Cancer Incidence and Mortality among Petroleum Industry Workers and Residents Living in Oil Producing Communities: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Information Sources
2.3. Assessment of Eligibility, Quality and Data Extraction
2.4. Statistical Analyses
3. Results
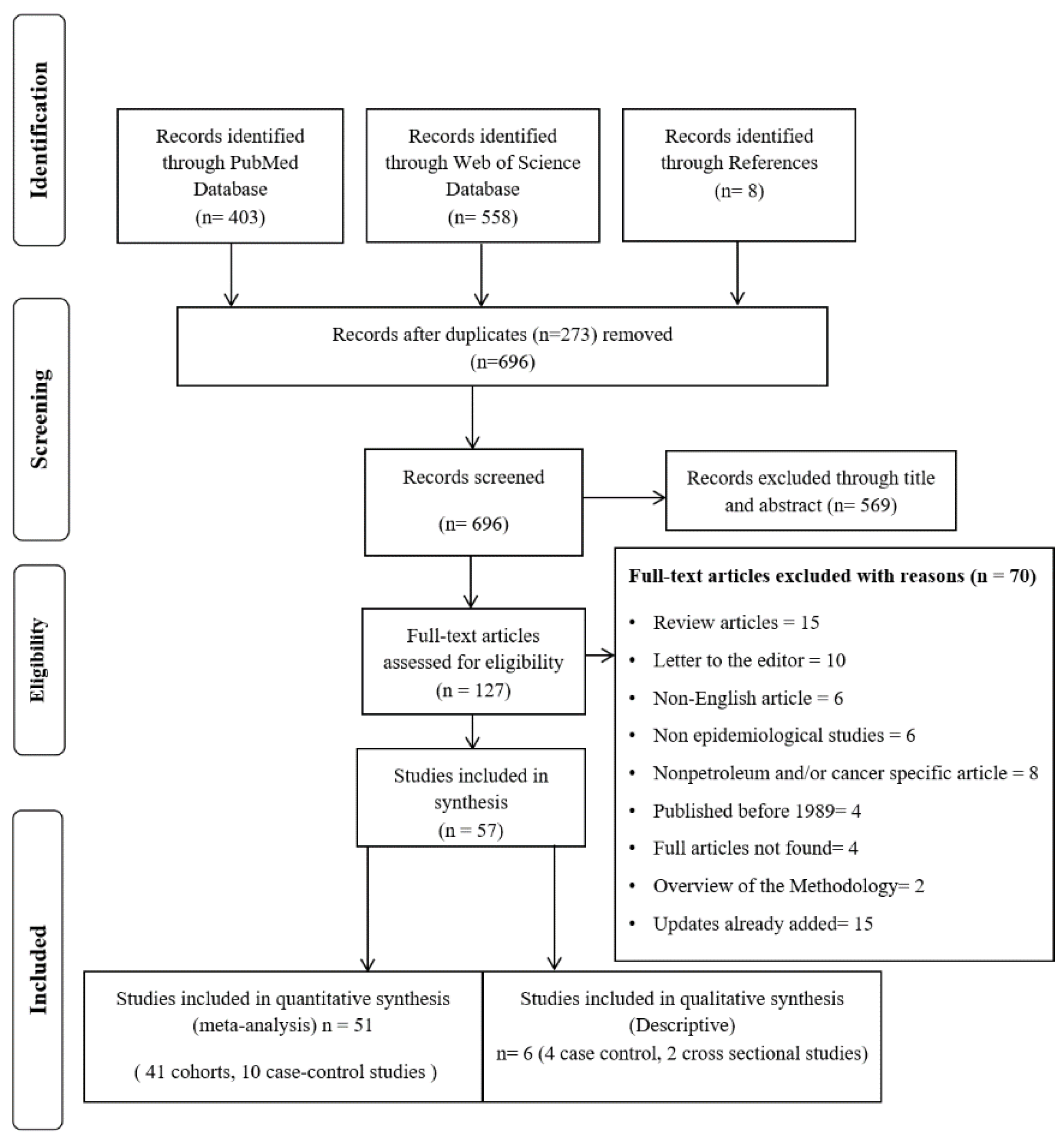
3.1. Search Strategy Outcome
3.2. Study Characteristics
3.3. Assessment of Risk of Bias
3.4. Review and Quantitative Analyses of Petroleum Workers
3.4.1. Respiratory Cancers
Lung Cancer
Mesothelioma
3.4.2. Skin Cancers
Skin Melanoma
Skin Cancer (Non-Specific)
3.4.3. Urinary Tract Cancers
Urinary Bladder Cancer
Kidney Cancer
3.4.4. Reproductive System Cancers
Prostate Cancer
Testicular Cancer
Breast Cancer
3.4.5. Digestive and Accessory Digestive organ Cancers
Esophageal and Gastric Cancers
Colon and Rectum Cancers
Liver, Pancreas and Gall Bladder Cancers
3.4.6. Central Nervous System
Brain Cancer
3.4.7. Hematological Cancers
Hodgkin’s Lymphoma
Non-Hodgkin’s Lymphoma
Multiple Myeloma
Leukaemia and Its Subtypes
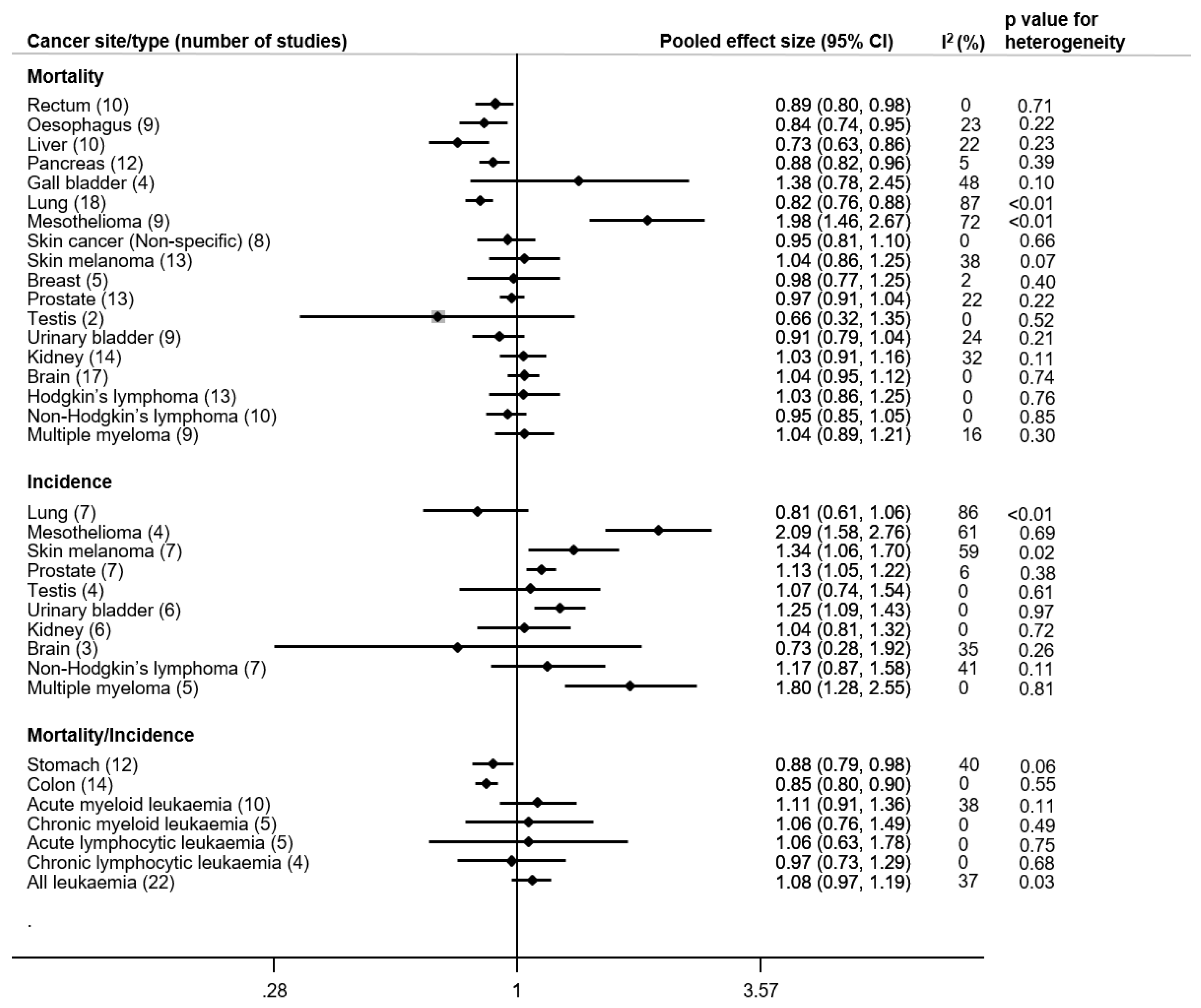
3.4.8. Stratified/Subgroup Analysis
3.5. Review and Quantitative Analyses of Residents Living Near Petroleum Facilities
3.5.1. Solid Cancers
3.5.2. Hematological Cancers
3.6. Review and Quantitative Analyses of Case-Control Studies of Workers and Residents Living Near Petroleum Facilities
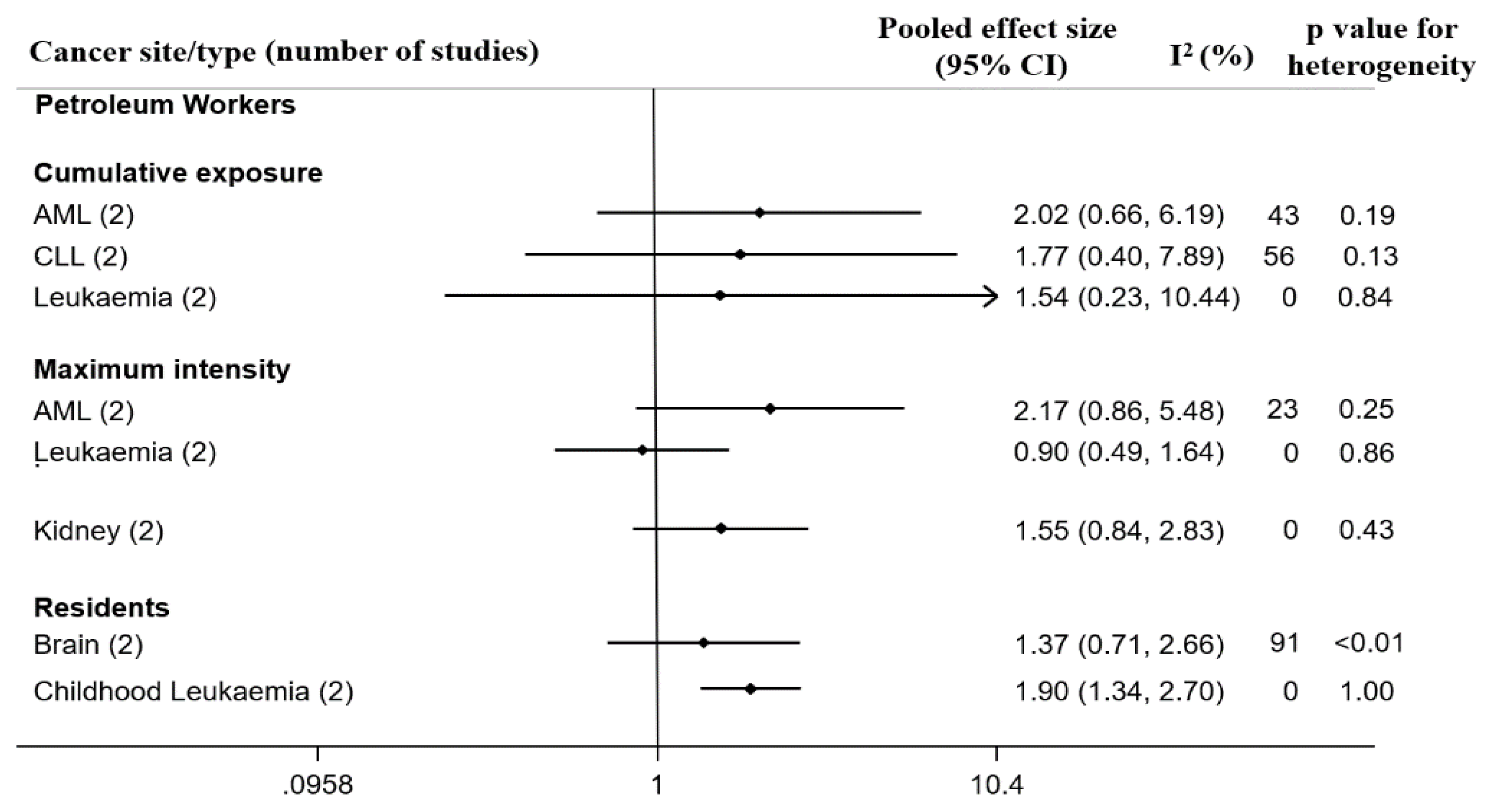
3.6.1. Petroleum Workers
Hematopoietic Cancers
Solid Cancers
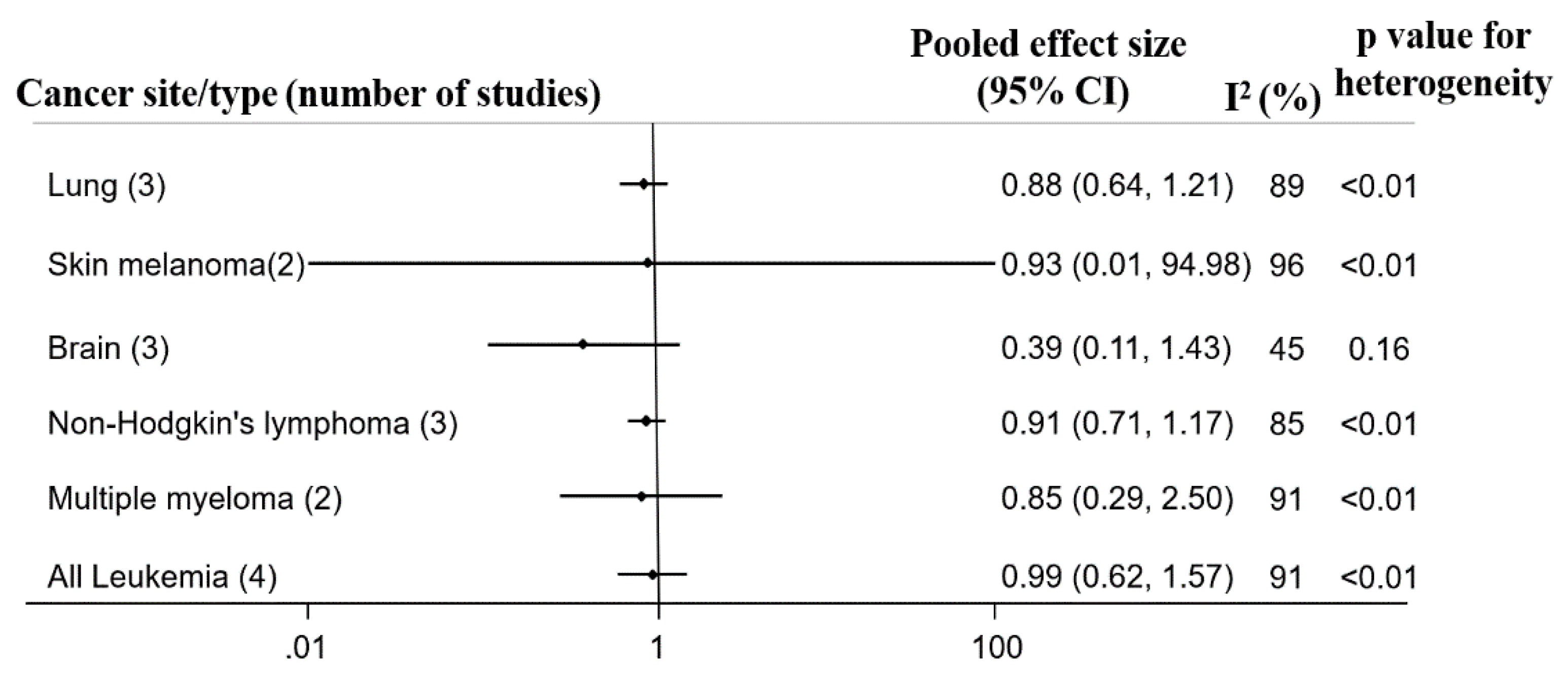
3.6.2. Residents Living Near Petroleum Facilities
Childhood Leukaemia and Other Hematopoietic Cancers
Solid Cancers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Disclaimer
References
- Vătavu, S.; Lobonț, O.-R.; Para, I.; Pelin, A. Addressing oil price changes through business profitability in oil and gas industry in the United Kingdom. PLoS ONE 2018, 13, e0199100. [Google Scholar] [CrossRef]
- Henry, B.; Laitala, K.; Klepp, I.G. Microfibres from apparel and home textiles: Prospects for including microplastics in environmental sustainability assessment. Sci. Total Environ. 2019, 652, 483–494. [Google Scholar] [CrossRef]
