Acute Effects of Nicotine on Physiological Responses and Sport Performance in Healthy Baseball Players
Abstract
1. Introduction
2. Materials and Methods
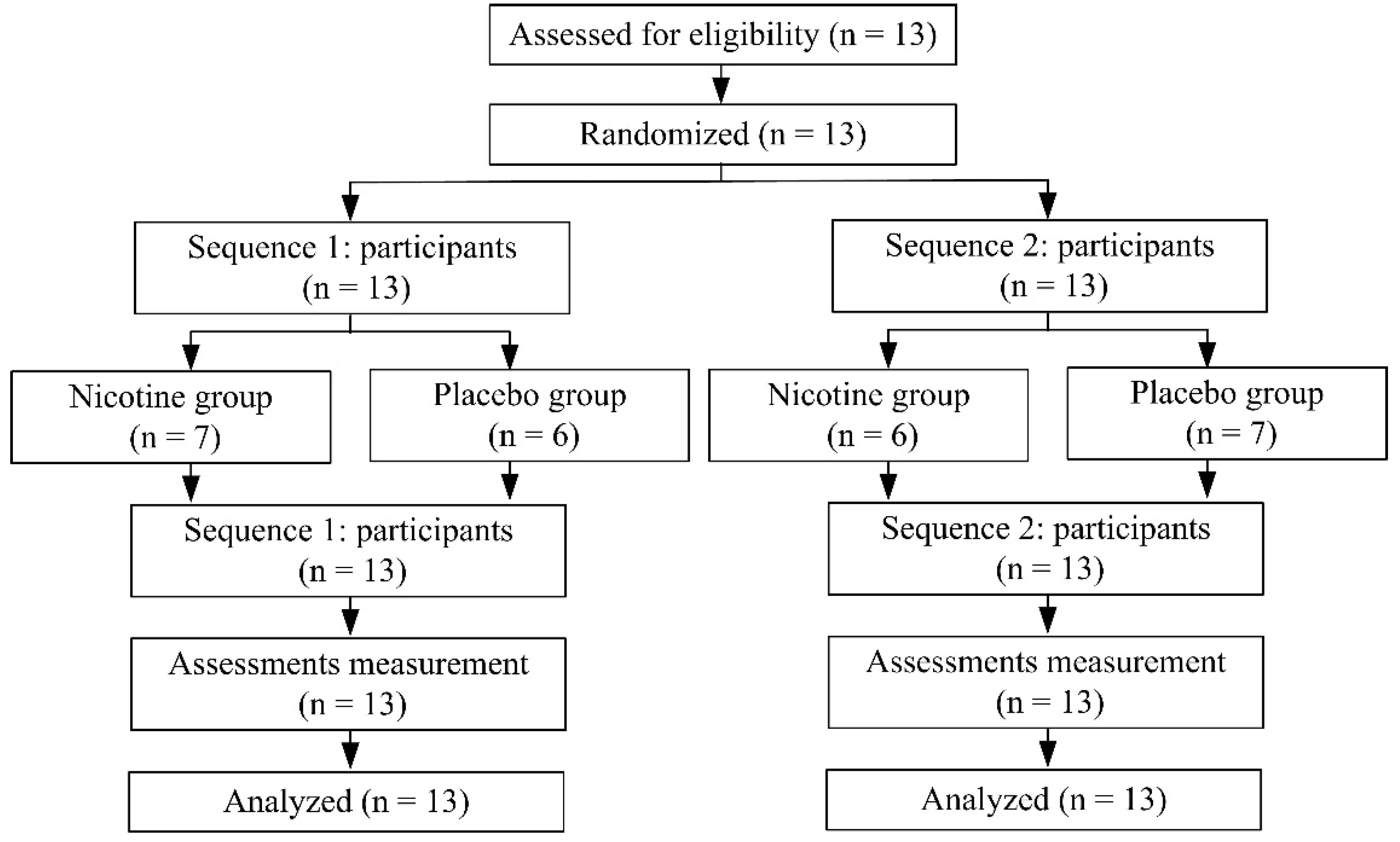
2.1. Participants
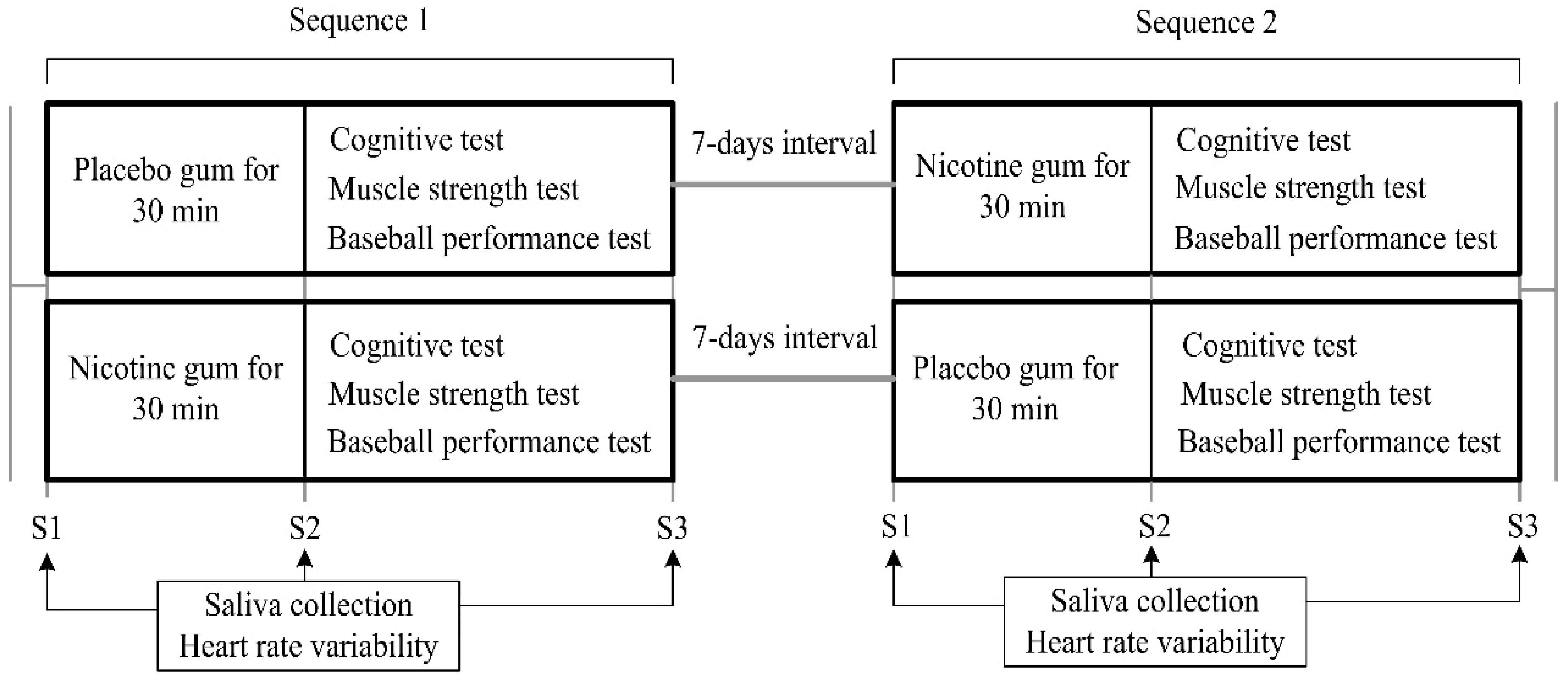
2.2. Experimental Protocol and Measures
2.3. Nicotine Intervention
2.4. Assessments
2.4.1. Heart Rate Variability Measurements
2.4.2. Saliva Collection and Assay
2.4.3. Cognitive Function Assessments
2.4.4. Muscle Strength Test
2.4.5. Baseball-Hitting Performance Test
2.5. Statistical Analyses
3. Results
3.1. Effects of Nicotine and Exercise on Heart Rate Variability (HRV) and Saliva α-Amylase Activity
3.2. Effects of Nicotine and Exercise on Salivary Testosterone and Cortisol
3.3. Effect of Nicotine on Cognitive Performance
3.4. Effect of Nicotine on Muscle Strength
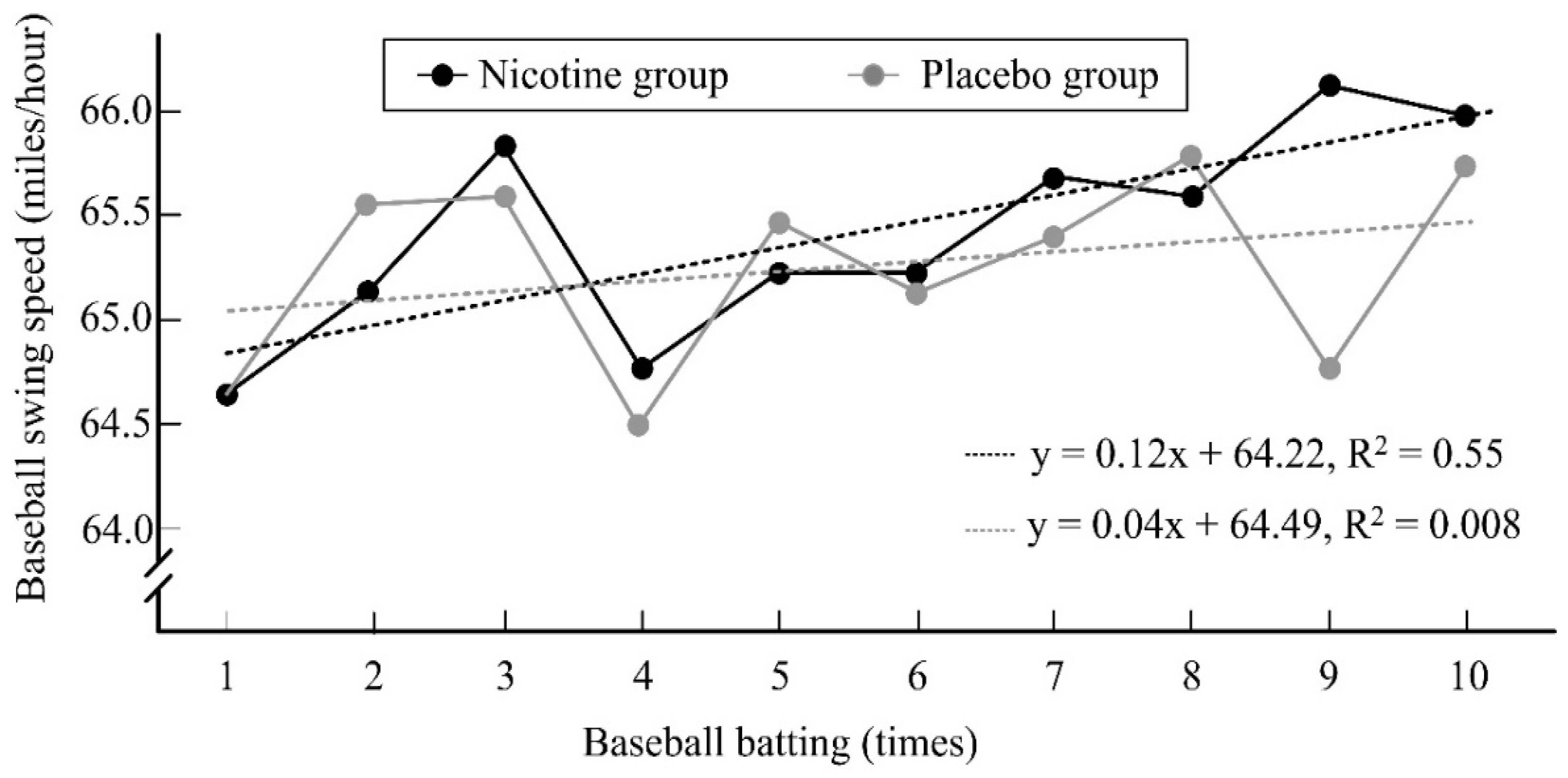
3.5. Effect of Nicotine on Baseball-Hitting Performance
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gil, S.M.; Metherate, R. Enhanced Sensory–Cognitive Processing by Activation of Nicotinic Acetylcholine Receptors. Nicotine Tob. Res. 2018, 21, 377–382. [Google Scholar] [CrossRef]
- Pickworth, W.B.; Fant, R.V. Endocrine effects of nicotine administration, tobacco and other drug withdrawal in humans. Psychoneuroendocrinology 1998, 23, 131–141. [Google Scholar] [CrossRef]
- Schumacher, S.; Kirschbaum, C.; Fydrich, T.; Ströhle, A. Is salivary alpha-amylase an indicator of autonomic nervous system dysregulations in mental disorders?—A review of preliminary findings and the interactions with cortisol. Psychoneuroendocrinology 2013, 38, 729–743. [Google Scholar] [CrossRef]
- Chagué, F.; Guenancia, C.; Gudjoncik, A.; Moreau, D.; Cottin, Y.; Zeller, M. Smokeless tobacco, sport and the heart. Arch. Cardiovasc. Dis. 2015, 108, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Mündel, T. Nicotine: Sporting Friend or Foe? A Review of Athlete Use, Performance Consequences and Other Considerations. Sports Med. 2017, 47, 2497–2506. [Google Scholar] [CrossRef]
- Johnston, R.; Doma, K.; Crowe, M. Nicotine effects on exercise performance and physiological responses in nicotine-naïve individuals: A systematic review. Clin. Physiol. Funct. Imaging 2017, 38, 527–538. [Google Scholar] [CrossRef]
