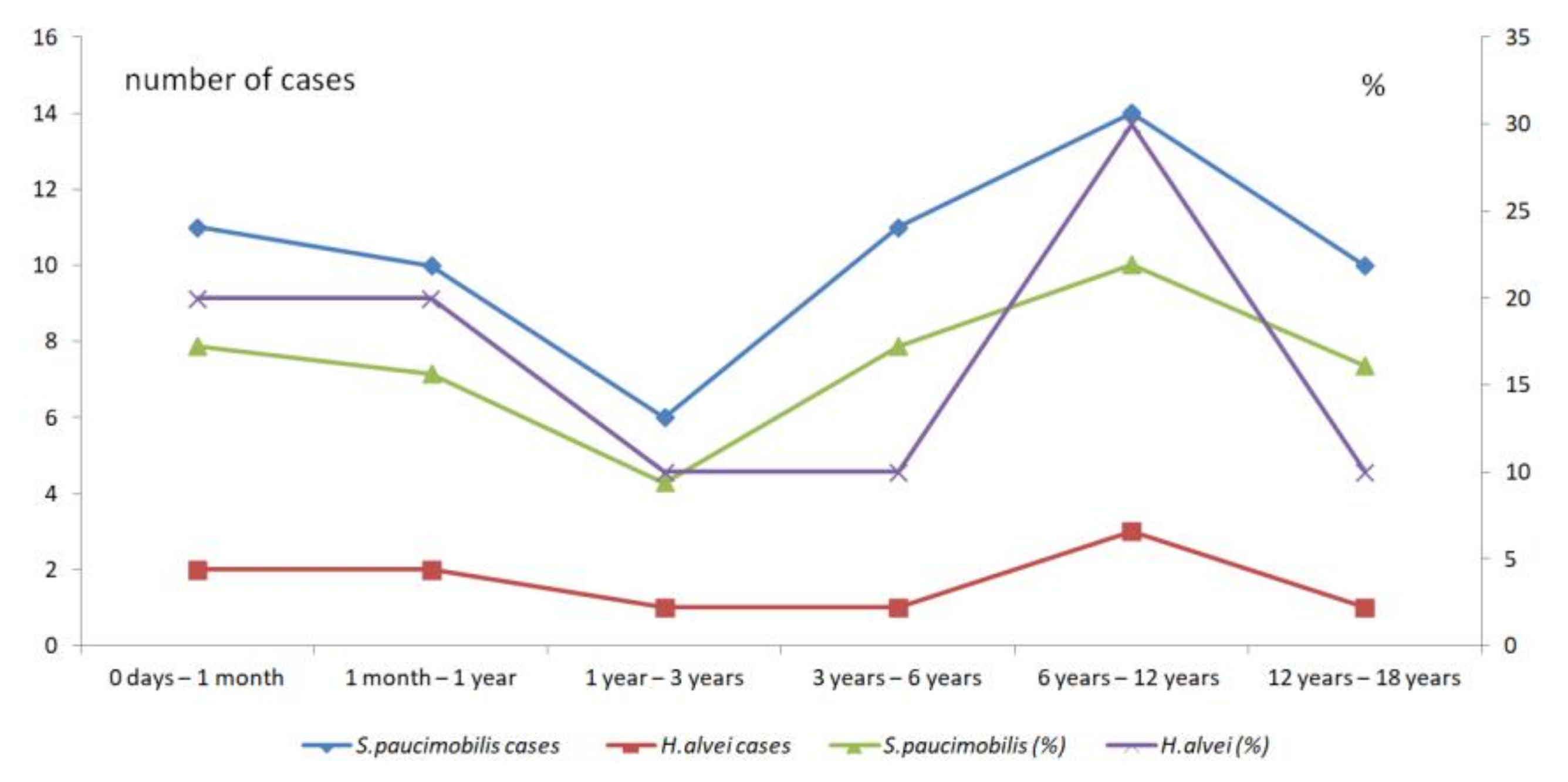
3.2.7. The Pediatric Infections
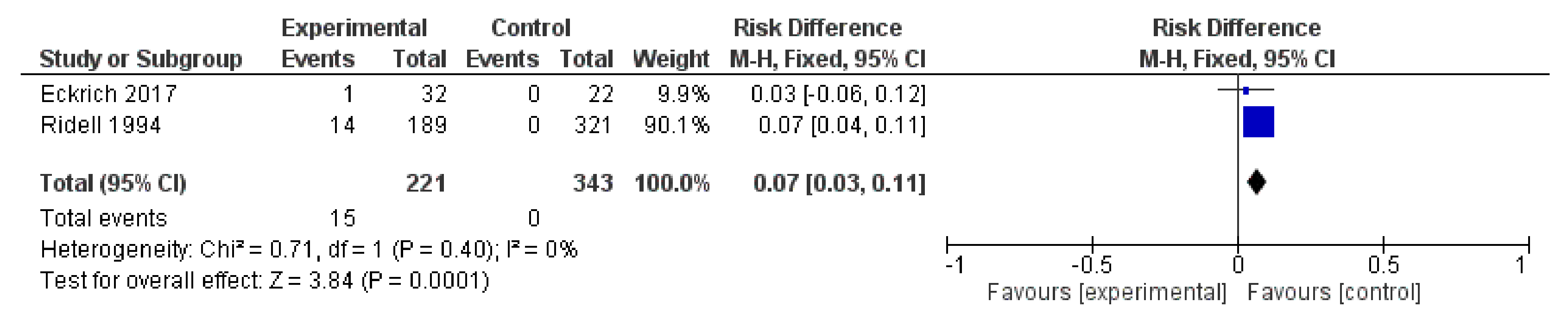
3.2.8. The Studies with Healthy-Control Groups
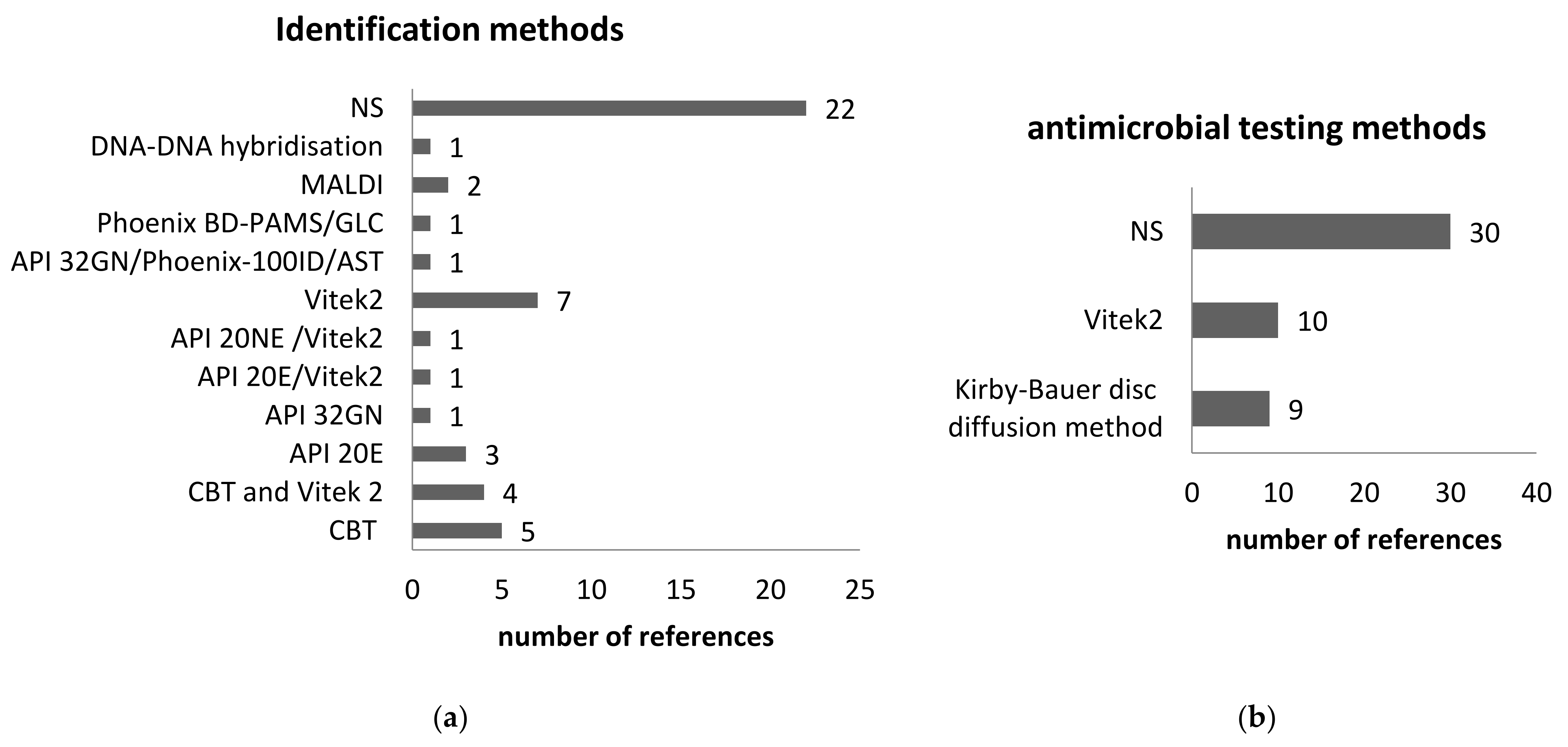
3.2.9. The Microbiological Diagnostic Methods
3.3. The Innate or Natural Antibiotic Resistance
3.4. The Limitations of the Study
3.5. The Strength of the Study
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Centers for Disease Control and Prevention; Infectious Disease Society of America; American Society of Blood and Marrow Transplantation. Guidelines for preventing opportunistic infections among hematopoietic stem cell transplant recipients MMWR. Recomm. Rep. Morb. Mortal. Wkly. Rep. Recomm. Rep. 2000, 49, 1–125, CE1-7. [Google Scholar]
- Kumar, R.; Ison, M.G. Opportunistic Infections in Transplant Patients. Infect. Dis. Clin. N. Am. 2019, 33, 1143–1157. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Gao, J.; Tang, W. Nosocomial infection and its molecular mechanisms of antibiotic resistance. Biosci. Trends 2016, 10, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Rizi, K.S.; Hasanzade, S.; Soleimanpour, S.; Youssefi, M.; Jamehdar, S.A.; Ghazvini, K.; Safdari, H.; Farsiani, H. Phenotypic and molecular characterization of antimicrobial resistance in clinical species of Enterobacter, Serratia, and Hafnia in Northeast Iran. Gene Rep. 2021, 25, 101352. [Google Scholar] [CrossRef]
- Ta, C.; Wong, G.; Cole, W.; Medvedev, G. Scrub sink contamination and transmission to operating room personnel. New Microbes New Infect. 2020, 37, 100754. [Google Scholar] [CrossRef]
- Quinn, G.A.; Banat, A.M.; Abdelhameed, A.M.; Banat, I.M. Streptomyces from traditional medicine: Sources of new innovations in antibiotic discovery. J. Med. Microbiol. 2020, 69, 1040–1048. [Google Scholar] [CrossRef]
- Awolope, O.K.; O’Driscoll, N.H.; Di Salvo, A.; Lamb, A.J. The complete genome sequence of Hafnia alvei A23BA; a potential antibiotic-producing rhizobacterium. BMC Res. Notes 2021, 14, 20–23. [Google Scholar] [CrossRef]
- Nishimura, K.; Ikarashi, M.; Yasuda, Y.; Sato, M.; Guerrero, M.C.; Galipon, J.; Arakawa, K. Complete Genome Sequence of Sphingomonas paucimobilis Strain Kira, Isolated from Human Neuroblastoma SH-SY5Y Cell Cultures Supplemented with Retinoic Acid. Microbiol. Resour. Announc. 2021, 10, e01156-20. [Google Scholar] [CrossRef]
- Bercea, S.; Năstase-Bucur, R.; Mirea, I.C.; Măntoiu, D.Ş.; Kenesz, M.; Petculescu, A.; Baricz, A.; Andrei, A.-Ş.; Banciu, H.L.; Papp, B.; et al. Novel approach to microbiological air monitoring in show caves. Aerobiologia 2018, 34, 445–468. [Google Scholar] [CrossRef]
- Jardine, J.L.; Abia, A.L.; Mavumengwana, V.; Ubomba-Jaswa, E. Phylogenetic Analysis and Antimicrobial Profiles of Cultured Emerging Opportunistic Pathogens (Phyla Actinobacteria and Proteobacteria) Identified in Hot Springs. Int. J. Environ. Res. Public Health 2017, 14, 1070. [Google Scholar] [CrossRef]
- Moldovan, O.T.; Bercea, S.; Năstase-Bucur, R.; Constantin, S.; Kenesz, M.; Mirea, I.C.; Petculescu, A.; Robu, M.; Arghir, R.A. Management of water bodies in show caves – A microbial approach. Tour. Manag. 2020, 78. [Google Scholar] [CrossRef]
- Bercea, S.; Năstase-Bucur, R.; Moldovan, O.T.; Kenesz, M.; Constantin, S. Yearly microbial cycle of human exposed surfaces in show caves. Subterr. Biol. 2019, 31, 1–14. [Google Scholar] [CrossRef]
- Leuko, S.; Koskinen, K.; Sanna, L.; D’Angeli, I.M.; De Waele, J.; Marcia, P.; Moissl-Eichinger, C.; Rettberg, P. The influence of human exploration on the microbial community structure and ammonia oxidizing potential of the Su Bentu limestone cave in Sardinia, Italy. PLoS ONE 2017, 12, e0180700. [Google Scholar]
- Man, B.; Wang, H.; Xiang, X.; Wang, R.; Yun, Y.; Gong, L. Phylogenetic diversity of culturable fungi in the Heshang Cave, central China. Front. Microbiol. 2015, 6, 1158. [Google Scholar] [CrossRef] [PubMed]
- Paduano, S.; Marchesi, I.; Casali, M.E.; Valeriani, F.; Frezza, G.; Vecchi, E.; Sircana, L.; Romano Spica, V.; Borella, P.; Bargellini, A. Characterisation of Microbial Community Associated with Different Disinfection Treatments in Hospital hot Water Networks. Int. J. Environ. Res. Public Health 2020, 17, 2158. [Google Scholar] [CrossRef]
- Oikonomou, O.; Sarrou, S.; Papagiannitsis, C.C.; Georgiadou, S.; Mantzarlis, K.; Zakynthinos, E.; Dalekos, G.N.; Petinaki, E. Rapid dissemination of colistin and carbapenem resistant Acinetobacter baumannii in Central Greece: Mechanisms of resistance, molecular identification and epidemiological data. BMC Infect. Dis. 2015, 15, 559. [Google Scholar] [CrossRef]
