Relationship between Attention Capacity and Hand–Eye Reaction Time in Adolescents between 15 and 18 Years of Age
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
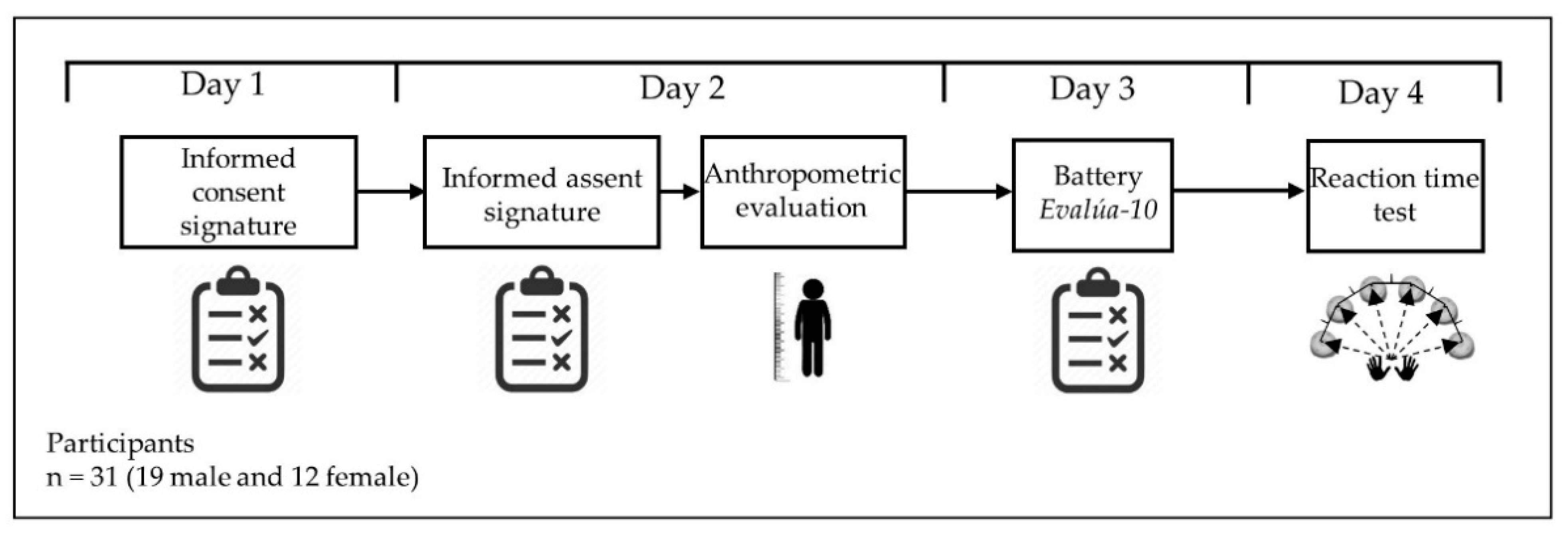
2.2. Research Design
2.3. Anthropometric Measurements
2.4. Psychopedagogical Battery Evalúa-10
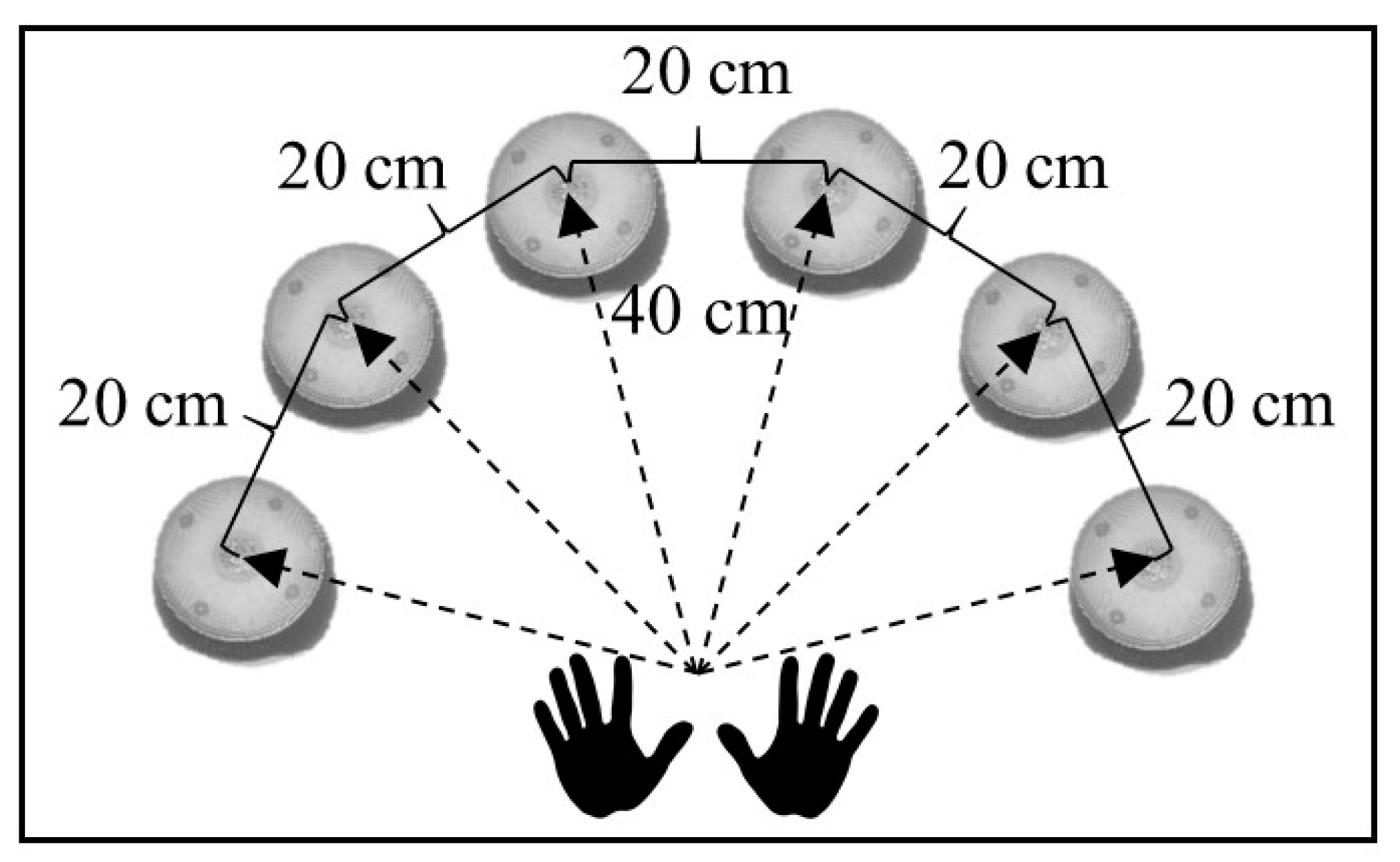
2.5. Reaction Time
2.6. Data Analysis
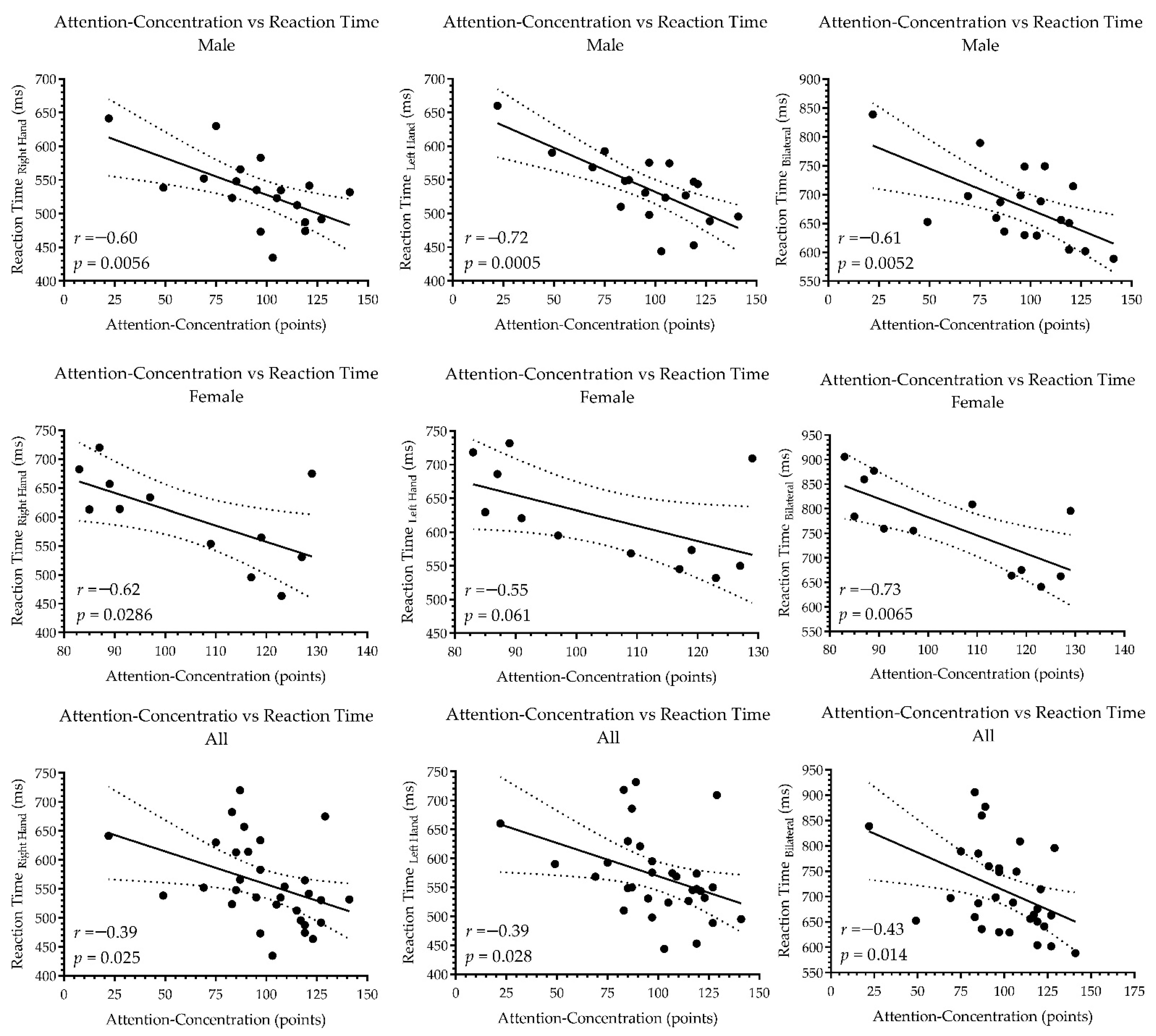
3. Results
4. Discussion
4.1. Attention and Concentration
4.2. Reaction Time
4.3. Physical Exercise and Brain Activation
4.4. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, Y.; Demnitz, N.; Yamamoto, S.; Yaffe, K.; Lawlor, B.; Leroi, I. Defining brain health: A concept analysis. Int. J. Geriatr. Psychiatry 2021, 37, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Okano, H.; Miyawaki, A.; Kasai, K. Brain/MINDS: Brain-mapping project in Japan. Philos. Trans. B 2015, 370, 20140310. [Google Scholar] [CrossRef] [PubMed]
- Ardila, A. Is intelligence equivalent to executive functions? Psicothema 2018, 30, 159–164. [Google Scholar] [CrossRef]