- Zandonai, T.; Tam, E.; Bruseghini, P.; Pizzolato, F.; Franceschi, L.; Baraldo, M.; Capelli, C.; Cesari, P.; Chiamulera, C. The effects of oral smokeless tobacco administration on endurance performance. J. Sport Health Sci. 2018, 7, 465–472. [Google Scholar] [CrossRef]
- Mündel, T.; Machal, M.; Cochrane, D.J.; Barnes, M.J. A Randomised, Placebo-Controlled, Crossover Study Investigating the Effects of Nicotine Gum on Strength, Power and Anaerobic Performance in nicotine-naïve, Active Males. Sports Med. Open 2017, 3, 5. [Google Scholar] [CrossRef] [PubMed]
- Johnston, R.; Crowe, M.; Doma, K. Effect of nicotine on repeated bouts of anaerobic exercise in nicotine naïve individuals. Eur. J. Appl. Physiol. 2018, 118, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Zandonai, T.; Pizzolato, F.; Tam, E.; Bruseghini, P.; Chiamulera, C.; Cesari, P. The effects of nicotine on cortical excitability after exercise: A double-blind randomized, placebo-controlled, crossover Study. J. Clin. Psychopharmacol. 2020, 40, 495–498. [Google Scholar] [CrossRef]
- Šaranović, S.Đ.; Vićić, J.; Pešić, I.; Tomović, M.; Batinić, Đ.; Antić, M.; Tadic, M.; Mazić, S. The Influence of Tobacco Use on Pulmonary Function in Elite Athletes. Int. J. Environ. Res. Public Health 2019, 16, 3515. [Google Scholar] [CrossRef]
- Guo, X.; Wang, H.; Wu, X.; Chen, X.; Chen, Y.; Guo, J.; Li, X.; Lian, Q.; Ge, R.-S. Nicotine affects rat Leydig cell function in vivo and vitro via down-regulating some key steroidogenic enzyme expressions. Food Chem. Toxicol. 2017, 110, 13–24. [Google Scholar] [CrossRef]
- Peak, T.C.; Haney, N.M.; Wang, W.; DeLay, K.J.; Hellstrom, W.J. Stem cell therapy for the treatment of Leydig cell dysfunction in primary hypogonadism. World J. Stem Cells 2016, 8, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Hung, B.-L.; Chen, L.-J.; Chen, Y.-Y.; Ou, J.-B.; Fang, S.-H. Nicotine supplementation enhances simulated game performance of archery athletes. J. Int. Soc. Sports Nutr. 2021, 18, 16. [Google Scholar] [CrossRef] [PubMed]
