Impact of the New Coronavirus Infection on the Immune System of Children and Adolescents in the Region of the Russian Federation
Abstract
1. Introduction
2. Materials and Methods
3. Research Results and Discussion
3.1. Study of the Pandemic in Perm Krai of the Russian Federation Subsection
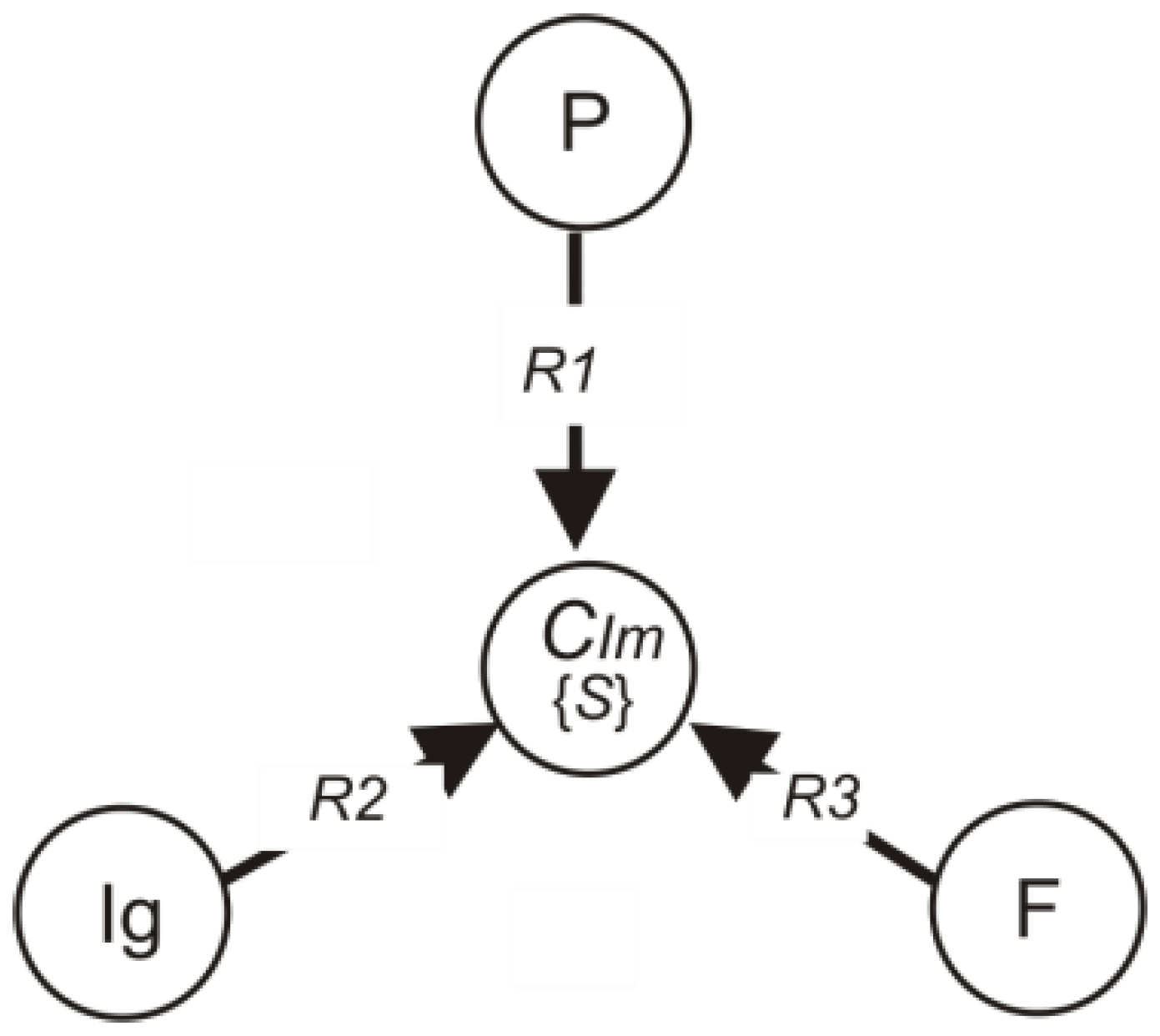
3.2. Development of the Immunogram Model
- P—the set of flow cytometry elements;
- F—the set of phagocytosis elements;
- Ig—the set of immunoglobulin elements;
- Im—the set of immunogram categories:
- k1—absence of disease (Immunoglobulins IgA, IgM, IgG are in the reference interval);
- k2—active stage of the disease (Immunoglobulins IgA, IgM, out of the reference interval);
- k3—passive stage of the disease (Immunoglobulins IgG and IgM, exceeded the reference interval);
- k4—patient’s recovery process (IgG immunoglobulin is out of the reference interval).
IgR20 [C2, Im0], C2 R21 Im1,
FR30 [C3, Im0], C3 R31 Im1.
