Concurrent Heavy Metal Exposures and Idiopathic Dilated Cardiomyopathy: A Case-Control Study from the Katanga Mining Area of the Democratic Republic of Congo
Abstract
1. Introduction
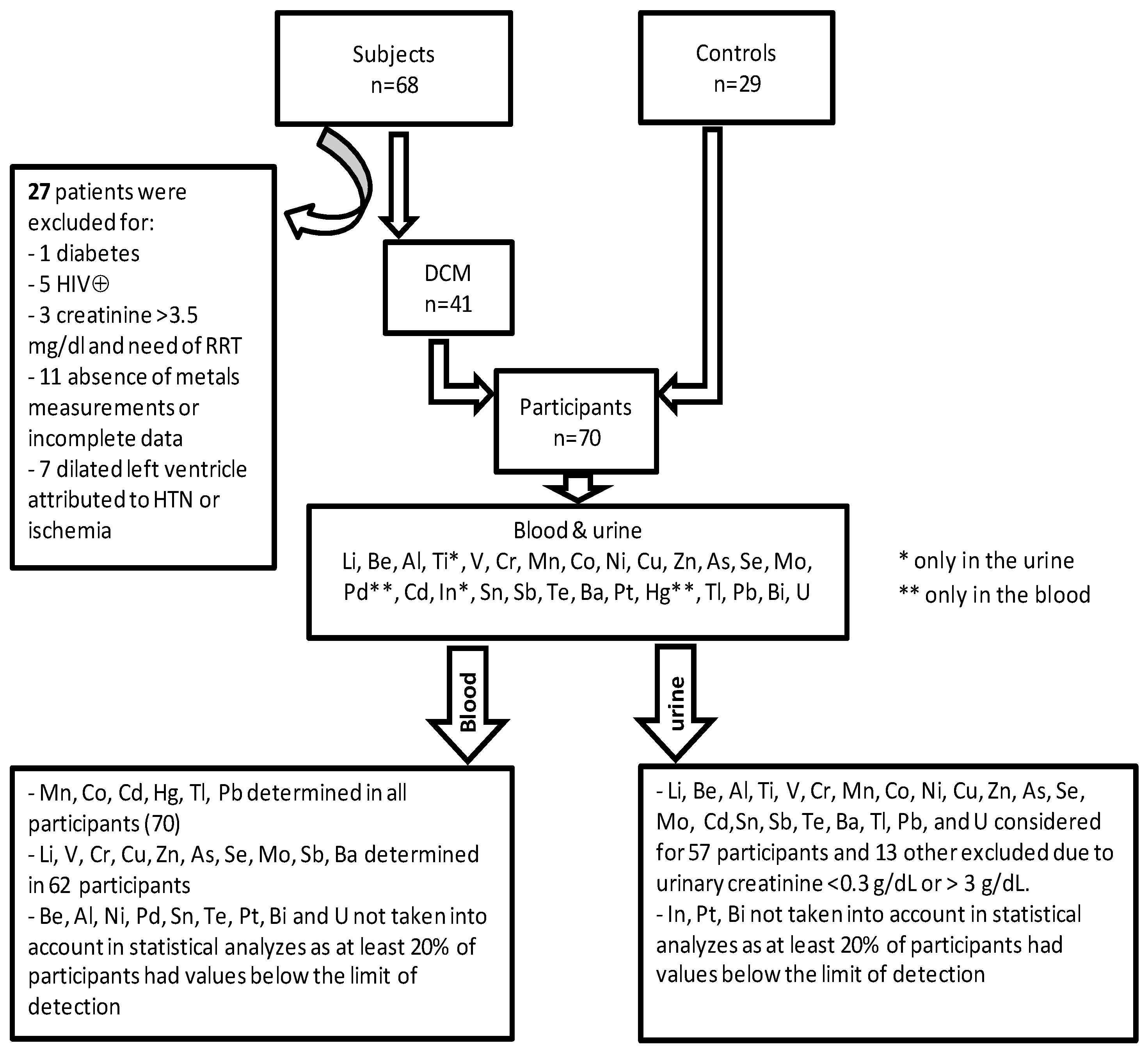
2. Subjects and Methods
2.1. Subjects
2.2. Readout Parameters and Measurements
2.3. Statistical Analysis
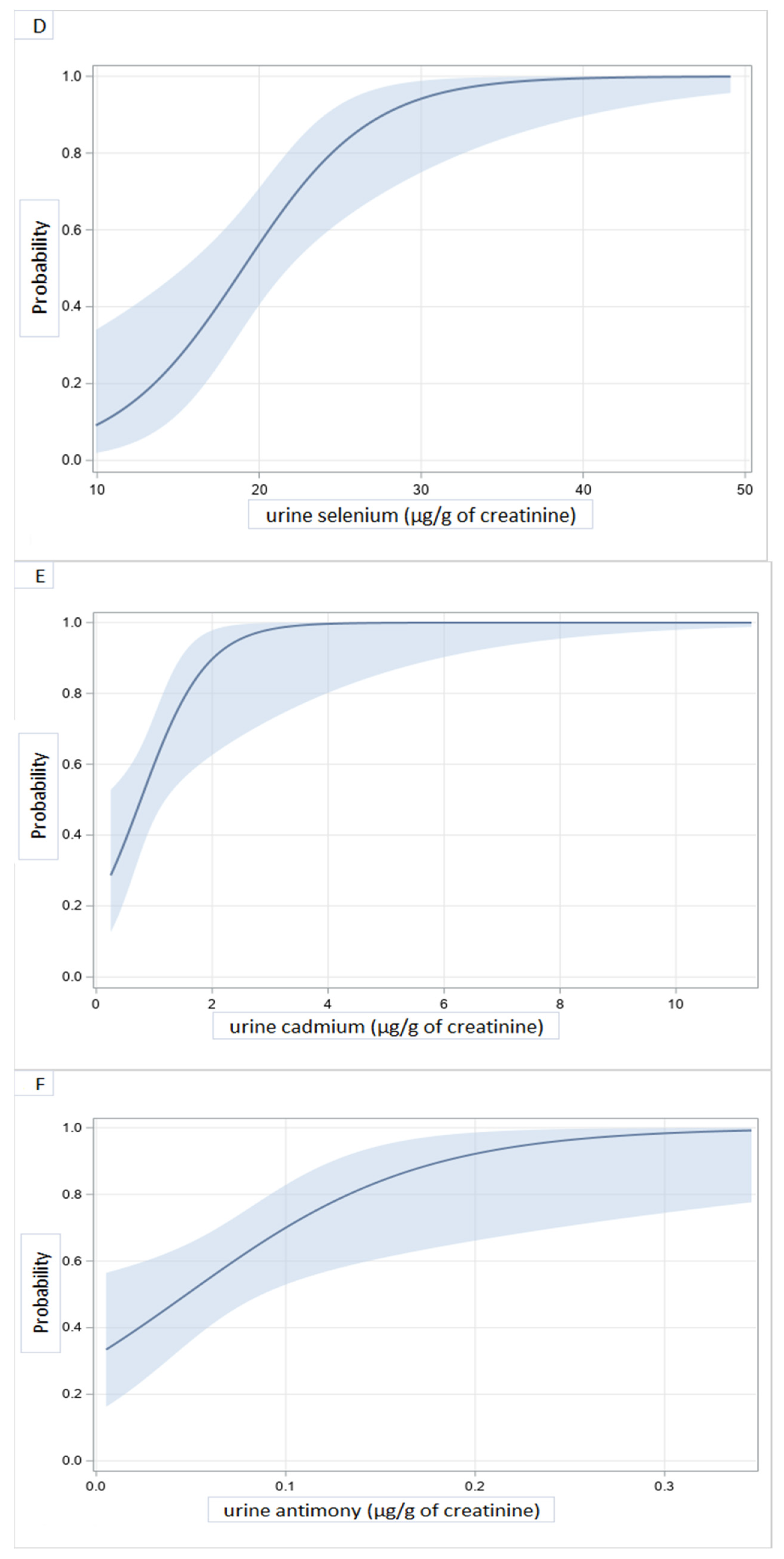
3. Results
3.1. General Characteristics of Study Participants
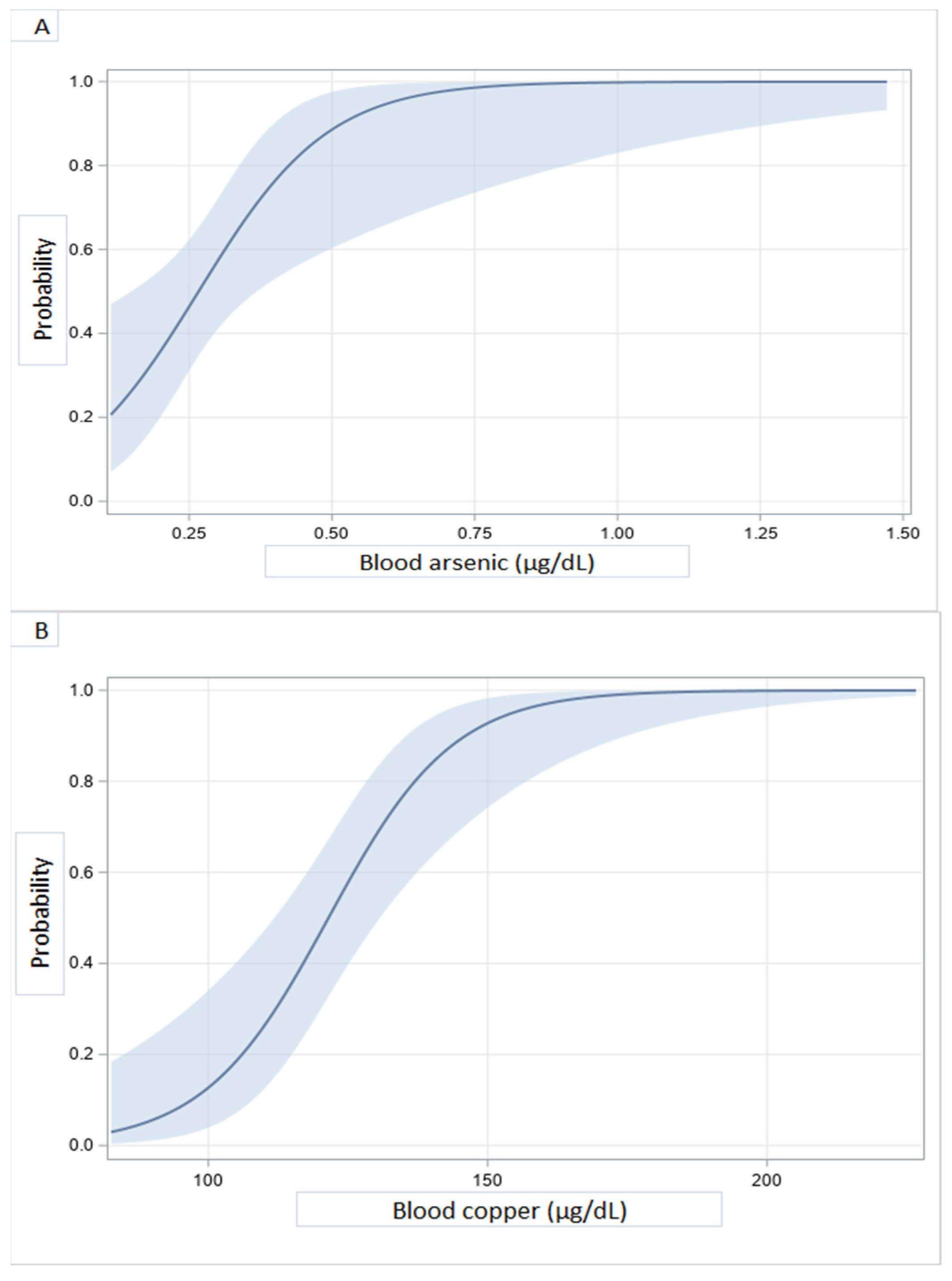
