EsReflux Protocol: Epidemiological Study of Heartburn and Reflux-like Symptoms in Spanish Community Pharmacies
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Setting
2.3. Study Population
2.4. Sample’s Size Calculation
2.5. Recruitment Procedure
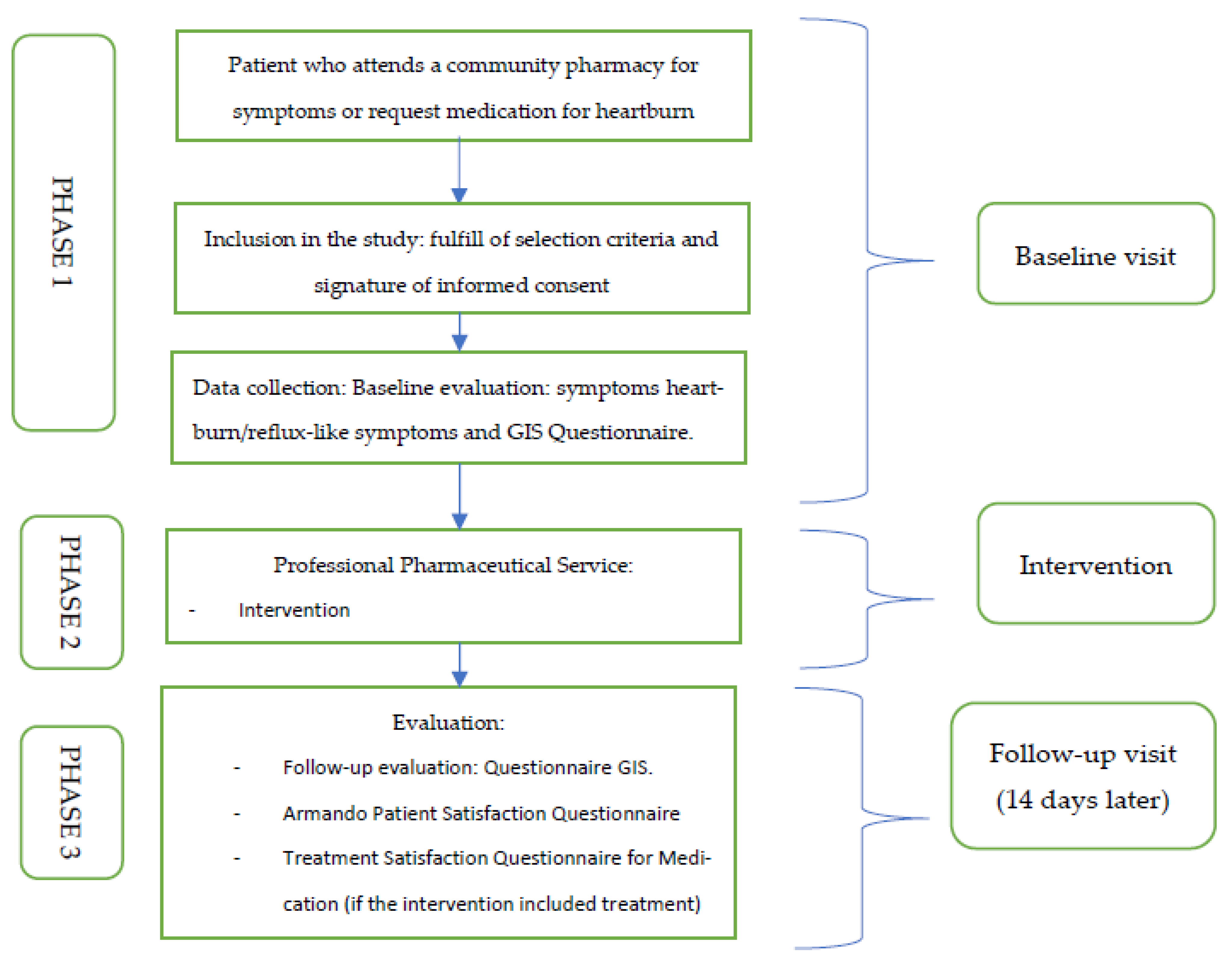
2.6. Data Collection Procedure
2.7. Baseline Visit
2.8. Outcome Measures
2.9. Data Analysis Plan
2.10. Ethics
2.11. Dissemination
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fletcher, J.; Wirz, A.; Young, J.; Vallance, R.; McColl, K.E. Unbuffered highly acidic gastric juice exists at the gastroesophageal junction after a meal. Gastroenterology 2001, 121, 775–783. [Google Scholar] [CrossRef] [PubMed]
- Serra Pueyo, J. Puesta al día en el reflujo gastroesofágico. Gastroenterol. Y Hepatol. 2014, 37, 73–82. [Google Scholar] [CrossRef] [PubMed]
- De Ruigh, A.; Roman, S.; Chen, J.; Pandolfino, J.E.; Kahrilas, P.J. Gaviscon Double Action Liquid (antacid & alginate) is more effective than antacid in controlling post-prandial oesophageal acid exposure in GERD patients: A double-blind crossover study. Aliment. Pharmacol. Ther. 2014, 40, 531–537. [Google Scholar] [PubMed]
- Mandel, K.G.; Daggy, B.P.; Brodie, A.D.; Jacoby, H.I. Review article: Alginate-raft formulations in the treatment of heartburn and acid reflux. Aliment. Pharmacol. Ther. 2000, 14, 669–690. [Google Scholar] [CrossRef]
- Quigley, E.M. Non-erosive reflux disease, functional heartburn and gastroesophageal reflux disease; insights into pathophysiology and clinical presentation. Chin. J. Dig. Dis. 2006, 7, 186–190. [Google Scholar] [CrossRef]
- Garrigues, V.; Ponce, J. Aspectos menos conocidos de la enfermedad por reflujo gastroesofágico: Pirosis funcional y reflujo no ácido. Gastroenterol. Hepatol. 2008, 31, 522–529. [Google Scholar] [CrossRef]
- Drossman, D.A. Functional Gastrointestinal Disorders: History, Pathophysiology, Clinical Features, and Rome IV. Gastroenterology 2016, 150, 1262–1279.e2. [Google Scholar] [CrossRef] [Green Version]