- Lesho, E.; Yoon, E.-J.; McGann, P.; Snesrud, E.; Kwak, Y.; Milillo, M.; Onmus-Leone, F.; Preston, L.; St Clair, K.; Nikolich, M.; et al. Emergence of colistin-resistance in extremely drug-resistant Acinetobacter baumannii containing a novel pmrCAB operon during colistin therapy of wound infections. J. Infect. Dis. 2013, 208, 1142–1151. [Google Scholar] [CrossRef]
- Adams, M.D.; Nickel, G.C.; Bajaksouzian, S.; Lavender, H.; Murthy, A.R.; Jacobs, M.R.; Bonomo, R.A. Resistance to colistin in Acinetobacter baumannii associated with mutations in the PmrAB two-component system. Antimicrob. Agents Chemother. 2009, 53, 3628–3634. [Google Scholar] [CrossRef]
- Caneiras, C.; Calisto, F.; Jorge da Silva, G.; Lito, L.; Melo-Cristino, J.; Duarte, A.; Caneiras, C.; Calisto, F.; Jorge da Silva, G.; Lito, L.; et al. First Description of Colistin and Tigecycline-Resistant Acinetobacter baumannii Producing KPC-3 Carbapenemase in Portugal. Antibiotics 2018, 7, 96. [Google Scholar] [CrossRef]
- Handschuh, H.; Ryan, M.P.; O’Dwyer, J.; Adley, C.C. Assessment of the Bacterial Diversity of Aircraft Water: Identification of the Frequent Fliers. PLoS One 2017, 12, e0170567. [Google Scholar] [CrossRef]
- Kulakov, L.A.; McAlister, M.B.; Ogden, K.L.; Larkin, M.J.; O’Hanlon, J.F. Analysis of bacteria contaminating ultrapure water in industrial systems. Appl. Environ. Microbiol. 2002, 68, 1548–1555. [Google Scholar] [CrossRef] [PubMed]
- Ryan, M.P.; Pembroke, J.T. Brevundimonas spp: Emerging global opportunistic pathogens. Virulence 2018, 9, 480–493. [Google Scholar] [CrossRef]
- Brenner, D.J. Characterization and clinical identification of Enterobacteriaceae by DNA hybridization. Prog. Clin. Pathol. 1978, 7, 71–117. [Google Scholar] [PubMed]
- Holmes, B.; Aucken, H.M. Citrobacter, Enterobacter, Klebsiella, Serratia and other members of the Enterobacteriaceae. In Topley & Wilson’s Microbiology an Microbial Infections, Systematic Bacteriology; Bawols, A., Duerden, B.I., Eds.; Arnold: London, UK, 1998; Volume 2, pp. 999–1020. ISBN 0340663170. [Google Scholar]
- Monnet, D.L.; Hansen, W.; Bollet, C.; Freney, J. Autres Enterobacteriaceae. In Manuel de Bactériologie Clinique; Elsevier: Paris, France, 1992; Volume 2, pp. 785–855. ISBN 2-906077-25-9. [Google Scholar]
- Klapholz, A.; Lessnau, K.D.; Huang, B.; Talavera, W.; Boyle, J.F. Hafnia alvei. Respiratory tract isolates in a community hospital over a three-year period and a literature review. Chest 1994, 105, 1098–1100. [Google Scholar] [CrossRef] [PubMed]
- Yabuuchi, E.; Yano, I.; Oyaizu, H.; Hashimoto, Y.; Ezaki, T.; Yamamoto, H. Proposals of Sphingomonas paucimobilis gen. nov. and comb. nov., Sphingomonas parapaucimobilis sp. nov., Sphingomonas yanoikuyae sp. nov., Sphingomonas adhaesiva sp. nov., Sphingomonas capsulata comb. nov., and two genospecies of the genus Sphingomonas. Microbiol. Immunol. 1990, 34, 99–119. [Google Scholar] [CrossRef]
- Pitt, T.L. Pseudomonas, Burkholderia and related genera. In Topley & Wilson’s Microbiology an Microbial Infections, Volume 2, Systematic Bacteriology; Bawols, A., Duerden, B.I., Eds.; Arnold: London, UK, 1999; pp. 1109–1138. ISBN 0340663170. [Google Scholar]
- Moffatt, J.H.; Harper, M.; Boyce, J.D. Mechanisms of Polymyxin Resistance. Adv. Exp. Med. Biol. 2019, 1145, 55–71. [Google Scholar]
- Olaitan, A.O.; Morand, S.; Rolain, J.-M. Mechanisms of polymyxin resistance: Acquired and intrinsic resistance in bacteria. Front. Microbiol. 2014, 5, 643. [Google Scholar] [CrossRef]
- Kawahara, K.; Kuraishi, H.; Zähringer, U. Chemical structure and function of glycosphingolipids of Sphingomonas spp and their distribution among members of the alpha-4 subclass of Proteobacteria. J. Ind. Microbiol. Biotechnol. 1999, 23, 408–413. [Google Scholar] [CrossRef]
- Sanger, F.; Nicklen, S.; Coulson, A.R. DNA sequencing with chain-terminating inhibitors. Proc. Natl. Acad. Sci. USA 1977, 74, 5463–5467. [Google Scholar] [CrossRef]
- CLSI; Wayne, P. M58: Methods for the Identification of Cultured Microorganisms Using Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry, 1st Edition Spectrometry; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2017; ISBN 1-56238-817-7. [Google Scholar]
- Maragakis, L.L.; Chaiwarith, R.; Srinivasan, A.; Torriani, F.J.; Avdic, E.; Lee, A.; Ross, T.R.; Carroll, K.C.; Perl, T.M.; LL, M.; et al. Sphingomonas paucimobilis bloodstream infections associated with contaminated intravenous fentanyl. Emerg. Infect. Dis. 2009, 15, 12–18. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing, 31st ed.; M100-S20; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2021. [Google Scholar]
