Abstract
SARS-CoV-2 has spread throughout the world, including remote areas such as those located at high altitudes. There is a debate about the role of hypobaric hypoxia on viral transmission and COVID-19 incidence. A descriptive cross-sectional analysis of SARS-CoV-2 infection and viral load among patients living at low (230 m) and high altitude (3800 m) in Ecuador was completed. Within these two communities, the total number of infected people at the time of the study was 108 cases (40.3%). The COVID-19 incidence proportion at low altitude was 64% while at high altitude was 30.3%. The mean viral load from those patients who tested positive was 3,499,184 copies/mL (SD = 23,931,479 copies/mL). At low altitude (Limoncocha), the average viral load was 140,223.8 copies/mL (SD = 990,840.9 copies/mL), while for the high altitude group (Oyacachi), the mean viral load was 6,394,789 copies/mL (SD = 32,493,469 copies/mL). We found no statistically significant differences when both results were compared (p = 0.056). We found no significant differences across people living at low or high altitude; however, men and younger populations had higher viral load than women older populations, respectively.
1. Introduction
Several months after the COVID-19 pandemic declaration on 11 March 2020, some interrogations still remain unsolved concerning viral transmission and the burden of COVID-19 at high-altitude locations [1,2,3]. The variability and susceptibility of COVID-19 infection throughout the world have been attributed to several factors such as chronic diseases, age, socioeconomic level, and access to medical care within different health care systems, among others [4]; however, the role of different environmental factors such as humidity, temperature, or altitude on the transmission of SARS-CoV-2 are not well-understood [4,5]. Since the onset of the pandemic, some researchers have attempted to elucidate the effects of living at a high altitude on the spread of the SARS-CoV-2 virus or on the severity and the COVID-19-related incidence and mortality rate [6,7,8].
A handful of investigations have suggested that living at high altitude is associated with lower COVID-19 attack and mortality rates; nevertheless, the explanations behind those findings have been poorly understood and still need to be elucidated [4,9,10].
At the planetary level, the pandemic has spread with greater virulence in places with higher population density and worse socioeconomic conditions [11,12,13]. This distribution does not apparently respond to environmental conditions but rather to social determinants. However, it is evident that living in remote places with low population density and harsher weathers could justify, in part, a proposed lower viral transmission and reduced SARS-CoV-2 infection rates at higher altitudes [1,6,7].
The hypotheses surrounding the role of high altitude on SARS-CoV-2 transmission and the impact of the pandemic in these populations can be summarized into three groups. The physiological and biological role of altitude-adapted organisms in relation to virus transmission (i.e., the role of ACE-2 receptors at high altitude), the direct or indirect effects of the environment on virulence (i.e., ozone, UV exposure, or cold on viral transmission), or the epidemiological relationship between sociodemographic factors and COVID-19 incidence (i.e., population density, overcrowding activities, or migration) [1,2,14].
In this sense, the least-studied factor is the role of high-altitude exposure on viral stability or viral transmissibility. A suitable indicator would be the evaluation of viral load using the reverse transcription polymerase chain reaction (RT-PCR) test [15,16].
2. Materials and Methods
2.1. Study Design
An observational, cross-sectional analysis of two populations located at two different altitudes was completed in Ecuador. The objective of this research project was to diagnose COVID-19 cases in remote areas of the country. Most of these areas had poor medical infrastructure and no access to molecular Rt-qPCR testing.
2.2. Settings
The study was carried out in two Kiwchas indigenous communities from Ecuador: Limoncocha, located at 230 m meters above sea level (low altitude), and Oyacachi, located at 3800 m meters above sea level (high altitude) (Figure 1).
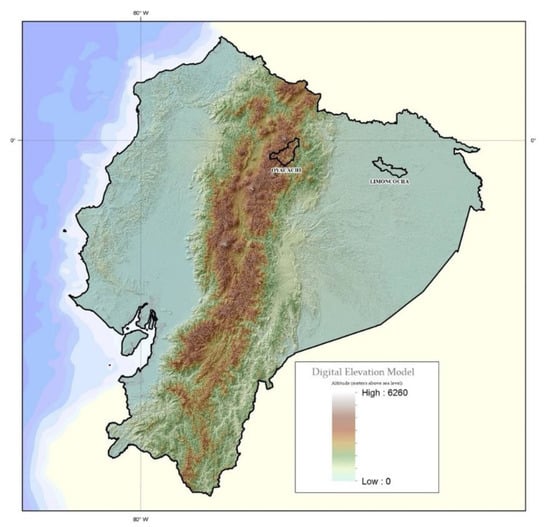
Figure 1.
