The Long-COVID Experience Changed People’s Vaccine Hesitancy but Not Their Vaccination Fear
Abstract
1. Introduction
1.1. How Long-COVID May Affect Vaccine Hesitancy
1.2. Hypotheses Development
2. Method
2.1. Participants
2.2. Procedure
2.3. Measures
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wu, F.; Zhao, S.; Yu, B.; Chen, Y.-M.; Wang, W.; Song, Z.-G.; Hu, Y.; Tao, Z.-W.; Tian, J.-H.; Pei, Y.-Y.; et al. A New Coronavirus Associated with Human Respiratory Disease in China. Nature 2020, 579, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Cucinotta, D.; Vanelli, M. WHO Declares COVID-19 a Pandemic. Acta Biomed. 2020, 91, 157–160. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization WHO. Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int (accessed on 3 October 2022).
- Böhning, D.; Rocchetti, I.; Maruotti, A.; Holling, H. Estimating the Undetected Infections in the Covid-19 Outbreak by Harnessing Capture–Recapture Methods. Int. J. Infect. Dis. 2020, 97, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Richards, R. Evidence on the Accuracy of the Number of Reported COVID-19 Infections and Deaths in Lower-Middle Income Countries; Institute of Development Studies: Brighton, UK, 2020. [Google Scholar]
- Mathieu, E.; Ritchie, H.; Ortiz-Ospina, E.; Roser, M.; Hasell, J.; Appel, C.; Giattino, C.; Rodés-Guirao, L. A Global Database of COVID-19 Vaccinations. Nat. Hum. Behav. 2021, 5, 947–953. [Google Scholar] [CrossRef]
- Duradoni, M.; Fiorenza, M.; Guazzini, A. When Italians Follow the Rules against COVID Infection: A Psychological Profile for Compliance. COVID 2021, 1, 246–262. [Google Scholar] [CrossRef]
- Koh, D. COVID-19 Lockdowns throughout the World. Occup. Med. 2020, 70, 322. [Google Scholar] [CrossRef]
- Aquino, E.M.L.; Silveira, I.H.; Pescarini, J.M.; Aquino, R.; de Souza-Filho, J.A.; dos Santos Rocha, A.; Ferreira, A.; Victor, A.; Teixeira, C.; Machado, D.B.; et al. Social Distancing Measures to Control the COVID-19 Pandemic: Potential Impacts and Challenges in Brazil. Ciênc. Saúde Coletiva 2020, 25, 2423–2446. [Google Scholar] [CrossRef]
- Liao, M.; Liu, H.; Wang, X.; Hu, X.; Huang, Y.; Liu, X.; Brenan, K.; Mecha, J.; Nirmalan, M.; Lu, J.R. A Technical Review of Face Mask Wearing in Preventing Respiratory COVID-19 Transmission. Curr. Opin. Colloid Interface Sci. 2021, 52, 101417. [Google Scholar] [CrossRef]
- Rundle, C.W.; Presley, C.L.; Militello, M.; Barber, C.; Powell, D.L.; Jacob, S.E.; Atwater, A.R.; Watsky, K.L.; Yu, J.; Dunnick, C.A. Hand Hygiene during COVID-19: Recommendations from the American Contact Dermatitis Society. J. Am. Acad. Dermatol. 2020, 83, 1730–1737. [Google Scholar] [CrossRef]
- Guazzini, A.; Fiorenza, M.; Panerai, G.; Duradoni, M. What Went Wrong? Predictors of Contact Tracing Adoption in Italy during COVID-19 Pandemic. Future Internet 2021, 13, 286. [Google Scholar] [CrossRef]
- Ahmed, N.; Michelin, R.A.; Xue, W.; Ruj, S.; Malaney, R.; Kanhere, S.S.; Seneviratne, A.; Hu, W.; Janicke, H.; Jha, S.K. A Survey of COVID-19 Contact Tracing Apps. IEEE Access 2020, 8, 134577–134601. [Google Scholar] [CrossRef]
- Ussai, S.; Pistis, M.; Missoni, E.; Formenti, B.; Armocida, B.; Pedrazzi, T.; Castelli, F.; Monasta, L.; Lauria, B.; Mariani, I. “Immuni” and the National Health System: Lessons Learnt from the COVID-19 Digital Contact Tracing in Italy. Int. J. Environ. Res. Public Health 2022, 19, 7529. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency—EMA. COVID-19 Vaccines: Authorised. Available online: https://www.ema.europa.eu/en/human-regulatory/overview/public-health-threats/coronavirus-disease-covid-19/treatments-vaccines/vaccines-covid-19/covid-19-vaccines-authorised (accessed on 3 October 2022).