- European Committee on Antimicrobial Susceptibility Testing (EUCAST). Breakpoint tables for interpretation of MICs and zone diameters. Version 11.0. CMI 2021, 12, P501–P503. [Google Scholar]
- Kahlmeter, G.; Brown, D.F.J.; Goldstein, F.W.; MacGowan, A.P.; Mouton, J.W.; Odenholt, I.; Rodloff, A.; Soussy, C.J.; Steinbakk, M.; Soriano, F.; et al. European Committee on Antimicrobial Susceptibility Testing (EUCAST) Technical Notes on antimicrobial susceptibility testing. Clin. Microbiol. Infect. 2006, 12, 501–503. [Google Scholar] [CrossRef]
- Benevides, G.N.; Hein, N.; Lo, D.S.; Ferronato, A.E.; Ragazzi, S.L.B.; Yoshioka, C.R.M.; Hirose, M.; Cardoso, D.M.; Regina Dos Santos, S.; Gilio, A.E. Otomastoiditis caused by Sphingomonas paucimobilis: Case report and literature review. Autops. Case Reports 2014, 4, 13–20. [Google Scholar] [CrossRef][Green Version]
- Lin, J.-N.; Lai, C.-H.; Chen, Y.-H.; Lin, H.-L.; Huang, C.-K.; Chen, W.-F.; Wang, J.-L.; Chung, H.-C.; Liang, S.-H.; Lin, H.-H. Sphingomonas paucimobilis bacteremia in humans: 16 case reports and a literature review. J. Microbiol. Immunol. Infect. 2010, 43, 35–42. [Google Scholar] [CrossRef]
- Liu, C.H.; Lin, W.J.; Wang, C.C.; Lee, K.L.; Tsai, M.C. Young-infant sepsis combined with urinary tract infection due to Hafnia alvei. J. Formos. Med. Assoc. 2007, 106, S39–S43. [Google Scholar] [CrossRef][Green Version]
- Nandy, S.; Dudeja, M.; Das, A.K.; Tiwari, R. Community Acquired Bacteremia by Sphingomonas paucimobilis: Two Rare Case Reports. J. Clin. Diagn. Res. 2013, 7, 2947–2949. [Google Scholar] [CrossRef] [PubMed]
- Rognrud, K.; Diaz, A.M.; Hill, C.; Kershaw, M.A. Bacterial Endocarditis Caused by Sphingomonas paucimobilis: A Case Report and Literature Review. Case Rep. Infect. Dis. 2020, 2020, 1–3. [Google Scholar] [CrossRef]
- Stanic, M.; Meusburger, E.; Hartmann, G.; Lhotta, K. Hafnia alvei Urosepsis in a Kidney Transplant Patient. Case Rep. Transpl. 2015, 2015, 863131. [Google Scholar]
- Yuen, L.C.; Jackson, T. A Case of Intra-abdominal abscess due to Sphingomonas paucimobilis in a patient on Peritoneal dialysis: A case report and review of literature. Indian J. Nephrol. 2020, 30, 196–200. [Google Scholar] [CrossRef]
- Albert, M.J.; Khorshed, A.; Islam, M.; Montanaro, J.; Rahaman, H.A.S.M.; Haider, K.; Hossain, M.A.; Kibriya, A.K.M.G.; Tzipori, S. Hafnia alvei, a probable cause of diarrhea in humans. Infect. Immun. 1991, 59, 1507–1513. [Google Scholar] [CrossRef]
- El Beaino, M.; Fares, J.; Malek, A.; Hachem, R. Sphingomonas paucimobilis-related bone and soft-tissue infections: A systematic review. Int. J. Infect. Dis. 2018, 77, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Göker, T.; Aşik, R.Z.; Yilmaz, M.B.; Çelik, İ.; Tekiner, A. Sphingomonas Paucimobilis: A Rare Infectious Agent Found in Cerebrospinal Fluid. J. Korean Neurosurg. Soc. 2017, 60, 481–483. [Google Scholar] [CrossRef] [PubMed]
- Lugito, H.; Pratama, N.; Kurniawan, A. A Lethal Case of Sphingomonas paucimobilis Bacteremia in an Immunocompromised Patient. Case Rep. Infect. Dis. 2016, 2016, 3294639. [Google Scholar]
- Hassan, E.A.; Elsherbiny, N.M.; Abd El-Rehim, A.S.; Soliman, A.M.A.; Ahmed, A.O. Health care-associated infections in pre-transplant liver intensive care unit: Perspectives and challenges. J. Infect. Public Health 2018, 11, 398–404. [Google Scholar] [CrossRef] [PubMed]
- Kelkar, A.S.; Kelkar, J.A.; Barve, P.M.; Mulay, A.; Sharma, S.; Amoaku, W. Post-clear corneal phacoemulsification endophthalmitis: Profile and management outcomes at a tertiary eye care center in western India. J. Ophthalmic. Inflamm. Infect. 2016, 6, 48. [Google Scholar] [CrossRef] [PubMed]
- Laupland, K.B.; Church, D.L.; Ross, T.; Pitout, J.D.D. Population-based laboratory surveillance of Hafnia alvei isolates in a large Canadian health region. Ann. Clin. Microbiol. Antimicrob. 2006, 5, 12. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.C.-W.; Siao-Ping Ong, N.D. Food-borne bacteremic illnesses in febrile neutropenic children. Hematol. Rep. 2011, 3, e11. [Google Scholar] [CrossRef]
- Lee, J.U.; Kim, J.K.; Yun, S.H.; Park, M.S.; Lee, N.E.; Sun, I.O.; Lee, K.Y. A case of peritoneal dialysis-associated peritonitis caused by Sphingomonas paucimobilis. Kidney Res. Clin. Pract. 2013, 32, 78–80. [Google Scholar] [CrossRef][Green Version]