- Diaz-Rubio, M.; Moreno-Elola-Olaso, C.; Rey, E.; Locke, G.R.; Rodriguez-Artalejo, F. Symptoms of gastro-oesophageal reflux: Prevalence, severity, duration and associated factors in a Spanish population. Aliment. Pharmacol. Ther. 2004, 19, 95–105. [Google Scholar] [CrossRef]
- Galmiche, J.P.; Clouse, R.E.; Bálint, A.; Cook, I.J.; Kahrilas, P.J.; Paterson, W.G.; Smout, A.J. Functional Esophageal Disorders. Gastroenterology 2006, 130, 1459–1465. [Google Scholar] [CrossRef] [Green Version]
- Ford, A.; Marwaha, A.; Sood, R.; Moayyedi, P. Global prevalence of, and risk factors for, uninvestigated dyspepsia: A meta-analysis. Gut 2015, 64, 1049–1057. [Google Scholar] [CrossRef]
- Ponce, J.; Beltrán, B.; Ponce, M.; Zapardiel, J.; Ortiz, V.; Vegazo, O.; Nuevo, J. Impact of gastroesophageal reflux disease on the quality of life of Spanish patients: The relevance of the biometric factors and the severity of symptoms. Eur. J. Gastroenterol. Hepatol. 2009, 21, 620–629. [Google Scholar] [CrossRef]
- Eusebi, L.H.; Ratnakumaran, R.; Yuan, Y.; Solaymani-Dodaran, M.; Bazzoli, F.; Ford, A.C. Global prevalence of, and risk factors for, gastro-oesophageal reflux symptoms: A meta-analysis. Gut 2018, 67, 430–440. [Google Scholar] [CrossRef]
- Gisbert, J.P.; Cooper, A.; Karagiannis, D.; Hatlebakk, J.; Agréus, L.; Jablonowski, H.; Nuevo, J. Impact of gastroesophageal reflux disease on work absenteeism, presenteeism and productivity in daily life: A European observational study. Health Qual. Life Outcomes 2009, 7, 90. [Google Scholar] [CrossRef] [Green Version]
- Actualización en El Manejo de Los Pacientes Con Acidez Y/O Reflujo en Atención Primaria Y Farmacia Comunitaria, 1st ed.; Medea, Medical Education Agency, S.L.: Madrid, Spain, 2019; ISBN 978-84-09-13267-6. Available online: https://www.campussefac.org/product/actualizacion-manejo-pacientes-con-acidez-o-reflujo-atencion-primaria-farmacia-comunitaria (accessed on 1 January 2022).
