Prophylactic Dose of Oxytocin for Uterine Atony during Caesarean Delivery: A Systematic Review
Abstract
1. Introduction
2. Methods
3. Results
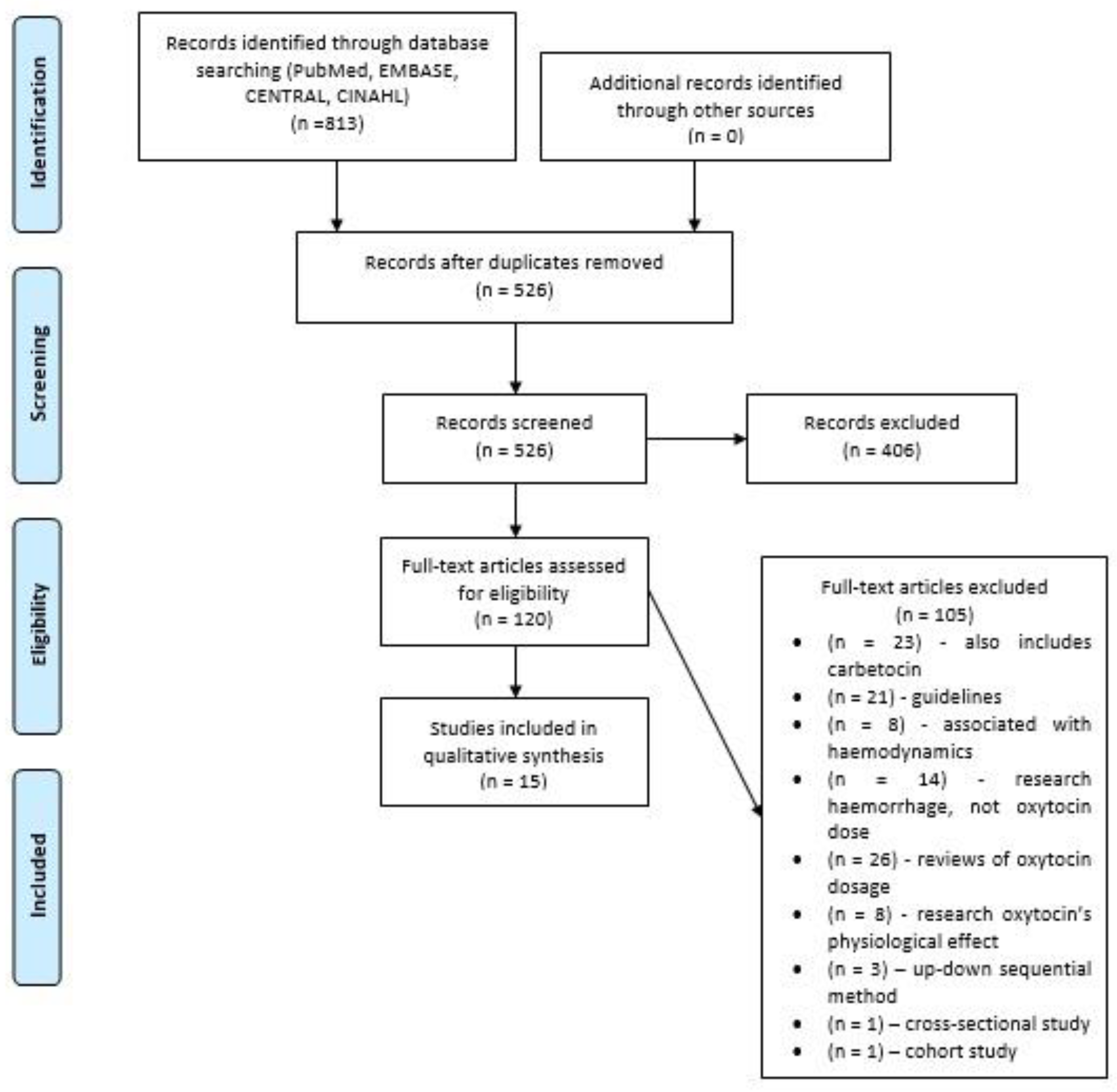
3.1. Study Selection Process
3.2. Characteristics of Included Studies
3.3. Synthesis of Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- American College of Obstetricians and Gynecologists. Obstetric Data Definitions (Version 1.0). Revitalize 2014. Available online: https://www.acog.org/-/media/project/acog/acogorg/files/pdfs/publications/revitalize-ob.pdf?la=en&hash=D9ECE1577C2A0B535F42C1B8DC492070 (accessed on 30 March 2021).
