Manual Therapy Effect in Placebo-Controlled Trials: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search
2.2. Study Selection
- Types of Studies. Parallel RTCs carried out in humans were included, however, cross-over designs were excluded to avoid the carry-over effect. In addition, letters, reports, and abstracts from congresses were also excluded.
- Types of population. All studies which were conducted with subjects suffering from pain were included, regardless of the characteristics of the participants. Nevertheless, when the pain was artificially induced in the subjects (i.e., experimental pain models), studies were excluded.
- Types of interventions. It was compulsory for the studies to be included, to have used as a unique technique one of the following manual therapy techniques: manipulation, mobilization, soft tissue techniques, and neural mobilization in at least one intervention group. Intervention groups that were complemented with other techniques (i.e., electrotherapy, exercise) were not included, to allow the analysis of the individual effect of each technique. In the same way, therapies that involved active subject participation (i.e., exercise) were excluded. Finally, alternative medicine therapies such as reflexology or holistic treatments were excluded.
- Types of comparison. To be included in the systematic review, the studies had to include at least one sham group, termed by the authors as: a placebo group, or sham group. If the subjects did not receive any intervention in the placebo or sham group, it was not considered a valid sham comparison group.
- Types of outcomes. Pain intensity had to be assessed with rating scales such as the Visual Analog Scale (VAS), Numerical Rating Scale (NRS), Numerical Pain Rating Scale (NPRS), or similar.
- Time points. For the quantitative analysis, the pre-treatment assessment and the first available post-treatment assessment were chosen.
2.3. Selection of Studies
2.4. Data Extraction
2.5. Blinding Quality Assessment
2.6. Data Analysis and Synthesis
2.7. Assessment of Heterogeneity
2.8. Subgroup Analysis
2.9. Assessment of the Level of Evidence
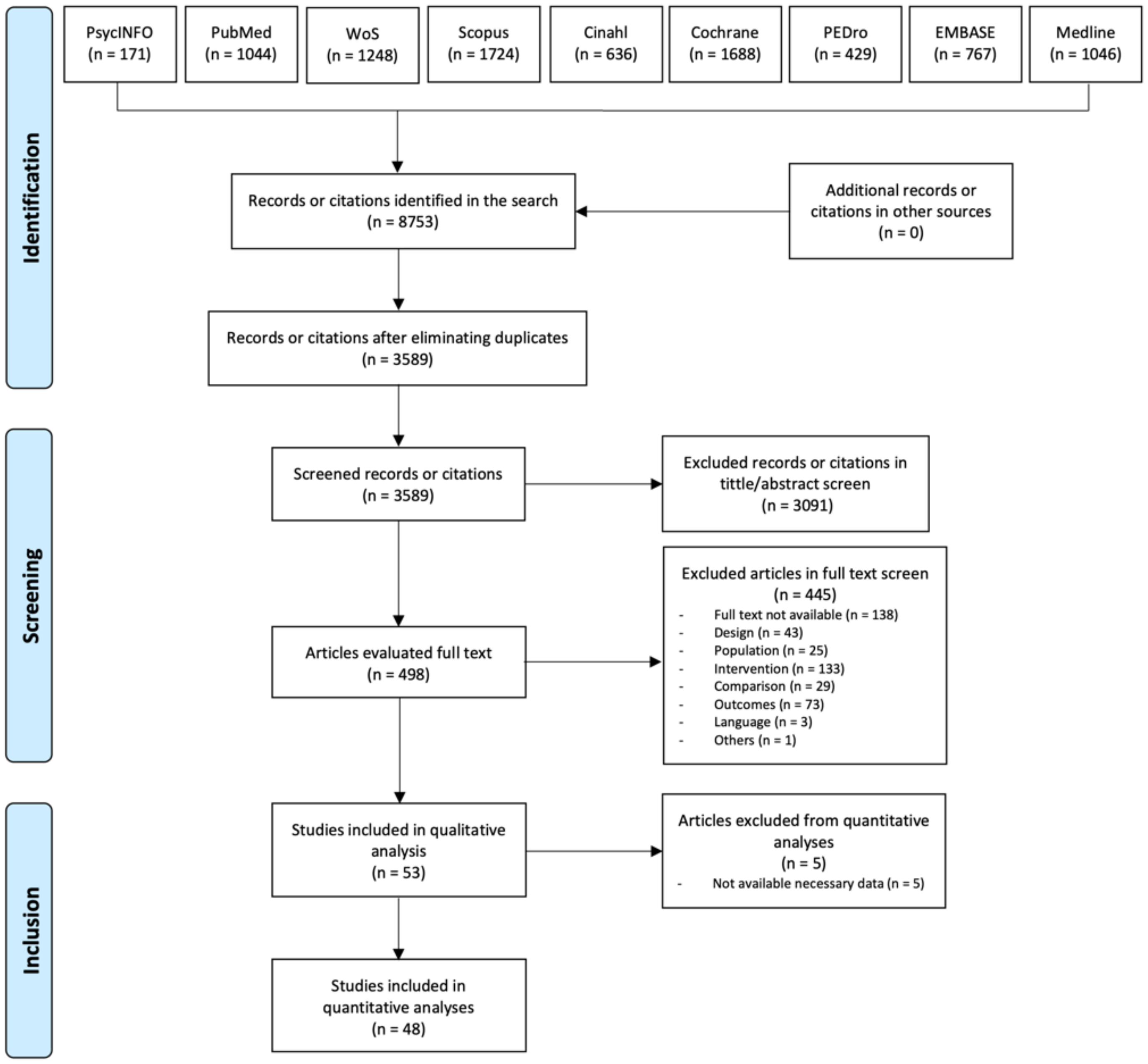
3. Results
3.1. Selection of Trials
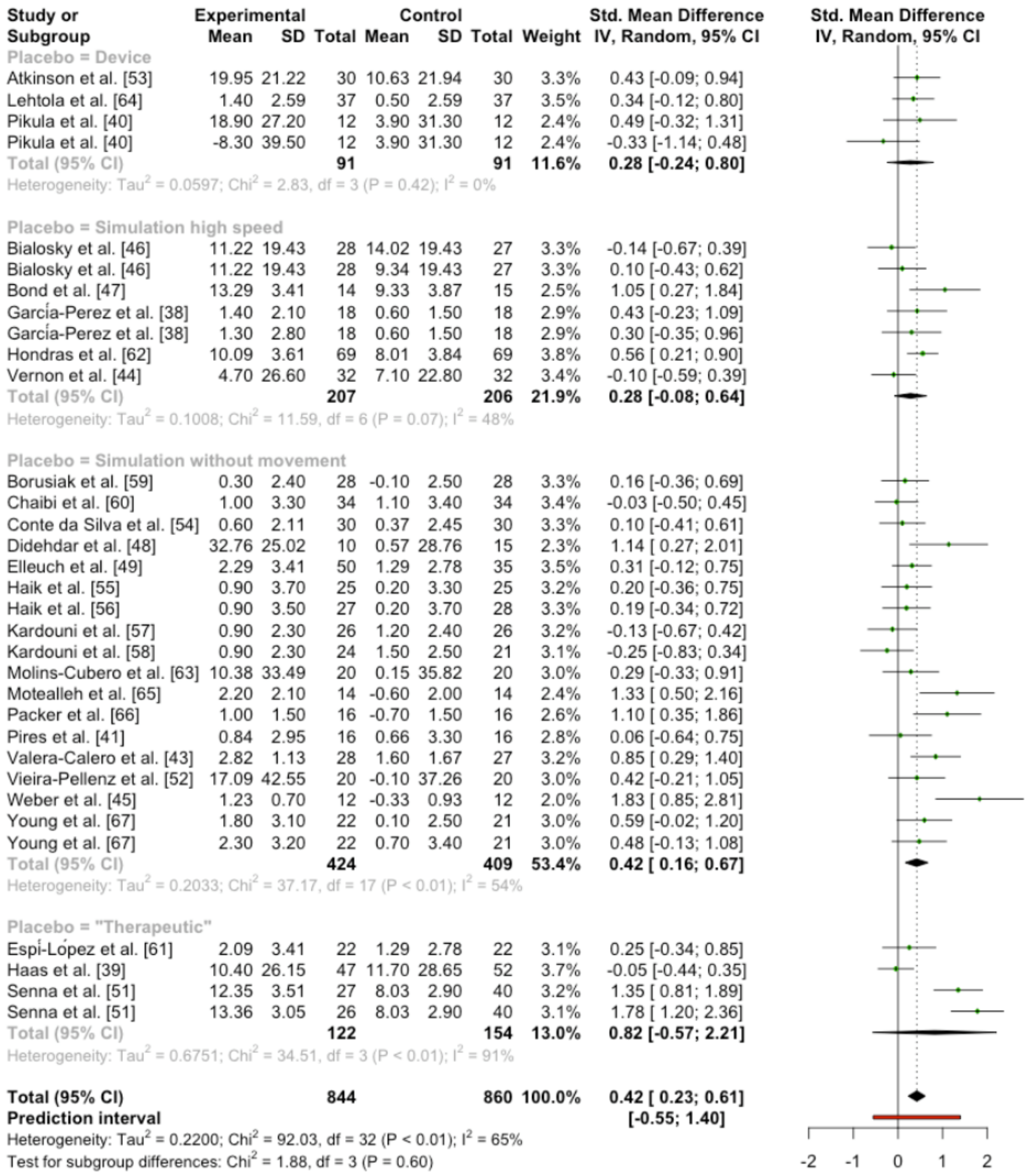
3.2. Trials Using Manipulation Techniques
3.2.1. Participants
3.2.2. Intervention Groups
3.2.3. Placebo
| Manipulation Techniques | |||||||
| Author/Year | Participants | Pathology | Groups | Intervention Group Description | Placebo Group Description | Pain Outcome | Measurements |
| García-Perez-Juana et al. [38] | n = 54 Female (n = 42) Age (years): 37 (8) | Neck pain (Chronic) | Manipulation I (n = 18) Manipulation II (n = 18) Placebo group (n = 18) 1 session | Manipulation I: One midcervical spine manipulation on the right side was performed. Manipulation II: One midcervical spine manipulation on the left side was performed. | Simulation of the technique and even reproduced the rapid application of motion but without inducing the thrust. | NPRS | Preintervention 1-week post-intervention |
| Haas et al. [39] | n = 99 Female (n = 63) Age (years): 42.55 (SD Not available) | Neck pain | Manipulation (n = 47) Placebo group (n = 52) 1 session | Cervical manipulation was performed in the “endfeel finding” | Real cervical manipulation was performed in a “sham endfeel finding”. | VAS | Preintervention Post-intervention |
| Pikula [40] | n = 36 Female (n = 28) Age (years): 42.10 (SD Not available) | Unilateral neck pain | Manipulation I (n = 12) Manipulation II (n = 12) Placebo group (n = 12) 1 session | Manipulation I: cervical manipulation was applied to the same side as the pain Manipulation II: cervical manipulation was applied to the side opposite the pain | A detuned ultrasound was used. | VAS | Preintervention Post-intervention |
| Pires et al. [41] | n = 32 Female (n = 32) Age (years): 24.7 (SD Not available) | Neck pain (Chronic) | Manipulation (n = 16) Placebo group (n = 16) 1 session | Upper thoracic spine manipulation was performed on vertebra T1. | Simulation of the procedure but without the rapid application of motion | VAS | Preintervention Post-intervention 48–72 h post-intervention |
| Sillevis et al. [42] | n = 100 Female (n = 77) Age (years): 44.77 (SD Not available) | Neck pain | Manipulation (n = 50) Placebo group (n = 50) 1 session | Thoracic manipulation in vertebral level T3-T4 was performed in a supine position. | Simulation of the procedure but without the rapid application of motion | VAS | Preintervention Post-intervention |
| Valera-Calero et al. [43] | n = 83 Female (n = 51) Age (years): 36.61 (SD Not available) | Neck pain (Chronic) | Manipulation (n = 28) Mobilization (n = 28) Placebo group (n = 27) 1 session | Cervical manipulation was performed on vertebral levels C5–C6. | Simulation of the procedure but without the rapid application of motion | VAS | Preintervention Post-intervention 1-week post-intervention |
| Vernon et al. [44] | n = 64 Female (n = 34) Age (years): 38.55 (SD Not available) | Neck pain | Manipulation (n = 32) Placebo group (n = 32) 1 session | Cervical manipulation was performed | Simulation of the technique and even reproduced the rapid application of motion but without inducing the thrust. | NPRS | Preintervention 5 min post-intervention 15 min pos-intervention |
| Weber et al. [45] | n = 24 Female (n = 16) Age (years): 38.0 (15.1) | Neck pain (Acute or subacute) | Manipulation (n = 12) Placebo group (n = 12) 1 session | Thoracic spinal manipulation was performed in the mid-thoracic spine. | Simulation of the procedure but without the rapid application of motion | NPRS | Preintervention Post-intervention |
| Bialosky et al. [46] | n = 110 Female (n = 77) Age (years): 31.68 (11.85) | Low back pain | Manipulation (n = 28) Placebo group I (n = 27) Placebo group II (n = 27) Control group: no treatment (n = 28) 5 sessions for 2 weeks | Lumbar manipulation was performed 2 times on each side. | Placebo group I: simulation of the technique and even reproduced the rapid application of motion but without inducing the thrust. Placebo group II: same simulation + increased expectations. | NRS | Preintervention Post-intervention |
| Bond et al. [47] | n = 29 Female: (n = 11) Age (years): 23.86 (5.74) | Low back pain (Chronic) | Manipulation (n = 14) Placebo group (n = 15) 3 sessions per week for 2 weeks | Two lumbopelvic manipulations were performed on both sides of the pelvis, alternating between the left and right sides. | Simulation of the technique and even reproduced the rapid application of motion but without inducing the thrust. | NPRS | Preintervention < 1-week post-intervention |
| Didehdar et al. [48] | n= 25 Female (n = 11) Age (years): 36.6 (SD Not available) | Low back pain (Chronic) | Manipulation (n = 10) Placebo group (n = 15) 3 sessions in 6 days | Sacroiliac and lumbar spine manipulations were performed. | Simulation of the procedure but without the rapid application of motion | NRS | Preintervention 5 weeks post-intervention |
| Elleuch and Ghroubi [49] | n = 85 Female (n = 68) Age (years): 38.34 (10.2) | Low back pain (Chronic) | Manipulation (n = 50) Placebo group (n = 35) 1 session per week for 4 weeks | 4 manipulations to the low back were performed | Simulation of the procedure but without the rapid application of motion | VAS | Preintervention 1-month post-intervention 2 months post-intervention |
| Kawchuk et al. [50] | n = 6 Female (n = 2) Age (years): 36.5 (SD Not available) | Low back pain (Acute) | Manipulation (n = 3) Placebo group (n = 3) 1 session | One lumbar manipulation was performed while the patients were under the effects of general anesthesia | In the placebo group, the patients were told that they were going to receive a manipulation, but no treatment was performed while the patients were under the effects of general anesthesia. | NRS | Preintervention 30 min post-recovery |
