A Community-Led Approach as a Guide to Overcome Challenges for Therapy Research in Congenital Disorders of Glycosylation
Abstract
:1. Introduction
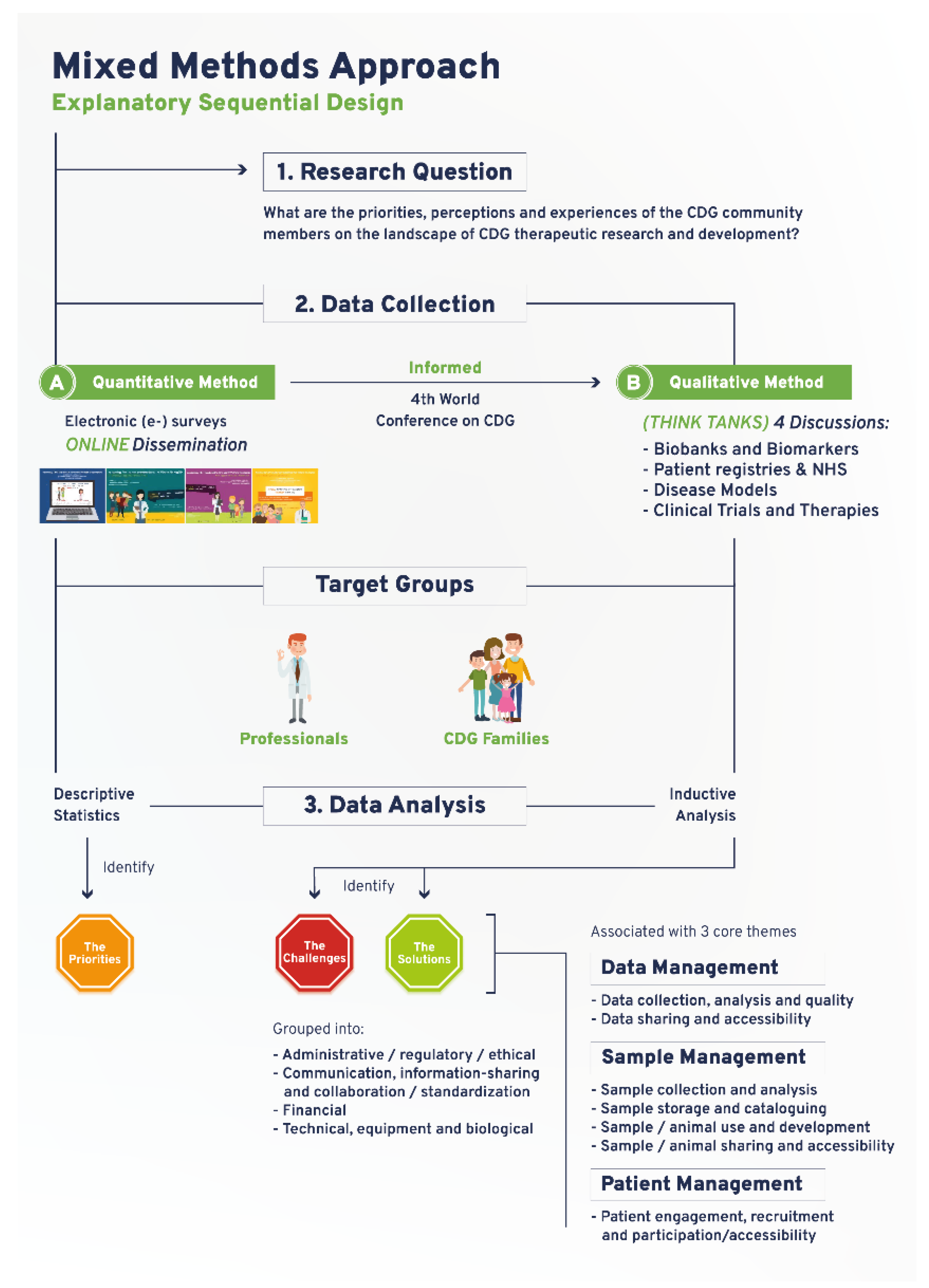
2. Methods
2.1. Quantitative Study
2.2. Qualitative Study
3. Results
3.1. A Community-Led Mixed Methods Approach Captures the CDG Community Research Priorities, Perspectives and Experiences
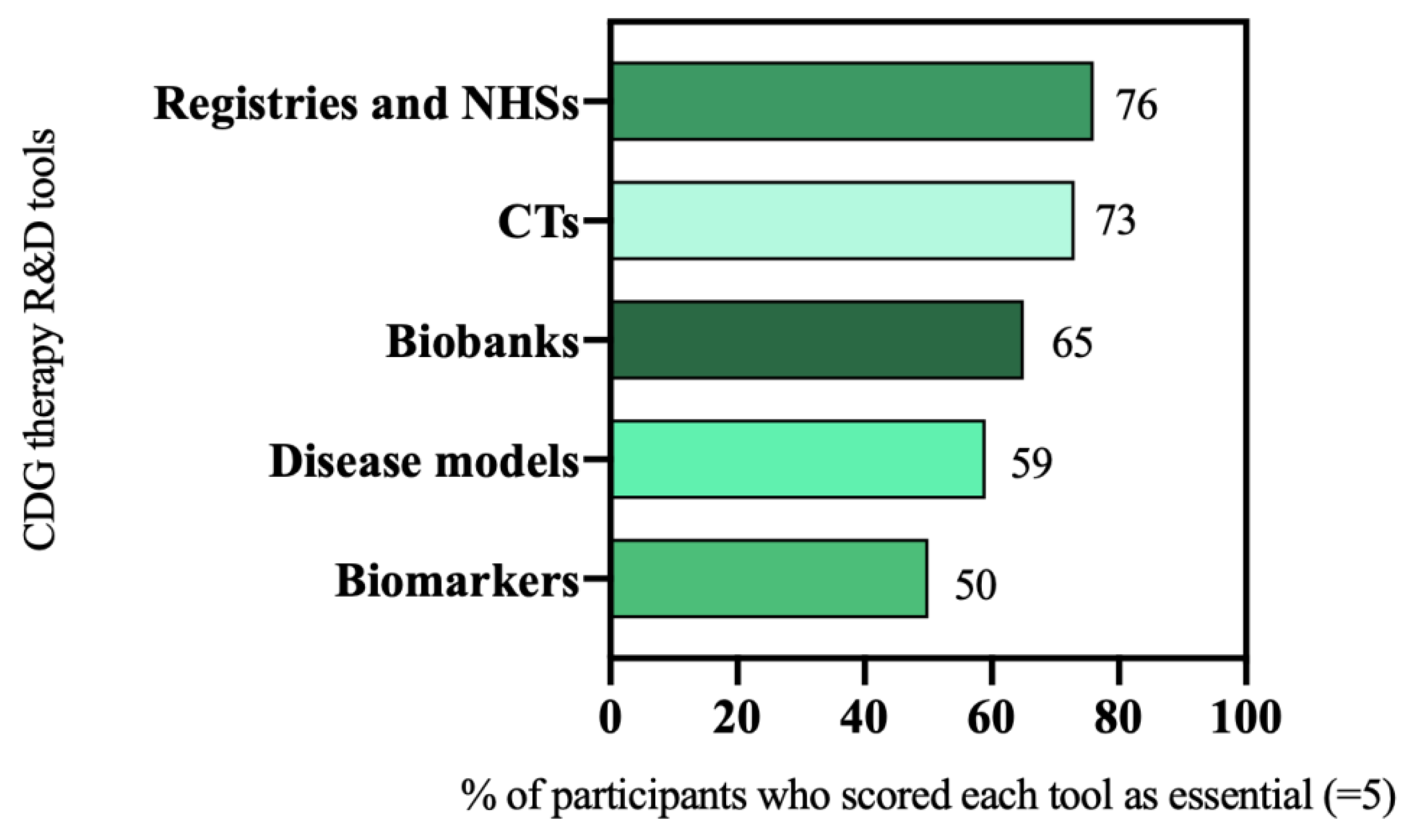
3.2. Six Therapy Research Tools Are Priorities for the CDG Community
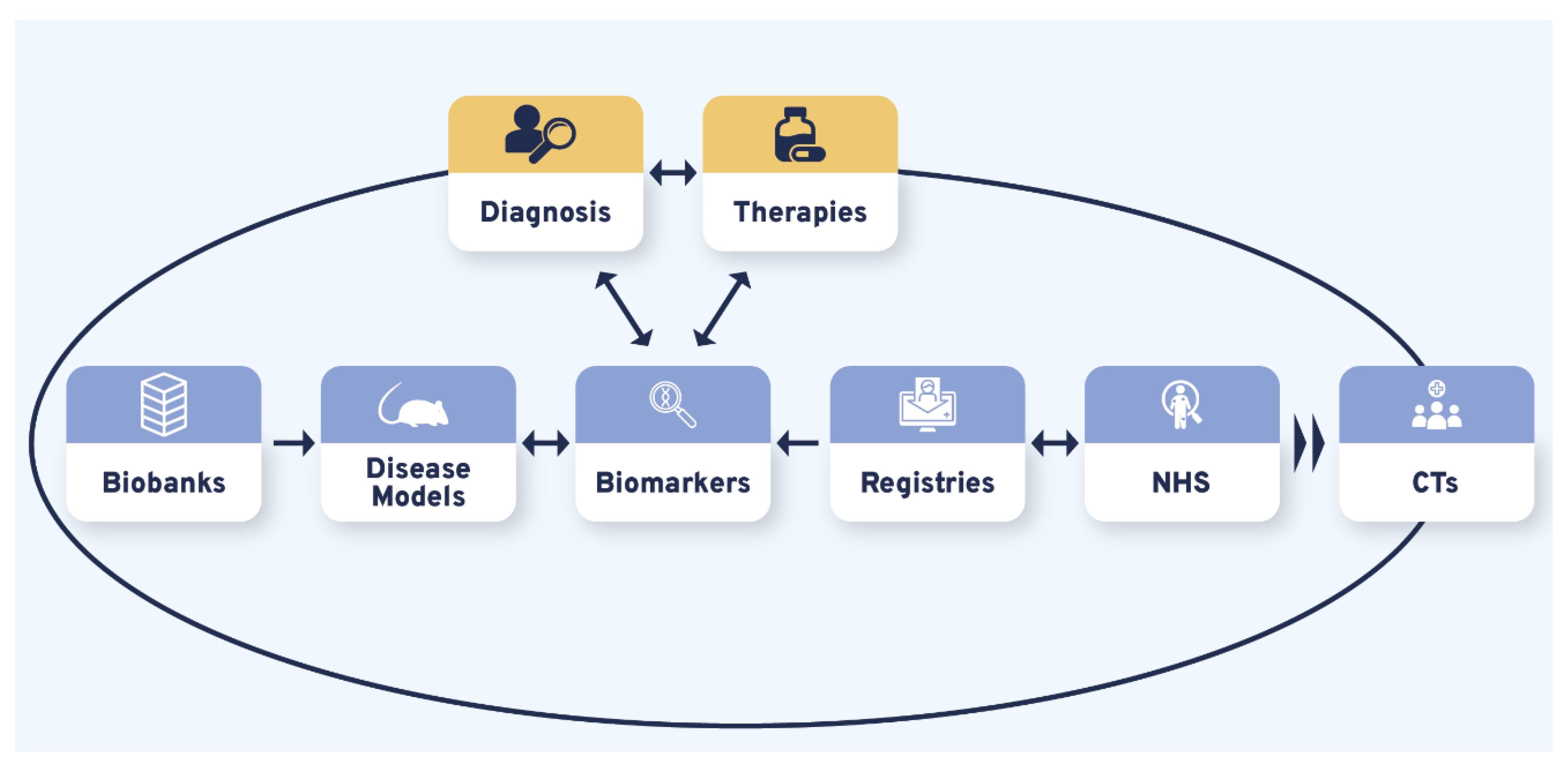
3.3. Data, Sample, and Patient Management Are the Core Elements of CDG Therapy R&D
3.3.1. CDG Biobanking (in) Success Is Defined by Sample and Data Sharing and Accessibility
Challenges of CDG Biobanks
“I feel that one of the main challenges we have is really how scattered the biobanks are (…) there’s actually lots of researcher specific biobanks. (…) in my institution we engage our patients in research and offer skin biopsies for them, and those skin biopsies are generally kept in our institution and so having those accessible to research around the world is a challenge.”(multiple roles)
Solutions for CDG Biobanks
“We don’t have many problems with parents when we say [that] we have to send blood to someone or [that] we have to send it to another country. They almost always say yes. There’s no problem to collect a little bit more of blood or even skin.”(HCP)
3.3.2. CDG Patient Registries Require Efficient Data Management
Challenges of CDG Patient Registries
Solutions for CDG Patient Registries
“(…) some sort of common language between patients and professionals”(Family member)