- Prevention and Management of Postpartum Haemorrhage: Green-Top Guideline No. 52. Bjog Int. J. Obstet. Gynaecol. 2017, 124, e106–e149. [CrossRef]
- Niino, Y. The Increasing Cesarean Rate Globally and What We Can Do about It. Biosci. Trends 2011, 5, 139–150. [Google Scholar] [CrossRef]
- Kambo, I.; Bedi, N.; Dhillon, B.S.; Saxena, N.C. A Critical Appraisal of Cesarean Section Rates at Teaching Hospitals in India. Int. J. Gynecol. Obstet. 2002, 79, 151–158. [Google Scholar] [CrossRef]
- Othman, E.R.; Fayez, M.F.; el Aal, D.E.M.A.; El-Dine Mohamed, H.S.; Abbas, A.M.; Ali, M.K. Sublingual Misoprostol versus Intravenous Oxytocin in Reducing Bleeding during and after Cesarean Delivery: A Randomized Clinical Trial. Taiwan. J. Obstet. Gynecol. 2016, 55, 791–795. [Google Scholar] [CrossRef]
- Evensen, A.; Anderson, J.M. Postpartum Hemorrhage: Prevention and Treatment. Am. Fam. Physician 2017, 95, 442–449. [Google Scholar]
- Feduniw, S.; Warzecha, D.; Szymusik, I.; Wielgos, M. Epidemiology, Prevention and Management of Early Postpartum Hemorrhage—A Systematic Review. Ginekol. Pol. 2020, 91, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Butwick, A.J.; Coleman, L.; Cohen, S.E.; Riley, E.T.; Carvalho, B. Minimum Effective Bolus Dose of Oxytocin during Elective Caesarean Delivery. Br. J. Anaesth. 2010, 104, 338–343. [Google Scholar] [CrossRef]
- Sartain, J.B.; Barry, J.J.; Howat, P.W.; McCormack, D.I.; Bryant, M. Intravenous Oxytocin Bolus of 2 Units Is Superior to 5 Units during Elective Caesarean Section. Br. J. Anaesth. 2008, 101, 822–826. [Google Scholar] [CrossRef] [PubMed]
- Somjit, M.; Surojananon, J.; Kongwattanakul, K.; Kasemsiri, C.; Sirisom, M.; Prawannoa, K.; Thepsuthammarat, K.; Komwilaisak, R. Comparison of Low Dose versus High Dose of Oxytocin for Initiating Uterine Contraction during Cesarean Delivery: A Randomized, Controlled, Non-Inferiority Trial. Int. J. Women’s Health 2020, 12, 667–673. [Google Scholar] [CrossRef] [PubMed]
- King, K.J.; Douglas, M.J.; Unger, W.; Wong, A.; King, R.A.R. Five Unit Bolus Oxytocin at Cesarean Delivery in Women at Risk of Atony: A Randomized, Double-Blind, Controlled Trial. Anesth. Analg. 2010, 111, 1460–1466. [Google Scholar] [CrossRef]
- Jonsson, M.; Hanson, U.; Lidell, C.; Nordén-Lindeberg, S. ST Depression at Caesarean Section and the Relation to Oxytocin Dose. A Randomised Controlled Trial. Bjog Int. J. Obstet. Gynaecol. 2010, 117, 76–83. [Google Scholar] [CrossRef]
- Kovacheva, V.P.; Soens, M.A.; Tsen, L.C. A Randomized, Double-Blinded Trial of a “Rule of Threes” Algorithm versus Continuous Infusion of Oxytocin during Elective Cesarean Delivery. Anesthesiology 2015, 123, 92–100. [Google Scholar] [CrossRef]
- Cecilia, M.; Vijayaselvi, R.; Bansal, R.; Lakshmi, L.; Jose, R. Ten Units Intravenous Oxytocin over 2–4 h Is as Effective as 30 Units over 8–12 h in Preventing Postpartum Hemorrhage after Cesarean Section: A Randomized Controlled Trial. Indian J. Pharm. 2018, 50, 279–283. [Google Scholar]
- Ghulmiyyah, L.M.; Usta, I.M.; Ghazeeri, G.; Taher, N.; Abu-Ghannam, G.; Tamim, H.; Nassar, A.H. Intravenous Oxytocin Use to Decrease Blood Loss during Scheduled Cesarean Delivery: A Randomized Double-Blinded Controlled Trial (OXYTRIAL). Am. J. Perinatol. 2017, 34, 379–387. [Google Scholar] [CrossRef]
- Duffield, A.; McKenzie, C.; Carvalho, B.; Ramachandran, B.; Yin, V.; El-Sayed, Y.Y.; Riley, E.T.; Butwick, A.J. Effect of a High-Rate Versus a Low-Rate Oxytocin Infusion for Maintaining Uterine Contractility during Elective Cesarean Delivery: A Prospective Randomized Clinical Trial. Anesth. Analg. 2017, 124, 857–862. [Google Scholar] [CrossRef]