3.3. Laboratory Investigation of Children Immunograms
3.4. Analysis of Immune Status and Immunograms from Biochemical Tests for Immunoglobulins
3.4.1. Immune Status k1
3.4.2. Immune Status k2
3.4.3. Immune Status k3
3.4.4. Immune Status k4
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Smith, K.A. Louis Pasteur, the father of immunology? Front. Immun. 2012, 3, 68. [Google Scholar] [CrossRef] [PubMed]
- Cavaillon, J.-M. The historical milestones in the understanding of leukocyte biology initiated by Elie Metchnikoff. J. Leukoc. Biol. 2011, 90, 413–424. [Google Scholar] [CrossRef] [PubMed]
- Boechat, J.L.; Chora, I.; Morais, A.; Delgado, L. The immune response to SARS-CoV-2 and COVID-19 immunopathology—Current perspectives. Pulmonology 2021, 27, 423–437. [Google Scholar] [CrossRef]
- Celardo, I.; Pace, L.; Cifaldi, L.; Gaudio, C.; Barnaba, V. The immune system view of the coronavirus SARS-CoV-2. Biol. Direct 2020, 15, 30. [Google Scholar] [CrossRef]
- Chen, Y.; Li, Q.; Gu, D. Emerging coronaviruses: Genome structure, replication, and pathogenesis. J. Med. Virol. 2020, 92, 418–423. [Google Scholar] [CrossRef]
- Ahn, D.G.; Choi, J.K.; Taylor, D.R.; Oh, J.W. Biochemical characterization of a recombinant SARS coronavirus nsp12 RNA-dependent RNA polymerase capable of copying viral RNA templates. Arch. Virol. 2012, 157, 2095–2104. [Google Scholar] [CrossRef]
- Tian, X.; Li, C.; Huang, A.; Xia, S.; Lu, S.; Shi, Z.; Lu, L.; Jiang, S.; Yang, Z.; Wu, Y.; et al. Potent binding of 2019 novel coronavirus spike protein by a SARS coronavirus specific human monoclonal antibody. Emerg. Microbes Infect. 2020, 9, 382–385. [Google Scholar] [CrossRef] [PubMed]
- Kostarev, S.N.; Kochetova, O.V.; Tatarnikova, N.A.; Sereda, T.G. Project design of a device for express analysis for Coronaviridae antigens based on Omron industrial programmable logic controller. Int. J. Biol. Biomed. Eng. 2020, 14, 21–31. [Google Scholar] [CrossRef]
- Kostarev, S.N.; Kochetova, O.V.; Tatarnikova, N.A.; Sereda, T.G. Study of the human infectious safety model under the influence of SARS-CoV-2 on the example of the Perm Krai of the Russian Federation. E3S Web Conf. 2021, 282, 06005. [Google Scholar] [CrossRef]
- Tatarnikova, N.A.; Prokhorova, T.S. Antiviral Immunity. Study Guide for the Course “Veterinary Virology”; Printing house of the PGSKHA: Perm, Russia, 1998; 26p. (In Russian) [Google Scholar]
- Kvartych, E.I. The state of the human immune system under the conditions of urbanization. Sci. Electron. J. Meridian 2020, 19, 12–14. [Google Scholar]
- Efimov, G.A. Interview with G.A. Efimov. “T-CELL immune is much better protected from virus mutations than humoral”. Nature 2021, 5, 3–8. [Google Scholar]
- Majumdar, S.; Verma, R.; Saha, A.; Bhattacharyya, P.; Maji, P.; Surjit, M.; Kundu, M.; Basu, J.; Saha, S. Perspectives about modulating host immune system in targeting SARS-CoV-2 in India. Front. Genet. 2021, 12, 637362. [Google Scholar] [CrossRef] [PubMed]
- Carsetti, R.; Quintarelli, C.; Quinti, I.; Piano Mortari, E.; Zumla, A.; Ippolito, G.; Locatelli, F. The immune system of children: The key to understanding SARS-CoV-2 susceptibility? Lancet Child Adolesc. Health 2020, 4, 414–416. [Google Scholar] [CrossRef]
- Rzyankina, M.F.; Potapova, K.E.; Ulyanova, E.A.; Korzinina, N.A. The prevalence of a new coronavirus infection among children. Literature review. Vestn. Obs. Zdorovya Zdr. Daln. Vost. Ross. 2021, 2, 48–58. [Google Scholar]
- Povorova, O.V. State of cellular immunity in children with recurrent respiratory diseases. Immunopathol. Allergol. Infectology 2020, 4, 64–74. [Google Scholar] [CrossRef]
- Manuilov, V.A.; Kudryashova, A.M.; Cherepovich, B.S.; Kharchenko, O.S.; Shatunova, P.O.; Semashko, M.A.; Vasina, D.V.; Grigoriev, I.V.; Mazunina, E.P.; Kleymenov, D.A.; et al. Differential diagnosis of immunity to SARS-CoV-2: Neutralizing antibodies, avidity, cellular immunity. In the Collection: Socially Significant and Especially Dangerous Infectious Diseases. In Proceedings of the VIII All-Russian Interdisciplinary Scientific and Practical Conference with International Participation, Sochi, Russia, 26–29 October 2021; pp. 124–125. [Google Scholar]
- Popova, A.Y.; Ezhlova, E.B.; Mel’nikova, A.A.; Danilova, T.E.; Bulanov, M.V.; Lyalina, L.V.; Smirnov, V.S.; Totolyan, A.A. SARS-CoV-2 seroprevalence among the population of the Belgorod region against the backdrop of the COVID-19 epidemic. Epidemiology and infectious diseases. Top. Issues 2021, 11, 18–24. [Google Scholar]
- Pogodina, E.A.; Lobov, A.V.; Ivanova, P.I.; Kazey, V.; Shubina, I. Induction of anti-SARS-CoV-2 immune reactions in immune compromised patients. Ross. Bioterapevticheskiy Zurnal Russ. J. Biother. 2021, 20, 18–25. [Google Scholar] [CrossRef]
- Röltgen, K.; Nielsen, S.C.; Silva, O.; Younes, S.F.; Zaslavsky, M.; Costales, C.; Yang, F.; Wirz, O.F.; Solis, D.; Hoh, R.A.; et al. Immune imprinting, breadth of variant recognition, and germinal center response in human SARS-CoV-2 infection and vaccination. Cell 2022, 185, 1025–1040. [Google Scholar] [CrossRef]
- Kharchenko, E.P. New method for immunoepitope recognition, markers of long-term immunity, immunosuppressive domains and COVID-19 vaccines. Epidemiol. Vaccin. 2022, 21, 4–20. [Google Scholar] [CrossRef]
- Korzhenevsky, A.A. Interpretation of the Immunogram in Inflammatory Processes: Textbook. Allowance; Ufa: Publishing House of FGBOU VO BSMU of the Ministry of Health of Russia: Moscow, Russia, 2017; p. 115.
