Raising Awareness on the Clinical and Forensic Aspects of Jellyfish Stings: A Worldwide Increasing Threat
Abstract
1. Introduction
2. Materials and Methods
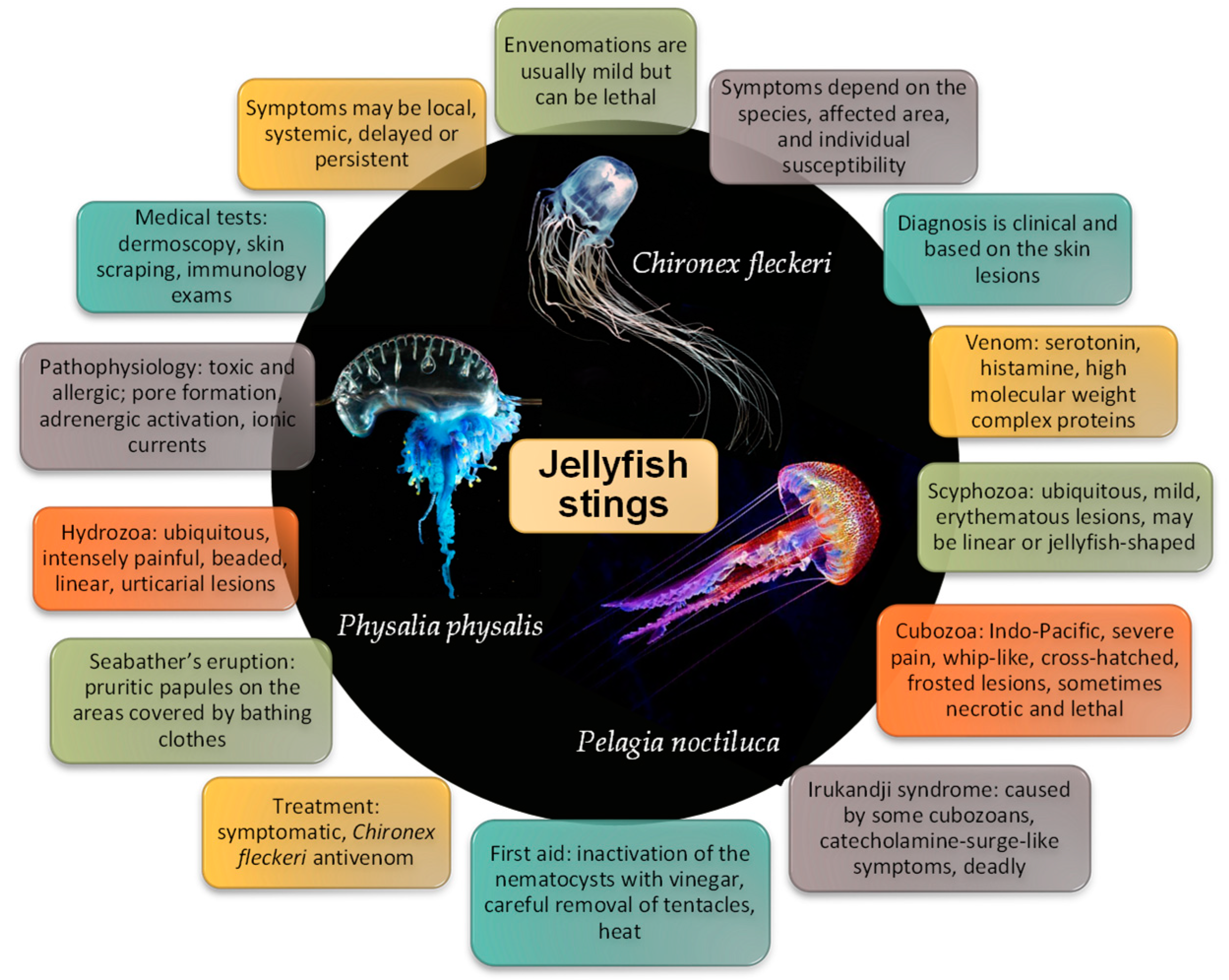
3. Jellyfish Biology
4. Toxicokinetics of Jellyfish Venom
5. Pathophysiology
5.1. Cardiotoxicity, Pore Formation, and Intracellular Ca2+ Overload
5.2. Induction of Na+ and K+ Currents
5.3. Targeting the Adrenergic System
5.4. Endothelial Nitric Oxide Synthase (eNOS) Induction
5.5. Tubule Length of the Nematocyst and Severity of the Pain
5.6. Hemostasis Disturbance
5.7. Other Reported Mechanisms of Toxicity
5.8. Delayed Reactions
5.9. Irukandji Syndrome
6. Envenomation Syndromes
6.1. Class Scyphozoa
6.2. Class Hydrozoa
6.3. Class Cubozoa
Irukandji Syndrome
6.4. Seabather’s Eruption
6.5. Delayed Reactions
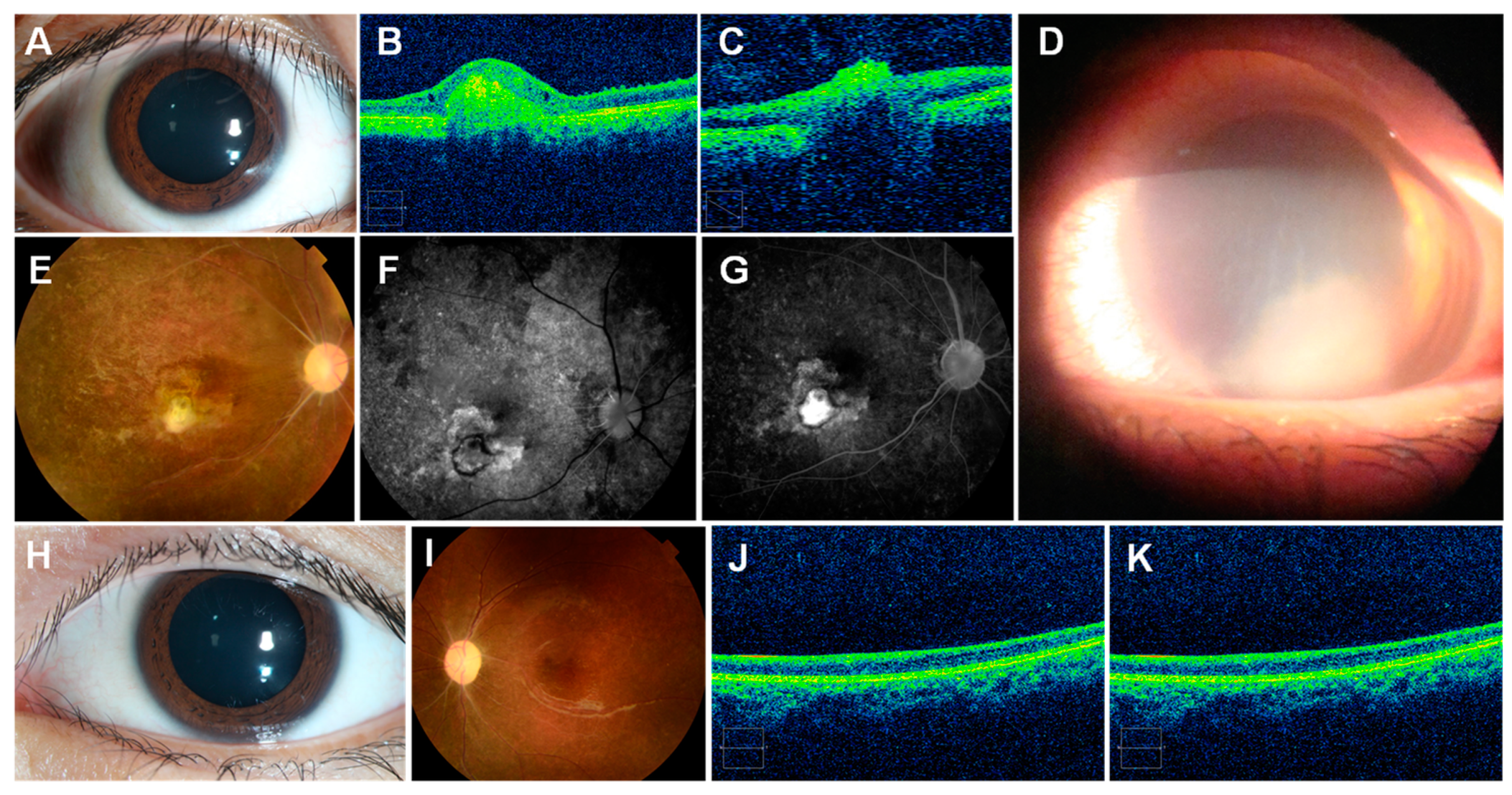
6.6. Eye Lesions
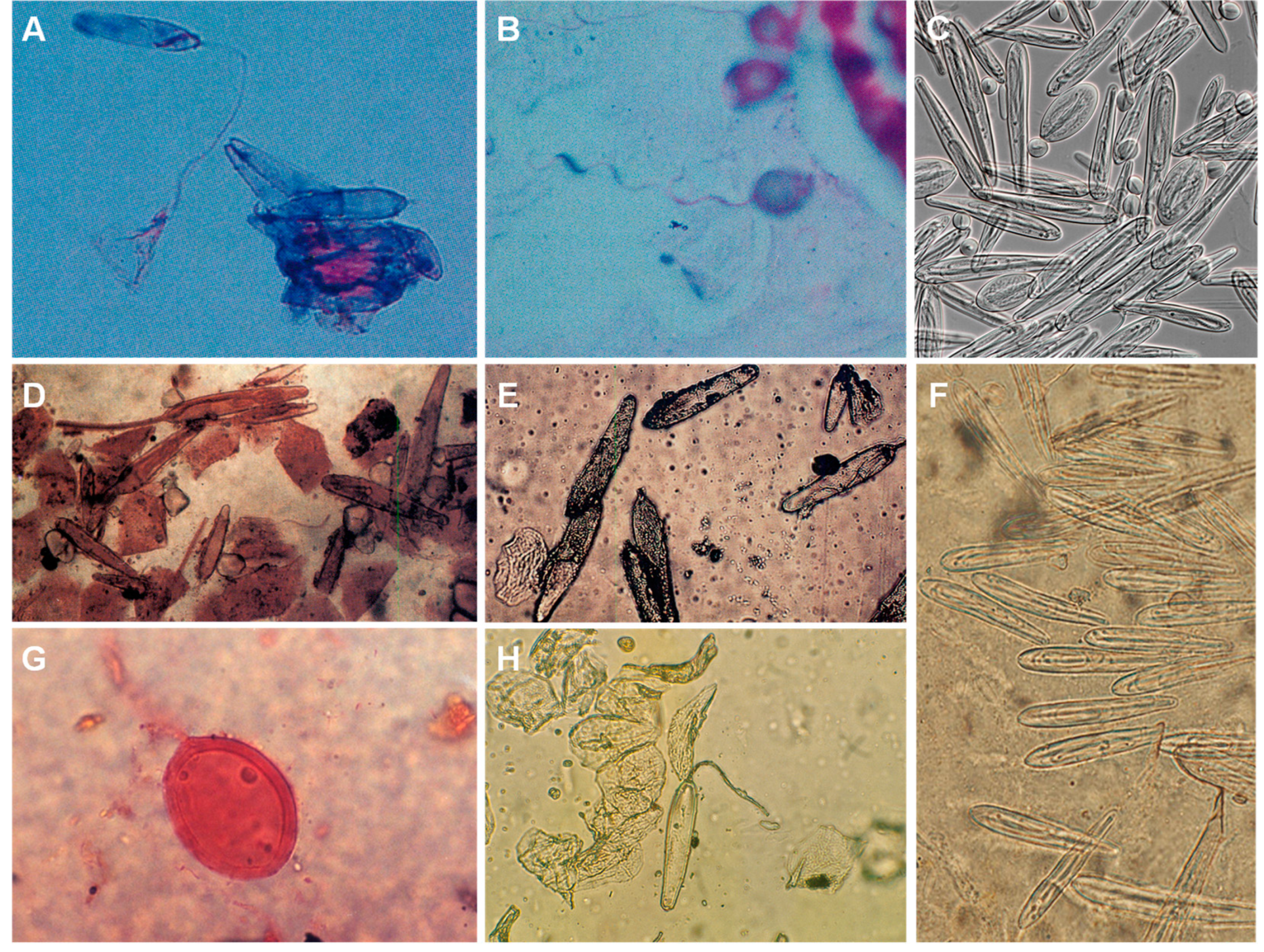
7. Diagnosis
7.1. Signs and Symptoms
7.2. Medical Exams
8. Treatment
- (a)
- Patient stabilization—victims should be removed from the water to prevent further stinging and drowning. If needed, life support maneuvers should be performed. The victim should be brought to emergency care if needed;
- (b)
- Preventing nematocyst discharge—care should be taken to minimize the venom load by preventing further discharge of the nematocysts. As such, vinegar is usually employed to prevent the discharge of the remaining nematocysts, but this use is based largely on empirical knowledge. Research regarding this use is conflicting. It is generally advised to use vinegar in Hydrozoan and Cubozoan stings and to avoid its use on Scyphozoan. Ballesteros et al. showed that vinegar leads to Pelagia noctiluca nematocysts discharge in vitro [201]. However, the most recent review on this topic suggested vinegar to be beneficial in Cyanea capillata and Pelagia noctiluca envenomation, so further research is needed [14,202]. After inactivating the nematocysts, adherent tentacles must be removed gently, using tweezers or a similar tool. Tentacle removal can also be attempted by rinsing with seawater [203], however, some studies recommend against this practice, as it can induce further venom delivery [202]. Freshwater must not be used for rinsing, as the osmotic challenge would induce nematocyst discharge [119]. An in vitro study found that lidocaine might block nematocyst discharge from Pelagia noctiluca [25]. Moreover, topical lidocaine was demonstrated to both reduce pain and prevent nematocyst release [25].