- Self, W.H.; Tenforde, M.W.; Rhoads, J.P.; Gaglani, M.; Ginde, A.A.; Douin, D.J.; Olson, S.M.; Talbot, H.K.; Casey, J.D.; Mohr, N.M.; et al. Comparative Effectiveness of Moderna, Pfizer-BioNTech, and Janssen (Johnson & Johnson) Vaccines in Preventing COVID-19 Hospitalizations Among Adults Without Immunocompromising Conditions—United States, March–August 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 1337–1343. [Google Scholar] [CrossRef] [PubMed]
- Rudan, I.; Adeloye, D.; Sheikh, A. COVID-19: Vaccines, Efficacy and Effects on Variants. Curr. Opin. Pulm. Med. 2022, 28, 180–191. [Google Scholar] [CrossRef]
- Gotlib, J.; Sobierajski, T.; Jaworski, M.; Wawrzuta, D.; Borowiak, E.; Dobrowolska, B.; Dyk, D.; Gaworska-Krzemińska, A.; Grochans, E.; Kózka, M.; et al. “Vaccinate, Do Not Hesitate!”. Vaccination Readiness against COVID-19 among Polish Nursing Undergraduate Students: A National Cross-Sectional Survey. Vaccines 2021, 9, 1029. [Google Scholar] [CrossRef]
- Simione, L.; Vagni, M.; Gnagnarella, C.; Bersani, G.; Pajardi, D. Mistrust and Beliefs in Conspiracy Theories Differently Mediate the Effects of Psychological Factors on Propensity for COVID-19 Vaccine. Front. Psychol. 2021, 12, 683684. [Google Scholar] [CrossRef]
- Simione, L.; Vagni, M.; Maiorano, T.; Giostra, V.; Pajardi, D. How Implicit Attitudes toward Vaccination Affect Vaccine Hesitancy and Behaviour: Developing and Validating the V-IRAP. Int. J. Environ. Res. Public Health 2022, 19, 4205. [Google Scholar] [CrossRef]
- Troiano, G.; Nardi, A. Vaccine Hesitancy in the Era of COVID-19. Public Health 2021, 194, 245–251. [Google Scholar] [CrossRef]
- Qiao, S.; Tam, C.C.; Li, X. Risk Exposures, Risk Perceptions, Negative Attitudes Toward General Vaccination, and COVID-19 Vaccine Acceptance Among College Students in South Carolina. Am. J. Health Promot. 2022, 36, 175–179. [Google Scholar] [CrossRef]
- Bendau, A.; Plag, J.; Petzold, M.B.; Ströhle, A. COVID-19 Vaccine Hesitancy and Related Fears and Anxiety. Int. Immunopharmacol. 2021, 97, 107724. [Google Scholar] [CrossRef]
- WHO Strategic Advisory Group on Experts—SAGE. Report of the SAGE Working Group on Vaccine Hesitancy; SAGE: Thousand Oaks, CA, USA, 2014; pp. 1–64. [Google Scholar]
- World Health Organization. Ten Threats to Global Health in 2019. Available online: https://www.who.int/news-room/spotlight/ten-threats-to-global-health-in-2019 (accessed on 3 October 2022).
- Larson, H.J.; Jarrett, C.; Schulz, W.S.; Chaudhuri, M.; Zhou, Y.; Dube, E.; Schuster, M.; MacDonald, N.E.; Wilson, R. Measuring Vaccine Hesitancy: The Development of a Survey Tool. Vaccine 2015, 33, 4165–4175. [Google Scholar] [CrossRef] [PubMed]
- Dubé, E.; Gagnon, D.; Vivion, M. Optimizing Communication Material to Address Vaccine Hesitancy. Can. Commun. Dis. Rep. 2020, 46, 48–52. [Google Scholar] [CrossRef] [PubMed]
- Diaz, P.; Zizzo, J.; Balaji, N.C.; Reddy, R.; Khodamoradi, K.; Ory, J.; Ramasamy, R. Fear about Adverse Effect on Fertility Is a Major Cause of COVID-19 Vaccine Hesitancy in the United States. Andrologia 2022, 54, e14361. [Google Scholar] [CrossRef] [PubMed]
- Montagni, I.; Ouazzani-Touhami, K.; Mebarki, A.; Texier, N.; Schück, S.; Tzourio, C.; The CONFINS Group. Acceptance of a COVID-19 Vaccine Is Associated with Ability to Detect Fake News and Health Literacy. J. Public Health 2021, 43, 695–702. [Google Scholar] [CrossRef] [PubMed]