- Liliav, B.; Yakoub, D.; Kasabian, A. Necrotizing fasciitis following endoscopic harvesting of the greater saphenous vein for coronary artery bypass graft. JSLS 2011, 15, 90–95. [Google Scholar] [CrossRef][Green Version]
- Mancini, M.; Panasiti, V.; Devirgiliis, V.; Pietropaolo, V.; Fioriti, D.; Nicosia, R.; Curzio, M.; Roberti, V.; Gobbi, S.; Bottoni, U.; et al. Bromhidrosis induced by sphingomonas paucimobilis: A case report. Int. J. Immunopathol. Pharmacol. 2009, 22, 845–848. [Google Scholar] [CrossRef]
- Bayram, N.; Devrim, I.; Apa, H.; Gülfidan, G.; Türkyilmaz, H.N.; Günay, I.; HN, T.; Günay, I. Sphingomonas paucimobilis infections in children: 24 case reports. Mediterr J Hematol Infect Dis 2013, 5, e2013040. [Google Scholar] [CrossRef] [PubMed]
- Mehmood, H.; Khan, N.; Ullah, S.; Ullah, A.; Marwat, A. A Rare Case of Sphingomonas paucimobilis Meningitis in the Absence of Cerebrospinal Fluid Pleocytosis. J. Investig. Med. High Impact Case Rep. 2018, 6, 2324709618756424. [Google Scholar]
- Mohan, D.; Railey, M. Sphingomonas paucimobilis peritonitis: A case report and review of the literature. Saudi J. Kidney Dis. Transpl. 2015, 26, 567–571. [Google Scholar] [CrossRef] [PubMed]
- Özdemir, M.; Pekcan, S.; Demircili, M.E.; Taşbent, F.E.; Feyzioğlu, B.; Pirinç, Ş.; Baykan, M. A rare cause of bacteremia in a pediatric patient with Down syndrome: Sphingomonas paucimobilis. Int. J. Med. Sci. 2011, 8, 537–539. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pascale, R.; Russo, E.; Esposito, I.; Leone, S.; Esposito, S. Sphingomonas paucimobilis osteomyelitis in an immunocompetent patient. A rare case report and literature review. New Microbiol. 2013, 36, 423–426. [Google Scholar]
- Rahman, S.R.; Ahmed, M.F.; Begum, A. Occurrence of urinary tract infection in adolescent and adult women of shanty town in Dhaka City, Bangladesh. Ethiop. J. Health Sci. 2014, 24, 145–152. [Google Scholar] [CrossRef]
- Refaat, M.; Zakka, P.; Khoury, M.; Chami, H.; Mansour, S.; Harbieh, B.; Abi-Saleh, B.; Bizri, A. Cardiac implantable electronic device infections: Observational data from a tertiary care center in Lebanon. Medicine 2019, 98, e14906. [Google Scholar] [CrossRef]
- Ridell, J.; Siitonen, A.; Paulin, L.; Mattila, L.; Korkeala, H.; Albert, M.J. Hafnia alvei in stool specimens from patients with diarrhea and healthy controls. J. Clin. Microbiol. 1994, 32, 2335–2337. [Google Scholar] [CrossRef]
- Roca, M.; García, A.; Peñas-Pardo, L.; Bosch-Aparicio, N.; Agustí, J. Sphingomonas paucimobilis keratitis in a patient with neurotrophic keratopathy and severe neurosensory hypoacusis: Treatment with penetrating keratoplasty and amniotic membrane grafting. Oman J. Ophthalmol. 2018, 11, 291–293. [Google Scholar] [CrossRef]
- Saboor, F.; Amin, F.; Nadeem, S. Community acquired sphingomonas paucimobilis in a child-a rare case. J. Pak. Med. Assoc. 2018, 68, 1714–1715. [Google Scholar]
- Seo, S.W.; Chung, I.Y.; Kim, E.; Park, J.M. A case of postoperative Sphingomonas paucimobilis endophthalmitis after cataract extraction. Korean J. Ophthalmol. 2008, 22, 63–65. [Google Scholar] [CrossRef] [PubMed]
- Chawla, K.; Vishwanath, S.; Munim, F.C. Nonfermenting Gram-negative Bacilli other than Pseudomonas aeruginosa and Acinetobacter Spp. Causing Respiratory Tract Infections in a Tertiary Care Center. J. Glob. Infect. Dis. 2013, 5, 144–148. [Google Scholar]
- Sirkhazi, M.; Sarriff, A.; Aziz, N.A.; Almana, F.; Arafat, O.; Shorman, M. Bacterial Spectrum, Isolation Sites and Susceptibility Patterns of Pathogens in Adult Febrile Neutropenic Cancer Patients at a Specialist Hospital in Saudi Arabia. World J. Oncol. 2014, 5, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Souto, A.; Guinda, M.; Mera, A.; Pardo, F. Septic arthritis caused by Sphingomonas paucimobilis in an immunocompetent patient. Reumatol. Clin. 2012, 8, 378–379. [Google Scholar] [CrossRef]
- Tai, M.L.S.; Velayuthan, R.D. Sphingomonas paucimobilis: An unusual cause of meningitis-case report. Neurol. Med. Chir. 2014, 54, 337–340. [Google Scholar] [CrossRef] [PubMed]
- Toh, H.S.; Tay, H.T.; Kuar, W.K.; Weng, T.C.; Tang, H.J.; Tan, C.K. Risk factors associated with Sphingomonas paucimobilis infection. J. Microbiol. Immunol. Infect. 2011, 44, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Vieira Colombo, A.P.; Magalhães, C.B.; Hartenbach, F.A.R.R.; Martins do Souto, R.; Maciel da Silva-Boghossian, C. Periodontal-disease-associated biofilm: A reservoir for pathogens of medical importance. Microb. Pathog. 2015, 94, 27–34. [Google Scholar] [CrossRef]
