Physiological and Psychological Effects of Volatile Organic Compounds from Dried Common Rush (Juncus effusus L. var. decipiens Buchen.) on Humans
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
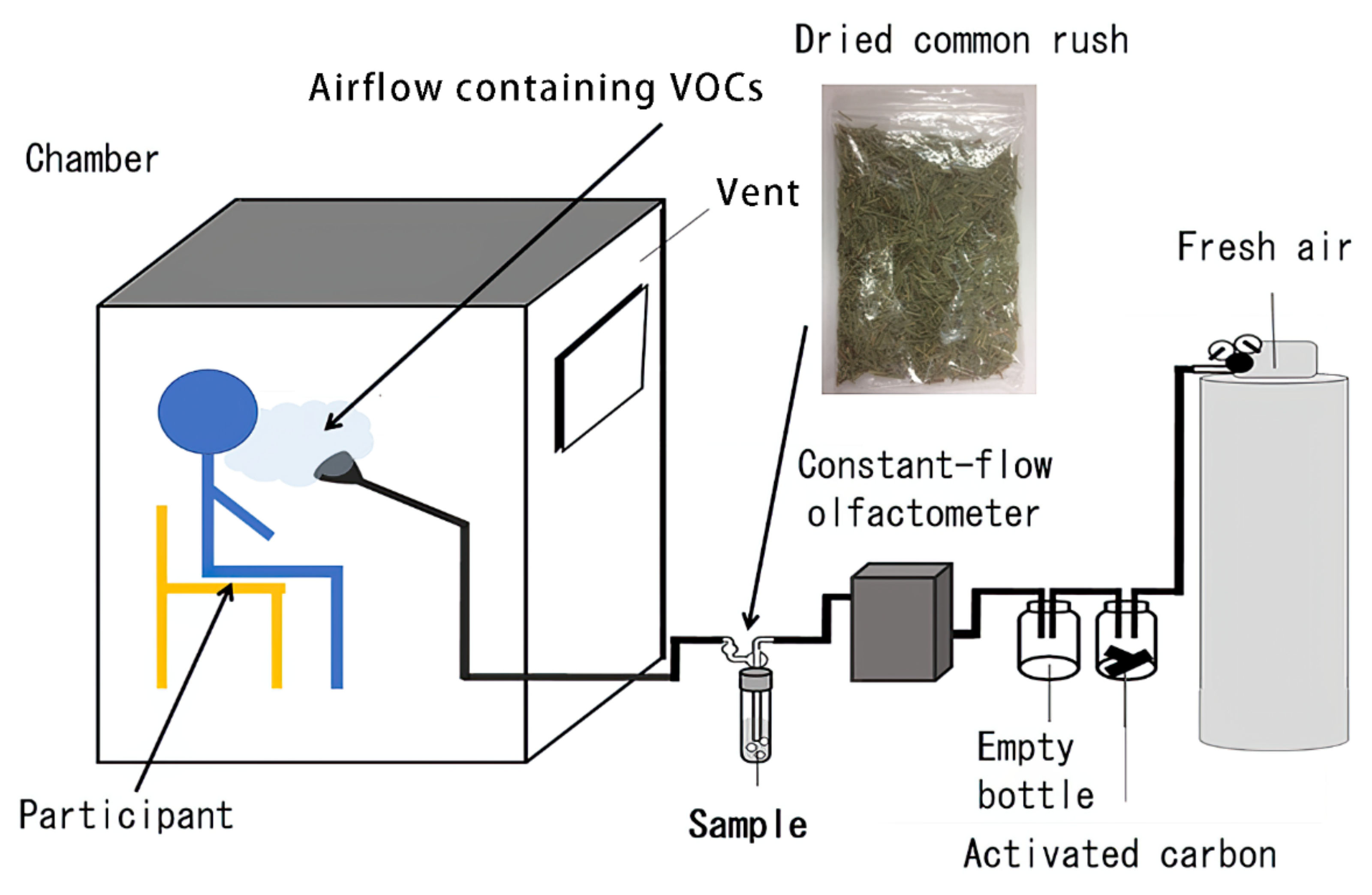
2.2. Experimental Room
2.3. Sample Determination
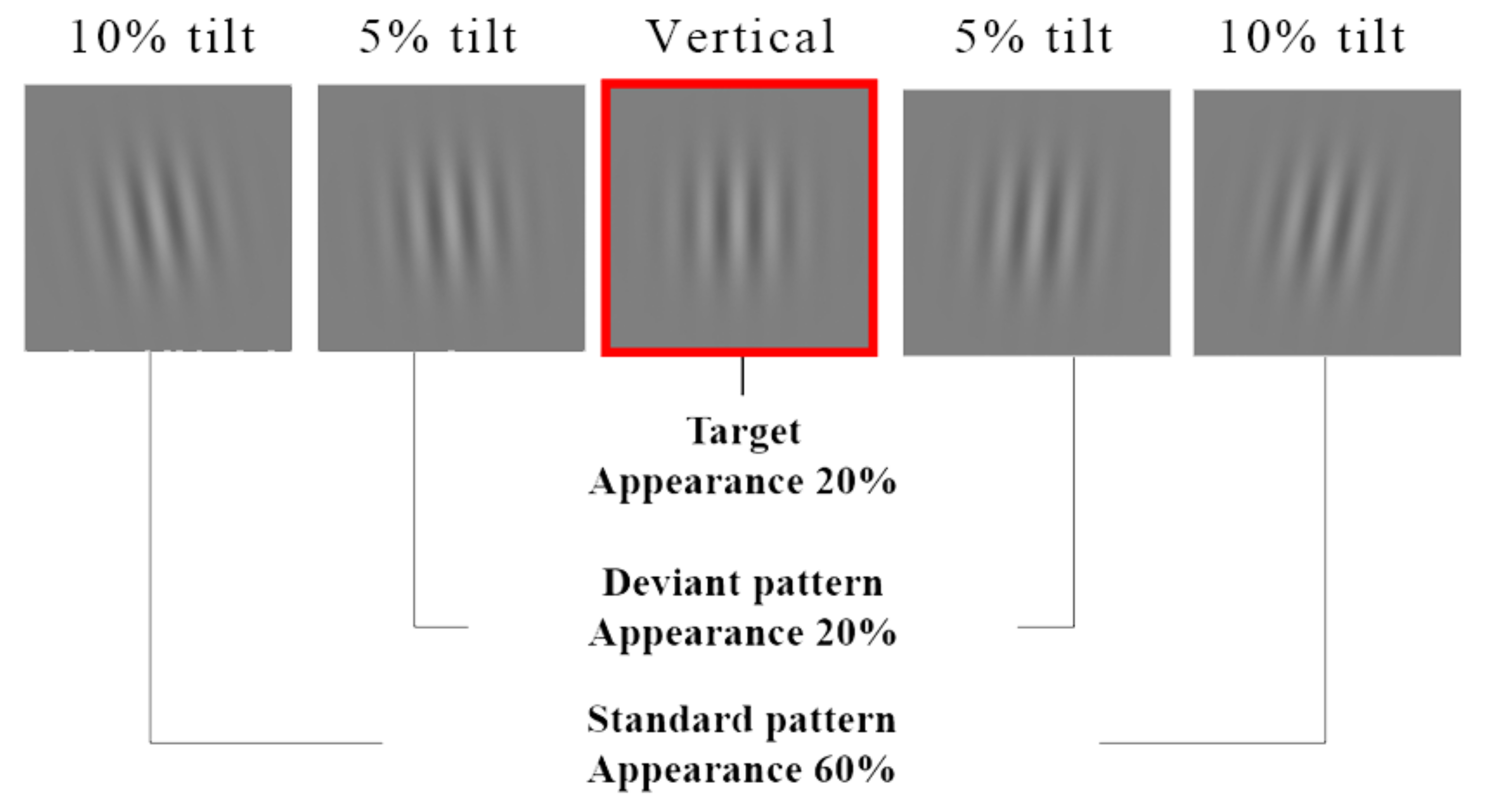
2.4. Visual Discrimination Task
2.5. Physiological Responses
2.6. Psychological Responses
2.7. Gas Chromatography-Mass Spectrometry Analysis
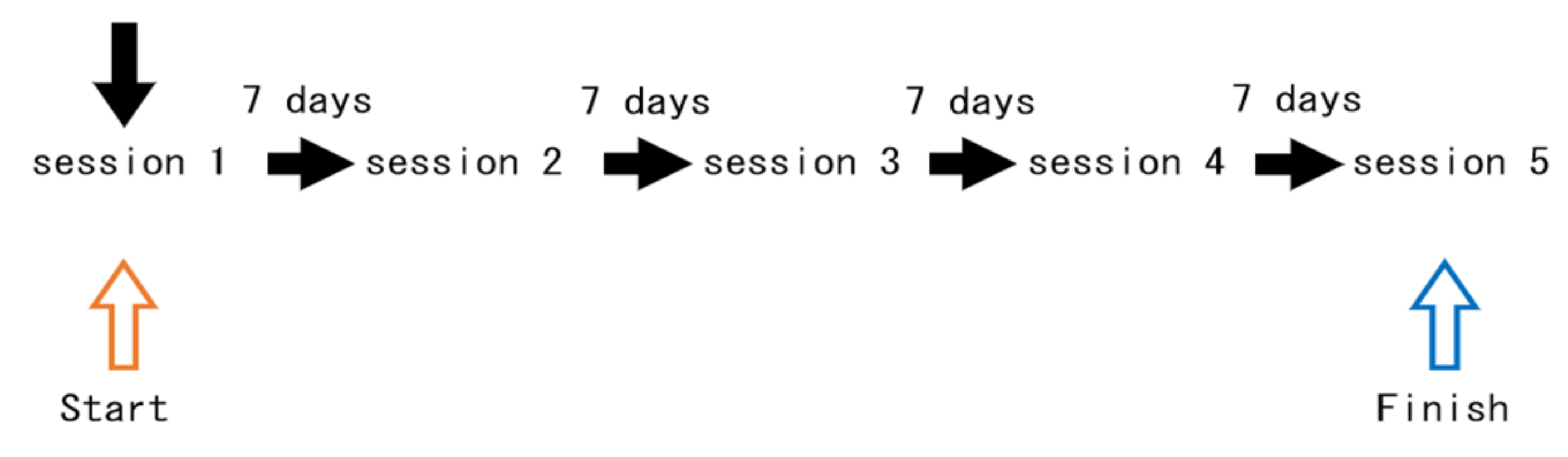
2.8. Experimental Setting
2.9. Data Analysis
3. Results
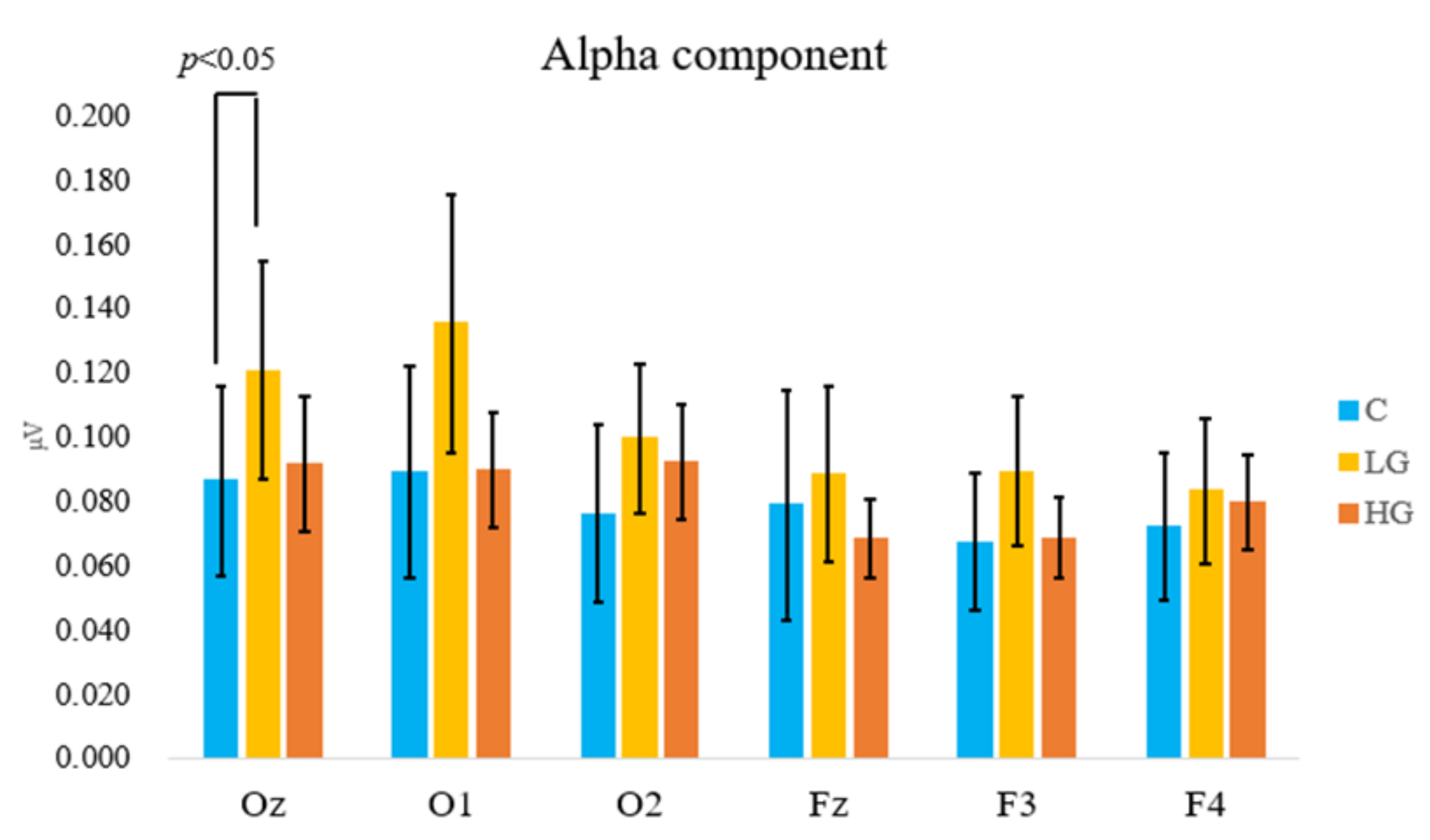
3.1. Physiological Responses
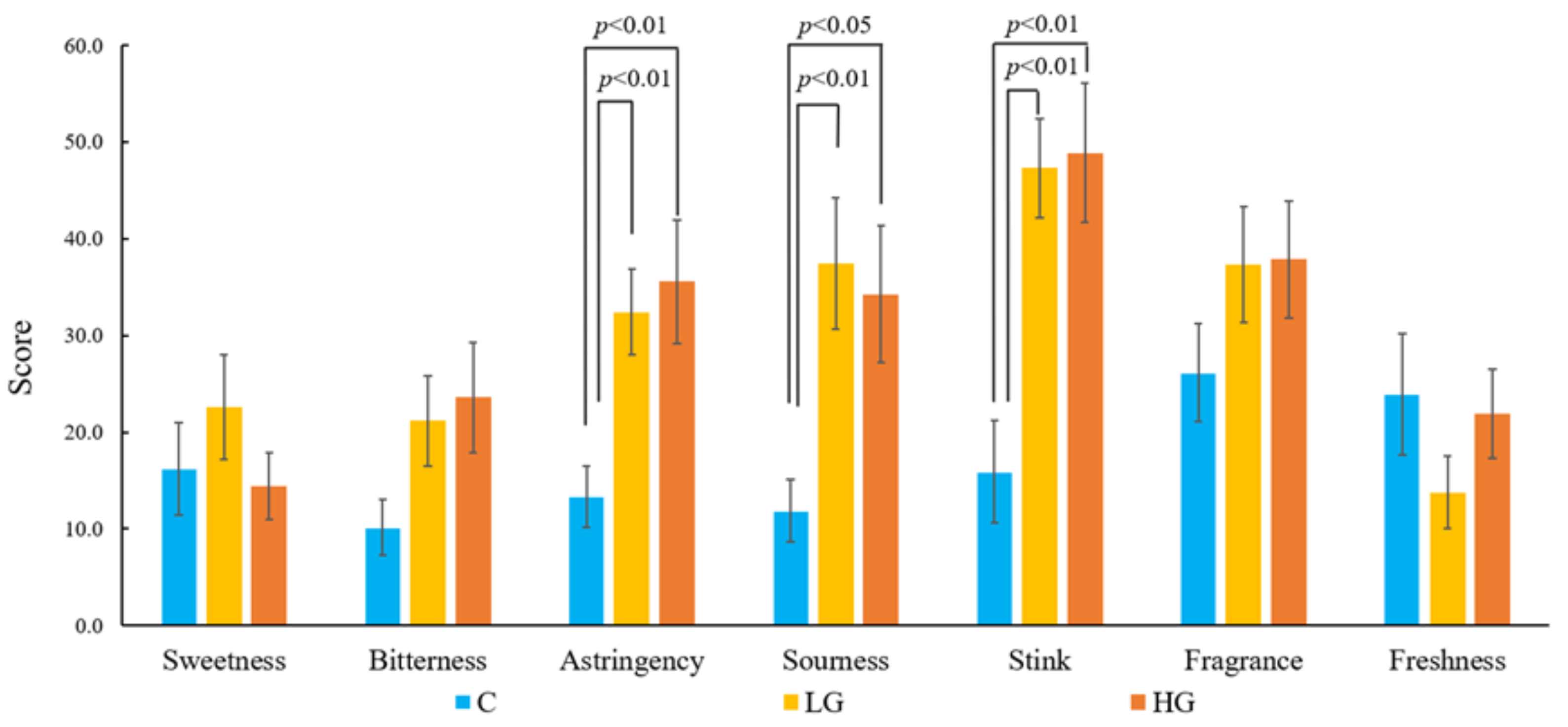
3.2. Psychological Responses
3.3. Visual Discrimination Task
3.4. Volatile Organic Compounds
4. Discussion
5. Conclusions
6. Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tsunetsugu, Y.; Miyazaki, Y.; Sato, H. Physiological effects in humans induces by visual stimulation of room interiors with different wood quantities. J. Wood Sci. 2007, 53, 11–16. [Google Scholar] [CrossRef]
- Sakuragawa, S.; Miyazaki, Y.; Kaneko, T.; Makita, T. Influence of wood wall panels on physiological and psychological responses. J. Wood Sci. 2005, 51, 136–140. [Google Scholar] [CrossRef]
- Kleber, M.; Wagner, A. Investigation of indoor thermal confort in warm-humid conditions at a German climate test facility. Build. Environ. 2018, 128, 216–224. [Google Scholar] [CrossRef]