- Sereda, T.G.; Kostarev, S.N.; Kochinov, Y.A.; Kochinova, T.V. Building a tool model for the study of the ecosystem “coronavirus–Vector–human–Environment”. IOP Conf. Ser. Earth Environ. Sci. 2020, 548, 042030. [Google Scholar] [CrossRef]
- Chen, T.-M.; Rui, J.; Wang, Q.-P.; Zhao, Z.-Y.; Cui, J.-A.; Yin, L. A mathematical model for simulating the phase-based transmissibility of a novel coronavirus. Infect. Dis. Poverty 2020, 9, 24. [Google Scholar] [CrossRef] [PubMed]
- Elfiky, A.A. Anti-HCV, nucleotide inhibitors, repurposing against COVID-19. Life Sci. 2020, 248, 117477. [Google Scholar] [CrossRef] [PubMed]
- Grech, V. Unknown unknowns—COVID-19 and potential global mortality. Early Hum. Dev. 2020, 144, 105026. [Google Scholar] [CrossRef] [PubMed]
- Sarkodie, S.A.; Owusu, P.A. Investigating the cases of novel coronavirus disease (COVID-19) in China using dynamic statistical techniques. Heliyon 2020, 6, e03747. [Google Scholar] [CrossRef] [PubMed]
- Enserink, M.; Kupferschmidt, K. With COVID-19, modeling takes on life and death importance. Science 2020, 367, 1414–1415. [Google Scholar] [CrossRef] [PubMed]
- Robson, B. Computers and viral diseases. Preliminary bioinformatics studies on the design of a synthetic vaccine and a preventative peptidomimetic antagonist against the SARS-CoV-2 (2019-nCoV, COVID-19) coronavirus. Comput. Biol. Med. 2020, 119, 103670. [Google Scholar] [CrossRef] [PubMed]
- Kurosh, A.A. Course of Higher Algebra: A Textbook for Universities, 22nd ed.; Lan: St. Petersburg, Russia, 2021; 432p, ISBN 978-581-146-851-5. (In Russian) [Google Scholar]