| Senna and MacHaly [51] | n = 88 Female (n = 22) Age (years): 41.41 (SD Not available) | Low back pain (Chronic) | Manipulation I (n = 27) Manipulation II (n = 26) Placebo group (n = 40) Manipulation I and sham manipulation: 3 sessions per week for 1 month. Manipulation II: same sessions plus after the first month 1 session every 2 weeks for the next 9 months. | Lumbar manipulation was performed. | Simulation of the technique and even reproduced the rapid application of motion but without inducing the thrust. | VAS | Preintervention Post-intervention 3 months post-intervention 6 months post-intervention 10 months post-intervention |
| Vieira-Pellenz et al. [52] | n = 40 No females Age (years): 38 (9.14) | Degenerative lumbar disease | Manipulation (n = 20) Placebo group (n = 20) 1 session | Lumbar manipulation was performed. | Simulation of the procedure but without the rapid application of motion | VAS | Preintervention Post-intervention |
| Atkinson et al. [53] | n = 60 Female (n = 17) Age (years): 41.76 (SD Not available) | Shoulder pain (Rotator cuff tendinopathy) | Manipulation (n = 30) Placebo group (n = 30) 6 sessions for 2 weeks | Shoulder manipulation was performed. | A detuned laser was used. | NRS | Preintervention Post-intervention third session Post-intervention sixth session |
| Conte-da-Silva et al. [54] | n = 60 Female (n = 41) Age (years): 45.26 (SD Not available) | Shoulder pain | Manipulation (n = 30) Placebo group (n = 30) 1 session | Thoracic manipulation was performed in prone. | Simulation of the procedure but without the rapid application of motion | VAS | Preintervention Post-intervention |
| Haik et al. [55] | n = 50 Female (n = 18) Age (years): 31.8 (10.9) | Shoulder impingement syndrome | Manipulation (n = 25) Placebo group (n = 25) 1 session | Thoracic manipulation was performed in a seated position. | Simulation of the procedure but without the rapid application of motion | NPRS | Preintervention Post-intervention |
| Haik et al. [56] | n = 61 Female (n = 23) Age (years): 31.9 (SD Not available) | Shoulder impingement syndrome | Manipulation (n = 27) Placebo group (n = 28) 2 sessions for 1 week | Thoracic manipulation was performed in a seated position. | Simulation of the procedure but without the rapid application of motion | NPRS | Preintervention first session Preintervention second session Post-intervention second session 3–4 days post-intervention |
| Kardouni et al. [57] | n = 52 Female (n = 24) Age (years): 32.0 (SD Not available) | Shoulder impingement syndrome | Manipulation (n = 26) Placebo group (n = 26) 1 session | 6 thoracic manipulations were performed per participant: 2 to the lower, 2 to the middle, and 2 to the upper thoracic spine. | Simulation of the procedure but without the rapid application of motion | NPRS | Preintervention Post-intervention 24–48 h post-intervention |
| Kardouni et al. [58] | n = 45 Female (n = 23) Age (years): 31.15 (SD Not available) | Shoulder impingement syndrome | Manipulation (n = 24) Placebo group (n = 21) 1 session | 6 thoracic manipulations were performed per participant: 2 to the lower, 2 to the middle, and 2 to the upper thoracic spine. | Simulation of the procedure but without the rapid application of motion | NPRS | Preintervention Post-intervention 24–48 h post-intervention |
| Borusiak et al. [59] | n = 52 Female (n = 31) Age (years): 11.6 (2.3) | Cervicogenic headache | Manipulation (n = 28) Placebo group (n = 28) 1 session | Cervical manipulation was performed | Simulation of the technique and even reproduced the rapid application of motion but without inducing the thrust. | VAS | Preintervention 2 months post-intervention |
| Chaibi et al. [60] | n = 98 Female (n = 83) Age (years): 39.8 (SD Not available) | Migraine | Manipulation (n = 34) Placebo group (n = 34) Control group (n = 29) 12 sessions for 3 months | Manipulation was performed on an unspecific vertebral level, wherever the therapist considered it necessary. | Simulation of the procedure but without the rapid application of motion | NRS | Preintervention Post-intervention 3 months post-intervention 6 months post-intervention 12 months post-intervention |
| Espí-López et al. [61] | n = 84 Female (n = 68) Age (years): 39.76 (SD Not available) | Tension-type headache | Soft tissue technique (n = 20) Manipulation (n = 22) Combined treatment intervention (n = 20) Placebo group (n = 22) 1 session per week for 4 weeks | Occiput-atlas-axis bilateral manipulation was performed in the manipulation group. | The control group underwent the artery test and remained for 10 min in a resting position. | VAS | Preintervention Post-intervention 1-month post-intervention |
| Hondras et al. [62] | n = 138 Female (n = 138) Age (years): 30.4 (SD Not available) | Primary dysmenorrhea | Manipulation (n = 69) Placebo group (n = 69) 1 session on day 1 of the menstrual cycle for 4 months | Manipulation was performed in all clinically relevant vertebral levels between T10-L5 and both sacroiliac joints. | Simulation of the technique and even reproduced the rapid application of motion but without inducing the thrust. | VAS | Preintervention first session Post-intervention first session Preintervention second session Post-intervention second session |
| Molins-Cubero et al. [63] | n = 40 Female (n = 40) Age (years): 30 (6.10) | Primary dysmenorrhea | Manipulation (n = 20) Placebo group (n = 20) 1 session | Manipulation of bilateral sacroiliac joint and vertebral level L5-S1 was performed. | Simulation of the procedure but without the rapid application of motion | VAS | Preintervention Post-intervention |
| Lehtola et al. [64] | n = 109 Female (n = 109) Age (years): 42.5 (SD Not available) | Thoracic spine pain | Manipulation (n = 37) Acupuncture (n = 35) Placebo group (n = 37) 4 sessions in 3 weeks | Thoracic manipulation was performed in a supine position, between the vertebral levels T3-T8 many times as the therapist considered. | Detuned electrotherapy with suction cups. | VAS | Preintervention first session Preintervention second session 1 week post-intervention |
| Motealleh et al. [65] | n = 28 Female (n = 16) Age (years): 26.5 (SD Not available) | Patellofemoral pain syndrome | Manipulation (n = 14) Placebo group (n = 14) 1 session | Lumbopelvic manipulation was performed once with the patient positioned supine. | Simulation of the procedure but without the rapid application of motion | VAS | Preintervention Post-intervention |
| Packer et al. [66] | n = 32 Female (n = 32) Age (years): 24.7 (SD Not available) | Temporomandibular disorder | Manipulation (n = 16) Placebo group (n = 16) 1 session | Upper thoracic spine manipulation was performed on vertebra T1. | Simulation of the procedure but without the rapid application of motion | VAS | Preintervention Post-intervention 48–72 h post-intervention |
| Young et al. [67] | n = 43 Female (n = 29) Age (years): 46 (SD Not available) | Cervical radiculopathy | Manipulation (n = 22) Placebo group (n = 21) 1 session | Thoracic spine manipulation was performed in the vertebral levels C7-L3 and T4-T9. | Simulation of the procedure but without the rapid application of motion | NPRS | Preintervention Post-intervention 48–72 h post-intervention |
| Soft Tissue Techniques | |||||||
| Author/Year | Participants | Pathology | Groups | Intervention Group Description | Placebo Group Description | Pain Outcome | Measurements |
| Espí-López et al. [61] | n = 84 Female (n = 68) Age (years): 39.76 (SD Not available) | Tension-type headache | Soft tissue technique (n = 20) Manipulation (n = 22) Combined treatment intervention (n = 20) Placebo group (n = 22) 1 session per week for 4 weeks | Soft tissue technique consisted of suboccipital inhibition for 10 min. | The subjects underwent the artery test and remained for 10 min in a resting position. | VAS | Preintervention Post-intervention 1-month post-intervention |
| Antolinos-Campillo et al. [71] | n = 40 Female (n = 17) Age (years): 34 (3.6) | Cervical whiplash | Soft tissue technique (n = 20) Placebo group (n = 20) 1 session | Suboccipital muscle inhibition was performed for 4 min. | Active movement of flexion/extension of the hip and knee joints for 4 min. | VAS | Preintervention Post-intervention |