- Walayat, S.; Malik, A.; Hussain, N.; Lynch, T. Sphingomonas paucimobilis presenting as acute phlebitis: A case report. IDCases 2018, 11, 6–8. [Google Scholar] [CrossRef]
- Wiström, J.; Myrnäs, T.; Lundgren, C.; Monsen, T.; Wistrom, J.; Myrnas, T.; Lundgren, C.; Monsen, T.; Wiström, J.; Myrnäs, T.; et al. A case of acute cholecystitis due to Aeromonas sobria and Hafnia alvei from northern Europe. Clin. Microbiol. Infect. 1998, 4, 607–609. [Google Scholar] [CrossRef][Green Version]
- Yarlagadda, K.; Shrimanker, I.; Nookala, V.K. Catheter-associated Hafnia alvei-induced Urosepsis. Cureus 2019, 11, e6471. [Google Scholar] [CrossRef]
- Yozgat, Y.; Kilic, A.; Karadeniz, C.; Ozdemir, R.; Doksoz, O.; Gulfidan, G.; Mese, T. Sphingomonas paucimobilis bacteraemia and shock in a patient with rheumatic carditis. Indian J. Med. Microbiol. 2014, 32, 451–454. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Treadwell, T. Sphingomonas paucimobilis empyema caused by remote foreign body aspiration. BMJ Case Rep. 2018, 2018, 2017–2019. [Google Scholar]
- Chowdhary, P.; Ranjan, R.; Pandey, A.; Kumar, R. Sphingomonas paucimobilis septicemia in a neonate: A rare case report. Indian J. Pathol. Microbiol. 2016, 59, 119–121. [Google Scholar]
- Zhang, Y.; Liu, Z.R.; Chen, H.; Dong, W.J.; Fan, Y.C.; Yu, H.; Wang, G.J.; Li, Y.C.; Cao, K. Comparative study of bacterial status from conjunctival sac of the elder Qiang minority and Han people with dry eye in Sichuan, China. Int J Ophthalmol 2012, 5, 343–347. [Google Scholar]
- Cutuli, S.L.; De Maio, F.; De Pascale, G.; Grieco, D.L.; Monzo, F.R.; Carelli, S.; Tanzarella, E.S.; Pintaudi, G.; Piervincenzi, E.; Cascarano, L.; et al. COVID-19 influences lung microbiota dynamics and favors the emergence of rare infectious diseases: A case report of Hafnia Alvei pneumonia. J. Crit. Care 2021, 64, 173–175. [Google Scholar] [CrossRef]
- Del Borgo, C.; Maneschi, F.; Belvisi, V.; Morelli, F.; Vetica, A.; Marocco, R.; Tieghi, T.; Lichtner, M.; Mastroianni, C.M. Postpartum fever in the presence of a fibroid: Sphingomonas paucimobilis sepsis associated with pyomyoma. BMC Infect. Dis. 2013, 13, 574. [Google Scholar] [CrossRef]
- Demir, T.; Dadali, M. Recurrent complicated urinary tract infection due to rare pathogen Sphingomonas paucimobilis: Contamination or real deal? Infez Med 2016, 24, 241–244. [Google Scholar] [PubMed]
- Droutsas, K.; Kalantzis, G.; Symeonidis, C.; Georgalas, I. Posttraumatic Sphingomonas paucimobilis Endophthalmitis. Case Rep. Ophthalmol. Med. 2015, 2015, 192864. [Google Scholar] [PubMed]
- Eckrich, J.; Zissler, U.M.; Serve, F.; Leutz, P.; Smaczny, C.; Schmitt-Grohé, S.; Fussbroich, D.; Schubert, R.; Zielen, S.; Eickmeier, O.; et al. Airway inflammation in mild cystic fibrosis. J. Cyst. Fibros. 2017, 16, 107–115. [Google Scholar] [CrossRef]
- Jayol, A.; Saly, M.; Nordmann, P.; Ménard, A.; Poirel, L.; Dubois, V. Hafnia, an enterobacterial genus naturally resistant to colistin revealed by three susceptibility testing methods. J. Antimicrob. Chemother. 2017, 72, 2507–2511. [Google Scholar] [CrossRef][Green Version]
- Yadav, K.; Sharma, M.; Agarwal, S.; Bhatia, N.; Yadav, N. Aortic pseudoaneurysm & endocarditis caused by Aerococcus viridans: A case report and literature review. Cardiovasc. Revasc. Med. 2018, 19, 201–203. [Google Scholar] [PubMed]
- Gutiérrez-Fernández, J.; Gámiz-Gámiz, A.; Navarro-Marí, J.M.; Santos-Pérez, J.L. Genitourinary tract infection in children due to Aerococcus other than Aerococcus viridans. Literature review and 3 case reports. Enfermedades Infecc. y Microbiol. Clin. 2021, 39, 156–158. (In English) [Google Scholar] [CrossRef]
- Oragui, J.I.; Mara, D.D. Investigation of the survival characteristics of Rhodococcus coprophilus and certain fecal indicator bacteria. Appl. Environ. Microbiol. 1983, 46, 356–360. [Google Scholar] [CrossRef] [PubMed]
- Glasset, B.; Sperry, M.; Dervyn, R.; Herbin, S.; Brisabois, A.; Ramarao, N. The cytotoxic potential of Bacillus cereus strains of various origins. Food Microbiol. 2021, 98, 103759. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Ai, F.; Ji, C.; Tu, P.; Gao, Y.; Wu, Y.; Yan, F.; Yu, T. A Rapid Screening Method of Candidate Probiotics for Inflammatory Bowel Diseases and the Anti-inflammatory Effect of the Selected Strain Bacillus smithii XY1. Front. Microbiol. 2021, 12, 760385. [Google Scholar] [CrossRef]
- Boonmak, C.; Takahasi, Y.; Morikawa, M. Draft Genome Sequence of Geobacillus thermoleovorans Strain B23. Genome Announc. 2013, 1. [Google Scholar] [CrossRef]