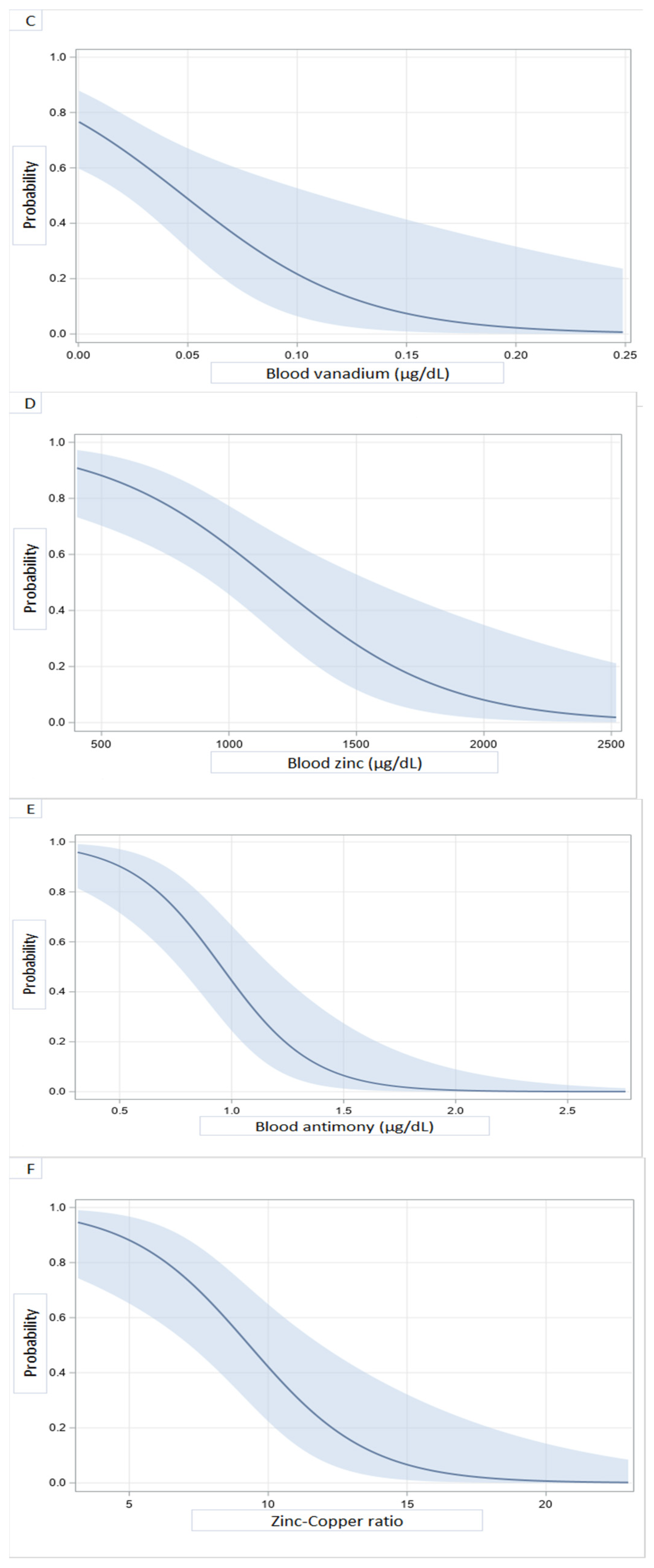
3.2. Blood Concentrations of Heavy Metals and Dilated Cardiomyopathy
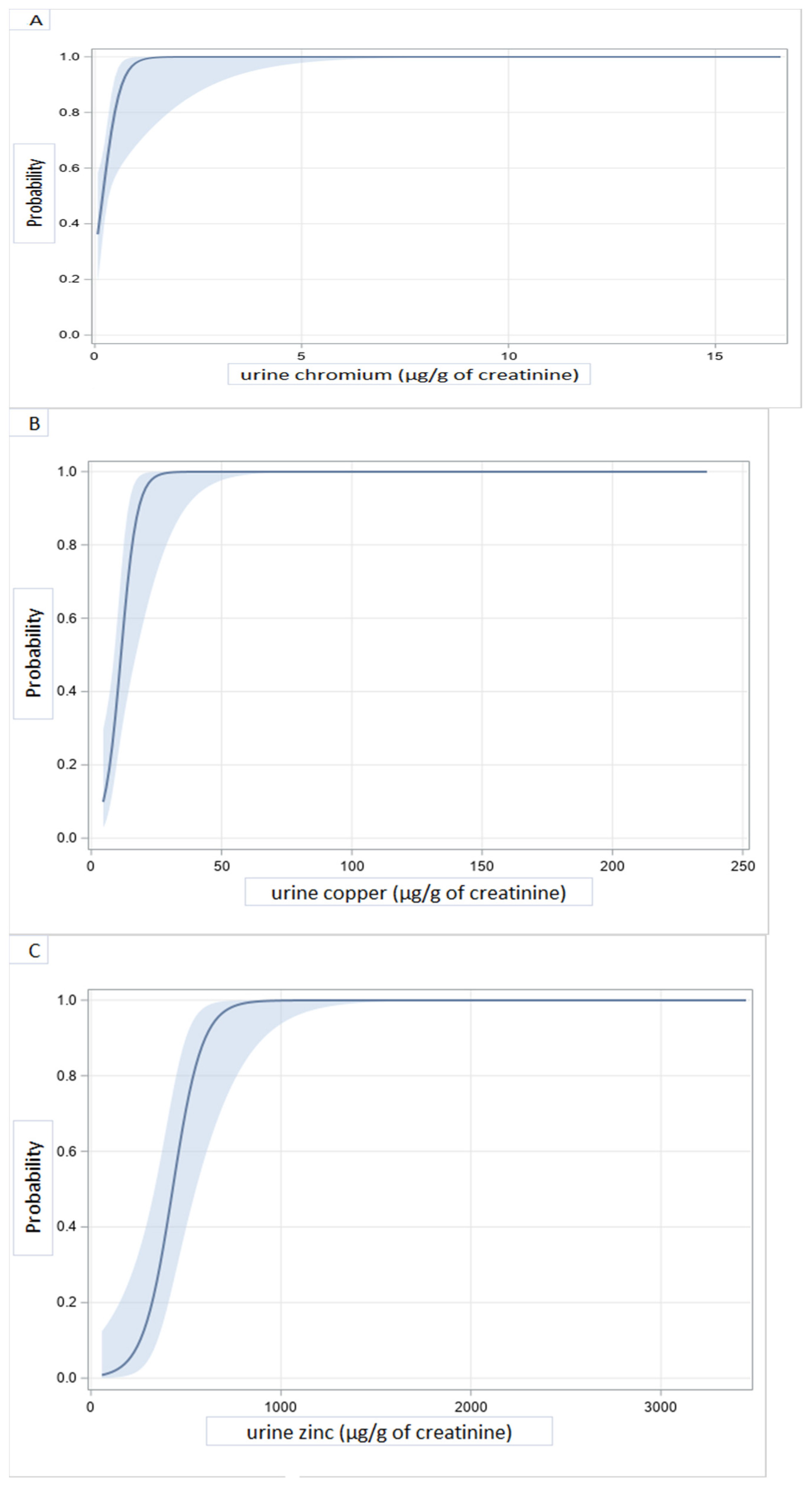
3.3. Urine Concentrations of Heavy Metals and Dilated Cardiomyopathy
4. Discussion
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Availability of Data and Materials
Acknowledgments
Conflicts of Interest
References
- Ober, J.A. Mineral Commodity Summaries 2018; Geological Survey: Liston, VA, USA, 2018.