- Aksha, S.; Preeti, S.; Ajay, S.; Richa, T.; Aparna, D. Hazardous Effects of Petrochemical Industries: A Review. Recent Adv. Petrochem. Sci. 2017, 3, 25–27. [Google Scholar] [CrossRef]
- IARC. List of Classifications by Cancer Sites with Sufficient or Limited Evidence in Humans; IARC: Lyon, France, 2019; Volume 1–125. [Google Scholar]
- Field, R.A.; Soltis, J.; Murphy, S. Air quality concerns of unconventional oil and natural gas production. Environ. Sci. Process. Impacts 2014, 16, 954–969. [Google Scholar] [CrossRef]
- Nelson, T.P. An examination of historical air pollutant emissions from us petroleum refineries. Environ. Prog. Sustain. Energy 2013, 32, 425–432. [Google Scholar] [CrossRef]
- Tuccella, P.; Thomas, J.L.; Law, K.S.; Raut, J.C.; Marelle, L.; Roiger, A.; Weinzierl, B.; van der Gon, H.; Schlager, H.; Onishi, T. Air pollution impacts due to petroleum extraction in the Norwegian Sea during the ACCESS aircraft campaign. Elem. Sci. Anthr. 2017, 5. [Google Scholar] [CrossRef]
- Tuomi, T.; Veijalainen, H.; Santonen, T. Managing Exposure to Benzene and Total Petroleum Hydrocarbons at Two Oil Refineries 1977–2014. Int. J. Environ. Res. Public Health 2018, 15, 197. [Google Scholar] [CrossRef]
- Gun, R.; Pratt, N.L.; Roder, D.M.; Ryan, P. Asbestos-related cancers in refinery workers in the Australian petroleum industry. Arch. Environ. Occup. Health 2006, 61, 11–16. [Google Scholar] [CrossRef]
- Schnatter, A.R.; Chen, M.; DeVilbiss, E.A.; Lewis, R.J.; Gallagher, E.M. Systematic Review and Meta-Analysis of Selected Cancers in Petroleum Refinery Workers. J. Occup. Environ. Med. 2018, 60, e329–e342. [Google Scholar] [CrossRef]
- Jephcote, C.; Brown, D.; Verbeek, T.; Mah, A. A systematic review and meta-analysis of haematological malignancies in residents living near petrochemical facilities. Environ. Health 2020, 19, 53. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; The, P.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Higgins, J.T.J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. (Eds.) Cochrane, 2019. In Cochrane Handbook for Systematic Reviews of Interventions, 2nd ed.; Wiley-Blackwell: Chichester, UK, 2019. [Google Scholar]