 reference interval,
reference interval,  exceeding the reference interval,
exceeding the reference interval,  subceeding the reference interval.
subceeding the reference interval.
 reference interval,
reference interval,  exceeding the reference interval,
exceeding the reference interval,  subceeding the reference interval.
subceeding the reference interval.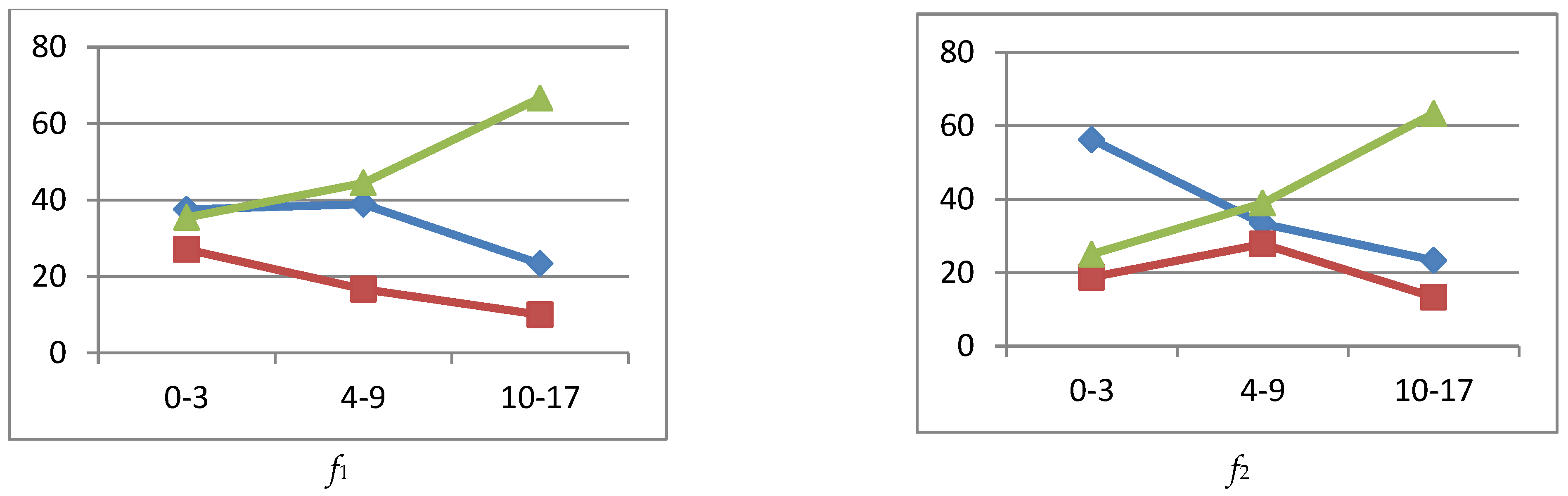
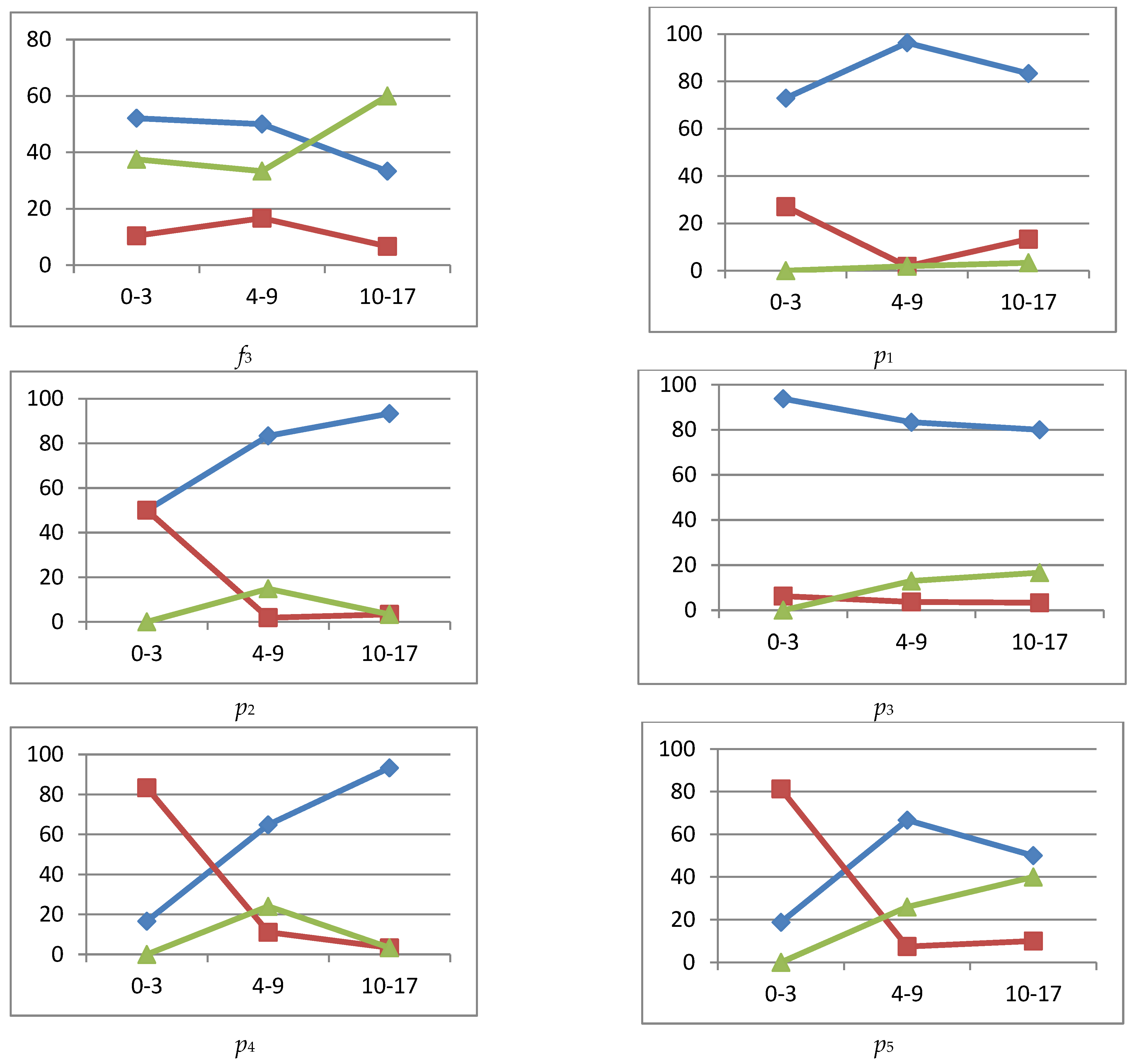
 reference interval,
reference interval,  more (exceeding the reference interval),
more (exceeding the reference interval),  smaller (subceeding the reference interval), IgA, IgM immunoglobulins are higher (more) or lower (smaller) if they are outside the reference interval.
smaller (subceeding the reference interval), IgA, IgM immunoglobulins are higher (more) or lower (smaller) if they are outside the reference interval.
 reference interval,
reference interval,  more (exceeding the reference interval),
more (exceeding the reference interval),  smaller (subceeding the reference interval), IgA, IgM immunoglobulins are higher (more) or lower (smaller) if they are outside the reference interval.
smaller (subceeding the reference interval), IgA, IgM immunoglobulins are higher (more) or lower (smaller) if they are outside the reference interval.
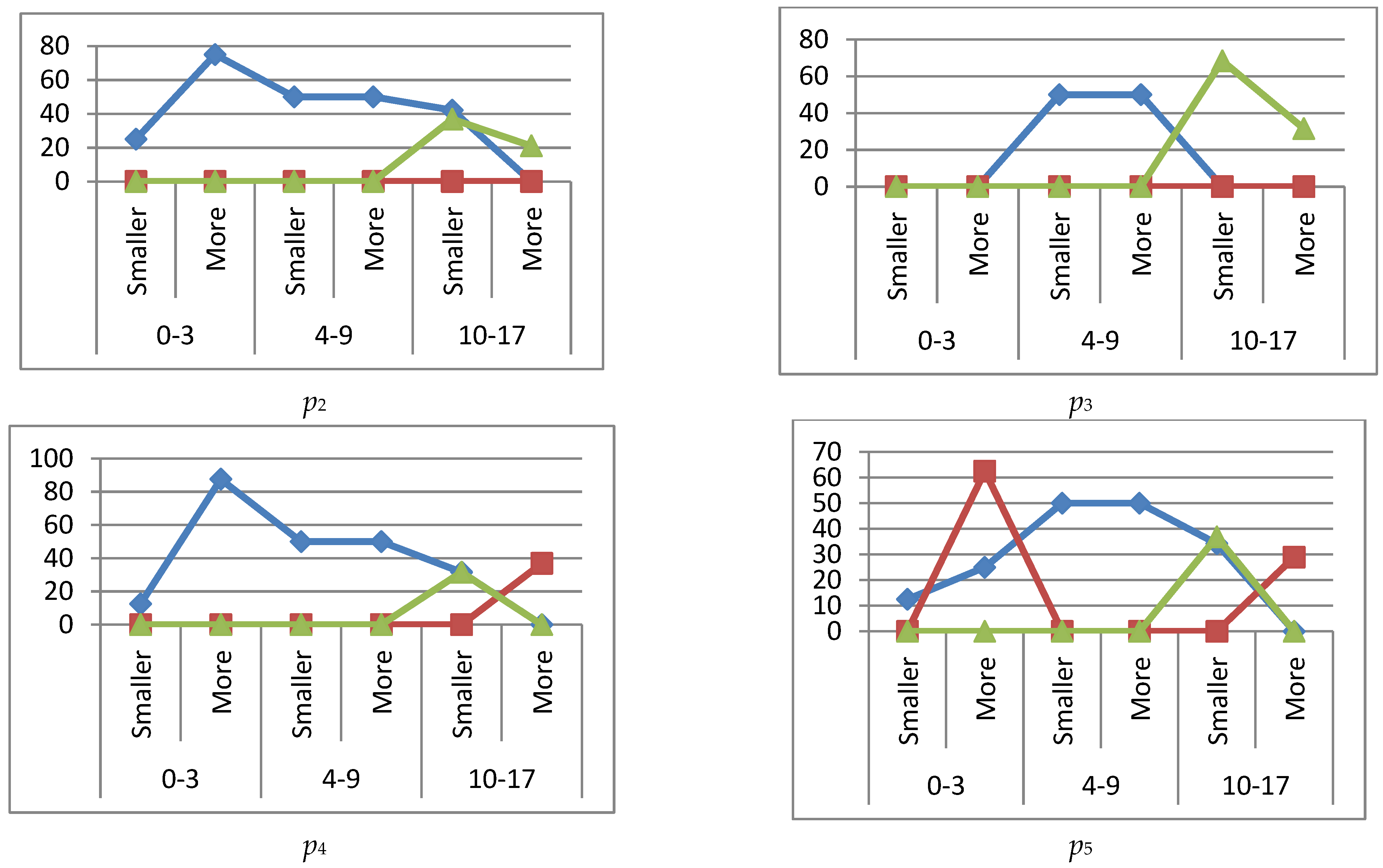
 reference interval,
reference interval,  more (exceeding the reference interval),
more (exceeding the reference interval),  smaller (subceeding the reference interval), when IgG, IgM immunoglobulins are higher (more) or lower (smaller) than the reference interval.
