Energy Metabolism in Gynecological Cancers: A Scoping Review
Abstract
:1. Introduction
2. Materials and Methods
3. Results
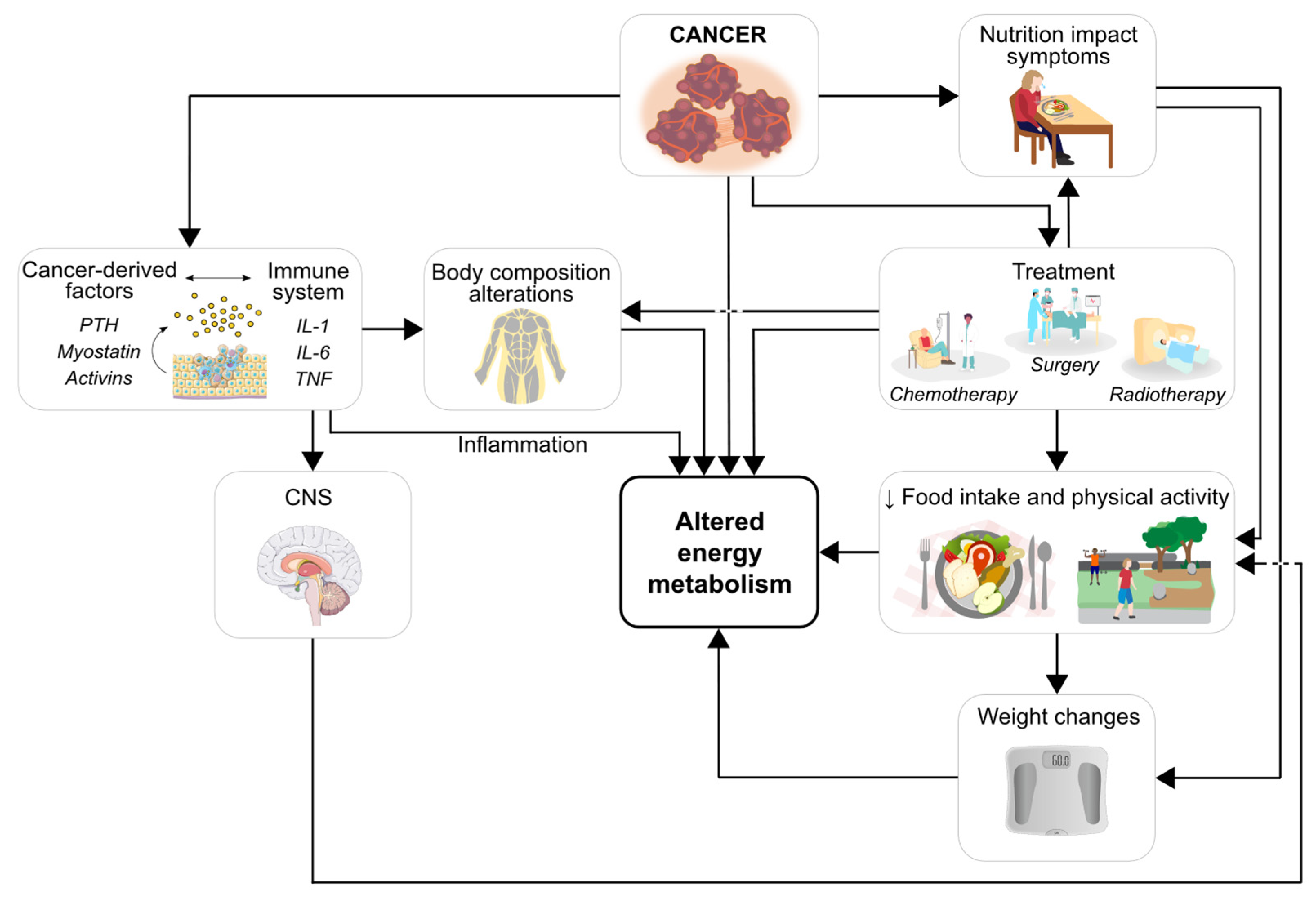
4. Energy Metabolism in Gynecological Cancers: Current Knowledge
5. Future Directions
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Purcell, S.A.; Elliott, S.A.; Baracos, V.E.; Chu, Q.S.C.; Prado, C.M. Key determinants of energy expenditure in cancer and implications for clinical practice. Eur. J. Clin. Nutr. 2016, 70, 1230–1238. [Google Scholar] [CrossRef] [PubMed]
- Ryan, A.M.; Power, D.G.; Daly, L.; Cushen, S.J.; Ní, E. Cancer-associated malnutrition, cachexia and sarcopenia: The skeleton in the hospital closet 40 years later Proceedings of the Nutrition Society Procee. Proc. Nutr. Soc. 2016, 75, 199–211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deutz, N.E.; Ashurst, I.; Ballesteros, M.D.; Bear, D.E.; Cruz-Jentoft, A.J.; Genton, L.; Landi, F.; Laviano, A.; Norman, K.; Prado, C.M. The underappreciated role of low muscle mass in the management of malnutrition. J. Am. Med. Dir. Assoc. 2019, 20, 22–27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prado, C.M.; Lieffers, J.R.; McCargar, L.J.; Reiman, T.; Sawyer, M.B.; Martin, L.; Baracos, V.E. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: A population-based study. Lancet Oncol. 2008, 9, 629–635. [Google Scholar] [CrossRef]
- Purcell, S.A.; Elliott, S.A.; Kroenke, C.H.; Sawyer, M.B.; Prado, C.M. Impact of Body Weight and Body Composition on Ovarian Cancer Prognosis. Curr. Oncol. Rep. 2016, 18, 8. [Google Scholar] [CrossRef]
- Goodrose-Flores, C.; Eke, H.; Bonn, S.E.; Björkhem-Bergman, L.; Trolle Lagerros, Y. Weight Loss in Advanced Cancer: Sex Differences in Health-Related Quality of Life and Body Image. Life 2022, 12, 105. [Google Scholar] [CrossRef]
- Lane, G. Obesity and gynaecological cancer. Menopause Int. 2008, 14, 33–37. [Google Scholar] [CrossRef]
- Donahoo, W.T.; Levine, J.A.; Melanson, E.L. Variability in energy expenditure and its components. Curr. Opin. Clin. Nutr. Metab. Care 2004, 7, 599–605. [Google Scholar] [CrossRef]
- Psota, T.; Chen, K.Y. Measuring energy expenditure in clinical populations: Rewards and challenges. Eur. J. Clin. Nutr. 2013, 67, 436–442. [Google Scholar] [CrossRef] [Green Version]
- Purcell, S.A.; Johnson-Stoklossa, C.; Tibaes, J.R.B.; Frankish, A.; Elliott, S.A.; Padwal, R.; Prado, C.M. Accuracy and reliability of a portable indirect calorimeter compared to whole-body indirect calorimetry for measuring resting energy expenditure. Clin. Nutr. ESPEN 2020, 39, 67–73. [Google Scholar] [CrossRef]