- Moola, S.; Munn, Z.; Tufanaru, C.; Aromataris, E.; Sears, K.; Sfetcu, R.; Currie, M.; Qureshi, R.; Mattis, P.; Lisy, K.; et al. Systematic Reviews of Etiology and Risk. JBI Manual for Evidence Synthesis. Aromataris, E., Munn, Z., Eds.; 2020. Available online: https://synthesismanual.jbi.global (accessed on 9 March 2021). [CrossRef]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Shi, L.; Chu, H.; Murad, M.H. The magnitude of small-study effects in the Cochrane Database of Systematic Reviews: An empirical study of nearly 30,000 meta-analyses. BMJ Evid. Based Med. 2020, 25, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Smith, G.D.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.; Egger, M.; Smith, G.D. Systematic reviews in health care: Investigating and dealing with publication and other biases in meta-analysis. BMJ 2001, 323, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Christie, D.; Robinson, K.; Gordon, I.; Bisby, J. A prospective study in the Australian petroleum industry. II. Incidence of cancer. Br. J. Ind. Med. 1991, 48, 511–514. [Google Scholar] [CrossRef][Green Version]
- Gun, R.T.; Pratt, N.L.; Griffith, E.C.; Adams, G.G.; Bisby, J.A.; Robinson, K.L. Update of a prospective study of mortality and cancer incidence in the Australian petroleum industry. Occup. Environ. Med. 2004, 61, 150–156. [Google Scholar] [CrossRef]
- Gun, R.T.; Pratt, N.; Ryan, P.; Roder, D. Update of mortality and cancer incidence in the Australian petroleum industry cohort. Occup. Environ. Med. 2006, 63, 476–481. [Google Scholar] [CrossRef] [PubMed]
- Schnatter, A.R.; Katz, A.M.; Nicolich, M.J.; Theriault, G. A retrospective mortality study among Canadian petroleum marketing and distribution workers. Environ. Health Perspect. 1993, 101, 85–99. [Google Scholar] [CrossRef]
- Lewis, R.J.; Schnatter, A.R.; Drummond, I.; Murray, N.; Thompson, F.S.; Katz, A.M.; Jorgensen, G.; Nicolich, M.J.; Dahlman, D.; Theriault, G. Mortality and cancer morbidity in a cohort of Canadian petroleum workers. Occup. Environ. Med. 2003, 60, 918–928. [Google Scholar] [CrossRef]
- Schnatter, A.R.; Nicolich, M.J.; Lewis, R.J.; Thompson, F.L.; Dineen, H.K.; Drummond, I.; Dahlman, D.; Katz, A.M.; Theriault, G. Lung cancer incidence in Canadian petroleum workers. Occup. Environ. Med. 2012, 69, 877–882. [Google Scholar] [CrossRef] [PubMed]