- Masumoto, Y.; Morinushi, T.; Kawasaki, H.; Takigawa, M. Spectral analysis of changes in electroencephalographic activity after the chewing of gum. Psychiatry Clin. Neurosci. 1998, 52, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N. Tobacco Use and Oral Cancer: A Global Perspective. J. Dent. Educ. 2001, 65, 328–339. [Google Scholar] [CrossRef] [PubMed]
- Connolly, G.N.; Orleans, C.T.; Kogan, M. Use of Smokeless Tobacco in Major-League Baseball. N. Engl. J. Med. 1988, 318, 1281–1285. [Google Scholar] [CrossRef] [PubMed]
- Conrad, A.K.; Hutton, S.B.; Munnelly, M.; Bay, R.C. Screening for smokeless tobacco use and presence of oral lesions in major league baseball athletes. J. Calif. Dent. Assoc. 2015, 43, 14–20. [Google Scholar]
- Silvette, H.; Hoff, E.C.; Larson, P.S.; Haag, H.B. The actions of nicotine on central nervous system functions. Pharmacol. Rev. 1962, 14, 137–173. [Google Scholar]
- Li, C.Y.; Hsu, G.S.; Suzuki, K.; Ko, M.H.; Fang, S.H. Salivary immuno factors, cortisol and testosterone responses in athletes of a competitive 5000 m race. Chin. J. Physiol. 2015, 58, 263–269. [Google Scholar] [CrossRef]
- Stubbs, B.; Ku, P.-W.; Chung, M.-S.; Chen, L.-J. Relationship Between Objectively Measured Sedentary Behavior and Cognitive Performance in Patients with Schizophrenia Vs Controls. Schizophr. Bull. 2016, 43, 566–574. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Yun, A.J.; Bazar, K.A.; Lee, P.Y.; Gerber, A.; Daniel, S.M. The smoking gun: Many conditions associated with tobacco expo-sure may be attributable to paradoxical compensatory autonomic responses to nicotine. Med. Hypotheses 2005, 64, 1073–1079. [Google Scholar] [CrossRef]
- Fernández, R.; Larraín, C.; Zapata, P. Acute ventilatory and circulatory reactions evoked by nicotine: Are they excitatory or depressant? Respir. Physiol. Neurobiol. 2002, 133, 173–182. [Google Scholar] [CrossRef]
- Druyan, A.; Atias, D.; Ketko, I.; Cohen-Sivan, Y.; Heled, Y. The effects of smoking and nicotine ingestion on exercise heat tolerance. J. Basic Clin. Physiol. Pharmacol. 2017, 28, 167–170. [Google Scholar] [CrossRef]
- Tracey, K.J. The inflammatory reflex. Nature 2002, 420, 853–859. [Google Scholar] [CrossRef] [PubMed]