- Purcell, S.A.; Elliott, S.A.; Baracos, V.E.; Chu, Q.S.C.; Sawyer, M.B.; Mourtzakis, M.; Easaw, J.C.; Spratlin, J.L.; Siervo, M.; Prado, C.M. Accuracy of Resting Energy Expenditure Predictive Equations in Patients with Cancer. Nutr. Clin. Pract. 2019, 34, 922–934. [Google Scholar] [CrossRef]
- Arends, J.; Bachmann, P.; Baracos, V.; Barthelemy, N.; Bertz, H.; Bozzetti, F.; Fearon, K.; Hütterer, E.; Isenring, E.; Kaasa, S.; et al. ESPEN guidelines on nutrition in cancer patients. Clin. Nutr. 2017, 36, 11–48. [Google Scholar] [CrossRef] [Green Version]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA extension for scoping reviews (PRISMA-ScR): Checklist and explanation. Ann. Intern Med. 2018, 169, 467–473. [Google Scholar] [CrossRef] [Green Version]
- Boothby, W.; Sandiford, I. Summary of the Basal Metabolism Data on 8,614 Subjects with Especial Reference to the Normal Standards for the Estimation of the Basal Metabolic Rate. J. Biol. Chem. 1992, 54, 783–803. [Google Scholar] [CrossRef]
- Harris, J.A.; Benedict, F.G. A Biometric Study of Human Basal Metabolism. Proc. Natl. Acad. Sci. USA 1918, 4, 370–373. [Google Scholar] [CrossRef] [Green Version]
- Bowman, R.O.; Pitts, H.C. Calcium and Phosphatase Studies in Cancer of the Female Sex with a Consideration of Basal Metabolic Rate and Urine pH. Am. J. Obstet. Gynecol. 1936, 32, 957–963. [Google Scholar] [CrossRef]
- Dickerson, R.; White, K.; Curcillo, P.; King, S.; Mullen, J. Resting Energy Expenditure of Patients with Gynecologic Malignancies. J. Am. Coll. Nutr. 1995, 14, 448–454. [Google Scholar] [CrossRef]
- de La Maza, M.P.; Agudelo, G.M.; Yudin, T.; Gattás, V.; Barrera, G.; Bunout, D.; Hirsch, S. Long-term nutritional and digestive consequences of pelvic radiation. J. Am. Coll. Nutr. 2004, 23, 102–107. [Google Scholar] [CrossRef]
- de la Maza, M.P.; Gotteland, M.; Ramírez, C.; Araya, M.; Yudin, T.; Bunout, D.; Hirsch, S. Acute nutritional and intestinal changes after pelvic radiation. J. Am. Coll. Nutr. 2001, 20, 637–642. [Google Scholar] [CrossRef]
- Macciò, A.; Madeddu, C.; Gramignano, G.; Mulas, C.; Floris, C.; Sanna, E.; Cau, M.C.; Panzone, F.; Mantovani, G. A randomized phase III clinical trial of a combined treatment for cachexia in patients with gynecological cancers: Evaluating the impact on metabolic and inflammatory profiles and quality of life. Gynecol. Oncol. 2012, 124, 417–425. [Google Scholar] [CrossRef]
- Hills, A.P.; Mokhtar, N.; Byrne, N.M. Assessment of Physical Activity and Energy Expenditure: An Overview of Objective Measures. Front. Nutr. 2014, 1, 5. [Google Scholar] [CrossRef]
- Nguyen, T.Y.V.; Batterham, M.J.; Edwards, C. Comparison of Resting Energy Expenditure Between Cancer Subjects and Healthy Controls: A Meta-Analysis. Nutr. Cancer 2016, 68, 374–387. [Google Scholar] [CrossRef] [Green Version]