- Elsadek, M.; Sun, M.K.; Fujii, E. Psycho-physiological responses to plant variegation as measured through eye movement, self-reported emotion and cerebral activity. Indoor Built Environ. 2017, 26, 758–770. [Google Scholar] [CrossRef]
- Sun, M.; Herrup, K.; Shi, B.; Hamano, Y.; Liu, C.; Goto, S. Changes in visual interaction: Viewing a Japanese garden directly, through glass or as a projected image. J. Environ. Psychol. 2018, 60, 116–121. [Google Scholar] [CrossRef]
- Nishimura, M.; Ikaga, S.; Hirata, J.; Ogawa, S.; Tsutiya, R. Influence of woody sleep scape to sleep and Intellectual productivity. Kanto Chapter Archit. Inst. Jpn. 2016, 86, 225–228. [Google Scholar]
- Kimura, A.; Sugiyama, H.; Sasaki, S.; Yatagai, M. Psychological and physiological effects in humans induced by the visual and olfactory stimulations of interior environment made of Hiba (Thujopsis dolabrata) wood. J. Jpn. Wood Res. Soc. 2011, 57, 150–159. [Google Scholar] [CrossRef]
- Okamura, R.; Ikaga, S.; Kimura, M.; Sugimoto, A.; Muragami, Y.; Shinotsuka, T. Quantification of influence of woody interior to study and sleep efficiency. Kanto Chapter Archit. Inst. Jpn. 2012, 82, 137–140. [Google Scholar]
- Tsunetsugu, Y.; Takeshi, M.; Yoshifumi, M. Relaxation Effect of Smell of Wood. Wood Ind. 2005, 60, 598–602. [Google Scholar]
- Matsubara, E.; Kawai, S. VOCs emitted from Japanese cedar (Cryptomeria japonica) interior walls induce physiological relaxation. Build. Environ. J. 2014, 72, 125–130. [Google Scholar] [CrossRef]