- Muric, G.; Wu, Y.; Ferrara, E. COVID-19 Vaccine Hesitancy on Social Media: Building a Public Twitter Data Set of Antivaccine Content, Vaccine Misinformation, and Conspiracies. JMIR Public Health Surveill. 2021, 7, e30642. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Haupert, S.R.; Zimmermann, L.; Shi, X.; Fritsche, L.G.; Mukherjee, B. Global Prevalence of Post-Coronavirus Disease 2019 (COVID-19) Condition or Long COVID: A Meta-Analysis and Systematic Review. J. Infect. Dis. 2022, 226, 1593–1607. [Google Scholar] [CrossRef]
- Yelin, D.; Margalit, I.; Yahav, D.; Runold, M.; Bruchfeld, J. Long COVID-19—It’s Not over Until? Clin. Microbiol. Infect. 2021, 27, 506–508. [Google Scholar] [CrossRef]
- Cabrera Martimbianco, A.L.; Pacheco, R.L.; Bagattini, Â.M.; Riera, R. Frequency, Signs and Symptoms, and Criteria Adopted for Long COVID-19: A Systematic Review. Int. J. Clin. Pract. 2021, 75, e14357. [Google Scholar] [CrossRef]
- Hastie, C.E.; Lowe, D.J.; McAuley, A.; Winter, A.J.; Mills, N.L.; Black, C.; Scott, J.T.; O’Donnell, C.A.; Blane, D.N.; Browne, S.; et al. Outcomes among Confirmed Cases and a Matched Comparison Group in the Long-COVID in Scotland Study. Nat. Commun. 2022, 13, 5663. [Google Scholar] [CrossRef]
- Ballering, A.V.; van Zon, S.K.R.; olde Hartman, T.C.; Rosmalen, J.G.M. Persistence of Somatic Symptoms after COVID-19 in the Netherlands: An Observational Cohort Study. Lancet 2022, 400, 452–461. [Google Scholar] [CrossRef]
- Zawilska, J.B.; Kuczyńska, K. Psychiatric and Neurological Complications of Long COVID. J. Psychiatr. Res. 2022, 156, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Global Burden of Disease Long COVID Collaborators; Wulf Hanson, S.; Abbafati, C.; Aerts, J.G.; Al-Aly, Z.; Ashbaugh, C.; Ballouz, T.; Blyuss, O.; Bobkova, P.; Bonsel, G.; et al. Estimated Global Proportions of Individuals With Persistent Fatigue, Cognitive, and Respiratory Symptom Clusters Following Symptomatic COVID-19 in 2020 and 2021. JAMA 2022, 328, 1604–1615. [Google Scholar] [CrossRef] [PubMed]
- Carfì, A.; Bernabei, R.; Landi, F.; For the Gemelli Against COVID-19 Post-Acute Care Study Group. Persistent Symptoms in Patients After Acute COVID-19. JAMA 2020, 324, 603–605. [Google Scholar] [CrossRef] [PubMed]
- del Rio, C.; Collins, L.F.; Malani, P. Long-Term Health Consequences of COVID-19. JAMA 2020, 324, 1723–1724. [Google Scholar] [CrossRef] [PubMed]
- Brüssow, H.; Timmis, K. COVID-19: Long Covid and Its Societal Consequences. Environ. Microbiol. 2021, 23, 4077–4091. [Google Scholar] [CrossRef]
- Fernández-de-las-Peñas, C.; Ryan-Murua, P.; Rodríguez-Jiménez, J.; Palacios-Ceña, M.; Arendt-Nielsen, L.; Torres-Macho, J. Serological Biomarkers at Hospital Admission Are Not Related to Long-Term Post-COVID Fatigue and Dyspnea in COVID-19 Survivors. RES 2022, 101, 658–665. [Google Scholar] [CrossRef]
- Raveendran, A.V.; Jayadevan, R.; Sashidharan, S. Long COVID: An Overview. Diabetes Metab. Syndr. Clin. Res. Rev. 2021, 15, 869–875. [Google Scholar] [CrossRef]
- Tenforde, M.W.; Kim, S.S.; Lindsell, C.J.; Rose, E.B.; Shapiro, N.I.; Files, D.C.; Gibbs, K.W.; Erickson, H.L.; Steingrub, J.S.; Smithline, H.A.; et al. Symptom Duration and Risk Factors for Delayed Return to Usual Health among Outpatients with COVID-19 in a Multistate Health Care Systems Network-United States, March-June 2020. MMWR. Morb. Mortal. Wkly. Rep. 2020, 69, 993–998. [Google Scholar] [CrossRef]
- Huang, C.; Huang, L.; Wang, Y.; Li, X.; Ren, L.; Gu, X.; Kang, L.; Guo, L.; Liu, M.; Zhou, X.; et al. 6-Month Consequences of COVID-19 in Patients Discharged from Hospital: A Cohort Study. Lancet 2021, 397, 220–232. [Google Scholar] [CrossRef]