- Cailteux, J.L.H.; Kampunzu, A.B.; Lerouge, C.; Kaputo, A.K.; Milesi, J.P. Genesis of sediment-hosted stratiform copper–cobalt deposits, central African Copperbelt. J. Afr. Earth Sci. 2005, 42, 134–158. [Google Scholar] [CrossRef]
- Sovacool, B.K. The precarious political economy of cobalt: Balancing prosperity, poverty, and brutality in artisanal and industrial mining in the Democratic Republic of the Congo. Extr. Ind. Soc. 2019. [Google Scholar] [CrossRef]
- Pourret, O.; Lange, B.; Bonhoure, J.; Colinet, G.; Decrée, S.; Mahy, G.; Séleck, M.; Shutcha, M.; Faucon, M.-P. Assessment of soil metal distribution and environmental impact of mining in Katanga (Democratic Republic of Congo). Appl. Geochem. 2016, 64, 43–55. [Google Scholar] [CrossRef]
- Prasad, M.S. Production of copper and cobalt at Gecamines, Zaire. Miner. Eng. 1989, 2, 521–541. [Google Scholar] [CrossRef]
- Edwards, D.P.; Sloan, S.; Weng, L.; Dirks, P.; Sayer, J.; Laurance, W.F. Mining and the African Environment. Conserv. Lett. 2014, 7, 302–311. [Google Scholar] [CrossRef]
- Atibu, E.K.; Lacroix, P.; Sivalingam, P.; Ray, N.; Giuliani, G.; Mulaji, C.K.; Otamonga, J.P.; Mpiana, P.T.; Slaveykova, V.I.; Pote, J. High contamination in the areas surrounding abandoned mines and mining activities: An impact assessment of the Dilala, Luilu and Mpingiri Rivers, Democratic Republic of the Congo. Chemosphere 2018, 191, 1008–1020. [Google Scholar] [CrossRef]
- Atibu, E.K.; Devarajan, N.; Thevenon, F.; Mwanamoki, P.M.; Tshibanda, J.B.; Mpiana, P.T.; Prabakar, K.; Mubedi, J.I.; Wildi, W.; Poté, J. Concentration of metals in surface water and sediment of Luilu and Musonoie Rivers, Kolwezi-Katanga, Democratic Republic of Congo. Appl. Geochem. 2013, 39, 26–32. [Google Scholar] [CrossRef]
- Muhaya, B.B.; wa Kayembe, M.K.; Kunyonga, C.Z.; Catherine, S. Assessment of Trace Metal Contamination of Sediments in the Lubumbashi River Basin, Kafubu, Kimilolo and Kinkalabwamba Rivers in Lubumbashi City, Democratic Republic of Congo. J. Environ. Sci. Eng. A 2017, 6, 167–177. [Google Scholar]
- Muhaya, B.B.; Kunyonga, C.Z.; Mulongo, S.C.; Mushobekwa, F.Z.; Bisimwa, A.M. Trace Metal Contamination of Sediments in Naviundu River Basin, Luano and Ruashi Rivers and Luwowoshi Spring in Lubumbashi City, Democratic Republic of Congo. J. Environ. Sci. Eng. B 2017. [Google Scholar] [CrossRef]
- Squadrone, S.; Burioli, E.; Monaco, G.; Koya, M.K.; Prearo, M.; Gennero, S.; Dominici, A.; Abete, M.C. Human exposure to metals due to consumption of fish from an artificial lake basin close to an active mining area in Katanga (D.R. Congo). Sci. Total Environ. 2016, 568, 679–684. [Google Scholar] [CrossRef]
- Cheyns, K.; Banza Lubaba Nkulu, C.; Ngombe, L.K.; Asosa, J.N.; Haufroid, V.; De Putter, T.; Nawrot, T.; Kimpanga, C.M.; Numbi, O.L.; Ilunga, B.K.; et al. Pathways of human exposure to cobalt in Katanga, a mining area of the D.R. Congo. Sci. Total Environ. 2014, 490, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Mudimbi Kalonda, D.; Kabamba Tshikongo, A.; Kodondi Kule Koto, F.; Kasongo Busambwa, C.; Kisunka Bwalya, Y.; Musola Cansa, H.; Kahambwe Tambwe, J.-L.; Lukumwena Kalala, Z.; Longanga Otshudi, A. Profile of heavy metals contained in food plants commonly consumed in a few mining areas of Katanga province. J. Appl. Biosci. 2015, 96, 9049–9054. [Google Scholar] [CrossRef]
- Banza, C.L.; Nawrot, T.S.; Haufroid, V.; Decree, S.; De Putter, T.; Smolders, E.; Kabyla, B.I.; Luboya, O.N.; Ilunga, A.N.; Mutombo, A.M.; et al. High human exposure to cobalt and other metals in Katanga, a mining area of the Democratic Republic of Congo. Environ. Res. 2009, 109, 745–752. [Google Scholar] [CrossRef] [PubMed]
- Narendrula, R.; Nkongolo, K.K.; Beckett, P. Comparative soil metal analyses in Sudbury (Ontario, Canada) and Lubumbashi (Katanga, DR-Congo). Bull. Environ. Contam. Toxicol. 2012, 88, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Atibu, E.K.; Devarajan, N.; Laffite, A.; Giuliani, G.; Salumu, J.A.; Muteb, R.C.; Mulaji, C.K.; Otamonga, J.-P.; Elongo, V.; Mpiana, P.T.; et al. Assessment of trace metal and rare earth elements contamination in rivers around abandoned and active mine areas. The case of Lubumbashi River and Tshamilemba Canal, Katanga, Democratic Republic of the Congo. Chem. Der Erdegeochem. 2016, 76, 353–362. [Google Scholar] [CrossRef]
- Nemery, B.; Banza Lubaba Nkulu, C. Assessing exposure to metals using biomonitoring: Achievements and challenges experienced through surveys in low- and middle-income countries. Toxicol. Lett. 2018, 298, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Mukendi, R.A.; Banza, C.L.N.; Mukeng, C.A.; Ngwe, J.T.M.; Mwembo, A.N.; Kalenga, P.M.K. Exposure of man to metal trace elements and alteration of sperm parameters: Study conducted in the mining areas of Haut-Katanga in the Democratic Republic of Congo. Pan Afr. Med. J. 2018, 30, 35. [Google Scholar] [CrossRef]
- Bora, B.K.; Ramos-Crawford, A.L.; Sikorskii, A.; Boivin, M.J.; Lez, D.M.; Mumba-Ngoyi, D.; Mukalay Wa Mukalay, A.; Okitundu-Luwa, D.; Tshala-Katumbay, D. Concurrent exposure to heavy metals and cognition in school-age children in Congo-Kinshasa: A complex overdue research agenda. Brain Res. Bull. 2019, 145, 81–86. [Google Scholar] [CrossRef]