- Kim, Y.K.; Hagino, I.; Shibayama, Y.; Watanuki, S. The effect of wooden essential oil’s odor on psychophysiological responses. Jpn. J. Physiol. Anthropol. 2000, 5, 20–21. [Google Scholar]
- Fukuda, H.; Nakamine, M. Change in Feelings by Perfume of Essential Oils: Sensory evaluation of odour preference for wood essential oils by children. Wood Ind. 2003, 58, 593–597. [Google Scholar]
- Sowndhararajan, K.; Kim, S. Influence of Fragrances on Human Psychophysiological Activity: With Special Reference to Human Electroencephalographic Response. Sci. Pharm. 2016, 84, 724. [Google Scholar] [CrossRef]
- Jo, H.; Rodiek, S.; Fujii, E.; Miyazaki, Y.; Park, B.J.; Ann, S.W. Physiological and Psychological Response to Floral Scent. Hortscience 2013, 48, 82–88. [Google Scholar] [CrossRef]
- Yatagai, M. Volatiles from Wood and Their Effects. Wood Ind. 2005, 60, 594–597. [Google Scholar]
- Yatagai, M. Function of plant aroma components. Res. J. Food Agric. 2004, 27, 5–10. [Google Scholar]
- Matsubara, E.; Shimizu, K.; Fukagawa, M.; Ishizi, Y.; Kakoi, C.; Hatayama, T.; Nagano, J.; Okamoto, T.; Ohnuki, K.; Kondo, R. Volatiles emitted from the roots of Vetiveria zizanioides suppress the decline in attention during a visual display terminal task. Biomed. Res. 2012, 33, 299–308. [Google Scholar] [CrossRef][Green Version]
- Song, X.; Li, H.; Li, C.; Xu, J.; Hu, D. Effects of VOCs from leaves of Acer truncatum Bunge and Cedrus deodara on human physiology and psychology. Urban For. Urban Green. 2016, 19, 29–34. [Google Scholar] [CrossRef]