- Thye, A.Y.-K.; Law, J.W.-F.; Tan, L.T.-H.; Pusparajah, P.; Ser, H.-L.; Thurairajasingam, S.; Letchumanan, V.; Lee, L.-H. Psychological Symptoms in COVID-19 Patients: Insights into Pathophysiology and Risk Factors of Long COVID-19. Biology 2022, 11, 61. [Google Scholar] [CrossRef]
- Davis, H.E.; Assaf, G.S.; McCorkell, L.; Wei, H.; Low, R.J.; Re’em, Y.; Redfield, S.; Austin, J.P.; Akrami, A. Characterizing Long COVID in an International Cohort: 7 Months of Symptoms and Their Impact. eClinicalMedicine 2021, 38, 101019. [Google Scholar] [CrossRef] [PubMed]
- Orrù, G.; Bertelloni, D.; Diolaiuti, F.; Mucci, F.; Di Giuseppe, M.; Biella, M.; Gemignani, A.; Ciacchini, R.; Conversano, C. Long-COVID Syndrome? A Study on the Persistence of Neurological, Psychological and Physiological Symptoms. Healthcare 2021, 9, 575. [Google Scholar] [CrossRef] [PubMed]
- Petty, R.E.; Wegener, D.T.; Fabrigar, L.R. Attitudes and Attitude Change. Annu. Rev. Psychol. 1997, 48, 609–647. [Google Scholar] [CrossRef] [PubMed]
- Janis, I.L.; King, B.T. The Influence of Role Playing on Opinion Change. J. Abnorm. Soc. Psychol. 1954, 49, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Albarracín, D. Action and Inaction in a Social World: Predicting and Changing Attitudes and Behavior; Cambridge University Press: Cambridge, UK, 2021; ISBN 978-1-108-87970-5. [Google Scholar]
- Bem, D.J. Self-Perception. In Advances in Experimental Social Psychology; Academic Press: Cambridge, MA, USA, 1972; Volume 6, pp. 1–62. [Google Scholar]
- Albarracín, D. Cognition in Persuasion: An Analysis of Information Processing in Response to Persuasive Communications. In Advances in Experimental Social Psychology; Academic Press: Cambridge, MA, USA, 2002; Volume 34, pp. 61–130. [Google Scholar]
- Haubrich, J.; Bernabo, M.; Nader, K. Noradrenergic Projections from the Locus Coeruleus to the Amygdala Constrain Fear Memory Reconsolidation. eLife 2020, 9, e57010. [Google Scholar] [CrossRef]
- Rachman, S. Systematic Desensitization. Psychol. Bull. 1967, 67, 93–103. [Google Scholar] [CrossRef]
- Abramowitz, J.S. Variants of Exposure and Response Prevention in the Treatment of Obsessive-Compulsive Disorder: A Meta-Analysis. Behav. Ther. 1996, 27, 583–600. [Google Scholar] [CrossRef]
- Festinger, L. A Theory of Cognitive Dissonance; Stanford University Press: Stanford, CA, USA, 1962; ISBN 978-0-8047-0911-8. [Google Scholar]
- Pham, P.N.; Weinstein, H.M.; Longman, T. Trauma and PTSD Symptoms in RwandaImplications for Attitudes Toward Justice and Reconciliation. JAMA 2004, 292, 602–612. [Google Scholar] [CrossRef]
- Shrigley, R.L. Attitude and Behavior Are Correlates. J. Res. Sci. Teach. 1990, 27, 97–113. [Google Scholar] [CrossRef]
- Cialdini, R.B.; Petty, R.E.; Cacioppo, J.T. Attitude and Attitude Change. Annu. Rev. Psychol. 1981, 32, 357–404. [Google Scholar] [CrossRef]
- Tourangeau, R.; Rasinski, K.A. Cognitive Processes Underlying Context Effects in Attitude Measurement. Psychol. Bull. 1988, 103, 299–314. [Google Scholar] [CrossRef]
- Torracinta, L.; Tanner, R.; Vanderslott, S. MMR Vaccine Attitude and Uptake Research in the United Kingdom: A Critical Review. Vaccines 2021, 9, 402. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Shekhar, R.; Kottewar, S.; Upadhyay, S.; Singh, M.; Pathak, D.; Kapuria, D.; Barrett, E.; Sheikh, A.B. COVID-19 Vaccine Hesitancy and Attitude toward Booster Doses among US Healthcare Workers. Vaccines 2021, 9, 1358. [Google Scholar] [CrossRef] [PubMed]