- Mbuyi-Musanzayi, S.; Kayembe, T.J.; Kashal, M.K.; Lukusa, P.T.; Kalenga, P.M.; Tshilombo, F.K.; Devriendt, K.; Reychler, H. Non-syndromic cleft lip and/or cleft palate: Epidemiology and risk factors in Lubumbashi (DR Congo), a case-control study. J. Cranio-Maxillo-Facial Surg. 2018, 46, 1051–1058. [Google Scholar] [CrossRef] [PubMed]
- Lubala, T.K.; Shongo, M.Y.; Munkana, A.N.; Mutombo, A.M.; Mbuyi, S.M.; wa Momat, F.K. Congenital malformations in Lubumbashi (Democratic Republic of Congo): About 72 cases observed and advocacy for the development of a National Registry of Congenital Malformations and a National Reference Center for Human Genetics. Pan Afr. Med. J. 2012, 13, 84. [Google Scholar] [CrossRef]
- Kayembe-Kitenge, T.; Kasole Lubala, T.; Musa Obadia, P.; Katoto Chimusa, P.; Katshiez Nawej, C.; Banza Lubaba Nkulu, C.; Devriendt, K.; Nemery, B. Holoprosencephaly: A case series from an area with high mining-related pollution. Birth Defects Res. 2019, 111, 1561–1563. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, R.; Ramond, A.; O’Keeffe, L.M.; Shahzad, S.; Kunutsor, S.K.; Muka, T.; Gregson, J.; Willeit, P.; Warnakula, S.; Khan, H.; et al. Environmental toxic metal contaminants and risk of cardiovascular disease: Systematic review and meta-analysis. BMJ 2018, 362, k3310. [Google Scholar] [CrossRef] [PubMed]
- Aneni, E.C.; Escolar, E.; Lamas, G.A. Chronic Toxic Metal Exposure and Cardiovascular Disease: Mechanisms of Risk and Emerging Role of Chelation Therapy. Curr. Atheroscler. Rep. 2016, 18, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Tellez-Plaza, M.; Jones, M.R.; Dominguez-Lucas, A.; Guallar, E.; Navas-Acien, A. Cadmium exposure and clinical cardiovascular disease: A systematic review. Curr. Atheroscler. Rep. 2013, 15, 356. [Google Scholar] [CrossRef] [PubMed]
- Ferreira de Mattos, G.; Costa, C.; Savio, F.; Alonso, M.; Nicolson, G.L. Lead poisoning: Acute exposure of the heart to lead ions promotes changes in cardiac function and Cav1.2 ion channels. Biophys. Rev. 2017, 9, 807–825. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Du, G.; Zhang, Y.; Zhou, F.; Wu, J.; Jiao, H.; Li, Y.; Chen, Y.; Ouyang, L.; Bo, D.; et al. ECG conduction disturbances and ryanodine receptor expression levels in occupational lead exposure workers. Occup. Environ. Med. 2019, 76, 151–156. [Google Scholar] [CrossRef]
- Shen, J.; Wang, X.; Zhou, D.; Li, T.; Tang, L.; Gong, T.; Su, J.; Liang, P. Modelling cadmium-induced cardiotoxicity using human pluripotent stem cell-derived cardiomyocytes. J. Cell. Mol. Med. 2018, 22, 4221–4235. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Zhang, S.L.; Liu, Z.Y.; Tian, Y.; Sun, Q. Cadmium toxicity induces ER stress and apoptosis via impairing energy homoeostasis in cardiomyocytes. Biosci. Rep. 2015, 35. [Google Scholar] [CrossRef]
- Ferramola, M.L.; Pérez Díaz, M.F.F.; Honoré, S.M.; Sánchez, S.S.; Antón, R.I.; Anzulovich, A.C.; Giménez, M.S. Cadmium-induced oxidative stress and histological damage in the myocardium. Effects of a soy-based diet. Toxicol. Appl. Pharmacol. 2012, 265, 380–389. [Google Scholar] [CrossRef]
- Furieri, L.B.; Fioresi, M.; Junior, R.F.R.; Bartolomé, M.V.; Fernandes, A.A.; Cachofeiro, V.; Lahera, V.; Salaices, M.; Stefanon, I.; Vassallo, D.V. Exposure to low mercury concentration in vivo impairs myocardial contractile function. Toxicol. Appl. Pharmacol. 2011, 255, 193–199. [Google Scholar] [CrossRef]
- Lorscheider, F.; Vimy, M. Mercury and idiopathic dilated cardiomyopathy. J. Am. Coll. Cardiol. 2000, 35, 819. [Google Scholar] [CrossRef][Green Version]
- Frustaci, A.; Magnavita, N.; Chimenti, C.; Caldarulo, M.; Sabbioni, E.; Pietra, R.; Cellini, C.; Possati, G.F.; Maseri, A. Marked elevation of myocardial trace elements in idiopathic dilated cardiomyopathy compared with secondary cardiac dysfunction. J. Am. Coll. Cardiol. 1999, 33, 1578–1583. [Google Scholar] [CrossRef]
- Hantson, P. Mechanisms of toxic cardiomyopathy. Clin. Toxicol. 2018, 57, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Perez, A.L.; Tang, W.H. Contribution of environmental toxins in the pathogenesis of idiopathic cardiomyopathies. Curr. Treat. Options Cardiovasc. Med. 2015, 17, 381. [Google Scholar] [CrossRef]
- Alexanian, I.; Parissis, J.; Farmakis, D.; Athanaselis, S.; Pappas, L.; Gavrielatos, G.; Mihas, C.; Paraskevaidis, I.; Sideris, A.; Kremastinos, D.; et al. Clinical and echocardiographic correlates of serum copper and zinc in acute and chronic heart failure. Clin. Res. Cardiol. 2014, 103, 938–949. [Google Scholar] [CrossRef] [PubMed]
- Malamba-Lez, D.; Ngoy-Nkulu, D.; Steels, P.; Tshala-Katumbay, D.; Mullens, W. Heart Failure Etiologies and Challenges to Care in the Developing World: An Observational Study in the Democratic Republic of Congo. J. Card Fail. 2018, 24, 854–859. [Google Scholar] [CrossRef]
- Banza Lubaba Nkulu, C.; Casas, L.; Haufroid, V.; De Putter, T.; Saenen, N.D.; Kayembe-Kitenge, T.; Musa Obadia, P.; Kyanika Wa Mukoma, D.; Lunda Ilunga, J.M.; Nawrot, T.S.; et al. Sustainability of artisanal mining of cobalt in DR Congo. Nat. Sustain. 2018, 1, 495–504. [Google Scholar] [CrossRef]
- Muhaya, B.B.; Mulongo, S.C.; Kunyonga, C.Z.; Mushobekwa, F.Z.; Wa Kayembe, M.K. Trace Metal Contamination of Water in Naviundu River Basin, Luano and Ruashi Rivers and Luwowoshi Spring in Lubumbashi City, Democratic Republic of Congo. J. Environ. Sci. Eng. A 2017, 6, 329–336. [Google Scholar]
- Initiative EEIT. The Democratic Republic of Congo. 2019. Available online: https://eiti.org/democratic-republic-of-congo (accessed on 17 July 2019).