smaller (subceeding the reference interval), when IgG, IgM immunoglobulins are higher (more) or lower (smaller) than the reference interval.
 reference interval,
reference interval,  more (exceeding the reference interval),
more (exceeding the reference interval),  smaller (subceeding the reference interval), when IgG, IgM immunoglobulins are higher (more) or lower (smaller) than the reference interval.
smaller (subceeding the reference interval), when IgG, IgM immunoglobulins are higher (more) or lower (smaller) than the reference interval.
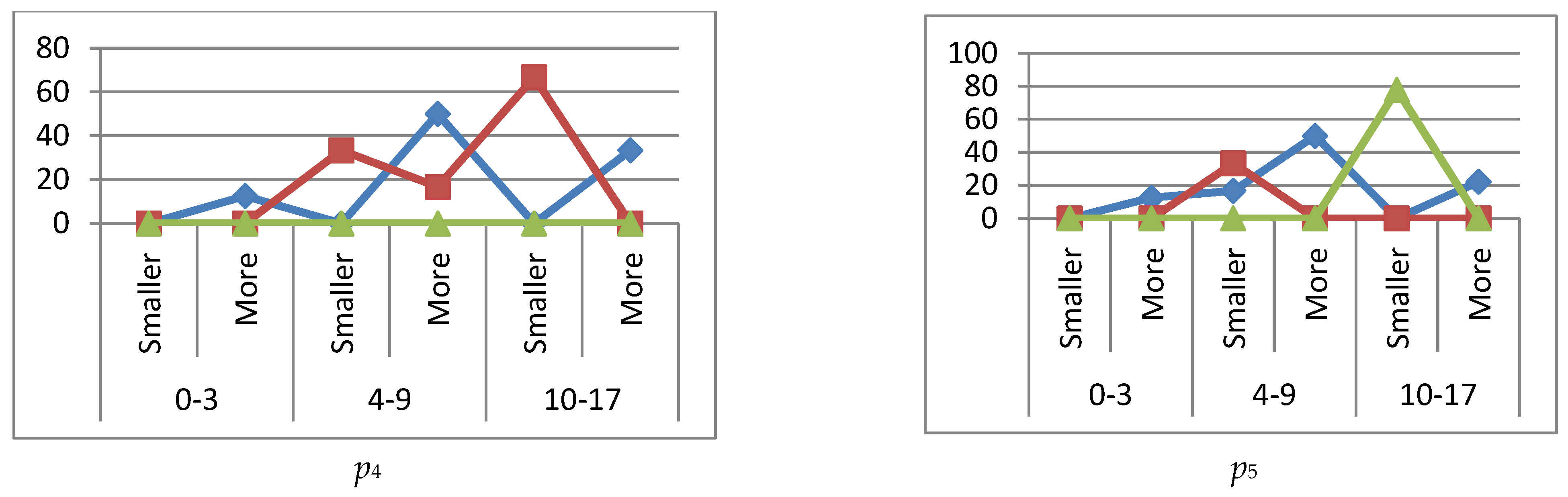
 reference interval,
reference interval,  more (exceeding the ref. interval),
more (exceeding the ref. interval),  smaller (subceeding the ref. interval), when immunoglobulin IgG is increased (more) or decreased (smaller) beyond the reference interval.
smaller (subceeding the ref. interval), when immunoglobulin IgG is increased (more) or decreased (smaller) beyond the reference interval.
 reference interval,
reference interval,  more (exceeding the ref. interval),
more (exceeding the ref. interval),  smaller (subceeding the ref. interval), when immunoglobulin IgG is increased (more) or decreased (smaller) beyond the reference interval.