- Schnatter, A.R.; Wojcik, N.C.; Jorgensen, G. Mortality Update of a Cohort of Canadian Petroleum Workers. J. Occup. Environ. Med. 2019, 61, 225–238. [Google Scholar] [CrossRef]
- Gennaro, V.; Ceppi, M.; Boffetta, P.; Fontana, V.; Perrotta, A. Pleural mesothelioma and asbestos exposure among Italian oil refinery workers. Scand. J. Work Environ. Health 1994, 20, 213–215. [Google Scholar] [CrossRef]
- Consonni, D.; Pesatori, A.C.; Tironi, A.; Bernucci, I.; Zocchetti, C.; Bertazzi, P.A. Mortality study in an Italian oil refinery: Extension of the follow-up. Am. J. Ind. Med. 1999, 35, 287–294. [Google Scholar] [CrossRef]
- Pasetto, R.; Zona, A.; Pirastu, R.; Cernigliaro, A.; Dardanoni, G.; Addario, S.P.; Scondotto, S.; Comba, P. Mortality and morbidity study of petrochemical employees in a polluted site. Environ. Health 2012, 11, 34. [Google Scholar] [CrossRef]
- Bonzini, M.; Grillo, P.; Consonni, D.; Cacace, R.; Ancona, C.; Forastiere, F.; Cocco, P.L.; Satta, G.; Boldori, L.; Carugno, M.; et al. Cancer risk in oil refinery workers: A pooled mortality study in Italy. Med. Lav. 2019, 110, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Koh, D.-H.; Kim, T.-W.; Yoon, Y.-H.; Shin, K.-S.; Yoo, S.-W. Lymphohematopoietic Cancer Mortality and Morbidity of Workers in a Refinery/Petrochemical Complex in Korea. Saf. Health Work 2011, 2, 26–33. [Google Scholar] [CrossRef]
- Koh, D.H.; Chung, E.K.; Jang, J.K.; Lee, H.E.; Ryu, H.W.; Yoo, K.M.; Kim, E.A.; Kim, K.S. Cancer incidence and mortality among temporary maintenance workers in a refinery/petrochemical complex in Korea. Int. J. Occup. Environ. Health 2014, 20, 141–145. [Google Scholar] [CrossRef]
- Aas, G.B.; Aagnes, B.; Strand, L.A.; Grimsrud, T.K. Suggested excess of occupational cancers in Norwegian offshore workers: Preliminary results from the Cancer Registry Offshore Cohort. Scand. J. Work Environ. Health 2009, 35, 397–399. [Google Scholar] [CrossRef][Green Version]
- Stenehjem, J.S.; Kjaerheim, K.; Rabanal, K.S.; Grimsrud, T.K. Cancer incidence among 41,000 offshore oil industry workers. Occup. Med. 2014, 64, 539–545. [Google Scholar] [CrossRef][Green Version]
