Healthy Sleep Every Day Keeps the Doctor Away
Abstract
1. Introduction
2. Inadequate Sleep Health and Obesity
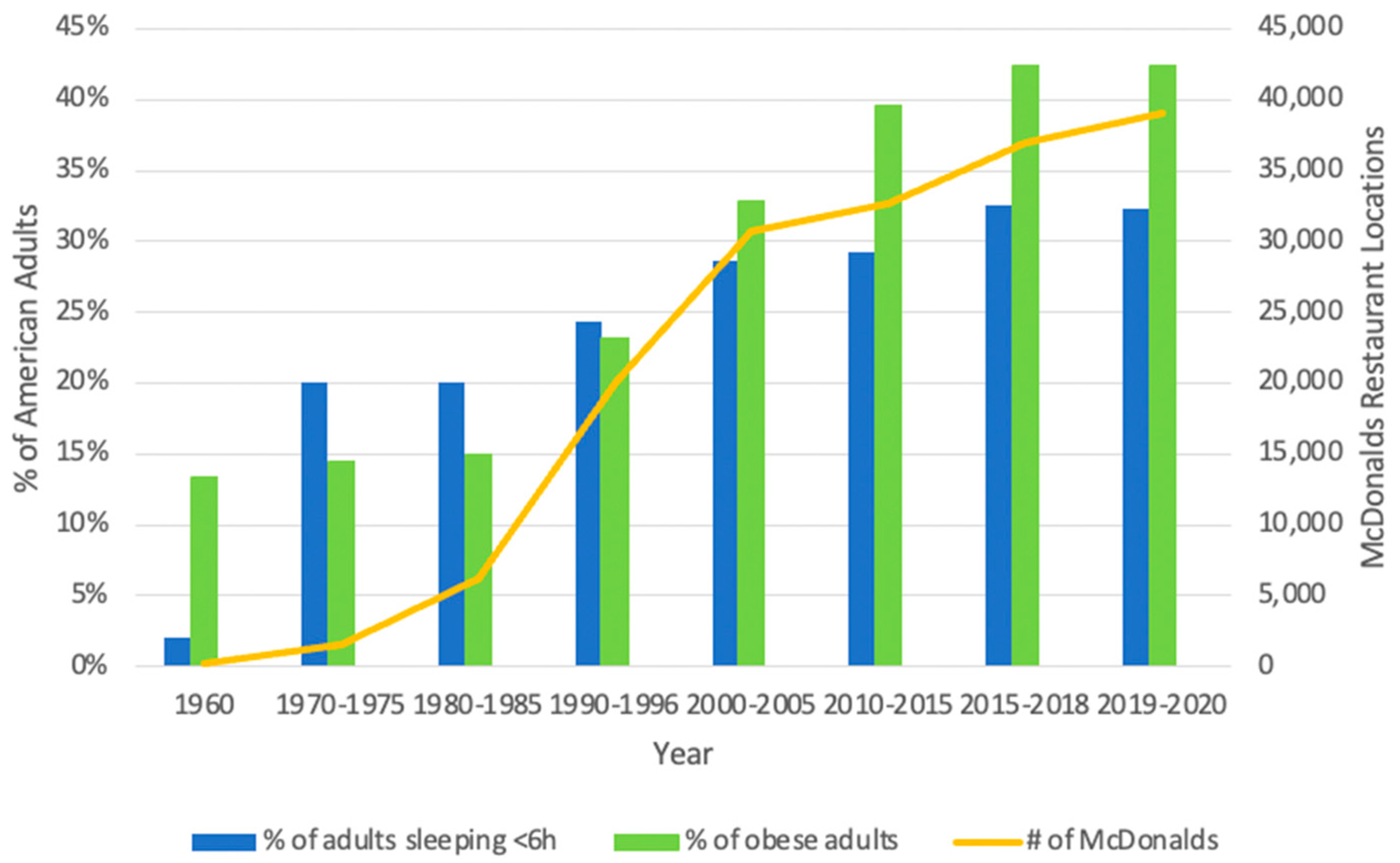
2.1. Trends in Sleep Duration, Obesity, and Dietary Behaviors over Time
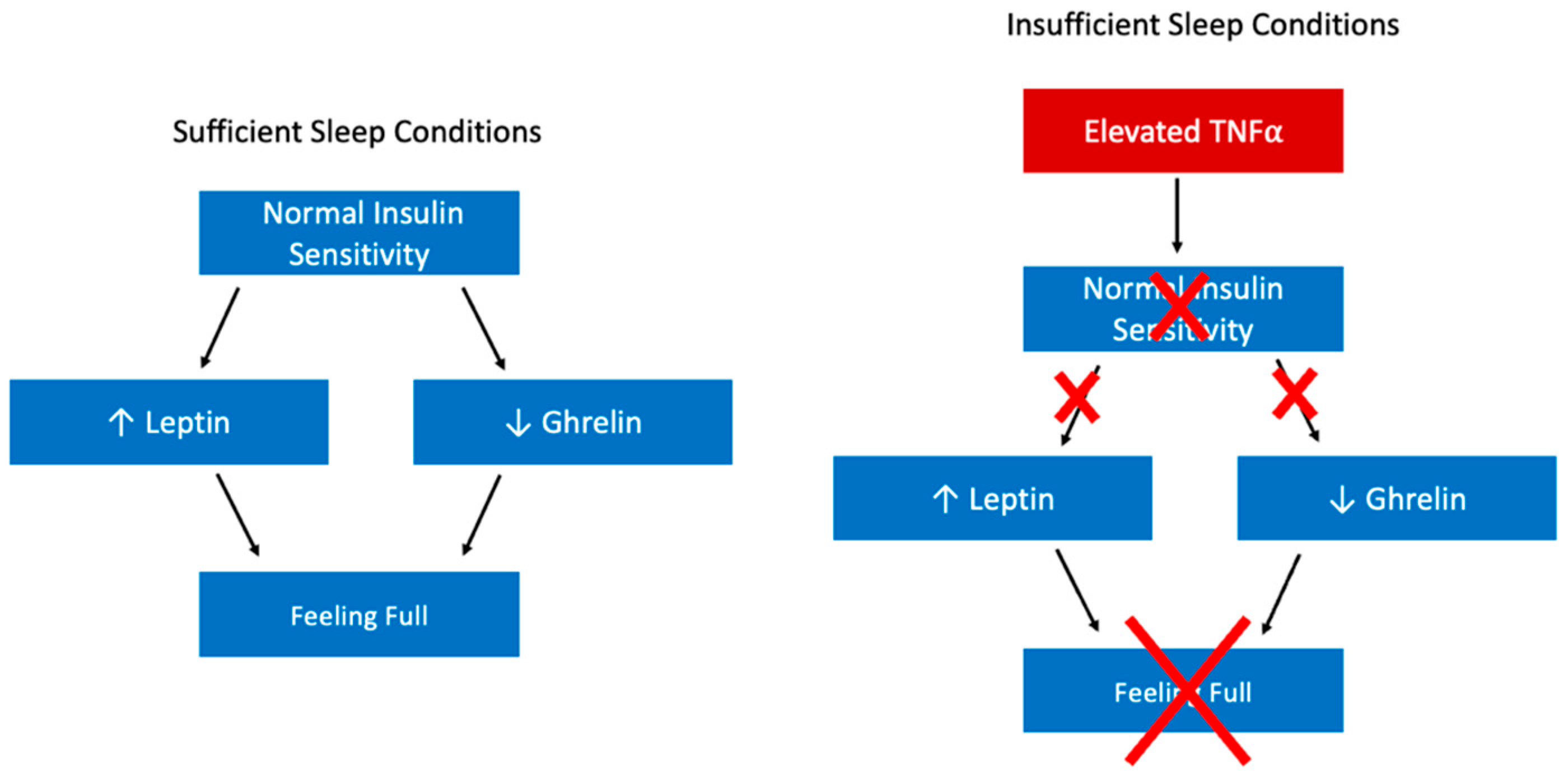
2.2. A Closer Look at the Relationship between Sleep Health and Obesity
2.3. Interaction among Sleep, Nutrition, and Physical Activity
3. Sleep Chronotype and Metabolic Syndrome
3.1. Sleep Fragmentation and Metabolic Syndrome
3.2. Circadian Misalignment and Metabolic Syndrome
3.3. Sleep Duration and Metabolic Syndrome
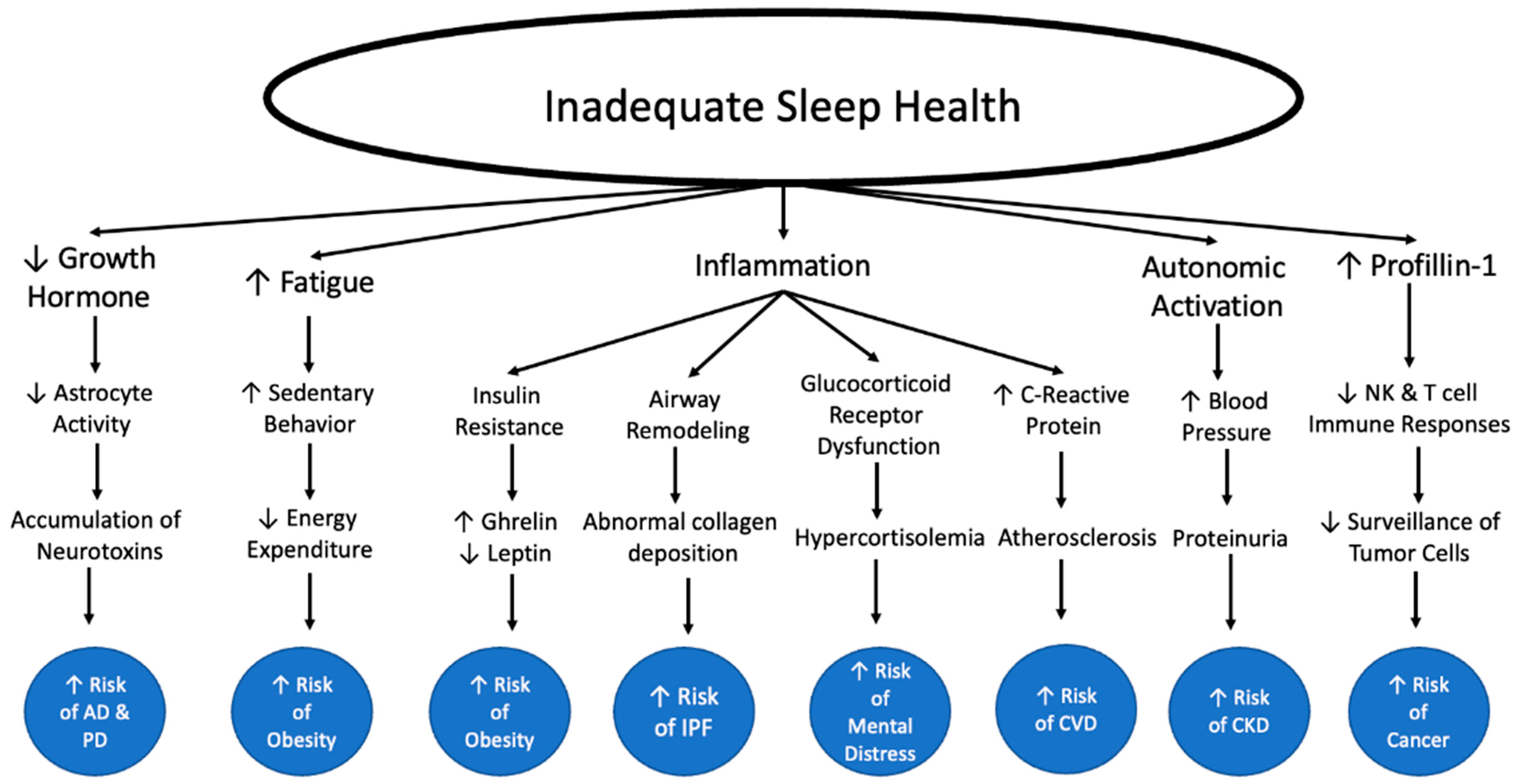
4. Sleep Health and Immunity: Inflammation
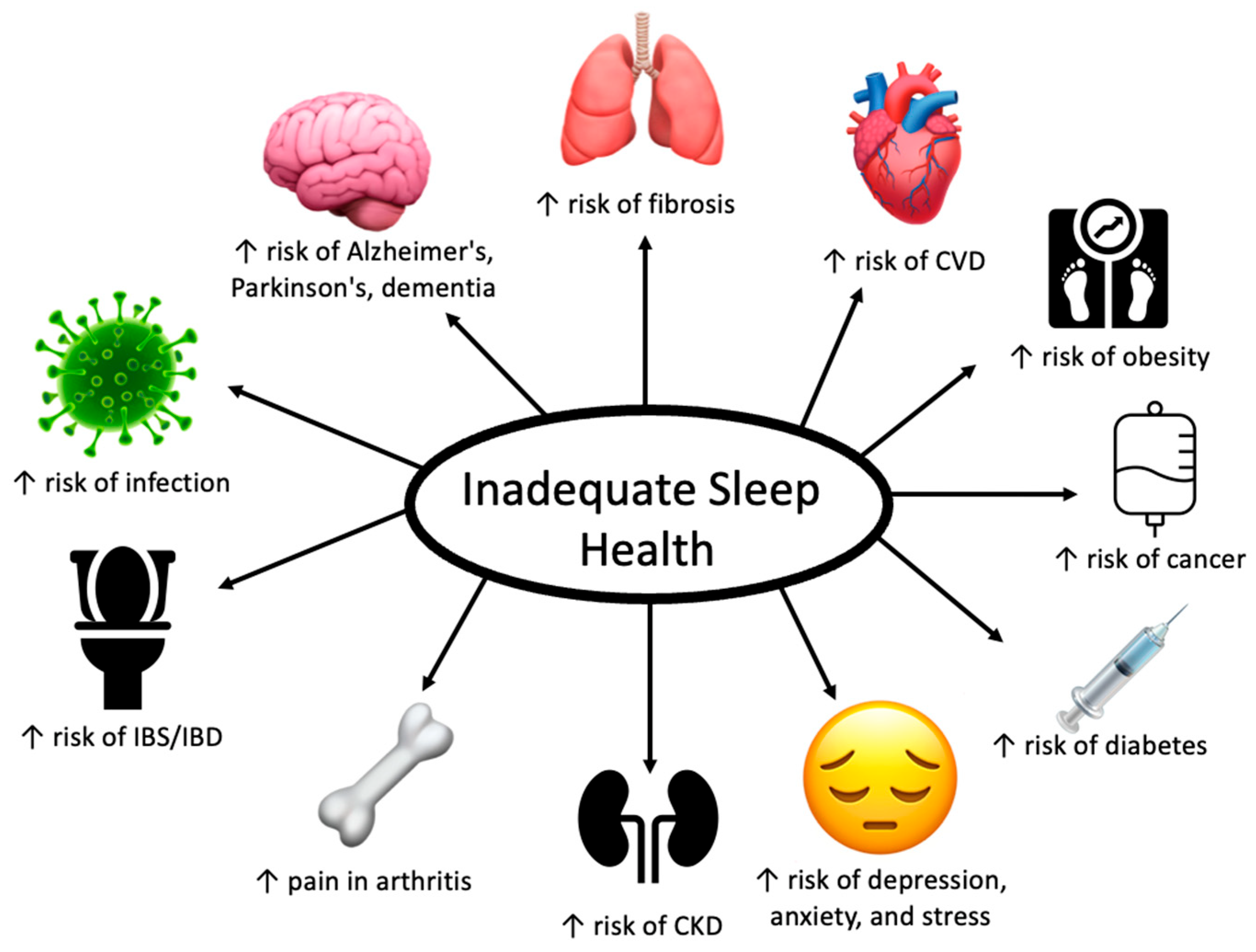
5. Contributions of Inadequate Sleep Health on Other Chronic Diseases
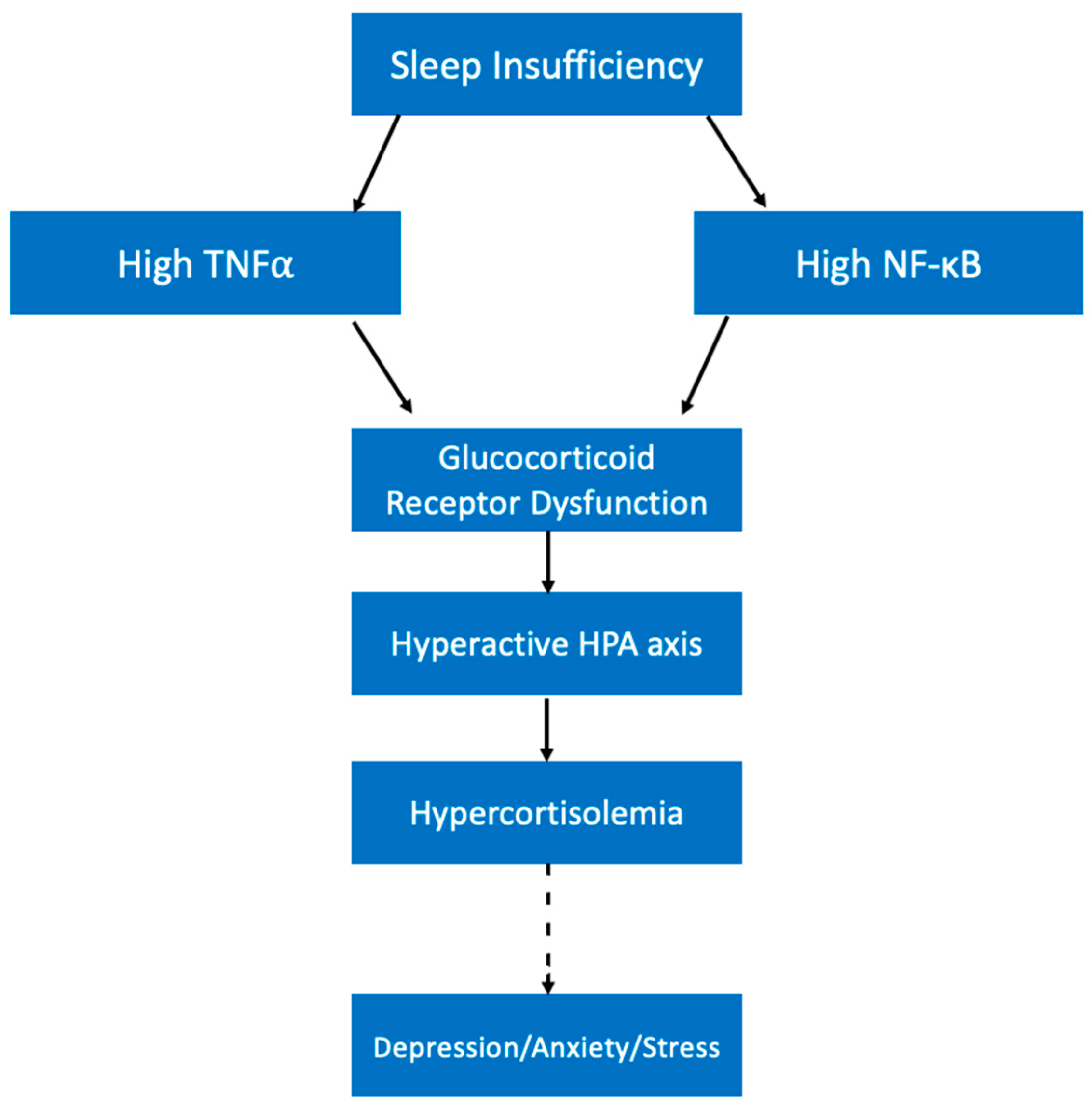
5.1. Sleep Health and Depression/Anxiety/Stress
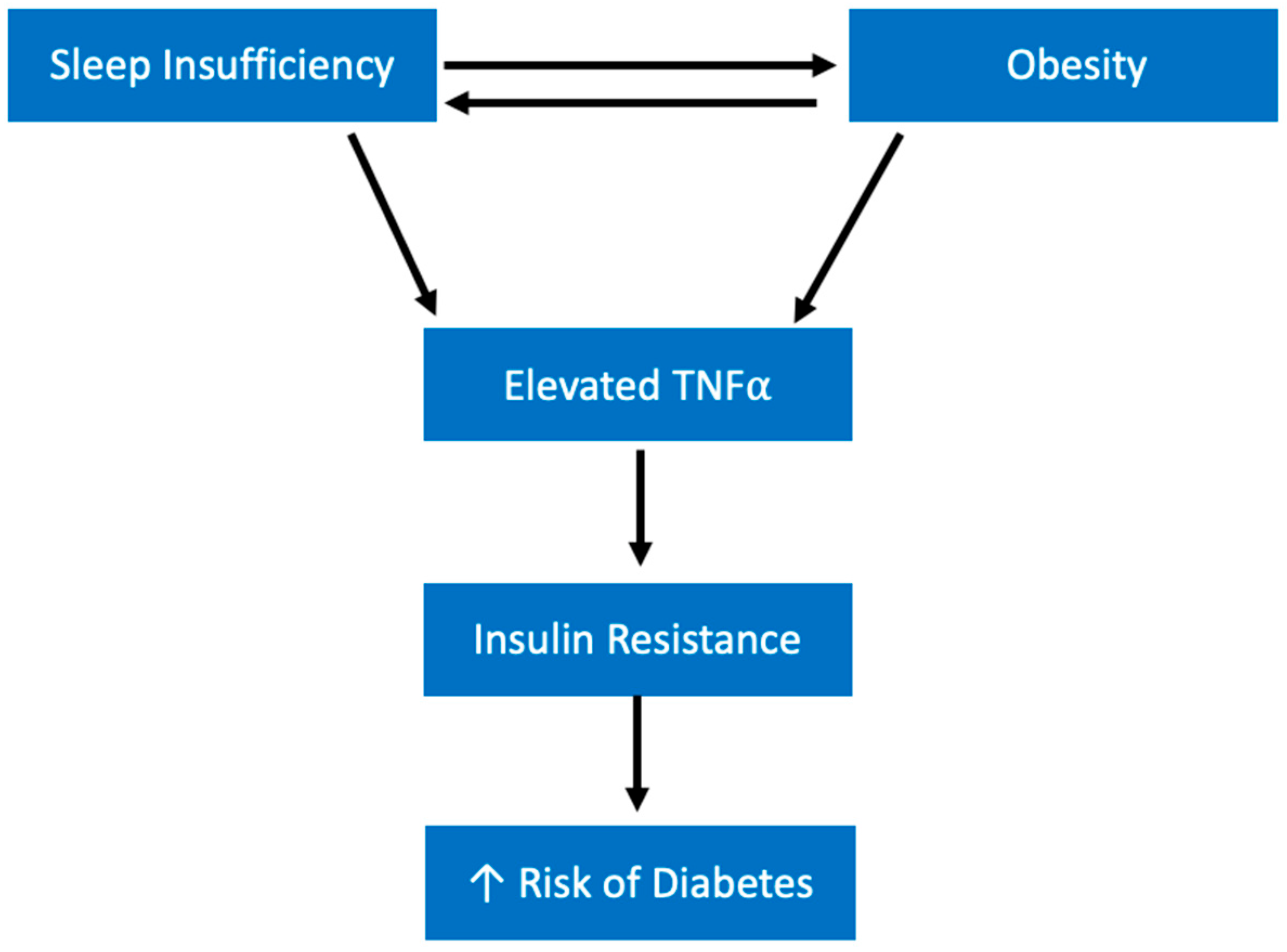
5.2. Diabetes Mellitus
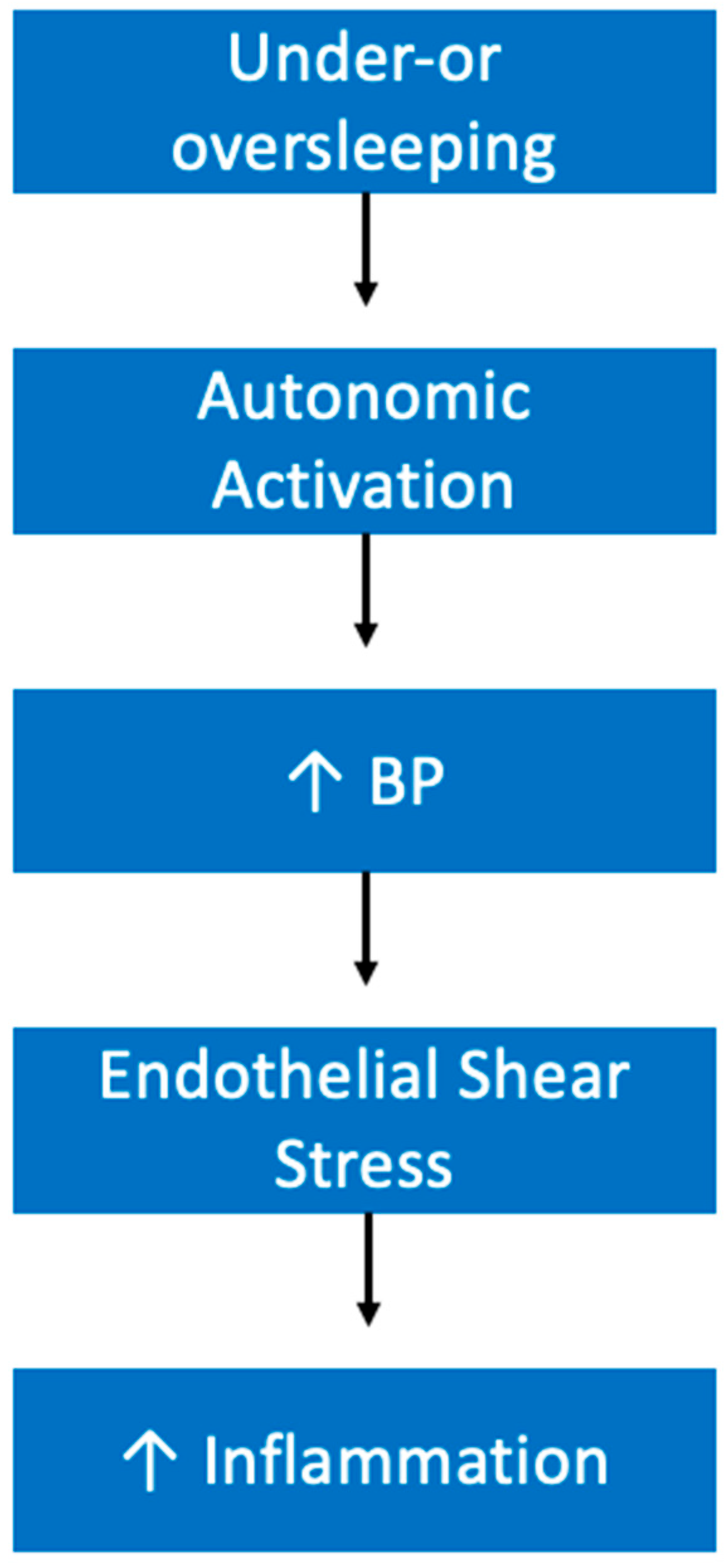
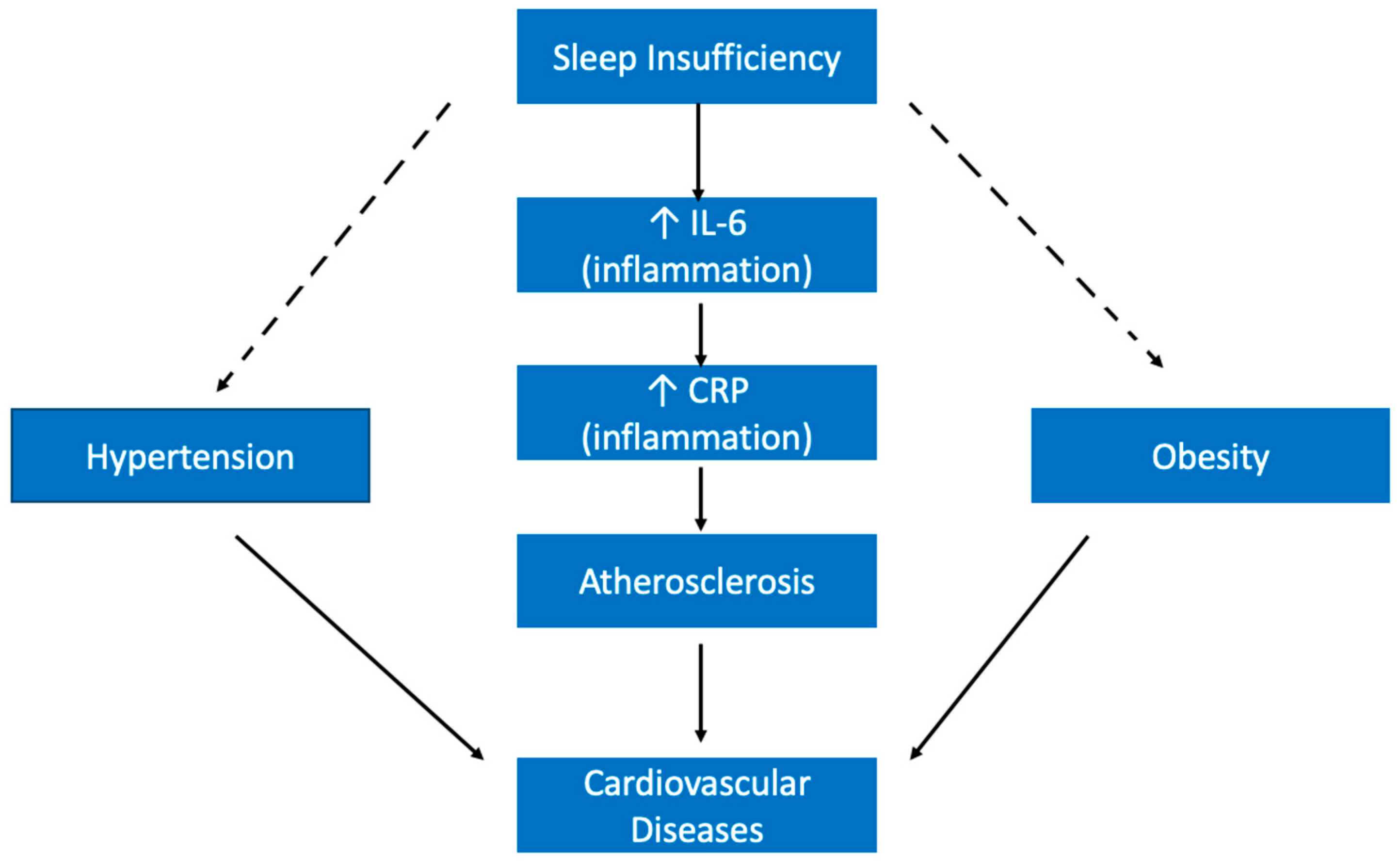
5.3. Hypertension
5.4. Other Cardiovascular Diseases
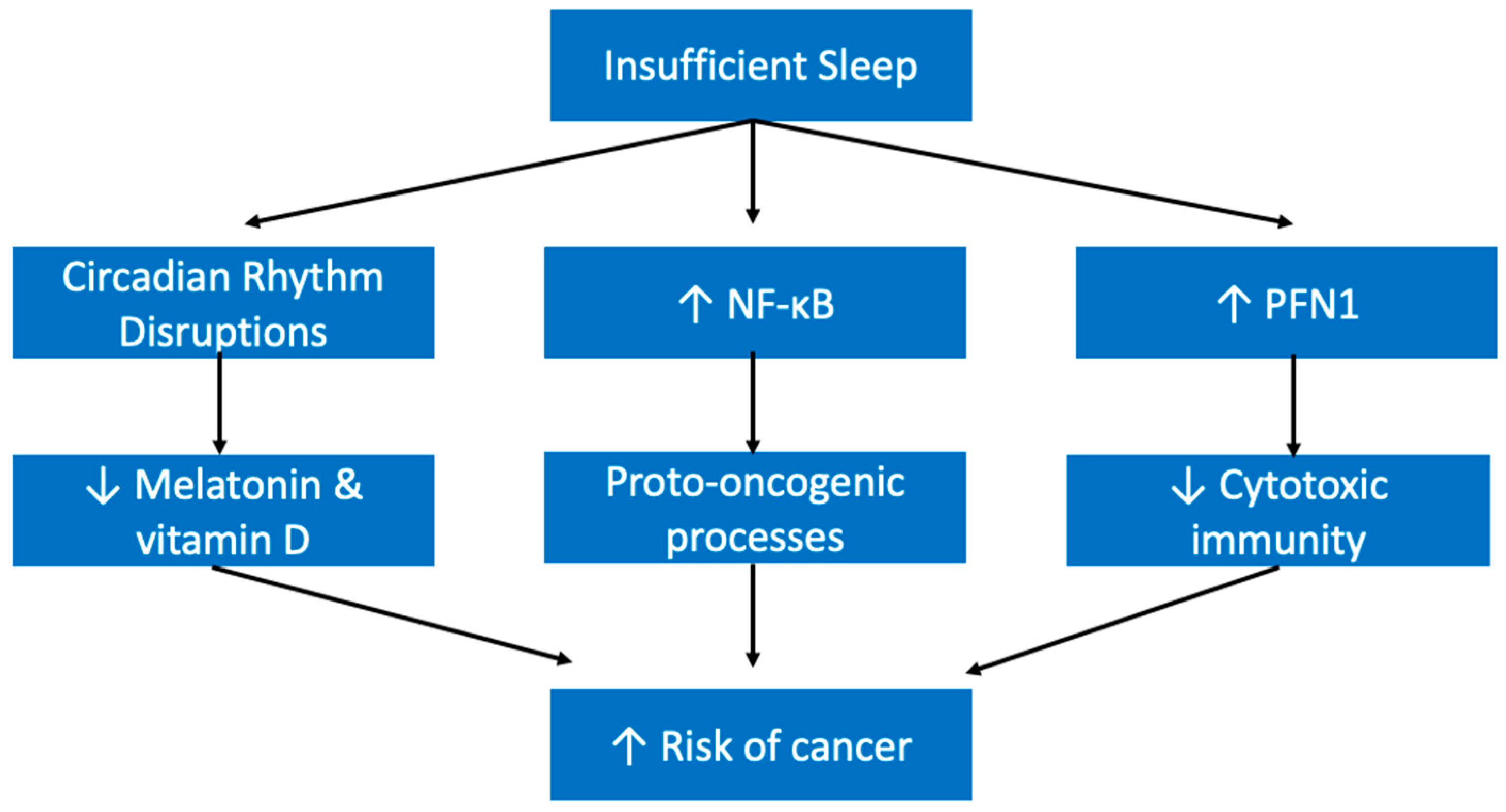
5.5. Cancers
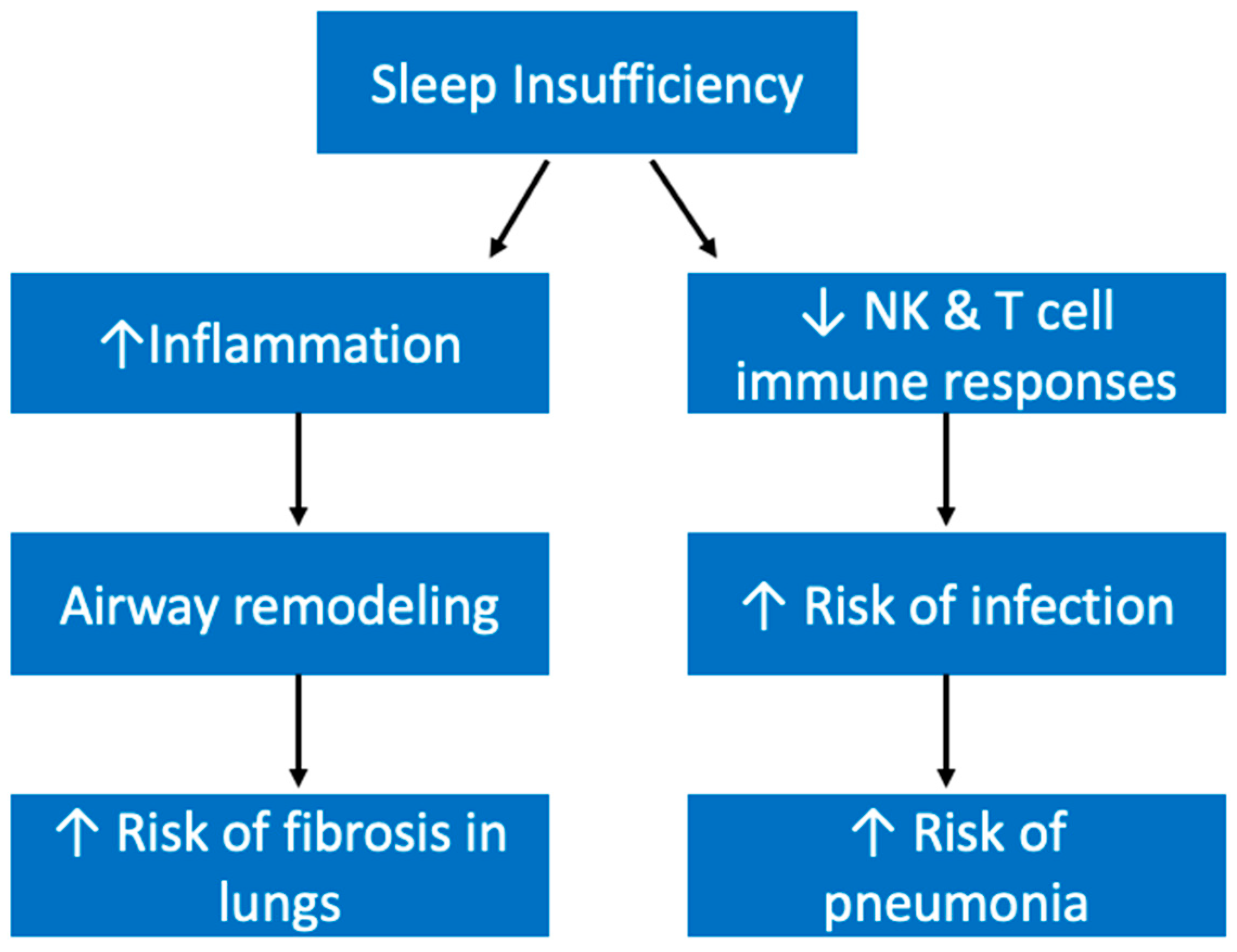
5.6. Pulmonary Diseases
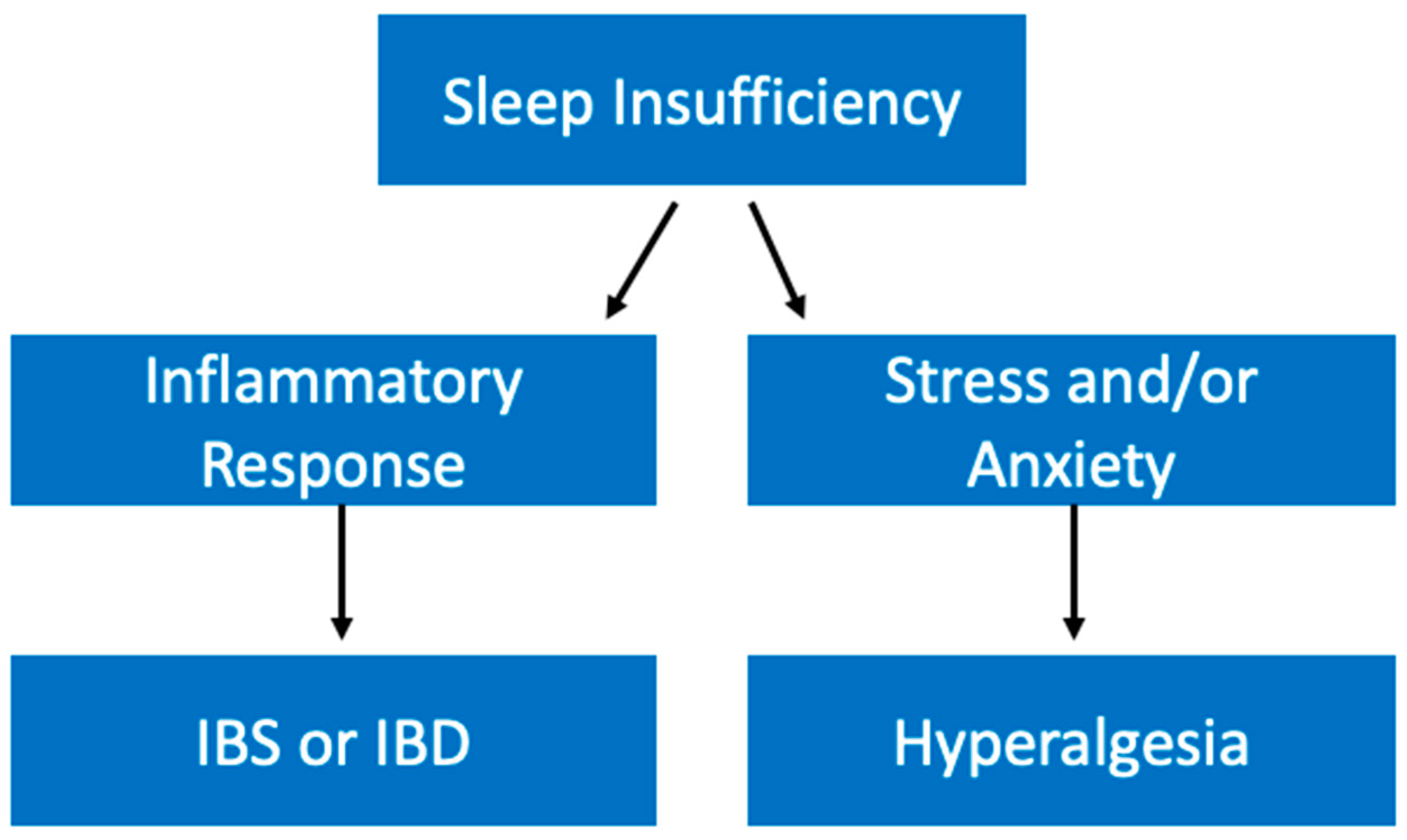
5.7. Gastrointestinal Diseases
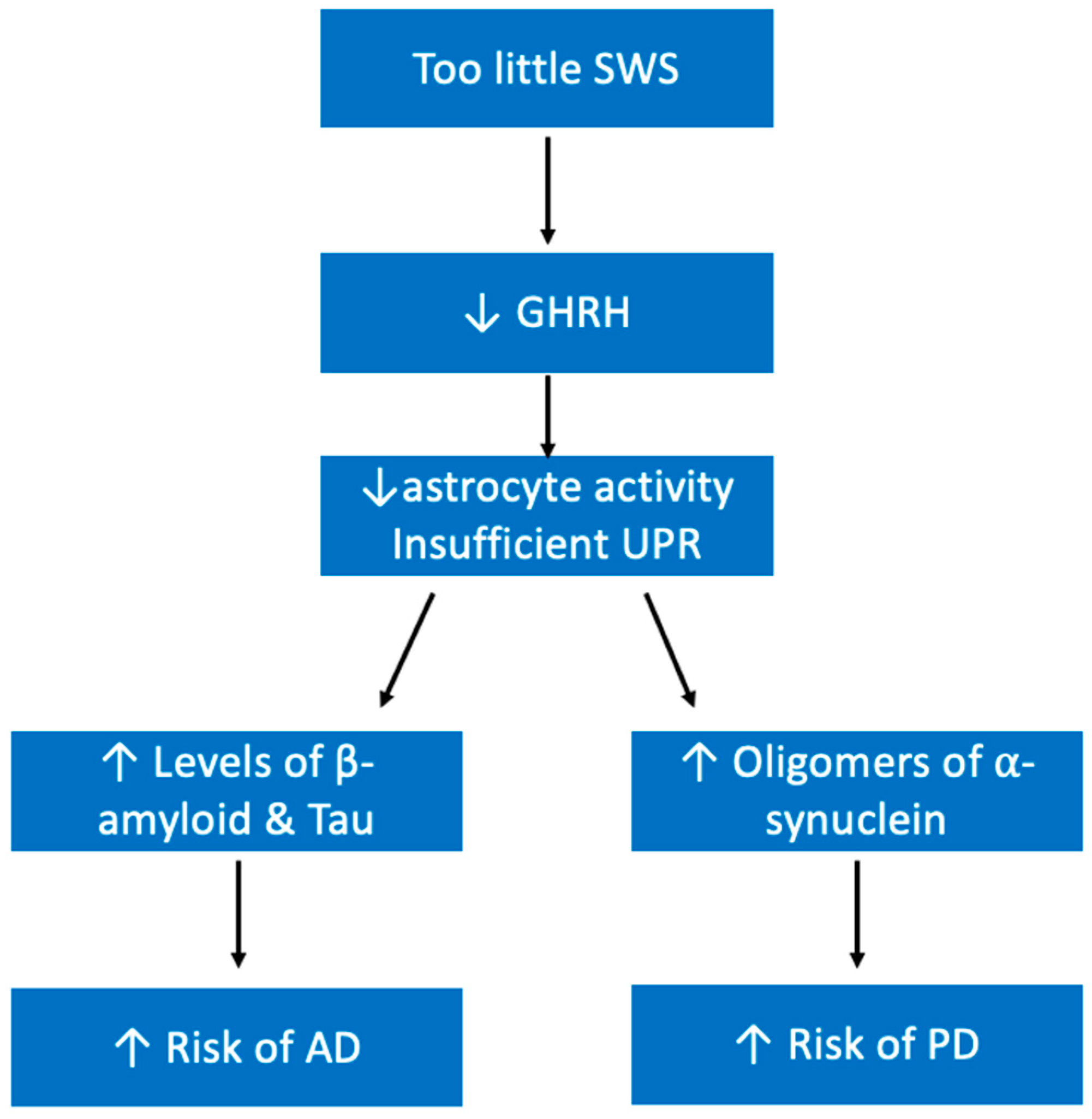
5.8. Neurological Diseases
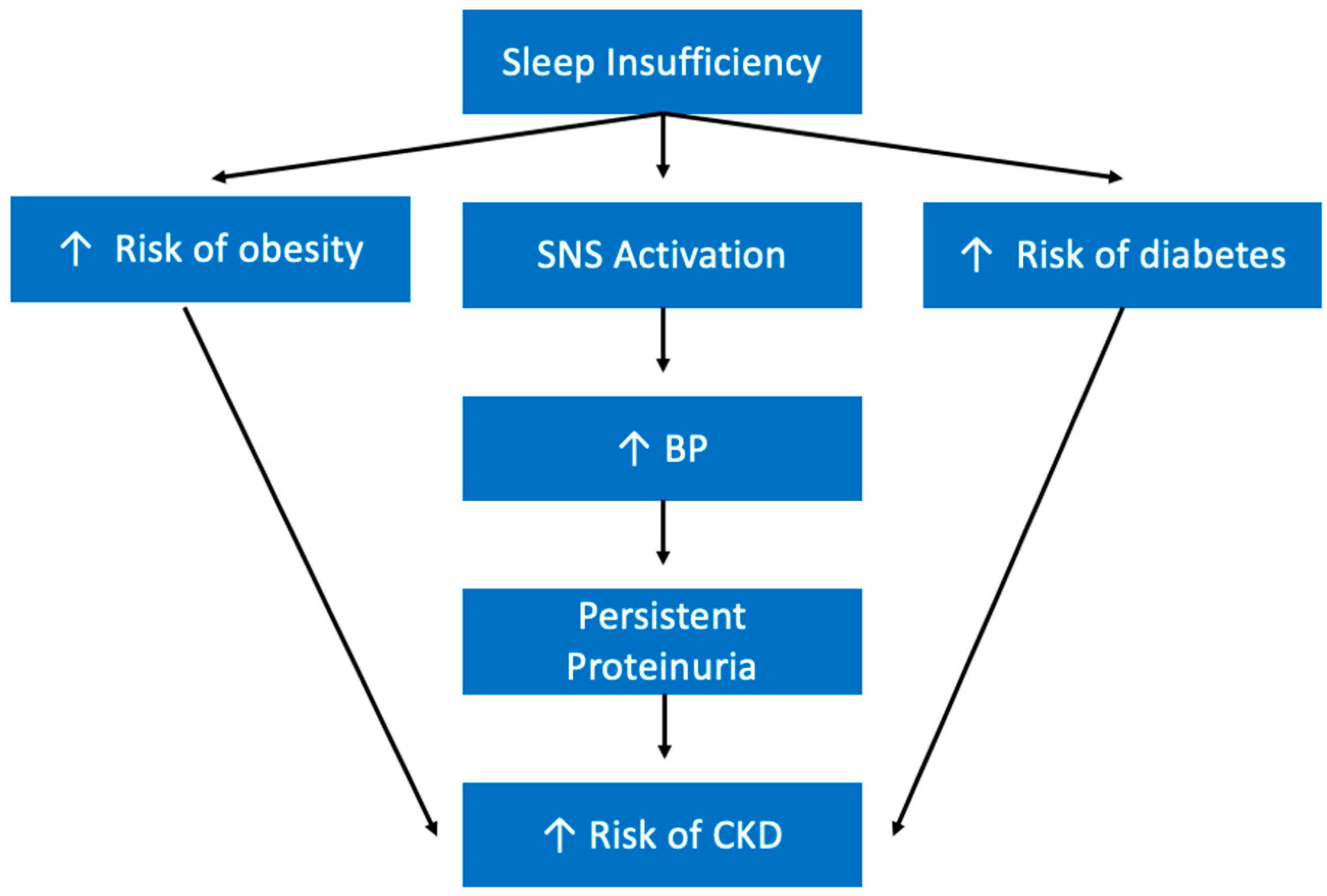
5.9. Renal Diseases
5.10. Musculoskeletal Diseases
5.11. An Additional Consideration
5.12. Sleep Schedule Variations
6. Benefits of Quality Sleep on Chronic Diseases
7. Relationship between Sleep Health and Wellness
8. Measuring Sleep Quality
9. Sleep Education
10. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- CDC QuickStats: Percentage of Currently Employed Adults Aged ≥18 Years Who Reported an Average of ≤6 Hours of Sleep per 24-h Period, by Employment Category—National Health Interview Survey, United States, 2008–2009 and 2017–2018. Available online: https://www.cdc.gov/mmwr/volumes/69/wr/mm6916a5.htm?s_cid=mm6916a5_w (accessed on 1 April 2022).
- Ford, E.S.; Cunninghamd, T.J.; Croft, J.B. Trends in Self-Reported Sleep Duration among US Adults from 1985 to 2012. Sleep 2015, 38, 829–832. [Google Scholar] [CrossRef] [PubMed]
- US Department of Health and Human Services. Your Guide to Healthy Sleep. Available online: https://www.nhlbi.nih.gov/files/docs/public/sleep/healthy_sleep.pdf (accessed on 1 April 2022).
- CDC. Adult Obesity Facts. Available online: https://www.cdc.gov/obesity/data/adult.html (accessed on 1 April 2022).
- Fryar, C.D.; Carroll, M.D.; Ogden, C.L. Prevalence of Overweight, Obesity, and Extreme Obesity among Adults: United States, Trends 1960–1962 through 2009–2010. Available online: https://www.cdc.gov/nchs/data/hestat/obesity_adult_09_10/obesity_adult_09_10.htm (accessed on 1 April 2022).
- Baglioni, C.; Battagliese, G.; Feige, B.; Spiegelhalder, K.; Nissen, C.; Voderholzer, U.; Loombardo, C.; Riemann, D. Insomnia as a Predictor of Depression: A Meta-Analytic Evaluation of Longitudinal Epidemiological Studies. J. Affect. Disord. 2011, 135, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Blackwelder, A.; Hoskins, M.; Huber, L. Effect of Inadequate Sleep on Frequent Mental Distress. Prev. Chronic Dis. 2021, 18, E61. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, L.; Xue, H.; Qu, W. A Review of the Growth of the Fast Food Industry in China and Its Potential Impact on Obesity. Int. J. Environ. Res. Public Health 2016, 13, 1112. [Google Scholar] [CrossRef]
- Mesarwi, O.; Polak, J.; Jun, J.; Polotsky, V.Y. Sleep Disorders and the Development of Insulin Resistance and Obesity. Endocrinol. Metab. Clin. N. Am. 2013, 42, 617–634. [Google Scholar] [CrossRef]
- Patel, S.R.; Hu, F.B. Short Sleep Duration and Weight Gain: A Systematic Review. Obesity 2008, 16, 643–653. [Google Scholar] [CrossRef]
- Zhu, G.; Cassidy, S.; Hiden, H.; Woodman, S.; Trenell, M.; Gunn, D.A.; Catt, M.; Birch-Machin, M.; Anderson, K.N. Exploration of Sleep as a Specific Risk Factor for Poor Metabolic and Mental Health: A UK Biobank Study of 84,404 Participants. Nat. Sci. Sleep 2021, 13, 1903–1912. [Google Scholar] [CrossRef]
- Cawley, J.; Biener, A.; Meyerhoefer, C.; Ding, Y.; Zvenyach, T.; Smolarz, B.G.; Ramasamy, A. Direct Medical Costs of Obesity in the United States and the Most Populous States. J. Manag. Care Spec. Pharm. 2021, 27, 354–366. [Google Scholar] [CrossRef]