smaller (subceeding the ref. interval), when immunoglobulin IgG is increased (more) or decreased (smaller) beyond the reference interval.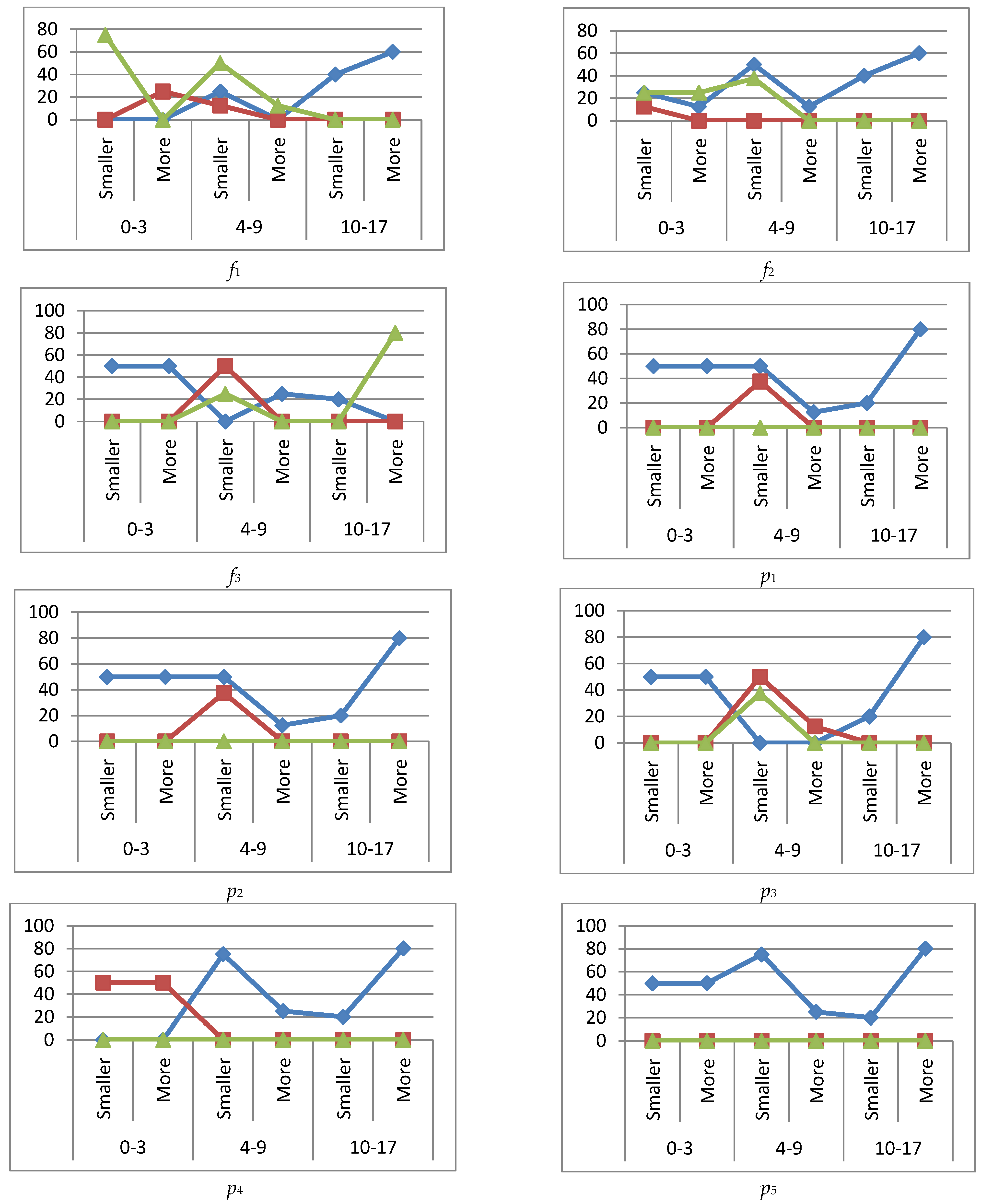
| Input Signals Indicators | Dimension | Mnemonics |
|---|---|---|
| Immunoglobulin A | g/L | A |
| Immunoglobulin M | g/L | M |
| Immunoglobulin G | g/L | G |
| Absolute value of phagocytosis | 109/L | f1 |
| Phagocytic number | f2 | |
| Phagocytic index | f3 | |
| Leukocytes | 109/L | p1 |
| Lymphocytes | 109/L | p2 |
| NK cells (CD16+CD56+) | 109/L | p3 |
| T helpers (CD3+CD4+) | 109/L | p4 |
| (CD4+/CD8+) immunoregulation index | p5 |
| Age Range, Years | The Number of Examined Samples | ||||||
|---|---|---|---|---|---|---|---|
| k1 | k2 | k3 | k4 | ||||
| ↓ | ↑ | ↓ | ↑ | ↓ | ↑ | ||
| 0–3 | 48 | 2 | 6 | 1 | 1 | 6 | 2 |
| 72.7% | 12.1% | 3% | 12.1% | ||||
| 4–9 | 54 | 24 | 10 | 2 | 4 | 4 | 1 |
| 70.1% | 15.6% | 7.8% | 6.5% | ||||
| 10–17 | 30 | 25 | 13 | 2 | 7 | 1 | 4 |
| 36.5% | 46.3% | 10.9% | 6.1% | ||||
| Total by subcategories | 132 | 51 | 29 | 5 | 12 | 11 | 7 |
| Total | 132 | 80 | 17 | 18 | |||
| 53.4% | 32.4% | 6.9% | 7.3% | ||||
| Indicator | Condition | Age Range, Years | |||||
|---|---|---|---|---|---|---|---|
| 0–3 | 4–9 | 10–17 | |||||
| RI | % | RI | % | RI | % | ||
| RI | 18 | 37.50 | 21 | 38.89 | 7 | 23.33 | |
| f1 | ↑ | 13 | 27.08 | 9 | 16.67 | 3 | 10.00 |
| ↓ | 17 | 35.42 | 24 | 44.44 | 20 | 66.67 | |
| RI | 27 | 56.25 | 18 | 33.33 | 7 | 23.33 | |
| f2 | ↑ | 9 | 18.75 | 15 | 27.78 | 4 | 13.33 |
| ↓ | 12 | 25.00 | 21 | 38.89 | 19 | 63.33 | |
| RI | 25 | 52.08 | 27 | 50.00 | 10 | 33.33 | |
| f3 | ↑ | 5 | 10.42 | 9 | 16.67 | 2 | 6.67 |
| ↓ | 18 | 37.50 | 18 | 33.33 | 18 | 60.00 | |
| RI | 35 | 72.92 | 52 | 96.30 | 25 | 83.33 | |
| p1 | ↑ | 13 | 27.08 | 1 | 1.85 | 4 | 13.33 |
| ↓ | 0.00 | 1 | 1.85 | 1 | 3.33 | ||
| RI | 24 | 50.00 | 45 | 83.33 | 28 | 93.33 | |
| p2 | ↑ | 24 | 50.00 | 1 | 1.85 | 1 | 3.33 |
| ↓ | 0.00 | 8 | 14.81 | 1 | 3.33 | ||
| RI | 45 | 93.75 | 45 | 83.33 | 24 | 80.00 | |
| p3 | ↑ | 3 | 6.25 | 2 | 3.70 | 1 | 3.33 |
| ↓ | 0.00 | 7 | 12.96 | 5 | 16.67 | ||
| RI | 8 | 16.67 | 35 | 64.81 | 28 | 93.33 | |
| p4 | ↑ | 40 | 83.33 | 6 | 11.11 | 1 | 3.33 |
| ↓ | 0.00 | 13 | 24.07 | 1 | 3.33 | ||
| RI | 9 | 18.75 | 36 | 66.67 | 15 | 50.00 | |
| p5 | ↑ | 39 | 81.25 | 4 | 7.41 | 3 | 10.00 |
| ↓ | 0.00 | 14 | 25.93 | 12 | 40.00 | ||
| Ind. | Cond. | Age Range, Years | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0–3 | 4–9 | 10–17 | |||||||||||
| ↓ * | % | ↑ | % | ↓ | % | ↑ | % | ↓ | % | ↑ | % | ||
| RI | 2 | 25 | 6 | 75 | 22 | 57.89 | 6 | 15.79 | |||||
| f1 | ↑ | 2 | 16.67 | 4 | 10.53 | 6 | 15.79 | ||||||
| ↓ | 0.00 | 10 | 83.33 | 0.00 | 0 | ||||||||
| RI | 3 | 37.5 | 5 | 62.5 | 3 | 25.00 | 0 | 22 | 57.89 | 5 | 13.16 | ||
| f2 | ↑ | 0.00 | 0 | 6 | 15.79 | 5 | 13.16 | ||||||
| ↓ | 0.00 | 9 | 75 | 0.00 | 0 | ||||||||
| RI | 0 | 0.00 | 0 | 0.00 | 0 | ||||||||