- Abebe, H.; Shitu, S.; Mose, A. Understanding of COVID-19 Vaccine Knowledge, Attitude, Acceptance, and Determinates of COVID-19 Vaccine Acceptance Among Adult Population in Ethiopia. Infect. Drug Resist. 2021, 14, 2015–2025. [Google Scholar] [CrossRef]
- Sallam, M. COVID-19 Vaccine Hesitancy Worldwide: A Concise Systematic Review of Vaccine Acceptance Rates. Vaccines 2021, 9, 160. [Google Scholar] [CrossRef]
- Biswas, M.R.; Alzubaidi, M.S.; Shah, U.; Abd-Alrazaq, A.A.; Shah, Z. A Scoping Review to Find Out Worldwide COVID-19 Vaccine Hesitancy and Its Underlying Determinants. Vaccines 2021, 9, 1243. [Google Scholar] [CrossRef]
- Aw, J.; Seng, J.J.B.; Seah, S.S.Y.; Low, L.L. COVID-19 Vaccine Hesitancy—A Scoping Review of Literature in High-Income Countries. Vaccines 2021, 9, 900. [Google Scholar] [CrossRef]
- Dror, A.A.; Eisenbach, N.; Taiber, S.; Morozov, N.G.; Mizrachi, M.; Zigron, A.; Srouji, S.; Sela, E. Vaccine Hesitancy: The next Challenge in the Fight against COVID-19. Eur. J. Epidemiol. 2020, 35, 775–779. [Google Scholar] [CrossRef]
- Piltch-Loeb, R.; Silver, D.R.; Kim, Y.; Norris, H.; McNeill, E.; Abramson, D.M. Determinants of the COVID-19 Vaccine Hesitancy Spectrum. PLoS ONE 2022, 17, e0267734. [Google Scholar] [CrossRef]
- Khubchandani, J.; Sharma, S.; Price, J.H.; Wiblishauser, M.J.; Webb, F.J. COVID-19 Morbidity and Mortality in Social Networks: Does It Influence Vaccine Hesitancy? Int. J. Environ. Res. Public Health 2021, 18, 9448. [Google Scholar] [CrossRef]
- Okoli, G.N.; Abou-Setta, A.M.; Neilson, C.J.; Chit, A.; Thommes, E.; Mahmud, S.M. Determinants of Seasonal Influenza Vaccine Uptake Among the Elderly in the United States: A Systematic Review and Meta-Analysis. Gerontol. Geriatr. Med. 2019, 5, 2333721419870345. [Google Scholar] [CrossRef] [PubMed]
- Duradoni, M.; Veloso, M.V.; Gamma, M.L.; Monciatti, A.M.; Guazzini, A. Italian Version of the Vaccination Fear Scale (VFS-6): Internal and External Validation. Mediterr. J. Clin. Psychol. 2022, 10. [Google Scholar] [CrossRef]
- Gazzetta Ufficiale della Repubblica Italiana. DECRETO-LEGGE 7 Gennaio 2022, n. 1 (Raccolta 2022). Misure Urgenti per Fronteggiare L’emergenza COVID-19, in Particolare Nei Luoghi Di Lavoro, Nelle Scuole e Negli Istituti Della Formazione Superiore. (22G00002) (GU Serie Generale n.4 Del 7 January 2022). Available online: https://www.gazzettaufficiale.it/eli/id/2022/01/07/22G00002/sg (accessed on 3 January 2022).
- Malas, O.; Tolsá, M.D. Vaccination Fear Scale (VFS-6): Development and Initial Validation. Mediterr. J. Clin. Psychol. 2021, 9, 19. [Google Scholar] [CrossRef]
- Betsch, C.; Schmid, P.; Heinemeier, D.; Korn, L.; Holtmann, C.; Böhm, R. Beyond Confidence: Development of a Measure Assessing the 5C Psychological Antecedents of Vaccination. PLoS ONE 2018, 13, e0208601. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Buchner, A.; Lang, A.-G. Statistical Power Analyses Using G*Power 3.1: Tests for Correlation and Regression Analyses. Behav. Res. Methods 2009, 41, 1149–1160. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Lang, A.-G.; Buchner, A. G*Power 3: A Flexible Statistical Power Analysis Program for the Social, Behavioral, and Biomedical Sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef]
- Coronavirus (COVID-19). Available online: https://news.google.com/covid19/map?hl=it&gl=IT&ceid=IT:it (accessed on 7 September 2022).