- Lang, R.M.; Badano, L.P.; Mor-Avi, V.; Afilalo, J.; Armstrong, A.; Ernande, L.; Flachskampf, F.A.; Foster, E.; Goldstein, S.A.; Kuznetsova, T.; et al. Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur. Heart J. Cardiovasc. Imaging 2015, 16, 233–270. [Google Scholar] [CrossRef]
- Nagueh, S.F.; Smiseth, O.A.; Appleton, C.P.; Byrd, B.F., 3rd; Dokainish, H.; Edvardsen, T.; Flachskampf, F.A.; Gillebert, T.C.; Klein, A.L.; Lancellotti, P.; et al. Recommendations for the Evaluation of Left Ventricular Diastolic Function by Echocardiography: An Update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 2016, 29, 277–314. [Google Scholar] [CrossRef]
- Prineas, J.R.; Crow, S.R.; Zhang, Z.-M. The Minnesota Code Manual of Electrocardiographic Findings: Standards and Procedures for Measurement and Classification, 2nd ed.; Springer: New York, NY, USA, 2010. [Google Scholar] [CrossRef]
- Hoet, P.; Jacquerye, C.; Deumer, G.; Lison, D.; Haufroid, V. Reference values and upper reference limits for 26 trace elements in the urine of adults living in Belgium. Clin. Chem. Lab. Med. 2013, 51, 839–849. [Google Scholar] [CrossRef]
- Salehifar, E.; Shokrzadeh, M.; Ghaemian, A.; Aliakbari, S.; Saeedi Saravi, S.S. The study of Cu and Zn serum levels in idiopathic dilated cardiomyopathy (IDCMP) patients and its comparison with healthy volunteers. Biol. Trace Elem. Res. 2008, 125, 97–108. [Google Scholar] [CrossRef]
- Shokrzadeh, M.; Ghaemian, A.; Salehifar, E.; Aliakbari, S.; Saravi, S.S.S.; Ebrahimi, P. Serum Zinc and Copper Levels in Ischemic Cardiomyopathy. Biol. Trace Elem. Res. 2009, 127, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Nisse, C.; Tagne-Fotso, R.; Howsam, M.; Richeval, C.; Labat, L.; Leroyer, A. Blood and urinary levels of metals and metalloids in the general adult population of Northern France: The IMEPOGE study, 2008–2010. Int. J. Hyg. Environ. Health 2017, 220, 341–363. [Google Scholar] [CrossRef]
- Wang, X.; Mukherjee, B.; Park, S.K. Associations of cumulative exposure to heavy metal mixtures with obesity and its comorbidities among U.S. adults in NHANES 2003–2014. Environ. Int. 2018, 121, 683–694. [Google Scholar] [CrossRef]
- Rehman, K.; Fatima, F.; Waheed, I.; Akash, M.S.H. Prevalence of exposure of heavy metals and their impact on health consequences. J. Cell. Biochem. 2018, 119, 157–184. [Google Scholar] [CrossRef]
- Nunes, J.A.; Batista, B.L.; Rodrigues, J.L.; Caldas, N.M.; Neto, J.A.; Barbosa, F., Jr. A simple method based on ICP-MS for estimation of background levels of arsenic, cadmium, copper, manganese, nickel, lead, and selenium in blood of the Brazilian population. J. Toxicol. Environ. Health Part A 2010, 73, 878–887. [Google Scholar] [CrossRef] [PubMed]
- Waseem, A.; Arshad, J. A review of Human Biomonitoring studies of trace elements in Pakistan. Chemosphere 2016, 163, 153–176. [Google Scholar] [CrossRef] [PubMed]
- Ding, C.; Pan, Y.; Zhang, A.; Wu, B.; Huang, H.; Zhu, C.; Liu, D.; Zhu, B.; Xu, G.; Shao, H.; et al. Study of distribution and influencing factors of arsenic in whole blood and urine among population in 8 provinces in China. Zhonghua Yu Fang Yi Xue Za Zhi [Chin. J. Prev. Med.] 2014, 48, 97–101. [Google Scholar] [PubMed]
- Das, A.K.; Sahu, R.; Dua, T.K.; Bag, S.; Gangopadhyay, M.; Sinha, M.K.; Dewanjee, S. Arsenic-induced myocardial injury: Protective role of Corchorus olitorius leaves. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2010, 48, 1210–1217. [Google Scholar] [CrossRef]
- Manna, P.; Sinha, M.; Sil, P.C. Arsenic-induced oxidative myocardial injury: Protective role of arjunolic acid. Arch. Toxicol. 2008, 82, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Alamolhodaei, N.S.; Shirani, K.; Karimi, G. Arsenic cardiotoxicity: An overview. Environ. Toxicol. Pharmacol. 2015, 40, 1005–1014. [Google Scholar] [CrossRef] [PubMed]
- Smeester, L.; Rager, J.E.; Bailey, K.A.; Guan, X.; Smith, N.; Garcia-Vargas, G.; Del Razo, L.M.; Drobna, Z.; Kelkar, H.; Styblo, M.; et al. Epigenetic changes in individuals with arsenicosis. Chem. Res. Toxicol. 2011, 24, 165–167. [Google Scholar] [CrossRef]
- Mo, J.; Xia, Y.; Wade, T.J.; DeMarini, D.M.; Davidson, M.; Mumford, J. Altered gene expression by low-dose arsenic exposure in humans and cultured cardiomyocytes: Assessment by real-time PCR arrays. Int. J. Environ. Res. Public Health 2011, 8, 2090–2108. [Google Scholar] [CrossRef] [PubMed]
- Domingo-Relloso, A.; Grau-Perez, M.; Briongos-Figuero, L.; Gomez-Ariza, J.L.; Garcia-Barrera, T.; Duenas-Laita, A.; Bobb, J.F.; Chaves, F.J.; Kioumourtzoglou, M.A.; Navas-Acien, A.; et al. The association of urine metals and metal mixtures with cardiovascular incidence in an adult population from Spain: The Hortega Follow-Up Study. Int. J. Epidemiol. 2019, 48, 1839–1849. [Google Scholar] [CrossRef]
- Oster, O. Trace element concentrations (Cu, Zn, Fe) in sera from patients with dilated cardiomyopathy. Clin. Chim. Acta Int. J. Clin. Chem. 1993, 214, 209–218. [Google Scholar] [CrossRef]
- Topuzoglu, G.; Erbay, A.R.; Karul, A.B.; Yensel, N. Concentrations of copper, zinc, and magnesium in sera from patients with idiopathic dilated cardiomyopathy. Biol. Trace Elem. Res. 2003, 95, 11–17. [Google Scholar] [CrossRef]
- Kosar, F.; Sahin, I.; Taskapan, C.; Kucukbay, Z.; Gullu, H.; Taskapan, H.; Cehreli, S. Trace element status (Se, Zn, Cu) in heart failure. Anadolu Kardiyol. Derg. Akd Anatol. J. Cardiol. 2006, 6, 216–220. [Google Scholar]
- Cenac, A.; Simonoff, M.; Djibo, A. Nutritional status and plasma trace elements in peripartum cardiomyopathy. A comparative study in Niger. J. Cardiovasc. Risk 1996, 3, 483–487. [Google Scholar] [CrossRef]
- Chou, H.T.; Yang, H.L.; Tsou, S.S.; Ho, R.K.; Pai, P.Y.; Hsu, H.B. Status of trace elements in patients with idiopathic dilated cardiomyopathy in central Taiwan. Zhonghua Yi Xue Za Zhi Chin. Med. J. Free China Ed. 1998, 61, 193–198. [Google Scholar]
- Malek, F.; Dvorak, J.; Jiresova, E.; Spacek, R. Difference of baseline serum copper levels between groups of patients with different one year mortality and morbidity and chronic heart failure. Cent. Eur. J. Public Health 2003, 11, 198–201. [Google Scholar]
- Gaetke, L.M.; Chow, C.K. Copper toxicity, oxidative stress, and antioxidant nutrients. Toxicology 2003, 189, 147–163. [Google Scholar] [CrossRef]
- Gaetke, L.M.; Chow-Johnson, H.S.; Chow, C.K. Copper: Toxicological relevance and mechanisms. Arch. Toxicol. 2014, 88, 1929–1938. [Google Scholar] [CrossRef] [PubMed]
- Fukai, T.; Ushio-Fukai, M.; Kaplan, J.H. Copper transporters and copper chaperones: Roles in cardiovascular physiology and disease. Am. J. Physiol. Cell Physiol. 2018, 315, C186–C201. [Google Scholar] [CrossRef]
- Grandis, D.J.; Nah, G.; Whitman, I.R.; Vittinghoff, E.; Dewland, T.A.; Olgin, J.E.; Marcus, G.M. Wilson’s Disease and Cardiac Myopathy. Am. J. Cardiol. 2017, 120, 2056–2060. [Google Scholar] [CrossRef] [PubMed]
- WHO. Trace Elements in Human Nutrition and Health. 1996. Available online: https://apps.who.int/iris/handle/10665/37931 (accessed on 17 February 2018).
- Canada, H. Report on Human Biomonitoring of Environmental Chemicals in Canada. Results of the Canadian Health Measures Survey Cycle 1 (2007–2009); Health Canada: Ottawa, ON, Canada, 2010.