- Masaoka, S.; Hisatsune, T. The Present Conditions and the Direction of Tatami-spaces of Houses in Matsue-City. Bull. Fac. Educ. Shimane Univ. 2009, 43, 97–101. [Google Scholar]
- Sun, M.; Nakashima, T.; Yoshimura, Y.; Honden, A.; Nakagawa, T.; Saijo, H.; Watanabe, Y.; Ajimi, T.; Yasunari, S.; Yamada, Y.; et al. Effects and interaction of different interior material treatment and personal preference on psychological and physiological responses in living environment. J. Wood Sci. 2020, 66, 63. [Google Scholar] [CrossRef]
- Matsuhara, R.; Fukawa, M.; Okamoto, T. Multifunctional biological activities of natural products (6) Multifunctional biological activities of essential oil of Abies sibirica: Abies sibirica with conifer leaf-specific odor reduces arousal level after visual display terminal work. Aroma Res. 2011, 12, 180–186. [Google Scholar]
- Beaty, R.E.; Benedek, M.; Wilkins, R.W.; Jauk, E.; Fink, A.; Silvia, P.J.; Hodges, D.A.; Koschutnig, K.; Neubauer, A.C. Creativity and the default network: A functional connectivity analysis of the creative brain at rest. Neuropsychologia 2014, 64C, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Sowndhararajana, K.; Cho, H.; Yu, B.; Song, J.; Kim, S. Effect of inhalation of essential oil from Inula helenium L. root on electroencephalographic (EEG) activity of the human brain. Eur. J. Integr. Med. 2016, 8, 453–457. [Google Scholar] [CrossRef]
- Popov, T.; Popova, P.; Harkotte, M.; Awiszus, B.; Rockstroh, B.; Miller, G.A. Cross-frequency interactions between frontal theta and posterior alpha control mechanisms foster working memory. NeuroImage 2018, 181, 728–733. [Google Scholar] [CrossRef]