Topographic map of Ecuador highlighting Limoncocha (230 m) and Oyacachi (3800 m).
2.3. Participants
We included 268 members from both communities to compare exposure at high altitude with those living at low altitude. These communities share the same ancestry background as previously reported, serving as a good genotyped-controlled population [17,18,19].
2.4. Data Source and Variables
Using an informed consent as well as an epidemiological data recollection sheet form, demographic variables including sex, age, jurisdiction of residence (Limoncocha and Oyacachi communities), as well as symptomology status were obtained. For nasopharyngeal swabs samples, the Center for Disease Control (CDC) 2019-Novel Coronavirus (2019-nCoV) RT-qPCR Diagnostic Panel was used to identify the presence of SARS-CoV-2.
2.5. Study and Sample Size
In this SARS-CoV-2-detection program, we used a non-probabilistic convenience sample technique that yielded a total of 77 volunteers from Limoncocha (low altitude) and 191 from Oyacachi (high altitude).
2.6. Statistical Methods
Measurements of frequency, central tendency, data dispersion, and absolute differences between groups were calculated for all categorical and continuous variables. An independent t-test analysis was used to assess differences between groups. We used a 95% confidence level (α = 0.05).
The hypotheses used can be explained as:
H 0.
“Viral load in both jurisdictions are equal”.
H 1.
“Viral load in both jurisdictions are different”.
2.7. RNA Extraction and RT-qPCR for SARS-CoV-2 Detection
Nasopharyngeal swabs were collected on 0.5 mL TE pH 8 buffer for SARS-CoV-2 detection by RT-qPCR following an adapted version of the CDC protocol by using PureLink Viral RNA/DNA Mini Kit (Invitrogen, Waltham, MA, USA) as an alternate RNA extraction method and CFX96 BioRad instrument [15,16,20,21,22,23,24,25]. Briefly, the CDC-designed RT-qPCR FDA EUA 2019-nCoV CDC kit (IDT, Newark, NJ, USA) is based on N1 and N2 probes to detect SARS-CoV-2 and RNase P as an RNA extraction quality control [21,23]. Additionally, negative controls (TE pH 8 buffer) were included as control for carryover contamination, one for each set of RNA extractions, to guarantee that only true positives were reported. For viral loads calculation, the 2019-nCoV N-positive control (IDT, Newark, NJ, USA) was used, providing at 200,000 genome equivalents/μL. This positive control is a plasmid including N1 and N2 viral gene targets sequences, and it is a SARS-CoV-2-positive control recommended by CDC guidelines [16,20,22].
2.8. Nasopharyngeal Sample Collection
Nasopharyngeal swab samples from each of the patients were performed by trained UDLA personnel. Two physicians were in charge of performing the swabs in both communities. The extraction of the genetic material was performed by well-trained laboratorists from the molecular medicine laboratory of the university [26,27].
2.9. Bias
The analysis and interpretation of the data was done by four of the investigators separately to look for discrepancies. Any new findings were reviewed by the entire team, and a unanimous decision was made in the event of differences in the results.
2.10. Ethics Statement
Informed written consent was obtained from every patient to use their anonymized results from the study in fulfillment of all legal requirements.
3. Results
3.1. Demographic Results
From the total sample, 56.3% (n = 151) were men, and 43.7% (n = 117) were women. The mean age from the entire cohort was 37.72 years (SD = 14.14). In Limoncocha, the mean age was 35.4 years (SD = 14.32) and in Oyacachi was 38.5 years (SD = 14.20).
3.2. Positive Testing Rates
The total number of positive cases was 108 (40.3%), and n = 50 were from Limoncocha and n = 58 from Oyacachi (Figure 2). The attack rate at low altitude was 64% while at high altitude was 30.3%.