- Kamal, R.; Hamed, S.; Mansour, S.; Mounir, Y.; Abdel Sallam, S. Ovarian cancer screening-ultrasound; impact on ovarian cancer mortality. Br. J. Radiol. 2018, 91, 20170571. [Google Scholar] [CrossRef]
- Rinninella, E.; Fagotti, A.; Cintoni, M.; Raoul, P.; Scaletta, G.; Quagliozzi, L.; Miggiano, G.A.D.; Scambia, G.; Gasbarrini, A.; Mele, M.C. Nutritional interventions to improve clinical outcomes in ovarian cancer: A systematic review of randomized controlled trials. Nutrients 2019, 11, 1404. [Google Scholar] [CrossRef] [Green Version]
- Fullmer, S.; Benson-Davies, S.; Earthman, C.P.; Frankenfield, D.C.; Gradwell, E.; Lee, P.S.P.; Piemonte, T.; Trabulsi, J. Evidence analysis library review of best practices for performing indirect calorimetry in healthy and non-critically ill individuals. J. Acad. Nutr. Diet 2015, 115, 1417–1446. [Google Scholar] [CrossRef]
- Henry, C.J. Basal metabolic rate studies in humans: Measurement and development of new equations. Public Health Nutr. 2005, 8, 1133–1152. [Google Scholar] [CrossRef]
- Compher, C.; Frankenfield, D.; Keim, N.; Roth-Yousey, L. Best Practice Methods to Apply to Measurement of Resting Metabolic Rate in Adults: A Systematic Review. J. Am. Diet Assoc. 2006, 106, 881–903. [Google Scholar] [CrossRef] [PubMed]
- Levine, J.A. Measurement of energy expenditure. Public Health Nutr. 2005, 8, 1123–1132. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, I.P.; Elliott, S.A.; Siervo, M.; Padwal, R.; Bertoli, S.; Battezzati, A.; Prado, C.M. Is Obesity Associated with Altered Energy Expenditure? Adv. Nutr. 2016, 7, 476–487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heymsfield, S.B.; Gallagher, D.; Poehlman, E.T.; Wolper, C.; Nonas, K.; Nelson, D.; Wang, Z.M. Menopausal changes in body composition and energy expenditure. Exp. Gerontol. 1994, 29, 377–389. [Google Scholar] [CrossRef]
- Webb, P. 24-hour energy expenditure and the menstrual cycle. Am. J. Clin. Nutr. 1986, 44, 614–619. [Google Scholar] [CrossRef] [Green Version]
- Frankenfield, D.; Roth-Yousey, L.; Compher, C. Comparison of predictive equations for resting metabolic rate in healthy nonobese and obese adults: A systematic review. J. Acad. Nutr. Diet 2005, 105, 775–789. [Google Scholar] [CrossRef]
- Fearon, K.; Strasser, F.; Anker, S.D.; Bosaeus, I.; Bruera, E.; Fainsinger, R.L.; Jatoi, A.; Loprinzi, C.; MacDonald, N.; Mantovani, G.; et al. Definition and classification of cancer cachexia: An international consensus. Lancet Oncol. 2011, 12, 489–495. [Google Scholar] [CrossRef]
- Martin, L.; Birdsell, L.; MacDonald, N.; Reiman, T.; Clandinin, M.T.; McCargar, L.J.; Murphy, R.; Ghosh, S.; Sawyer, M.B.; Baracos, V.E. Cancer cachexia in the age of obesity: Skeletal muscle depletion is a powerful prognostic factor, independent of body mass index. J. Clin. Oncol. 2013, 31, 1539–1547. [Google Scholar] [CrossRef]