- (c)
- (d)
- Controlling systemic effects—analgesics, antihistamines, corticosteroids, anti-hypertensive drugs are some of the treatments that may be required, depending on the symptoms;
- (e)
- Preventing complications—if a sting is at risk of becoming infected, antibiotics should cover Streptococcus spp., Staphylococcus spp., and marine organisms, such as Vibrio spp. According to Center for Disease Control recommendations, tetanus prophylaxis is indicated whenever there is a minor wound, and the patient is not up to date with the tetanus vaccination scheme. As such, tetanus prophylaxis must also be considered [6,76,206].
8.1. Particularities of Each Class
8.2. Irukandji Syndrome
8.3. Seabather´s Eruption
8.4. Corneal Lesions
8.5. Other Treatments
9. Forensic and Toxicological Aspects
10. Climate Changes and the Increased Risk of Jellyfish Stings
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- D’Ambra, I.; Lauritano, C. A Review of Toxins from Cnidaria. Mar. Drugs 2020, 18, 507. [Google Scholar] [CrossRef] [PubMed]
- Needleman, R.K.; Neylan, I.P.; Erickson, T.B. Environmental and Ecological Effects of Climate Change on Venomous Marine and Amphibious Species in the Wilderness. Wilderness Environ. Med. 2018, 29, 343–356. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, C.; De Haro, L. Clinical marine toxicology: A European perspective for clinical toxicologists and poison centers. Toxins 2013, 5, 1343–1352. [Google Scholar] [CrossRef]
- Kim, E.; Lee, S.; Kim, J.S.; Yoon, W.D.; Lim, D.; Hart, A.J.; Hodgson, W.C. Cardiovascular effects of Nemopilema nomurai (Scyphozoa: Rhizostomeae) jellyfish venom in rats. Toxicol. Lett. 2006, 167, 205–211. [Google Scholar] [CrossRef]
- Boulware, D.R. A randomized, controlled field trial for the prevention of jellyfish stings with a topical sting inhibitor. J. Travel Med. 2006, 13, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Killi, N.; Mariottini, G.L. Cnidarian Jellyfish: Ecological Aspects, Nematocyst Isolation, and Treatment Methods of Sting. Mar. Org. Model Syst. Biol. Med. 2018, 65, 477–513. [Google Scholar] [CrossRef]
- Dinis-Oliveira, R.J. One image is worth more than a thousand words: Producing an atlas of medical signs for teaching clinical and forensic toxicology. Forensic Sci. Res. 2022, in press. [CrossRef]
- Dinis-Oliveira, R.J.; Carvalho, F.; Duarte, J.A.; Proença, J.B.; Santos, A.; Magalhães, T. Clinical and forensic signs related to cocaine abuse. Curr. Drug Abus. Rev. 2012, 5, 64–83. [Google Scholar] [CrossRef]
- Dinis-Oliveira, R.J.; Carvalho, F.; Moreira, R.; Duarte, J.A.; Proenca, J.B.; Santos, A.; Magalhaes, T. Clinical and forensic signs related to opioids abuse. Curr. Drug Abus. Rev. 2012, 5, 273–290. [Google Scholar] [CrossRef]
- Dinis-Oliveira, R.J. Metabolic Profiles of Propofol and Fospropofol: Clinical and Forensic Interpretative Aspects. Biomed. Res. Int. 2018, 2018, 6852857. [Google Scholar] [CrossRef]
- Dinis-Oliveira, R.J.; Magalhães, T.; Moreira, R.; Proença, J.B.; Pontes, H.; Santos, A.; Duarte, J.A.; Carvalho, F. Clinical and forensic signs related to ethanol abuse: A mechanistic approach. Toxicol. Mech. Methods 2014, 24, 81–110. [Google Scholar] [CrossRef]
- Horton, T.; Kroh, A.; Ahyong, S.; Bailly, N.; Bieler, R.; Boyko, C.B.; Brandão, S.N.; Gofas, S.; Hooper, J.N.A.; Hernandez, F.; et al. World Register of Marine Species (WoRMS); WoRMS Editorial Board: Ostend, Belgium, 2022. [Google Scholar]
- Sujanitha, V.; Sivansuthan, S.; Luckshman, W.V.; Gnaneswaran, R.; Jeyakanth, T.; Gunarathna, U. The clinical manifestations, outcome and identification of jellyfish stings in Jaffna, Sri Lanka. Trop. Dr. 2017, 47, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Remigante, A.; Costa, R.; Morabito, R.; La Spada, G.; Marino, A.; Dossena, S. Impact of Scyphozoan Venoms on Human Health and Current First Aid Options for Stings. Toxins 2018, 10, 133. [Google Scholar] [CrossRef] [PubMed]
- Burnett, J.W.; Calton, G.J.; Burnett, H.W.; Mandojana, R.M. Local and systemic reactions from jellyfish stings. Clin. Dermatol. 1987, 5, 14–28. [Google Scholar] [CrossRef]
- Carrette, T.J.; Underwood, A.H.; Seymour, J.E. Irukandji syndrome: A widely misunderstood and poorly researched tropical marine envenoming. Diving Hyperb. Med. 2012, 42, 214–223. [Google Scholar]
- Horiike, T.; Nagai, H.; Kitani, S. Identification of allergens in the box jellyfish Chironex yamaguchii that cause sting dermatitis. Int. Arch. Allergy Immunol. 2015, 167, 73–82. [Google Scholar] [CrossRef]
- Haddad, V., Jr. Environmental dermatology: Skin manifestations of injuries caused by invertebrate aquatic animals. An. Bras. Dermatol. 2013, 88, 496–506. [Google Scholar] [CrossRef]
- Tibballs, J.; Yanagihara, A.A.; Turner, H.C.; Winkel, K. Immunological and toxinological responses to jellyfish stings. Inflamm Allergy Drug Targets 2011, 10, 438–446. [Google Scholar] [CrossRef]
- Mariottini, G.L.; Pane, L. Mediterranean jellyfish venoms: A review on scyphomedusae. Mar. Drugs 2010, 8, 1122–1152. [Google Scholar] [CrossRef]
- Xiao, L.; Liu, G.S.; Wang, Q.Q.; He, Q.; Liu, S.H.; Li, Y.; Zhang, J.; Zhang, L.M. The lethality of tentacle-only extract from jellyfish Cyanea capillata is primarily attributed to cardiotoxicity in anaesthetized SD rats. Toxicon 2010, 55, 838–845. [Google Scholar] [CrossRef]
- Liang, X.; Beilei, W.; Ying, L.; Qianqian, W.; Sihua, L.; Yang, W.; Guoyan, L.; Jia, L.; Xuting, Y.; Liming, Z. Cardiovascular effect is independent of hemolytic toxicity of tentacle-only extract from the jellyfish Cyanea capillata. PLoS ONE 2012, 7, e43096. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, Q.; Xiao, L.; Zhang, L. Intervention effects of five cations and their correction on hemolytic activity of tentacle extract from the jellyfish Cyanea capillata. PeerJ 2017, 5, e3338. [Google Scholar] [CrossRef] [PubMed]
- Mariottini, G.L. Hemolytic venoms from marine cnidarian jellyfish—An overview. J. Venom. Res. 2014, 5, 22–32. [Google Scholar] [PubMed]
- Morabito, R.; Marino, A.; Dossena, S.; La Spada, G. Nematocyst discharge in Pelagia noctiluca (Cnidaria, Scyphozoa) oral arms can be affected by lidocaine, ethanol, ammonia and acetic acid. Toxicon 2014, 83, 52–58. [Google Scholar] [CrossRef]
- Ponce, D.; Brinkman, D.L.; Potriquet, J.; Mulvenna, J. Tentacle Transcriptome and Venom Proteome of the Pacific Sea Nettle, Chrysaora fuscescens (Cnidaria: Scyphozoa). Toxins 2016, 8, 102. [Google Scholar] [CrossRef]
- Badré, S. Bioactive toxins from stinging jellyfish. Toxicon 2014, 91, 114–125. [Google Scholar] [CrossRef]
- Morabito, R.; Condello, S.; Currò, M.; Marino, A.; Ientile, R.; La Spada, G. Oxidative stress induced by crude venom from the jellyfish Pelagia noctiluca in neuronal-like differentiated SH-SY5Y cells. Toxicol. In Vitro 2012, 26, 694–699. [Google Scholar] [CrossRef]
- Mc, D.T.D.; Pereira, P.; Seymour, J.; Winkel, K.D. A sting from an unknown jellyfish species associated with persistent symptoms and raised troponin I levels. Emerg. Med. 2002, 14, 175–180. [Google Scholar] [CrossRef]
- Bianchi, R.; Torella, D.; Spaccarotella, C.; Mongiardo, A.; Indolfi, C. Mediterranean jellyfish sting-induced Tako-Tsubo cardiomyopathy. Eur. Heart J. 2011, 32, 18. [Google Scholar] [CrossRef][Green Version]
- Salam, A.M.; Albinali, H.A.; Gehani, A.A.; Al Suwaidi, J. Acute myocardial infarction in a professional diver after jellyfish sting. Mayo Clin. Proc. 2003, 78, 1557–1560. [Google Scholar] [CrossRef]