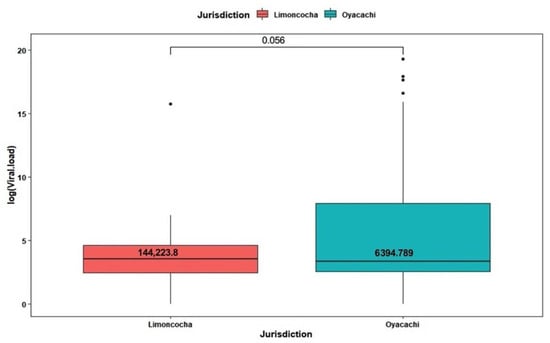
Figure 2.
Box plot of viral load composition across jurisdiction (communities) of SARS-CoV-2 test positivity among 108 tested people, 50 from Limoncocha and 58 from Oyacachi.
3.3. Viral Load Analysis by Altitude
The mean viral load of the 40.3% (n = 108) patients who tested positive was 3,499,184 copies/mL (SD = 23,931,479 copies/mL). For Limoncocha, the average viral load was 144,223.8 copies/mL (SD = 990,840.9 copies/mL) and for Oyacachi, 6,394,789 copies/mL (SD = 32,493,469 copies/mL). We found no statistically significant differences between communities (p = 0.056) (Figure 2).
3.4. Viral Load Analysis by Age and Sex
Among female patients, no difference in viral load were found (viral load mean = 6,165,595 copies/mL (p-value of 0.13)). When comparing low- versus high-altitude viral load values, men from Oyacachi had a higher viral load (mean 2,016,226 copies/mL) than those men living in Limoncocha (mean 241,700 copies/mL), with this difference being statistically significant at a confidence level of α = 0.05 (p-value of 0.018) (Figure 3).
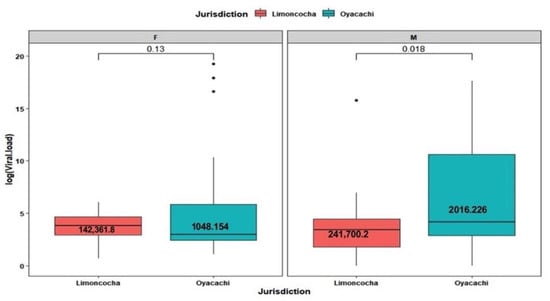
Figure 3.
Box plot of viral load composition across jurisdiction (communities) and gender of SARS-CoV-2 test positivity among 108 tested people, 21 women and 29 men from Limoncocha and 30 women and 28 men from Oyacachi.
3.5. Viral Load Analysis by Age
In general terms, there were no statistically significant differences when comparing age groups; however, we found significant differences between those aged 25 to 29 years living at high altitude, whose samples had a higher viral load than those from the low-altitude group (Table 1).

Table 1.
Viral loads composition across age groups among 108 positive tests for SARS-CoV-2 infection.
When we grouped the individuals into age groups according to the WHO classification, we found a statistically significant difference between the high-altitude young adult men who had higher viral loads than their low-altitude counterparts (p-value of 0.017) (Figure 4).
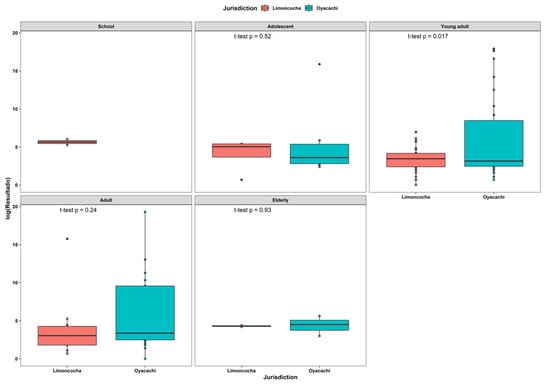
Figure 4.
Box plot of viral load composition across age groups among 108 positive tests for SARS-CoV-2 infection. Age groups classification is according to the United Nations guidelines as follows: school (<12 years), adolescent (13–20 years), young adult (21 to 39 years), adult (40 to 64 years), and elderly (>65 years).
4. Discussion
Our exploratory results found no significant difference between people living at high altitude and those living at low altitude in terms of SARS-CoV-2 viral load. While acknowledging the importance of measuring viral load to understand the hypothesized difference across altitudes, our study involves several uncontrolled variables that undermine our ability to conclude if viral loads are different at higher altitudes. Our report is the first one available in terms of exploring this issue, and according to our results, it seems that altitude might not play an important role affecting viral load among patients living at different altitudes.