- Rihm, J.S.; Menz, M.M.; Schultz, H.; Bruder, L.; Schillbach, L.; Schmid, S.M.; Peters, J. Sleep Deprivation Selectively Upregulates an Amygdala–Hypothalamic Circuit Involved in Food Reward. J. Neurosci. 2019, 39, 888–899. [Google Scholar] [CrossRef]
- Sargent, C.; Zhou, X.; Matthews, R.W.; Darwent, D.; Roach, G.D. Daily Rhythms of Hunger and Satiety in Healthy Men during One Week of Sleep Restriction and Circadian Misalignment. Int. J. Environ. Res. Public Health 2016, 13, 170. [Google Scholar] [CrossRef]
- Nedeltcheva, A.V.; Kilkus, J.M.; Imperial, J.; Kasza, K.; Schoeller, D.A.; Penev, P.D. Sleep Curtailment Is Accompanied by Increased Intake of Calories from Snacks. Am. J. Clin. Nutr. 2009, 89, 126–133. [Google Scholar] [CrossRef]
- Imaki, M.; Hatanaka, Y.; Ogawa, Y.; Yoshida, Y.; Tanada, S. An Epidemiological Study on Relationship between the Hours of Sleep and Life Style Factors in Japanese Factory Workers. J. Physiol. Anthropol. Appl. Hum. Sci. 2002, 21, 115–120. [Google Scholar] [CrossRef]
- Lakerveld, J.; Mackenbach, J.D.; Horvath, E.; Rutters, F.; Compernolle, S.; Bárdos, H.; De Bourdeaudhuij, I.; Charreire, H.; Rutter, H.; Oppert, J.M.; et al. The Relation between Sleep Duration and Sedentary Behaviours in European Adults. Obes. Rev. 2016, 17, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Watenpaugh, D.E. The Role of Sleep Dysfunction in Physical Inactivity and Its Relationship to Obesity. Curr. Sports Med. Rep. 2009, 8, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.R.; Malhotra, A.; White, D.P.; Gottlieb, D.J.; Hu, F.B. Association between Reduced Sleep and Weight Gain in Women. Am. J. Epidemiol. 2006, 154, 947–954. [Google Scholar] [CrossRef] [PubMed]
- Schmid, S.M.; Hallschmid, M.; Juach-Chara, K.; Born, J.; Schultes, B. A Single Night of Sleep Deprivation Increases Ghrelin Levels and Feelings of Hunger in Normal-Weight Healthy Men. J. Sleep Res. 2008, 17, 331–334. [Google Scholar] [CrossRef]
- Taheria, S.; Lin, L.; Austin, D.; Young, T.; Mignot, E. Short Sleep Duration Is Associated with Reduced Leptin, Elevated Ghrelin, and Increased Body Mass Index. PLoS Med. 2004, 1, e62. [Google Scholar] [CrossRef]
- Mullington, J.M.; Chan, J.L.; Van Dongen, H.P.A.; Szuba, M.P.; Samaras, J.; Price, N.J.; Meier-Ewer, H.K.; Dinges, D.F.; Mantzoros, C.S. Sleep Loss Reduces Diurnal Rhythm Amplitude of Leptin in Healthy Men. J. Neuroendocrinol. 2003, 15, 851–854. [Google Scholar] [CrossRef]
- Spiegel, K.; Leproult, R.; L’Hermite-Baleriaux, M.; Copinschi, G.; Penev, P.D.; Cauter, E.V. Leptin Levels Are Dependent on Sleep Duration: Relationships with Sympathovagal Balance, Carbohydrate Regulation, Cortisol, and Thyrotropin. J. Clin. Endocrinol. Metab. 2004, 89, 5762–5771. [Google Scholar] [CrossRef]
- Batterham, R.L.; Cowley, M.A.; Small, C.J.; Herzog, H.; Cohen, M.A.; Dakin, C.L.; Wren, A.M.; Brynes, A.E.; Low, M.J.; Ghatei, M.A.; et al. Hormone PYY(3–36) Physiologically Inhibits Food Intake. Nature 2002, 418, 650–654. [Google Scholar] [CrossRef]
- Hibi, M.; Kubota, C.; Mizuno, T.; Aritake, S.; Mitsui, Y.; Katashima, M.; Uchida, S. Effect of Shortened Sleep on Energy Expenditure, Core Body Temperature, and Appetite: A Human Randomised Crossover Trial. Sci. Rep. 2017, 7, 39640. [Google Scholar] [CrossRef] [PubMed]
- Holst, J. The Physiology of Glucagon-like Peptide 1. Physiol. Rev. 2007, 87, 1409–1439. [Google Scholar] [CrossRef] [PubMed]
- Flint, A.; Raben, A.; Astrup, A.; Holst, J. Glucagon-Like Peptide 1 Promotes Satiety and Suppresses Energy Intake in Humans. J. Clin. Investig. 1998, 101, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Benedict, C.; Barclay, J.; Ott, V.; Oster, H.; Hallschmid, M. Acute Sleep Deprivation Delays the Glucagon-like Peptide 1 Peak Response to Breakfast in Healthy Men. Nutr. Diabetes 2013, 3, e78. [Google Scholar] [CrossRef]
- Broussard, J.L.; Ehrmann, D.A.; Cauter, E.V.; Tasali, E.; Brady, M.J. Impaired Insulin Signaling in Human Adipocytes after Experimental Sleep Restriction: A Randomized, Crossover Study. Ann. Intern. Med. 2012, 157, 549–557. [Google Scholar] [CrossRef]
- Donga, E.; van Dijk, M.; van Dijk, J.G.; Biermasz, N.R.; Lammers, G.-J.; van Kralingen, K.W.; Corssmit, E.P.M.; Romjin, J.A. A Single Night of Partial Sleep Deprivation Induces Insulin Resistance in Multiple Metabolic Pathways in Healthy Subjects. J. Endocrinol. Metab. 2010, 95, 2963–2968. [Google Scholar] [CrossRef]