- Kirkeleit, J.; Riise, T.; Bratveit, M.; Moen, B.E. Increased risk of acute myelogenous leukemia and multiple myeloma in a historical cohort of upstream petroleum workers exposed to crude oil. Cancer Causes Control 2008, 19, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Jarvholm, B.; Mellblom, B.; Norrman, R.; Nilsson, R.; Nordlinder, R. Cancer incidence of workers in the Swedish petroleum industry. Occup. Environ. Med. 1997, 54, 686–691. [Google Scholar] [CrossRef] [PubMed]
- Rushton, L. A 39-year follow-up of the UK oil refinery and distribution center studies-results for kidney cancer and leukemia. Environ. Health Perspect. 1993, 101, 77–84. [Google Scholar] [CrossRef]
- Sorahan, T. Mortality of UK oil refinery and petroleum distribution workers, 1951–2003. Occup. Med. 2007, 57, 177–185. [Google Scholar] [CrossRef]
- Satin, K.P.; Wong, O.; Yuan, L.A.; Bailey, W.J.; Newton, K.L.; Wen, C.P.; Swencicki, R.E. A 50-year mortality follow-up of a large cohort of oil refinery workers in Texas. J. Occup. Environ. Med. 1996, 38, 492–506. [Google Scholar] [CrossRef]
- Divine, B.J.; Hartman, C.M.; Wendt, J.K. Update of the Texaco Mortality Study 1947-93: Part II. Analyses of Specific Causes of Death for White Men Employed in Refining, Research, and Petrochemicals. Occup. Environ. Med. 1999, 56, 174–180. [Google Scholar] [CrossRef]
- Gamble, J.F.; Lewis, R.J.; Jorgensen, G. Mortality among three refinery/petrochemical plant cohorts. II. Retirees. J. Occup. Environ. Med. 2000, 42, 730–736. [Google Scholar] [CrossRef]
- Huebner, W.W.; Chen, V.W.; Friedlander, B.R.; Wu, X.C.; Jorgensen, G.; Bhojani, F.A.; Friedmann, C.H.; Schmidt, B.A.; Sales, E.A.; Joy, J.A.; et al. Incidence of lymphohaematopoietic malignancies in a petrochemical industry cohort: 1983-94 follow up. Occup. Environ. Med. 2000, 57, 605–614. [Google Scholar] [CrossRef] [PubMed]
- Sathiakumar, N.; Delzell, E.; Rodu, B.; Beall, C.; Myers, S. Cancer incidence among employees at a petrochemical research facility. J. Occup. Environ. Med. 2001, 43, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Wong, O.; Harris, F.; Rosamilia, K.; Raabe, G.K. Updated mortality study of workers at a petroleum refinery in Torrance, California, 1959 to 1997. J. Occup. Environ. Med. 2001, 43, 1089–1102. [Google Scholar] [CrossRef] [PubMed]