In some investigations, the role that hypobaric hypoxia could have on the viability of the SARS-CoV-2 virus has been analyzed [1,25,28]. In very few reports, it is mentioned that ozone could affect the viability of the virus, humidity could affect transmissibility, and ultraviolet rays (UV lights) could eliminate the virus faster than in other locations where there is less UV light exposure than at high altitude [29,30] (Table 2).

Table 2.
Comparative table of main climatological and meteorological differences between Oyacachi and Limoncocha [17,31,32,33,34].
Another factor often linked to the transmission of the virus may be cold weather. Several studies have suggested that the susceptibility of the virus to temperature may be affected by climate, but it is more logical to think that climate influences the behavior of people, the way people socialize, and the places people visit (open or closed spaces), as these factors are more feasible when exploring the association between cold and SARS-CoV-2 transmission [35,36,37].
Although socio-demographic factors are the most relevant in terms of viral transmission, incidence, and COVID-19-related mortality, is plausible that altitude may have an important role in improving the survival of some seriously ill patients who live at high altitudes [38]. Even with the presence of comorbidities, survival among severely ill COVID-19 patients at high altitude seems to be improved when compared to low-altitude patients, probably linked to their adaptation status that might improves oxygenation [38,39].
Most of the reports that have described reduced mortality are observational, population-based studies rather than individual-based [3,6,7,10,40], in other words, on populations but not on individuals. This confers a limitation since important data, such as viral load, have never been measured in high altitudes. A recent report by Arias-Reyes et al., 2021 [9], investigated whether the transmission rate of SARS-CoV-2 differs between low and high altitudes [9]. They found that after using a mathematical SEIR model, the probability of viral transmission is lower at high altitude, concluding that their findings strongly support the hypothesis of decreased SARS-CoV-2 virulence in highlands compared to lowlands [9].
Although the available results suggest lower COVID-19-related mortality at high altitudes, lower viral load cannot be attributed as one causal factor. Viral load differences among low- and high-altitude dwellers is unlikely, mainly since the idiopathic response of each organism towards viral replication depends on immunological and biological factors more than in environmental or socio-demographic differences [41,42,43].
The question of whether the SARS-CoV-2 viral load among high-altitude dwellers is different from that of low-altitude dwellers remains unresolved yet. Although our exploratory results indicates that men at high altitude have higher viral load than men at lower altitudes, the differences in viral load are more a function of age and sex rather than altitude.
5. Limitations
The study analyzed viral load from COVID-19 patients at low and high altitude in Ecuador. However, there are multiple factors affecting viral load, including age, timing of the samples, disease severity, and other factors that we were not able to control. Days of sampling relative to infection or symptom onset might have a significant impact on viral load. However, this information was not available. The representativeness and recruitment when using a convenience sampling technique cannot be guaranteed; however, we believe that the results provide us with important data that were not available elsewhere.
6. Conclusions
This is the first exploratory study that attempts to identify differences in SARS-CoV-2 viral loads between low- and high-altitude populations. We found that there are no significant differences in terms of viral load between the two populations.
We believe that air quality and its composition, such as the ozone concentration, particulate matter, UV lights, humidity (%), and hypoxia, have no direct relationship with the viral load found in SARS-CoV-2-positive patients; rather, we believe that the differences depend on other factors, such as the stage at which the sample collection was taken and what stage of the viral load curve each patient was in.
Finally, we conclude that men have more viral load than women and that young adults have more viral load than the elderly, and this could be due to a greater and more prolonged exposure to aerosols than those older people or women who surely take more care of themselves and may be more vigilant in the use of biosecurity measures.
Author Contributions
E.O.-P. was fully responsible for the conceptualization and the analysis of the study; K.S.-R. and R.F.-N. were responsible for the data analysis, construction of figures and tables, as well as the statistics analysis; J.E.V. with the elaboration of the first draft of the manuscript; E.O.-P. wrote the final version of the manuscript; G.V. and M.A.G.-B. critically reviewed the entire manuscript and produced several comments prior to the submission; T.L., A.R.H.-T., A.P.V.-J., and I.A.R.-O. collected or analyzed the samples as part of the diagnosis program directed by Universidad de las Americas in coordination with the Minister of Public Health of Ecuador. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Universidad de Las Américas and by Fundación CRISFE (Fondo “Sumar juntos”).