3.3.3. CDG Biomarker Discovery and Implementation Rely on Robust Sample and Data Collection
Challenges of CDG Biomarkers
“(…) we have been talking about good biomarkers, right? We don’t have them.”(researcher)
Solutions for CDG Biomarkers
3.3.4. CDG Disease Models Require Further Development and Use
Challenges of CDG Disease Models
“(…) when you’re trying to recapitulate the disease, the animals don’t survive. And when the animals do survive you don’t recapitulate the disease. So, that’s just a fundamental problem that we have”(industry representative)
Solutions for CDG Disease Models
“(…) the N-linked, the O-linked, the GPI [glycosylphosphatidylinositol] anchors, perhaps you can learn from one model of one disease, that can apply that to other models within that same pathway”(multiple roles)
3.3.5. Patient and Data Management Are Key Elements for Successful NHS
Challenges of CDG Natural History Studies
Solutions for CDG Natural History Studies
3.3.6. Patients and Data Realities Challenge CDG CTs
Challenges of CDG Clinical Trials
“(…) all of my patients that don’t have PMM2-CDG think PMM2-CDG is getting way too much attention (…) All CTs are for PMM2-CDG, but that doesn’t actually serve us well, because if we want to say that we’re a CDG group, then we need to be inclusive”(HCP)
Solutions for CDG Clinical Trials
“(…) grouping small groups of patients together (…) with a similar pathogenesis to PMM2”(industry representative)
3.4. Diagnosis and Therapies Are the Bidirectional Triggers of CDG Therapy Research Tool Interdependence
