Effects of Mental Fatigue on Reaction Time in Sportsmen
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
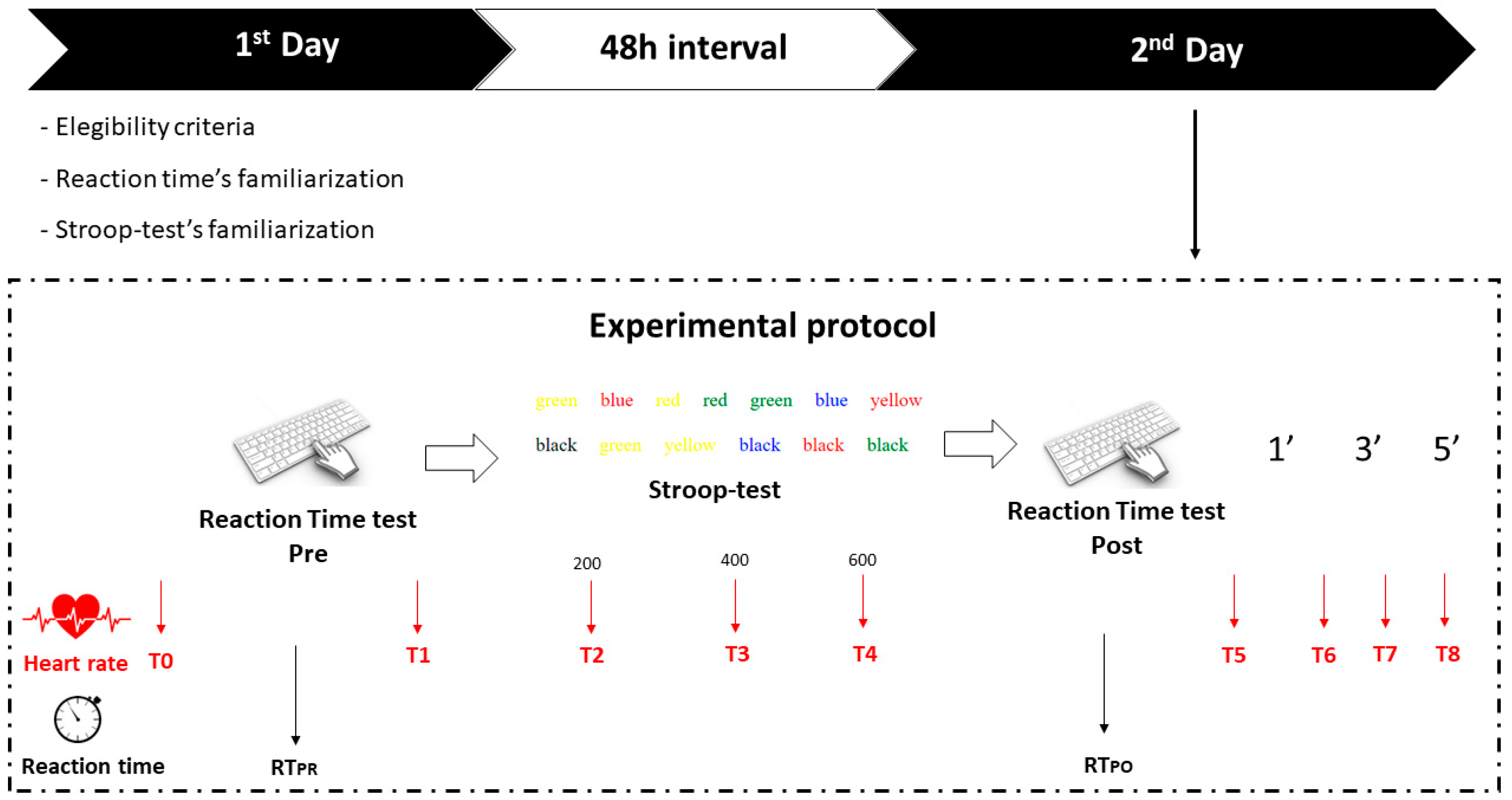
2.2. Experimental Setting
- C key combined with yellow color
- V key combined with red color
- Button B combined with green color
- N button combined with blue color
- M button combined with white color
- T0—At the initial trial
- T1—At the end of the Reaction test (PRE)
- T2—After 200 questions of the Stroop test
- T3—After 400 questions of the Stroop test
- T4—At the end of the Stroop test, or 600 questions
- T5—At the end of the Reaction test (POST)
- T6—At rest, after 1’
- T7—At rest, after 3’
- T8—At rest, after 5’
- Heart rate (HR)
- Resting heart rate (HRR)
- Maximum heart rate (HRMAX)
- Reaction time (RT)
- Heart Rate after Reaction Time before Stroop test (HRPRTR)
- Heart Rate after Reaction Time after Stroop test (HRPOTR)
- Reaction time before the Stroop test (RTPR)
- Reaction time after Stroop test (RTPO)
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ishii, A.; Tanaka, M.; Watanabe, Y. Neural mechanisms of mental fatigue. Rev. Neurosci. 2014, 25, 469–479. [Google Scholar] [CrossRef] [PubMed]
- Boksem, M.A.S.; Tops, M. Mental fatigue: Costs and benefits. Brain Res. Rev. 2008, 59, 125–139. [Google Scholar] [CrossRef] [PubMed]
- Marcora, S.M.; Staiano, W.; Manning, V. Mental fatigue impairs physical performance in humans. J. Appl. Physiol. 2009, 106, 857–864. [Google Scholar] [CrossRef]
- Habay, J.; Van Cutsem, J.; Verschueren, J.; De Bock, S.; Proost, M.; De Wachter, J.; Tassignon, B.; Meeusen, R.; Roelands, B. Mental fatigue and sport-specific psychomotor performance: A systematic review. Sports Med. 2021, 51, 1527–1548. [Google Scholar] [CrossRef] [PubMed]
- Hemamalini, R. Influence of rotating shift work on visual reaction time and visual evoked potential. JCDR 2014, 8, BC04–BC07. [Google Scholar]
- Kowalski, K.L.; Tierney, B.C.; Christie, A.D. Mental fatigue does not substantially alter neuromuscular function in young, healthy males and females. Physiol. Behav. 2022, 11, 38–55. [Google Scholar] [CrossRef]