- Suzuki, H. Peculiarities and biotechnological potential of environmental adaptation by Geobacillus species. Appl. Microbiol. Biotechnol. 2018, 102, 10425–10437. [Google Scholar] [CrossRef]
- Dragomirescu, C.C.; Lixandru, B.E.; Coldea, I.L.; Corneli, O.N.; Pana, M.; Palade, A.M.; Cristea, V.C.; Suciu, I.; Suciu, G.; Manolescu, L.S.C.; et al. Antimicrobial Susceptibility Testing for Corynebacterium Species Isolated from Clinical Samples in Romania. Antibiotics 2020, 9, 31. [Google Scholar] [CrossRef]
- Kalt, F.; Schulthess, B.; Sidler, F.; Herren, S.; Fucentese, S.F.; Zingg, P.O.; Berli, M.; Zinkernagel, A.S.; Zbinden, R.; Achermann, Y. Corynebacterium Species Rarely Cause Orthopedic Infections. J. Clin. Microbiol. 2018, 56. [Google Scholar] [CrossRef]
- Dykhuizen, R.S.; Douglas, G.; Weir, J.; Gould, I.M. Corynebacterium afermentans subsp. lipophilum: Multiple abscess formation in brain and liver. Scand. J. Infect. Dis. 1995, 27, 637–639. [Google Scholar] [CrossRef]
- Kumari, P.; Tyagi, A.; Marks, P.; Kerr, K.G. Corynebacterium afermentans spp. afermentans sepsis in a neurosurgical patient. J. Infect. 1997, 35, 201–202. [Google Scholar] [CrossRef]
- Yu, Z.; Zhu, F.; Tao, X.; Zhang, L.; Wu, S.; Dong, C.; Dong, Y.; Chen, G.; Zhou, X.; Fang, Y.; et al. Vogesella perlucida-induced bacteremia in an advanced-age patient: First case report. BMC Infect. Dis. 2020, 20, 687. [Google Scholar] [CrossRef] [PubMed]
- Briegel, I.; Trautnitz, M.; Behr, J. Rare Cause of Lung Tumour - Pulmonary Infection with Hafnia alvei: A Case Report. Pneumologie 2022, 1718–2521. [Google Scholar]
- Ryan, M.P.; Adley, C.C. Sphingomonas paucimobilis: A persistent Gram-negative nosocomial infectious organism. J. Hosp. Infect. 2010, 75, 153–157. [Google Scholar] [CrossRef]
- Kathayat, D.; Antony, L.; Deblais, L.; Helmy, Y.A.; Scaria, J.; Rajashekara, G. Small molecule adjuvants potentiate Colistin activity and attenuate resistance development in Escherichia coli by affecting pmrAB system. Infect. Drug Resist. 2020, 13, 2205. [Google Scholar] [CrossRef] [PubMed]
- Matuschek, E.; Brolund, A.; Karlsson Lindsjö, O.; Giske, C.G.; Byfors, S.; Kahlmeter, G. Revisiting colistin susceptibility testing: Will adding calcium to Mueller–Hinton agar improve the detection of colistin resistance? Clin. Microbiol. Infect. 2021, 27, 1172.e1–1172.e5. [Google Scholar] [CrossRef] [PubMed]


| Sample Code | Cave | Bacterial Strain | Probability (%) * |
|---|---|---|---|
| PTDF1 | Topolniţa | Aerococcus viridans | 88 |
| PLDF1 | Apă din Valea Leșului | Bacillus cereus/thuringiensis/mycoides | 89 |
| PFDF5 | Ferice | Rhodococcus coprophilus/erythropolis/globerulus | 90 |
| PFDF3 | Ferice | Bacillus cereus/thuringiensis/mycoides | 85 |
| PCDF1 | Cloşani | Sphingomonas paucimobilis Geobacillus thermoleovorans | 94 |
| 92 | |||
| PTSDF2 | Tăuşoare | Hafnia alvei Corynebacterium afermentans | 86 |
| 90 | |||
| PMDF11 | Muierilor | Geobacillus toebii | 91 |
| PMDF11A | Muierilor | Bacillus smithii | 88 |
| PMDF11B | Muierilor | Bacillus cereus/thuringiensis/mycoides | 86 |
| PMOS1 | Muierilor | Sphingomonas paucimobilis Bacillus smithii | 93 |
| 97 | |||
| PMOS2 | Muierilor | Geobacillus toebi | 95 |
| Antibiotic | S. paucimobilis PCDF1 | S. paucimobilis PMOS1 | H. alvei PTSDF2 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| MIC 1 | CLSI 2 | EUCAST 8 | MIC | CLSI 2 | EUCAST 8 | MIC | CLSI | EUCAST 8 | |
| Ticarcillin | 32 | I | <=8 | S | S | <=8 | - | S 5 | |
| Ticarcillin/Clavulanic Acid | 16 | S | S | <=8 | - | S | <=8 | S | S |
| Piperacillin | 32 | I 6 | I | <=4 | S | S | <=4 | S | S |
| Piperacillin/Tazobactam | 16 | S | S | <=4 | S | S | 8 | S | S |
| Ceftazidime | >=64 | R | R | 2 | S | S | 32 | R 7 | R |
| Cefepime | >=64 | R | R | <=1 | S | S | <=1 | S | S |
| Aztreonam | >=64 | - | (-) | 16 | - | I | 8 | R | |
| Imipenem | 1 | S | S | 1 | S | 0.5 | S | S | |
| Meropenem | <=0.25 | S | S | 0.5 | S | 4 | R | I | |
| Amikacin | <=2 | S | S | <=2 | S | <=2 | S | S | |
| Gentamicin | <=1 | S | IE 3 | <=1 | IE 3 | <=1 | S | S | |
| Tobramycin | <=1 | S | S | <=1 | S | <=1 | S | S | |
| Ciprofloxacin | <=0.25 | S | I | 2 | R | <=0.25 | S | S | |
| Pefloxcin | - | - | - | - | - | - | - | - | - |
| Minocycline | <=1 | S | S | <=1 | S | 8 | I | - | |
| Colistin 4 | >=16 | R | R 9 | 8 | R | R 9 | >=16 | R | R 9 |
| Rifampicin | - | - | - | - | - | - | - | - | - |
| Trimethoprim/Sulfamethoxazole | <=20 | S | S | <=20 | S | <=20 | S | S | |