- Reiner, M.; Rozengurt, R.; Barne, A. Better than sleep: Theta neurofeedback training accelerates memory consolidation. Biol. Psychol. 2014, 95, 45–53. [Google Scholar] [CrossRef]
- Yurugi, Y.; Iwatsuki, H.; Suzuki, T. Effects of lavender inhalation on the excitability of spinal motor neurons. Jpn. J. Clin. Neurophysiol. 2015, 43, 111–120. [Google Scholar]
- Barbierim, R.; Matten, E.C.; Alabi, A.A.; Brown, E.N. A point-process model of human heartbeat intervals: New definitions of heart rate and heart rate variability. Am. J. Physiol. Heart Circ. Physiol. 2005, 288, 424–435. [Google Scholar] [CrossRef]
- Kanaya, N.; Hirata, N.; Kurosawa, S.; Nakayama, M.; Namiki, A. Differential effects of propofol and sevoflurane on heart rate variability. Anesthesiology 2003, 98, 34–40. [Google Scholar] [CrossRef]
- Cacioppo, J.T.; Berntson, G.G.; Binkley, P.F.; Quigley, K.S.; Uchino, B.N.; Fieldstone, A. Autonomic cardiac control. II. Noninvasive indices and basal response as revealed by autonomic blockades. Psychophysiology 1994, 31, 586–598. [Google Scholar] [CrossRef]
- Kobayashi, H.; Park, B.J.; Miyazaki, Y. Normative references of heart rate variability and salivary alpha-amylase in a healthy young males population. J. Physiol. Anthropol. 2012, 31, 9. [Google Scholar] [CrossRef]
- Kasuhito, Y. Instruction and Case Commentary of Short Version of POMS; Kaneko Shobo: Tokyo, Japan, 2005; pp. 1–9. [Google Scholar]