| Blikstad and Gemmell [72] | n = 45 Female (n = 25) Age (years): 23.8 (1.153) | Non-specific neck pain | Activator device (n = 15) Soft tissue technique (n = 15) Placebo group (n = 15) 1 session | Myofascial band therapy consisted of a slow stroking motion from the lateral shoulder to the mastoid process along the upper trapezius muscle. | A detuned ultrasound was used. | NRS | Preintervention Post-intervention |
| Briem et al. [73] | n = 40 Female (n = 31) Age (years): 34.7 (SD Not available) | Neck pain | Soft tissue technique (n = 20) Placebo group (n = 20) 1 session | Inhibitive distraction was applied for 3 to 3.5 min onto the suboccipital musculotendinous structures. | Gently positioning the hands on the massage area in the absence of movement or pressure. | NPRS | Preintervention Post-intervention |
| Buttagat et al. [74] | n = 50 Female (n = 43) Age (years): 22.24 (SD Not available) | Neck pain (Myofascial trigger points) | Soft tissue technique (n = 25) Placebo group (n = 25) 1 session | Massage was performed for 30 min on the upper neck and upper back. | A detuned microwave diathermy was used for 30 min. | VAS | Preintervention Post-intervention |
| Capó-Juan et al. [75] | n = 75 Female (n = 60) Age (years): 38.28(0.68) | Neck pain (caused by sternocleidomastoid) | Kinesiotape (n = 25) Soft tissue technique (n = 25) Placebo group (n = 25) 1 session | Pressure release was applied on the active myofascial trigger points in the stermocleidomastoid. | Application of algometric bilateral pressure for the placebo group. | NRS | Preintervention 1-week post-intervention |
| Gemmell et al. [76] | n = 45 Female (not available) Age (years): 23.67 (SD Not available) | Neck pain (caused by upper trapezius) | Soft tissue technique I (n = 15) Soft tissue technique II (n = 15) Placebo group (n = 15) 1 session | Soft tissue technique I: Ischemic compression was applied with the thumb to the upper trapezius myofascial trigger point for 30 s to 1 min. Soft tissue technique II: Pressure release was applied with the thumb to the upper trapezius myofascial trigger point for 90 s. | A detuned ultrasound was used. | VAS | Preintervention Post-intervention |
| Chatchawan et al. [77] | n = 72 Female (n = 55) Age (years): 27.37 (SD Not available) | Chronic tension-type and migraine headaches. | Soft tissue technique (n = 36) Placebo group (n = 36) 9 sessions for 3 weeks | The massage group consisted of 25 min of massage on the upper neck, and upper back. | A detuned ultrasound was used. | VAS | Preintervention Post-intervention 3 weeks post-intervention 9 weeks post-intervention |
| Ferragut-Garcías et al. [78] | n = 97 Female (n = 78) Age (years): 39.4 (SD Not available) | Tension-type headache | Soft tissue technique (n = 23) Neural technique (n = 25) Combined treatment intervention (n = 25) Placebo group (n = 24) 2 sessions in the first 2 weeks and 1 session per week in the next 2 weeks | Soft tissue techniques were performed on the sternocleidomastoid muscle, temporal muscle, suboccipital musculature, masster muscle, and upper trapezius muscle. The protocol lasted 15 min. | Superficial massage was performed for 15 min using ultrasound gel to minimize skin stimulation. | VAS | Preintervention Post-intervention 15 days post-intervention 30 days post-intervention |
| Arguisuelas et al. [79] | n = 54 Female (n = 33) Age (years): 46.6 (10.3) | Low back pain (Chronic) | Soft tissue technique (n = 26) Placebo group (n = 26) 2 sessions per week for 2 weeks | Massotherapy was applied to lumbar paravertebral muscles, thoracolumbar fascia, quadratus lumborum, and psoas muscle. (45 min) | Gently positioning the hands on the massage area in absence of movement or pressure. | VAS | Preintervention Post-intervention 10 weeks post-intervention |
| Celenay et al. [80] | n = 63 Female (n = 51) Age (years): 52 (SD Not available) | Low back pain (Chronic) | Soft tissue technique (n = 21) Placebo group (n = 21) Control group: Standardized physiotherapy program (n = 21) 5 sessions per week for 3 weeks | Connective tissue massage was performed for 10 to 15 min each session on the lumbar area. | In the sham connective tissue massage general slow and slight strokes and effleurage on the lower back area were applied using no specific technique and specific muscles. | VAS | Preintervention Post-intervention |
| Field et al. [81] | n = 20 Female (n = 16) Age (years): 47 (SD Not available) | Chronic fatigue syndrome | Soft tissue technique (n = 10) Placebo group (n = 10) 2 sessions per week for 5 weeks | Full body massage was performed for 30 min. | A detuned TENS was used. | VAS | Preintervention first session Post-intervention first session Preintervention last session Post-intervention last session |
| Nourbakhsh and Fearon [82] | n = 23 Female (n = 9) Age (years): 52.55 (SD Not available) | Lateral epicondylitis | Soft tissue technique (n = 11) Placebo group (n = 12) 6 sessions in 2–3 week | Inhibition pressure release for 30 s to 2 min was performed in the most painful myofascial trigger point | Gently positioning the hands on the massage area in absence of movement or pressure. | NRS | Preintervention Post-intervention |
| Tanaka et al. [83] | n = 32 Female (n = 29) Age (years): 81 (SD Not available) | Knee Osteoarthritis | Soft tissue technique (n = 16) Placebo group (n = 16) 1 session | Continuous compression for 5 min was performed on the anterior and distal portions of the medial thigh. | Gently positioning the hands on the massage area in absence of movement or pressure. | VAS | Preintervention Post-intervention |
| Mobilization Techniques | |||||||
| Author/Year | Participants | Pathology | Groups | Intervention Group Description | Placebo Group Description | Pain Outcome | Measurements |
| Valera-Calero et al. [43] | n = 83 Female (n = 51) Age (years): 36.61 (SD Not available) | Neck pain (Chronic) | Manipulation (n = 28) Mobilization (n = 28) Placebo group (n = 27) 1 session | Cervical mobilization was performed on vertebral level C5-C6 for three sets of one min. | Simulation of the manipulation but without the rapid application of motion | VAS | Preintervention Post-intervention 1-week post-intervention |
| Snodgrass et al. [84] | n = 64 Female (n = 48) Age (years): 33.4 (SD Not available) | Neck pain (Chronic) | Mobilization I (n = 22) Mobilization II (n = 21) Placebo group (n = 21) 1 session | Mobilization I: low force posterior-to-anterior cervical mobilization (31± 1 N) was performed in 3 sets of 1 min on the most painful vertebral level. Mobilization II: high force posterior-to-anterior cervical mobilization (89 ± 3 N) was performed in 3 sets of 1 min on the most painful vertebral level. | A detuned laser was used. | VAS | Preintervention Post-intervention 4 days post-intervention |
| Kogure et al. [85] | n = 179 Female (n = 111) Age (years): 59.8 (13.1) | Low back pain (Chronic) | Mobilization (n = 90) Placebo group (n = 89) 1 session per month for 6 months | Sacroiliac mobilizations consisting of upward gliding, downward gliding, superior distraction, and inferior distraction were performed for 15–20 min. | The therapist simulated the treatment, giving a light force on the joint, but did not actually produce movement. | VAS | Preintervention first session Preintervention second session Preintervention third session Preintervention fourth session Preintervention fifth session Preintervention sixth session |
| Krekoukias et al. [86] | n = 75 Female (n = 33) Age (years): 47.51 (SD Not available) | Low back pain | Mobilization (n = 25) Placebo group (n = 25) Conventional physiotherapy (Stretching exercises, TENS, and massage) (n = 25) 1 session per week for 5 weeks | Spinal lumbar mobilization for 10 min: passive accessory intervertebral movements and passive physiological intervertebral movements. | The therapist mimicked the grip and the procedure of mobilization without performing any force. | NPRS | Preintervention Post-intervention |