- Spiegel, K.; Leproult, R.; Van Cauter, E. Impact of Sleep Debt on Metabolic and Endocrine Function. Lancet 1999, 354, 1435–1439. [Google Scholar] [CrossRef]
- Reynolds, A.C.; Dorrian, J.; Liu, P.Y.; Van Dongen, H.P.A.; Wittert, G.A.; Harmer, L.J.; Banks, S. Impact of Five Nights of Sleep Restriction on Glucose Metabolism, Leptin and Testosterone in Young Adult Men. PLoS ONE 2012, 7, e41218. [Google Scholar] [CrossRef]
- Broussard, J.L.; Wroblewski, K.; Kilkus, J.M. Two Nights of Recovery Sleep Reverses the Effects of Short-Term Sleep Restriction on Diabetes Risk. Diabetes Care 2016, 39, e40–e41. [Google Scholar] [CrossRef]
- Leproult, R.; Deliens, G.; Gilson, M.; Peigneux, P. Beneficial Impact of Sleep Extension on Fasting Insulin Sensitivity in Adults with Habitual Sleep Restriction. Sleep 2015, 38, 707–715. [Google Scholar] [CrossRef]
- Moreno-Aliaga, M.J.; Stanhope, K.L.; Havel, P.J. Transcriptional Regulation of the Leptin Promoter by Insulin-Stimulated Glucose Metabolism in 3T3-L1 Adipocytes. Biochem. Biophys. Res. Commun. 2001, 282, 544–548. [Google Scholar] [CrossRef] [PubMed]
- Tsubai, T.; Noda, Y.; Ito, K.; Makoto, N.; Seino, Y.; Oiso, Y.; Hamada, Y. Insulin Elevates Leptin Secretion and MRNA Levels via Cyclic AMP in 3T3-L1 Adipocytes Deprived of Glucose. Heliyon 2016, 2, e00194. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zeigerer, A.; Rodeheffer, M.S.; McGraw, T.E.; Friedman, J.M. Insulin Regulates Leptin Secretion from 3T3–L1 Adipocytes by a PI 3 Kinase Independent Mechanism. Exp. Cell Res. 2008, 314, 2249–2256. [Google Scholar] [CrossRef] [PubMed]
- Cummings, D.; Purnell, J.Q.; Frayo, R.S.; Schmidova, K.; Wisse, B.E.; Weigle, D.S. A Preprandial Rise in Plasma Ghrelin Levels Suggests a Role in Meal Initiation in Humans. Diabetes 2001, 50, 1714–1719. [Google Scholar] [CrossRef]
- Chabot, F.; Caron, A.; Laplante, M.; St-Pierre, D.H. Interrelationships between Ghrelin, Insulin and Glucose Homeostasis: Physiological Relevance. World J. Diabetes 2015, 5, 328–341. [Google Scholar] [CrossRef]
- Atwater, A.Q.; Immergluck, L.C.; Davidson, A.J.; Castanon-Cervantes, O. Shift Work Predicts Increases in Lipopolysaccharide-Binding Protein, Interleukin-10, and Leukocyte Counts in a Cross-Sectional Study of Healthy Volunteers Carrying Low-Grade Systemic Inflammation. Int. J. Environ. Res. Public Health 2021, 18, 131518. [Google Scholar] [CrossRef]
- Chennaoui, M.; Sauvet, F.; Drogou, C.; Van Beers, P.; Langrume, C.; Guillard, M.; Gourby, B.; Bourrilhon, C.; Florence, G.; Gomez-Merrino, D. Effect of One Night of Sleep Loss on Changes in Tumor Necrosis Factor Alpha (TNF-α) Levels in Healthy Men. Cytokine 2011, 56, 318–324. [Google Scholar] [CrossRef]
- Hotamisligil, G.S.; Shargill, N.S.; Spiegelman, B.M. Adipose Expression of Tumor Necrosis Factor-α: Direct Role in Obesity-Linked Insulin Resistance. Science 1993, 259, 87–91. [Google Scholar] [CrossRef]
- Hotamisligil, G.S.; Arner, P.; Caro, J.F.; Atkinson, R.L.; Spiegelman, B.M. Increased Adipose Tissue Expression of Tumor Necrosis Factor-Alpha in Human Obesity and Insulin Resistance. J. Clin. Investig. 1995, 95, 2409–2415. [Google Scholar] [CrossRef]
- Kern, P.A. Potential Role of TNFalpha and Lipoprotein Lipase as Candidate Genes for Obesity. J. Nutr. 1997, 127, 1917–1922. [Google Scholar] [CrossRef]
- Uysal, K.T.; Wiesbrock, S.M.; Marino, M.W.; Hotamisligil, G.S. Protection from Obesity-Induced Insulin Resistance in Mice Lacking TNF-α Function. Nature 1997, 389, 610–614. [Google Scholar] [CrossRef] [PubMed]
- Plomgaard, P.; Bouzakri, K.; Krogh-Madsen, R.; Mittendorfer, B.; Zierath, J.R.; Pedersen, B.K. Tumor Necrosis Factor-α Induces Skeletal Muscle Insulin Resistance in Healthy Human Subjects via Inhibition of Akt Substrate 160 Phosphorylation. Diabetes 2005, 54, 2939–2945. [Google Scholar] [CrossRef] [PubMed]
- Benedict, C.; Brooks, S.J.; O’Daly, O.G.; Almen, M.S.; Morell, A.; Aberg, K.; Gingnell, M.; Schultes, B.; Hallschmid, M.; Broman, J.-E.; et al. Acute Sleep Deprivation Enhances the Brain’s Response to Hedonic Food Stimuli: An FMRI Study. J. Clin. Endocrinol. Metab. 2012, 97, E443–E447. [Google Scholar] [CrossRef] [PubMed]
- Greer, S.M.; Goldstein, A.N.; Walker, M.P. The Impact of Sleep Deprivation on Food Desire in the Human Brain. Nat. Commun. 2013, 4, 2259. [Google Scholar] [CrossRef]