- Little, M.; Pereira, P.; Mulcahy, R.; Cullen, P.; Carrette, T.; Seymour, J. Severe cardiac failure associated with presumed jellyfish sting. Irukandji syndrome? Anaesth. Intensive Care 2003, 31, 642–647. [Google Scholar] [CrossRef] [PubMed]
- Tibballs, J.; Li, R.; Tibballs, H.A.; Gershwin, L.A.; Winkel, K.D. Australian carybdeid jellyfish causing “Irukandji syndrome”. Toxicon 2012, 59, 617–625. [Google Scholar] [CrossRef]
- Mustafa, M.R.; White, E.; Hongo, K.; Othman, I.; Orchard, C.H. The mechanism underlying the cardiotoxic effect of the toxin from the jellyfish Chironex fleckeri. Toxicol. Appl. Pharm. 1995, 133, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Burnett, J.W.; Gean, C.J.; Calton, G.J.; Warnick, J.E. The effect of verapamil on the cardiotoxic activity of Portuguese man-o’war (Physalia physalis) and sea nettle (Chrysaora quinquecirrha) venoms. Toxicon 1985, 23, 681–689. [Google Scholar] [CrossRef]
- Azuma, H.; Ishikawa, M.; Nakajima, T.; Satoh, A.; Sekizaki, S. Calcium-dependent contractile response of arterial smooth muscle to a jellyfish toxin (pCrTX: Carybdea rastonii). Br. J. Pharmacol. 1986, 88, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, H.; Karaki, H.; Nagase, H.; Urakawa, N.; Azuma, H.; Nakajima, T. Contractile effects of jellyfish toxin extracted from Carybdea rastonii on isolated rabbit aorta. Jpn. J. Pharmacol. 1986, 42, 425–430. [Google Scholar] [CrossRef]
- Burnett, J.W.; Calton, G.J. Response of the box-jellyfish (Chironex fleckeri) cardiotoxin to intravenous administration of verapamil. Med. J. Aust. 1983, 2, 192–194. [Google Scholar] [CrossRef]
- Beilei, W.; Lin, Z.; Qian, H.; Qianqian, W.; Tao, W.; Jia, L.; Xiaojuan, W.; Xuting, Y.; Liang, X.; Liming, Z. Direct cardiac toxicity of the tentacle-only extract from the jellyfish Cyanea capillata demonstrated in isolated rat heart. J. Cardiovasc. Pharm. 2012, 59, 331–338. [Google Scholar] [CrossRef]
- Bailey, P.M.; Bakker, A.J.; Seymour, J.E.; Wilce, J.A. A functional comparison of the venom of three Australian jellyfish—Chironex fleckeri, Chiropsalmus sp., and Carybdea xaymacana—On cytosolic Ca2+, haemolysis and Artemia sp. lethality. Toxicon 2005, 45, 233–242. [Google Scholar] [CrossRef]
- Chung, J.J.; Ratnapala, L.A.; Cooke, I.M.; Yanagihara, A.A. Partial purification and characterization of a hemolysin (CAH1) from Hawaiian box jellyfish (Carybdea alata) venom. Toxicon 2001, 39, 981–990. [Google Scholar] [CrossRef]
- Nagai, H.; Takuwa, K.; Nakao, M.; Ito, E.; Miyake, M.; Noda, M.; Nakajima, T. Novel proteinaceous toxins from the box jellyfish (Sea Wasp) Carybdea rastoni. Biochem. Biophys. Res. Commun. 2000, 275, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Nagai, H.; Takuwa-Kuroda, K.; Nakao, M.; Oshiro, N.; Iwanaga, S.; Nakajima, T. A novel protein toxin from the deadly box jellyfish (Sea Wasp, Habu-kurage) Chiropsalmus quadrigatus. Biosci. Biotechnol. Biochem. 2002, 66, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Brinkman, D.L.; Aziz, A.; Loukas, A.; Potriquet, J.; Seymour, J.; Mulvenna, J. Venom proteome of the box jellyfish Chironex fleckeri. PLoS ONE 2012, 7, e47866. [Google Scholar] [CrossRef] [PubMed]
- Koyama, T.; Noguchi, K.; Matsuzaki, T.; Sakanashi, M.; Nakasone, J.; Miyagi, K.; Sakanashi, M.; Sakanashi, M. Haemodynamic effects of the crude venom from nematocysts of the box-jellyfish Chiropsalmus quadrigatus (Habu-kurage) in anaesthetized rabbits. Toxicon 2003, 41, 621–631. [Google Scholar] [CrossRef]
- Morabito, R.; Costa, R.; Rizzo, V.; Remigante, A.; Nofziger, C.; La Spada, G.; Marino, A.; Paulmichl, M.; Dossena, S. Crude venom from nematocysts of Pelagia noctiluca (Cnidaria: Scyphozoa) elicits a sodium conductance in the plasma membrane of mammalian cells. Sci. Rep. 2017, 7, 41065. [Google Scholar] [CrossRef]
- Frazão, B.; Campos, A.; Osório, H.; Thomas, B.; Leandro, S.; Teixeira, A.; Vasconcelos, V.; Antunes, A. Analysis of Pelagia noctiluca proteome Reveals a Red Fluorescent Protein, a Zinc Metalloproteinase and a Peroxiredoxin. Protein J. 2017, 36, 77–97. [Google Scholar] [CrossRef]
- Lee, H.; Jung, E.S.; Kang, C.; Yoon, W.D.; Kim, J.S.; Kim, E. Scyphozoan jellyfish venom metalloproteinases and their role in the cytotoxicity. Toxicon 2011, 58, 277–284. [Google Scholar] [CrossRef]
- Ramasamy, S.; Isbister, G.K.; Seymour, J.E.; Hodgson, W.C. The in vivo cardiovascular effects of the Irukandji jellyfish (Carukia barnesi) nematocyst venom and a tentacle extract in rats. Toxicol. Lett. 2005, 155, 135–141. [Google Scholar] [CrossRef]
- Winkel, K.D.; Tibballs, J.; Molenaar, P.; Lambert, G.; Coles, P.; Ross-Smith, M.; Wiltshire, C.; Fenner, P.J.; Gershwin, L.A.; Hawdon, G.M.; et al. Cardiovascular actions of the venom from the Irukandji (Carukia barnesi) jellyfish: Effects in human, rat and guinea-pig tissues in vitro and in pigs in vitro. Clin. Exp. Pharmacol. Physiol. 2005, 32, 777–788. [Google Scholar] [CrossRef]
- Li, R.; Wright, C.E.; Winkel, K.D.; Gershwin, L.A.; Angus, J.A. The pharmacology of Malo maxima jellyfish venom extract in isolated cardiovascular tissues: A probable cause of the Irukandji syndrome in Western Australia. Toxicol. Lett. 2011, 201, 221–229. [Google Scholar] [CrossRef]
- Ramasamy, S.; Isbister, G.K.; Seymour, J.E.; Hodgson, W.C. The in vivo cardiovascular effects of an Australasian box jellyfish (Chiropsalmus sp.) venom in rats. Toxicon 2005, 45, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Ramasamy, S.; Isbister, G.K.; Seymour, J.E.; Hodgson, W.C. Pharmacologically distinct cardiovascular effects of box jellyfish (Chironex fleckeri) venom and a tentacle-only extract in rats. Toxicol. Lett. 2005, 155, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Hughes, R.J.; Angus, J.A.; Winkel, K.D.; Wright, C.E. A pharmacological investigation of the venom extract of the Australian box jellyfish, Chironex fleckeri, in cardiac and vascular tissues. Toxicol. Lett. 2012, 209, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhang, H.; Wang, B.; Wang, C.; Xiao, L.; Zhang, L. β adrenergic receptor/cAMP/PKA signaling contributes to the intracellular Ca(2+) release by tentacle extract from the jellyfish Cyanea capillata. BMC Pharmacol. Toxicol. 2017, 18, 60. [Google Scholar] [CrossRef]
- Bueno, T.C.; Collaço, R.C.; Cardoso, B.A.; Bredariol, R.F.; Escobar, M.L.; Cajado, I.B.; Gracia, M.; Antunes, E.; Zambelli, V.O.; Picolo, G.; et al. Neurotoxicity of Olindias sambaquiensis and Chiropsalmus quadrumanus extracts in sympathetic nervous system. Toxicon 2021, 199, 127–138. [Google Scholar] [CrossRef]
- Wang, B.; Liu, D.; Wang, C.; Wang, Q.; Zhang, H.; Liu, G.; Tao, X.; Zhang, L. Mechanism of endothelial nitric oxide synthase phosphorylation and activation by tentacle extract from the jellyfish Cyanea capillata. PeerJ 2017, 5, e3172. [Google Scholar] [CrossRef]
- Kitatani, R.; Yamada, M.; Kamio, M.; Nagai, H. Length Is Associated with Pain: Jellyfish with Painful Sting Have Longer Nematocyst Tubules than Harmless Jellyfish. PLoS ONE 2015, 10, e0135015. [Google Scholar] [CrossRef]
- Cuypers, E.; Yanagihara, A.; Karlsson, E.; Tytgat, J. Jellyfish and other cnidarian envenomations cause pain by affecting TRPV1 channels. FEBS Lett. 2006, 580, 5728–5732. [Google Scholar] [CrossRef]
- Cromer, B.A.; McIntyre, P. Painful toxins acting at TRPV1. Toxicon 2008, 51, 163–173. [Google Scholar] [CrossRef]
- Rastogi, A.; Sarkar, A.; Chakrabarty, D. Partial purification and identification of a metalloproteinase with anticoagulant activity from Rhizostoma pulmo (Barrel Jellyfish). Toxicon 2017, 132, 29–39. [Google Scholar] [CrossRef]