- Simon, M.A.; Luginbuhl, R.D.; Parker, R. Reduced Incidence of Long-COVID Symptoms Related to Administration of COVID-19 Vaccines Both Before COVID-19 Diagnosis and Up to 12 Weeks After. medRxiv 2021. [Google Scholar] [CrossRef]
- Mahase, E. Covid-19: Vaccinated People Are Less Likely to Get Long Covid, Review Finds. BMJ 2022, 376, o407. [Google Scholar] [CrossRef]
- Yasmin, F.; Najeeb, H.; Moeed, A.; Naeem, U.; Asghar, M.S.; Chughtai, N.U.; Yousaf, Z.; Seboka, B.T.; Ullah, I.; Lin, C.-Y.; et al. COVID-19 Vaccine Hesitancy in the United States: A Systematic Review. Front. Public Health 2021, 9, 770985. [Google Scholar] [CrossRef]
- Eberhardt, J.; Ling, J. Predicting COVID-19 Vaccination Intention Using Protection Motivation Theory and Conspiracy Beliefs. Vaccine 2021, 39, 6269–6275. [Google Scholar] [CrossRef]
- Graziani, A.R.; Botindari, L.; Menegatti, M.; Moscatelli, S. So Far, So Close: Identification with Proximal and Distal Groups as a Resource in Dealing with the COVID-19 Pandemic. Int. J. Environ. Res. Public Health 2022, 19, 11231. [Google Scholar] [CrossRef] [PubMed]
- Moscatelli, S.; Graziani, A.R.; Botindari, L.; Ciaffoni, S.; Menegatti, M. “Everything Will Be All Right!” National and European Identification as Predictors of Positive Expectations for the Future During the COVID-19 Emergency. Front. Psychol. 2021, 12, 723518. [Google Scholar] [CrossRef] [PubMed]
- Mohamed-Hussein, A.A.R.; Amin, M.T.; Makhlouf, H.A.; Makhlouf, N.A.; Galal, I.; Abd-Elaal, H.K.; Abdeltawab, D.; Kholief, K.M.S.; Hashem, M.K. Non-Hospitalised COVID-19 Patients Have More Frequent Long COVID-19 Symptoms. Int. J. Tuberc. Lung Dis. 2021, 25, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Szlamka, Z.; Kiss, M.; Bernáth, S.; Kámán, P.; Lubani, A.; Karner, O.; Demetrovics, Z. Mental Health Support in the Time of Crisis: Are We Prepared? Experiences With the COVID-19 Counselling Programme in Hungary. Front. Psychiatry 2021, 12, 655211. [Google Scholar] [CrossRef] [PubMed]
- Wiederhold, B.K. Social Media Use During Social Distancing. Cyberpsychol. Behav. Soc. Netw. 2020, 23, 275–276. [Google Scholar] [CrossRef] [PubMed]
- Guazzini, A.; Gursesli, M.C.; Serritella, E.; Tani, M.; Duradoni, M. Obsessive-Compulsive Disorder (OCD) Types and Social Media: Are Social Media Important and Impactful for OCD People? Eur. J. Investig. Health Psychol. Educ. 2022, 12, 1108–1120. [Google Scholar] [CrossRef]