- GÜngÖrdÜk, K.; Asicioglu, O.; Celikkol, O.; Olgac, Y.; Ark, C. Use of Additional Oxytocin to Reduce Blood Loss at Elective Caesarean Section A Randomised Control Trial. Aust. N. Zealand J. Obstet. Gynaecol. 2010, 50, 36–39. [Google Scholar] [CrossRef] [PubMed]
- Kajendran, J.; Silva, G.R.C.; Ranaraja, S.K. Effects of 5 IU Oxytocin Bolus and 20 IU Oxytocin Infusion Compared to 5 IU Oxytocin Bolus and Normal Saline Infusion in the Control of Blood Loss during and after Ante-Partum Lower Segment Caesarean Section: A Randomized Controlled Trial. Ceylon Med. J. 2017, 62, 121. [Google Scholar] [CrossRef]
- McLeod, G.; Munishankar, B.; MacGregor, H.; Murphy, D.J. Maternal Haemodynamics at Elective Caesarean Section: A Randomised Comparison of Oxytocin 5-Unit Bolus and Placebo Infusion with Oxytocin 5-Unit Bolus and 30-Unit Infusion. Int. J. Obstet. Anesth. 2010, 19, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Murphy, D.J.; MacGregor, H.; Munishankar, B.; McLeod, G. A Randomised Controlled Trial of Oxytocin 5IU and Placebo Infusion versus Oxytocin 5IU and 30IU Infusion for the Control of Blood Loss at Elective Caesarean Section-Pilot Study. ISRCTN 40302163. Eur. J. Obstet. Gynecol. Reprod. Biol. 2009, 142, 30–33. [Google Scholar] [CrossRef]
- Sheehan, S.R.; Montgomery, A.A.; Carey, M.; McAuliffe, F.M.; Gleeson, R.; Geary, M.; Murphy, D.J. Oxytocin Bolus versus Oxytocin Bolus and Infusion for Control of Blood Loss at Elective Caesarean Section: Double Blind, Placebo Controlled, Randomised Trial. BMJ 2011, 343, 46–61. [Google Scholar] [CrossRef] [PubMed]
- Qian, X.W.; Drzymalski, D.M.; Lv, C.C.; Guo, F.H.; Wang, L.Y.; Chen, X.Z. The ED50 and ED95 of Oxytocin Infusion Rate for Maintaining Uterine Tone during Elective Caesarean Delivery: A Dose-Finding Study. BMC Pregnancy Childbirth 2020, 20, 6. [Google Scholar] [CrossRef] [PubMed]
- Gallos, I.D.; Williams, H.M.; Price, M.J.; Merriel, A.; Gee, H.; Lissauer, D.; Moorthy, V.; Tobias, A.; Deeks, J.J.; Widmer, M.; et al. Uterotonic agents for preventing postpartum haemorrhage: A network meta-analysis. Cochrane Database Syst. Rev. 2018, 12. [Google Scholar] [CrossRef]
- World Health Organization. WHO recommendations: Uterotonics for the prevention of postpartum haemorrhage. In WHO Recommendations: Uterotonics for the Prevention of Postpartum Haemorrhage; World Health Organization: Geneva, Switzerland, 2018; pp. 1–53. [Google Scholar]
- Thorneloe, B.; Carvalho, J.C.A.; Downey, K.; Balki, M. Uterotonic Drug Usage in Canada: A Snapshot of the Practice in Obstetric Units of University-Affiliated Hospitals. Int. J. Obstet. Anesth. 2019, 37, 45–51. [Google Scholar] [CrossRef]
- Leduc, D.; Senikas, V.; Lalonde, A.B.; Ballerman, C.; Biringer, A.; Delaney, M.; Duperron, L.; Girard, I.; Jones, D.; Lee, L.S.Y.; et al. Active Management of the Third Stage of Labour: Prevention and Treatment of Postpartum Hemorrhage. J. Obstet. Gynaecol. Can. 2009, 31, 980–993. [Google Scholar] [CrossRef]
- American College of Obstetricians and Gynecologists. Postpartum Hemorrhage. Obs. Gynecol. 2017, 130, e168–e186. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, M.; Stensballe, J.; Ducloy-Bouthors, A.S.; Bonnet, M.P.; de Robertis, E.; Fornet, I.; Goffinet, F.; Hofer, S.; Holzgreve, W.; Manrique, S.; et al. Patient Blood Management in Obstetrics: Prevention and Treatment of Postpartum Haemorrhage. A NATA Consensus Statement: A Multidisciplinary Consensus Statement. Blood Transfus. 2019, 17, 112–136. [Google Scholar] [CrossRef]