- Demos, K.; Sweet, L.; Hart, C.; McCaffery, J.; Williams, S.; Mailloux, K.; Trautvetter, J.; Owens, M.; Wing, B. The Effects of Experimental Manipulation of Sleep Duration on Neural Response to Food Cues. Sleep 2017, 40, zsx125. [Google Scholar] [CrossRef]
- Goldstone, A.P.; Prechtl, C.G.; Scholtz, S.; Miras, A.D.; Chhina, N.; Durighel, G.; Deliran, S.S.; Beckmann, C.; Ghatei, M.A.; Ashby, D.R.; et al. Ghrelin Mimics Fasting to Enhance Human Hedonic, Orbitofrontal Cortex, and Hippocampal Responses to Food. Am. J. Clin. Nutr. 2014, 99, 1319–1330. [Google Scholar] [CrossRef]
- Solinas, M.; Goldberg, S.; Piomelli, D. The Endocannabinoid System in Brain Reward Processes. Br. J. Pharmacol. 2008, 154, 369–383. [Google Scholar] [CrossRef]
- Kirkham, T. Endocannabinoids in the Regulation of Appetite and Body Weight. Behav. Pharmacol. 2005, 16, 297–313. [Google Scholar] [CrossRef]
- Scheer, F.A.J.L. Hungry for Sleep: A Role for Endocannabinoids? Sleep 2016, 39, 495–496. [Google Scholar] [CrossRef][Green Version]
- Chaput, J.-P. Sleep Patterns, Diet Quality and Energy Balance. Physiol. Behav. 2014, 134, 86–91. [Google Scholar] [CrossRef]
- St-Onge, M.-P. The Role of Sleep Duration in the Regulation of Energy Balance: Effects on Energy Intakes and Expenditure. J. Clin. Sleep Med. 2013, 9, 73–80. [Google Scholar] [CrossRef]
- Pearson, N.J.; Johnson, L.L.; Nahin, R.L. Insomnia, Trouble Sleeping, and Complementary and Alternative Medicine Analysis of the 2002 National Health Interview Survey Data. Arch. Intern. Med. 2006, 166, 1775–1782. [Google Scholar] [CrossRef]
- Malhotra, A.; White, D.P. Obstructive Sleep Apnoea. Lancet 2002, 360, 237–245. [Google Scholar] [CrossRef]
- Rodrigues, G.D.; Fiorelli, E.M.; Furlan, L.; Montano, N.; Tobaldini, E. Obesity and Sleep Disturbances: The “Chicken of the Egg” Question. Eur. J. Intern. Med. 2021, 92, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Young, T.; Skatrud, J.; Peppard, P.E. Risk Factors for Obstructive Sleep Apnea in Adults. JAMA 2005, 291, 2013–2016. [Google Scholar] [CrossRef] [PubMed]
- Yannakoulia, M.; Anastasiou, C.A.; Karfopoulou, E.; Pehlivanidis, A.; Panagiotakos, D.B.; Vgontzas, A. Sleep Quality Is Associated with Weight Loss Maintenance Status: The MedWeight Study. Sleep Med. 2017, 34, 242–245. [Google Scholar] [CrossRef]
- Vitale, K.; Owens, R.; Hopkins, S.; Malhotra, A. Sleep Hygiene for Optimizing Recovery in Athletes: Review and Recommendations. Int. J. Sports Med. 2019, 40, 535–543. [Google Scholar] [CrossRef]
- Huang, K.; Ihm, J. Sleep and Injury Risk. Curr. Sports Med. Rep. 2021, 20, 286–290. [Google Scholar] [CrossRef]
- Dolezal, B.; Neufeld, E.; Boland, D.; Martin, J.; Cooper, C. Interrelationship between Sleep and Exercise: A Systematic Review. Adb. Prev. Med. 2017, 2018, 1364387. [Google Scholar] [CrossRef]
- Kredlow, M.; Capozzoli, M.; Hearon, B.; Calkins, A.; Otto, M. The Effects of Physical Activity on Sleep: A Meta-Analytic Review. J. Behav. Med. 2015, 38, 427–449. [Google Scholar] [CrossRef]
- Beck, K.; Thomson, J.; Swift, R.; von Hurst, P. Role of Nutrition in Performance Enhancement and Postexercise Recovery. Open Access J. Sports Med. 2015, 6, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Azzolino, D.; Arosio, B.; Marzetti, E.; Calvani, R.; Cesari, M. Nutritional Status as a Mediator of Fatigue and Its Underlying Mechanisms in Older People. Nutrients 2020, 12, 444. [Google Scholar] [CrossRef] [PubMed]
- Zuraikat, F.; Wood, R.; Barragán, R.; St-Onge, M.-P. Sleep and Diet: Mounting Evidence of a Cyclical Relationship. Annu. Rev. Nutr. 2021, 41, 309–332. [Google Scholar] [CrossRef] [PubMed]
- Saidi, O.; Rochette, E.; Bovet, M.; Merlin, E.; Duché, P. Acute Intense Exercise Improves Sleep and Decreases Next Morning Consumption of Energy-Dense Food in Adolescent Girls with Obesity and Evening Chronotype. Pediatr. Obes. 2020, 15, e12613. [Google Scholar] [CrossRef]
- Smiley, A.; Wolter, S.; Nissan, D. Mechanisms of Association of Sleep and Metabolic Syndrome. J. Med.-Clin. Res. Rev. 2019, 3, 1–9. [Google Scholar] [CrossRef]
- Song, Q.; Liu, X.; Zhou, W.; Wang, X.; Wu, S. Changes in Sleep Duration and Risk of Metabolic Syndrome: The Kailuan Prospective Study. Sci. Rep. 2016, 6, 36861. [Google Scholar] [CrossRef]
- Reiter, A.M.; Roach, G.D.; Sargent, C. The Night before Night Shift: Chronotype Impacts Total Sleep and Rapid Eye Movement Sleep during a Strategically Delayed Sleep. J. Sleep Res. 2022. [Google Scholar] [CrossRef]