“(…) it’s a bit of a vicious circle, (…) we don’t have enough biomarkers to accurately diagnose, but without an accurate diagnosis families can’t get into the system, to be enrolled in the biobanks, to permit the development of biomarkers (…) it’s shortening that “diagnostic odyssey” (…) so that I as a patient can get to see you as an expert quickly, and you can take my samples and put them into the biobank, to do the research to find the biomarkers (…)”(multiple roles)
“(…) we don’t even have [CDG] newborn screening, because most clinicians think it’s not treatable (…)”(HCP)
“(…) a lot of older patients (…) can benefit from [management therapies] whereas a cure should be preferably implemented by early diagnosis”(family member)
4. Discussion
4.1. Study Limitations
- -
- -
- Think tanks were also exclusively conducted in English. They may have led to the underrepresentation of participants’ views with limited English proficiency while favoring native speakers expressing their opinion and sharing their insights. In order to tackle this potential limitation, command of English and other languages (according to the nationality reported in the registration form) proficiency was a weighed factor when assigning participants to the think tank groups. Additionally, during the think tank discussions, participants were stimulated to express themselves in their mother tongue when necessary, benefiting from translation into English by another discussion group member;
- -
- Our study may have been subject to selection bias, particularly regarding recruitment for the think tanks, which occurred in connection with the 4th World Conference on CDG. Conference participants tend to be more engaged, academically educated, and/or financially empowered participants when compared to the overall CDG community [25].