| Type of Infections (n = 447) | S. paucimobilis (n = 262) n (%) | H. alvei (n = 185) n (%) |
|---|---|---|
| BSI | 116 (44.27) | 20 (10.81) |
| UTI | 4 (1.52) | 121 (65.41) |
| respiratory tract infections | 17 (6.49) | 11 (5.94) |
| bone or soft-tissue infections | 18 (6.87) | 7 (3.78) |
| Intra-abdominal infections | 15 (5.72) | 25 (13.51) |
| Head and neck infections | 20 (7.63) | 0 |
| Ocular infections | 17 (6.49) | 0 |
| Cardiovascular infections | 3 (1.15) | 0 |
| Other types of infections | 52 (19.85) | 1 (0.54) |
| t(14) = 0.49, p = 0.32 | ||
| The Acquired Source (n = 136) | SP | HA |
|---|---|---|
| n (%) | n = 116 (%) | n = 20 (%) |
| Hospital 50 (36.76) | 50 (43.10) | - |
| Community 43 (31.62) | 43 (37.06) | - |
| Not specified * 43 (31.62) | 23 (19.82) | 20 (100) |
| The Underlying Conditions (n = 136) | SP | HA |
|---|---|---|
| n (%) | n = 116 (%) | n = 20 (%) |
| malignancy 45 (33.08) | 44 (37.93) | 1 (5) |
| diabetes mellitus 15 (11.02) | 15 (12.93) | - |
| bacteremia associated with contaminated iv fentanyl 6 (4.41) | 6 (5.17) | - |
| kidney transplant 6 (4.41) | - | 6 (30) |
| prematurity 5 (3.67) | 4 (3.44) | 1 ** (5) |
| surgery 3 (2.21) | 3 (2.59) | - |
| Streptococcus pyogenes infections 2 (1.47) | 2 (1.72) | - |
| end-stage renal disease 3 (2.21) | 3 (2.59) | |
| HIV 2 (1.47) | 1 (0.86) | 1 (5) |
| neonatal sepsis 2 (1.47) | 2 (1.72) | - |
| burn injury 2 (1.47) | 2 (1.72) | - |
| chronic obstructive pulmonary disease and steroid use 1 (0.74) | 1 (0.86) | - |
| chronic steroid use 1 (0.74) | 1 (0.86) | - |
| chylothorax 1 (0.74) | 1 (0.86) | - |
| Down syndrome 1 (0.74) | 1 (0.86) | - |
| duodenal atresia 1 (0.74) | 1 (0.86) | - |
| epilepsy 1 (0.74) | 1 (0.86) | - |
| imperforate anus 1 (0.74) | 1 (0.86) | - |
| liver cirrhosis and alcoholism 1 (0.74) | 1 (0.86) | - |
| liver transplantation 1 (0.74) | - | 1 (5) |
| perforated appendicitis 1 (0.74) | 1 (0.86) | - |
| pulmonary embolization and atrial fibrillation 1 (0.74) | 1 (0.86) | - |
| septic arthritis 1 (0.74) | 1 (0.86) | - |
| urethral stone 1 (0.74) | 1 (0.86) | - |
| unclear 9 (6.62) | 3 (2.59) | 6 (30) |
| No* 23 (16.91) | 19 (16.38) | 4 (20) |
| Age Range | S. paucimobilis (n = 62) n (%) | H. alvei (n = 10) n (%) |
|---|---|---|
| 0 day to 1 month | 11 (17.19) | 2 (20) |
| 1 month to 1 year | 10 (15.63) | 2 (20) |
| 1 to 3 years | 6 (9.38) | 1 (10) |
| 3 to 6 years | 11 (17.19) | 1 (10) |
| 6 to 12 years | 14 (21.88) | 3 (30) |
| 12 to 18 years | 10 (16.12) | 1 (10) |
| t(10) = 7.84, p < 0.00001 | ||
| Type of Infections (n = 447) | Pediatric (n = 72) | Adult (n = 375) | ||
|---|---|---|---|---|
| n (%) | SP n = 62 (%) | HA n = 10 (%) | SP n = 200 (%) | HA n = 175 (%) |
| BSI 136 (30.42) | 55 (88.71) | 8 (80) | 61 (30.5) | 12 (6.85) |
| primary bacteremia 69 (15.44) | 34 (54.83) | 4 (40) | 25 (12.5) | 6 (3.43) |
| CR-BSI 19 (4.25) | 7 (11.29) | 0 | 12 (6) | 0 |
| bacteremia associated with contaminated iv fentanyl 6 (1.34) | 0 | 0 | 6 (3) | 0 |
| urosepsis 10 (2.24) | 3 (4.83) | 1 (10) | 0 | 6 (3.43) |
| pneumonia 4 (0.89) | 1 (1.61) | 0 | 3 (1.5) | 0 |
| meningitis 2 (0.45) | 0 | 1 (10) | 1 (0.5) | 0 |
| bone or soft tissue infections 5 (1.12) | 0 | 0 | 5 (2.5) | 0 |
| intra-abdominal infections * 6 (1.34) | 1 (1.61) | 2 (20) | 3 (1.5) | 0 |
| unspecified 15 (3.35) | 9 (14.52) | 0 | 6 (3) | 0 |
| UTI 125 (27.96) | 0 | 0 | 4 (2) | 121 (69.14) |
| Respiratory tract infections 28 (6.26) | 1 (1.61) | 0 | 16 (8) | 11 (6.29) |
| ventilator-associated pneumonia 1(0.22) | 0 | 0 | 0 | 1 (0.57) |
| empyema 1(0.22) | 0 | 0 | 1 (0.5) | 0 |
| airway inflammation 1(0.22) | 1 (1.61) | 0 | 0 | 0 |
| unspecified 25 (5.6) | 0 | 0 | 15 (7.5) | 10 (5.71) |
| Bone or soft tissue infections 25 (5.6) | 2 (3.23) | 1 (10) | 16 (8) | 6 (3.43) |
| cellulitis 2 (0.45) | 0 | 0 | 2 (1) | 0 |
| otomastoiditis 1 (0.22) | 1 (1.61) | 0 | 0 | 0 |
| osteomyelitis and septic arthritis 1 (0.22) | 1 (1.61) | 0 | 0 | 0 |
| septic arthritis 1 (0.22) | 0 | 0 | 1 (0.5) | 0 |
| osteomyelitis 2 (0.45) | 0 | 0 | 2 (1) | 0 |
| deep infection, open fracture 1(0.22) | 0 | 1 | 0 | 0 |
| focal myositis 1(0.22) | 0 | 0 | 1 (0.5) | 0 |
| necrotizing fasciitis 1(0.22) | 0 | 0 | 0 | 1 (0.57) |
| unspecified soft tissue infections 15(3.36) | 0 | 0 | 10 (5) | 5 (2.86) |
| Intra-abdominal infections 40 (8.94) | 1 (1.61) | 1 (10) | 14 (7) | 24 (13.71) |
| peritoneal dialysis-associated peritonitis 14 (3.13) | 1 (1.61) | 0 | 13 (6.5) | 0 |
| diarrhea 19 (4.25) | 0 | 1 (10) | 0 | 18 (10.29) |
| cholecystitis 1(0.22) | 0 | 0 | 0 | 1 (0.57) |