- Venegas-Vera, A.V.; Colbert, G.B.; Lerma, E.V. Positive and Negative Impact of Social Media in the COVID-19 Era. Rev. Cardiovasc. Med. 2020, 21, 561–564. [Google Scholar] [CrossRef]
- Gottlieb, M.; Dyer, S. Information and Disinformation: Social Media in the COVID-19 Crisis. Acad. Emerg. Med. 2020, 27, 640–641. [Google Scholar] [CrossRef]
- Goel, A.; Gupta, L. Social Media in the Times of COVID-19. J. Clin. Rheumatol. 2020, 26, 220–223. [Google Scholar] [CrossRef]
- Wilson, S.L.; Wiysonge, C. Social Media and Vaccine Hesitancy. BMJ Glob. Health 2020, 5, e004206. [Google Scholar] [CrossRef]
- Clark, S.E.; Bledsoe, M.C.; Harrison, C.J. The Role of Social Media in Promoting Vaccine Hesitancy. Curr. Opin. Pediatrics 2022, 34, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Piedrahita-Valdés, H.; Piedrahita-Castillo, D.; Bermejo-Higuera, J.; Guillem-Saiz, P.; Bermejo-Higuera, J.R.; Guillem-Saiz, J.; Sicilia-Montalvo, J.A.; Machío-Regidor, F. Vaccine Hesitancy on Social Media: Sentiment Analysis from June 2011 to April 2019. Vaccines 2021, 9, 28. [Google Scholar] [CrossRef] [PubMed]
- Pulido, C.M.; Redondo-Sama, G.; Sordé-Martí, T.; Flecha, R. Social Impact in Social Media: A New Method to Evaluate the Social Impact of Research. PLoS ONE 2018, 13, e0203117. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, S.; Singh, T. Social Media Its Impact with Positive and Negative Aspects. IJCATR 2016, 5, 71–75. [Google Scholar] [CrossRef]
- Modi, C.; Böhm, V.; Ferraro, S.; Stein, G.; Seljak, U. Estimating COVID-19 Mortality in Italy Early in the COVID-19 Pandemic. Nat. Commun. 2021, 12, 2729. [Google Scholar] [CrossRef] [PubMed]
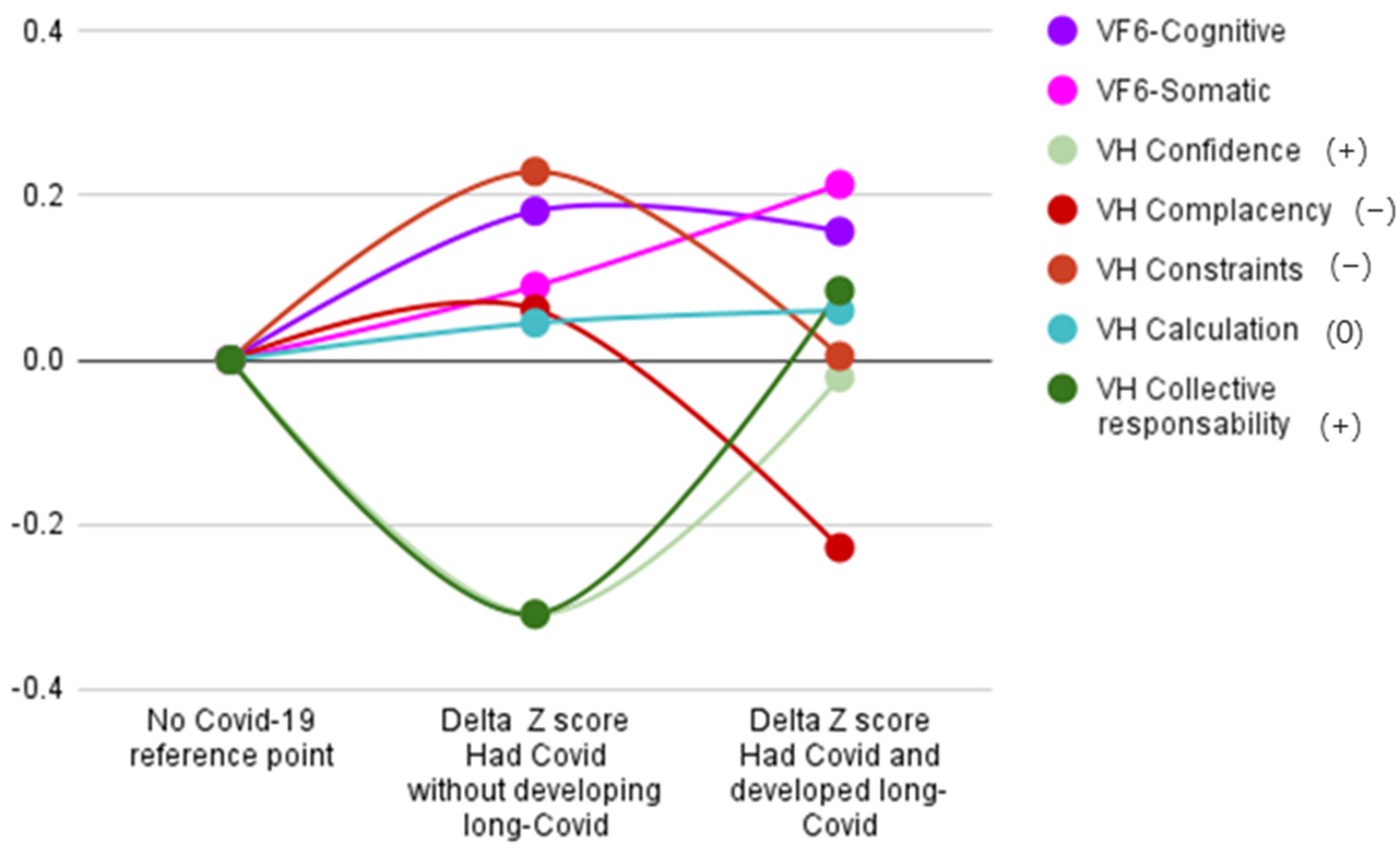
| Had No COVID-19 (N = 795) | Had COVID without Developing Long-COVID (N = 167) | Had COVID-19 and Developed Long-COVID (N = 149) | ||||
|---|---|---|---|---|---|---|
| Variable | M | s.d. | M | s.d. | M | s.d. |
| VFS-6 cognitive | 7.42 | 4.04 | 8.15 | 4.38 | 8.05 | 3.98 |
| VFS-6 somatic | 4.88 | 2.91 | 5.14 | 3.24 | 5.50 | 3.25 |
| VH confidence | 10.39 | 3.72 | 9.24 | 3.75 | 10.31 | 2.88 |
| VH complacency | 5.35 | 2.76 | 5.52 | 3.24 | 4.72 | 2.36 |
| VH constraints | 4.69 | 2.23 | 5.2 | 2.46 | 4.70 | 2.37 |
| VH calculation | 10.99 | 2.68 | 11.11 | 2.60 | 11.15 | 2.54 |
| VH collective responsibility | 12.05 | 2.98 | 11.13 | 3.29 | 12.3 | 2.63 |
| Criterion Variable | Mean Squares | d.f. | F | p. |
|---|---|---|---|---|
| VFS-6 cognitive | 53.89 | 2; 1110 | 3.23 | 0.05 |
| VFS-6 somatic | 25.65 | 2; 1110 | 2.83 | 0.06 |
| VH confidence | 92.23 | 2; 1110 | 8.32 | <0.001 |