4.2. Study Strengths
- -
- The development and implementation of a sustained participatory, community-led study including lay and professional stakeholders from diverse backgrounds and nationalities enabled the creation of a comprehensive map of the current CDG therapeutic research landscape and the offering of orientation for future therapy R&D. Importantly, the reproducibility of this approach offers the possibility of its adaptation by other disease communities and/or for other biological questions;
- -
- The adoption of mixed research methods enriched the completeness and depth of the data. At the same time, it made data collection a stepwise process, guiding data analysis and strengthening study conclusions;
- -
- The e-survey methodology used to gather quantitative data, including survey development and dissemination, has been optimized by our research team in previous works [23,24]. Regarding the qualitative study, the key strength of the think tank methodology is based on the possibility of participants meeting face-to-face in a safe environment which stimulates sharing, reflection, and co-learning. The previous identification of the World Conference on CDG for Families and Professionals as a collaborative platform aided and added another level of innovation to the study design [25]. Framing the study around the World Conference on CDG also allowed the community to co-develop the conference agenda according to their needs and preferences. These multiple study outcomes underscore the adaptability and positive impact of people-centricity in research.
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jaeken, J.; Vanderschueren-Lodeweyckx, M.; Casaer, P. Familial psychomotor retardation with markedly fluctuating serum prolactin, FSH and GH levels, partial TBG deficiency, increased serum arylsulphatase A and increased CSF protein: A new syndrome? Pediatr Res. 1980, 14, 179. [Google Scholar] [CrossRef] [Green Version]
- Ferreira, C.R.; Altassan, R.; Marques-Da-Silva, D.; Francisco, R.; Jaeken, J.; Morava, E. Recognizable phenotypes in CDG. J. Inherit. Metab. Dis. 2018, 41, 541–553. [Google Scholar] [CrossRef] [PubMed]
- Péanne, R.; de Lonlay, P.; Foulquier, F.; Kornak, U.; Lefeber, D.J.; Morava, E.; Pérez, B.; Seta, N.; Thiel, C.; Van Schaftingen, E.; et al. Congenital disorders of glycosylation (CDG): Quo vadis? Eur. J. Med. Genet. 2018, 61, 643–663. [Google Scholar] [CrossRef]
- Francisco, R.; Pascoal, C.; Marques-Da-Silva, D.; Morava, E.; Gole, G.A.; Coman, D.; Jaeken, J.; Ferreira, V.D.R. Keeping an eye on congenital disorders of Oglycosylation: A systematic literature review. J. Inherit. Metab. Dis. 2019, 42, 29–48. [Google Scholar] [CrossRef] [PubMed]
- Marques-Da-Silva, D.; Francisco, R.; Webster, D.; Ferreira, V.D.R.; Jaeken, J.; Pulinilkunnil, T. Cardiac complications of congenital disorders of glycosylation (CDG): A systematic review of the literature. J. Inherit. Metab. Dis. 2017, 40, 657–672. [Google Scholar] [CrossRef]
- Marques-Da-Silva, D.; Ferreira, V.D.R.; Monticelli, M.; Janeiro, P.; Videira, P.; Witters, P.; Jaeken, J.; Cassiman, D. Liver involvement in congenital disorders of glycosylation (CDG). A systematic review of the literature. J. Inherit. Metab. Dis. 2017, 40, 195–207. [Google Scholar] [CrossRef]
- Pascoal, C.; Francisco, R.; Ferro, T.; Ferreira, V.D.R.; Jaeken, J.; Videira, P.A. CDG and immune response: From bedside to bench and back. J. Inherit. Metab. Dis. 2020, 43, 90–124. [Google Scholar] [CrossRef]
- Brasil, S.; Pascoal, C.; Francisco, R.; Marques-Da-Silva, D.; Andreotti, G.; Videira, P.A.; Morava, E.; Jaeken, J.; Ferreira, V.D.R. CDG Therapies: From Bench to Bedside. Int. J. Mol. Sci. 2018, 19, 1304. [Google Scholar] [CrossRef] [Green Version]
- Witters, P.; Cassiman, D.; Morava, E. Nutritional Therapies in Congenital Disorders of Glycosylation (CDG). Nutrients 2017, 9, 1222. [Google Scholar] [CrossRef] [Green Version]
- Iyer, S.; Sam, F.S.; DiPrimio, N.; Preston, G.; Verheijen, J.; Murthy, K.; Parton, Z.; Tsang, H.; Lao, J.; Morava, E.; et al. Repurposing the aldose reductase inhibitor and diabetic neuropathy drug epalrestat for the congenital disorder of glycosylation PMM2-CDG. Dis. Model. Mech. 2019, 12, dmm040584. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Monseny, A.F.; Bolasell, M.; Callejón-Póo, L.; Cuadras, D.; Freniche, V.; Itzep, D.C.; Gassiot, S.; Arango, P.; Casas-Alba, D.; De La Morena, E.; et al. AZATAX: Acetazolamide safety and efficacy in cerebellar syndrome in PMM2 congenital disorder of glycosylation (PMM2-CDG). Ann. Neurol. 2019, 85, 740–751. [Google Scholar] [CrossRef] [PubMed]
- Word CDG Organization. CDG Pipeline. 2021. Available online: https://worldcdg.org/drug-development/pipeline (accessed on 1 July 2021).