| Silva et al. [87] | n = 38 Female (n = 22) Age (years): 40.8 (2.0) | Ankle injury (subacute or chronic) | Mobilization (n = 19) Placebo group (n = 19) 3 sessions per week for 2 weeks | Cyclic and rhythmic mobilizations were applied to the talus in the anteroposterior direction. | The therapist simulated the treatment, giving a light force on the joint, but did not actually produce movement. | VAS | Preintervention Post-intervention first session Post-intervention sixth session 2 weeks post-intervention |
| La Touche et al. [88] | n = 32 Female (n = 21) Age (years): 33.87 (SD Not available) | Cervico-craniofacial pain | Mobilization (n = 16) Placebo group (n = 16) 3 sessions in 2 weeks | Mobilization for 6 min on the 3 upper cervical segments. | The therapist mimicked the grip and the procedure of mobilization without performing any force. | VAS | Preintervention first session Post-intervention first session Preintervention second session Post-intervention second session Preintervention third session Post-intervention third session |
| Pecos-Martin et al. [89] | n = 34 Female (n = 19) Age (years): 24 (3) | Thoracic spine pain | Mobilization (n = 17) Placebo group (n = 17) 1 session | Mobilization on T7 was performed using a pisiform grip three times for 1 min, with a 20-s rest between sets. | The therapist simulated the treatment, giving a light force on the joint, but did not actually produce movement. | NRS | Preintervention Post-intervention |
| Surenkok et al. [90] | n = 39 Female (n = 22) Age (years): 54.30 (14.16) | Shoulder pain | Mobilization (n = 13) Placebo group (n = 13) Control group: no treatment (n = 13) 1 session | Sets of 10 repetitions of scapular mobilization were performed with a rest interval of 30 s between sets. | The therapist simulated the treatment, giving a light force on the joint, but did not actually produce movement. | VAS | Preintervention Post-intervention |
| Neural Techniques | |||||||
| Author/Year | Participants | Pathology | Groups | Intervention Group Description | Placebo Group Description | Pain Outcome | Measurements |
| Ferragut-Garcías et al. [78] | n = 97 Female (n = 78) Age (years): 39.4 (SD Not available) | Tension-type headache | Soft tissue technique (n = 23) Neural technique (n = 25) Combined treatment intervention (n = 25) Placebo group (n = 24) 2 sessions in the first 2 weeks and 1 session per week in the next 2 weeks | Neural techniques consisted of mobilization in craniocervical flexion, lateral cervical sliding, and opening the mouth in craniocervical flexion. The protocol lasted 15 min (5 min per technique). | Superficial massage was performed for 15 min using ultrasound gel to minimize skin stimulation. | VAS | Preintervention Post-intervention 15 days post-intervention 30 days post-intervention |
| Bialosky et al. [91] | n = 40 Female (n = 40) Age (years): 46.90 (10.25) | Carpal tunnel syndrome | Neural technique (n = 20) Placebo group (n = 20) 2 sessions per week for 3 weeks | A neurodynamic technique for the median nerve was applied. 5 sets of 10 cycles for the first 3 sessions and 7 sets of 10 cycles for sessions 4 through 6. | Sham group received a sham technique that minimized anatomical stress across the median nerve. | VAS | Preintervention first session Post-intervention first session Preintervention sixth session Post-intervention sixth session |
| Wolny and Linek [92] | n = 150 Female (n = 135) Age (years): 53.2 (SD Not available) | Carpal tunnel syndrome | Neural technique (n = 78) Placebo group (n = 72) 2 sessions per week for 10 weeks | Neurodynamic technique for the median nerve was performed in standard protocol consisting of 3 series of 60 repetitions of glide and tension neurodynamic techniques separated by inter-series intervals of 15 s. | Sham group received a sham technique that minimized anatomical stress across the median nerve. | NPRS | Preintervention Post-intervention |
| Fernández-Carnero et al. [93] | n = 54 Female (n = 41) Age (years): 20.91 (2.64) | Neck pain (Chronic) | Neural technique (n = 27) Placebo group (n = 27) 1 session | Neurodynamic technique for the median nerve was applied. Treatment duration was seven min (frequency of 0.5 Hz) for two min and repeated three times with 30 s of rest time between each mobilization. | Sham group received a sham technique that minimized anatomical stress across the median nerve. | VAS | Preintervention Post-intervention |
3.2.4. Outcomes
3.2.5. Risk of Bias’ Blinding Assessment
3.2.6. Quantitative Analysis
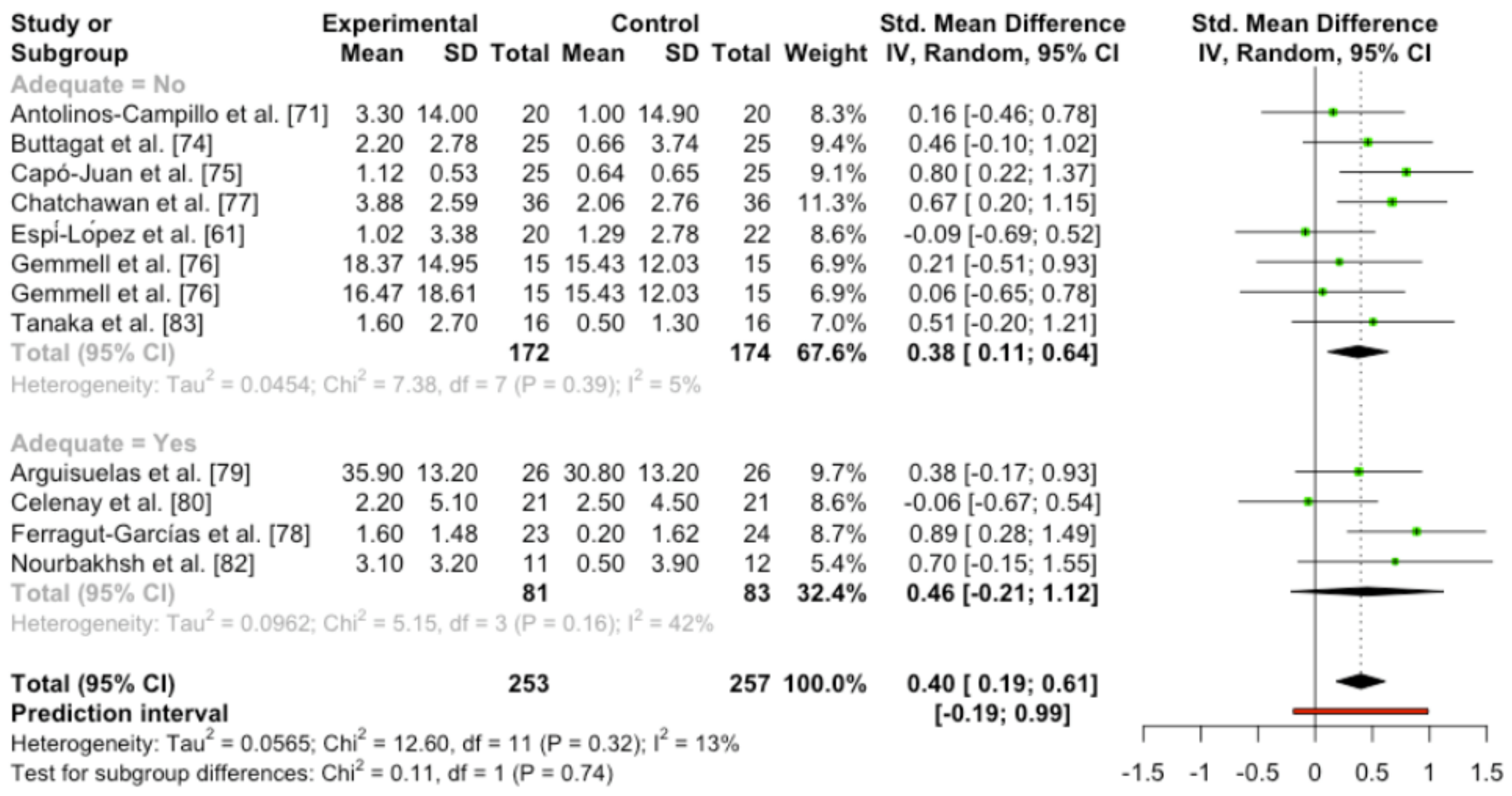
3.3. Trials Using Soft Tissue Techniques
3.3.1. Participants
3.3.2. Intervention Groups
3.3.3. Placebo
3.3.4. Outcomes
3.3.5. Risk of Bias Blinding Assessment
3.3.6. Quantitative Analysis
3.4. Trials Using Mobilization Techniques
3.4.1. Participants
3.4.2. Intervention Groups
3.4.3. Placebo
3.4.4. Outcomes
3.4.5. Risk of Bias’ Blinding Assessment
3.4.6. Quantitative Analysis
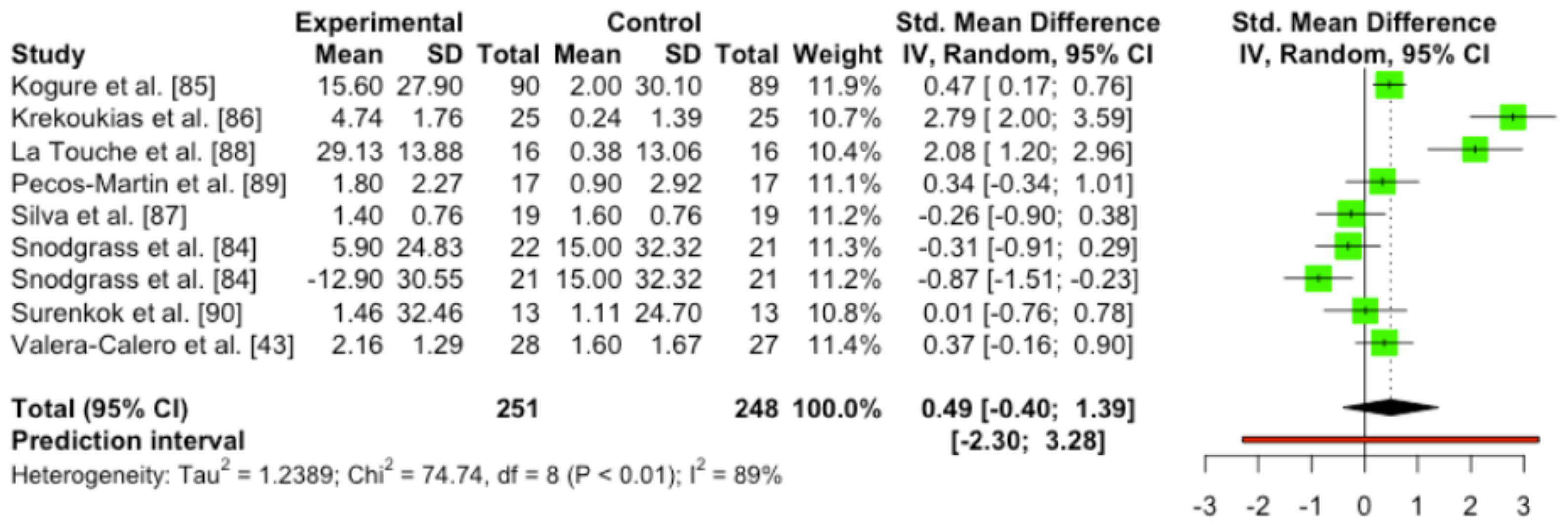
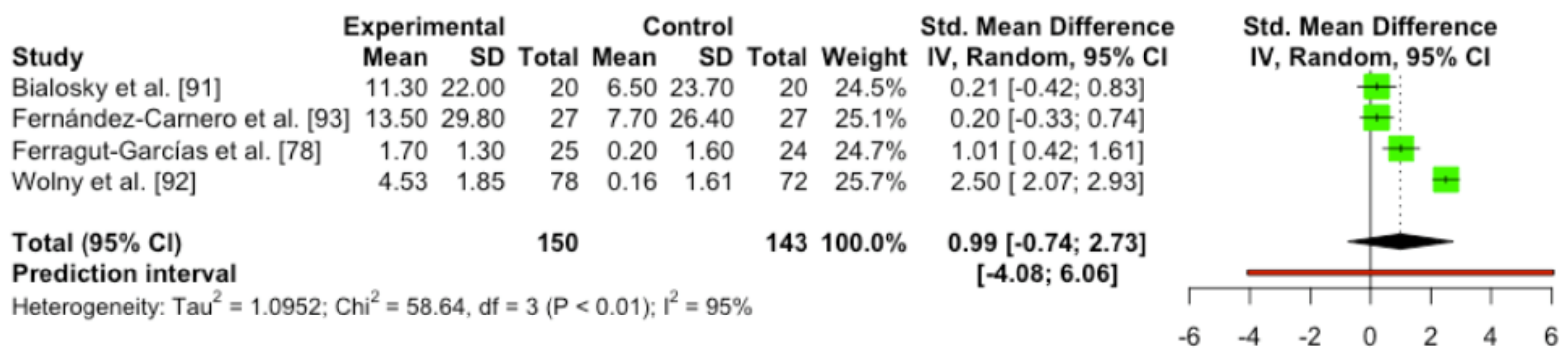
3.5. Trials Using Neurodynamic Techniques
3.5.1. Participants
3.5.2. Intervention Groups
3.5.3. Placebo
3.5.4. Outcomes
3.5.5. Risk of Bias’ Blinding Assessment
3.5.6. Quantitative Analysis
3.6. Quality of Evidence
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mills, S.E.E.; Nicolson, K.P.; Smith, B.H. Chronic pain: A review of its epidemiology and associated factors in population-based studies. Br. J. Anaesth. 2019, 123, e273–e283. [Google Scholar] [CrossRef]
- IsHak, W.W.; Wen, R.Y.; Naghdechi, L.; Vanle, B.; Dang, J.; Knosp, M.; Dascal, J.; Marcia, L.; Gohar, Y.; Eskander, L. Pain and Depression: A Systematic Review. Harv. Rev. Psychiatry 2018, 26, 352–363. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, Z.; Zajacova, A.; Grol-Prokopczyk, H. Trends in Pain Prevalence among Adults Aged 50 and Older across Europe, 2004 to 2015. J. Aging Health 2020, 32, 1419–1432. [Google Scholar] [CrossRef]
- Breivik, H.; Collett, B.; Ventafridda, V.; Cohen, R.; Gallacher, D. Survey of chronic pain in Europe: Prevalence, impact on daily life, and treatment. Eur. J. Pain 2006, 10, 287. [Google Scholar] [CrossRef] [PubMed]
- Saxen, M.; Rosenquist, R.W. The Epidemiology and Economic Impact of Spine Pain. In Spine Pain Care, A Comprehensive Clinical Guide; Springer: Berlin/Heidelberg, Germany, 2020; pp. 7–11. [Google Scholar] [CrossRef]
- Korr, I.M. The Neurobiologic Mechanisms in Manipulative Therapy. In The Neurobiologic Mechanisms in Manipulative Therapy; Springer: Berlin/Heidelberg, Germany, 1978. [Google Scholar] [CrossRef]
- Montañez-Aguilera, F.J.; Valtueña-Gimeno, N.; Pecos-Martín, D.; Arnau-Masanet, R.; Barrios-Pitarque, C.; Bosch-Morell, F. Changes in a patient with neck pain after application of ischemic compression as a trigger point therapy. J. Back Musculoskelet. Rehabil. 2010, 23, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Montañez-Aguilera, F.J.; Pecos-Martín, D.; Arnau-Masanet, R.; Camps-Botella, A.; Borja-Soler, L.; Bosh-Morell, F. Immediate Effect of Ultrasound and Ischemic Compression Techniques for the Treatment of Trapezius Latent Myofascial Trigger Points in Healthy Subjects: A Randomized Controlled Study. J. Manip. Physiol. Ther. 2009, 32, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Uemoto, L.; Nascimento-de-Azevedo, R.; Almedia-Alfaya, T.; Nunes-Jardim-Reis, R.; Depes-de-Gouvêa, C.V.; Cavalcanti-Garcia, M.A. Myofascial trigger point therapy: Laser therapy and dry needling. Curr. Pain Headache Rep. 2003, 17, 357. [Google Scholar] [CrossRef] [PubMed]
- Hall, T.M.; Elvey, R.L. Nerve trunk pain: Physical diagnosis and treatment. Man. Ther. 1999, 4, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Elvey, R.L. Physical evaluation of the peripheral nervous system in disorders of pain and dysfunction. J. Hand Ther. 1997, 10, 122–129. [Google Scholar] [CrossRef]
- Bokarius, A.V.; Bokarius, V. Evidence-Based Review of Manual Therapy Efficacy in Treatment of Chronic Musculoskeletal Pain. Pain Pract. 2010, 10, 451–458. [Google Scholar] [CrossRef]