| intra-abdominal abscess and peritoneal dialysis related peritonitis 1 (0.22) | 0 | 0 | 1 (0.5) | 0 |
| unspecified 5 (1.12) | 0 | 0 | 0 | 5 (2.86) |
| Head and neck infections 20 (4.47) | 3 (4.84) | 0 | 17 (8.5) | 0 |
| brain abscess 1 (0.22) | 1 (1.61) | 0 | 0 | 0 |
| cervical adenitis 1 (0.22) | 1 (1.61) | 0 | 0 | 0 |
| central nervous system infections 6 (1.34) | 1 (1.61) | 0 | 5 (2.5) | 0 |
| unspecified 12 (2.68) | 0 | 0 | 12 (6) | 0 |
| Ocular infections 17 (3.8) | 0 | 0 | 17 (8.5) | 0 |
| endophthalmitis 3 (0.67) | 0 | 0 | 3 (1.5) | 0 |
| neurotrophic keratitis 1 (0.22) | 0 | 0 | 1 (0.5) | 0 |
| ocular contaminations 13 (2.9) | 0 | 0 | 13 (6.5) | 0 |
| Cardiovascular infections 3 (0.67) | 0 | 0 | 3 (1.5) | 0 |
| cardiac implantable electronic device infection 1 (0.22) | 0 | 0 | 1 (0.5) | 0 |
| endocarditis 1 (0.22) | 0 | 0 | 1 (0.5) | 0 |
| acute phlebitis 1(0.22) | 0 | 0 | 1 (0.5) | 0 |
| Other types of infections 53 (11.86) | 0 | 0 | 52 (26) | 1 (0.57) |
| periodontal disease 51 (11.41) | 0 | 0 | 51 (25.5) | 0 |
| bromhidrosis 1 (0.22) | 0 | 0 | 1 (0.5) | 0 |
| unclear (body fluid) 1 (0.22) | 0 | 0 | 0 | 1 (0.57) |
| t-test, p-value | t(16) = 0.95, p = 0.18 | t(16) = 0.19, p = 0.42 | ||
| F-ratio, p-value | F = 1.61, p = 0.21 | |||
| Reported Underlying Conditions (n = 447) | Pediatric (n = 72) | Adult (n = 375) | ||
|---|---|---|---|---|
| n (%) | SP n = 62 (%) | HA n = 10 (%) | SP n = 200 (%) | HA n = 175 (%) |
| Malignancy 50 (11.19) | 22 (35.48) | 1 (10) | 27 (13.5) | 0 |
| Chronic heart disease 8 (1.78) | 0 | 0 | 8 (4) | 0 |
| Chronic renal disease 25 (5.59) | 2 (3.22) | 0 | 16 (8) | 7 (4) |
| Chronic liver disease 5 (1.12) | 0 | 1 (10) | 4 (2) | 0 |
| Chronic pulmonary disease 7 (1.57) | 0 | 0 | 7 (3.5) | 0 |
| Congenital malformation 2 (0.45) (duodenal atresia, imperforate anus) | 2 (3.22) | 0 | 0 | 0 |
| Genetic disorders 3 (0.67) (Down syndrome, cystic fibrosis) | 2 (3.22) | 0 | 1(0.5) | 0 |
| Diabetes mellitus 15 (3.36) | 0 | 0 | 15 (7.5) | 0 |
| Surgery 4 (0.89) (cardiovascular surgery, neurosurgery, cataract extraction) | 0 | 0 | 3 (1.5) | 1 (0.57) |
| Trauma 7 (1.57) (burn injury, penetrating globe injury, history of fracture, remote foreign body aspiration during the dental procedure) | 1 (1.61) | 1 (10) | 5 (2.5) | 0 |
| Prematurity * 6 (1.34) | 5 (8.06) | 1 (10) | 0 | 0 |
| Multiple conditions ** 8 (1.79) | 0 | 0 | 8 (4) | 0 |
| other infections 11 (2.46) (HIV, Streptococcus pyogenes, abdominal infections due to perforated appendicitis, septic arthritis, neonatal sepsis, significant dysbiosis associated COVID-19) | 4 (6.45) | 1 (10) | 5 (2.5) | 1 (0.57) |
| Other conditions 16 (3.58) (red eye, chylothorax, epilepsy, respiratory distress) | 1(1.61) | 0 | 15 (7.5) | 0 |
| Chronic steroid use or alcohol abuse *** 10 (2.24) | 0 | 0 | 10 (5) | 0 |
| No underlying conditions reported 80 (17.9) | 22 (35.48) | 5 (50) | 1(0.5) | 52 (29.71) |
| Unclear or not specify 190 (42.51) | 1(1.61) | 0 | 75 (37.5) | 114 (65.14) |
| t-test, p-value | t(32) = −0.503, p = 0.31 | t(32) = 0.37, p = 0.35 | ||
| F-ratio, p-value | F = 1.56, p = 0.21 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ionescu, M.I.; Neagoe, D.Ș.; Crăciun, A.M.; Moldovan, O.T. The Gram-Negative Bacilli Isolated from Caves—Sphingomonas paucimobilis and Hafnia alvei and a Review of Their Involvement in Human Infections. Int. J. Environ. Res. Public Health 2022, 19, 2324. https://doi.org/10.3390/ijerph19042324
Ionescu MI, Neagoe DȘ, Crăciun AM, Moldovan OT. The Gram-Negative Bacilli Isolated from Caves—Sphingomonas paucimobilis and Hafnia alvei and a Review of Their Involvement in Human Infections. International Journal of Environmental Research and Public Health. 2022; 19(4):2324. https://doi.org/10.3390/ijerph19042324
Chicago/Turabian StyleIonescu, Mihaela Ileana, Dan Ștefan Neagoe, Alexandra Marioara Crăciun, and Oana Teodora Moldovan. 2022. "The Gram-Negative Bacilli Isolated from Caves—Sphingomonas paucimobilis and Hafnia alvei and a Review of Their Involvement in Human Infections" International Journal of Environmental Research and Public Health 19, no. 4: 2324. https://doi.org/10.3390/ijerph19042324
APA StyleIonescu, M. I., Neagoe, D. Ș., Crăciun, A. M., & Moldovan, O. T. (2022). The Gram-Negative Bacilli Isolated from Caves—Sphingomonas paucimobilis and Hafnia alvei and a Review of Their Involvement in Human Infections. International Journal of Environmental Research and Public Health, 19(4), 2324. https://doi.org/10.3390/ijerph19042324