- Satin, K.P.; Bailey, W.J.; Newton, K.L.; Ross, A.Y.; Wong, O. Updated epidemiological study of workers at two California petroleum refineries, 1950–1995. Occup. Environ. Med. 2002, 59, 248–256. [Google Scholar] [CrossRef]
- Tsai, S.S.; Yu, H.S.; Liu, C.C.; Yang, C.Y. Increased incidence of preterm delivery in mothers residing in an industrialized area in Taiwan. J. Toxicol. Environ. Health Part A Curr. Issues 2003, 66, 987–994. [Google Scholar] [CrossRef]
- Huebner, W.W.; Wojcik, N.C.; Rosamilia, K.; Jorgensen, G.; Milano, C.A. Mortality updates (1970–1997) of two refinery/petrochemical plant cohorts at Baton Rouge, Louisiana, and Baytown, Texas. J. Occup. Environ. Med. 2004, 46, 1229–1245. [Google Scholar] [PubMed]
- Buffler, P.A.; Kelsh, M.; Chapman, P.; Wood, S.; Lau, E.; Golembesky, A.; Wood, R.; Kalmes, R.; Brorby, G. Primary brain tumor mortality at a petroleum exploration and extraction research facility. J. Occup. Environ. Med. 2004, 46, 257–270. [Google Scholar] [CrossRef] [PubMed]
- Tsai, S.P.; Chen, V.W.; Fox, E.E.; Wendt, J.K.; Wu, X.C.; Foster, D.E.; Fraser, A.E. Cancer incidence among refinery and petrochemical employees in Louisiana, 1983–1999. Ann. Epidemiol. 2004, 14, 722–730. [Google Scholar] [CrossRef] [PubMed]
- Tsai, S.P.; Ahmed, F.S.; Wendt, J.K.; Foster, D.E.; Donnelly, R.P.; Strawmyer, T.R. A 56-Year Mortality Follow-Up of Texas Petroleum Refinery and Chemical Employees, 1948–2003. J. Occup. Environ. Med. 2007, 49, 557–567. [Google Scholar] [CrossRef]
- Huebner, W.W.; Wojcik, N.C.; Jorgensen, G.; Marcella, S.P.; Nicolich, M.J. Mortality patterns and trends among 127,266 U.S.-based men in a petroleum company: Update 1979–2000. J. Occup. Environ. Med. 2009, 51, 1333–1348. [Google Scholar] [CrossRef]
- Divine, B.J.; Hartman, C.M.; Wendt, J.K. Update of the Texaco mortality study 1947-93: Part I. Analysis of overall patterns of mortality among refining, research, and petrochemical workers. Occup. Environ. Med. 1999, 56, 167. [Google Scholar] [CrossRef]
- Wong, O.; Harris, F.; Rosamilia, K.; Raabe, G.K. An Updated Mortality Study of Workers at a Petroleum Refinery in Beaumont, Texas, 1945 to 1996. J. Occup. Environ. Med. 2001, 43, 384–401. [Google Scholar] [CrossRef]