- Prado, C.M.; Baracos, V.E.; McCargar, L.J.; Reiman, T.; Mourtzakis, M.; Tonkin, K.; Mackey, J.R.; Koski, S.; Pituskin, E.; Sawyer, M.B. Sarcopenia as a determinant of chemotherapy toxicity and time to tumor progression in metastatic breast cancer patients receiving capecitabine treatment. Clin. Cancer Res. 2009, 15, 2920–2926. [Google Scholar] [CrossRef] [Green Version]
- Prado, C.M.; Lieffers, J.R.; Bowthorpe, L.; Baracos, V.E.; Mourtzakis, M.; McCargar, L.J. Sarcopenia and physical function in overweight patients with advanced cancer. Can. J. Diet Pract. Res. 2013, 74, 69–74. [Google Scholar] [CrossRef]
- Purcell, S.A.; Elliott, S.A.; Ryan, A.M.; Sawyer, M.B.; Prado, C.M. Accuracy of a Portable Indirect Calorimeter for Measuring Resting Energy Expenditure in Individuals With Cancer. JPEN J. Parenter Enteral. Nutr. 2019, 43, 145–151. [Google Scholar] [CrossRef]
- Purcell, S.A.; Johnson-Stoklossa, C.; Tibaes, J.R.B.; Frankish, A.; Elliott, S.A.; Padwal, R.; Prado, C.M. Accuracy of the MedGem® portable indirect calorimeter for measuring resting energy expenditure in adults with class II or III obesity. Clin. Nutr. ESPEN 2020, 40, 408–411. [Google Scholar] [CrossRef]

| Author, Year | Mean Age (Years) | Cancer Type (n) | Cancer Stage 1 | Cancer Treatment | Energy Expenditure (kcal/Day) Measured Predicted | Abnormalities in Energy Metabolism | Technique/Equation | |
|---|---|---|---|---|---|---|---|---|
| Bowman et al., 1936 [17] | 54 | Ovarian (n = 2); Cervical (n = 27); Tubal (n = 1); Uterine (n = 5); Vaginal (n = 2); Vulvar (n = 1) | Not reported | Not reported | Not reported | Not reported | Hypermetabolism 2 Deviation from “normal 3” ranged from −13.6% to +53.5%; average deviation: +9.14% | Benedict–Roth apparatus/not specified |
| Dickerson et al., 1995 [18] | Cervical: 55 Ovarian: 58 | Cervical (n = 30); Ovarian (n = 31) | I-II: Cervical (n = 15); Ovarian (n = 1) III-IV: Cervical (n = 10); Ovarian (n = 27) Unknown: Cervical (n = 5); Ovarian (n = 3) | Not reported | Ovarian: 1332 ± 214 Cervical: 1179 ± 181 | Ovarian: 1234 ± 115 Cervical: 1203 ± 118 | Hypometabolic: 21% Hypermetabolic: 34% Normometabolic: 45% | Indirect calorimetry (device not specified)/Harris–Benedict |
| de la Maza et al., 2001 [20] | n/A 4 | Cervical (n = 10); Endometrial (n = 3); Uterine (n = 1); Vaginal cupula (n = 1) | IB: Cervical (n = 5) IC: Endometrial (n = 2) II: Endometrial (n = 1) IIB: Cervical (n = 3) IIIB: Cervical (n = 2) Unknown: Uterine (n = 1); Vaginal cupula (n = 1) | 45–50 Gy of pelvic external radiation over 5 weeks | Pre-treatment 5: 1673 ± 488 Post-treatment 5: 1585 ± 275 | Not reported or possible to calculate | Pre-treatment: Hypermetabolism (measured REE 125% of predicted) Post-treatment: Not reported | Sensor Medic model 2900 calorimeter/Harris–Benedict |