- Bae, S.K.; Lee, H.; Heo, Y.; Pyo, M.J.; Choudhary, I.; Han, C.H.; Yoon, W.D.; Kang, C.; Kim, E. In vitro characterization of jellyfish venom fibrin (ogen) olytic enzymes from Nemopilema nomurai. J. Venom. Anim. Toxins Incl. Trop. Dis. 2017, 23, 35. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, A.; Biswas, S.; Sarkar, A.; Chakrabarty, D. Anticoagulant activity of Moon jellyfish (Aurelia aurita) tentacle extract. Toxicon 2012, 60, 719–723. [Google Scholar] [CrossRef] [PubMed]
- Yue, Y.; Yu, H.; Li, R.; Li, P. Topical Exposure to Nemopilema nomurai Venom Triggers Oedematogenic Effects: Enzymatic Contribution and Identification of Venom Metalloproteinase. Toxins 2021, 13, 44. [Google Scholar] [CrossRef] [PubMed]
- Bruschetta, G.; Impellizzeri, D.; Morabito, R.; Marino, A.; Ahmad, A.; Spanò, N.; Spada, G.L.; Cuzzocrea, S.; Esposito, E. Pelagia noctiluca (Scyphozoa) crude venom injection elicits oxidative stress and inflammatory response in rats. Mar. Drugs 2014, 12, 2182–2204. [Google Scholar] [CrossRef]
- Wang, T.; Wen, X.J.; Mei, X.B.; Wang, Q.Q.; He, Q.; Zheng, J.M.; Zhao, J.; Xiao, L.; Zhang, L.M. Lipid peroxidation is another potential mechanism besides pore-formation underlying hemolysis of tentacle extract from the jellyfish Cyanea capillata. Mar. Drugs 2013, 11, 67–80. [Google Scholar] [CrossRef]
- Ayed, Y.; Chayma, B.; Hayla, A.; Abid, S.; Bacha, H. Is cell death induced by nematocysts extract of medusa Pelagia noctiluca related to oxidative stress? Environ. Toxicol. 2013, 28, 498–506. [Google Scholar] [CrossRef]
- Nevalainen, T.J.; Peuravuori, H.J.; Quinn, R.J.; Llewellyn, L.E.; Benzie, J.A.; Fenner, P.J.; Winkel, K.D. Phospholipase A2 in cnidaria. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2004, 139, 731–735. [Google Scholar] [CrossRef]
- Cormier, S.M. Exocytotic and cytolytic release of histamine from mast cells treated with Portuguese man-of-war (Physalia physalis) venom. J. Exp. Zool. 1984, 231, 1–10. [Google Scholar] [CrossRef]
- Uvnas, B. A toxic (histamine-releasing) principle from tentacles of Cyanea capillata (the stinging jelly-fish). J. Med. Pharm. Chem. 1961, 4, 511–515. [Google Scholar] [CrossRef]
- Guevara, B.E.; Dayrit, J.F.; Haddad, V., Jr. Delayed allergic dermatitis presenting as a keloid-like reaction caused by sting from an Indo-Pacific Portuguese man-o’-war (Physalia utriculus). Clin. Exp. Derm. 2017, 42, 182–184. [Google Scholar] [CrossRef]
- Tiong, K. Irukandji syndrome, catecholamines, and mid-ventricular stress cardiomyopathy. Eur. J. Echocardiogr. 2009, 10, 334–336. [Google Scholar] [CrossRef] [PubMed]
- Tibballs, J. Australian venomous jellyfish, envenomation syndromes, toxins and therapy. Toxicon 2006, 48, 830–859. [Google Scholar] [CrossRef] [PubMed]
- Burnett, J.W.; Calton, G.J. Jellyfish envenomation syndromes updated. Ann. Emerg. Med. 1987, 16, 1000–1005. [Google Scholar] [CrossRef]
- Kim, J.H.; Han, S.B.; Durey, A. Fatal Pulmonary Edema in a Child After Jellyfish Stings in Korea. Wilderness Environ. Med. 2018, 29, 527–530. [Google Scholar] [CrossRef]
- Sridhar, S.C.; Deo, S.C. Marine and Other Aquatic Dermatoses. Indian J. Dermatol. 2017, 62, 66–78. [Google Scholar] [CrossRef] [PubMed]
- Auerbach, P.S.; Gupta, D.; Van Hoesen, K.; Zavala, A. Dermatological Progression of a Probable Box Jellyfish Sting. Wilderness Environ. Med. 2019, 30, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Tønseth, K.A. Health damage after jellyfish stings. Tidsskr. Den Nor. Legeforen. 2007, 127, 1777–1778. [Google Scholar]
- Shabani, M.; Saffaei, A.; Asghari, M.; Sahraei, Z. Jellyfish Stings Rarely Induced Infectious Cellulitis: First Aid Remedies as Double-Edged Sword. Adv. J. Emerg. Med. 2020, 4, e33. [Google Scholar] [CrossRef] [PubMed]
- Thaikruea, L.; Siriariyaporn, P. Severe Dermatonecrotic Toxin and Wound Complications Associated With Box Jellyfish Stings 2008–2013. J. Wound Ostomy Cont. Nurs. 2015, 42, 599–604. [Google Scholar] [CrossRef]
- Rossetto, A.L.; Cruz, C.C.B.; Pereira, I.C.C.; Nunes, J.A.; Martins, M.M.; Nicolacópulos, T.; Rossetto, A.L.; Haddad Junior, V. Diagnostic confusion between seabather’s eruption as well as dermatophytosis and parasitic infestations. Rev. Soc. Bras. Med. Trop. 2020, 53, e20190462. [Google Scholar] [CrossRef]
- Desax-Willer, D.; Krebs, T.; Christen, S. Delayed deep dermal necrosis after jellyfish sting in a 4-year-old female infant. Case Rep. Plast Surg. Hand Surg. 2018, 5, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Giavedoni, P.; Velasco, V.; Morgado-Carrasco, D.; Nogué, S.; Mascaró Galy, J.M. Delayed Allergic Reactions to Jellyfish Stings: Usefulness of Ultrasonographic Evaluation. J. Ultrasound Med. 2018, 37, 2721–2724. [Google Scholar] [CrossRef] [PubMed]
- Manabe, Y.; Mabuchi, T.; Kawai, M.; Ota, T.; Ikoma, N.; Ozawa, A.; Horita, T. A Case of Delayed Flare-up Allergic Dermatitis Caused by Jellyfish Sting. Tokai J. Exp. Clin. Med. 2014, 39, 90–94. [Google Scholar] [PubMed]
- Veraldi, S.; Carrera, C. Delayed cutaneous reaction to jellyfish. Int. J. Dermatol. 2000, 39, 28–29. [Google Scholar] [CrossRef]
- Ohtaki, N.; Satoh, A.; Azuma, H.; Nakajima, T. Delayed flare-up reactions caused by jellyfish. Dermatologica 1986, 172, 98–103. [Google Scholar] [CrossRef]
- Ulrich, H.; Landthaler, M.; Vogt, T. Granulomatous jellyfish dermatitis. J. Dtsch Dermatol. Ges. 2007, 5, 493–495. [Google Scholar] [CrossRef] [PubMed]
- Yaffee, H.S. A delayed cutaneous reaction following contact with jellyfish. Dermatol. Int. 1968, 7, 75–77. [Google Scholar] [CrossRef] [PubMed]
- Di Costanzo, L.; Balato, N.; Zagaria, O.; Balato, A. Successful management of a delayed and persistent cutaneous reaction to jellyfish with pimecrolimus. J. Dermatol. Treat. 2009, 20, 179–180. [Google Scholar] [CrossRef]
- Rallis, E.; Limas, C. Recurrent dermatitis after solitary envenomation by jellyfish partially responded to tacrolimus ointment 0.1%. J. Eur. Acad. Dermatol. Venereol. 2007, 21, 1287–1288. [Google Scholar] [CrossRef]
- Burnett, J.W.; Calton, G.J. Recurrent eruption following a solitary envenomation by the cnidarian Stomolophous meleagris. Toxicon 1985, 23, 1010–1014. [Google Scholar] [CrossRef]
- Menahem, S.; Shvartzman, P. Recurrent dermatitis from jellyfish envenomation. Can. Fam. Phys. 1994, 40, 2116–2118. [Google Scholar]
- Dellanna, L.; Hirche, F.; Capra, V. Successful Treatment of Recurrent Dermatitis after Physalia physalis (Portuguese Man O’ War) Envenomation with Extracorporeal Shock Wave Therapy. Case Rep. Derm. 2021, 13, 202–208. [Google Scholar] [CrossRef]
- Chand, R.P.; Selliah, K. Reversible parasympathetic dysautonomia following stinging attributed to the box jelly fish (Chironex fleckeri). Aust. New Zealand J. Med. 1984, 14, 673–675. [Google Scholar] [CrossRef] [PubMed]
- Ponampalam, R. An unusual case of paralytic ileus after jellyfish envenomation. Emerg. Med. J. 2002, 19, 357–358. [Google Scholar] [CrossRef]
- Burnett, J.W. Prolonged urinary incontinence and biliary dyskinesia following abdominal contact with jellyfish tentacles. Wilderness Environ. Med. 2006, 17, 180–186. [Google Scholar] [CrossRef]
- Adiga, K.M. Brachial artery spasm as a result of a sting. Med. J. Aust. 1984, 140, 181–182. [Google Scholar] [CrossRef]
- Williamson, J.A.; Burnett, J.W.; Fenner, P.J.; Hach-Wunderle, V.; Hoe, L.Y.; Adiga, K.M. Acute regional vascular insufficiency after jellyfish envenomation. Med. J. Aust. 1988, 149, 698–701. [Google Scholar] [CrossRef] [PubMed]
- Choong, C.; Chan, H.Z.; Faruk, N.A.; Bea, K.C.; Zulkiflee, O. Jellyfish Envenomation Resulting In Vascular Insufficiency And Neurogenic Injury of Upper Limb. Malays. Orthop. J. 2015, 9, 49–51. [Google Scholar] [CrossRef]