- Koibuchi, E.; Suzuki, Y. Exercise upregulates salivary amylase in humans. Exp. Ther. Med. 2014, 7, 773–777. [Google Scholar] [CrossRef]
- Tasaka, A.; Takeuchi, K.; Sasaki, H.; Yoshii, T.; Soeda, R.; Ueda, T.; Sakurai, K. Influence of chewing time on salivary stress markers. J. Prosthodont. Res. 2014, 58, 48–54. [Google Scholar] [CrossRef]
- Rohleder, N.; Wolf, J.M.; Maldonado, E.F.; Kirschbaum, C. The psychosocial stress-induced increase in salivary al-pha-amylase is independent of saliva flow rate. Psychophysiology 2006, 43, 645–652. [Google Scholar] [CrossRef] [PubMed]
- Takai, N.; Yamaguchi, M.; Aragaki, T.; Eto, K.; Uchihashi, K.; Nishikawa, Y. Effect of psychological stress on the salivary cortisol and amylase levels in healthy young adults. Arch. Oral Biol. 2004, 49, 963–968. [Google Scholar] [CrossRef]
- Cohen, L.M.; Britt, D.M.; Collins, F.L.; Al’Absi, M.; McChargue, D.E. Multimodal assessment of the effect of chewing gum on nicotine withdrawal. Addict. Behav. 2001, 26, 289–295. [Google Scholar] [CrossRef][Green Version]
- Istvan, J.A.; Buist, A.S.; Hess, D.L.; Voelker, H. Relationship of smoking cessation and nicotine gum use to salivary andros-tenedione and testosterone in middle-aged men. Metabolism 1995, 44, 90–95. [Google Scholar] [CrossRef]
- Dai, W.S.; Gutai, J.P.; Kuller, L.H.; Cauley, J.A. Cigarette smoking and serum sex hormones in men. Am. J. Epidemiol. 1988, 128, 796–805. [Google Scholar] [CrossRef]
- Meikle, A.W.; Bishop, D.T.; Stringham, J.D.; Ford, M.H.; West, D.W.; Rao, D.C. Relationship between body mass index, cigarette smoking, and plasma sex steroids in normal male twins. Genet. Epidemiol. 1989, 6, 399–412. [Google Scholar] [CrossRef]
- Zhao, X.; Xu, W.; Wu, J.; Zhang, D.; Abou-Shakra, A.; Di, L.; Wang, Z.; Wang, L.; Yang, F.; Qiao, Z. Nicotine induced autoph-agy of Leydig cells rather than apoptosis is the major reason of the decrease of serum testosterone. Int. J. Biochem. Cell Biol. 2018, 100, 30–41. [Google Scholar] [CrossRef]