| de la Maza et al., 2004 [19] | 49 | Cervical (n = 10); Endometrial (n = 3); Uterine (n = 1); Vaginal cupula (n = 1) | IB: Cervical (n = 5) IC: Endometrial (n = 2) II: Endometrial (n = 1) IIB: Cervical (n = 3) IIIB: Cervical (n = 2) Unknown: Uterine (n = 1); Vaginal cupula (n = 1) | 45–50 Gy of pelvic external radiation over 5 weeks (n = 15) and post-external ratiation intracavitary radiotherapy (n = 14) and/or surgery prior to external radiation (n = 7) | Pre-treatment 5,6: 1690 ± 231 Post-treatment: 1644 ± 292 Two years post-treatment: 1287 ± 175 | Pre-treatment 5,6: 1363 Post-treatment: 1359 Two years post-treatment: 1399 | Hypermetabolism (pre- and post-treatment); normometabolism two years post-treatment | Sensor Medic model 2900 calorimeter/Harris–Benedict |
| Macciò et al., 2012 [21] | 60 | Ovarian (n = 50); Endometrial (n = 49); Cervical (n = 25) | IIIC: Ovarian (n = 5) IV: Ovarian (n = 45); Endometrial (n = 49); Cervical (n = 25) | Previous chemotherapy (n = 124) and/or ongoing palliative chemotherapy (n = 90) | Baseline (prior to intervention): Arm 1: 1166 ± 440 Arm 2: 1157 ± 279 | Baseline (prior to intervention) 7: Arm 1: 1146 Arm 2: 1155 | Normometabolism | Medgem®/Harris–Benedict |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pagano, A.P.; Ford, K.L.; Porter Starr, K.N.; Kiss, N.; Steed, H.; Kung, J.Y.; Elango, R.; Prado, C.M. Energy Metabolism in Gynecological Cancers: A Scoping Review. Int. J. Environ. Res. Public Health 2022, 19, 6419. https://doi.org/10.3390/ijerph19116419
Pagano AP, Ford KL, Porter Starr KN, Kiss N, Steed H, Kung JY, Elango R, Prado CM. Energy Metabolism in Gynecological Cancers: A Scoping Review. International Journal of Environmental Research and Public Health. 2022; 19(11):6419. https://doi.org/10.3390/ijerph19116419
Chicago/Turabian StylePagano, Ana Paula, Katherine L. Ford, Kathryn N. Porter Starr, Nicole Kiss, Helen Steed, Janice Y. Kung, Rajavel Elango, and Carla M. Prado. 2022. "Energy Metabolism in Gynecological Cancers: A Scoping Review" International Journal of Environmental Research and Public Health 19, no. 11: 6419. https://doi.org/10.3390/ijerph19116419
APA StylePagano, A. P., Ford, K. L., Porter Starr, K. N., Kiss, N., Steed, H., Kung, J. Y., Elango, R., & Prado, C. M. (2022). Energy Metabolism in Gynecological Cancers: A Scoping Review. International Journal of Environmental Research and Public Health, 19(11), 6419. https://doi.org/10.3390/ijerph19116419