- Koomhin, P.; Sattayakhom, A.; Chandharakool, S.; Sinlapasorn, J.; Suanjan, S.; Palipoch, S.; Na-ek, P.; Punsawad, C.; Matan, N. Michelia Essential Oil Inhalation Increases Fast Alpha Wave Activity. Sci. Pharm. 2020, 88, 23. [Google Scholar] [CrossRef]
- Omaiye, E.E.; Whirter, K.J.; Luo, W.; Pankow, J.F.; Talbot, P. High-Nicotine Electronic Cigarette Products: Toxicity of JUUL Fluids and Aerosols Correlates Strongly with Nicotine and Some Flavor Chemical Concentrations. Chem. Res. Toxicol. 2019, 32, 1058–1069. [Google Scholar] [CrossRef] [PubMed]
- Masago, R.; Matsuda, T.; Kikuchi, Y.; Miyazaki, Y.; Iwanaga, K.; Harada, H.; Katsuura, T. Effects of Inhalation of Essential Oils on EEG Activity and Sensory Evaluation. J. Physiol. Anthropol. Appl. Hum. Sci. 2000, 19, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, R.S. Natural versus urban scenes some psychophysiological effects. Environ. Behav. 1981, 13, 523–556. [Google Scholar] [CrossRef]
- Lin, C.T.; Huang, K.C.; Chao, C.F.; Chen, J.A.; Chiu, T.W.; Ko, L.W.; Jung, T.P. Tonic and phasic EEG and behavioral changes induced by arousing feedback. NeuroImage 2010, 52, 633–642. [Google Scholar] [CrossRef]
- Berger, A.M.; Davelaar, E.J. Frontal Alpha Oscillations and Attentional Control: A Virtual Reality Neurofeedback Study. Neuroscience 2018, 378, 189–197. [Google Scholar] [CrossRef]
- Fink, A.; Graif, B.; Neubauer, A.C. Brain correlates underlying creative thinking: EEG alpha activity in professional vs. novice dancers. NeuroImage 2009, 46, 854–862. [Google Scholar] [CrossRef]