| VH complacency | 138.73 | 2; 1110 | 17.82 | <0.001 |
| VH constraints | 18.44 | 2; 1110 | 3.54 | 0.03 |
| VH calculation | 2.42 | 2; 1110 | 0.34 | 0.71 |
| VH collective responsibility | 70.16 | 2; 1110 | 7.88 | <0.001 |
| Had No COVID-19 vs. Had COVID without Developing Long-COVID | Had No COVID-19 vs. Had COVID and Developed Long-COVID | Had COVID without Developing Long-COVID vs. Had COVID and Developed Long-COVID | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Criterion Variable | t | d.f. | d | t | d.f. | d | t | d.f. | d |
| VFS-6 cognitive | −1.99 | 229.25 | n.c. | −1.79 | 209.13 | n.c. | 0.20 | 313.89 | n.c. |
| VFS-6 somatic | −0.97 | 226.10 | n.c. | −2.16 * | 195.24 | −0.20 | −0.97 | 309.65 | n.c. |
| VH confidence | 3.70 *** | 225.13 | 0.33 | 0.31 | 228.20 | n.c. | −2.87 ** | 308.22 | −0.32 |
| VH complacency | −4.35 *** | 219.61 | −0.39 | 2.89 ** | 230.81 | 0.25 | 5.67 *** | 302.43 | 0.64 |
| VH constraints | −2.48 * | 226.63 | −0.22 | −0.02 | 199.99 | n.c. | 1.86 | 312.15 | n.c. |
| VH calculation | −0.55 | 245.40 | n.c. | −0.74 | 241.59 | n.c. | −0.16 | 311.60 | n.c. |
| VH collective responsibility | 3.36 ** | 226.67 | 0.29 | −1.04 | 225.37 | n.c. | −3.53 *** | 310.33 | −0.40 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duradoni, M.; Gursesli, M.C.; Materassi, L.; Serritella, E.; Guazzini, A. The Long-COVID Experience Changed People’s Vaccine Hesitancy but Not Their Vaccination Fear. Int. J. Environ. Res. Public Health 2022, 19, 14550. https://doi.org/10.3390/ijerph192114550
Duradoni M, Gursesli MC, Materassi L, Serritella E, Guazzini A. The Long-COVID Experience Changed People’s Vaccine Hesitancy but Not Their Vaccination Fear. International Journal of Environmental Research and Public Health. 2022; 19(21):14550. https://doi.org/10.3390/ijerph192114550
Chicago/Turabian StyleDuradoni, Mirko, Mustafa Can Gursesli, Letizia Materassi, Elena Serritella, and Andrea Guazzini. 2022. "The Long-COVID Experience Changed People’s Vaccine Hesitancy but Not Their Vaccination Fear" International Journal of Environmental Research and Public Health 19, no. 21: 14550. https://doi.org/10.3390/ijerph192114550
APA StyleDuradoni, M., Gursesli, M. C., Materassi, L., Serritella, E., & Guazzini, A. (2022). The Long-COVID Experience Changed People’s Vaccine Hesitancy but Not Their Vaccination Fear. International Journal of Environmental Research and Public Health, 19(21), 14550. https://doi.org/10.3390/ijerph192114550