- Wu, Y.; Wu, Y.; Deng, J.; Chu, L.; Yang, H.; Wang, W.; Liao, J.; Cheng, Y.; Deng, H. Screening and identification of salivary biomarkers for assessing the effects of exogenous testosterone administration on HPG and HPA axes and ECS. Steroids 2020, 158, 108604. [Google Scholar] [CrossRef] [PubMed]
- Hartmann-Boyce, J.; Chepkin, S.C.; Ye, W.; Bullen, C.; Lancaster, T. Nicotine replacement therapy versus control for smoking cessation. Cochrane Database Syst. Rev. 2018, 5, CD000146. [Google Scholar] [CrossRef]
- Hahn, B.; Shrieves, M.E.; Olmstead, C.K.; Yuille, M.B.; Chiappelli, J.J.; Pereira, E.F.R.; Albuquerque, E.X.; Fawcett, W.P. Evidence for positive allosteric modulation of cognitive-enhancing effects of nicotine in healthy human subjects. Psychopharmacology 2020, 237, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Heishman, S.J.; Kleykamp, B.A.; Singleton, E.G. Meta-analysis of the acute effects of nicotine and smoking on human performance. Psychopharmacology 2010, 210, 453–469. [Google Scholar] [CrossRef]
- Philpot, R.M. Potential Use of Nicotinic Receptor Agonists for the Treatment of Chemotherapy-Induced Cognitive Deficits. Neurochem. Res. 2015, 40, 2018–2031. [Google Scholar] [CrossRef] [PubMed]
- Newhouse, P.A.; Potter, A.; Singh, A. Effects of nicotinic stimulation on cognitive performance. Curr. Opin. Pharmacol. 2004, 4, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Hahn, B. Nicotinic receptors and attention. Curr. Top. Behav. Neurosci. 2015, 23, 103–135. [Google Scholar] [PubMed]
- Ettinger, U.; Faiola, E.; Kasparbauer, A.-M.; Petrovsky, N.; Chan, R.C.K.; Liepelt, R.; Kumari, V. Effects of nicotine on response inhibition and interference control. Psychopharmacology 2017, 234, 1093–1111. [Google Scholar] [CrossRef]
- Escher, S.A.; Tucker, A.M.; Lundin, T.M.; Grabiner, M.D. Smokeless tobacco, reaction time, and strength in athletes. Med. Sci. Sports Exerc. 1998, 30, 1548–1551. [Google Scholar] [CrossRef] [PubMed]
- Siafaka, A.; Angelopoulos, E.; Kritikos, K.; Poriazi, M.; Basios, N.; Gerovasili, V.; Andreou, A.; Roussos, C.; Nanas, S. Acute Effects of Smoking on Skeletal Muscle Microcirculation Monitored by Near-Infrared Spectroscopy. Chest 2007, 131, 1479–1485. [Google Scholar] [CrossRef]