- Chen, J.; Li, X.; Edmondson, A.; Meyers, G.D.; Izumi, K.; Ackermann, A.M.; Morava, E.; Ficicioglu, C.; Bennett, M.J.; He, M. Increased Clinical Sensitivity and Specificity of Plasma Protein N-Glycan Profiling for Diagnosing Congenital Disorders of Glycosylation by Use of Flow Injection–Electrospray Ionization–Quadrupole Time-of-Flight Mass Spectrometry. Clin. Chem. 2019, 65, 653–663. [Google Scholar] [CrossRef] [PubMed]
- Bruneel, A.; Cholet, S.; Tran, N.T.; Mai, T.D.; Fenaille, F. CDG biochemical screening: Where do we stand? Biochim. Biophys. Acta (BBA)-Gen. Subj. 2020, 1864, 129652. [Google Scholar] [CrossRef] [PubMed]
- Haouari, W.; Dubail, J.; Lounis-Ouaras, S.; Prada, P.; Bennani, R.; Roseau, C.; Huber, C.; Afenjar, A.; Colin, E.; Vuillaumier-Barrot, S.; et al. Serum bikunin isoforms in congenital disorders of glycosylation and linkeropathies. J. Inherit. Metab. Dis. 2020, 43, 1349–1359. [Google Scholar] [CrossRef]
- Ligezka, A.N.; Radenkovic, S.; Saraswat, M.; Garapati, K.; Ranatunga, W.; Krzysciak, W.; Yanaihara, H.; Preston, G.; Brucker, W.; BA, R.M.M.; et al. Sorbitol Is a Severity Biomarker for PMM2-CDG with Therapeutic Implications. Ann. Neurol. 2021, 90, 887–900. [Google Scholar] [CrossRef]
- Griggs, R.C.; Batshaw, M.; Dunkle, M.; Gopal-Srivastava, R.; Kaye, E.; Krischer, J.; Nguyen, T.; Paulus, K.; Merkel, P.A. Clinical research for rare disease: Opportunities, challenges, and solutions. Mol. Genet. Metab. 2009, 96, 20–26. [Google Scholar] [CrossRef] [Green Version]
- Augustine, E.F.; Adams, H.R.; Mink, J.W. Clinical Trials in Rare Disease. J. Child Neurol. 2013, 28, 1142–1150. [Google Scholar] [CrossRef]
- Hemati, M. Stakeholder Engagement, 1st ed.; Dahinden, M., Paschke, M., Eds.; Zurich-Basel Plant Science Center: Zürich, Switzerland, 2020. [Google Scholar] [CrossRef]
- Dwyer, A.A.; Quinton, R.; Morin, D.; Pitteloud, N. Identifying the unmet health needs of patients with congenital hypogonadotropic hypogonadism using a web-based needs assessment: Implications for online interventions and peer-to-peer support. Orphanet, J. Rare Dis. 2014, 9, 83. [Google Scholar] [CrossRef] [Green Version]
- Goodspeed, K.; Bliss, G.; Linnehan, D. Bringing everyone to the table–findings from the 2018 Phelan-McDermid Syndrome Foundation International Conference. Orphanet, J. Rare Dis. 2020, 15, 152. [Google Scholar] [CrossRef]
- de Freitas, C.; Amorim, M.; Machado, H.; Teles, E.L.; Baptista, M.J.; Renedo, A.; Provoost, V.; Silva, S. Public and patient involvement in health data governance (DATAGov): Protocol of a people-centred, mixed-methods study on data use and sharing for rare diseases care and research. BMJ Open 2021, 11, e044289. [Google Scholar] [CrossRef]
- Marques-Da-Silva, D.; Francisco, R.; Ferreira, V.D.R.; Forbat, L.; Lagoa, R.; Videira, P.A.; Witters, P.; Jaeken, J.; Cassiman, D. An Electronic Questionnaire for Liver Assessment in Congenital Disorders of Glycosylation (LeQCDG): A Patient-Centered Study. JIMD Rep. 2019, 44, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Francisco, R.; Pascoal, C.; Marques-Da-Silva, D.; Brasil, S.; Pimentel-Santos, F.M.; Altassan, R.; Jaeken, J.; Grosso, A.R.; Ferreira, V.D.R.; Videira, P.A. New Insights into Immunological Involvement in Congenital Disorders of Glycosylation (CDG) from a People-Centric Approach. J. Clin. Med. 2020, 9, 2092. [Google Scholar] [CrossRef] [PubMed]