- Santa-Cruz, C.; Rosas, R. Mapping of Executive Functions/Cartografía de las Funciones Ejecutivas. Estud. Psicol. 2017, 38, 284–310. [Google Scholar] [CrossRef]
- Spinella, M. Self-rated executive function: Development of the executive function index. Int. J. Neurosci. 2005, 115, 649–667. [Google Scholar] [CrossRef] [PubMed]
- Lozano, A.; Ostrosky, F. Desarrollo de las funciones ejecutivas y la corteza prefrontal. Rev. Neuropsicol. Neuropsiquiatría Neurocienc. 2011, 11, 159–172. [Google Scholar]
- Chan, R.C.K.; Shum, D.; Toulopoulou, T.; Chen, E.Y.H. Assessment of executive functions: Review of instruments and identification of critical issues. Arch. Clin. Neuropsychol. 2008, 23, 201–216. [Google Scholar] [CrossRef]
- Rebollo, M.A.; Montiel, S. Atención y funciones ejecutivas. Rev. Neurol. 2006, 42, 3–7. [Google Scholar] [CrossRef]
- Singer, R.N.; Hausenblas, H.A.; Janelle, C.M. Handbook of Sport Psychology, 2nd ed.; John Wiley & Sons Inc.: New York, NY, USA, 2001. [Google Scholar]