- Boudreau, P.; Yeh, W.-H.; Dumont, G.A.; Boivin, D.B. Circadian Variation of Heart Rate Variability across Sleep Stages. Sleep 2013, 36, 1919–1928. [Google Scholar] [CrossRef]
- Harfmann, B.; Swalve, N.; Mitrzyk, J.; Montoye, A.H.K. Effect of Chronotype on Sleep Quality in a Laboratory Setting. Int. J. Exerc. Sci. 2020, 13, 1283–1294. [Google Scholar]
- Tassi, P.; Muzet, A. Sleep Inertia. Sleep Med. Rev. 2000, 4, 341–353. [Google Scholar] [CrossRef]
- Nauha, L.; Jurvelin, H.; Ala-Marsula, L.; Niemela, M.; Jamsa, T.; Kangas, M.; Korpelainen, R. Chronotypes and Objectively Measured Physical Activity and Sedentary Time at Midlife. Scand. J. Med. Sci. Sports 2020, 30, 1930–1938. [Google Scholar] [CrossRef] [PubMed]
- De Amicis, R.; Galasso, L.; Leone, A.; Vignati, L.; De Carlo, G.; Foppiani, A.; Montaruli, A.; Roveda, E.; Cé, E.; Eposito, F. Is Abdominal Fat Distribution Associated with Chronotype in Adults Independently of Lifestyle Factors? Nutrients 2020, 12, 592. [Google Scholar] [CrossRef]
- Kuo, T.; McQueen, A.; Chen, T.-C.; Wang, J.-C. Regulation of Glucose Homeostasis by Glucocorticoids. Adv. Exp. Med. Biol. 2015, 872, 99–126. [Google Scholar] [CrossRef] [PubMed]
- Sapolsky, R.M.; Romero, L.M.; Munck, A.U. How Do Glucocorticoids Influence Stress Responses? Integrating Permissive, Suppressive, Stimulatory, and Preparative Actions. Endocr. Rev. 2000, 21, 55–89. [Google Scholar] [CrossRef] [PubMed]
- Sassin, J.F.; Parker, D.C.; Mace, J.W.; Gotlin, R.W.; Johnson, L.C.; Rossman, L.G. Human Growth Hormone Release: Relation to Slow-Wave Sleep and Sleep-Walking Cycles. Science 1969, 165, 513–515. [Google Scholar] [CrossRef]
- Lavrnja, I.; Ajdzanovic, V.; Trifunovic, S.; Savic, D.; Milosevic, V.; Stojiljkovic, M.; Pekovic, S. Cortical Ablation Induces Time-Dependent Changes in Rat Pituitary Somatotrophs and Upregulates Growth Hormone Receptor Expression in the Injured Cortex. J. Neurosci. 2014, 92, 1338–1349. [Google Scholar] [CrossRef]
- Mander, B.A.; Marks, S.M.; Vogel, J.W.; Rao, V.; Lu, B.; Saletin, J.M.; Ancoli-Israel, S.; Jagust, W.J.; Walker, M.P. β-Amyloid Disrupts Human NREM Slow Waves and Related Hippocampus-Dependent Memory Consolidation. Nat. Neurosci. 2015, 18, 1051–1057. [Google Scholar] [CrossRef]
- Pedrolli, F.; Banfi, D.; Gesmundo, I.; Guglielmotto, M.; Tamagno, E.; Schally, A.; Ghigo, E.; Granata, R. Growth Hormone-Releasing Hormone (GHRH) Promotes Survival and Proliferation of Neural Stem Cells and Reduces Amyloid-β-Induced Toxicity. Pituit. Neuroendocrinol. 2022, 81, 436. [Google Scholar] [CrossRef]
- Lucey, B.P.; Hicks, T.J.; McLelan, J.S.; Toedebusch, C.D.; Boyd, J.; Elbert, D.L.; Patterson, B.W.; Baty, J.; Morris, J.C.; Ovod, V.; et al. Effect of Sleep on Overnight Cerebrospinal Fluid Amyloid β Kinetics. Ann. Neurol. 2018, 83, 197–204. [Google Scholar] [CrossRef]
- Walker, M.P. Cognitive Consequences of Sleep and Sleep Loss. Sleep Med. 2008, 9, S29–S34. [Google Scholar] [CrossRef]
- Walker, M.P. The Role of Slow Wave Sleep in Memory Processing. J. Clin. Sleep Med. 2009, 5, S20–S26. [Google Scholar] [CrossRef] [PubMed]
- Thapa, N.; Kim, B.; Yang, J.-G.; Park, H.-J.; Jang, M.; Son, H.-E.; Kim, G.; Park, H. The Relationship between Chronotype, Physical Activity and the Estimated Risk of Dementia in Community-Dwelling Older Adults. Int. J. Environ. Res. Public Health 2020, 17, 3701. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Liu, Y.; Xiang, Y.; Lin, M.; Gao, J. Ghrelin Protects Human Umbilical Vein Endothelial Cells against Advanced Glycation End Products-Induced Apoptosis via NO/CGMP Signaling. Int. J. Clin. Exp. Med. 2015, 8, 15269–15275. [Google Scholar] [PubMed]
- Wu, X.; She, W.; Niu, X.; Chen, X. Association between Serum Level of Advanced Glycation End Products and Obstructive Sleep Apnea-Hypopnea Syndrome: A Meta-Analysis. J. Int. Med. Res. 2018, 46, 4377–4385. [Google Scholar] [CrossRef]
- Liu, X.; Forbes, E.; Ryan, N.; Rofey, D.; Hannon, T.; Dahl, R. Rapid Eye Movement Sleep in Relation to Overweight in Children and Adolescents. Arch. Gen. Psychiatry 2008, 65, 924–932. [Google Scholar] [CrossRef]
- Pase, M.; Himali, J.; Grima, N.; Beiser, A.; Satizabal, C.; Aparicio, H.; Thomas, R.; Gottlieb, D.; Auerbach, S.; Seshadri, S. Sleep Architecture and the Risk of Incident Dementia in the Community. Neurology 2017, 89, 1244–1250. [Google Scholar] [CrossRef]
- Heid, M. Time. 2017. Available online: https://time.com/3183183/best-time-to-sleep/ (accessed on 1 May 2022).