- Mündel, T.; Jones, D.A. Effect of transdermal nicotine administration on exercise endurance. Exp. Physiol. 2006, 91, 705–713. [Google Scholar] [CrossRef]
- Pysný, L.; Petru, D.; Pysna, J.; Cihlar, D. The acute effect of nicotine on anaerobic exercise performance. J. Phys. Educ. Sport 2015, 15, 103–107. [Google Scholar]
- Poltavski, D.V.; Petros, T.V.; Holm, J.E. Lower but not higher doses of transdermal nicotine facilitate cognitive performance in smokers on gender non-preferred tasks. Pharmacol. Biochem. Behav. 2012, 102, 423–433. [Google Scholar] [CrossRef] [PubMed]
- England, L.J.; Bunnell, R.E.; Pechacek, T.F.; Tong, V.T.; McAfee, T.A. Nicotine and the developing human: A neglected ele-ment in the electronic cigarette debate. Am. J. Prev. Med. 2015, 49, 286–293. [Google Scholar] [CrossRef]
- Henley, S.J.; Asman, K.; Momin, B.; Gallaway, M.S.; Culp, M.B.; Ragan, K.R.; Richards, T.B.; Babb, S. Smoking cessation be-haviors among older U.S. adults. Prev. Med. Rep. 2019, 16, 100978. [Google Scholar] [CrossRef]
- Gallaway, M.S.; Henley, S.J.; Steele, C.B.; Momin, B.; Thomas, C.C.; Jamal, A.; Trivers, K.F.; Singh, S.D.; Stewart, S.L. Surveil-lance for cancers associated with tobacco use—United States, 2010–2014. MMWR Surveill. Summ. 2018, 67, 1–42. [Google Scholar] [CrossRef]
- Bruijnzeel, A.W. Tobacco addiction and the dysregulation of brain stress systems. Neurosci. Biobehav. Rev. 2012, 36, 1418–1441. [Google Scholar] [CrossRef] [PubMed]
- Sachs, D.P. Nicotine polacrilex: Practical use requirements. Curr. Pulmonol. 1989, 10, 141–158. [Google Scholar]
| Participants (n = 13) | |
|---|---|
| Gender (male), n (%) | 13 (100%) |
| Age (years) | 20.69 ± 0.75 |
| Height (m) | 174.46 ± 3.89 |
| Weight (kg) | 78.12 ± 10.34 |
| Body mass index (kg/m2) | 25.61 ± 2.77 |
| Baseball age (years) | 11.31 ± 1.38 |
| Nicotine Group | Placebo Group | Condition Effect | Time Effect | Condition * Time | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| S1 | S2 | S3 | S1 | S2 | S3 | p | ηp2 | p | ηp2 | p | ηp2 | |
| Heart rate variability | ||||||||||||
| HR (bpm) | 74.11 ± 10.88 | 80.77 ± 10.12 ***, # | 93.95 ± 11.49 *** | 76.69 ± 7.21 | 74.55 ± 7.56 | 92.49 ± 12.26 *** | 0.35 | 0.04 | 0.001 | 0.73 | 0.04 | 0.15 |