- Russell, S.; Jenkins, D.; Rynne, S.; Halson, S.L.; Kelly, V. What is mental fatigue in elite sport? Perceptions from athletes and staff. EJSS 2019, 19, 1367–1376. [Google Scholar] [CrossRef]
- Tomei, G.; Rosati, M.V.; Martini, A.; Tarsitani, L.; Biondi, M.; Pancheri, P.; Monti, C.; Ciarrocca, M.; Capozzella, A.; Tomei, F. Assessment of subjective stress in video display terminal workers. Ind. Health 2006, 44, 291–295. [Google Scholar] [CrossRef]
- Li, G.; Huang, S.; Xu, W.; Jiao, W.; Jiang, Y.; Gao, Z.; Zhang, J. The impact of mental fatigue on brain activity: A comparative study both in resting state and task state using EEG. BMC Neurosci. 2020, 21, 20. [Google Scholar] [CrossRef]
- Skarpsno, E.S.; Nilsen, T.I.L.; Sand, T.; Hagen, K.; Mork, P.J. Work-related mental fatigue, physical activity and risk of insomnia symptoms: Longitudinal data from the Norwegian HUNT Study. Behav. Sleep Med. 2020, 18, 488–499. [Google Scholar] [CrossRef]
- Penna, E.M.; Filho, E.; Wanner, S.P.; Campos, B.T.; Quinan, G.R.; Mendes, T.T.; Smith, M.R.; Prado, L.S. Mental fatigue impairs physical performance in young swimmers. Pediatr. Exerc. Sci. 2018, 30, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Rauch, W.A.; Schmitt, K. Fatigue of cognitive control in the Stroop-Task. In Proceedings of the 31st Annual Conference of the Cognitive Science Society, Amsterdam, The Netherlands, 29 July–1 August 2009; pp. 750–755. [Google Scholar]
- Scarpina, F.; Tagini, S. The Stroop color and word test. Front. Psychol. 2017, 8, 557. [Google Scholar] [CrossRef] [PubMed]
- Slimani, M.; Znazen, H.; Bragazzi, N.; Zguira, M.; Tod, D. The effect of mental fatigue on cognitive and aerobic performance in adolescent active endurance athletes: Insights from a randomized counterbalanced, cross-over trial. J. Clin. Med. 2018, 7, 510. [Google Scholar] [CrossRef] [PubMed]
- Wright, R.A.; Junious, T.R.; Neal, C.; Avello, A.; Graham, C.; Herrmann, L.; Junious, S.; Walton, N. Mental fatigue influence on effort-related cardiovascular response: Difficulty effects and extension across cognitive performance domains. Motiv. Emot. 2007, 31, 219–231. [Google Scholar] [CrossRef]