- Yu, X.; Huang, L.; Zhao, J.; Wang, Z.; Yao, W.; Wu, X.; Huang, J.; Bian, B. The Relationship between Serum Zinc Level and Heart Failure: A Meta-Analysis. Biomed. Res. Int. 2018, 2018, 2739014–2739019. [Google Scholar] [CrossRef]
- Weber, K.T.; Weglicki, W.B.; Simpson, R.U. Macro- and micronutrient dyshomeostasis in the adverse structural remodelling of myocardium. Cardiovasc. Res. 2009, 81, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Liu, X.; Pan, Z. Zinc deficiency and cellular oxidative stress: Prognostic implications in cardiovascular diseases. Acta Pharmacol. Sin. 2018, 39, 1120–1132. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.; Vidal, A.; Bhattacharya, S.K.; Ahokas, R.A.; Sun, Y.; Gerling, I.C.; Weber, K.T. Zinc dyshomeostasis in rats with aldosteronism. Response to spironolactone. Am. J. Physiol. Heart Circ. Physiol. 2007, 293, H2361–H2366. [Google Scholar] [CrossRef] [PubMed]
- Ripa, S.; Ripa, R.; Giustiniani, S. Are failured cardiomyopathies a zinc-deficit related disease? A study on Zn and Cu in patients with chronic failured dilated and hypertrophic cardiomyopathies. Minerva Med. 1998, 89, 397–403. [Google Scholar] [PubMed]
- Gandhi, M.S.; Deshmukh, P.A.; Kamalov, G.; Zhao, T.; Zhao, W.; Whaley, J.T.; Tichy, J.R.; Bhattacharya, S.K.; Ahokas, R.A.; Sun, Y.; et al. Causes and consequences of zinc dyshomeostasis in rats with chronic aldosteronism. J. Cardiovasc. Pharmacol. 2008, 52, 245–252. [Google Scholar] [CrossRef]
- Brzoska, M.M.; Moniuszko-Jakoniuk, J. Interactions between cadmium and zinc in the organism. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2001, 39, 967–980. [Google Scholar] [CrossRef]
- Prasad, A.S.; Bao, B. Molecular Mechanisms of Zinc as a Pro-Antioxidant Mediator: Clinical Therapeutic Implications. Antioxidants 2019, 8, 164. [Google Scholar] [CrossRef]
- Soudani, N.; Troudi, A.; Bouaziz, H.; Ben Amara, I.; Boudawara, T.; Zeghal, N. Cardioprotective effects of selenium on chromium (VI)-induced toxicity in female rats. Ecotoxicol. Environ. Saf. 2011, 74, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Peters, J.L.; Perlstein, T.S.; Perry, M.J.; McNeely, E.; Weuve, J. Cadmium exposure in association with history of stroke and heart failure. Environ. Res. 2010, 110, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Smetana, R.H.; Glogar, D.H. Role of cadmium and magnesium in pathogenesis of idiopathic dilated cardiomyopathy. Am. J. Cardiol. 1986, 58, 364–366. [Google Scholar] [CrossRef]
- Sundar, S.; Chakravarty, J. Antimony toxicity. Int. J. Environ. Res. Public Health 2010, 7, 4267–4277. [Google Scholar] [CrossRef]
- Alvarez, M.; Malécot, C.O.; Gannier, F.; Lignon, J.M. Antimony-induced cardiomyopathy in guinea-pig and protection by L-carnitine. Br. J. Pharm. 2005, 144, 17–27. [Google Scholar] [CrossRef][Green Version]
- Cvjetko, P.; Cvjetko, I.; Pavlica, M. Thallium toxicity in humans. Arh. Za Hig. Rada I Toksikol. 2010, 61, 111–119. [Google Scholar] [CrossRef]
- Osorio-Rico, L.; Santamaria, A.; Galvan-Arzate, S. Thallium Toxicity: General Issues, Neurological Symptoms, and Neurotoxic Mechanisms. Adv. Neurobiol. 2017, 18, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, R.S. Thallium toxicity and the role of Prussian blue in therapy. Toxicol. Rev. 2003, 22, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Lin, G.; Yuan, L.; Bai, L.; Liu, Y.; Wang, Y.; Qiu, Z. Successful treatment of a patient with severe thallium poisoning in a coma using Prussian blue and plasma exchange: A case report. Medicine 2019, 98, e14629. [Google Scholar] [CrossRef]
- Xiao, T.; Yang, F.; Li, S.; Zheng, B.; Ning, Z. Thallium pollution in China: A geo-environmental perspective. Sci. Total Environ. 2012, 421–422, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Keith, S.; Faroon, O.; Roney, N.; Scinicariello, F.; Wilbur, S.; Ingerman, L.; Llados, F.; Plewak, D.; Wohlers, D.; Diamond, G. Toxicological Profile for Uranium. In Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological Profiles; Agency for Toxic Substances and Disease Registry (US): Atlanta, GA, USA, 2013. [Google Scholar]
- Bjorklund, G.; Christophersen, O.A.; Chirumbolo, S.; Selinus, O.; Aaseth, J. Recent aspects of uranium toxicology in medical geology. Environ. Res. 2017, 156, 526–533. [Google Scholar] [CrossRef]
- Pavlakis, N.; Pollock, C.A.; McLean, G.; Bartrop, R. Deliberate overdose of uranium: Toxicity and treatment. Nephron 1996, 72, 313–317. [Google Scholar] [CrossRef] [PubMed]

| Metal (Reference Value) | Controls (n = 29) GM (95% CL) Mean ± SD Median (IQR) | DCM (n = 41) Ø GM (95% CL) Mean ± SD Median (IQR) | GM Ratio (95% CL) | p |
|---|---|---|---|---|
| Mn 0.77 ¥ | 1.59 (1.41–1.81) | 1.56 (1.39–1.76) | 1.02 (0.86–1.22) | 0.8094 |
| 1.69 ± 0.61 | 1.67 ± 0.69 | |||
| 1.59 (0.58) | 1.64 (0.80) | |||
| Co 0.03 ¥ | 0.06 (0.04–0.08) | 0.12 (0.08–0.16) | 0.50 (0.31–0.82) | 0.0063 |
| 0.09 ± 0.08 | 0.22 ± 0.31 | |||
| 0.05 (0.07) | 0.09 (0.14) | |||
| Cd 0.04 ¥ | 0.16 (0.15–0.17) | 0.13 (0.08–0.21) | 1.24 (0.86–1.31) | 0.3636 |
| 0.17 ± 0.04 | 0.21 ± 0.28 | |||
| 0.16 (0.04) | 0.14 (0.09) | |||
| Hg | 0.26 (0.22–0.31) | 0.26 (0.22–0.31) | 0.99 (0.77–1.28) | 0.9663 |
| 0.29 ± 0.15 | 0.31 ± 0.24 | |||
| 0.27 (0.13) | 0.26 (0.13) | |||
| Tl 0.002 ¥ | 0.0027 (0.0024–0.0031) | 0.0028 (0.002–0.0039) | 0.96 (0.67–1.38) | 0.8273 |
| 0.0029 ± 0.0012 | 0.0050 ± 0.0082 | |||
| 0.0027 (0.0008) | 0.0036 (0.0027) | |||
| Pb 1.88 ¥ | 5.65 (4.83–6.61) | 6.99 (5.94–8.22) | 0.81 (0.64–1.02) | 0.0686 |
| 6.13 ± 2.59 | 8.02 ± 4.94 | |||
| 5.83 (2.81) | 6.76 (2.93) | |||
| Li | 0.21 (0.08–0.61) | 0.10 (0.07–0.16) | 2.08 (0.67–6.45) | 0.1981 |
| 1.11 ± 1.05 | 0.26 ± 0.54 | |||
| 1.22 (1.89) | 0.10 (0.11) | |||
| V 0.005 * | 0.02 (0.01–0.05) | 0.006 (0.004–0.01) | 3.5 (1.40–8.98) | 0.0086 |
| 0.07 ± 0.07 | 0.02 ± 0.31 | |||
| 0.52 (0.13) | 0.007 (0.008) | |||
| Cr 0.04 | 0.20 (0.18–0.22) | 0.18 (0.15–0.21) | 1.12 (0.94–1.33) | 0.2136 |
| 0.21 ± 0.06 | 0.20 ± 0.13 | |||