| pNN50 (%) | 23.33 ± 22.71 | 17.28 ± 17.37 | 5.27 ± 4.03 ** | 17.36 ± 16.90 | 20.64 ± 15.12 | 12.54 ± 18.44 | 0.54 | 0.01 | 0.01 | 0.22 | 0.007 | 0.20 |
| LF (n.u.) | 60.74 ± 13.00 | 64.81 ± 16.17 | 73.58 ± 15.57 ** | 61.79 ± 12.59 | 65.10 ± 10.96 | 69.11 ± 17.86 | 0.77 | 0.004 | 0.002 | 0.24 | 0.69 | 0.01 |
| HF (n.u.) | 39.09 ± 12.88 | 35.04 ± 16.05 | 26.21 ± 15.37 ** | 38.03 ± 12.56 | 34.79 ± 10.95 | 30.68 ± 17.64 | 0.76 | 0.004 | 0.006 | 0.24 | 0.68 | 0.01 |
| LF/HF | 1.84 ± 0.95 | 2.67 ± 2.21 * | 4.63 ± 3.88 ** | 1.97 ± 1.17 | 2.16 ± 1.05 | 4.70 ± 5.94 * | 0.98 | 0.001 | 0.006 | 0.27 | 0.54 | 0.02 |
| α-amylase (U/mL) | 129.59 ± 69.17 | 158.03 ± 85.58 *, # | 170.62 ± 99.11 *, # | 139.33 ± 94.31 | 122.30 ± 78.46 | 143.60 ± 103.17 | 0.08 | 0.13 | 0.05 | 0.13 | 0.009 | 0.20 |
| Testosterone(nmole/L) | 0.78 ± 0.31 | 0.53 ± 0.30 **, # | 0.79 ± 0.43 | 0.86 ± 0.36 | 0.81 ± 0.35 | 0.81 ± 0.40 | 0.10 | 0.11 | 0.11 | 0.09 | 0.10 | 0.09 |
| Cortisol (nmole/L) | 3.48 ± 2.10 | 3.54 ± 1.75 # | 2.33 ± 1.22 ** | 3.53 ± 1.58 | 2.59 ± 0.99 ** | 2.37 ± 1.22 ** | 0.37 | 0.03 | 0.001 | 0.34 | 0.02 | 0.14 |
| T/C ratio | 0.30 ± 0.21 | 0.18 ± 0.13 *, ## | 0.52 ± 0.50 ** | 0.27 ± 0.11 | 0.34 ± 0.17 | 0.43 ± 0.29 * | 0.83 | 0.002 | 0.004 | 0.27 | 0.02 | 0.16 |
| Nicotine Group | Placebo Group | d | p | |
|---|---|---|---|---|
| Reaction time | ||||
| Reaction (msec) | 267.00 ± 49.89 | 270.70 ± 46.07 | −0.07 | 0.53 |
| Motor (msec) | 134.80 ± 33.78 | 151.70 ± 38.04 # | −0.47 | 0.03 |
| Cognitrone | ||||
| Correct rejection (msec) | 0.70 ± 0.05 | 0.73 ± 0.06 | −0.54 | 0.19 |
| Total of correct rejections | 44.20 ± 3.19 | 44.70 ± 3.80 | −0.14 | 0.63 |
| GPT (sec) | 121.30 ± 15.03 | 128.66 ± 15.81 # | −0.47 | 0.04 |
| Nicotine Group | Placebo Group | d a | p a | d b | p b | |||
|---|---|---|---|---|---|---|---|---|
| Left | Right | Left | Right | |||||
| Absolute strength (Ib) | ||||||||
| Deltoid | 62.17 ± 8.76 | 61.53 ± 8.61 | 59.20 ± 8.45 | 63.24 ± 7.82 | 0.34 | 0.24 | −0.20 | 0.09 |
| Biceps brachii | 73.39 ± 13.62 | 68.80 ± 13.47 | 73.22 ± 13.60 | 67.39 ± 12.33 | 0.01 | 0.30 | 0.10 | 0.94 |