- De Freitas, C.; Dos Reis, V.; Silva, S.; Videira, P.A.; Morava, E.; Jaeken, J. Public and patient involvement in needs assessment and social innovation: A people-centred approach to care and research for congenital disorders of glycosylation. BMC Health Serv. Res. 2017, 17, 682. [Google Scholar] [CrossRef] [Green Version]
- Strauss, A.; Corbin, J. Basics of Qualitative Research: Techniques and Procedures for Developing Grounded Theory, 2nd ed.; Sage Publications, Inc.: Thousand Oaks, CA, USA, 1998. [Google Scholar]
- Mitchell, D.; Geissler, J.; Parry-Jones, A.; Keulen, H.; Schmitt, D.C.; Vavassori, R.; Matharoo-Ball, B. Biobanking from the patient perspective. Res. Involv. Engag. 2015, 1, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mora, M.; Angelini, C.; Bignami, F.; Bodin, A.-M.; Crimi, M.; Di Donato, J.H.; Felice, A.; Jaeger, C.; Karcagi, V.; LeCam, Y.; et al. The EuroBioBank Network: 10 years of hands-on experience of collaborative, transnational biobanking for rare diseases. Eur. J. Hum. Genet. 2015, 23, 1116–1123. [Google Scholar] [CrossRef] [PubMed]
- McGettigan, P.; Olmo, C.A.; Plueschke, K.; Castillon, M.; Zondag, D.N.; Bahri, P.; Kurz, X.; Mol, P.G.M. Patient Registries: An Underused Resource for Medicines Evaluation. Drug Saf. 2019, 42, 1343–1351. [Google Scholar] [CrossRef] [Green Version]
- Strimbu, K.; Tavel, J.A. What Are Biomarkers? Curr. Opin. HIV AIDS 2010, 5, 463–466. [Google Scholar] [CrossRef]
- Tambuyzer, E.; Vandendriessche, B.; Austin, C.P.; Brooks, P.J.; Larsson, K.; Needleman, K.I.M.; Valentine, J.; Davies, K.; Groft, S.C.; Preti, R.; et al. Therapies for rare diseases: Therapeutic modalities, progress and challenges ahead. Nat. Rev. Drug Discov. 2020, 19, 93–111. [Google Scholar] [CrossRef]
- McGonigle, P.; Ruggeri, B. Animal models of human disease: Challenges in enabling translation. Biochem. Pharmacol. 2014, 87, 162–171. [Google Scholar] [CrossRef]
- Huml, R.A.; Dawson, J.; Bailey, M.; Nakas, N.; Williams, J.; Kolochavina, M.; Huml, J.R. Accelerating Rare Disease Drug Development: Lessons Learned from Muscular Dystrophy Patient Advocacy Groups. Ther. Innov. Regul. Sci. 2021, 55, 370–377. [Google Scholar] [CrossRef]
- Lauritsen, S.M.; Kristensen, M.; Olsen, M.V.; Larsen, M.S.; Lauritsen, K.M.; Jørgensen, M.; Lange, J.; Thiesson, B. Explainable artificial intelligence model to predict acute critical illness from electronic health records. Nat. Commun. 2020, 11, 3852. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Monseny, A.; Cuadras, D.; Bolasell, M.; Muchart, J.; Arjona, C.; Borregan, M.; Algrabli, A.; Montero, R.; Artuch, R.; Velázquez-Fragua, R.; et al. From gestalt to gene: Early predictive dysmorphic features of PMM2-CDG. J. Med. Genet. 2019, 56, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Applequist, J.; Burroughs, C.; Jr, A.R.; Merkel, P.A.; Rothenberg, M.E.; Trapnell, B.; Desnick, R.J.; Sahin, M.; Krischer, J.P. A novel approach to conducting clinical trials in the community setting: Utilizing patient-driven platforms and social media to drive web-based patient recruitment. BMC Med. Res. Methodol. 2020, 20, 58. [Google Scholar] [CrossRef] [PubMed]
- Gulhan, I. A unique e-health and telemedicine implementation: European Reference Networks for rare diseases. J. Public Health 2020, 28, 223–225. [Google Scholar] [CrossRef]