- Hengeveld, E.; Banks, K. Maitland’s Vertebral Manipulation: Management of Neuromusculoskeletal Disorders; Elsevier Health Sciences: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Walser, R.F.; Meserve, B.B.; Boucher, T.R. The effectiveness of thoracic spine manipulation for the management of musculoskeletal conditions: A systematic review and meta-analysis of randomized clinical trials. J. Man. Manip. Ther. 2009, 17, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, C.; Machado-Caetano, J.; Cunha, L.; Rebouta, P.; Kaptchuk, T.J.; Kirsch, I. Open-Label placebo treatment in chronic low back pain: A randomized controlled trial. Pain 2016, 157, 2766–2772. [Google Scholar] [CrossRef] [PubMed]
- Testa, M.; Rossettini, G. Enhance placebo, avoid nocebo: How contextual factors affect physiotherapy outcomes. Man. Ther. 2016, 24, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Bizzarri, P.; Buzzatti, L.; Cattrysse, E.; Scafoglieri, A. Thoracic manual therapy is not more effective than placebo thoracic manual therapy in patients with shoulder dysfunctions: A systematic review with meta-analysis. Musculoskelet. Sci. Pract. 2018, 33, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Stewart-Williams, S.; Podd, J. The Placebo Effect: Dissolving the Expectancy Versus Conditioning Debate. Psychol. Bull. 2004, 130, 324–340. [Google Scholar] [CrossRef]
- Colloca, L.; Klinger, R.; Flor, H.; Bingel, U. Placebo analgesia: Psychological and neurobiological mechanisms. Pain 2013, 154, 511–514. [Google Scholar] [CrossRef]
- Vase, L.; Wartolowska, K. Pain, placebo, and test of treatment efficacy: A narrative review. Br. J. Anaesth. 2019, 123, e254–e262. [Google Scholar] [CrossRef]
- Wang, V.C.; Mullally, W.J. Pain Neurology. Am. J.Med. 2020, 133, 273–280. [Google Scholar] [CrossRef]
- Wan, M.; Orlu-Gul, M.; Legay, H.; Tuleu, C. Blinding in pharmacological trials: The devil is in the details. Arch. Dis. Child. 2013, 98, 656–659. [Google Scholar] [CrossRef]
- Druart, L.; Graham-Lomgsworth, S.E.; Rolland, C.; Dolgopoloff, M.; Terrisse, H.; Bosson, J.L.; Pinsault, N. Can an Open-Label Placebo Be as Effective as a Deceptive Placebo? Methodological Considerations of a Study Protocol. Medicines 2020, 7, 3. [Google Scholar] [CrossRef]
- Vernon, H.; Puhl, A.; Reinhart, C. Systematic review of clinical trials of cervical manipulation: Control group procedures and pain outcomes. Chiropr. Man. Ther. 2011, 19, 3. [Google Scholar] [CrossRef] [PubMed]
- Hancock, M.J.; Maher, C.G.; Latimer, J.; McAuley, J.H. Selecting an appropriate placebo for a trial of spinal manipulative therapy. Aust. J. Physiother. 2006, 52, 135–138. [Google Scholar] [CrossRef]
- Puhl, A.A.; Reinhart, C.J.; Doan, J.B.; Vernon, H. The quality of placebos used in randomized, controlled trials of lumbar and pelvic joint thrust manipulation—A systematic review. Spine J. 2017, 17, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. Cochrane Handbook for Systematic Reviews of Interventions, 2nd ed.; John Wiley Sons: Hoboken, NJ, USA, 2019. [Google Scholar] [CrossRef]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Steward, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savocic, J.; Shulz, K.F.; Weeks, L.; Sterne, J.A.C.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 314, d5928. [Google Scholar] [CrossRef]
- Braithwaite, F.A.; Walters, J.L.; Li, L.S.K.; Moseley, G.L.; Williams, M.T.; McEvoy, M.P. Effectiveness and adequacy of blinding in the moderation of pain outcomes: Systematic review and meta-analyses of dry needling trials. PeerJ 2018, 6, e5318. [Google Scholar] [CrossRef]
- McHugh, M.L. Interrater reliability: The kappa statistic. Biochem. Med. 2012, 22, 276. [Google Scholar] [CrossRef]
- Viechtbauer, W. Conducting meta-analyses in R with the metafor. J. Stat. Softw. 2010, 36, 1–48. [Google Scholar] [CrossRef]
- Harrer, M.; Cuijpers, P.; Furukawa, T.A.; Ebert, D. Doing Meta-Analysis with R: A Hands-On Guide; Chapmann & Hall/CRC: Boca Raton, FL, USA, 2021. [Google Scholar]
- Kinney, A.R.; Eakman, A.M.; Graham, J.E. Novel Effect Size Interpretation Guidelines and an Evaluation of Statistical Power in Rehabilitation Research. Arch. Phys. Med. Rehabil. 2020, 12, 2219–2226. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Andrews, J.C.; Schunemann, H.J.; Oxman, A.D.; Pottie, K.; Meerppohl, J.J.; Coello, P.A.; Rind, D.; Montori, V.M.; Brito, J.P.; Norris, S.; et al. GRADE guidelines: 15. Going from evidence to recommendation-determinants of a recommendation’s direction and strength. J. Clin. Epidemiol. 2013, 66, 726–735. [Google Scholar] [CrossRef] [PubMed]
- García-Pérez-Juana, D.; Fernández-de-las-Peñas, C.; Arias-buría, J.L.; Cleland, J.A.; Plaza-Manzano, G.; Ortega-Santiago, R. Changes in Cervicocephalic Kinesthetic Sensibility, Widespread Pressure Pain Sensitivity, and Neck Pain After Cervical Thrust Manipulation in Patients With Chronic Mechanical Neck Pain: A Randomized Clinical Trial. J. Manip. Physiol. Ther. 2018, 41, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Haas, M.; Groupp, E.; Panzer, D.; Partna, L.; Lumsden, S.; Aickin, M. Efficacy of cervical endplay assessment as an indicator for spinal manipulation. Spine 2003, 28, 1091–1096. [Google Scholar] [CrossRef]
- Pikula, J.R. The effect of spinal manipulative therapy (SMT) on pain reduction and range of motion in patients with acute unilateral neck pain: A pilot study. J. Can. Chiropr. Assoc. 1999, 43, 111–119. [Google Scholar]
- Pires, P.F.; Packer, A.C.; Dibai-Filho, A.V.; Rodrigues-Bigaton, D. Immediate and Short-Term Effects of Upper Thoracic Manipulation on Myoelectric Activity of Sternocleidomastoid Muscles in Young Women With Chronic Neck Pain: A Randomized Blind Clinical Trial. J. Manip. Physiol. Ther. 2015, 38, 555–563. [Google Scholar] [CrossRef]
- Sillevis, R.; Cleland, J.; Hellman, M.; Beekhuizen, K. Immediate effects of a thoracic spine thrust manipulation on the autonomic nervous system: A randomized clinical trial. J. Man. Manip. Ther. 2010, 18, 181–190. [Google Scholar] [CrossRef]
- Valera-Calero, A.; Lluch-Girbés, E.; Gallego-Izquierdo, T.; Malfiet, A.; Pecos-Martín, D. Endocrine response after cervical manipulation and mobilization in people with chronic mechanical neck pain: A Randomized controlled trial. Eur. J. Phys. Rehabil. Med. 2019, 55, 792–805. [Google Scholar] [CrossRef]
- Vernon, H.; Triano, J.; Ross, K.; Tran, S.K.; Soave, S.K.; Dinulos, M.D. Validation of a novel sham cervical manipulation procedure. Clin. Chiropr. 2011, 14, 177. [Google Scholar] [CrossRef][Green Version]
- Weber, K.A.; Wager, T.D.; Mackey, S.; Elliott, J.M.; Liu, W.C.; Sparks, C.L. Evidence for decreased Neurologic Pain Signature activation following thoracic spinal manipulation in healthy volunteers and participants with neck pain. NeuroImage Clin. 2019, 24, 102042. [Google Scholar] [CrossRef]
- Bialosky, J.E.; George, S.Z.; Horn, M.E.; Price, D.D.; Staud, R.; Robinson, M.E. Spinal manipulative therapy-specific changes in pain sensitivity in individuals with low back pain (NCT01168999). J. Pain. 2014, 15, 136–148. [Google Scholar] [CrossRef]
- Bond, B.M.; Kinslow, C.D.; Yoder, A.W.; Liu, W. Effect of spinal manipulative therapy on mechanical pain sensitivity in patients with chronic nonspecific low back pain: A pilot randomized, controlled trial. J. Man. Manip. Ther. 2020, 28, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Didehdar, D.; Kamali, F.; Yoosefinejad, A.K.; Lofti, M. The effect of spinal manipulation on brain neurometabolites in chronic nonspecific low back pain patients: A randomized clinical trial. Ir. J. Med. Sci. 2020, 189, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Elleuch, M.H.; Ghroubi, S. Vertebral manipulation in chronic low back pain: A prospective randomised study of 85 cases. Int. Musculoskelet. Med. 2009, 31, 57–62. [Google Scholar] [CrossRef]
- Kawchuk, G.N.; Haugen, R.; Fritz, J.A. True Blind for Subjects Who Receive Spinal Manipulation Therapy. Arch. Phys. Med. Rehabil. 2009, 90, 366–368. [Google Scholar] [CrossRef] [PubMed]