- Rominger, C.; Papousek, I.; Perchtold, C.; Weber, B.; Weiss, E.M.; Fink, A. The creative brain in the figural domain: Distinct patterns of EEG alpha power during idea generation and idea elaboration. Neuropsychologia 2018, 118, 13–19. [Google Scholar] [CrossRef]
- Cona, G.; Chiossi, F.; Tomasso, S.D.; Pellegrino, G.; Piccione, F.; Bisiacchi, P.; Arcarac, G. Theta and alpha oscillations as signatures of internal and external attention to delayed intentions: A magnetoencephalography (MEG) study. NeuroImage 2020, 205, 116295. [Google Scholar] [CrossRef]
- Kako, H.; Kobayashi, Y.; Yokogoshi, H. Effects of n-hexanal on dopamine release in the striatum of living rats. Eur. J. Pharmacol. 2011, 651, 77–82. [Google Scholar] [CrossRef]
- Fornal, C.; Wojcik, W.J.; Radulovacki, M. α-Flupenthixol increases slow-wave sleep in rats: Effect of dopamine receptor blockade. Neuropharmacology 1982, 21, 323–325. [Google Scholar] [CrossRef]


| Preparation (50 min) | Pre-Test (10 min) | Task (20 min) | Post-Test (15 min) | Remove Device (30 min) | |
|---|---|---|---|---|---|
| Device fixing | ● | ||||
| Condition check | ● | ||||
| Blood pressure and pulse | ● | ● | |||
| POMS | ● | ● | |||
| EEG and HRV during visual discrimination task | ● | ||||
| VAS and POMS | ● | ||||
| Remove device | ● |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, M.; Nakashima, T.; Yoshimura, Y.; Honden, A.; Nakagawa, T.; Nakashima, Y.; Kawaguchi, M.; Takamori, Y.; Koshi, Y.; Sawada, R.; et al. Physiological and Psychological Effects of Volatile Organic Compounds from Dried Common Rush (Juncus effusus L. var. decipiens Buchen.) on Humans. Int. J. Environ. Res. Public Health 2022, 19, 1856. https://doi.org/10.3390/ijerph19031856