- Sentilhes, L.; Vayssière, C.; Deneux-Tharaux, C.; Aya, A.G.; Bayoumeu, F.; Bonnet, M.P.; Djoudi, R.; Dolley, P.; Dreyfus, M.; Ducroux-Schouwey, C.; et al. Postpartum Hemorrhage: Guidelines for Clinical Practice from the French College of Gynaecologists and Obstetricians (CNGOF): In Collaboration with the French Society of Anesthesiology and Intensive Care (SFAR). Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 198, 12–21. [Google Scholar] [CrossRef]
- Carvalho, J.C.A.; Balki, M.; Kingdom, J.; Windrim, R. Oxytocin Requirements at Elective Cesarean Delivery: A Dose-Finding Study. Am. Coll. Obstet. Gynecol. 2004, 104, 1005–1010. [Google Scholar] [CrossRef]
- Balki, M.; Ronayne, M.; Davies, S.; Fallah, S.; Kingdom, J.; Windrim, R.; Carvalho, J. Minimum Oxytocin Dose Requirement After Cesarean Delivery for Labor Arrest. Obstet. Gynecol. 2006, 107, 45–50. [Google Scholar] [CrossRef]
- Beiranvand, S.; Karimi, A.; Vahabi, S.; Amin-Bidokhti, A. Comparison of the Mean Minimum Dose of Bolus Oxytocin for Proper Uterine Contraction during Cesarean Section. Curr. Clin. Pharmacol. 2019, 14, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Svanström, M.C.; Biber, B.; Hanes, M.; Johansson, G.; Näslund, U.; Bålfors, E.M. Signs of Myocardial Ischaemia after Injection of Oxytocin: A Randomized Double-Blind Comparison of Oxytocin and Methylergometrine during Caesarean Section. Br. J. Anaesth. 2008, 100, 683–689. [Google Scholar] [CrossRef]
- Robinson, C.; Schumann, R.; Zhang, P.; Young, R.C. Oxytocin-Induced Desensitization of the Oxytocin Receptor. Am. J. Obstet. Gynecol. 2003, 188, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Heesen, M.; Carvalho, B.; Carvalho, J.C.A.; Duvekot, J.J.; Dyer, R.A.; Lucas, D.N.; McDonnell, N.; Orbach-Zinger, S.; Kinsella, S.M. International Consensus Statement on the Use of Uterotonic Agents during Caesarean Section. Anaesthesia 2019, 74, 1305–1319. [Google Scholar] [CrossRef] [PubMed]
- Japundžić-Žigon, N.; Lozić, M.; Šarenac, O.; Murphy, D. Vasopressin & Oxytocin in Control of the Cardiovascular System: An Updated Review. Curr. Neuropharmacol. 2019, 18, 14–33. [Google Scholar] [CrossRef]
- Thomas, J.S.; Koh, S.H.; Cooper, G.M. Haemodynamic Effects of Oxytocin given as i.v. Bolus or Infusion on Women Undergoing Caesarean Section. Br. J. Anaesth. 2007, 98, 116–119. [Google Scholar] [CrossRef]
- Langesæter, E.; Rosseland, L.A.; Stubhaug, A. Hemodynamic Effects of Oxytocin during Cesarean Delivery. Int. J. Gynecol. Obstet. 2006, 95, 46–47. [Google Scholar] [CrossRef]
- Langesæter, E.; Rosseland, L.A.; Stubhaug, A. Haemodynamic Effects of Oxytocin in Women with Severe Preeclampsia. Int. J. Obstet. Anesth. 2011, 20, 26–29. [Google Scholar] [CrossRef]
- Archer, T.L.; Knape, K.; Liles, D.; Wheeler, A.S.; Carter, B. The Hemodynamics of Oxytocin and Other Vasoactive Agents during Neuraxial Anesthesia for Cesarean Delivery: Findings in Six Cases. Int. J. Obstet. Anesth. 2008, 17. [Google Scholar] [CrossRef]
| Primary Author | Country, Year of Publishing | Trial Type | No. of Patients | Type of Anaesthesia | Inclusion Criteria | Exclusion Criteria | Elective CD |
|---|---|---|---|---|---|---|---|