Institutional Review Board Statement
In relation to local legislation, in Ecuador any study that includes secondary source data that has been subsequently anonymized and is impossible to identify any individual is exempt from a full IRB review. In that sense, and in line with international legislation such as the Helsinki Declaration and good research practice (GCP), this study did not require full approval by the UDLA IRB (CEISH).
Informed Consent Statement
Written consent was obtained for all the individuals enrolling SARS-CoV-2 testing at UDLA. As non personal information was used and data was anonymized before analysis, this study falls under the category of “exempted” by the Bioethics Commission from UDLA.
Data Availability Statement
All data is anonymized and available in the following open access repository https://github.com/covid19ec/Altitude (accessed on 30 April 2022).
Acknowledgments
We would like to thank the community health service center from Limoncocha and Oyacachi for their help while organizing, visiting, and collecting nasopharyngeal swabs. We also thank the Molecular Biology Laboratory at Universidad de Las Americas for providing free SARS-CoV-2 testing to all study participants, and the UDLA COVID-19 team: Byron Freire-Paspuel, Bárbara Coronel, Heberson Galvis, Tatiana Jaramillo, María Belén Rodríguez Paredes, Angel S. Rodríguez Pazmiño, Diana Morales-Jadán, Daniela Santander Gordón, Gabriel Alfredo Iturralde, Julio Alejandro Terán, Karen Marcela Vásquez, Jonathan Darío Rondal, Genoveva Granda, Ana Cecilia Santamaría, Cynthia Lorena Pino, Oscar Lenin Espinosa, Angie Buitrón, David Sánchez Grisales, Karina Beatriz Jimenez, Vanessa Bastidas, Dayana Marcela Aguilar, Inás María Paredes, Christian David Bilvao, Henry Herrera, Pablo Marcelo Espinosa, Edison Andres Galárraga, Marlon Steven Zambrano-Mila, Ana Maria Tito, Nelson David Zapata.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Millet, G.P.; Debevec, T.; Brocherie, F.; Burtscher, M.; Burtscher, J. Altitude and COVID-19: Friend or Foe? A Narrative Review. Physiol. Rep. 2021, 8, e14615. [Google Scholar] [CrossRef]
- Srivastava, A. COVID-19 and Air Pollution and Meteorology-an Intricate Relationship: A Review. Chemosphere 2021, 263, 128297. [Google Scholar] [CrossRef]
- Arias-Reyes, C.; Zubieta-DeUrioste, N.; Poma-Machicao, L.; Aliaga-Raduan, F.; Carvajal-Rodriguez, F.; Dutschmann, M.; Schneider-Gasser, E.M.; Zubieta-Calleja, G.; Soliz, J. Does the Pathogenesis of SARS-CoV-2 Virus Decrease at High-Altitude? Respir. Physiol. Neurobiol. 2020, 277, 103443. [Google Scholar] [CrossRef]
- Pun, M.; Turner, R.; Strapazzon, G.; Brugger, H.; Swenson, E.R. Lower Incidence of COVID-19 at High Altitude: Facts and Confounders. High Alt. Med. Biol. 2020, 21, 217–222. [Google Scholar] [CrossRef]
- Castilla, J.; Fresán, U.; Trobajo-Sanmartín, C.; Guevara, M. Altitude and SARS-CoV-2 Infection in the First Pandemic Wave in Spain. Int. J. Environ. Res. Public Health 2021, 18, 2578. [Google Scholar] [CrossRef]
- Ortiz-Prado, E.; Simbaña-Rivera, K.; Barreno, L.G.; Diaz, A.M.; Barreto, A.; Moyano, C.; Arcos, V.; Vásconez-González, E.; Paz, C.; Simbaña-Guaycha, F.; et al. Epidemiological, Socio-Demographic and Clinical Features of the Early Phase of the COVID-19 Epidemic in Ecuador. PLoS Negl. Trop. Dis. 2021, 15, e0008958. [Google Scholar] [CrossRef]
- Cano-Pérez, E.; Torres-Pacheco, J.; Fragozo-Ramos, M.C.; García-Díaz, G.; Montalvo-Varela, E.; Pozo-Palacios, J.C. Negative Correlation between Altitude and COVID-19 Pandemic in Colombia: A Preliminary Report. Am. J. Trop. Med. Hyg. 2020, 103, 2347–2349. [Google Scholar] [CrossRef]