- Glasser, D.B.; Noell, M.J.; Burnett, J.W.; Kathuria, S.S.; Rodrigues, M.M. Ocular jellyfish stings. Ophthalmology 1992, 99, 1414–1418. [Google Scholar] [CrossRef]
- Burnett, H.W.; Burnett, J.W. Prolonged blurred vision following coelenterate envenomation. Toxicon 1990, 28, 731–733. [Google Scholar] [CrossRef]
- Little, M.; Seymour, J. Another cause of “Irukandji stingings”. Med. J. Aust. 2003, 179, 654. [Google Scholar] [CrossRef] [PubMed]
- de Pender, A.M.; Winkel, K.D.; Ligthelm, R.J. A probable case of Irukandji syndrome in Thailand. J. Travel Med. 2006, 13, 240–243. [Google Scholar] [CrossRef] [PubMed]
- Fenner, P.J.; Lippmann, J. Severe Irukandji-like jellyfish stings in Thai waters. Diving Hyperb Med. 2009, 39, 175–177. [Google Scholar] [PubMed]
- Grady, J.D.; Burnett, J.W. Irukandji-like syndrome in South Florida divers. Ann. Emerg. Med. 2003, 42, 763–766. [Google Scholar] [CrossRef]
- Sando, J.J.; Usher, K. Case review: A 28-year-old Korean man with Irukandji syndrome. Int. Emerg. Nurs. 2009, 17, 72–76. [Google Scholar] [CrossRef] [PubMed]
- McIver, L.J.; Tjhung, I.G.; Parish, S.T.; Derkenne, R.C.; Kippin, A.N. Irukandji sydrome in the Torres Strait: A series of 8 cases. Wilderness Environ. Med. 2011, 22, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Thaikruea, L. Irukandji-like syndrome caused by single-tentacle box jellyfish found in Thailand, 2007–2019. Int. Marit. Health 2020, 71, 91–96. [Google Scholar] [CrossRef]
- Filling-Katz, M.R. Mononeuritis multiplex following jellyfish stings. Ann. Neurol. 1984, 15, 213. [Google Scholar] [CrossRef]
- Burnett, J.W.; Williamson, J.A.; Fenner, P.J. Mononeuritis multiplex after coelenterate sting. Med. J. Aust. 1994, 161, 320–322. [Google Scholar] [CrossRef]
- Peel, N.; Kandler, R. Localized neuropathy following jellyfish sting. Postgrad. Med. J. 1990, 66, 953–954. [Google Scholar] [CrossRef]
- Al-Ajmi, A.M.; Jayappa, S.; Rousseff, R.T. Isolated severe median mononeuropathy caused by a jellyfish sting. J. Clin. Neuromuscul. Dis. 2013, 14, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Hach-Wunderle, V.; Mebs, D.; Frederking, K.; Breddin, H.K. Jellyfish poisoning. Dtsch. Med. Wochenschr. 1987, 112, 1865–1868. [Google Scholar] [CrossRef] [PubMed]
- Laing, J.H.; Harrison, D.H. Envenomation by the box-jellyfish--an unusual cause of ulnar nerve palsy. J. R. Soc. Med. 1991, 84, 115–116. [Google Scholar] [CrossRef]
- Pang, K.A.; Schwartz, M.S. Guillain-Barré syndrome following jellyfish stings (Pelagia noctiluca). J. Neurol. Neurosurg. Psychiatry 1993, 56, 1133. [Google Scholar] [CrossRef]
- Devere, R. Guillain-Barré syndrome after a jellyfish sting. J. Clin. Neuromuscul. Dis. 2011, 12, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Azari, P.; Lu, Y.; Clarke, C.F.; Collins, T.; Briones, D.; Huh, B. Pathophysiology of the spreading of complex regional pain syndrome revisited: A case report. Neuromodulation 2011, 14, 428–431; discussion 431. [Google Scholar] [CrossRef] [PubMed]
- Burnett, J.W. Dysphonia: A new addition to jellyfish envenomation syndromes. Wilderness Environ. Med. 2005, 16, 117–118. [Google Scholar] [CrossRef]
- Cegolon, L.; Heymann, W.C.; Lange, J.H.; Mastrangelo, G. Jellyfish stings and their management: A review. Mar. Drugs 2013, 11, 523–550. [Google Scholar] [CrossRef]
- Spielman, F.J.; Bowe, E.A.; Watson, C.B.; Klein, E.F., Jr. Acute renal failure as a result of Physalia physalis sting. South Med. J. 1982, 75, 1425–1426. [Google Scholar] [CrossRef]
- Pereira, J.C.C.; Szpilman, D.; Haddad Junior, V. Anaphylactic reaction/angioedema associated with jellyfish sting. Rev. Soc. Bras. Med. Trop. 2018, 51, 115–117. [Google Scholar] [CrossRef]
- Maldonado, E.; Maillaud, C.; Barguil, Y.; Labadie, M. Rhabdomyolysis during envenomation by Physalia sp. envenomation in New Caldonia. Med. St. Trop. 2017, 27, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Rengel, A.C.; Davel, S.; O’Connor, S.; Medcraft, N. Rhabdomyolysis due to jellyfish envenomation in Western Australian waters. Med. J. Aust. 2022, 216, 397–399. [Google Scholar] [CrossRef] [PubMed]
- Auerbach, P.S.; Hays, J.T. Erythema nodosum following a jellyfish sting. J. Emerg. Med. 1987, 5, 487–491. [Google Scholar] [CrossRef]
- Al-Ebrahim, K.; Tahir, M.Z.; Rustom, M.; Shafei, H. Jellyfish-venom-induced deep venous thrombosis. A case report. Angiology 1995, 46, 449–451. [Google Scholar] [CrossRef] [PubMed]
- Tahmassebi, J.F.; O’Sullivan, E.A. A case report of an unusual mandibular swelling in a 4-year-old child possibly caused by a jellyfish sting. Int. J. Paediatr. Dent. 1998, 8, 51–54. [Google Scholar] [CrossRef]
- Tortell, A.; Baldacchino, R.V.; Grech, J.A. The Boy With the Jellyfish Tattoo and Facial Swelling. Wilderness Environ. Med. 2021, 32, 545–547. [Google Scholar] [CrossRef]
- Binnetoglu, F.K.; Kizildag, B.; Topaloglu, N.; Kasapcopur, O. Severe digital necrosis in a 4-year-old boy: Primary Raynaud’s or jellyfish sting. Case Rep. 2013, 2013, bcr2013201478. [Google Scholar] [CrossRef]
- Lam, S.C.; Hung, Y.W.; Chow, E.C.; Wong, C.W.; Tse, W.L.; Ho, P.C. Digital ischaemia: A rare but severe complication of jellyfish sting. Hong Kong Med. J. 2014, 20, 460–463. [Google Scholar] [CrossRef]
- Lo, A.H.; Chan, Y.C.; Law, Y.; Cheng, S.W. Successful treatment of jellyfish sting-induced severe digital ischemia with intravenous iloprost infusion. J. Vasc. Surg. Cases 2016, 2, 31–33. [Google Scholar] [CrossRef]
- Chiang, F.; Castillo, M. Seastrokes: A new threat for north Carolina swimmers? A case report. Neuroradiol. J. 2014, 27, 499–502. [Google Scholar] [CrossRef]
- Li, R.; Yu, H.; Xue, W.; Yue, Y.; Liu, S.; Xing, R.; Li, P. Jellyfish venomics and venom gland transcriptomics analysis of Stomolophus meleagris to reveal the toxins associated with sting. J. Proteom. 2014, 106, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Haddad, V., Jr.; Virga, R.; Bechara, A.; Silveira, F.L.; Morandini, A.C. An outbreak of Portuguese man-of-war (Physalia physalis—Linnaeus, 1758) envenoming in Southeastern Brazil. Rev. Soc. Bras. Med. Trop. 2013, 46, 641–644. [Google Scholar] [CrossRef] [PubMed]
- Kapamajian, M.A.; Ahmad, A.; Burnett, J.W.; Burnett, H.W.; Frenkel, S. Unilateral orbital inflammation in a child after a jellyfish sting to the lower extremities. Ophthalmic Plast Reconstr. Surg. 2009, 25, 239–241. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.K.; Bandyopadhyay, D.; Haldar, S. Lichen planus-like eruption resulting from a jellyfish sting: A case report. J. Med. Case Rep. 2009, 3, 7421. [Google Scholar] [CrossRef]
- Del Pozo, L.J.; Knöpfel, N.; Martín-Santiago, A.; Escudero-Góngora, M.M.; Saus, C.; Izquierdo-Herce, N.; Bauzà-Alonso, A. Dermoscopic Findings of Jellyfish Stings Caused by Pelagia noctiluca. Actas Dermosifiliogr. 2016, 107, 509–515. [Google Scholar] [CrossRef]
- Gunn, M.A. Localized fat atrophy after jellyfish sting. Br. Med. J. 1949, 2, 687. [Google Scholar] [CrossRef]
- Drury, J.K.; Noonan, J.D.; Pollock, J.G.; Reid, W.H. Jelly fish sting with serious hand complications. Injury 1980, 12, 66–68. [Google Scholar] [CrossRef]
- Tamanaha, R.H.; Izumi, A.K. Persistent cutaneous hypersensitivity reaction after a Hawaiian box jellyfish sting (Carybdea alata). J. Am. Acad. Dermatol. 1996, 35, 991–993. [Google Scholar] [CrossRef]
- Pérez-López, I.; Garrido-Colmenero, C.; Blasco-Morente, G.; Tercedor-Sánchez, J. Koebner phenomenon in a patient with lichen sclerosus following a jellyfish sting: An exceptional morphology. Actas Dermosifiliogr. 2015, 106, 238–239. [Google Scholar] [CrossRef]