| Butwick [8] | USA, 2010 | Double-blind, randomized | 75 | Spinal anesthesia | ASA I or II, age between 18 and 40 yr, singleton pregnancies, and elective CD with a pfannensteil incision, spinal anesthesia | Active labor, ruptured membranes, known drug allergy to oxytocin, multiple gestation, significant obstetric disease, known risk factors for postpartum hemorrhage, inherited or acquired coagulation disorder and thrombocytopenia | Yes |
| Sartain [9] | Australia, 2008 | Double-blind, randomized | 80 | CSE | Elective caesarean section under regional anesthesia | Patients at increased risk of uterine atony or excessive bleeding (more than two previous Caesarean sections, a history of previous post-partum hemorrhage, known placenta praevia or accreta, twin pregnancy, and polyhydramnios) or cardiovascular instability (pre-eclampsia or essential hypertension) | Yes |
| Somjit [10] | Thailand, 2020 | Double-blind, randomized | 155 | Spinal anesthesia | Singleton pregnancy, age 18–40 years, 37–41 completed weeks of gestational age, ASA class II and scheduled caesarean delivery under spinal anesthesia | Spinal anesthesia had failed or was inadequate, previous uterine surgery other than caesarean section, high risk of uterine atony (macrosomia, chorioamnionitis, polyhydramnion,, uterine mass) or postpartum hemorrhage (placenta praevia or other placenta disorders, history of postpartum hemorrhage, coagulopathy, thrombocytopenia, or pre-eclampsia), or known allergies to oxytocin | Yes |
| King [11] | Canada, 2010 | Double-blind, randomized | 143 | Epidural/spinal/CSE/GA/neuraxial + GA | Patients scheduled for elective and or emergency caesarean delivery at a time when an investigator was available were approached | Cardiac disease, hemodynamic instability before commencement of surgery, bleeding disorders, or younger than 19 years, or could not understand or read English | Yes/No |
| Jonsson [12] | Sweden, 2009 | Double-blind, randomized | 103 | Spinal anesthesia | Elective caesarean section under spinal anesthesia, ≥18 years old | Multiple pregnancy, obesity (body mass index > 35), complications to the pregnancy or nonproficiency in the Swedish language | Yes |
| Kovacheva [13] | USA, 2015 | Double-blind, randomized | 60 | Spinal anesthesia | ASA I or II, between 18 and 40 yrs of age, with singleton pregnancies, and undergoing an elective caesarean delivery with a pfannenstiel incision and a spinal anesthesia. | Presence of labor, ruptured membranes, maternal or fetal risk factors for uterine atony, previous uterine surgery (except for one previous caesarean delivery with a low-transverse uterine incision), maternal risks for hemorrhage, contraindications to spinal anesthesia or any of the uterotonic agents, and maternal or obstetrician refusal. | Yes |
| Cecilia [14] | India, 2018 | Double-blind, randomized | 271 | Not known | All the women who underwent elective and emergency CD during the study period were included in the study if they gave informed consent. | Anaemia, placenta previa, abruptio placentae, haemolysis, elevated liver enzymes, and low platelet syndrome, presence of bleeding disorders, intraoperative atony of uterus requiring additional uterotonics or severe intraoperative blood loss requiring blood transfusion, severe fetal distress, previous PPH | Yes |
| Ghulmiyyah [15] | Lebanon, 2016 | Double-blind, randomized | 189 | Not known | Singleton gestation, elective CD at term with no obstetric or medical complication | Multifetal gestation, hypertensive disorders, chorioamnionitis, suspected macrosomia, polyhydramnios, history of PPH, clotting disorders, antecedent intake of magnesium sulphate, history of uterine fibroids, placenta previa/abruption/accrete or those who were in labor | Yes |