- Canales-Gutiérrez, A.; Canales-Manchuria, G.P.-M.; Canales-Manchuria, F. Adaptation to Hypobaric Hypoxia of Residents at High Altitude, to Counteract COVID-19 Disease. Enferm. Clin. Engl. Ed. 2021, 31, 130–131. [Google Scholar] [CrossRef]
- Arias-Reyes, C.; Carvajal-Rodriguez, F.; Poma-Machicao, L.; Aliaga-Raduán, F.; Marques, D.A.; Zubieta-DeUrioste, N.; Accinelli, R.A.; Schneider-Gasser, E.M.; Zubieta-Calleja, G.; Dutschmann, M.; et al. Decreased Incidence, Virus Transmission Capacity, and Severity of COVID-19 at Altitude on the American Continent. PLoS ONE 2021, 16, e0237294. [Google Scholar] [CrossRef]
- Segovia-Juarez, J.; Castagnetto, J.M.; Gonzales, G.F. High Altitude Reduces Infection Rate of COVID-19 but Not Case-Fatality Rate. Respir. Physiol. Neurobiol. 2020, 281, 103494. [Google Scholar] [CrossRef]
- Bhadra, A.; Mukherjee, A.; Sarkar, K. Impact of Population Density on COVID-19 Infected and Mortality Rate in India. Model. Earth Syst. Environ. 2020, 7, 623–629. [Google Scholar] [CrossRef]
- Calderón-Larrañaga, A.; Vetrano, D.L.; Rizzuto, D.; Bellander, T.; Fratiglioni, L.; Dekhtyar, S. High Excess Mortality in Areas with Young and Socially Vulnerable Populations during the COVID-19 Outbreak in Stockholm Region, Sweden. BMJ Glob. Health 2020, 5, e003595. [Google Scholar] [CrossRef] [PubMed]
- Cooper, L.A.; Williams, D.R. Excess Deaths from COVID-19, Community Bereavement, and Restorative Justice for Communities of Color. JAMA 2020, 324, 1491–1492. [Google Scholar] [CrossRef] [PubMed]
- Choquenaira-Quispe, C.; Saldaña-Bobadilla, V.; Ramirez, J.K. Factors Involved in Low Susceptibility to COVID-19: An Adaptation of High Altitude Inhabitants. Med. Hypotheses 2020, 143, 110068. [Google Scholar] [CrossRef]
- Pujadas, E.; Chaudhry, F.; McBride, R.; Richter, F.; Zhao, S.; Wajnberg, A.; Nadkarni, G.; Glicksberg, B.S.; Houldsworth, J.; Cordon-Cardo, C. SARS-CoV-2 Viral Load Predicts COVID-19 Mortality. Lancet Respir. Med. 2020, 8, e70. [Google Scholar] [CrossRef]
- Freire-Paspuel, B.; Vega-Mariño, P.; Velez, A.; Castillo, P.; Gomez-Santos, E.E.; Cruz, M.; Garcia-Bereguiain, M.A. Cotton-Tipped Plastic Swabs for SARS-CoV-2 RT-QPCR Diagnosis to Prevent Supply Shortages. Front. Cell. Infect. Microbiol. 2020, 10, 356. [Google Scholar] [CrossRef]
- Ortiz-Prado, E.; Portilla, D.; Mosquera, J.; Simbaña-Rivera, K.L.; Duta, D.; Ochoa, I.; Burgos, G.; Izquierdo-Condoy, J.S.; Vasconez, E.; Calvopiña, M. Hematological Parameters, Lipid Profile and Cardiovascular Risk Analysis among Genotype Controlled Indigenous Kichwa Men and Women Living at Low and High Altitude. Front. Physiol. 2021, 1769, 749006. [Google Scholar] [CrossRef]
- Ortiz-Prado, E.; Simbaña-Rivera, K.; Duta, D.; Ochoa, I.; Izquierdo-Condoy, J.S.; Vasconez, E.; Carrasco, K.; Calvopiña, M.; Viscor, G.; Paz, C. Optimism and Health Self-Perception-Related Differences in Indigenous Kiwchas of Ecuador at Low and High Altitude: A Cross-Sectional Analysis. High Alt. Med. Biol. 2022, 23, 1. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Prado, E.; Encalada, S.; Mosquera, J.; Simbaña-Rivera, K.; Gomez-Barreno, L.; Duta, D.; Ochoa, I.; Izquierdo-Condoy, J.S.; Vasconez, E.; Burgos, G. A Comparative Analysis of Lung Function and Spirometry Parameters in Genotype-Controlled Natives Living at Low and High Altitude. BMC Pulm. Med. 2022, 22, 100. [Google Scholar] [CrossRef]
- Freire-Paspuel, B.; Vega-Mariño, P.; Velez, A.; Cruz, M.; Garcia-Bereguiain, M.A. Sample Pooling of RNA Extracts to Speed up SARS-CoV-2 Diagnosis Using CDC FDA EUA RT-QPCR Kit. Virus Res. 2020, 290, 198173. [Google Scholar] [CrossRef] [PubMed]
- CDC Labs. Available online: https://www.cdc.gov/coronavirus/2019-ncov/lab/guidelines-clinical-specimens.html (accessed on 13 November 2021).