- Talarico, R.; Aguilera, S.; Alexander, T.; Amoura, Z.; Antunes, A.M.; Arnaud, L.; Avcin, T.; Beretta, L.; Bombardieri, S.; Burmester, G.R.; et al. The impact of COVID-19 on rare and complex connective tissue diseases: The experience of ERN ReCONNET. Nat. Rev. Rheumatol. 2021, 17, 177–184. [Google Scholar] [CrossRef]
- Courbier, S.; Dimond, R.; Bros-Facer, V. Share and protect our health data: An evidence based approach to rare disease patients’ perspectives on data sharing and data protection-quantitative survey and recommendations. Orphanet J. Rare Dis. 2019, 14, 175. [Google Scholar] [CrossRef]
- Wilkinson, M.D.; Dumontier, M.; Aalbersberg, I.J.; Appleton, G.; Axton, M.; Baak, A.; Blomberg, N.; Boiten, J.W.; da Silva Santos, L.B.; Bourne, P.E.; et al. The FAIR Guiding Principles for scientific data management and stewardship. Sci. Data 2016, 3, 160018. [Google Scholar] [CrossRef] [Green Version]
- Cornel, M.C.; Rigter, T.; Jansen, M.E.; Henneman, L. Neonatal and carrier screening for rare diseases: How innovation challenges screening criteria worldwide. J. Community Genet. 2020, 12, 257–265. [Google Scholar] [CrossRef]
- Kaufmann, P.; Pariser, A.R.; Austin, C. From scientific discovery to treatments for rare diseases–the view from the National Center for Advancing Translational Sciences–Office of Rare Diseases Research. Orphanet J. Rare Dis. 2018, 13, 196. [Google Scholar] [CrossRef]
- Héon-Klin, V. European Reference networks for rare diseases: What is the conceptual framework? Orphanet J. Rare Dis. 2017, 12, 1–9. [Google Scholar] [CrossRef] [Green Version]
- RDCRN. The Frontiers in CDG Consortium. 2021. Available online: https://www.rarediseasesnetwork.org/fcdgc (accessed on 16 May 2021).
- CDG & Allies-PPAIN C & A-P and PAIN. 2021. Available online: https://www.researchcdg.com/ (accessed on 16 May 2021).
- Miller, K.L.; Mueller, C.; Liu, G.; Needleman, K.I.M.; Maynard, J. FDA orphan products clinical trial grants: Assessment of outcomes and impact on rare disease product development. Orphanet J. Rare Dis. 2020, 15, 234. [Google Scholar] [CrossRef] [PubMed]
- Julkowska, D.; Austin, C.P.; Cutillo, C.M.; Gancberg, D.; Hager, C.; Halftermeyer, J.; Jonker, A.H.; Lau, L.P.L.; Norstedt, I.; Rath, A.; et al. The importance of international collaboration for rare diseases research: A European perspective. Gene Ther. 2017, 24, 562–571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- CDG CARE; CDG Canada; INVITAE. CDG Connect. 2021. Available online: https://connect.invitae.com/org/cdg (accessed on 14 July 2021).
- Pascoal, C.; Brasil, S.; Francisco, R.; Marques-Da-Silva, D.; Rafalko, A.; Jaeken, J.; Videira, P.A.; Barros, L.; Ferreira, V.D.R. Patient and observer reported outcome measures to evaluate health-related quality of life in inherited metabolic diseases: A scoping review. Orphanet J. Rare Dis. 2018, 13, 215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
 —bidirectional flow; |→—flow interruption or blockage; X—blockage, low levels or quantity, and/or inaccessibility; √—availability, increased levels or quantity and accessibility.
—bidirectional flow; |→—flow interruption or blockage; X—blockage, low levels or quantity, and/or inaccessibility; √—availability, increased levels or quantity and accessibility.
 —bidirectional flow; |→—flow interruption or blockage; X—blockage, low levels or quantity, and/or inaccessibility; √—availability, increased levels or quantity and accessibility.