| Duffield [16] | USA, 2017 | Double-blind, randomized | 51 | Intrathecal anesthesia using a spinal or combined spinal-epidural technique | ASA physical class 2, singleton pregnancies, ≥ 37 weeks’ gestational age, elective CD with a pfannansteil incision, and aged between 18 and 40 yrs. | Patients with significant medical or obstetric disease, active labor or ruptured membranes, placenta previa or other placental disorders, multiple gestation, known uterine abnormalities, and allergies to oxytocin. | Yes |
| Gungorduk [17] | Turkey, 2010 | Double-blind, randomized | 720 | General anesthesia | Estimated gestational age over 38 weeks and required elective caesarean section | Any risk factor for postpartum hemorrhage, anemia, multiple gestation, antepartum hemorrhage, uterine fibroids, polyhydramnion, emergency CD, a history of uterine atony and postpartum bleeding, current or previous history of significant disease including heart disease, liver, renal disorders or known coagulopathy | Yes |
| Kajendran [18] | Sri Lanka, 2017 | Double-blind, randomized | 92 | Spinal anesthesia | Pregnant women, who were at term, with singleton pregnancies and had a planned elective caesarean section | Women who were in established labor, had multiple pregnancies, established or suspected cases of chorioamnionitis, both minor and major degree placenta praevia and established or suspected cases of placental abruption, previous history of postpartum hemorrhage and coagulation disorders, and women with a history of or had ultrasonically proven fibroids. | Yes |
| McLeods [19] | UK, 2010 | Double-blind, randomized | 74 | Spinal anesthesia | Elective caesarean section | Placenta praevia, multiple pregnancy, known bleeding disorder or use of anticoagulant therapy, a history of major obstetric hemorrhage or if the surgeon felt that participation was not appropriate, technical problems in the time leading up to administration of oxytocin. | Yes |
| Murphy [20] | UK, 2009 | Double-blind, randomized | 110 | Regional anesthesia | Elective lower segment caesarean section | Placenta praevia, multiple pregnancy, known bleeding disorder or use of anti-coagulant therapy, a past history of a major obstetric hemorrhage or if the surgeon felt that participation was not appropriate for any reason. | Yes |
| Sheehan [21] | Ireland, 2011 | Double-blind, randomized | 2058 | Spinal anesthesia | Healthy women at term (>36 weeks) with singleton pregnancy booked for elective CD | Placenta praevia, thrombocytopenia, coagulopathies, previous major obstetric hemorrhage (>1000 mL), known fibroids, or women who received anticoagulant treatment, did not understand English, younger than 18 years. | Yes |
| Qian [22] | China, 2020 | Triple-blind, randomized | 150 | Epidural anesthesia | ASA II, aged 18–40 years old, body mass index < 40 kg/m2, singleton pregnancy, ≥37 weeks’ gestation age, elective CDplanned with a pannenstiel incision, and planning epidural anesthesia. | Maternal refusal, emergency CD, active labor, ruptured membranes, pregnancy-induced hypertension, placental abnormalities, multiple gestation, uterine fibroids, history of prior peripartum hemorrhage, coagulation disorders, oxytocin allergy, contraindication to epidural anesthesia, and the need for pharmacological anxiolysis | Yes |
| Investigation Object | Primary Author | Placebo or Other Group Treatment | Investigative Group Treatment | Results | |||