- Freire-Paspuel, B.; Garcia-Bereguiain, M.A. Analytical Sensitivity and Clinical Performance of a Triplex RT-QPCR Assay Using CDC N1, N2, and RP Targets for SARS-CoV-2 Diagnosis. Int. J. Infect. Dis. 2021, 102, 14–16. [Google Scholar] [CrossRef] [PubMed]
- Mancini, F.; Barbanti, F.; Scaturro, M.; Fontana, S.; Di Martino, A.; Marsili, G.; Puzelli, S.; Calzoletti, L.; Facchini, M.; Di Mario, G.; et al. Multiplex Real-Time Reverse-Transcription Polymerase Chain Reaction Assays for Diagnostic Testing of Severe Acute Respiratory Syndrome Coronavirus 2 and Seasonal Influenza Viruses: A Challenge of the Phase 3 Pandemic Setting. J. Infect. Dis. 2021, 223, 765–774. [Google Scholar] [CrossRef]
- Semple, J.L.; Moore, G.W.K. High Levels of Ambient Ozone (O3) May Impact COVID-19 in High Altitude Mountain Environments. Respir. Physiol. Neurobiol. 2020, 280, 103487. [Google Scholar] [CrossRef]
- Haque, S.E.; Rahman, M. Association between Temperature, Humidity, and COVID-19 Outbreaks in Bangladesh. Environ. Sci. Policy 2020, 114, 253–255. [Google Scholar] [CrossRef]
- Ortiz-Prado, E.; Rivera-Olivero, I.A.; Freire-Paspuel, B.; Lowe, R.; Lozada, T.; Henriquez-Trujillo, A.R.; Garcia-Bereguiain, M.A. Testing for SARS-CoV-2 at the Core of Voluntary Collective Isolation: Lessons from the Indigenous Populations Living in the Amazon Region in Ecuador. Int. J. Infect. Dis. 2021, 105, 234–235. [Google Scholar] [CrossRef]
- Santander-Gordon, D.; Iturralde, G.A.; Freire-Paspuel, B.; Zambrano-Mila, M.S.; Morales-Jadan, D.; Vallejo-Janeta, P.A.; Coronel, B.; Galvis, H.; Jaramillo-Vivanco, T.; Bilvao, C.D. Crucial Contribution of the Universities to SARS-CoV-2 Surveillance in Ecuador: Lessons for Developing Countries. One Health 2021, 13, 100267. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.; Garg, I.; Bansal, A.; Kumar, B. SARS-CoV-2 Infection: Physiological and Environmental Gift Factors at High Altitude. Virus Dis. 2020, 31, 450. [Google Scholar] [CrossRef] [PubMed]
- Nicastro, F.; Sironi, G.; Antonello, E.; Bianco, A.; Biasin, M.; Brucato, J.R.; Ermolli, I.; Pareschi, G.; Salvati, M.; Tozzi, P.; et al. Solar UV-B/A Radiation Is Highly Effective in Inactivating SARS-CoV-2. Sci. Rep. 2021, 11, 14805. [Google Scholar] [CrossRef]
- Seyer, A.; Sanlidag, T. Solar Ultraviolet Radiation Sensitivity of SARS-CoV-2. Lancet Microbe 2020, 1, e8–e9. [Google Scholar] [CrossRef]
- AccuWeather. Air Quality in Limoncocha and Oyacachi. Available online: https://www.accuweather.com/es/ec/limoncocha/1239184/air-quality-index/1239184 (accessed on 30 April 2022).