- Rodríguez, M.C.; Montoya, J.C. Entrenamiento en el mantenimiento de la atención en deportistas y su afectividad en el rendimiento [Training in the maintenance of attention in sportsmen and women, and their effectiveness of yield]. Acta Colomb. Psicol. 2006, 9, 99–112. [Google Scholar]
- Kerr, A.; Zelazo, P.D. Development of “hot” executive function: The children’s gambling task. Brain Cogn. 2004, 55, 148–157. [Google Scholar] [CrossRef]
- Bernabéu, E. La atención y la memoria como claves del proceso de aprendizaje. Aplicaciones para el entorno escolar. ReiDoCrea Rev. Electrónica Investig. Docencia Creat. 2017, 6, 16–23. [Google Scholar] [CrossRef]
- Estévez-González, A.; García-Sánchez, C.; Junqué, C. La atención: Una compleja función cerebral. Rev. Neurol. 1997, 25, 1989–1997. [Google Scholar] [PubMed]
- Reigal, R.E.; Barrero, S.; Martín, I.; Morales-Sánchez, V.; Juárez-Ruiz de Mier, R.; Hernández-Mendo, A. Relationships between reaction time, selective attention, physical activity, and physical fitness in children. Front. Psychol. 2019, 10, 2278. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, A.; Silva, A. Sistema para medir el tiempo de reacción. Jóvenes Ciencia. Rev. Divulg. Científica 2018, 4, 567–572. [Google Scholar]
- Hall, J.E. Guyton y Hall. Tratado de Fisiología Médica; Elsevier: Amsterdam, The Netherlands, 2006. [Google Scholar]
- Vences de Brito, A.; Silva, C.; Cid, L.; Ferreira, D.; Marques, A. Atención y tiempo de reacción en practicantes de kárate shotokan. Rev. Artes Marciales Asiáticas 2011, 6, 141. [Google Scholar] [CrossRef][Green Version]
- Valdés, J.L.; Torrealba, F. La corteza prefrontal medial controla el alerta conductual y vegetativo. Implicancias en desórdenes de la conducta. Rev. Chil. Neuropsiquiatr. 2006, 44, 195–204. [Google Scholar] [CrossRef]
- Hülsdünker, T.; Ostermann, M.; Mierau, A. The speed of neural visual motion perception and processing determines the visuomotor reaction time of young elite table tennis athletes. Front. Behav. Neurosci. 2019, 13, 165. [Google Scholar] [CrossRef]
- Faul, F. G*Power 2020. Available online: https://www.psychologie.hhu.de/arbeitsgruppen/allgemeine-psychologie-und-arbeitspsychologie/gpower (accessed on 14 July 2022).