- Clarrett, D.M.; Hachem, C. Gastroesophageal Reflux Disease (GERD). Mo. Med. 2018, 115, 214–218. [Google Scholar]
- Tytgat, G.N.; Heading, R.C.; Müller-Lissner, S.; Kamm, M.A.; Schölmerich, J.; Berstad, A.; Fried, M.; Chaussade, S.; Jewell, D.; Briggs, A. Contemporary understanding and management of reflux and constipation in the general population and pregnancy: A consensus meeting. Aliment. Pharmacol. Ther. 2003, 18, 291–301. [Google Scholar] [CrossRef]
- Tytgat, G.N.; McColl, K.; Tack, J.; Holtmann, G.; Hunt, R.H.; Malfertheiner, P.; Hungin, A.P.; Batchelor, H.K. New algorithm for the treatment of gastro-oesophageal reflux disease. Aliment. Pharmacol. Ther. 2008, 27, 249–256. [Google Scholar] [CrossRef]
- LAW 16/1997, of 25 April 1997, Regulating Pharmacy Office Services. B.O.E. nº 100 of 26 April 1997:13450-2. Available online: https://www.boe.es/diario_boe/txt.php?id=BOE-A-1997-9022 (accessed on 6 August 2022).
- Louis, E.; Tack, J.; Vandenhoven, G.; Taeter, C. Evaluation of the GERD Impact Scale, an international, validated patient questionnaire, in daily practice. Results of the ALEGRIA study. Acta Gastro-Enterol. Belg. 2009, 72, 3–8. [Google Scholar]
- Nuevo, J.; Tafalla, M.; Zapardiel, J. Validation of the Reflux Disease Questionnaire (RDQ) and Gastrointestinal Impact Scale (GIS) in patients with gastroesophageal reflux disease in the Spanish population. Gastroenterol. Hepatol. 2009, 32, 264–273. [Google Scholar] [CrossRef]
- Vakil, N.B.; Halling, K.; Becher, A.; Rydén, A. Systematic review of patient-reported outcome instruments for gastroesophageal reflux disease symptoms. Eur. J. Gastroenterol. Hepatol. 2013, 25, 2–14. [Google Scholar] [CrossRef]
- Fuller, G.; Bolus, R.; Whitman, C.; Talley, J.; Erder, M.H.; Joseph, A.; Silberg, D.G.; Spiegel, B. PRISM, a Patient-Reported Outcome Instrument, Accurately Measures Symptom Change in Refractory Gastroesophageal Reflux Disease. Dig. Dis. Sci. 2017, 62, 593–606. [Google Scholar] [CrossRef]
- Chassany, O.; Shaheen, N.J.; Karlsson, M.; Hughes, N.; Rydén, A. Systematic review: Symptom assessment using patient-reported outcomes in gastroesophageal reflux disease and dyspepsia. Scand. J. Gastroenterol. 2012, 47, 1412–1421. [Google Scholar] [CrossRef] [PubMed]
- Becher, A.; El-Serag, H. Systematic review: The association between symptomatic response to proton pump inhibitors and health-related quality of life in patients with gastro-oesophageal reflux disease. Aliment. Pharmacol. Ther. 2011, 34, 618–627. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, K.H.; Musial, F.; Eypasch, E.; Meining, A. Gastrointestinal Quality of Life in Gastroesophageal Reflux Disease: A Systematic Review. Digestion 2022, 103, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Carle, P.D.; Uema, S.A.; Pérez, S.R.; Pallarés, M.M.; Uthurry, N.H.; Dáder, M.J. Validation of a questionnaire on patient satisfaction with the dispensing service in community pharmacies. Atención Primaria 2007, 39, 591–596. [Google Scholar]