- Finkelstein, M.M. Asbestos-associated cancers in the Ontario refinery and petrochemical sector. Am. J. Ind. Med. 1996, 30, 610–615. [Google Scholar] [CrossRef]
- Schnatter, A.R.; Armstrong, T.W.; Nicolich, M.J.; Thompson, F.S.; Katz, A.M.; Huebner, W.W.; Pearlman, E.D. Lymphohaematopoietic malignancies and quantitative estimates of exposure to benzene in Canadian petroleum distribution workers. Occup. Environ. Med. 1996, 53, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Anttila, A.; Pokhrel, A.; Heikkila, P.; Viinanen, R.; Pukkala, E. Kidney cancer risk in oil refining in Finland: A nested case-referent study. J. Occup. Environ. Med. 2015, 57, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Rushton, L.; Schnatter, A.R.; Tang, G.; Glass, D.C. Acute myeloid and chronic lymphoid leukaemias and exposure to low-level benzene among petroleum workers. Br. J. Cancer 2014, 110, 783–787. [Google Scholar] [CrossRef] [PubMed]
- Glass, D.C.; Schnatter, A.R.; Tang, G.; Irons, R.D.; Rushton, L. Risk of myeloproliferative disease and chronic myeloid leukaemia following exposure to low-level benzene in a nested case-control study of petroleum workers. Occup. Environ. Med. 2014, 71, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Stenehjem, J.S.; Kjaerheim, K.; Bratveit, M.; Samuelsen, S.O.; Barone-Adesi, F.; Rothman, N.; Lan, Q.; Grimsrud, T.K. Benzene exposure and risk of lymphohaematopoietic cancers in 25,000 offshore oil industry workers. Br. J. Cancer 2015, 112, 1603–1612. [Google Scholar] [CrossRef] [PubMed]
- Rushton, L.; Romaniuk, H. A case-control study to investigate the risk of leukaemia associated with exposure to benzene in petroleum marketing and distribution workers in the United Kingdom. Occup. Environ. Med. 1997, 54, 152–166. [Google Scholar] [CrossRef]
- Poole, C.; Dreyer, N.A.; Satterfield, M.H.; Levin, L.; Rothman, K.J. Kidney cancer and hydrocarbon exposures among petroleum refinery workers. Environ. Health Perspect. 1993, 101, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Hurtig, A.K.; San Sebastian, M. Geographical differences in cancer incidence in the Amazon basin of Ecuador in relation to residence near oil fields. Int. J. Epidemiol. 2002, 31, 1021–1027. [Google Scholar] [CrossRef][Green Version]