- Thaikruea, L.; Siriariyaporn, P.; Wutthanarungsan, R.; Smithsuwan, P. Review of fatal and severe cases of box jellyfish envenomation in Thailand. Asia Pac. J. Public Health 2015, 27, NP1639–NP1651. [Google Scholar] [CrossRef]
- Anziani Vente, A.; Jaloux, C.; Morand, J.J.; Bel, S.L.; Bosdure, E.; Dubus, J.C.; Morand, A. Severe box jellyfish envenomation in 2 children. Travel Med. Infect. Dis. 2022, 45, 102217. [Google Scholar] [CrossRef] [PubMed]
- Tiemensma, M.; Currie, B.J.; Byard, R.W. Fatal jellyfish envenoming-Pediatric and geographic vulnerabilities. J. Forensic Sci. 2021, 66, 2006–2009. [Google Scholar] [CrossRef] [PubMed]
- Togias, A.G.; Burnett, J.W.; Kagey-Sobotka, A.; Lichtenstein, L.M. Anaphylaxis after contact with a jellyfish. J. Allergy Clin. Immunol. 1985, 75, 672–675. [Google Scholar] [CrossRef]
- Friedel, N.; Scolnik, D.; Adir, D.; Glatstein, M. Severe anaphylactic reaction to mediterranean jellyfish (Ropilhema nomadica) envenomation: Case report. Toxicol. Rep. 2016, 3, 427–429. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Armoni, M.; Ohali, M.; Hay, E. Severe dyspnea due to jellyfish envenomation. Pediatr. Emerg. Care 2003, 19, 84–86. [Google Scholar] [CrossRef] [PubMed]
- Fenner, P.J.; Williamson, J.A.; Burnett, J.W.; Colquhoun, D.M.; Godfrey, S.; Gunawardane, K.; Murtha, W. The “Irukandji syndrome” and acute pulmonary oedema. Med. J. Aust. 1988, 149, 150–156. [Google Scholar] [CrossRef]
- Fenner, P.J.; Hadok, J.C. Fatal envenomation by jellyfish causing Irukandji syndrome. Med. J. Aust 2002, 177, 362–363. [Google Scholar] [CrossRef]
- Pereira, P.; Barry, J.; Corkeron, M.; Keir, P.; Little, M.; Seymour, J. Intracerebral hemorrhage and death after envenoming by the jellyfish Carukia barnesi. Clin. Toxicol. 2010, 48, 390–392. [Google Scholar] [CrossRef]
- Bastos, D.M.; Haddad, V.J.; Nunes, J.L. Human envenomations caused by Portuguese man-of-war (Physalia physalis) in urban beaches of São Luis City, Maranhão State, Northeas.st Coast of Brazil. Rev. Soc. Bras. Med. Trop. 2017, 50, 130–134. [Google Scholar] [CrossRef]
- Moleiro, S.; Pereira, A.; Paiva Lopes, M.J. Marine dermatitis following exposure to a Portuguese-man-of-war. Acta Med. Port. 2013, 26, 66–69. [Google Scholar]
- Endean, R.; Monks, S.A.; Cameron, A.M. Toxins from the box-jellyfish Chironex fleckeri. Toxicon 1993, 31, 397–410. [Google Scholar] [CrossRef]
- Flecker, H. Fatal stings to North Queensland bathers. Med. J. Aust. 1952, 1, 35–38. [Google Scholar] [CrossRef] [PubMed]
- Lumley, J.; Williamson, J.A.; Fenner, P.J.; Burnett, J.W.; Colquhoun, D.M. Fatal envenomation by Chironex fleckeri, the north Australian box jellyfish: The continuing search for lethal mechanisms. Med. J. Aust. 1988, 148, 527–534. [Google Scholar] [CrossRef] [PubMed]
- Kong, E.L.; Nappe, T.M. Irukandji Syndrome. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2021. [Google Scholar]
- Nickson, C.P.; Waugh, E.B.; Jacups, S.P.; Currie, B.J. Irukandji syndrome case series from Australia’s Tropical Northern Territory. Ann. Emerg. 2009, 54, 395–403. [Google Scholar] [CrossRef]
- Little, M.; Pereira, P.; Carrette, T.; Seymour, J. Jellyfish responsible for Irukandji syndrome. J. Assoc. Phys. 2006, 99, 425–427. [Google Scholar] [CrossRef]
- Bouyer-Monot, D.; Pelczar, S.; Ferracci, S.; Boucaud-Maitre, D. Retrospective study of jellyfish envenomation in emergency wards in Guadeloupe between 2010 and 2016: When to diagnose Irukandji syndrome? Toxicon 2017, 137, 73–77. [Google Scholar] [CrossRef]
- Flecker, H. Irukandji sting to North Queensland bathers without production of weals but with severe general symptoms. J. Aust. 1952, 2, 89–91. [Google Scholar] [CrossRef]
- Yoshimoto, C.M.; Yanagihara, A.A. Cnidarian (coelenterate) envenomations in Hawai’i improve following heat application. Trans. R Soc. Trop. Med. Hyg. 2002, 96, 300–303. [Google Scholar] [CrossRef]
- Cheng, A.C.; Winkel, K.D.; Hawdon, G.M.; McDonald, M. Irukandji-like syndrome in Victoria. Aust. N. Z. J. Med. 1999, 29, 835. [Google Scholar] [CrossRef]
- Gershwin, L.A.; Richardson, A.J.; Winkel, K.D.; Fenner, P.J.; Lippmann, J.; Hore, R.; Avila-Soria, G.; Brewer, D.; Kloser, R.J.; Steven, A.; et al. Biology and ecology of Irukandji jellyfish (Cnidaria: Cubozoa). Adv. Mar. Biol. 2013, 66, 1–85. [Google Scholar] [CrossRef]
- Greenland, P.; Hutchinson, D.; Park, T. Irukandji Syndrome: What nurses need to know. Nurs. Health Sci. 2006, 8, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Fenner, P.J.; Harrison, S.L. Irukandji and Chironex fleckeri jellyfish envenomation in tropical Australia. Wilderness Environ. Med. 2000, 11, 233–240. [Google Scholar] [CrossRef]
- Fenner, P.J.; Williamson, J.; Callanan, V.I.; Audley, I. Further understanding of, and a new treatment for, “Irukandji” (Carukia barnesi) stings. Med. J. Aust. 1986, 145, 569–574. [Google Scholar] [CrossRef] [PubMed]
- Hadok, J.C. “Irukandji” syndrome: A risk for divers in tropical waters. Med. J. Aust. 1997, 167, 649–650. [Google Scholar] [CrossRef] [PubMed]
- Rathbone, J.; Franklin, R.; Gibbs, C.; Williams, D. Review article: Role of magnesium sulphate in the management of Irukandji syndrome: A systematic review. Emerg. Med. Australas. 2017, 29, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Sando, J.J.; Usher, K.; Buettner, P. ‘To swim or not to swim’: The impact of jellyfish stings causing Irukandji Syndrome in Tropical Queensland. J. Clin. Nurs. 2010, 19, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.C.; Audley, I. Cardiac failure following Irukandji envenomation. Med. J. Aust. 1990, 153, 164–166. [Google Scholar] [CrossRef]
- Huynh, T.T.; Seymour, J.; Pereira, P.; Mulcahy, R.; Cullen, P.; Carrette, T.; Little, M. Severity of Irukandji syndrome and nematocyst identification from skin scrapings. Med. J. Aust. 2003, 178, 38–41. [Google Scholar] [CrossRef]
- Little, M.; Mulcahy, R.F. A year’s experience of Irukandji envenomation in far north Queensland. Med. J. Aust 1998, 169, 638–641. [Google Scholar] [CrossRef]
- Segura Puertas, L.; Burnett, J.W.; de la Cotera, E.H. The medusa stage of the coronate scyphomedusa Linuche unguiculata (‘thimble jellyfish’) can cause seabather’s eruption. Dermatology 1999, 198, 171–172. [Google Scholar] [CrossRef]
- Segura-Puertas, L.; Ramos, M.E.; Aramburo, C.; De La Cotera, E.P.H.; Burnett, J.W. One Linuche mystery solved: All 3 stages of the coronate scyphomedusa Linuche unguiculata cause seabather’s eruption. J. Am. Acad. Dermatol. 2001, 44, 624–628. [Google Scholar] [CrossRef] [PubMed]
- Wong, D.E.; Meinking, T.L.; Rosen, L.B.; Taplin, D.; Hogan, D.J.; Burnett, J.W. Seabather’s eruption. Clinical, histologic, and immunologic features. J. Am. Acad. Dermatol. 1994, 30, 399–406. [Google Scholar] [CrossRef]
- Rossetto, A.L.; Dellatorre, G.; Silveira, F.L.; Haddad Júnior, V. Seabather’s eruption: A clinical and epidemiological study of 38 cases in Santa Catarina State, Brazil. Rev. Do Inst. De Med. Trop. De São Paulo 2009, 51, 169–175. [Google Scholar] [CrossRef] [PubMed][Green Version]
- O’Donnell, B.F.; Tan, C.Y. Persistent contact dermatitis from jellyfish sting. Contact Dermat. 1993, 28, 112–113. [Google Scholar] [CrossRef]
- Uri, S.; Marina, G.; Liubov, G. Severe delayed cutaneous reaction due to Mediterranean jellyfish (Rhopilema nomadica) envenomation. Contact Dermat. 2005, 52, 282–283. [Google Scholar] [CrossRef]
- Mao, C.; Hsu, C.C.; Chen, K.T. Ocular Jellyfish Stings: Report of 2 Cases and Literature Review. Wilderness Environ. Med. 2016, 27, 421–424. [Google Scholar] [CrossRef]