- Harriss, D.; Macsween, A.; Atkinson, G. Ethical standards in sport and exercise science research: 2020 update. Int. J. Sports Med. 2019, 40, 813–817. [Google Scholar] [CrossRef]
- Ato, M.; López, J.J.; Benavente, A. A classification system for research designs in psychology. An. Psicol. 2013, 29, 1038–1059. [Google Scholar] [CrossRef]
- Barrera, A.; Gladys, M. Estandáres Antropométricos para Evaluación del Estado Nutritivo; INTA: Santiago, Chile, 2004. [Google Scholar]
- García Vidal, J.; González Manjón, D.; Martínez García, M. Batería Psicopedagógica Evalúa-10, 4th ed.; Instituto de Evaluación Psicopedagógica EOS: Madrid, España, 2019; ISBN 978-84-9727-819-5. [Google Scholar]
- Hopkins, W.G.; Marshall, S.W.; Batterham, A.M.; Hanin, J. Progressive statistics for studies in sports medicine and exercise science. Med. Sci. Sports Exerc. 2009, 41, 3–12. [Google Scholar] [CrossRef]
- Hopkins, W.G. Measures of reliability in sports medicine and science. Sport. Med. 2000, 30, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Hu, P.; Fan, J.; Wang, K. Gender differences associated with orienting attentional networks in healthy subjects. Chin. Med. J. 2013, 126, 2308–2312. [Google Scholar]
- Sebastián, M.V.; Arana, M.V.; Arcos, C.; Navascués, M.A.; Idiazábal, M.Á.; Ruiz, C.; Iso, J.M. Procesos atencionales en tareas de simulación militar: Diferencias relacionadas con el género. Rev. Científica Soc. Española Enfermería Neurológica 2018, 48, 2–8. [Google Scholar] [CrossRef]
- Gutiérrez-Ruiz, K.; Mejía, K.; López, A.; Cuadros, U.; Suarez, D. Influencia del sexo y la dificultad de la tarea en el cambio atencional. Anu. Psicol. 2017, 47, 80–86. [Google Scholar] [CrossRef]
- Luna, F.G.; Marino, J.; Macbeth, G.; Foa Torres, G. ¿Existen diferencias entre sexos en las redes atencionales? Una revisión sistemática sobre el alerta fásica, vigilancia, orientación y control ejecutivo. Psychologia 2016, 10, 63–71. [Google Scholar] [CrossRef]
- Neto, O.; Pacheco, M.; Bolander, R.; Bir, C. Force, reaction time, and precision of Kung Fu strikes. Percept. Mot. Skills 2009, 109, 295–303. [Google Scholar] [CrossRef]
- Szabo, D.A.; Neagu, N.; Teodorescu, S.; Panait, C.M.; Sopa, I.S. Study on the influence of proprioceptive control versus visual control on reaction speed, hand coordination, and lower limb balance in young students 14–15 years old. Int. J. Environ. Res. Public Health 2021, 18, 10356. [Google Scholar] [CrossRef]
- Akarsu, S.; Caliskan, E.; Dane, S. Athletes have faster eye-hand visual reaction times and higher scores on visuospatial intelligence than non-athletes. Turk. J. Med. Sci. 2009, 39, 871–874. [Google Scholar] [CrossRef]
- Adleman, N.E.; Chen, G.; Reynolds, R.C.; Frackman, A.; Razdan, V.; Weissman, D.H.; Pine, D.S.; Leibenluft, E. Age-related differences in the neural correlates of trial-to-trial variations of reaction time. Dev. Cogn. Neurosci. 2016, 19, 248–257. [Google Scholar] [CrossRef]
- Hülsdünker, T.; Strüder, H.K.; Mierau, A. The athletes’ visuomotor system-Cortical processes contributing to faster visuomotor reactions. Eur. J. Sport Sci. 2018, 18, 955–964. [Google Scholar] [CrossRef] [PubMed]
- Semeijn, E.; Korten, N.C.; Comijs, H.; Michielsen, M.; Deeg, D.; Beekman, A.; Kooij, J. No lower cognitive functioning in older adults with attention-deficit/hyperactivity disorder. Int. Psychogeriatr. 2015, 27, 1467–1476. [Google Scholar] [CrossRef] [PubMed]