|---|---|---|---|---|---|---|---|
| Uterine Tone | Blood Loss | PPH Incidence | ECG And Hemodynamics | ||||
| Bolus dose | Butwick [8] | Normal saline bolus IV over 15 s and 10 IU oxytocin in 250 mL 0.9% normal saline over 2 h | 0.5/1/3/5 IU IV oxytocin bolus over 15 s and 10 IU oxytocin in 250 mL 0.9% normal saline over 2 h | No difference observed at 2 min between all groups. | No difference observed | No data | Hypotension occurred more often in 5 IU group vs. 0 at 1 min. |
| Bolus dose | Somjit [10] | 5 IU IV oxytocin bolus over 15 s followed by 20 IU oxytocin in 1 L of Ringer’s lactate at over 8 h | 10 IU IV oxytocin bolus over 15 s followed by 20 IU oxytocin in 1 L of Ringer’s lactate at over 8 h | No difference observed | No difference observed | 0 | No difference observed |
| Bolus dose | King [11] | 3 mL normal saline IV over 30 s followed by 40 IU oxytocin in 500 mL of normal saline over 30 min via infusion pump, then a second infusion of 20 IU oxytocin in 1 L normal saline over 8 h | 5 IU IV oxytocin bolus over 30 s followed by 40 IU oxytocin in 500 mL of normal saline over 30 min via infusion pump, then a second infusion of 20 IU oxytocin in 1 L normal saline over 8 h | Uterine tone score was bigger in investigative group, but disappeared after 5 min | No difference observed | No data | No difference observed |
| Bolus dose | Sartain [9] | 2 IU IV oxytocin bolus over 5–10 s and 40 IU oxytocin in 1 L of Hartmann’s solution over 4 h | 5 IU IV oxytocin bolus over 5–10 s and 40 IU oxytocin in 1 L of Hartmann’s solution over 4 h | No difference observed | No difference observed | No data | After oxytocin bolus increased HR was observed <1 min, at 1 min MAP decreased and was greater in the 5 IU, not 2 IU group. 57.5 per cent of 5 IU group HR increase over 30 beats/min 15 per cent in the same group experienced MAP decrease more than 30 mm Hg |
| Bolus dose | Jonsson [12] | 5 IU IV oxytocin bolus over 1 min followed by 30 IU oxytocin in 500 mL 0.9% saline at a rate sufficient to control uterine atony | 10 IU IV oxytocin bolus over 1 min followed by 30 IU oxytocin in 500 mL 0.9% saline at a rate sufficient to control uterine atony | 9 women in 5 IU group needed additional uterotonic agent | No difference observed | 6 (4 in 5 IU and 2 in 10 IU groups) | Less frequently ST depression and decrease in MAP at 2 min was observed in 5 IU oxytocin group |
| Infusion dose | Cecilia [14] | 30 IU oxytocin IV in 1500 mL IV fluids over 8–12 h | 10 IU oxytocin IV in 500 mL of IV fluids over 2–4 h | Atonic uterus in 7 women (2.5 per cent) in 30 IU group | No difference observed | 2 (1 in each group) | No difference observed |
| Infusion dose | Ghulmiyyah [15] | 20/30/40 IU IV oxytocin in 500 mL of lactated Ringer solution over 30 min followed by consecutively 30 IU then 20 IU then 10 IU of oxytocin in each 1000 mL of lactated Ringer solution (a total of 3 L) for 24 h postpartum. | No difference observed | No difference observed | 0 | No difference observed | |
| Infusion dose | Duffield [16] | 1 IU IV oxytocin bolus and 10 IU oxytocin in 1000 mL lactated Ringer’s solution for 4 h. | 1 IU IV oxytocin bolus and 60 IU oxytocin in 1000 mL lactated Ringer’s solution for 4 h. | No difference observed | No difference observed | 8 (4 in each group) | No difference observed |