| 0.19 (0.04) | 0.17 (0.07) | |||
| Cu 70–140 ‡ | 110.6 (105–116.4) | 143.7 (135.1–153) | 0.77 (0.71–0.83) | <0.0001 |
| 111.51 ± 14.83 | 145.88 ± 25.74 | |||
| 109.71 (20.50) | 146.84 (31.15) | |||
| Zn 580.5 ¥ | 1196.9 (992.1–1444) | 823.4 (740.3–915.8) | 1.5 (1.2–1.8) | 0.0009 |
| 1331.38 ± 577.13 | 864.21 ± 307.60 | |||
| 1509.84 (906.91) | 797.38 (230.11) | |||
| Zn/Cu | 10.8 (8.8–13.2) | 5.7 (5.1–6.4) | 1.9 (1.5–2.4) | <0.0001 |
| 12.18 ± 5.44 | 6.08 ± 2.47 | |||
| 13.33 (8.02) | 5.39 (1.95) | |||
| As 0.17 ¥ | 0.23 (0.20–0.26) | 0.39 (0.32–0.49) | 0.58 (0.46–0.74) | <0.0001 |
| 0.25 ± 0.09 | 0.47 ± 0.33 | |||
| 0.22 (0.09) | 0.38 (0.23) | |||
| Se 12.5 ** | 12.6 (11.8–13.4) | 11.9 (11.1–12.8) | 1.05 (0.9–1.2) | 0.2820 |
| 12.78 ± 2.12 | 12.20 ± 2.51 | |||
| 13.12 (3.34) | 11.82 (3.28) | |||
| Mo 0.1–0.3 ‡ | 0.18 (0.16–0.21) | 0.21 (0.18–0.25) | 0.87 (0.69–1.09) | 0.2385 |
| 0.20 ± 0.11 | 0.25 ± 0.23 | |||
| 0.17 (0.07) | 0.19 (0.08) | |||
| Sb 0.005 | 1.11 (0.96–1.29) | 0.56 (0.49–0.64) | 1.98 (1.63–2.42) | <0.0001 |
| 1.19 ± 0.46 | 0.61 ± 0.29 | |||
| 1.18 (0.47) | 0.57 (0.27) | |||
| Ba 0.05–0.25 | 0.13 (0.09–0.16) | 0.31 (0.2–0.48) | 2.45 (1.50–4.01) | 0.0006 |
| 0.53 ± 0.53 | 0.18 ± 0.29 | |||
| 0.33 (0.68) | 0.11 (0.05) |
| Variable | Coefficient | SE | Wald 95% Confidence Limits | Wald X2 | p-Value | |
|---|---|---|---|---|---|---|
| Arsenic Model | ||||||
| Intercept | −2.84 | 1.65 | −6.06 | 0.39 | 2.97 | 0.0850 |
| As | 9.04 | 3.44 | 2.28 | 15.79 | 6.88 | 0.0087 |
| Age | −0.001 | 0.03 | −0.06 | 0.06 | 0.00 | 0.9639 |
| Male sex | 0.48 | 0.70 | −0.89 | 1.85 | 0.47 | 0.4909 |
| Education X | −1.14 | 0.74 | −2.59 | 0.29 | 2.43 | 0.1192 |
| GFR < 60 | 2.21 | 1.00 | 0.24 | 4.17 | 4.83 | 0.0279 |
| Copper model | ||||||
| Intercept | −13.49 | 3.98 | −21.29 | −5.68 | 11.48 | 0.0007 |
| Cu | 0.09 | 0.02 | 0.04 | 0.14 | 13.07 | 0.0003 |
| Age | 0.02 | 0.03 | −0.04 | 0.09 | 0.50 | 0.4776 |
| Male sex | 1.46 | 0.87 | −0.24 | 3.15 | 2.85 | 0.0915 |
| Education | −0.79 | 0.80 | −2.37 | 0.77 | 0.99 | 0.3195 |
| GFR < 60 | 1.18 | 1.15 | −1.07 | 3.44 | 1.06 | 0.3042 |
| Vanadium model | ||||||
| Intercept | 1.39 | 1.49 | −1.53 | 4.31 | 0.87 | 0.3517 |
| Vanadium | −23.91 | 8.93 | −41.42 | −6.40 | 7.16 | 0.0074 |
| Age | −0.01 | 0.03 | −0.08 | 0.05 | 0.22 | 0.6394 |
| Male sex | 0.30 | 0.68 | −1.04 | 1.65 | 0.19 | 0.6589 |
| Education | −1.18 | 0.70 | −2.55 | 0.19 | 2.83 | 0.0925 |
| GFR < 60 | 2.75 | 1.13 | 0.54 | 4.96 | 5.95 | 0.0147 |
| Zinc model | ||||||
| Intercept | 3.36 | 1.86 | −0.28 | 7.00 | 3.28 | 0.0701 |
| Zinc | −0.003 | 0.0009 | −0.005 | −0.001 | 9.53 | 0.0020 |
| Age | −0.02 | 0.03 | −0.08 | 0.05 | 0.21 | 0.6437 |
| Male sex | 0.79 | 0.73 | −0.63 | 2.22 | 1.18 | 0.2769 |
| Education | −1.39 | 0.74 | −2.84 | 0.06 | 3.54 | 0.0598 |
| GFR < 60 | 3.32 | 1.31 | 0.75 | 5.88 | 6.43 | 0.0112 |
| Antimony model | ||||||
| Intercept | 4.89 | 2.16 | 0.66 | 9.13 | 5.12 | 0.0236 |
| Antimony | −4.65 | 1.25 | −7.09 | −2.20 | 13.87 | 0.0002 |
| Age | −0.02 | 0.04 | −0.09 | 0.05 | 0.40 | 0.5284 |
| Male sex | 0.58 | 0.82 | −1.01 | 2.19 | 0.52 | 0.4720 |
| Education | −1.42 | 0.84 | −3.07 | 0.22 | 2.89 | 0.0892 |
| GFR < 60 | 3.58 | 1.66 | 0.33 | 6.83 | 4.65 | 0.0310 |
| Barium model | ||||||
| Intercept | 1.32 | 1.40 | −1.43 | 4.06 | 0.89 | 0.3459 |
| Barium | −2.73 | 1.24 | −5.17 | −0.29 | 4.81 | 0.0284 |
| Age | −0.01 | 0.03 | −0.07 | 0.05 | 0.15 | 0.6997 |
| Male sex | 0.19 | 0.65 | −1.09 | 1.48 | 0.09 | 0.7673 |
| Education | −1.18 | 0.67 | −2.49 | 0.13 | 3.13 | 0.0769 |
| GFR < 60 | 2.54 | 1.01 | 0.57 | 4.51 | 6.36 | 0.0117 |
| Ratio Zinc/Copper | ||||||
| Intercept | 3.63 | 2.03 | −0.35 | 7.61 | 3.20 | 0.0738 |
| Zinc/copper | −0.46 | 0.14 | −0.74 | −0.19 | 11.01 | 0.0009 |
| Age | −0.01 | 0.04 | −0.09 | 0.07 | 0.06 | 0.8053 |
| Male sex | 1.44 | 0.89 | −0.31 | 3.20 | 2.59 | 0.1077 |
| Education | −1.31 | 0.81 | −2.89 | 0.27 | 2.63 | 0.1050 |
| GFR < 60 | 3.73 | 1.71 | 0.38 | 7.07 | 4.76 | 0.0291 |
| Metal (Reference Value) | Controls (n = 25) GM (95% CL) Mean ± SD Median (IQR) | DCM (n = 32) GM (95% CL) Mean ± SD Median (IQR) | GM Ratio (95% CL) | p |
|---|---|---|---|---|
| Li 21.5 | 13.93 (11.98–16.19) | 16.06 (12.55–20.57) | 0.87 (0.65–1.15) | 0.3189 |
| 14.92 ± 6.14 | 20.52 ± 18.51 | |||
| 13.44 (6.04) | 15.25 (13.44) | |||
| Be | 0.0009 (0007–0.0012) | 0.0029 (0.0015–0.0056) | 0.31 (0.16–0.62) | 0.0015 |
| 0.0011 ± 0009 | 0.041 ± 0.15 | |||
| 0.0008 (0004) | 0.0022 (0.0059) | |||
| Al 2.04 | 10.62 (7.97–14.15) | 15.62 (10.22–23.88) | 0.68 (0.41–1.12) | 0.1292 |
| 14.23 ± 15.39 | 97.42 ± 438.12 | |||
| 9.59 (6.42) | 11.25 (10.62) | |||
| Ti | 30.87 (25.19–37.82) | 31.13 (23.07–42.02) | 0.99 (0.69–1.41) | 0.9612 |
| 34.49 ± 16.15 | 39.95 ± 25.09 | |||
| 29.40 (25.48) | 34.43 (29.65) | |||
| V 0.22 | 1.46 (0.73–2.93) | 1.53 (0.91–2.57) | 0.96 (0.42–2.19) | 0.9178 |
| 3.33 ± 3.49 | 2.95 ± 3.08 | |||
| 2.07 (3.24) | 1.92 (2.67) | |||
| Cr 0.11 | 0.18 (0.14–0.22) | 0.36 (0.24–0.54) | 0.49 (0.32–0.78) | 0.0033 |
| 0.20 ± 0.13 | 1.00 ± 2.91 | |||
| 0.16 (0.08) | 0.29 (0.39) | |||
| Mn <0.043 | 0.12 (0.06–0.21) | 0.39 (0.19–0.81) | 0.29 (0.11–0.78) | 0.0145 |
| 0.28 ± 0.37 | 3.36 ± 8.65 | |||
| 0.11 (0.27) | 0.29 (1.05) | |||
| Co 0.2 | 1.18 (0.73–1.92) | 2.75 (1.98–3.82) | 0.43 (0.25–0.75) | 0.0034 |
| 2.45 ± 3.58 | 4.31 ± 5.08 | |||
| 0.97 (1.84) | 2.24 (3.46) | |||
| Ni 1.79 | 1.19 (0.94–1.51) | 1.49 (1.16–1.92) | 0.79 (0.57–1.13) | 0.1967 |
| 1.39 ± 0.75 | 1.87 ± 1.33 | |||
| 1.18 (1.24) | 1.57 (1.44) | |||
| Cu 6.84 | 7.38 (6.45–8.43) | 25.76 (19.93–33.29) | 0.29 (0.22–0.38) | <0.0001 |
| 7.78 ± 2.81 | 32.48 ± 23.22 | |||
| 7.06 (2.48) | 28.52 (25.87) | |||
| Zn 246 | 221.10 (176.70–276.07) | 1033.80 (818.5–1305.7) | 0.21 (0.15–0.29) | <0.0001 |
| 250.38 ± 116.76 | 1256.08 ± 820.55 | |||
| 256.67 (167.09) | 1007.14 (821.22) | |||
| As 13.7 | 20.36 (14.99–27.65) | 27.06 (20.41–35.89) | 0.75 (0.49–1.13) | 0.1689 |
| 27.19 ± 24.59 | 35.21 ± 24.55 | |||