- Vermersch, P.; Hobart, J.; Dive-Pouletty, C.; Bozzi, S.; Hass, S.; Coyle, P.K. Measuring treatment satisfaction in MS: Is the Treatment Satisfaction Questionnaire for Medication fit for purpose? Mult. Scler. 2017, 23, 604–613. [Google Scholar] [CrossRef] [Green Version]
- Westerlund, T.; Allebeck, P.; Marklund, B.; Andersson, I.L.; Brånstad, J.O.; Sjöblom, M. Evaluation of a model for counseling patients with dyspepsia in Swedish community pharmacies. Am. J. Health Syst. Pharm. 2003, 60, 1336–1341. [Google Scholar] [CrossRef]
- Branham, A.R.; Katz, A.J.; Moose, J.S.; Ferreri, S.P.; Farley, J.F.; Marciniak, M.W. Retrospective analysis of estimated cost avoidance following pharmacist-provided medication therapy management services. J. Pharm. Pract. 2013, 26, 420–427. [Google Scholar] [CrossRef]

| 1. How often have you had the following symptoms: a. Pain in your chest or behind the breastbone? b. Burning sensation in your chest or behind the breastbone? c. Regurgitation or acid taste in your mouth? d. Pain or burning in your upper stomach? e. Sore throat or hoarseness that is related to your heartburn or acid reflux? 2. How often have you had difficulty getting a good night’s sleep because of your symptoms? 3. How often have your symptoms prevented you from eating or drinking any of the foods you like? 4. How frequently have your symptoms kept you from being fully productive in your job or daily activities? 5. How often do you take additional medication other than what the clinician told you to take? |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-Pintor, E.; Puig-Moltó, M.; Lumbreras, B. EsReflux Protocol: Epidemiological Study of Heartburn and Reflux-like Symptoms in Spanish Community Pharmacies. Int. J. Environ. Res. Public Health 2022, 19, 9807. https://doi.org/10.3390/ijerph19169807
López-Pintor E, Puig-Moltó M, Lumbreras B. EsReflux Protocol: Epidemiological Study of Heartburn and Reflux-like Symptoms in Spanish Community Pharmacies. International Journal of Environmental Research and Public Health. 2022; 19(16):9807. https://doi.org/10.3390/ijerph19169807
Chicago/Turabian StyleLópez-Pintor, Elsa, María Puig-Moltó, and Blanca Lumbreras. 2022. "EsReflux Protocol: Epidemiological Study of Heartburn and Reflux-like Symptoms in Spanish Community Pharmacies" International Journal of Environmental Research and Public Health 19, no. 16: 9807. https://doi.org/10.3390/ijerph19169807
APA StyleLópez-Pintor, E., Puig-Moltó, M., & Lumbreras, B. (2022). EsReflux Protocol: Epidemiological Study of Heartburn and Reflux-like Symptoms in Spanish Community Pharmacies. International Journal of Environmental Research and Public Health, 19(16), 9807. https://doi.org/10.3390/ijerph19169807