| Triceps brachii | 48.07 ± 7.87 | 50.52 ± 9.39 | 49.70 ± 6.20 | 51.98 ± 9.58 | −0.23 | 0.47 | −0.15 | 0.32 |
| Wrist flexor | 50.30 ± 11.81 | 48.03 ± 9.04 | 46.60 ± 9.38 | 49.87 ± 8.50 | 0.34 | 0.48 | −0.21 | 0.20 |
| Wrist extensor | 67.59 ± 12.20 | 64.79 ± 13.83 | 67.11 ± 9.66 | 67.78 ± 10.31 | 0.04 | 0.31 | −0.24 | 0.82 |
| Relative strength (Ib/kg) | ||||||||
| Deltoid | 0.80 ± 0.11 | 0.79 ± 0.10 | 0.77 ± 0.12 | 0.81 ± 0.09 | 0.26 | 0.23 | −0.21 | 0.07 |
| Biceps brachii | 0.94 ± 0.12 | 0.88 ± 0.12 | 0.94 ± 0.14 | 0.86 ± 0.12 | 0.01 | 0.32 | 0.16 | 0.93 |
| Triceps brachii | 0.62 ± 0.11 | 0.65 ± 0.09 | 0.64 ± 0.10 | 0.67 ± 0.14 | −0.19 | 0.30 | −0.16 | 0.35 |
| Wrist flexor | 0.65 ± 0.15 | 0.62 ± 0.11 | 0.60 ± 0.13 | 0.65 ± 0.15 | 0.35 | 0.34 | −0.22 | 0.22 |
| Wrist extensor | 0.87 ± 0.14 | 0.83 ± 0.13 | 0.87 ± 0.13 | 0.87 ± 0.12 | 0.01 | 0.24 | −0.31 | 0.88 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fang, S.-H.; Lu, C.-C.; Lin, H.-W.; Kuo, K.-C.; Sun, C.-Y.; Chen, Y.-Y.; Chang, W.-D. Acute Effects of Nicotine on Physiological Responses and Sport Performance in Healthy Baseball Players. Int. J. Environ. Res. Public Health 2022, 19, 515. https://doi.org/10.3390/ijerph19010515
Fang S-H, Lu C-C, Lin H-W, Kuo K-C, Sun C-Y, Chen Y-Y, Chang W-D. Acute Effects of Nicotine on Physiological Responses and Sport Performance in Healthy Baseball Players. International Journal of Environmental Research and Public Health. 2022; 19(1):515. https://doi.org/10.3390/ijerph19010515
Chicago/Turabian StyleFang, Shih-Hua, Chi-Cheng Lu, Hua-Wei Lin, Kuan-Chen Kuo, Chen-Yu Sun, Yi-Ying Chen, and Wen-Dien Chang. 2022. "Acute Effects of Nicotine on Physiological Responses and Sport Performance in Healthy Baseball Players" International Journal of Environmental Research and Public Health 19, no. 1: 515. https://doi.org/10.3390/ijerph19010515
APA StyleFang, S.-H., Lu, C.-C., Lin, H.-W., Kuo, K.-C., Sun, C.-Y., Chen, Y.-Y., & Chang, W.-D. (2022). Acute Effects of Nicotine on Physiological Responses and Sport Performance in Healthy Baseball Players. International Journal of Environmental Research and Public Health, 19(1), 515. https://doi.org/10.3390/ijerph19010515