- Narang, I.; Manlhiot, C.; Davies-Shaw, J.; Gibson, D.; Chahal, N.; Srearne, K.; Fisher, A.; Dobbin, S.; McCrindle, B. Sleep Disturbance and Cardiovascular Risk in Adolescents. Can. Med. Assoc. J. 2012, 184, E913–E920. [Google Scholar] [CrossRef]
- Lauderdale, D.; Knutson, K.; Rathouz, P.; Yan, L.; Hulley, S.; Liu, K. Cross-Sectional and Longitudinal Associations between Objectively Measured Sleep Duration and Body Mass Index: The CARDIA Sleep Study. Am. J. Epidemiol. 2009, 170, 805–813. [Google Scholar] [CrossRef]
- Stamatakis, K.; Punjabi, N. Effects of Sleep Fragmentation on Glucose Metabolism in Normal Subjects. Chest 2010, 137, 95–101. [Google Scholar] [CrossRef]
- Medic, G.; Wille, M.; Hemels, M. Short- and Long-Term Health Consequences of Sleep Disruption. Nat. Sci. Sleep 2017, 9, 151–161. [Google Scholar] [CrossRef]
- Rosa, D.; Marot, L.; de Mello, M.; Narciso, F.; Goncalves, B.; Marqueze, E.; Crispim, C. Shift Rotation, Circadian Misalignment and Excessive Body Weight Influence Psychomotor Performance: A Prospective and Observational Study under Real Life Conditions. Sci. Rep. 2019, 9, 19333. [Google Scholar] [CrossRef] [PubMed]
- Broussard, J.L.; Cauter, E. Disturbances of Sleep and Circadian Rhythms: Novel Risk Factors for Obesity. Curr. Opin. Endocrinol. Diabetes Obes. 2016, 23, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Morris, C.; Purvis, T.; Hu, K.; Scheer, F. Circadian Misalignment Increases Cardiovascular Disease Risk Factors in Humans. Proc. Natl. Acad. Sci. USA 2016, 113, e1402–e1411. [Google Scholar] [CrossRef] [PubMed]
- Hampton, S.; Morgan, L.; Lawrence, N.; Anastasiadou, T.; Norris, F.; Deacon, S.; Ribeiro, D.; Arendt, J. Postprandial Hormone and Metabolic Responses in Simulated Shift Work. J. Endocrinol. 1996, 151, 259–267. [Google Scholar] [CrossRef]
- Karlsson, B.; Knutsson, A.; Lindahl, B. Is There an Association between Shift Work and Having a Metabolic Syndrome? Results from a Population Based Study of 27,485 People. BMJ Occup. Environ. Med. 2001, 58, 747–752. [Google Scholar] [CrossRef]
- Fárková, E.; Smotek, M.; Bendová, Z.; Manková, D.; Koprivova, J. Chronotype and Social Jet-Lag in Relation to Body Weight, Apetite, Sleep Quality and Fatigue. Biol. Rhythm Res. 2021, 52, 1205–1216. [Google Scholar] [CrossRef]
- Smiley, A.; King, D.; Bidulescu, A. The Association between Sleep Duration and Metabolic Syndrome: The NHANES 2013/2014. Nutrients 2019, 11, 2582. [Google Scholar] [CrossRef]
- Smiley, A. Association of Sleep and Metabolic Syndrome/Metabolic Syndrome Components among Participants in Jackson Heart Study and REGARDS (Doctoral Dissertation); ProQuest LLC: Ann Arbor, MI, USA, 2019. [Google Scholar]
- Smiley, A.; King, D.; Harezlak, J.; Dinh, P.; Bidulescu, A. The Association between Sleep Duration and Lipid Profiles: The NHANES 2013–2014. J. Diabetes Metab. Disord. 2019, 18, 315–322. [Google Scholar] [CrossRef]
- Irwin, M.R.; Wang, M.; Ribeiro, D.; Cho, H.J.; Olmstead, R.; Breen, E.C.; Martinez-Maza, O.; Cole, S. Sleep Loss Activates Cellular Inflammatory Signaling. Biol. Psychiatry 2008, 64, 538–540. [Google Scholar] [CrossRef]
- Shearer, W.T.; Reuben, J.M.; Mullington, J.M.; Price, N.J.; Lee, B.-N.; Smith, E.O.; Szuba, M.P.; Van Dongen, H.P.A.; Dinges, D.F. Soluble TNF-α Receptor 1 and IL-6 Plasma Levels in Humans Subjected to the Sleep Deprivation Model of Spaceflight. J. Allergy Clin. Immunol. 2001, 107, 165–170. [Google Scholar] [CrossRef]
- Vgontzas, A.N.; Papanicolaou, D.A.; Bixler, E.O.; Lotsikas, A.; Zachman, K.; Kales, A.; Prolo, P.; Wong, M.-L.; Licinio, J.; Gold, P.W.; et al. Circadian Interleukin-6 Secretion and Quantity and Depth of Sleep. J. Clin. Endocrinol. Metab. 1999, 84, 2603–2607. [Google Scholar] [CrossRef] [PubMed]
- Castell, J.V.; Gómez-lechón, M.J.; David, M.; Fabra, R.; Trullenque, R.; Heinrich, P.C. Acute-Phase Response of Human Hepatocytes: Regulation of Acute-Phase Protein Synthesis by Interleukin-6. Hepatology 1990, 12, 1179–1186. [Google Scholar] [CrossRef] [PubMed]
- Okun, M.; Coussons-Read, M.; Hall, M. Disturbed Sleep Is Associated with Increased C-Reactive Protein in Young Women. Brain Behav. Immun. 2010, 23, 351–354. [Google Scholar] [CrossRef]
- Sauvet, F.; Leftheriotis, G.; Gomez-Merrino, D.; Drogou, C.; Van Beers, P.; Bourrilhon, C.; Florence, G.; Chennaoui, M. Effect of Acute Sleep Deprivation on Vascular Function in Healthy Subjects. J. Appl. Physiol. 2010, 108, 68–75. [Google Scholar] [CrossRef]
- Chae, C.U.; Lee, R.T.; Rifai, N.; Ridker, P.M. Blood Pressure and Inflammation in Apparently Healthy Men. Hypertension 2001, 38, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Safhi, M.A.; Alafif, R.A.; Alamoudi, N.M.; Alghamdi, W.A.; Albishri, S.F.; Rizk, H. The Association of Stress with Sleep Quality among Medical Students at King Abdulaziz University. J. Fam. Med. Prim. Care 2020, 9, 1662–1667. [Google Scholar] [CrossRef] [PubMed]
- Buysse, D.J.; Angst, J.; Gamma, A.; Ajdacic, V.; Eich, D.; Rossler, W. Prevalence, Course, and Comorbidity of Insomnia and Depression in Young Adults. Sleep 2008, 31, 473–480. [Google Scholar] [CrossRef]
- Nishitani, N.; Kawasaki, Y.; Sakakibara, H. Insomnia and Depression: Risk Factors for Development of Depression in Male Japanese Workers during 2011–2013. Int. J. Public Health 2018, 63, 49–55. [Google Scholar] [CrossRef]
- Hellhammer, D.K.; Wust, S.; Kudielka, B.M. Salivary Cortisol as a Biomarker in Stress Research. Psychoneuroendocrinology 2009, 34, 163–171. [Google Scholar] [CrossRef]
- Gamble, K.L.; Berry, R.; Frank, S.J.; Young, M.E. Circadian Clock Control of Endocrine Factors. Nat. Rev. Endocrinol. 2014, 10, 466–475. [Google Scholar] [CrossRef]
- Leproult, R.; Copinschi, G.; Buxton, O.; Van Cauter, E. Sleep Loss Results in an Elevation of Cortisol Levels the Next Evening. Sleep 1997, 20, 865–870. [Google Scholar] [PubMed]
- Bertollo, A.G.; Grolli, R.E.; Plissari, M.; Gasparin, V.A.; Quevedo, J.; Réus, G.Z.; Bagatini, M.D.; Ignácio, Z.M. Stress and Serum Cortisol Levels in Major Depressive Disorder: A Cross-Sectional Study. AIMS Neurosci. 2020, 7, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Halbreich, U.; Asnis, G.M.; Shindledecker, R. Cortisol Secretion in Endogenous Depression I. Basal Plasma Levels. Arch. Gen. Psychiatry 1985, 42, 909–914. [Google Scholar] [CrossRef] [PubMed]
- Young, E.A.; Carlson, N.E.; Brown, M.B. Twenty-Four-Hour ACTH and Cortisol Pulsatility in Depressed Women. Neuropsychopharmacology 2001, 25, 267–276. [Google Scholar] [CrossRef]
- Linkowski, P.; Mendlewicz, J.; Leclercq, R.; Brasseur, M.; Hubain, P.; Goldstein, J.; Copinschi, G.; Cauter, E.V. The 24-Hour Profile of Adrenocorticotropin and Cortisol in Major Depressive Illness Get Access Arrow. J. Clin. Endocrinol. Metab. 1985, 61, 429–438. [Google Scholar] [CrossRef]
- Nandam, L.S.; Brazel, M.; Zhou, M.; Jhaveri, D.J. Cortisol and Major Depressive Disorder—Translating Findings from Humans to Animal Models and Back. Front. Psychiatry 2019, 10, 974. [Google Scholar] [CrossRef]
- Pace, T.W.W.; Hu, F.; Miller, A.H. Cytokine-Effects on Glucocorticoid Receptor Function: Relevance to Glucocorticoid Resistance and the Pathophysiology and Treatment of Major Depression. Brain Behav. Immun. 2007, 21, 9–19. [Google Scholar] [CrossRef]
- Dendoncker, K.; Timmermans, S.; Vandewalle, J.; Eggermont, M.; Lempiainen, J.; Paakinaho, V.; Van Hamme, E.; Libert, C. TNF-α Inhibits Glucocorticoid Receptor-Induced Gene Expression by Reshaping the GR Nuclear Cofactor Profile. Proc. Natl. Acad. Sci. USA 2019, 116, 15745. [Google Scholar] [CrossRef]
- Kim, E.-J.; Dimsdale, J.E. The Effect of Psychosocial Stress on Sleep: A Review of Polysomnographic Evidence. Behav. Sleep Med. 2007, 5, 256–278. [Google Scholar] [CrossRef]
- Gupta, R.; Lahan, V. Insomnia Associated with Depressive Disorder: Primary, Secondary, or Mixed? Indian J. Psychol. Med. 2011, 33, 123–128. [Google Scholar] [CrossRef]
- Mayers, A.G.; van Hooff, J.C.; Baldwin, D.S. Quantifying Subjective Assessment of Sleep and Life-Quality in Antidepressant-Treated Depressed Patients. Hum. Psychopharmacol. 2002, 18, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Nutt, D.; Wilson, S.; Paterson, L. Sleep Disorders as Core Symptoms of Depression. Dialogues Clin. Neurosci. 2008, 10, 329–336. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, E.M.; Chelminski, I.; Young, D.; Dalrymple, K.; Hrabosly, J.; Zimmerman, M. Severe Insomnia Is Associated with More Severe Presentation and Greater Functional Deficits in Depression. J. Psychiatr. Res. 2011, 45, 1101–1105. [Google Scholar] [CrossRef] [PubMed]
- Klonoff, D.C. The Increasing Incidence of Diabetes in the 21st Century. J. Diabetes Sci. Technol. 2009, 3, 1–2. [Google Scholar] [CrossRef] [PubMed]
- CDC. Long-Term Trends in Diabetes. Available online: https://www.cdc.gov/diabetes/statistics/slides/long_term_trends.pdf (accessed on 1 April 2022).
- CDC. National Diabetes Statistics Report. 2020. Available online: https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf (accessed on 1 April 2022).