| 19.97 (20.48) | 29.06 (29.68) | |||
| Se 21.6 | 17.42 (15.48–19.59) | 24.53 (21.70–27.73) | 0.71 (0.59–0.84) | 0.0001 |
| 18.09 ± 5.07 | 25.98 ± 9.23 | |||
| 18.54 (5.63) | 22.85 (12.10) | |||
| Mo 29.8 | 60.46 (41.95–87.13) | 55.17 (38.23–79.60) | 1.09 (0.65–1.83) | 0.4882 |
| 84.94 ± 74.05 | 92.35 ± 137.29 | |||
| 65.18 ± 38.00 | 49.98 (54.58) | |||
| Cd 0.22 | 0.61 (0.48–0.76) | 1.48 (1.15–1.90) | 0.41 (0.29–0.58) | <0.0001 |
| 0.72 ± 0.54 | 1.94 ± 1.96 | |||
| 0.57 (0.41) | 1.44 (1.29) | |||
| Sn 0.35 | 0.23 (0.19–0.29) | 3.59 (1.79–7.19) | 0.07 (0.03–0.13) | <0.0001 |
| 0.26 ± 0.14 | 11.24 ± 14.55 | |||
| 0.21 (0.11) | 6.86 (16.66) | |||
| Sb 0.04 | 0.05 (0.04–0.06) | 0.08 (0.05–0.11) | 0.64 (0.42–0.97) | 0.0351 |
| 0.05 ± 0.04 | 0.11 ± 0.09 | |||
| 0.046 (0.019) | 0.08 (0.14) | |||
| Te 0.14 | 0.23 (0.19–0.27) | 0.23 (0.19–0.28) | 1.01 (0.78–1.32) | 0.9417 |
| 0.25 ± 0.09 | 0.27 ± 0.17 | |||
| 0.21 (0.14) | 0.21 (0.24) | |||
| Ba 1.86 | 1.27 (0.94–1.72) | 1.65 (1.04–2.57) | 0.77 (0.45–1.33) | 0.3442 |
| 1.61 ± 1.19 | 3.54 ± 4.96 | |||
| 1.16 (0.74) | 1.89 (3.98) | |||
| Tl 0.18 | 0.14 (0.11–0.17) | 0.23 (0.18–0.29) | 0.59 (0.42–0.83) | 0.0031 |
| 0.16 ± 0.11 | 0.29 ± 0.19 | |||
| 0.13 (0.05) | 0.25 (0.16) | |||
| Pb 1.78 | 1.76 (1.38–2.26) | 1.24 (0.90–1.69) | 1.42 (0.95–2.15) | 0.0888 |
| 2.09 ± 1.28 | 1.91 ± 2.50 | |||
| 1.68 (1.28) | 1.24 (1.22) | |||
| U <0.007 | 0.008 (0.006–0.0105) | 0.02 (0.016–0.031) | 0.37 (0.24–0.55) | <0.0001 |
| 0.11 ± 0.01 | 0.03 ± 0.04 | |||
| 0.007 (0.004) | 0.02 (0.03) |
| Variable | Coefficient | SE | Wald 95% Confidence Limits | Wald X2 | p-Value | |
|---|---|---|---|---|---|---|
| Chromium model | ||||||
| Intercept | −2.32 | 1.29 | −4.85 | 0.21 | 3.24 | 0.0717 |
| Chromium | 4.69 | 2.09 | 0.60 | 8.78 | 5.05 | 0.0246 |
| Age | 0.02 | 0.02 | −0.02 | 0.07 | 0.90 | 0.3435 |
| Male sex | 1.04 | 0.69 | −0.32 | 2.40 | 2.24 | 0.1344 |
| Education X | −0.86 | 0.65 | −2.13 | 0.41 | 1.76 | 0.1847 |
| Copper model | ||||||
| Intercept | −5.87 | 2.15 | −10.10 | −1.65 | 7.44 | 0.0064 |
| Copper | 0.32 | 0.11 | 0.10 | 0.54 | 8.32 | 0.0039 |
| Age | 0.03 | 0.04 | −0.04 | 0.10 | 0.69 | 0.4058 |
| Male sex | 0.88 | 0.95 | −0.98 | 2.74 | 0.85 | 0.3556 |
| Education | 0.08 | 0.94 | −1.76 | 1.93 | 0.01 | 0.9313 |
| Zinc model | ||||||
| Intercept | −4.88 | 2.65 | −10.07 | 0.29 | 3.41 | 0.0648 |
| Zinc | 0.02 | 0.01 | 0.01 | 0.03 | 9.45 | 0.0021 |
| Age | −0.05 | 0.06 | −0.17 | 0.07 | 0.67 | 0.4146 |
| Male sex | −0.81 | 1.26 | −3.28 | 1.67 | 0.41 | 0.5231 |
| Education | 0.95 | 1.31 | −1.62 | 3.52 | 0.53 | 0.4684 |
| Selenium model | ||||||
| Intercept | −7.39 | 2.41 | −12.13 | −2.66 | 9.38 | 0.0022 |
| Selenium | 0.24 | 0.08 | 0.09 | 0.39 | 9.84 | 0.0017 |
| Age | 0.05 | 0.03 | −0.01 | 0.10 | 3.11 | 0.0780 |
| Male sex | 1.51 | 0.78 | −0.02 | 3.05 | 3.74 | 0.0533 |
| High education | −1.28 | 0.74 | −2.73 | 0.16 | 3.03 | 0.0818 |
| Cadmium model | ||||||
| Intercept | −2.19 | 1.35 | −4.83 | 0.46 | 2.63 | 0.1051 |
| Cadmium | 2.47 | 0.87 | 0.78 | 4.17 | 8.19 | 0.0042 |
| Age | −0.01 | 0.03 | −0.07 | 0.05 | 0.16 | 0.6850 |
| Male sex | 1.39 | 0.77 | −0.11 | 2.91 | 3.29 | 0.0697 |
| Education | −0.83 | 0.70 | −2.20 | 0.55 | 1.38 | 0.2401 |
| Antimony model | ||||||
| Intercept | −2.39 | 1.33 | −4.99 | −0.22 | 3.23 | 0.0723 |
| Antimony | 14.59 | 6.11 | 2.61 | 26.56 | 5.70 | 0.0170 |
| Age | 0.03 | 0.02 | −00.1 | 0.08 | 1.97 | 0.1602 |
| Male sex | 0.56 | 0.64 | −0.69 | 1.82 | 0.78 | 0.3785 |
| Education | −0.77 | 0.65 | −2.04 | 0.51 | 1.39 | 0.2380 |
| Thallium model | ||||||
| Intercept | −3.13 | 1.40 | −5.88 | −0.38 | 4.96 | 0.0259 |
| Thallium | 8.51 | 3.32 | 2.01 | 15.01 | 6.58 | 0.0103 |
| Age | 0.03 | 0.03 | −0.02 | 0.08 | 1.43 | 0.2324 |
| Male sex | 1.36 | 0.74 | −0.08 | 2.80 | 3.42 | 0.0645 |
| Education | −1.25 | 0.69 | −2.59 | 0.09 | 3.33 | 0.0681 |
| Uranium model | ||||||
| Intercept | −3.78 | 1.62 | −6.95 | −0.61 | 5.48 | 0.0193 |
| Uranium | 85.43 | 35.37 | 16.11 | 154.74 | 5.83 | 0.0157 |
| Age | 0.04 | 0.03 | −0.01 | 0.09 | 2.62 | 0.1055 |
| Male sex | 1.59 | 0.78 | 0.07 | 3.10 | 4.19 | 0.0408 |
| Education | −0.92 | 0.71 | −2.32 | 0.47 | 1.68 | 0.1944 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malamba-Lez, D.; Tshala-Katumbay, D.; Bito, V.; Rigo, J.-M.; Kipenge Kyandabike, R.; Ngoy Yolola, E.; Katchunga, P.; Koba-Bora, B.; Ngoy-Nkulu, D. Concurrent Heavy Metal Exposures and Idiopathic Dilated Cardiomyopathy: A Case-Control Study from the Katanga Mining Area of the Democratic Republic of Congo. Int. J. Environ. Res. Public Health 2021, 18, 4956. https://doi.org/10.3390/ijerph18094956
Malamba-Lez D, Tshala-Katumbay D, Bito V, Rigo J-M, Kipenge Kyandabike R, Ngoy Yolola E, Katchunga P, Koba-Bora B, Ngoy-Nkulu D. Concurrent Heavy Metal Exposures and Idiopathic Dilated Cardiomyopathy: A Case-Control Study from the Katanga Mining Area of the Democratic Republic of Congo. International Journal of Environmental Research and Public Health. 2021; 18(9):4956. https://doi.org/10.3390/ijerph18094956
Chicago/Turabian StyleMalamba-Lez, Didier, Désire Tshala-Katumbay, Virginie Bito, Jean-Michel Rigo, Richie Kipenge Kyandabike, Eric Ngoy Yolola, Philippe Katchunga, Béatrice Koba-Bora, and Dophra Ngoy-Nkulu. 2021. "Concurrent Heavy Metal Exposures and Idiopathic Dilated Cardiomyopathy: A Case-Control Study from the Katanga Mining Area of the Democratic Republic of Congo" International Journal of Environmental Research and Public Health 18, no. 9: 4956. https://doi.org/10.3390/ijerph18094956
APA StyleMalamba-Lez, D., Tshala-Katumbay, D., Bito, V., Rigo, J.-M., Kipenge Kyandabike, R., Ngoy Yolola, E., Katchunga, P., Koba-Bora, B., & Ngoy-Nkulu, D. (2021). Concurrent Heavy Metal Exposures and Idiopathic Dilated Cardiomyopathy: A Case-Control Study from the Katanga Mining Area of the Democratic Republic of Congo. International Journal of Environmental Research and Public Health, 18(9), 4956. https://doi.org/10.3390/ijerph18094956