- Ortiz-Prado, E.; Dunn, J.F.; Vasconez, J.; Castillo, D.; Viscor, G. Partial Pressure of Oxygen in the Human Body: A General Review. Am. J. Blood Res. 2019, 9, 1. [Google Scholar]
- GAD Oyacachi. Actualización Del Plan de Desarrollo y Ordenamiento Terrritorial de Oyacachi 2014–2019. 2019. Available online: http://app.sni.gob.ec/sni-link/sni/PORTAL_SNI/data_sigad_plus/sigadplusdocumentofinal/1768098760001_PDyOT%20DIAGNOSTICO%20OYACACHI%201_30-10-2015_23-37-32.pdf (accessed on 30 April 2022).
- GAG Limoncocha. Actualización Del Plan de Desarrollo y Ordenamiento Territorial de Limoncocha 2014–2019. 2019. Available online: http://app.sni.gob.ec/sni-link/sni/PORTAL_SNI/data_sigad_plus/sigadplusdocumentofinal/1768086160001_ACTUALIZACION%20PDOT%20LIMONCOCHA%202015%20-%202019_29-10-2015_15-41-36.pdf (accessed on 30 April 2022).
- Sobral, M.F.F.; Duarte, G.B.; da Penha Sobral, A.I.G.; Marinho, M.L.M.; de Souza Melo, A. Association between Climate Variables and Global Transmission oF SARS-CoV-2. Sci. Total Environ. 2020, 729, 138997. [Google Scholar] [CrossRef]
- Adedokun, K.A.; Olarinmoye, A.O.; Mustapha, J.O.; Kamorudeen, R.T. A Close Look at the Biology of SARS-CoV-2, and the Potential Influence of Weather Conditions and Seasons on COVID-19 Case Spread. Infect. Dis. Poverty 2020, 9, 77. [Google Scholar] [CrossRef]
- Scafetta, N. Distribution of the SARS-CoV-2 Pandemic and Its Monthly Forecast Based on Seasonal Climate Patterns. Int. J. Environ. Res. Public Health 2020, 17, 3493. [Google Scholar] [CrossRef]
- Jaramillo, P.R.M.; Simbaña-Rivera, K.; Silva, J.V.V.; Gómez-Barreno, L.; Campoverde, A.B.V.; Cevallos, J.F.N.; Guanoquiza, W.E.A.; Guevara, S.L.C.; Castro, L.G.I.; Puerta, N.A.M.; et al. High-Altitude Is Associated with Better Short-Term Survival in Critically Ill COVID-19 Patients Admitted to the ICU. PLoS ONE 2022, 17, e0262423. [Google Scholar] [CrossRef]
- Collaco, J.M.; Aoyama, B.C.; Rice, J.L.; McGrath-Morrow, S.A. Influences of Environmental Exposures on Preterm Lung Disease. Expert Rev. Respir. Med. 2021, 15, 1271–1279. [Google Scholar] [CrossRef]
- Woolcott, O.O.; Bergman, R.N. Mortality Attributed to COVID-19 in High-Altitude Populations. High Alt. Med. Biol. 2020, 21, 409–416. [Google Scholar] [CrossRef]
- Fajnzylber, J.; Regan, J.; Coxen, K.; Corry, H.; Wong, C.; Rosenthal, A.; Worrall, D.; Giguel, F.; Piechocka-Trocha, A.; Atyeo, C.; et al. SARS-CoV-2 Viral Load Is Associated with Increased Disease Severity and Mortality. Nat. Commun. 2020, 11, 5493. [Google Scholar] [CrossRef]
- Wu, Y.; Cheng, X.; Jiang, G.; Tang, H.; Ming, S.; Tang, L.; Lu, J.; Guo, C.; Shan, H.; Huang, X. Altered Oral and Gut Microbiota and Its Association with SARS-CoV-2 Viral Load in COVID-19 Patients during Hospitalization. npj Biofilm. Microbiomes 2021, 7, 61. [Google Scholar] [CrossRef]
- Mahallawi, W.H.; Alsamiri, A.D.; Dabbour, A.F.; Alsaeedi, H.; Al-Zalabani, A.H. Association of Viral Load in SARS-CoV-2 Patients with Age and Gender. Front. Med. 2021, 8, 39. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).