Sun M, Nakashima T, Yoshimura Y, Honden A, Nakagawa T, Nakashima Y, Kawaguchi M, Takamori Y, Koshi Y, Sawada R, et al. Physiological and Psychological Effects of Volatile Organic Compounds from Dried Common Rush (Juncus effusus L. var. decipiens Buchen.) on Humans. International Journal of Environmental Research and Public Health. 2022; 19(3):1856. https://doi.org/10.3390/ijerph19031856
Chicago/Turabian StyleSun, Minkai, Taisuke Nakashima, Yuri Yoshimura, Akiyoshi Honden, Toshinori Nakagawa, Yu Nakashima, Makoto Kawaguchi, Yukimitsu Takamori, Yoshitaka Koshi, Rimpei Sawada, and et al. 2022. "Physiological and Psychological Effects of Volatile Organic Compounds from Dried Common Rush (Juncus effusus L. var. decipiens Buchen.) on Humans" International Journal of Environmental Research and Public Health 19, no. 3: 1856. https://doi.org/10.3390/ijerph19031856
APA StyleSun, M., Nakashima, T., Yoshimura, Y., Honden, A., Nakagawa, T., Nakashima, Y., Kawaguchi, M., Takamori, Y., Koshi, Y., Sawada, R., Nishida, S., Ohnuki, K., & Shimizu, K. (2022). Physiological and Psychological Effects of Volatile Organic Compounds from Dried Common Rush (Juncus effusus L. var. decipiens Buchen.) on Humans. International Journal of Environmental Research and Public Health, 19(3), 1856. https://doi.org/10.3390/ijerph19031856