| f3 | ↑ | 0 | 0.00 | 0 | 0.00 | 0 | |||||||
| ↓ | 0 | 5 | 62.5 | 0.00 | 8 | 66.67 | 8 | 21.05 | 7 | 18.42 | |||
| RI | 1 | 12.5 | 7 | 87.5 | 6 | 50.00 | 6 | 50 | 0.00 | 0 | |||
| p1 | ↑ | 0 | 0 | 0.00 | 0 | 38 | 100.00 | 0 | |||||
| ↓ | 0 | 0 | 0.00 | 0 | 0.00 | 0 | |||||||
| RI | 2 | 25 | 6 | 75 | 6 | 50.00 | 6 | 50 | 16 | 42.11 | 0 | ||
| p2 | ↑ | 0 | 0 | 0.00 | 0 | 0.00 | 0 | ||||||
| ↓ | 0 | 0 | 0.00 | 0 | 14 | 36.84 | 8 | 21.05 | |||||
| RI | 0 | 0 | 0 | 0 | 6 | 50.00 | 6 | 50 | 0.00 | 0 | |||
| p3 | ↑ | 0 | 0 | 0.00 | 0 | 0.00 | 0 | ||||||
| ↓ | 0 | 0 | 0.00 | 0 | 26 | 68.42 | 12 | 31.58 | |||||
| RI | 1 | 12.5 | 7 | 87.5 | 6 | 50.00 | 6 | 50 | 12 | 31.58 | 0 | ||
| p4 | ↑ | 0 | 0 | 0.00 | 0 | 0.00 | 14 | 36.84 | |||||
| ↓ | 0 | 0 | 0.00 | 0 | 12 | 31.58 | 0 | ||||||
| RI | 1 | 12.5 | 2 | 25 | 6 | 50.00 | 6 | 50 | 13 | 34.21 | 0 | ||
| p5 | ↑ | 0 | 5 | 62.5 | 0.00 | 0 | 0.00 | 11 | 28.95 | ||||
| ↓ | 0 | 0 | 0.00 | 0 | 14 | 36.84 | 0 | ||||||
| Ind. | Cond. | Age Range, Years | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0–3 | 4–9 | 10–17 | |||||||||||
| ↓ * | % | ↑ | % | ↓ | % | ↑ | % | ↓ | % | ↑ | % | ||
| RI | 1 | 50.00 | 1 | 0.00 | 2 | 33.33 | 0.00 | 0.00 | |||||
| f1 | ↑ | 0.00 | 2 | 33.33 | 2 | 33.33 | 0.00 | 2 | 22.22 | ||||
| ↓ | 0.00 | 0.00 | 0.00 | 0.00 | 7 | 77.78 | 0.00 | ||||||
| RI | 1 | 1 | 12.50 | 0.00 | 2 | 33.33 | 0.00 | 0.00 | |||||
| f2 | ↑ | 0.00 | 0.00 | 4 | 66.67 | 0.00 | 0.00 | 2 | 22.22 | ||||
| ↓ | 0.00 | 0.00 | 0.00 | 0.00 | 7 | 77.78 | 0.00 | ||||||
| RI | 1 | 1 | 12.50 | 3 | 50.00 | 3 | 50.00 | 0.00 | 0.00 | ||||
| f3 | ↑ | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 3 | 33.33 | |||||
| ↓ | 0.00 | 0.00 | 0.00 | 0.00 | 6 | 66.67 | 0.00 | ||||||
| RI | 1 | 1 | 12.50 | 3 | 50.00 | 3 | 50.00 | 0.00 | 0.00 | ||||
| p1 | ↑ | 0.00 | 0.00 | 0.00 | 0.00 | 7 | 77.78 | 2 | 22.22 | ||||
| ↓ | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | |||||||
| RI | 1 | 1 | 12.50 | 3 | 50.00 | 3 | 50.00 | 6 | 66.67 | 3 | 33.33 | ||
| p2 | ↑ | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | ||||||
| ↓ | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | |||||||
| RI | 1 | 1 | 12.50 | 3 | 50.00 | 3 | 50.00 | 5 | 55.56 | 24 | 266.67 | ||
| p3 | ↑ | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | ||||||
| ↓ | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | |||||||
| RI | 1 | 1 | 12.50 | 0.00 | 3 | 50.00 | 0.00 | 3 | 33.33 | ||||
| p4 | ↑ | 0.00 | 0.00 | 2 | 33.33 | 1 | 16.67 | 6 | 66.67 | 0.00 | |||
| ↓ | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | |||||||
| RI | 1 | 1 | 12.50 | 1 | 16.67 | 3 | 50.00 | 0.00 | 2 | 22.22 | |||
| p5 | ↑ | 0.00 | 0.00 | 2 | 33.33 | 0.00 | 0.00 | 0.00 | |||||
| ↓ | 0.00 | 0.00 | 0.00 | 0.00 | 7 | 77.78 | 0.00 | ||||||
| Ind. | Cond. | Age Range, Years | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0–3 | 4–9 | 10–17 | |||||||||||