- Williamson, J.W.; Fadel, P.J.; Mitchell, J.H. New insights into central cardiovascular control during exercise in humans: A central command update. Exp. Physiol. 2006, 91, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Stewart, C.C.; Wright, R.A.; Azor Hui, S.K.; Simmons, A. Outcome expectancy as a moderator of mental fatigue influence on cardiovascular response. Psychophysiology 2009, 46, 1141–1149. [Google Scholar] [CrossRef] [PubMed]
- Bull, F.C.; Al-Ansari, S.S.; Biddle, S.; Borodulin, K.; Buman, M.P.; Cardon, G.; Carty, C.; Chaput, J.P.; Chastin, S.; Chou, R.; et al. World Health Organization 2020 guidelines on physical activity and sedentary behaviour. BJSM 2020, 54, 1451–1462. [Google Scholar] [CrossRef]
- Tanaka, H.; Monahan, K.D.; Seals, D.R. Age-predicted maximal heart rate revisited. J. Am. Coll. Cardiol. 2001, 37, 153–156. [Google Scholar] [CrossRef]
- Nakagawa, S.; Schielzeth, H. A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol. Evol. 2013, 4, 133–142. [Google Scholar] [CrossRef]
- De Giorgio, A.; Iuliano, E.; Turner, A.; Millevolte, C.; Cular, D.; Ardigò, L.P.; Padulo, J. Validity and reliability of a light-based electronic target for testing response time in fencers. J. Strength Cond. Res. 2021, 35, 2636–2644. [Google Scholar] [CrossRef]
- Padulo, J.; Pizzolato, F.; Rodrigues, S.T.; Migliaccio, G.M.; Attene, G.; Curcio, R.; Zagatto, A.M. Task complexity reveals expertise of table tennis players. J. Sport. Med. Phys. Fit. 2016, 56, 149–156. [Google Scholar]
- Padulo, J.; Attene, G.; Migliaccio, G.M.; Cuzzolin, F.; Vando, S.; Ardigò, L.P. Metabolic optimisation of the basketball free throw. J. Sports Sci. 2015, 33, 1454–1458. [Google Scholar] [CrossRef]
- Padulo, J.; Nikolaidis, P.T.; Cular, D.; Iacono, A.D.; Vando, S.; Galasso, M.; Lo Storto, D.; Ardigò, L.P. The effect of heart rate on jump-shot accuracy of adolescent basketball players. Front. Physiol. 2018, 3, 1065. [Google Scholar] [CrossRef]
- Ardigò, L.P.; Kuvacic, G.; Iacono, A.D.; Dascanio, G.; Padulo, J. Effect of heart rate on basketball three-point shot accuracy. Front. Physiol. 2018, 6, 75. [Google Scholar] [CrossRef]


| All Participants (n = 64) | Male (n = 34) | Female (n = 30) | p (Male × Female) | |
|---|---|---|---|---|
| Heart Rate | ||||
| HRMAX (bpm) | 185.8 ± 4.3 | 187.6 ± 2.2 | 184.3 ± 5.1 | 0.006 |