- Senna, M.K.; MacHaly, S.A. Does maintained spinal manipulation therapy for chronic nonspecific low back pain result in better long-term outcome? Spine 2011, 36, 1427–1437. [Google Scholar] [CrossRef]
- Vieira-Pellenz, F.; Oliva-Pascual-Vaca, A.; Rodríguez-Blanco, C.; Heredia-Rizo, A.M.; Ricard, F.; Almazán-Camps, G. Short-term effect of spinal manipulation on pain perception, spinal mobility, and full height recovery in male subjects with degenerative disk disease: A randomized controlled trial. Arch. Phys. Med. Rehabil. 2014, 95, 1613–1619. [Google Scholar] [CrossRef]
- Atkinson, M.; Mathews, R.; Brantingham, J.W.; Globe, G.; Cassa, T.; Bonnefin, D.; Korporaal, C. A Randomized Controlled Trial to Assess the Efficacy of Shoulder Manipulation vs. Placebo in the Treatment of Shoulder Pain Due to Rotator Cuff Tendinopathy. J. Am. Chiropr. Assoc. 2008, 45, 11–26. [Google Scholar]
- Conte-da-Silva, A.; Moraes-Santos, G.; de-Godoy-Marques, C.M.; Brum-Marques, J.L. Immediate effects of spinal manipulation on shoulder motion range and pain in individuals with shoulder pain: A randomized trial. J. Chiropr. Med. 2019, 18, 19–26. [Google Scholar] [CrossRef]
- Haik, M.N.; Albuquerque-Sendín, F.; Silva, C.Z.; Siqueira-Junior, A.L.; Ribeiro, I.L.; Camargo, P.R. Scapular kinematics pre- And post- Thoracic thrust manipulation in individuals with and without shoulder impingement symptoms: A randomized controlled study. J. Orthop. Sports Phys. Ther. 2014, 44, 475–487. [Google Scholar] [CrossRef]
- Haik, M.N.; Alburquerque-Sendín, F.; Camargo, P.R. Short-Term Effects of Thoracic Spine Manipulation on Shoulder Impingement Syndrome: A Randomized Controlled Trial. Arch. Phys. Med. Rehabil. 2017, 98, 1594–1605. [Google Scholar] [CrossRef] [PubMed]
- Kardouni, J.R.; Pidcoe, P.E.; Shaffer, S.W.; Finucane, S.D.; Cheatham, S.A.; Sousa, C.O.; Michener, L.A. Thoracic Spine Manipulation in Individuals with Subacromial Impingement Syndrome Does Not Immediately Alter Thoracic Spine Kinematics, Thoracic Excursion, or Scapular Kinematics: A Randomized Controlled Trial. J. Orthop. Sport. Phys. Ther. 2015, 45, 527–538. [Google Scholar] [CrossRef] [PubMed]
- Kardouni, J.R.; Shaffer, S.W.; Pidcoe, P.E.; Finucane, S.D.; Cheatham, S.A.; Michener, L.A. Immediate changes in pressure pain sensitivity after thoracic spinal manipulative therapy in patients with subacromial impingement syndrome: A randomized controlled study. Man. Ther. 2015, 20, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Borusiak, P.; Biedermann, H.; Boßerhoff, S.; Opp, J. Lack of efficacy of manual therapy in children and adolescents with suspected cervicogenic headache: Results of a prospective, randomized, placebo-controlled, and blinded trial: Research submission. Headache 2010, 50, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Chaibi, A.; Benth, J.; Tuchin, P.J.; Russell, M.B. Chiropractic spinal manipulative therapy for migraine: A three-armed, single-blinded, placebo, randomized controlled trial. Eur. J. Neurol. 2017, 24, 143–153. [Google Scholar] [CrossRef]
- Espí-López, G.V.; Gómez-Conesa, A.; Arnal-Gómez, A.; Benítez-Martínez, J.; Oliva-Pascual-Vaca, A.; Rodríguez-Blanco, C. Treatment of tension-type headache with articulatory and suboccipital soft tissue therapy: A double-blind, randomized, placebo-controlled clinical trial. J. Bodyw. Mov. Ther. 2014, 18, 576–585. [Google Scholar] [CrossRef]
- Hondras, M.A.; Long, C.R.; Brennan, P.C. Spinal manipulative therapy versus a low force mimic maneuver for women with primary dysmenorrhea: A randomized, observer-blinded, clinical trial. Pain 1999, 81, 105–114. [Google Scholar] [CrossRef]
- Molins-Cubero, S.; Rodríguez-Blanco, C.; Oliva-Pascual-Vaca, A.; Heredia-Rizo, A.M.; Boscá-Gandía, J.J.; Ricard, F. Changes in pain perception after pelvis manipulation in women with primary dysmenorrea: A randomized controlled trial. Pain Med. 2014, 15, 1455–1463. [Google Scholar] [CrossRef] [PubMed]
- Lehtola, V.; Korhonen, I.; Airaksinen, O. A randomised, placebo-controlled, clinical trial for the short-term effectiveness of manipulative therapy and acupuncture on pain caused by mechanical thoracic spine dysfunction. Int. Musculoskelet. Med. 2010, 32, 25–32. [Google Scholar] [CrossRef]
- Motealleh, A.; Gheysari, E.; Shokri, E.; Sobhani, S. The immediate effect of lumbopelvic manipulation on EMG of vasti and gluteus medius in athletes with patellofemoral pain syndrome: A randomized controlled trial. Man. Ther. 2016, 22, 16–21. [Google Scholar] [CrossRef]
- Packer, A.C.; Pires, P.F.; Dibai-Filho, A.V.; Rodriguez-Bigaton, D. Effects of upper thoracic manipulation on pressure pain sensitivity in women with temporomandibular disorder: A randomized, double-blind, clinical trial. Am. J. Phys. Med. Rehabil. 2014, 93, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Young, I.A.; Pozzi, F.; Dunning, J.; Linkonis, R.; Michener, L.A. Immediate and short-term effects of thoracic spine manipulation in patients with cervical radiculopathy: A randomized controlled trial. J. Orthop. Sports Phys. Ther. 2019, 49, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Cleland, J.A.; Childs, M.J.D.; McRae, M.; Palmer, J.A.; Stowell, T. Immediate effects of thoracic manipulation in patients with neck pain: A randomized clinical trial. Man. Ther. 2005, 10, 127–135. [Google Scholar] [CrossRef]
- Michener, L.A.; Kardouni, J.R.; Sousa, C.; Ely, J.M. Validation of a sham comparator for thoracic spinal manipulation in patients with shoulder pain. Man. Ther. 2015, 20, 171–175. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Vernon, H.; MacAdam, K.; Marshall, V.; Pion, M.; Sadowska, M. Validation of a sham manipulative procedure for the cervical spine for use in clinical trials. J. Manip. Physiol. Ther. 2005, 28, 662–666. [Google Scholar] [CrossRef] [PubMed]
- Antolinos-Campillo, P.J.; Oliva-Pascua-Vaca, A.; Rodríguez-Blanco, C.; Heredia-Rizo, A.M.; Espí-López, G.V.; Ricard, F. Short-term changes in median nerve neural tension after a suboccipital muscle inhibition technique in subjects with cervical whiplash: A randomised controlled trial. Physiotherapy 2014, 100, 249–255. [Google Scholar] [CrossRef]
- Blikstad, A.; Gemmell, H. Immediate effect of activator trigger point therapy and myofascial band therapy on non-specific neck pain in patients with upper trapezius trigger points compared to sham ultrasound: A randomised controlled trial. Clin. Chiropr. 2008, 11, 23–29. [Google Scholar] [CrossRef]
- Briem, K.; Huijbregts, P.; Thorsteinsdottir, M. Immediate effects of inhibitive distraction on active range of cervical flexion in patients with neck pain: A pilot study. J. Man. Manip. Ther. 2007, 15, 82–92. [Google Scholar] [CrossRef]
- Buttagat, V.; Narktro, T.; Onsrira, K.; Pobsamai, C. Short-term effects of traditional Thai massage on electromyogram, muscle tension and pain among patients with upper back pain associated with myofascial trigger points. Complement. Ther. Med. 2016, 28, 8–12. [Google Scholar] [CrossRef]
- Capó-Juan, M.A.; Grávalos-Gasull, A.; Bennasar-Veny, M.; Arguiló-Pons, A.; Gamundi-Gamundi, A.; de-Pedro-Gómez, J.E. Short term effectiveness of Pressure Release and Kinesiotaping in Cervical Myofascial Pain caused by sternocleidomastoid muscle: A randomized clinical trial. Fisioterapia 2017, 39, 68–74. [Google Scholar] [CrossRef]
- Gemmell, H.; Miller, P.; Nordstrom, H. Immediate effect of ischaemic compression and trigger point pressure release on neck pain and upper trapezius trigger points: A randomised controlled trial. Clin. Chiropr. 2008, 11, 30–36. [Google Scholar] [CrossRef]