- Budde, H.; Voelcker-Rehage, C.; Pietraßyk-Kendziorra, S.; Ribeiro, P.; Tidow, G. Acute coordinative exercise improves attentional performance in adolescents. Neurosci. Lett. 2008, 441, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Gentier, I.; Augustijn, M.; Deforche, B.; Tanghe, A.; De Bourdeaudhuij, I.; Lenoir, M.; D’Hondt, E. A comparative study of performance in simple and choice reaction time tasks between obese and healthy-weight children. Res. Dev. Disabil. 2013, 34, 2635–2641. [Google Scholar] [CrossRef] [PubMed]
- Kuang, S. Is reaction time an index of white matter connectivity during training? Cogn. Neurosci. 2017, 8, 126–128. [Google Scholar] [CrossRef] [PubMed]
| All (n = 31) | Male (n = 19) | Female (n = 12) | Mean Diff | 95% CI of Diff | t | ES | p-Value | |
|---|---|---|---|---|---|---|---|---|
| Age (years) | 16.4 ± 0.99 | 16.2 ± 1.08 | 16.7 ± 0.78 | −0.45 | −46.04 to 45.13 | 0.029 | 0.49 | ns |
| Weight (kg) | 69.1 ± 14.5 | 71.4 ± 16.6 | 65.4 ± 9.86 | 6.02 | −39.57 to 51.61 | 0.391 | 0.45 | ns |
| Height (m) | 1.65 ± 0.10 | 1.71 ± 0.08 | 1.55 ± 0.05 | 0.15 | −45.44 to 45.74 | 0.009 | 2.41 | ns |
| BMI (kg/m2) | 25.4 ± 4.45 | 24.4 ± 4.75 | 27.0 ± 3.56 | −2.56 | −48.16 to 43.02 | 0.166 | 0.62 | ns |
| Fat (%) | 25.4 ± 9.35 | 20.6 ± 7.96 | 33.2 ± 5.31 | −12.62 | −58.21 to 32.97 | 0.820 | 1.90 | ns |
| DH | R = 26 (83.8%) L = 5 (16.2%) | R = 15 (78.9%) L = 4 (21.1%) | R = 11 (91.6%) L = 1 (8.4%) | - | - | - | - | - |
| Non-DH | R = 5 (16.2%) L = 26 (83.8%) | R = 4 (21.1%) L = 15 (78.9%) | R = 1 (8.4%) L = 11 (91.6%) | - | - | - | - | - |
| All (n = 31) | Male (n = 19) | Female (n = 12) | Mean Diff | 95% CI of Diff | t | ES | p-Value | |
|---|---|---|---|---|---|---|---|---|
| E-10 (points) | 99.1 ± 24.8 | 95.6 ± 28.4 | 104.7 ± 17.7 | −10.47 | −54.68 to 36.50 | 0.590 | 0.39 | ns |
| Reaction Time Right hand (ms) | 558.9 ± 70.6 | 532.7 ± 50.9 | 600.3 ± 79.4 | −67.18 | −113.2 to −21.99 | 4.392 | 1.04 | 0.0002 |
| Reaction Time Left hand (ms) | 570.4 ± 72.5 | 538.0 ± 51.6 | 621.8 ± 72.7 | −77.37 | −129.4 to −38.20 | 5.446 | 1.35 | 0.0001 |
| Reaction Time Bilateral (ms) | 713.2 ± 85.9 | 679.9 ± 66.0 | 766.0 ± 89.8 | −77.02 | −131.6 to −40.45 | 5.592 | 1.10 | 0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huerta Ojeda, Á.; Lizama Tapia, P.; Pulgar Álvarez, J.; González-Cruz, C.; Yeomans-Cabrera, M.-M.; Contreras Vera, J. Relationship between Attention Capacity and Hand–Eye Reaction Time in Adolescents between 15 and 18 Years of Age. Int. J. Environ. Res. Public Health 2022, 19, 10888. https://doi.org/10.3390/ijerph191710888
Huerta Ojeda Á, Lizama Tapia P, Pulgar Álvarez J, González-Cruz C, Yeomans-Cabrera M-M, Contreras Vera J. Relationship between Attention Capacity and Hand–Eye Reaction Time in Adolescents between 15 and 18 Years of Age. International Journal of Environmental Research and Public Health. 2022; 19(17):10888. https://doi.org/10.3390/ijerph191710888
Chicago/Turabian StyleHuerta Ojeda, Álvaro, Patricio Lizama Tapia, Jaime Pulgar Álvarez, Claudia González-Cruz, María-Mercedes Yeomans-Cabrera, and Juan Contreras Vera. 2022. "Relationship between Attention Capacity and Hand–Eye Reaction Time in Adolescents between 15 and 18 Years of Age" International Journal of Environmental Research and Public Health 19, no. 17: 10888. https://doi.org/10.3390/ijerph191710888
APA StyleHuerta Ojeda, Á., Lizama Tapia, P., Pulgar Álvarez, J., González-Cruz, C., Yeomans-Cabrera, M.-M., & Contreras Vera, J. (2022). Relationship between Attention Capacity and Hand–Eye Reaction Time in Adolescents between 15 and 18 Years of Age. International Journal of Environmental Research and Public Health, 19(17), 10888. https://doi.org/10.3390/ijerph191710888