| T0: HRR (%HRMAX) | 35.1 ± 5.4 | 34.9 ± 6.0 | 35.2 ± 4.9 | 0.603 |
| T1: HRPRTR (%HRMAX) | 38.6 ± 7.6 * | 37.8 ± 8.1 | 39.4 ± 7.2 * | 0.250 |
| T2: (%HRMAX) | 37.0 ± 5.7 *† | 36.8 ± 6.7 | 37.2 ± 4.8 *† | 0.406 |
| T3: (%HRMAX) | 40.2 ± 6.4 * | 40.0 ± 6.9 * | 40.4 ± 5.9 * | 0.758 |
| T4: (%HRMAX) | 42.5 ± 7.1 *† | 42.0 ± 7.5 *† | 42.9 ± 6.8 * | 0.559 |
| T5: HRPOTR (%HRMAX) | 38.5 ± 7.1 * | 39.0 ± 7.8 * | 38.1 ± 6.5 * | 0.831 |
| T6: (%HRMAX) | 37.5 ± 6.7 * | 37.6 ± 7.6 * | 37.3 ± 5.9 † | 0.998 |
| T7: (%HRMAX) | 37.4 ± 6.0 * | 37.6 ± 6.9 * | 37.2 ± 5.3 † | 0.352 |
| T8: (%HRMAX) | 37.0 ± 6.6 * | 37.3 ± 7.9 * | 36.8 ± 5.4 † | 0.169 |
| Reaction Time | ||||
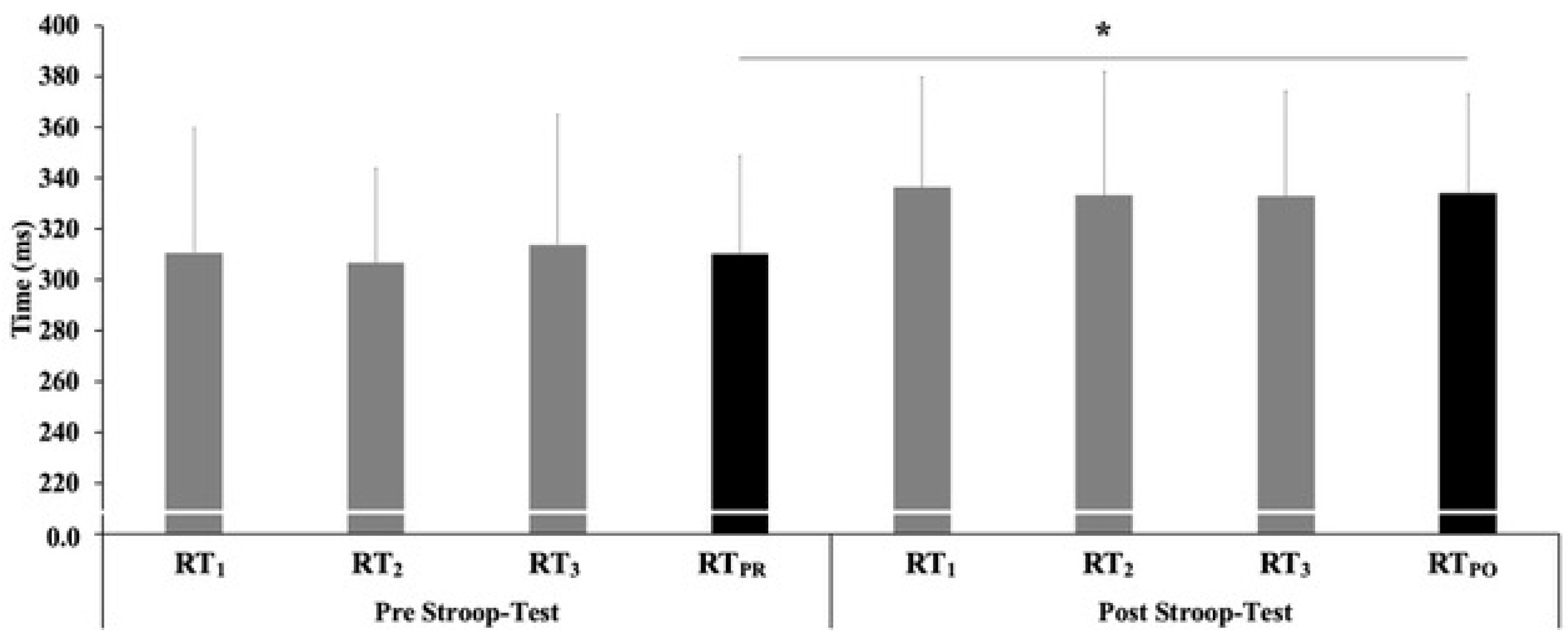
| RTPR (ms) | 308.3 ± 40.1 | 316.5 ± 39.9 | 301.1 ± 39.4 | 0.410 |
| RTPO (ms) | 334.1 ± 40.7 § | 344.3 ± 39.9 § | 325.1 ± 39.9 § | 0.067 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Migliaccio, G.M.; Di Filippo, G.; Russo, L.; Orgiana, T.; Ardigò, L.P.; Casal, M.Z.; Peyré-Tartaruga, L.A.; Padulo, J. Effects of Mental Fatigue on Reaction Time in Sportsmen. Int. J. Environ. Res. Public Health 2022, 19, 14360. https://doi.org/10.3390/ijerph192114360
Migliaccio GM, Di Filippo G, Russo L, Orgiana T, Ardigò LP, Casal MZ, Peyré-Tartaruga LA, Padulo J. Effects of Mental Fatigue on Reaction Time in Sportsmen. International Journal of Environmental Research and Public Health. 2022; 19(21):14360. https://doi.org/10.3390/ijerph192114360
Chicago/Turabian StyleMigliaccio, Gian Mario, Gloria Di Filippo, Luca Russo, Tania Orgiana, Luca Paolo Ardigò, Marcela Zimmermann Casal, Leonardo Alexandre Peyré-Tartaruga, and Johnny Padulo. 2022. "Effects of Mental Fatigue on Reaction Time in Sportsmen" International Journal of Environmental Research and Public Health 19, no. 21: 14360. https://doi.org/10.3390/ijerph192114360
APA StyleMigliaccio, G. M., Di Filippo, G., Russo, L., Orgiana, T., Ardigò, L. P., Casal, M. Z., Peyré-Tartaruga, L. A., & Padulo, J. (2022). Effects of Mental Fatigue on Reaction Time in Sportsmen. International Journal of Environmental Research and Public Health, 19(21), 14360. https://doi.org/10.3390/ijerph192114360