- Chatchawan, U.; Eungpinichpong, W.; Sooktho, S.; Tiamkao, S.; Yamauchi, J. Effects of thai traditional massage on pressure pain threshold and headache intensity in patients with chronic tension-type and migraine headaches. J. Altern. Complement. Med. 2014, 20, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Ferragut-Garcías, A.; Plaza-Manzano, G.; Rodríguez-Blanco, C.; Velasco-Roldán, O.; Pecos-Martín, D.; Oliva-Pascual-Vaca, C.; Llabrés-Bennasar, B.; Oliva-Pascual-Vaca, J. Effectiveness of a Treatment Involving Soft Tissue Techniques and/or Neural Mobilization Techniques in the Management of Tension-Type Headache: A Randomized Controlled Trial. Arch. Phys. Med. Rehabil. 2017, 98, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Arguisuelas, M.D.; Lisón, J.F.; Sánchez-Zuriaga, D.; Martínez-Hurtado, I.; Doménech-Fernández, J. Effects of Myofascial Release in Non-specific Chronic Low Back Pain: A Randomized Clinical Trial. Spine 2017, 42, 627–634. [Google Scholar] [CrossRef] [PubMed]
- Celenay, S.T.; Kaya, D.O.; Ucurum, S.G. Adding connective tissue manipulation to physiotherapy for chronic low back pain improves pain, mobility, and well-being: A randomized controlled trial. J. Exerc. Rehabil. 2019, 15, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Field, T.M.; Sunshine, W.; Hernandezreif, M.; Quintino, O.; Shanberg, S.; Kuhn, C.; Burman, I. Massage therapy effects on depression and somatic symptoms in chronic fatigue syndrome. J. Chronic Fatigue Syndr. 1997, 3, 43–51. [Google Scholar] [CrossRef]
- Nourbakhsh, M.R.; Fearon, F.J. The Effect of Oscillating-energy Manual Therapy on Lateral Epicondylitis: A Randomized, Placebo-control, Double-blinded Study. J. Hand Ther. 2008, 21, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, R.; Umehara, T.; Kawabata, Y.; Sakuda, T. Effect of Continuous Compression Stimulation on Pressure-Pain Threshold and Muscle Spasms in Older Adults With Knee Osteoarthritis: A Randomized Trial. J. Manip. Physiol. Ther. 2018, 41, 315–322. [Google Scholar] [CrossRef]
- Snodgrass, S.J.; Rivett, D.A.; Sterling, M.; Vicenzino, B. Dose optimization for spinal treatment effectiveness: A randomized controlled trial investigating the effects of high and low mobilization forces in patients with neck pain. J. Orthop. Sports Phys. Ther. 2014, 44, 141–152. [Google Scholar] [CrossRef]
- Kogure, A.; Kotani, K.; Katada, S.; Takagi, H.; Kamikozuru, M.; Isaji, T.; Hakata, S. A randomized, single-blind, placebo-controlled study on the efficacy of the arthrokinematic approach-hakata method in patients with chronic nonspecific low back pain. PLoS ONE 2015, 10, e0144325. [Google Scholar] [CrossRef] [PubMed]
- Krekoukias, G.; Gelalis, I.D.; Xenakis, T.; Gioftsos, G.; Dimitriadis, Z.; Sakellari, V. Spinal mobilization vs conventional physiotherapy in the management of chronic low back pain due to spinal disk degeneration: A randomized controlled trial. J. Man. Manip. Ther. 2017, 25, 66–73. [Google Scholar] [CrossRef]
- Silva, R.D.; Teixeira, L.M.; Moreira, T.S.; Teixeira-Salmela, L.F.; de-Resende, M.A. Effects of Anteroposterior Talus Mobilization on Range of Motion, Pain, and Functional Capacity in Participants With Subacute and Chronic Ankle Injuries: A Controlled Trial. J. Manip. Physiol. Ther. 2017, 40, 273–283. [Google Scholar] [CrossRef] [PubMed]
- La-Touche, R.; París-Alemany, A.; Mannheimer, J.S.; Angulo-Díaz-Parreño, S.; Bishop, M.D.; López-Valverde-Centeno, A.; von-Piekartz, H.; Fernández-Carnero, J. Does Mobilization of the Upper Cervical Spine Affect Pain Sensitivity and Autonomic Nervous System Function in Patients With Cervico-craniofacial Pain?: A randomized-controlled trial. Clin. J. Pain 2013, 29, 205–215. [Google Scholar] [CrossRef]
- Pecos-Martín, D.; de-Melo-Aroeira, A.E.; Verás-Silva, R.L.; Martínez-de-Tejada-Pozo, G.; Rodríguez-Solano, L.M.; Plaza-Manzano, G.; Gallego-Izquierdo, T.; Falla, D. Immediate effects of thoracic spinal mobilisation on erector spinae muscle activity and pain in patients with thoracic spine pain: A preliminary randomised controlled trial. Physiotherapy 2017, 103, 90–97. [Google Scholar] [CrossRef]
- Surenkok, O.; Aytar, A.; Baltaci, G. Acute effects of scapular mobilization in shoulder dysfunction: A double-blind randomized placebo-controlled trial. J. Sport Rehabil. 2009, 18, 493–501. [Google Scholar] [CrossRef]
- Bialosky, J.E.; Bishop, M.D.; Price, D.D.; Robinson, M.E.; Vincent, K.R.; George, S.Z. A randomized sham-controlled trial of a neurodynamic technique in the treatment of carpal tunnel syndrome. J. Orthop. Sports Phys. Ther. 2009, 39, 709–723. [Google Scholar] [CrossRef]
- Wolny, T.; Linek, P. Neurodynamic Techniques Versus “Sham” Therapy in the Treatment of Carpal Tunnel Syndrome: A Randomized Placebo-Controlled Trial. Arch. Phys. Med. Rehabil. 2018, 99, 843–854. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Carnero, J.; Sierra-Silvestre, E.; Beltran-Alacreu, H.; Gil-Martínez, A.; La-Touche, R. Neural tension technique improves immediate conditioned pain modulation in patients with chronic neck pain: A randomized clinical trial. Pain Med. 2019, 20, 1227–1235. [Google Scholar] [CrossRef]
- Bialosky, J.E.; Bishop, M.D.; Penza, C. Placebo mechanisms of manual therapy: A sheep in wolf’s clothing? J. Orthop. Sports Phys. Ther. 2017, 47, 301–304. [Google Scholar] [CrossRef]
- Fergusson, D.; Glass, C.; Waring, D.; Shapiro, S. Turning a blind eye: The success of blinding reported in a random sample of randomised, placebo controlled trials. BMJ 2004, 328, 432. [Google Scholar] [CrossRef] [PubMed]
- Villamar, M.F.; Contreras, V.S.; Kuntz, R.E.; Fregni, F. The reporting of blinding in physical medicine and rehabilitation randomized controlled trials: A systematic review. J. Rehabil. Med. 2013, 45, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Cerritelli, F.; Verzella, M.; Cicchitti, L.; D’Alessandro, G.; Vanacore, N. The paradox of sham therapy and placebo effect in osteopathy A systematic review. Medicine 2016, 95, e4728. [Google Scholar] [CrossRef] [PubMed]
- Bang, H.; Ni, L.; Davis, C.E. Assessment of blinding in clinical trials. Control. Clin. Trials 2004, 25, 143–156. [Google Scholar] [CrossRef] [PubMed]
- Bang, H.; Flaherty, S.P.; Kolahi, J.; Park, J. Blinding assessment in clinical trials: A review of statistical methods and a proposal of blinding assessment protocol. Clin. Res. Regul. Aff. 2010, 27, 42–51. [Google Scholar] [CrossRef]
- Armijo-Olivo, S.; Fuentes, J.; da-Costa, B.R.; Saltaji, H.; Ha, C.; Cummings, G.G. Blinding in Physical Therapy Trials and Its Association with Treatment Effects: A Meta-epidemiological Study. Am. J. Phys. Med. Rehabil. 2017, 96, 34–44. [Google Scholar] [CrossRef]
- Chaibi, A.; Benth, J.Š.; Russell, M.B. Validation of placebo in a manual therapy randomized controlled trial. Sci. Rep. 2015, 5, 1–8. [Google Scholar] [CrossRef]
- Michener, L.A.; Kardouni, J.R.; Lopes-Albers, A.D.; Ely, J.M. Development of a sham comparator for thoracic spinal manipulative therapy for use with shoulder disorders. Man. Ther. 2013, 18, 60–64. [Google Scholar] [CrossRef]
- Lavazza, C.; Margherita, G.; Abenavoli, A.; Maggiani, A. Sham treatment effects in manual therapy trials on back pain patients: Systematic review and pairwise meta-analysis. BMJ Open 2021, 11, e045106. [Google Scholar] [CrossRef]
- Ruddock, J.K.; Sallis, H.; Ness, A.; Perry, R.E. Spinal Manipulation Vs Sham Manipulation for Nonspecific Low Back Pain: A Systematic Review and Meta-analysis. J. Chiropr. Med. 2016, 15, 165–183. [Google Scholar] [CrossRef]
- Galasso, A.; Urits, I.; An, D.; Nguyen, D.; Borchart, M.; Yazdi, C.; Manchikanti, L.; Kaye, R.J.; Kaye, A.L.; Mancuso, K.F.; et al. A comprehensive review of the treatment and management of Myofascial Pain Syndrome. Ther. Adv. Musculoskelet. Dis. 2020, 24, 43. [Google Scholar] [CrossRef]
- Conte-da-Silva, A.; de-Noronha, M.; Liberatori-Junior, R.M.; Aily, J.B.; Gonçalves, G.H.; Arrais-Lima, C.; de-Araújo-Vieira, L.M.S.M.; Mattiello, S.M. The Effectiveness of Ischemic Compression Technique on Pain and Function in Individuals With Shoulder Pain: A Systematic Review. J. Manip. Physiol. Ther. 2020, 43, 234–246. [Google Scholar] [CrossRef] [PubMed]
- Basson, A.; Oliver, B.; Ellis, R.; Coppieters, M.; Stewart, A.; Mudzi, W. The effectiveness of neural mobilization for neuromusculoskeletal conditions: A systematic review and meta-Analysis. J. Orthop. Sports Phys. Ther. 2017, 47, 593–615. [Google Scholar] [CrossRef]