- Yaggi, H.K.; Araujo, A.B.; McKinlay, J.B. Sleep Duration as a Risk Factor for the Development of Type 2 Diabetes. Cardiovasc. Metab. Risk 2006, 29, 657–661. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.T.; Nguyen, X.T.; Lane, J.; Wang, P. Relationship between Obesity and Diabetes in a US Adult Population: Findings from the National Health and Nutrition Examination Survey, 1999–2006. Obes. Surg. 2011, 21, 351–355. [Google Scholar] [CrossRef]
- Frey, D.J.; Fleshner, M.; Wright, K.P. The Effects of 40 Hours of Total Sleep Deprivation on Inflammatory Markers in Healthy Young Adults. Brain Behav. Immun. 2007, 21, 1050–1057. [Google Scholar] [CrossRef]
- Haack, M.; Sanchez, E.; Mullington, J.M. Elevated Inflammatory Markers in Response to Prolonged Sleep Restriction Are Associated with Increased Pain Experience in Healthy Volunteers. Sleep 2007, 30, 1145–1152. [Google Scholar] [CrossRef]
- Chirwa, S.; Nwabuisi, C.R.; Ladson, G.M.; Korley, L.; Whitty, J.E.; Atkinson, R.; Clark, J.T. Poor Sleep Quality Is Associated with Higher Hemoglobin A1c in Pregnant Women: A Pilot Observational Study. Int. J. Environ. Res. Public Health 2018, 15, 2287. [Google Scholar] [CrossRef]
- Kass, L.; Sanderson, J.C.; Desai, T.; Hurst, R. The Relationship between the Elevation of Haemoglobin A1c Level, Sleep Quality and Sleep Duration in Clinically Diagnosed Pre-Diabetic Patients in a Nationally Representative Sample. Diabetes Vasc. Dis. Res. 2022, 19, 14791641211067421. [Google Scholar] [CrossRef]
- Rizza, S.; Luzi, A.; Mavilio, M.; Ballanti, M.; Massimi, A.; Porzio, O.; Magrini, A.; Hannemann, J.; Menghini, R.; Lehrke, M.; et al. Alterations in Rev-ERBα/BMAL1 Ratio and Glycated Hemoglobin in Rotating Shift Workers: The EuRhythDia Study. Acta Diebtologica 2021, 58, 1111–1117. [Google Scholar] [CrossRef] [PubMed]
- Burt, V.L.; Whelton, P.; Roccella, E.J.; Brown, C.; Cutler, J.A.; Higgins, M.; Horan, M.J.; Labarthe, D. Results from the Third National Health and Nutrition Examination Survey, 1988–1991. Hypertension 1995, 25, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, D.J.; Redline, S.; Nieto, F.J.; Baldwin, C.M.; Newman, A.B.; Resnick, H.E.; Punjabi, N.M. Association of Usual Sleep Duration with Hypertension: The Sleep Heart Health Study. Sleep 2006, 29, 1009–1014. [Google Scholar] [CrossRef] [PubMed]
- Matre, D.; Sirnes, P.A.; Goffeng, E.; Skare, O.; Skogstad, M. Sleep Duration, Number of Awakenings and Arterial Stiffness in Industrial Shift Workers: A Five-Week Follow-Up Study. Int. J. Environ. Res. Public Health 2022, 19, 1964. [Google Scholar] [CrossRef]
- Tochikubo, O.; Ikeda, A.; Miyajima, E.; Ishii, M. Effects of Insufficient Sleep on Blood Pressure Monitored by a New Multibiomedical Recorder. Hypertension 1996, 27, 1318–1324. [Google Scholar] [CrossRef]
- Kato, M.; Phillips, B.G.; Sigurdsson, G.; Narkiewicz, K.; Pesek, C.A.; Somers, V.K. Effects of Sleep Deprivation on Neural Circulatory Control. Hypertension 2000, 35, 1173–1175. [Google Scholar] [CrossRef]
- Palma, B.D.; Gabrial, A.; Bignotto, M.; Tufik, S. Paradoxical Sleep Deprivation Increases Plasma Endothelin Levels. Braz. J. Med. Biol. Res. 2002, 35, 75–79. [Google Scholar] [CrossRef]
- Weil, B.R.; Mestek, M.L.; Westby, C.M.; Van Guilder, G.P.; Greiner, J.J.; Stauffer, B.L.; DeSouza, C.A. Short Sleep Duration Is Associated with Enhanced Endothelin-1 Vasoconstrictor Tone. Can. J. Physiol. Pharacol. 2010, 88, 777–781. [Google Scholar] [CrossRef]
- Chang, H.-M.; Wu, U.-I.; Lin, T.-B.; Lan, C.-T.; Chien, W.-C.; Huang, W.-L.; Shieh, J.-Y. Total Sleep Deprivation Inhibits the Neuronal Nitric Oxide Synthase and Cytochrome Oxidase Reactivities in the Nodose Ganglion of Adult Rats. J. Anat. 2006, 209, 239–250. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Underlying Cause of Death, 1999–2018; CDC WONDER Online Database: Atlanta, GA, USA, 2018. [Google Scholar]
- Ayas, N.T.; White, D.P.; Manson, J.E.; Stampfer, M.J.; Speizer, F.E.; Malhotra, A.; Hu, F.B. A Prospective Study of Sleep Duration and Coronary Heart Disease in Women. JAMA Intern. Med. 2003, 163, 205–209. [Google Scholar] [CrossRef]
- Lao, X.Q.; Liu, X.; Deng, H.-B.; Chan, T.-C.; Ho, K.F.; Wang, F.; Vermeulen, R.; Tam, T.; Wong, M.; Tse, L.A.; et al. Sleep Quality, Sleep Duration, and the Risk of Coronary Heart Disease: A Prospective Cohort Study with 60,586 Adults. J. Clin. Sleep Med. 2018, 14, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Ferrie, J.E.; Shipley, M.J.; Cappuccio, F.P.; Brunner, E.; Miller, M.A.; Kumari, M.; Marmot, M.G. A Prospective Study of Change in Sleep Duration: Associations with Mortality in the Whitehall II Cohort. Sleep 2007, 30, 1659–1666. [Google Scholar] [CrossRef] [PubMed]
- Meier-Ewert, H.K.; Ridker, P.M.; Rifai, N.; Regan, M.M.; Price, N.J.; Dinges, D.F.; Mullington, J.M. Effect of Sleep Loss on C-Reactive Protein, an Inflammatory Marker of Cardiovascular Risk. J. Am. Coll. Cardiol. 2004, 43, 678–683. [Google Scholar] [CrossRef]
- Danesh, J.; Collins, R.; Appleby, P.; Peto, R. Association of Fibrinogen, C-Reactive Protein, Albumin, or Leukocyte Count with Coronary Heart Disease. JAMA 1998, 279, 1477–1482. [Google Scholar] [CrossRef]
- Hashimoto, H.; Kitagawa, K.; Hougaku, H.; Shimizu, Y.; Sakaguchi, M.; Nagai, Y.; Lyama, S.; Yamanishi, H.; Matsumoto, M.; Hori, M. C-Reactive Protein Is an Independent Predictor of the Rate of Increase in Early Carotid Atherosclerosis. Circulation 2001, 104, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Ridker, P.M.; Maseri, A. Inflammation and Atherosclerosis. Circulation 2002, 105, 1135–1143. [Google Scholar] [CrossRef]
- Frostegard, J. Immunity, Atherosclerosis and Cardiovascular Disease. BMC Med. 2013, 11, 117. [Google Scholar] [CrossRef] [PubMed]
- Poirier, P.; Giles, T.D.; Bray, G.A.; Hong, Y.; Stern, J.S.; Pi-Sunyer, F.X.; Eckel, R.H. Obesity and Cardiovascular Disease: Pathophysiology, Evaluation, and Effect of Weight Loss. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 968–976. [Google Scholar] [CrossRef]
- Song, C.; Zhang, R.; Wang, C.; Fu, R.; Song, W.; Dou, K.; Wang, S. Sleep Quality and Risk of Cancer: Findings from the English Longitudinal Study of Aging. Sleep 2021, 44, zsaa192. [Google Scholar] [CrossRef]
- Shi, T.; Min, M.; Sun, C.; Zhang, Y.; Liang, M.; Sun, Y. Does Insomnia Predict a High Risk of Cancer? A Systematic Review and Meta-Analysis of Cohort Studies. J. Sleep Res. 2020, 29, e12876. [Google Scholar] [CrossRef]
- Walker, W.H., II; Borniger, J.C. Molecular Mechanisms of Cancer-Induced Sleep Disruption. Int. J. Mol. Sci. 2019, 20, 2780. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Rosales-Corral, S.A.; Tan, D.-X.; Acuna-Castroviejo, D.; Qin, L.; Yang, S.-F.; Xu, K. Melatonin, a Full Service Anti-Cancer Agent: Inhibition of Initiation, Progression and Metastasis. Int. J. Mol. Sci. 2017, 18, 843. [Google Scholar] [CrossRef] [PubMed]
- Fritschi, L.; Glass, D.C.; Heyworth, J.S.; Aronson, K.; Girschik, J.; Boyle, T.; Grundy, A.; Erren, T.C. Hypotheses for Mechanisms Linking Shiftwork and Cancer. Med. Hypotheses 2011, 77, 430–436. [Google Scholar] [CrossRef]
- Khan, Q.J.; Kimler, B.F.; Fabian, C.J. The Relationship between Vitamin D and Breast Cancer Incidence and Natural History. Curr. Oncol. Rep. 2010, 12, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Coussens, L.M.; Werb, Z. Inflammation and Cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef]
- Liu, X.; Chen, B.; Huang, Z.; Duan, R.; Li, H.; Wang, R.; Li, Z.; Gao, Y.; Zheng, Y.; Su, W. Effects of Poor Sleep on the Immune Cell Landscape as Assessed by Single-Cell Analysis. Commun. Biol. 2021, 4, 1325. [Google Scholar] [CrossRef]
- Ji, Z.; He, L.; Regev, A.; Struhl, K. Inflammatory Regulatory Network Mediated by the Joint Action of NF-KB, STAT3, and AP-1 Factors Is Involved in Many Human Cancers. Proc. Natl. Acad. Sci. USA 2019, 116, 9453–9462. [Google Scholar] [CrossRef]
- Punjabi, N.M. The Epidemiology of Adult Obstructive Sleep Apnea. Proc. Am. Thorac. Soc. 2007, 5, 136–143. [Google Scholar] [CrossRef]
- Agusti, A.; Hedner, J.; Marin, J.M.; Barbé, F.; Cazzola, M.; Rennard, S. Night-Time Symptoms: A Forgotten Dimension of COPD. Eur. Respir. Rev. 2011, 20, 183–194. [Google Scholar] [CrossRef]
- Fuller-Thomson, E.; Howden, K.E.; Fuller-Thomson, L.R.; Agbeyaka, S. A Strong Graded Relationship between Level of Obesity and COPD: Findings from a National Population-Based Study of Lifelong Nonsmokers. J. Obes. 2018, 2018, 6149263. [Google Scholar] [CrossRef]
- Bringardner, B.D.; Baran, C.P.; Eubank, T.D.; Marsh, C.B. The Role of Inflammation in the Pathogenesis of Idiopathic Pulmonary Fibrosis. Antioxid. Redox Signal. 2008, 10, 287–301. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Xiong, X.; Lin, Y.; Zheng, B.; Cheng, D. Association between Serum Interleukin-6 Concentrations and Chronic Obstructive Pulmonary Disease: A Systematic Review and Meta-Analysis. PeerJ 2015, 3, e1199. [Google Scholar] [CrossRef]
- Knight, D.A.; Ernst, M.; Anderson, G.P.; Moodley, Y.P.; Mutsaers, S.E. The Role of Gp130/IL-6 Cytokines in the Development of Pulmonary Fibrosis: Critical Determinants of Disease Susceptibility and Progression? Pharmacol. Ther. 2003, 99, 327–338. [Google Scholar] [CrossRef]
- Patel, S.R.; Malhotra, A.; Gao, X.; Hu, F.B.; Neuman, M.I.; Fawzi, W.W. A Prospective Study of Sleep Duration and Pneumonia Risk in Women. Sleep 2012, 35, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Irwin, M.; McClintick, J.; Costlow, C.; Fortner, M.; White, J.; Gillin, J.C. Partial Night Sleep Deprivation Reduces Natural Killer and Celhdar Immune Responses in Human. FASEB J. 1996, 10, 643–653. [Google Scholar] [CrossRef] [PubMed]
- Bollinger, T.; Bolinger, A.; Skrum, L.; Dimitrov, S.; Lange, T.; Solbach, W. Sleep-Dependent Activity of T Cells and Regulatory T Cells. Clin. Exp. Immunol. 2009, 155, 231–238. [Google Scholar] [CrossRef]
- Mitselou, A.; Grammaeniatis, V.; Varouktsi, A.; Papadatos, S.S.; Katsanos, K.; Galani, V. Proinflammatory Cytokines in Irritable Bowel Syndrome: A Comparison with Inflammatory Bowel Disease. Intest. Res. 2020, 18, 115–120. [Google Scholar] [CrossRef]
- Kim, H.I.; Jung, S.-A.; Choi, J.Y.; Kim, S.-E.; Jung, H.-K.; Shim, K.-N.; Yoo, K. Impact of Shiftwork on Irritable Bowel Syndrome and Functional Dyspepsia. J. Korean Med. Sci. 2013, 28, 431–437. [Google Scholar] [CrossRef]
- Tauseef, A.; Choe, J.; Awab, A.; Wagener, T.L.; Orr, W.C. Sleep, Immunity and Inflammation in Gastrointestinal Disorders. World J. Gestroenterol. 2013, 19, 9231–9239. [Google Scholar] [CrossRef]
- Schey, R.; Dickman, R.; Parthasarathy, S.; Quan, S.F.; Wendel, C.; Merchant, J.; Powers, J.; Han, B.; Van Handel, D.; Fass, R. Sleep Deprivation Is Hyperalgesic in Patients with Gastroesophageal Reflux Disease. Gastroenterology 2007, 133, 1787–1795. [Google Scholar] [CrossRef]
- Dickhaus, B.; Mayer, E.A.; Firooz, N.; Stains, J.; Conde, F.; Olivas, T.I.; Fass, R.; Chang, L.; Mayer, M.; Naliboff, B.D. Irritable Bowel Syndrome Patients Show Enhanced Modulation of Visceral Perception by Auditory Stress. Am. J. Gastroenterol. 2003, 98, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Holtzman, D.M. Bidirectional Relationship between Sleep and Alzheimer’s Disease: Role of Amyloid, Tau, and Other Factors. Neuropsychopharmacology 2020, 45, 104–120. [Google Scholar] [CrossRef] [PubMed]
- Brown, B.M.; Rainey-Smith, S.R.; Villemagne, V.L.; Weinborn, M.; Bucks, R.S.; Sohrabi, H.R.; Laws, S.M.; Taddei, K.; Macaulay, S.L.; Ames, D.; et al. The Relationship between Sleep Quality and Brain Amyloid Burden. Sleep 2016, 39, 1063–1068. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.-Y.; Sunwoo, J.-S.; Kim, M.-W.; Kim, M. The Neuroprotective Effects of Human Growth Hormone as a Potential Treatment for Amyotrophic Lateral Sclerosis. Neural Regen. Res. 2015, 10, 1201–1203. [Google Scholar] [CrossRef] [PubMed]