| Infusion dose | Gungorduk [17] | 5 IU IV oxytocin bolus over 5–10 s and a 500 mL of lactated Ringer’s solution for 4 h. | 5 IU IV oxytocin bolus over 5–10 s and a 30 IU oxytocin infusion in 500 mL of lactated Ringer’s solution for 4 h | Placebo group required more frequent additional uterotonic agent | The amount of lost blood smaller in the investigative group. Placebo group required more frequent blood transfusion | 46 in placebo group and 18 in oxytocin group (12.8 and 5 per cent respectively) | No difference observed |
| Infusion dose | Kajendran [18] | 5 IU IV oxytocin bolus over 5–10 s and 500 mL of 0.9% normal saline for 4 h | 5 IU IV oxytocin bolus over 5–10 s and 20 IU oxytocin in 500 mL 0.9% normal saline solution for 4 h | No need for additional uterotonic agent in both groups | Investigative group amount of blood loss was smaller | No data | No data |
| Infusion dose | Murphy [20] | 5 IU IV oxytocin bolus and 500 mL of Hartmann’s solution for 4 h | 5 IU IV oxytocin bolus and 30 IU oxytocin in 500 mL of Hartmann’s solution for 4 h | Investigative group—almost none needed additional uterotonic agent | Investigative group amount of lost blood was lower | 3 (2 in placebo and 1 in oxytocin group) | No data |
| Infusion dose | McLeods [19] | 5 IU IV oxytocin bolus over 3 min and a placebo infusion of Hartmann’s solution 500 mL over 4 h. | 5 IU IV oxytocin bolus over 3 min and a 30 IU oxytocin infusion in 500 mL | No data | No data | No data | No difference observed |
| Infusion dose | Sheehan [21] | 5 IU oxytocin IV bolus over 1 min and 500 mL of 0.9% saline IV over 4 h | 5 IU oxytocin IV bolus over 1 min and 40 IU oxytocin in 500 mL 0.9% saline solution IV over 4 h | Placebo group—more frequently needed additional uterotonic agent | No difference observed | 317 (159 in bolus group and 158 in bolus and infusion group) | No data |
| Infusion dose | Qian [22] | 1 IU IV oxytocin bolus over 15 s and 50 mL normal saline over 1 h | 1 IU IV oxytocin bolus over 15 s and 1/2/3/5/8 IU oxytocin in 50 mL normal saline over 1 h | 3,5,8 IU oxytocin groups required rescue oxytocin bolus or uterotonic agent | No difference observed | 0 | No difference observed |
| Bolus dose, infusion dose | Kovacheva [13] | 3 mL of 0.9% saline bolus over 15 s and 30 IU oxytocin in 500 mL 0.9% saline, wide-open infusion flow rate | 3 IU in 3 mL IV oxytocin bolus over 15 s and 500 mL 0.9% saline, wide-open infusion flow rate | Investigative group required less additional oxytocin | No difference observed | No data | No difference observed |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baliuliene, V.; Vitartaite, M.; Rimaitis, K. Prophylactic Dose of Oxytocin for Uterine Atony during Caesarean Delivery: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 5029. https://doi.org/10.3390/ijerph18095029
Baliuliene V, Vitartaite M, Rimaitis K. Prophylactic Dose of Oxytocin for Uterine Atony during Caesarean Delivery: A Systematic Review. International Journal of Environmental Research and Public Health. 2021; 18(9):5029. https://doi.org/10.3390/ijerph18095029
Chicago/Turabian StyleBaliuliene, Vilda, Migle Vitartaite, and Kestutis Rimaitis. 2021. "Prophylactic Dose of Oxytocin for Uterine Atony during Caesarean Delivery: A Systematic Review" International Journal of Environmental Research and Public Health 18, no. 9: 5029. https://doi.org/10.3390/ijerph18095029
APA StyleBaliuliene, V., Vitartaite, M., & Rimaitis, K. (2021). Prophylactic Dose of Oxytocin for Uterine Atony during Caesarean Delivery: A Systematic Review. International Journal of Environmental Research and Public Health, 18(9), 5029. https://doi.org/10.3390/ijerph18095029