- Neto, T.; Freitas, S.R.; Marques, M.; Gomes, L.; Andrade, R.; Oliveira, R. Effects of lower body quadrant neural mobilization in healthy and low back pain populations: A systematic review and meta-analysis. Musculoskelet. Sci. Pract. 2017, 27, 14–22. [Google Scholar] [CrossRef]
- Rubinstein, S.M.; de Zoete, A.; van Middelkoop, M.; Assendelft, W.J.J.; de-Boer, M.R.; van-Tulder, M.W. Benefits and harms of spinal manipulative therapy for the treatment of chronic low back pain: Systematic review and meta-analysis of randomised controlled trials. BMJ 2019, 364, l689. [Google Scholar] [CrossRef] [PubMed]


| Manipulation Tecniques | |||||||||||
| Author | Selection Bias | Performance Bias | Detection Bias | Attrition Bias | Reporting Bias | Other Bias | OVERALL | Adequated | |||
| Random Sequence Generation | Allocation Concealment | Participant Blinding | Research Personnel Blinding | Therapist Blinding | |||||||
| Atkinson et al. [53] | x | ? | x | ? | x | x | x | ✓ | x | x | No |
| Bialosky et al. [46] | ✓ | ✓ | ✓ | ? | x | x | ✓ | ✓ | ✓ | x | No |
| Bond et al. [47] | ✓ | ✓ | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Borusiak et al. [59] | ✓ | ✓ | ✓ | ✓ | x | ✓ | x | ✓ | ✓ | ✓ | Yes |
| Chaibi et al. [60] | ✓ | ? | ✓ | ? | x | ? | x | ✓ | ✓ | ? | No |
| Conte-da-Silva et al. [54] | ✓ | ✓ | ✓ | ✓ | x | x | ✓ | ✓ | ✓ | x | No |
| Didehdar et al. [48] | ✓ | ? | ✓ | ? | x | ✓ | x | ✓ | x | ✓ | Yes |
| Elleuch and Ghroubi [49] | ✓ | ? | ✓ | ? | x | ✓ | x | ✓ | x | ✓ | Yes |
| Espí-López et al. [61] | ✓ | ? | ? | ? | x | ? | ✓ | ✓ | ✓ | x | No |
| García-Perez-Juana et al. [38] | ✓ | ✓ | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Haas et al. [39] | ✓ | ✓ | ✓ | ✓ | ✓ | x | ✓ | ✓ | ✓ | x | No |
| Haik et al. [55] | ✓ | ? | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Haik et al. [56] | ✓ | ? | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Hondras et al. [62] | ✓ | ✓ | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Kardouni et al. [57] | ✓ | ✓ | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Kardouni et al. [58] | ✓ | ✓ | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Kawchuk et al. [50] | ? | ? | ✓ | ? | x | ✓ | ✓ | x | ✓ | ? | No |
| Lehtola et al. [64] | ✓ | ? | x | ? | x | ✓ | ✓ | ✓ | ✓ | x | No |
| Molins-Cubero et al. [63] | ✓ | ✓ | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Motealleh et al. [65] | ✓ | ? | ? | ? | x | ? | ✓ | ✓ | x | x | No |
| Packer et al. [66] | ? | ✓ | ✓ | ✓ | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Pikula [40] | ✓ | ? | x | ? | x | x | ✓ | ✓ | ✓ | x | No |
| Pires et al. [41] | ? | ✓ | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Senna and MacHaly [51] | ✓ | ✓ | ✓ | ? | x | ✓ | x | ✓ | x | ✓ | Yes |
| Sillevis et al. [42] | ✓ | ✓ | ? | ? | x | ? | ✓ | ✓ | ✓ | ? | No |
| Valera-Calero et al. [43] | ✓ | ✓ | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Vernon et al. [44] | ✓ | ✓ | ✓ | ? | x | ? | ✓ | ✓ | ✓ | ✓ | Yes |
| Vieira-Pellenz et al. [52] | ✓ | ? | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Weber et al. [45] | ✓ | ? | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Young et al. [67] | ✓ | ✓ | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Soft Tissue Techniques | |||||||||||
| Author | Selection Bias | Performance Bias | Detection Bias | Attrition Bias | Reporting Bias | Other Bias | Overall | Adequated | |||
| Random Sequence Generation | Allocation Concealment | Participant Blinding | Research Personnel Blinding | Therapist Blinding | |||||||
| Antolinos-Campillo et al. [71] | ✓ | ? | x | ✓ | x | ✓ | ✓ | ✓ | ✓ | ? | No |
| Arguisuelas et al. [79] | ✓ | ? | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Blikstad and Gemmell [72] | ✓ | ✓ | x | ? | x | ✓ | ✓ | ? | x | x | No |
| Briem et al. [73] | ✓ | ✓ | ✓ | ? | x | ✓ | ✓ | x | ✓ | ✓ | Yes |
| Buttagat et al. [74] | ✓ | ✓ | x | ✓ | x | ✓ | ✓ | ✓ | ✓ | x | No |
| Capó-Juan et al. [75] | ✓ | ? | ? | ✓ | x | ? | ✓ | ✓ | ✓ | x | No |
| Celenay et al. [80] | ✓ | ? | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ? | Yes |
| Chatchawan et al. [77] | ✓ | ✓ | x | ✓ | x | ✓ | ✓ | x | ✓ | x | No |
| Espí-López et al. [61] | ✓ | ? | ? | ? | x | ? | ✓ | ✓ | ✓ | x | No |
| Ferragut-Garcías et al. [78] | ✓ | ✓ | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Field et al. [81] | ✓ | ? | x | ✓ | x | ✓ | ? | ✓ | ✓ | x | No |
| Gemmell et al. [76] | ✓ | ✓ | x | ? | x | ✓ | ✓ | ✓ | ✓ | x | No |
| Nourbakhsh and Fearon [82] | ✓ | ? | ✓ | ? | x | ✓ | ? | ✓ | ✓ | ✓ | Yes |
| Tanaka et al. [83] | ✓ | x | ? | ? | x | ✓ | ✓ | ✓ | ✓ | x | No |
| Mobilization Techniques | |||||||||||
| Author | Selection Bias | Performance Bias | Detection Bias | Attrition Bias | Reporting Bias | Other Bias | Overall | Adequated | |||
| Random Sequence Generation | Allocation Concealment | Participant Blinding | Research Personnel Blinding | Therapist Blinding | |||||||
| Kogure et al. [85] | ✓ | ? | ✓ | x | x | x | ✓ | ✓ | ✓ | x | No |
| Krekoukias et al. [86] | ✓ | ? | ✓ | ? | x | x | ✓ | ✓ | ✓ | x | No |
| La Touche et al. [88] | ✓ | ? | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Pecos-Martin et al. [89] | ? | ✓ | ✓ | ? | x | ✓ | ? | ✓ | ✓ | ✓ | Yes |
| Silva et al. [87] | x | ? | ✓ | ✓ | x | x | ✓ | ✓ | ✓ | x | No |
| Snodgrass et al. [84] | ✓ | ✓ | x | ? | x | ✓ | ✓ | ✓ | ✓ | x | No |
| Surenkok et al. [90] | ? | ? | ? | ? | x | ✓ | ✓ | ✓ | ✓ | x | No |
| Valera-Calero et al. [43] | ✓ | ✓ | x | ? | x | ✓ | ✓ | ✓ | ✓ | x | No |
| Neural Techniques | |||||||||||
| Author | Selection Bias | Performance Bias | Detection Bias | Attrition Bias | Reporting Bias | Other Bias | Overall | Adequated | |||
| Random Sequence Generation | Allocation Concealment | Participant Blinding | Research Personnel Blinding | Therapist Blinding | |||||||
| Bialosky et al. [91] | ✓ | ✓ | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Fernández-Carnero et al. [93] | ✓ | ✓ | ✓ | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | Yes |
| Ferragut-Garcías et al. [78] | ✓ | ✓ | x | ? | x | ✓ | ✓ | ✓ | ✓ | ✓ | No |
| Wolny and Linek [92] | ✓ | ? | ✓ | ? | x | ✓ | x | ✓ | ✓ | ✓ | Yes |
| GRADE Assessment | |||||||
|---|---|---|---|---|---|---|---|
| Outcomes | Study Design | Risk of Bias −1 Serious −2 Very Serious | Inconsistency −1 Serious −2 Very Serious | Indirectness −1 Serious −2 Very Serious | Imprecision −1 Serious −2 Very Serious | Large Effect +1 Large +1 Very Large | Quality of Evidence |
| Manipulation techniques | RCT | −1 | −1 | 0 | 0 | 0 | Low |
| Soft tissue techniques | RCT | −2 | 0 | 0 | 0 | 0 | Low |
| Mobilization techniques | RCT | −2 | −2 | 0 | −1 | 0 | Very low |
| Neurodynamic techniques | RCT | −1 | −2 | 0 | −1 | 1 | Very low |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molina-Álvarez, M.; Arribas-Romano, A.; Rodríguez-Rivera, C.; García, M.M.; Fernández-Carnero, J.; Armijo-Olivo, S.; Goicoechea Garcia, C. Manual Therapy Effect in Placebo-Controlled Trials: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2022, 19, 14021. https://doi.org/10.3390/ijerph192114021
Molina-Álvarez M, Arribas-Romano A, Rodríguez-Rivera C, García MM, Fernández-Carnero J, Armijo-Olivo S, Goicoechea Garcia C. Manual Therapy Effect in Placebo-Controlled Trials: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health. 2022; 19(21):14021. https://doi.org/10.3390/ijerph192114021
Chicago/Turabian StyleMolina-Álvarez, Miguel, Alberto Arribas-Romano, Carmen Rodríguez-Rivera, Miguel M. García, Josué Fernández-Carnero, Susan Armijo-Olivo, and Carlos Goicoechea Garcia. 2022. "Manual Therapy Effect in Placebo-Controlled Trials: A Systematic Review and Meta-Analysis" International Journal of Environmental Research and Public Health 19, no. 21: 14021. https://doi.org/10.3390/ijerph192114021
APA StyleMolina-Álvarez, M., Arribas-Romano, A., Rodríguez-Rivera, C., García, M. M., Fernández-Carnero, J., Armijo-Olivo, S., & Goicoechea Garcia, C. (2022). Manual Therapy Effect in Placebo-Controlled Trials: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health, 19(21), 14021. https://doi.org/10.3390/ijerph192114021