—bidirectional flow; |→—flow interruption or blockage; X—blockage, low levels or quantity, and/or inaccessibility; √—availability, increased levels or quantity and accessibility.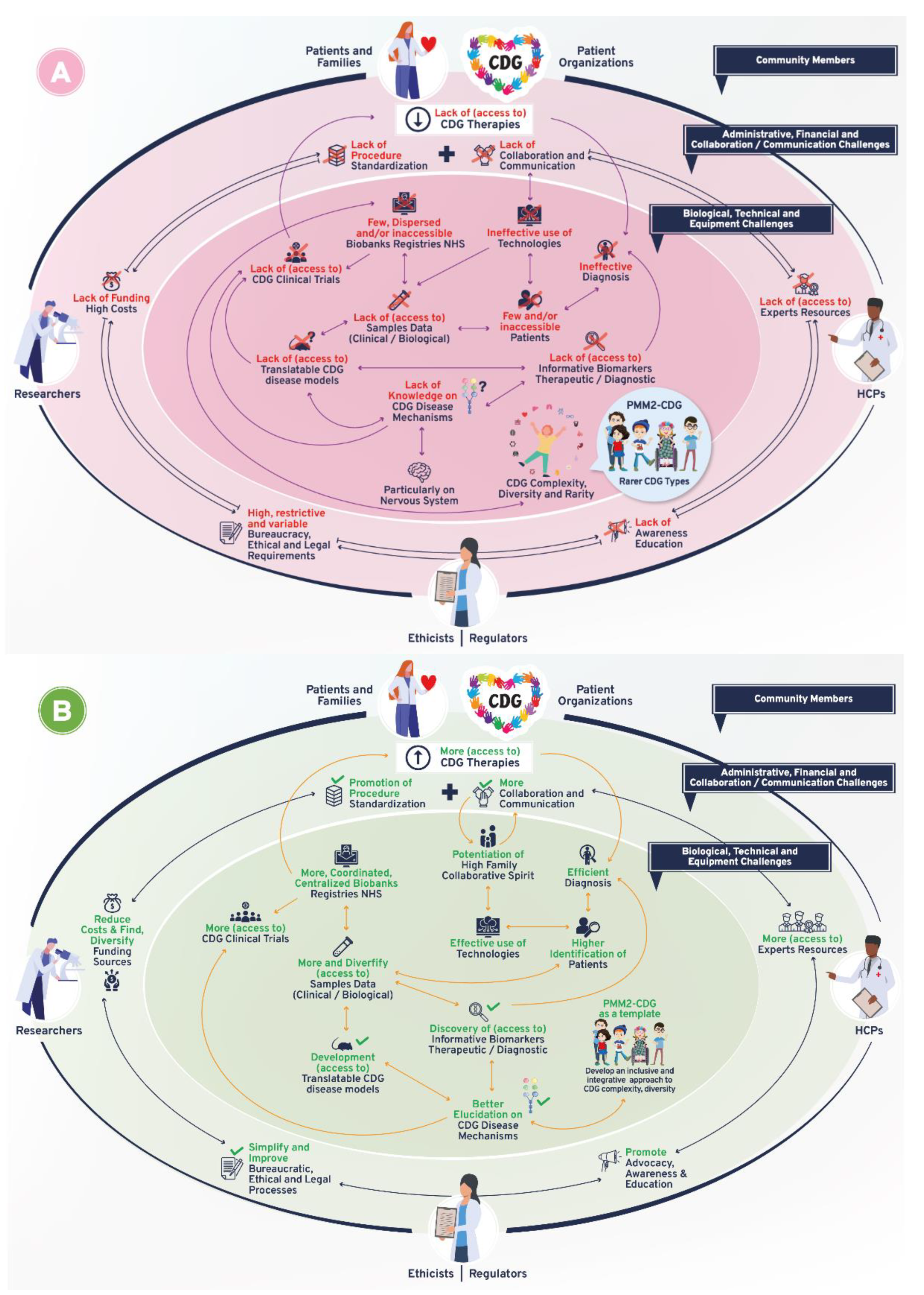

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Francisco, R.; Brasil, S.; Pascoal, C.; Edmondson, A.C.; Jaeken, J.; Videira, P.A.; de Freitas, C.; Ferreira, V.d.R.; Marques-da-Silva, D. A Community-Led Approach as a Guide to Overcome Challenges for Therapy Research in Congenital Disorders of Glycosylation. Int. J. Environ. Res. Public Health 2022, 19, 6829. https://doi.org/10.3390/ijerph19116829
Francisco R, Brasil S, Pascoal C, Edmondson AC, Jaeken J, Videira PA, de Freitas C, Ferreira VdR, Marques-da-Silva D. A Community-Led Approach as a Guide to Overcome Challenges for Therapy Research in Congenital Disorders of Glycosylation. International Journal of Environmental Research and Public Health. 2022; 19(11):6829. https://doi.org/10.3390/ijerph19116829
Chicago/Turabian StyleFrancisco, Rita, Sandra Brasil, Carlota Pascoal, Andrew C. Edmondson, Jaak Jaeken, Paula A. Videira, Cláudia de Freitas, Vanessa dos Reis Ferreira, and Dorinda Marques-da-Silva. 2022. "A Community-Led Approach as a Guide to Overcome Challenges for Therapy Research in Congenital Disorders of Glycosylation" International Journal of Environmental Research and Public Health 19, no. 11: 6829. https://doi.org/10.3390/ijerph19116829
APA StyleFrancisco, R., Brasil, S., Pascoal, C., Edmondson, A. C., Jaeken, J., Videira, P. A., de Freitas, C., Ferreira, V. d. R., & Marques-da-Silva, D. (2022). A Community-Led Approach as a Guide to Overcome Challenges for Therapy Research in Congenital Disorders of Glycosylation. International Journal of Environmental Research and Public Health, 19(11), 6829. https://doi.org/10.3390/ijerph19116829