- Winkel, K.D.; Hawdon, G.M.; Ashby, K.; Ozanne-Smith, J. Eye injury after jellyfish sting in temperate Australia. Wilderness Environ. Med. 2002, 13, 203–205. [Google Scholar] [CrossRef]
- Mitchell, J.H. Eye injuries due to jellyfish (Cyanea annaskala). Med. J. Aust. 1962, 49, 303–305. [Google Scholar] [CrossRef]
- Chui, J.J.; Ooi, K.G.; Reeves, D.; Francis, I.C. Bluebottle envenomation-induced crystalline keratopathy. Cornea 2011, 30, 835–837. [Google Scholar] [CrossRef]
- Zheng, X.Y.; Cheng, D.J.; Lian, L.H.; Zhang, R.T.; Yu, X.Y. Severe fundus lesions induced by ocular jellyfish stings: A case report. World J. Clin. Cases 2020, 8, 4544–4549. [Google Scholar] [CrossRef]
- Li, R.; Yu, H.; Yue, Y.; Liu, S.; Xing, R.; Chen, X.; Li, P. Combined proteomics and transcriptomics identifies sting-related toxins of jellyfish Cyanea nozakii. J. Proteom. 2016, 148, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Abellaneda, C.; Navarra, R.; Martín-Urda, M.T.; Gómez, M. Jellyfish sting or tattoo? Actas Dermo-Sifili. 2012, 103, 243–244. [Google Scholar] [CrossRef] [PubMed]
- Pearn, J. The sea, stingers, and surgeons: The surgeon’s role in prevention, first aid, and management of marine envenomations. J. Pediatr. Surg. 1995, 30, 105–110. [Google Scholar] [CrossRef]
- Law, Y.H. Stopping the sting. Science 2018, 362, 631–635. [Google Scholar] [CrossRef] [PubMed]
- Thaikruea, L.; Siriariyaporn, P. The magnitude of severe box jellyfish cases on Koh Samui and Koh Pha-ngan in the Gulf of Thailand. BMC Res. Notes 2016, 9, 108. [Google Scholar] [CrossRef] [PubMed]
- Mebs, D. Jellyfish sting injuries. Hautarzt 2014, 65, 873–878. [Google Scholar] [CrossRef]
- Haddad, V., Jr.; Cardoso, J.L.; Silveira, F.L. Seabather’s eruption: Report of five cases in southeast region of Brazil. Rev. Inst. Med. Trop. Sao Paulo 2001, 43, 171–172. [Google Scholar] [CrossRef]
- Haddad Junior, V.; Silveira, F.L.; Migotto, A.E. Skin lesions in envenoming by cnidarians (Portuguese man-of-war and jellyfish): Etiology and severity of accidents on the Brazilian coast. Rev. Inst. Med. Trop. Sao Paulo 2010, 52, 47–50. [Google Scholar] [CrossRef]
- Sil, A.; Panigrahi, A.; Chakraborty, S. Seabather’s Eruption. Am. J. Med. Sci. 2020, 360, 81. [Google Scholar] [CrossRef]
- Ahmad, A. Jelly fish eye stings. J. Egypt. Ophthalmol. Soc. 2016, 109, 172–173. [Google Scholar] [CrossRef]
- Fu, J.; Koo, K.; Sang, A.X.; Shisler, D.C. Jellyfish envenomation in an ocean swimmer. Intern. Emerg. Med. 2014, 9, 103–104. [Google Scholar] [CrossRef] [PubMed]
- Bengtson, K.; Nichols, M.M.; Schnadig, V.; Ellis, M.D. Sudden death in a child following jellyfish envenomation by Chiropsalmus quadrumanus. Case report and autopsy findings. JAMA 1991, 266, 1404–1406. [Google Scholar] [CrossRef] [PubMed]
- Currie, B.J.; Wood, Y.K. Identification of Chironex fleckeri envenomation by nematocyst recovery from skin. Med. J. Aust. 1995, 162, 478–480. [Google Scholar] [CrossRef] [PubMed]
- Thaikruea, L.; Santidherakul, S. The public health impact of a new simple practical technique for collection and transfer of toxic jellyfish specimens and for nematocyst identification. J. Public Health Policy 2018, 39, 143–155. [Google Scholar] [CrossRef]
- Hartman, K.R.; Calton, G.J.; Burnett, J.W. Use of the radioallergosorbent test for the study of coelenterate toxin-specific immunoglobulin E. Int. Arch. Allergy Appl. Immunol. 1980, 61, 389–393. [Google Scholar] [CrossRef]
- Fenner, P.J.; Lippmann, J.; Gershwin, L.A. Fatal and nonfatal severe jellyfish stings in Thai waters. J. Travel Med. 2010, 17, 133–138. [Google Scholar] [CrossRef]
- Tønseth, K.A.; Andersen, T.S.; Pripp, A.H.; Karlsen, H.E. Prophylactic treatment of jellyfish stings—A randomised trial. Idsskrift Den Nor. Legeforen. 2012, 132, 1446–1449. [Google Scholar] [CrossRef]
- Kimball, A.B.; Arambula, K.Z.; Stauffer, A.R.; Levy, V.; Davis, V.W.; Liu, M.; Rehmus, W.E.; Lotan, A.; Auerbach, P.S. Efficacy of a jellyfish sting inhibitor in preventing jellyfish stings in normal volunteers. Wilderness Environ. Med. 2004, 15, 102–108. [Google Scholar] [CrossRef]
- Ballesteros, A.; Marambio, M.; Fuentes, V.; Narda, M.; Santín, A.; Gili, J.M. Differing Effects of Vinegar on Pelagia noctiluca (Cnidaria: Scyphozoa) and Carybdea marsupialis (Cnidaria: Cubozoa) Stings-Implications for First Aid Protocols. Toxins 2021, 13, 509. [Google Scholar] [CrossRef]
- Doyle, T.K.; Headlam, J.L.; Wilcox, C.L.; MacLoughlin, E.; Yanagihara, A.A. Evaluation of Cyanea capillata Sting Management Protocols Using Ex Vivo and In Vitro Envenomation Models. Toxins 2017, 9, 215. [Google Scholar] [CrossRef]
- Pyo, M.J.; Lee, H.; Bae, S.K.; Heo, Y.; Choudhary, I.; Yoon, W.D.; Kang, C.; Kim, E. Modulation of jellyfish nematocyst discharges and management of human skin stings in Nemopilema nomurai and Carybdea mora. Toxicon 2016, 109, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Loten, C.; Stokes, B.; Worsley, D.; Seymour, J.E.; Jiang, S.; Isbister, G.K. A randomised controlled trial of hot water (45 degrees C) immersion versus ice packs for pain relief in bluebottle stings. Med. J. Aust. 2006, 184, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Buckley, N.A.; Isbister, G.K. ACP Journal Club. Review: Application of heat or hot water reduces pain from jellyfish stings. Ann. Intern. Med. 2012, 157, Jc6-12. [Google Scholar] [CrossRef] [PubMed]
- Staggs, R.; Pay, J.L. Cnidaria Toxicity. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2022. [Google Scholar]
- Wilcox, C.L.; Headlam, J.L.; Doyle, T.K.; Yanagihara, A.A. Assessing the Efficacy of First-Aid Measures in Physalia sp. Envenomation, Using Solution- and Blood Agarose-Based Models. Toxins 2017, 9, 149. [Google Scholar] [CrossRef]
- Li, L.; McGee, R.G.; Isbister, G.; Webster, A.C. Interventions for the symptoms and signs resulting from jellyfish stings. Cochrane Database Syst. Rev. 2013, 10, CD009688. [Google Scholar] [CrossRef]
- Li, L.; McGee, R.G.; Webster, A.C. Pain from bluebottle jellyfish stings. J. Paediatr. Child. Health 2015, 51, 734–737. [Google Scholar] [CrossRef]
- Yanagihara, A.A.; Wilcox, C.; King, R.; Hurwitz, K.; Castelfranco, A.M. Experimental Assays to Assess the Efficacy of Vinegar and Other Topical First-Aid Approaches on Cubozoan (Alatina alata) Tentacle Firing and Venom Toxicity. Toxins 2016, 8, 19. [Google Scholar] [CrossRef]
- Yanagihara, A.A.; Wilcox, C.L. Cubozoan Sting-Site Seawater Rinse, Scraping, and Ice Can Increase Venom Load: Upending Current First Aid Recommendations. Toxins 2017, 9, 105. [Google Scholar] [CrossRef]
- Hartwick, R.; Callanan, V.; Williamson, J. Disarming the box-jellyfish: Nematocyst inhibition in Chironex fleckeri. Med. J. Aust. 1980, 1, 15–20. [Google Scholar] [CrossRef]
- Little, M.; Fitzpatrick, R.; Seymour, J. Successful use of heat as first aid for tropical Australian jellyfish stings. Toxicon 2016, 122, 142–144. [Google Scholar] [CrossRef]
- Isbister, G.K.; Palmer, D.J.; Weir, R.L.; Currie, B.J. Hot water immersion v icepacks for treating the pain of Chironex fleckeri stings: A randomised controlled trial. Med. J. Aust. 2017, 206, 258–261. [Google Scholar] [CrossRef] [PubMed]
- Lakkis, N.A.; Maalouf, G.J.; Mahmassani, D.M. Jellyfish Stings: A Practical Approach. Wilderness Environ. Med. 2015, 26, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Macrokanis, C.J.; Hall, N.L.; Mein, J.K. Irukandji syndrome in northern Western Australia: An emerging health problem. Med. J. Aust. 2004, 181, 699–702. [Google Scholar] [CrossRef] [PubMed]
- Berling, I.; Isbister, G. Marine envenomations. Aust. Fam. Phys. 2015, 44, 28–32. [Google Scholar]