| ↓ * | % | ↑ | % | ↓ | % | ↑ | % | ↓ | % | ↑ | % | ||
| RI | 2 | 25.00 | 0 | 2 | 40.00 | 3 | 60 | ||||||
| f1 | ↑ | 2 | 25 | 1 | 12.50 | 0 | 0.00 | 0 | |||||
| ↓ | 6 | 75 | 0 | 4 | 50.00 | 1 | 12.5 | 0.00 | 0 | ||||
| RI | 2 | 25 | 1 | 12.5 | 4 | 50.00 | 1 | 12.5 | 2 | 40.00 | 3 | 60 | |
| f2 | ↑ | 1 | 12.5 | 0 | 0.00 | 0 | 0.00 | 0 | |||||
| ↓ | 2 | 25 | 2 | 25 | 3 | 37.50 | 0 | 0.00 | 0 | ||||
| RI | 4 | 50 | 4 | 50 | 0.00 | 2 | 25 | 1 | 20.00 | 0 | |||
| f3 | ↑ | 0 | 0 | 4 | 50.00 | 0 | 0.00 | 0 | |||||
| ↓ | 0 | 0 | 2 | 25.00 | 0 | 0.00 | 4 | 80 | |||||
| RI | 4 | 50 | 4 | 50 | 4 | 50.00 | 1 | 12.5 | 1 | 20.00 | 4 | 80 | |
| p1 | ↑ | 0 | 0 | 3 | 37.50 | 0 | 0.00 | 0 | |||||
| ↓ | 0 | 0 | 0.00 | 0 | 0.00 | 0 | |||||||
| RI | 4 | 50 | 4 | 50 | 4 | 50.00 | 1 | 12.5 | 1 | 20.00 | 4 | 80 | |
| p2 | ↑ | 0 | 0 | 3 | 37.50 | 0 | 0.00 | 0 | |||||
| ↓ | 0 | 0 | 0.00 | 0 | 0.00 | 0 | |||||||
| RI | 4 | 50 | 4 | 50 | 0.00 | 0 | 1 | 20.00 | 4 | 80 | |||
| p3 | ↑ | 0 | 0 | 4 | 50.00 | 1 | 12.5 | 0.00 | 0 | ||||
| ↓ | 0 | 0 | 3 | 37.50 | 0 | 0.00 | 0 | ||||||
| RI | 0 | 0 | 6 | 75.00 | 2 | 25 | 1 | 20.00 | 4 | 80 | |||
| p4 | ↑ | 4 | 50 | 4 | 50 | 0.00 | 0 | 0.00 | 0 | ||||
| ↓ | 0 | 0 | 0.00 | 0 | 0.00 | 0 | |||||||
| RI | 4 | 50 | 4 | 50 | 6 | 75.00 | 2 | 25 | 1 | 20.00 | 4 | 80 | |
| p5 | ↑ | 0 | 0 | 0.00 | 0 | 0.00 | 0 | ||||||
| ↓ | 0 | 0 | 0.00 | 0 | 0.00 | 0 | |||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kostarev, S.; Komyagina, O.; Fayzrakhmanov, R.; Kurushin, D.; Tatarnikova, N.; Novikova, O.; Sereda, T. Impact of the New Coronavirus Infection on the Immune System of Children and Adolescents in the Region of the Russian Federation. Int. J. Environ. Res. Public Health 2022, 19, 13669. https://doi.org/10.3390/ijerph192013669
Kostarev S, Komyagina O, Fayzrakhmanov R, Kurushin D, Tatarnikova N, Novikova O, Sereda T. Impact of the New Coronavirus Infection on the Immune System of Children and Adolescents in the Region of the Russian Federation. International Journal of Environmental Research and Public Health. 2022; 19(20):13669. https://doi.org/10.3390/ijerph192013669
Chicago/Turabian StyleKostarev, Sergey, Oksana Komyagina, Rustam Fayzrakhmanov, Daniel Kurushin, Natalya Tatarnikova, Oksana Novikova (Kochetova), and Tatyana Sereda. 2022. "Impact of the New Coronavirus Infection on the Immune System of Children and Adolescents in the Region of the Russian Federation" International Journal of Environmental Research and Public Health 19, no. 20: 13669. https://doi.org/10.3390/ijerph192013669
APA StyleKostarev, S., Komyagina, O., Fayzrakhmanov, R., Kurushin, D., Tatarnikova, N., Novikova, O., & Sereda, T. (2022). Impact of the New Coronavirus Infection on the Immune System of Children and Adolescents in the Region of the Russian Federation. International Journal of Environmental Research and Public Health, 19(20), 13669. https://doi.org/10.3390/ijerph192013669