- Chahine, L.M.; Amara, A.W.; Videnovic, A. A Systematic Review of the Literature on Disorders of Sleep and Wakefulness in Parkinson’s Disease from 2005 to 2015. Sleep Med. Rev. 2017, 35, 33–50. [Google Scholar] [CrossRef]
- Comella, C.L. Sleep Disturbances and Excessive Daytime Sleepiness in Parkinson Disease: An Overview. J. Neural Transm. 2006, 70, 349–355. [Google Scholar] [CrossRef]
- Hsiao, Y.-H.; Chen, Y.-T.; Tseng, C.-M.; Wu, L.-A.; Perng, D.-W.; Chen, Y.-M.; Chen, T.-J.; Chang, S.-C.; Chou, K.-T. Sleep Disorders and an Increased Risk of Parkinson’s Disease in Individuals with Non-Apnea Sleep Disorders: A Population-Based Cohort Study. J. Sleep Res. 2017, 26, 623–628. [Google Scholar] [CrossRef]
- Stefanis, L. α-Synuclein in Parkinson’s Disease. Cold Spring Harb. Perspect. Med. 2012, 2, a009399. [Google Scholar] [CrossRef]
- Bo, Y.; Yeoh, E.-K.; Guo, C.; Zhang, Z.; Tam, T.; Chan, T.-C.; Change, L.-Y.; Lao, X.Q. Sleep and the Risk of Chronic Kidney Disease: A Cohort Study. J. Clin. Sleep Med. 2019, 15, 393–400. [Google Scholar] [CrossRef]
- Hering, D.; Esler, M.D.; Schlaich, M.P. Chronic Kidney Disease: Role of Sympathetic Nervous System Activation and Potential Benefits of Renal Denervation. Eurointervention 2013, 9, 127–135. [Google Scholar] [CrossRef]
- Yamamoto, R.; Nagasawa, Y.; Iwatani, H.; Shinzawa, M.; Obi, Y.; Teranishi, J.; Ishigami, T.; Yamauchi-Takihara, K.; Nishida, M.; Rakugi, H.; et al. Self-Reported Sleep Duration and Prediction of Proteinuria: A Retrospective Cohort Study. Am. J. Kidney Dis. 2012, 59, 343–355. [Google Scholar] [CrossRef] [PubMed]
- Shibata, M.; Sato, K.K.; Uehara, S.; Koh, H.; Kinuhata, S.; Oue, K.; Kambe, H.; Morimoto, M.; Hayashi, T. Blood Pressure Components and the Risk for Proteinuria in Japanese Men: The Kansai Healthcare Study. J. Epidemiol. 2017, 27, 505–510. [Google Scholar] [CrossRef]
- Wong, C.S.; Pierce, C.B.; Cole, S.R.; Warady, B.A.; Mak, R.H.K.; Benador, N.M.; Kaskel, F.; Furth, S.L.; Shwartz, G.J. Association of Proteinuria with Race, Cause of Chronic Kidney Disease, and Glomerular Filtration Rate in the Chronic Kidney Disease in Children Study. Clin. J. Am. Soc. Nephrol. 2009, 4, 812–819. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.T.; Quartana, P.J.; Okonkwo, R.M.; Nasir, A. Mechanisms by Which Sleep Disturbance Contributes to Osteoarthritis Pain: A Conceptual Model. Curr. Pain Headache Rep. 2009, 13, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Goldring, M.B.; Otero, M. Inflammation in Osteoarthritis. Curr. Opin. Rheumatol. 2011, 23, 471–478. [Google Scholar] [CrossRef]
- Malemud, C.J. The Biological Basis of Osteoarthritis: State of the Evidence. Curr. Opin. Rheumatol. 2015, 27, 289–294. [Google Scholar] [CrossRef]
- Yang, M.; Cen, X.; Xie, Q.; Zuo, C.; Yin, G. Serum Interleukin-6 Expression Level and Its Clinical Significance in Patients with Dermatomyositis. Clin. Dev. Immunol. 2013, 2013, 717808. [Google Scholar] [CrossRef]
- Grabovac, I.; Haider, S.; Berner, C.; Lamprecht, T.; Fenzl, K.-H.; Erlacher, L.; Quittan, M.; Dorner, T.E. Sleep Quality in Patients with Rheumatoid Arthritis and Associations with Pain, Disability, Disease Duration, and Activity. J. Clin. Med. 2018, 7, 336. [Google Scholar] [CrossRef]
- Doghramji, K. Melatonin and Its Receptors: A New Class of Sleep-Promoting Agents. J. Clin. Sleep Med. 2007, 3, S17–S23. [Google Scholar] [CrossRef]
- Suzuki, M.; Taniguchi, T.; Furihata, R.; Yoshita, K.; Arai, Y.; Yoshiike, N.; Uchiyama, M. Seasonal Changes in Sleep Duration and Sleep Problems: A Prospective Study in Japanese Community Residents. PLoS ONE 2019, 14, e0215345. [Google Scholar] [CrossRef]
- Paul, M.A.; Love, R.J.; Hawton, A.; Arendt, J. Sleep and the Endogenous Melatonin Rhythm of High Arctic Residents during the Summer and Winter. Physiol. Behav. 2015, 141, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Dong, Q.; Gentry, N.W.; McMahon, T.; Yamazaki, M.; Bernitez-Rivera, L.; Wang, T.; Ptacek, L.; Fu, Y.-H. Familial Natural Short Sleep Mutations Reduce Alzheimer Pathology in Mice. iScience 2022, 25, 103964. [Google Scholar] [CrossRef] [PubMed]
- Shi, G.; Yin, C.; Fan, Z.; Xing, L.; Mostovoy, Y.; Kwok, P.; Ashbrook, L.H.; Krystal, A.D.; Ptacek, L.J.; Fu, Y. Mutations in Metabotropic Glutamate Receptor 1 Contribute to Natural Short Sleep Trait. Curr. Biol. 2021, 31, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Xing, L.; Shi, G.; Mostovoy, Y.; Gentry, N.W.; Fan, Z.; McMahon, T.B.; Kwok, P.; Jones, C.R.; Fu, Y. Mutant Neuropeptide S Receptor Reduces Sleep Duration with Preserved Memory Consolidation. Sci. Transl. Med. 2019, 11, eaax2014. [Google Scholar] [CrossRef] [PubMed]
- Aeschbach, D.; Cajochen, C.; Landolt, H.; Borbely, A.A. Homeostatic Sleep Regulation in Habitual Short Sleepers and Long Sleepers. Am. J. Physiol. Regul. Integr. Comp. Physiol. 1996, 270, R41–R53. [Google Scholar] [CrossRef]
- Aeschbach, D.; Sher, L.; Postolache, T.T.; Matthews, J.R.; Jackson, M.A.; Wehr, T.A. A Longer Biological Night in Long Sleepers Than in Short Sleepers. J. Clin. Endocrinol. Metab. 2003, 88, 26–30. [Google Scholar] [CrossRef]
- Irwin, M.R. Sleep and Inflammation: Partners in Sickness and in Health. Nat. Rev. 2019, 19, 702–715. [Google Scholar] [CrossRef]
- Lange, T.; Dimitrov, S.; Fehm, H.-L.; Westermann, J.; Born, J. Shift of Monocyte Function toward Cellular Immunity during Sleep. JAMA Intern. Med. 2006, 166, 1695–1700. [Google Scholar] [CrossRef]
- Hahn, J.; Gunter, M.; Schumacher, J.; Bieber, K.; Poschel, S.; Schutz, M.; Engelhardt, B.; Oster, H.; Sina, C.; Lange, T.; et al. Sleep Enhances Numbers and Function of Monocytes and Improves Bacterial Infection Outcome in Mice. Brain Behav. Immun. 2020, 87, 329–338. [Google Scholar] [CrossRef]
- Scott, A.J.; Webb, T.L.; Martyn-St James, M.; Rowse, G.; Weich, S. Improving Sleep Quality Leads to Better Mental Health: A Meta-Analysis of Randomised Controlled Trials. Sleep Med. Rev. 2021, 60, 101556. [Google Scholar] [CrossRef]
- Franzen, P.L.; Buysse, D.J. Sleep Disturbances and Depression: Risk Relationships for Subsequent Depression and Therapeutic Implications. Dialogues Clin. Neurosci. 2008, 10, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Kline, C.E.; Chasens, E.R.; Bizhanova, Z.; Sereika, S.M.; Buysse, D.J.; Imes, C.C.; Kariuku, J.K.; Mendez, D.D.; Cajita, M.I.; Rathbun, S.L.; et al. The Association between Sleep Health and Weight Change during a 12-Month Behavioral Weight Loss Intervention. Int. J. Obes. 2021, 45, 639–649. [Google Scholar] [CrossRef] [PubMed]
- Thomson, C.A.; Morrow, K.L.; Flatt, S.W.; Wetheim, B.C.; Perfect, M.M.; Ravia, J.J.; Sherwood, N.E.; Karanja, N.; Rock, C.L. Relationship between Sleep Quality and Quantity and Weight Loss in Women Participating in a Weight-Loss Intervention Trial. Obes. Res. J. 2012, 20, 1419–1425. [Google Scholar] [CrossRef] [PubMed]
- Tasali, E.; Wroblewski, K.; Kahn, E. Effect of Sleep Extension on Objectively Assessed Energy Intake among Adults with Overweight in Real-Life Settings. JAMA Intern. Med. 2022, 182, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Pizinger, T.M.; Aggarwal, B.; St-Onge, M.-P. Sleep Extension in Short Sleepers: An Evaluation of Feasibility and Effectiveness for Weight Management and Cardiometabolic Disease Prevention. Front. Endocrinol. 2018, 9, 392. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Wang, M.; Viswanath, K.; Lai, A.; Fong, D.; Lin, C.-C.; Chan, S.S.-C.; Lam, T. Short Sleep Duration and Insomnia Symptoms Were Associated with Lower Happiness Levels in Chinese Adults in Hong Kong. Int. J. Environ. Res. Public Health 2019, 16, 2079. [Google Scholar] [CrossRef]
- Ruggeri, K.; Garcia-Garzon, E.; Maguire, Á.; Matz, S.; Huppert, F. Well-Being Is More than Happiness and Life Satisfaction: A Multidimensional Analysis of 21 Countries. Health Qual. Life Outcomes 2020, 18, 192. [Google Scholar] [CrossRef]
- Diener, E.; Pressman, S.; Hunter, J.; Delgadillo-Chase, D. If, Why, and When Subjective Well-Being Influences Health, and Future Needed Research. Appl. Psychol. Health Wellbeing 2017, 9, 133–167. [Google Scholar] [CrossRef]
- Smiley, A.; Ramos, W.; Elliot, L.; Wolter, S. Comparing the Trail Users with Trail Non-Users on Physical Activity, Sleep, Mood and Well-Being Index. Int. J. Environ. Res. Public Health 2020, 17, 6225. [Google Scholar] [CrossRef]
- Smiley, A.; Ramos, W.; Elliot, L.; Wolter, S. Association between Trail Use and Self-Rated Wellness and Health. BMC Public Health 2020, 20, 128. [Google Scholar] [CrossRef]
- Triantafillou, S.; Saeb, S.; Lattie, E.G.; Mohr, D.C.; Kording, K.P. Relationship between Sleep Quality and Mood: Ecological Momentary Assessment Study. JMIR Ment. Health 2019, 6, e12613. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.-E.; Kim, J.K. How a Good Sleep Predicts Life Satisfaction: The Role of Zero-Sum Beliefs about Happiness. Front. Psychol. 2018, 9, 1589. [Google Scholar] [CrossRef] [PubMed]
- Fabbri, M.; Beracci, A.; Martoni, M.; Meneo, D.; Tonetti, L.; Natale, V. Measuring Subjective Sleep Quality: A Review. Int. J. Environ. Res. Public Health 2021, 18, 1082. [Google Scholar] [CrossRef] [PubMed]
- Natale, V.; Fabbri, M.; Tonetti, L.; Martoni, M. Psychometric Goodness of the Mini Sleep Questionnaire. Psychiatry Clin. Neurosci. 2014, 68, 568–573. [Google Scholar] [CrossRef]
- Masoumi, M.; Shokraee, K.; Mohammadi, S.; Moradi, S.; Bagherzade, M.; Balasi, J.; Smiley, A. Sleep Duration as the Main Indicator of Self-Rated Wellness and Health among Healthcare Workers Involved in the COVID-19 Pandemic. Int. J. Environ. Res. Public Health 2021, 19, 136. [Google Scholar] [CrossRef]
- Masoumi, M.; Tabaraii, R.; Shakiba, S.; Shakeri, M.; Smiley, A. Association of Lifestyle Elements with Self-Rated Wellness and Health Status in Patients with Behcet’s Disease. BMC Rheumatol. 2020, 4, 49. [Google Scholar] [CrossRef] [PubMed]
- Tabaraii, R.; Masoumi, M.; Bagherzadeh-Ford, M.; Yazdanifar, M.A.; Balasi, J.; Smiley, A. Association of Lifestyle and Disease Characteristics with Self-Rated Wellness/Health Score in Patients with Rheumatoid Arthritis. BMC Rheumatol. 2021, 5, 55. [Google Scholar] [CrossRef]
- Hershner, S.; O’Brien, L.M. The Impact of a Randomized Sleep Education Intervention for College Students. J. Clin. Sleep Med. 2018, 14, 337–347. [Google Scholar] [CrossRef]
- Tucker, R.M.; Contreras, D.A.; Carlson, B.R.; Carter, A.; Drake, C.L. Sleep Education for Elders Program (SLEEP):Promising Pilot Results of a Virtual, Health Educator-Led, Community-Delivered Sleep Behavior Change Intervention. Nat. Sci. Sleep 2021, 13, 625–633. [Google Scholar] [CrossRef]
- Chawla, S.P.S.; Kaur, S.; Bharty, A.; Ravinder, G.; Manjeet, K.; Soin, D.; Ghosh, A.; Pal, R. Impact of Health Education on Knowledge, Attitude, Practices and Glycemic Control in Type 2 Diabetes Mellitus. J. Fam. Med. Prim. Care 2019, 8, 261–268. [Google Scholar] [CrossRef]
- Al-Naggar, R.A.; Musa, R.; Al-Jashamy, K.; Isa, Z.M. Knowledge, Attitude, and Practice (KAP) towards Sleep among Medical Students of International Islamic University Malaysia (IIUM). ASEAN J. Psychiatry 2010, 11, 6–12. [Google Scholar]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feingold, C.L.; Smiley, A. Healthy Sleep Every Day Keeps the Doctor Away. Int. J. Environ. Res. Public Health 2022, 19, 10740. https://doi.org/10.3390/ijerph191710740
Feingold CL, Smiley A. Healthy Sleep Every Day Keeps the Doctor Away. International Journal of Environmental Research and Public Health. 2022; 19(17):10740. https://doi.org/10.3390/ijerph191710740
Chicago/Turabian StyleFeingold, Cailan Lindsay, and Abbas Smiley. 2022. "Healthy Sleep Every Day Keeps the Doctor Away" International Journal of Environmental Research and Public Health 19, no. 17: 10740. https://doi.org/10.3390/ijerph191710740
APA StyleFeingold, C. L., & Smiley, A. (2022). Healthy Sleep Every Day Keeps the Doctor Away. International Journal of Environmental Research and Public Health, 19(17), 10740. https://doi.org/10.3390/ijerph191710740