- Pereira, P.; Corkeron, M.; Little, M.; Farlow, D. Irukandji Taskforce guidelines for the emergency management of Irukandji syndrome. Diving Hyperb. Med. South Pac. Underw. Med. Soc. 2007, 37, 48. [Google Scholar]
- McCullagh, N.; Pereira, P.; Cullen, P.; Mulcahy, R.; Bonin, R.; Little, M.; Gray, S.; Seymour, J. Randomised trial of magnesium in the treatment of Irukandji syndrome. Emerg. Med. Australas 2012, 24, 560–565. [Google Scholar] [CrossRef]
- Guevara, B.E.K.; Dayrit, J.F.; Haddad, V., Jr. Seabather’s eruption caused by the thimble jellyfish (Linuche aquila) in the Philippines. Clin. Exp. Dermatol. 2017, 42, 808–810. [Google Scholar] [CrossRef]
- Sonmez, B.; Beden, U.; Yeter, V.; Erkan, D. Jellyfish sting injury to the cornea. Ophthalmic Surg. Lasers Imaging Retin. 2008, 39, 415–417. [Google Scholar] [CrossRef]
- Yu Yao, H.; Cho, T.H.; Lu, C.H.; Lin, F.C.; Horng, C.T. Hypertonic saline in the treatment of corneal jellyfish stings. J. Travel Med. 2016, 23, tav030. [Google Scholar] [CrossRef]
- Olivieri, L.; Mazzarelli, D.; Cappella, A.; De Angelis, D.; Piscitelli, V.; Cattaneo, C. The importance of “secondary methods” in the identification of the victims of the 3rd of October 2013 shipwreck. La Rev. De Med. Leg. 2017, 8, 190–191. [Google Scholar] [CrossRef]
- Stein, M.R.; Marraccini, J.V.; Rothschild, N.E.; Burnett, J.W. Fatal Portuguese man-o’-war (Physalia physalis) envenomation. Ann. Emerg. Med. 1989, 18, 312–315. [Google Scholar] [CrossRef]
- Purcell, J.E.; Atienza, D.; Fuentes, V.; Olariaga, A.; Tilves, U.; Colahan, C.; Gili, J.-M. Temperature effects on asexual reproduction rates of scyphozoan species from the northwest Mediterranean Sea. Hydrobiologia 2012, 690, 169–180. [Google Scholar] [CrossRef]
- Widmer, C.L.; Fox, C.J.; Brierley, A.S. Effects of temperature and salinity on four species of northeastern Atlantic scyphistomae (Cnidaria: Scyphozoa). Mar. Ecol. Prog. Ser. 2016, 559, 73–88. [Google Scholar] [CrossRef]
- Schiariti, A.; Melica, V.; Kogovšek, T.; Malej, A. Density-dependent effects control the reproductive strategy and population growth of Aurelia aurita s.l. scyphistomae. Mar. Biol. 2015, 162, 1665–1672. [Google Scholar] [CrossRef]
- Sokołowski, A.; Brulińska, D.; Olenycz, M.; Wołowicz, M. Does temperature and salinity limit asexual reproduction of Aurelia aurita polyps (Cnidaria: Scyphozoa) in the Gulf of Gdańsk (southern Baltic Sea)? An experimental study. Hydrobiologia 2016, 773, 49–62. [Google Scholar] [CrossRef]
- Treible, L.; Condon, R. Temperature-driven asexual reproduction and strobilation in three scyphozoan jellyfish polyps. J. Exp. Mar. Biol. Ecol. 2019, 520, 151204. [Google Scholar] [CrossRef]
- Goldstein, J.; Steiner, U.K. Ecological drivers of jellyfish blooms—The complex life history of a ‘well-known’ medusa (Aurelia aurita). J. Anim. Ecol. 2020, 89, 910–920. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Yu, H.; Li, A.; Yu, C.; Li, P. Preparation and Neutralization Efficacy of Novel Jellyfish Antivenoms against Cyanea nozakii Toxins. Toxins 2021, 13, 165. [Google Scholar] [CrossRef]
- Li, R.; Yu, H.; Li, A.; Yu, C.; Li, P. Refinement and Neutralization Evaluation of the F(ab’)2 Type of Antivenom against the Deadly Jellyfish Nemopilema nomurai Toxins. Int. J. Mol. Sci. 2021, 22, 12672. [Google Scholar] [CrossRef]
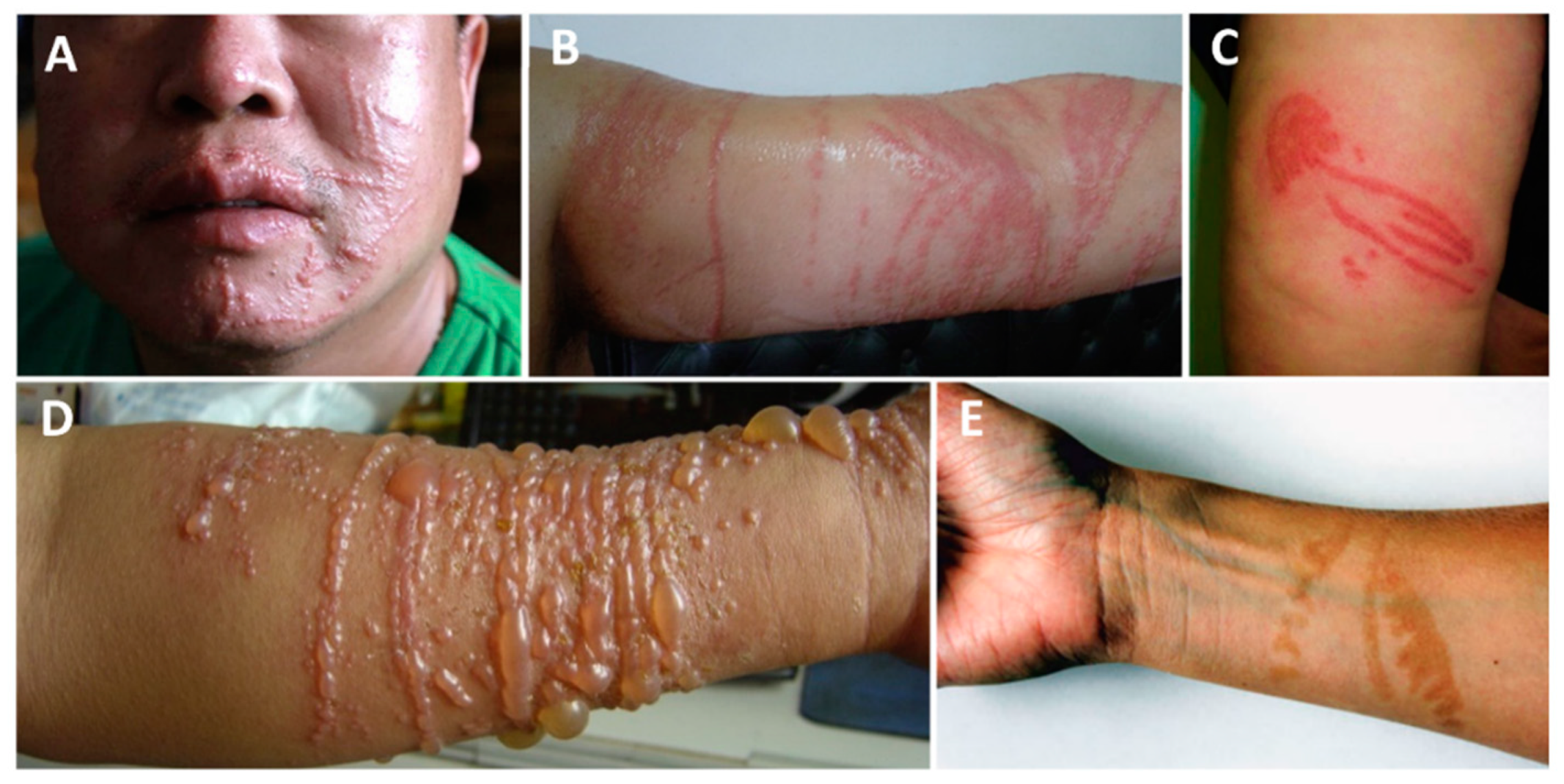
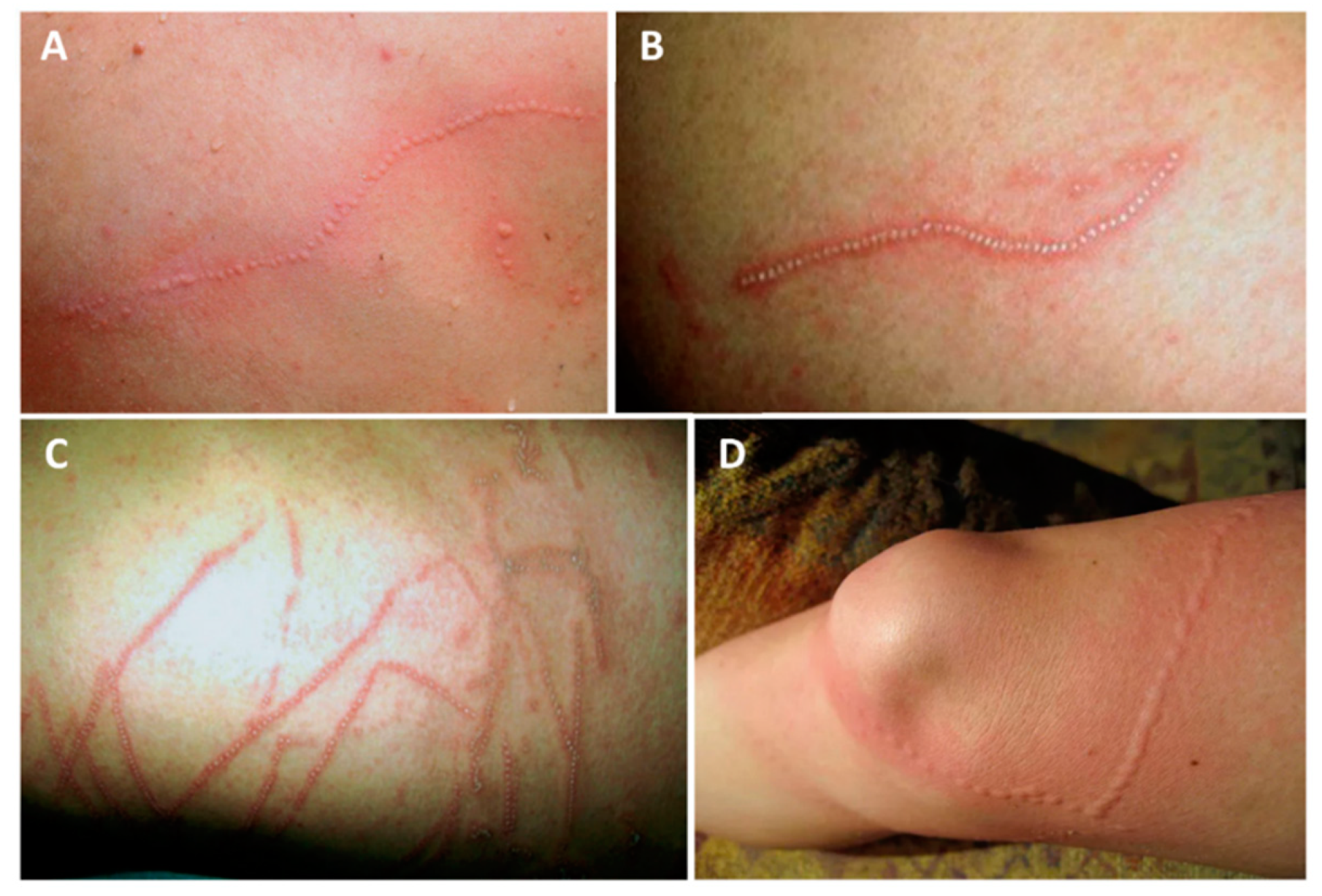
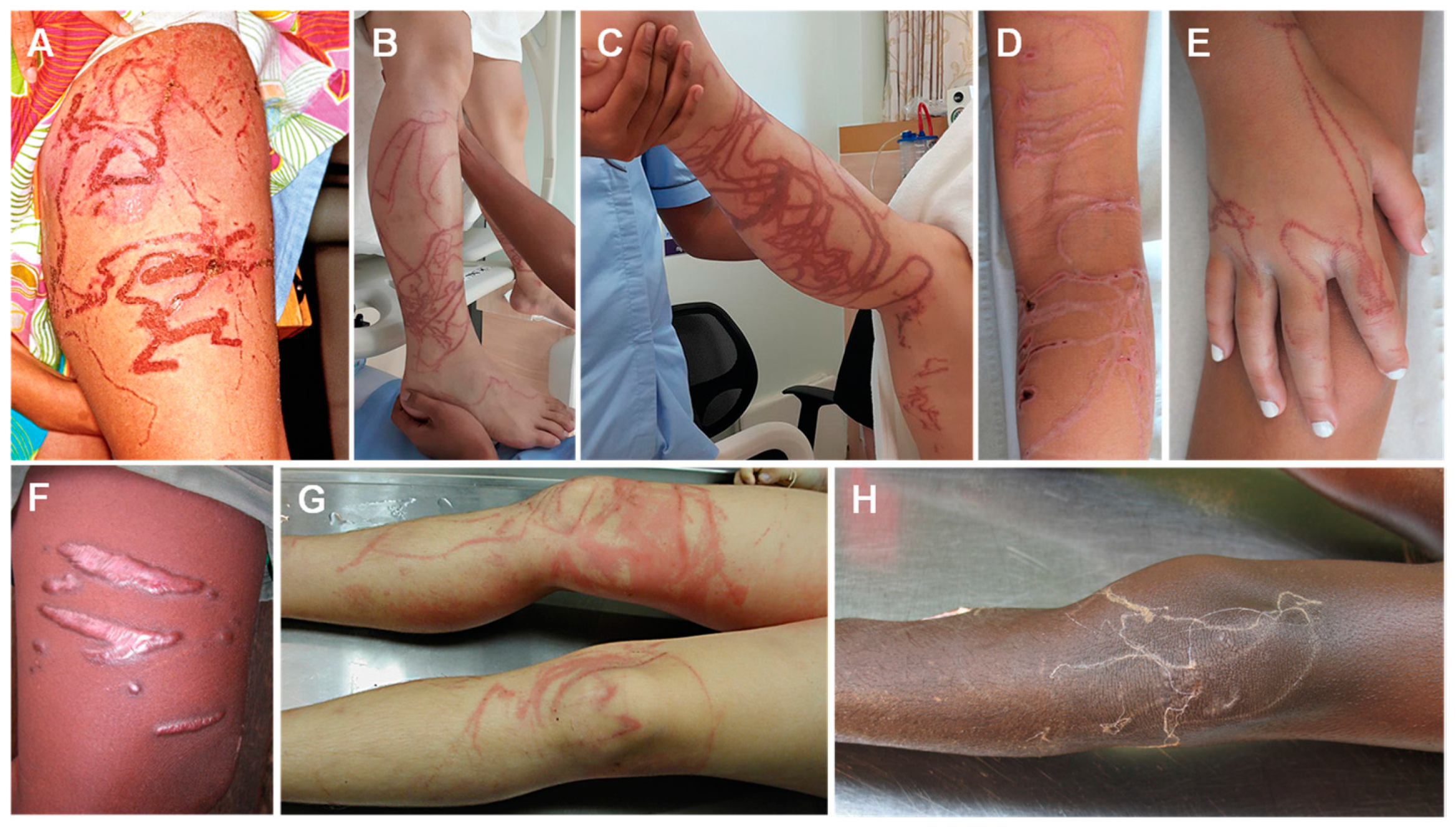
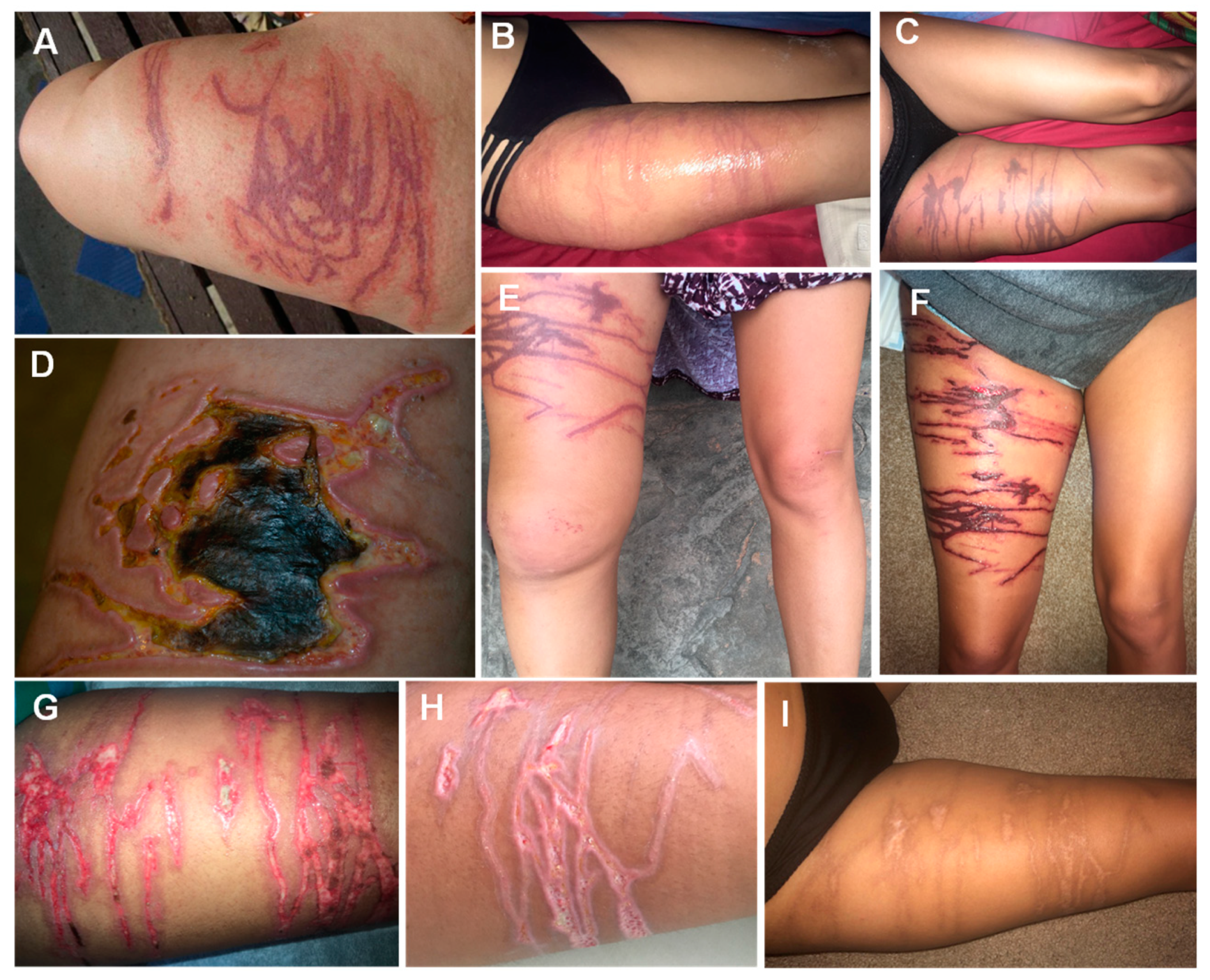
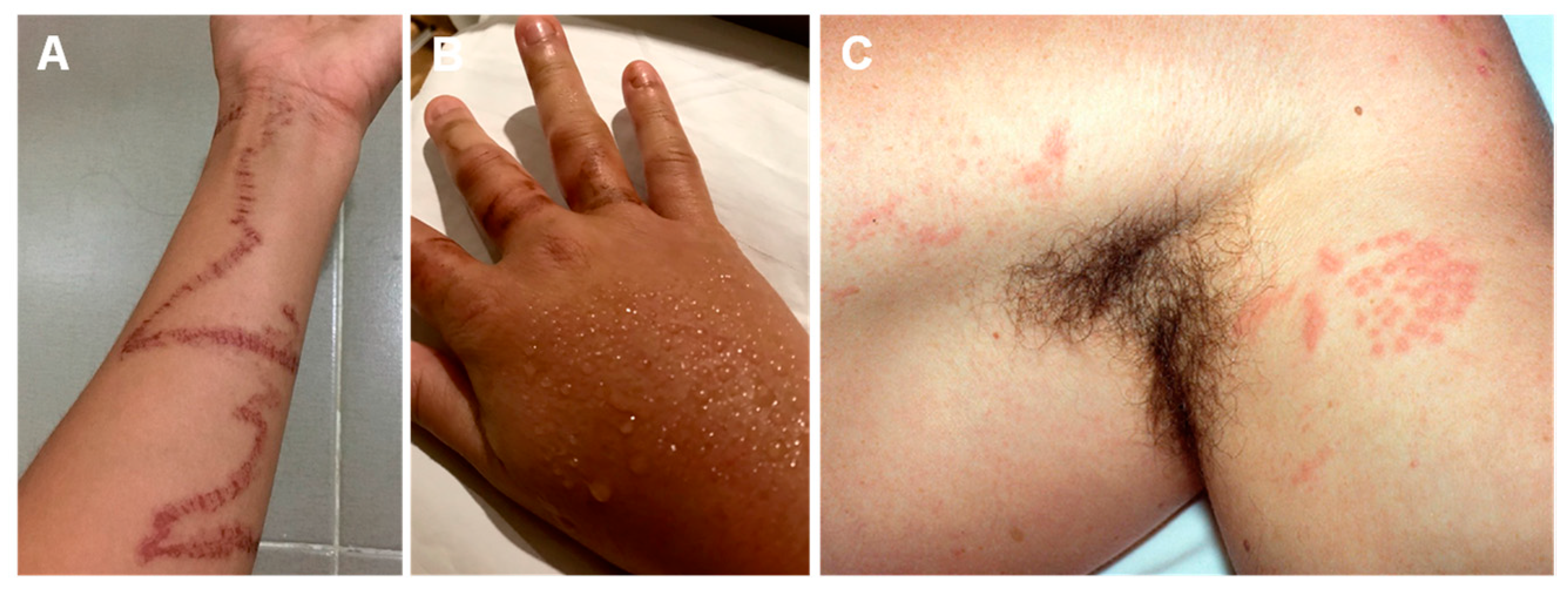


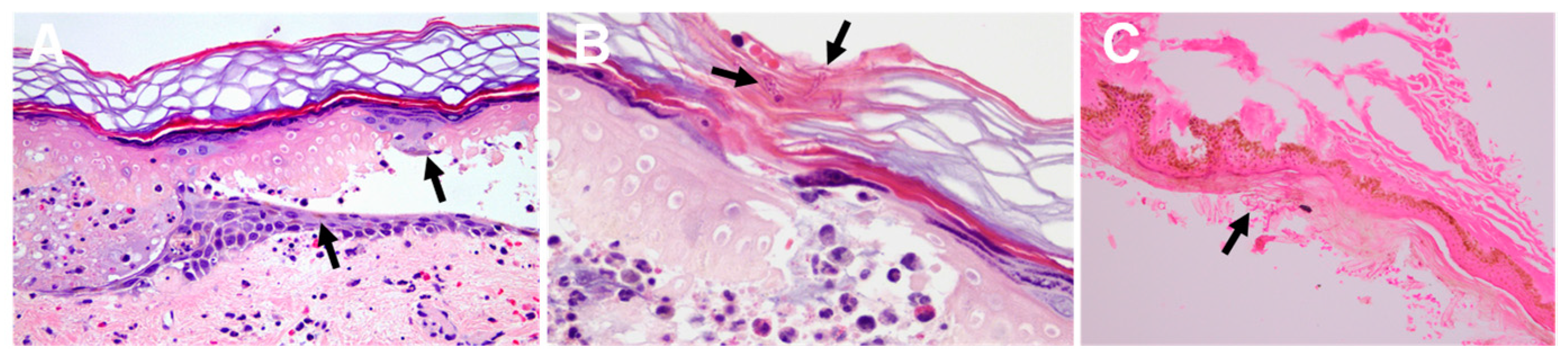
| Type of Reaction | Reaction | Class Implied | References |
|---|---|---|---|
| Local reactions | Vesiculourticarial, painful dermatitis | Scyphozoa, Hydrozoa, Cubozoa | [14,76] |
| Scarring | Scyphozoa, Cubozoa | [77,78] | |
| Infection | Scyphozoa, Cubozoa | [79,80] | |
| Pruritus | Scyphozoa | [81] | |
| Delayed dermatitis | Hydrozoa, Scyphozoa, Cubozoa | [82,83,84,85,86,87,88,89] | |
| Recurrent dermatitis | Scyphozoa, Hydrozoa | [90,91,92,93] | |
| Parasympathetic actions | Parasympathetic dysautonomia | Cubozoa | [94] |
| Ileus paraliticus | unknown | [95] | |
| Urinary incontinence | Cubozoa | [94,96] | |
| Biliary dyskinesia | Scyphozoa | [96] | |
| Vascular spasm | Hydrozoa | [97,98,99] | |
| Sympathetic actions | Mydriasis | Scyphozoa | [100] |
| Blurred vision | Scyphozoa | [101] | |
| Stress cardiomyopathy | Cubozoa, Scyphozoa | [30,72] | |
| Irukandji syndrome | Cubozoa | [102,103,104,105,106,107,108] | |
| Neuromuscular actions | Mononeuritis multiplex | unknown | [109,110] |
| Peripheral neuropathy | Scyphozoa, Cubozoa | [99,111,112,113,114] | |
| Guillain-Barré | Scyphozoa | [115,116] | |
| Complex Regional Pain Syndrome | unknown | [117] | |
| Dysphonia | Hydrozoa, Scyphozoa | [13,118,119] | |
| Other systemic reactions | Acute renal failure | Hydrozoa | [120] |
| Arrythmia | Hydrozoa | [121] | |
| Rhabdomyolysis | Hydrozoa | [122,123] | |
| Erythema nodosum | Hydrozoa | [124] | |
| Deep venous thrombosis | Scyphozoa | [125] | |
| Facial swelling | unknown | [126,127] | |
| Digital necrosis | unknown | [128,129,130] | |
| Spinal cord infarct | unknown | [131] | |
| Cellulitis | Scyphozoa | [79] | |
| Myalgia | Scyphozoa, Cubozoa | [132] | |
| Abdominal cramping | Scyphozoa, Cubozoa | [13,119] | |
| Cough | Scyphozoa | [13,119] | |
| Nausea and vomiting | Hydrozoa | [133] | |
| Cold sweating | Hydrozoa | [133] | |
| Delayed eye swelling | unknown | [134] | |
| Long term reactions | Keloids | Cubozoa | [71,73] |
| Lichen planus | unknown | [135] | |
| Hyperpigmentation | Scyphozoa, Hydrozoa, Cubozoa | [78,86,136] | |
| Fat atrophy | unknown | [137] | |
| Delayed necrosis | Cubozoa | [82] | |
| Gangrene | Cubozoa | [138] | |
| Persistent hypersensitivity | Scyphozoa | [139] | |
| Koebner Phenomenon | unknown | [140] | |
| Persistent neuropathy | unknown | [111] | |
| Granulomatous infiltration | Cubozoa | [87] | |
| Nodular papular eruption | Cubozoa | [84] | |
| Potentially fatal reactions | Immediate cardiac arrest | Cubozoa | [141,142,143] |
| Acute myocardial infarction | unknown | [29,31] | |
| Rapid respiratory arrest | Cubozoa, Scyphozoa | [75,141] | |
| Anaphylaxis | Scyphozoa, Hydrozoa, Cubozoa | [121,144,145] | |
| Angioedema | Scyphozoa | [121,146] | |
| Pulmonary edema | Scyphozoa, Cubozoa | [75,146,147] | |
| Shock | Scyphozoa, Hydrozoa | [132] | |
| Intracerebral hemorrhage after severe hypertension | Cubozoa | [148,149] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cunha, S.A.; Dinis-Oliveira, R.J. Raising Awareness on the Clinical and Forensic Aspects of Jellyfish Stings: A Worldwide Increasing Threat. Int. J. Environ. Res. Public Health 2022, 19, 8430. https://doi.org/10.3390/ijerph19148430
Cunha SA, Dinis-Oliveira RJ. Raising Awareness on the Clinical and Forensic Aspects of Jellyfish Stings: A Worldwide Increasing Threat. International Journal of Environmental Research and Public Health. 2022; 19(14):8430. https://doi.org/10.3390/ijerph19148430
Chicago/Turabian StyleCunha, Sara Almeida, and Ricardo Jorge Dinis-Oliveira. 2022. "Raising Awareness on the Clinical and Forensic Aspects of Jellyfish Stings: A Worldwide Increasing Threat" International Journal of Environmental Research and Public Health 19, no. 14: 8430. https://doi.org/10.3390/ijerph19148430
APA StyleCunha, S. A., & Dinis-Oliveira, R. J. (2022). Raising Awareness on the Clinical and Forensic Aspects of Jellyfish Stings: A Worldwide Increasing Threat. International Journal of Environmental Research and Public Health, 19(14), 8430. https://doi.org/10.3390/ijerph19148430