- Hurtig, A.K.; San Sebastian, M. Incidence of childhood leukemia and oil exploitation in the Amazon basin of Ecuador. Int. J. Occup. Environ. Health 2004, 10, 245–250. [Google Scholar] [CrossRef]
- Kelsh, M.A.; Morimoto, L.; Lau, E. Cancer mortality and oil production in the Amazon Region of Ecuador, 1990-2005. Int. Arch. Occup. Environ. Health 2009, 82, 381–395. [Google Scholar] [CrossRef]
- Ramis, R.; Diggle, P.; Boldo, E.; Garcia-Perez, J.; Fernandez-Navarro, P.; Lopez-Abente, G. Analysis of matched geographical areas to study potential links between environmental exposure to oil refineries and non-Hodgkin lymphoma mortality in Spain. Int. J. Health Geogr. 2012, 11, 4. [Google Scholar] [CrossRef] [PubMed]
- Barregard, L.; Holmberg, E.; Sallsten, G. Leukaemia incidence in people living close to an oil refinery. Environ. Res. 2009, 109, 985–990. [Google Scholar] [CrossRef] [PubMed]
- Sans, S.; Elliott, P.; Kleinschmidt, I.; Shaddick, G.; Pattenden, S.; Walls, P.; Grundy, C.; Dolk, H. Cancer incidence and mortality near the Baglan Bay petrochemical works, South Wales. Occup. Environ. Med. 1995, 52, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.H.; Park, M.S.; Ha, M.; Hur, J.I.; Cheong, H.K. Cancer Incidence Trend in the Hebei Spirit Oil Spill Area, from 1999 to 2014: An Ecological Study. Int. J. Environ. Res. Public Health 2018, 15, 1006. [Google Scholar] [CrossRef]
- Lyons, R.A.; Monaghan, S.P.; Heaven, M.; Littlepage, B.N.; Vincent, T.J.; Draper, G.J. Incidence of leukaemia and lymphoma in young people in the vicinity of the petrochemical plant at Baglan Bay, South Wales, 1974 to 1991. Occup. Environ. Med. 1995, 52, 225–228. [Google Scholar] [CrossRef] [PubMed]
- Tsai, S.S.; Tiao, M.M.; Kuo, H.W.; Wu, T.N.; Yang, C.Y. Association of bladder cancer with residential exposure to petrochemical air pollutant emissions in Taiwan. J. Toxicol. Environ. Health Part A 2009, 72, 53–59. [Google Scholar] [CrossRef]
- Weng, H.H.; Tsai, S.S.; Chiu, H.F.; Wu, T.N.; Yang, C.Y. Association of childhood leukemia with residential exposure to petrochemical air pollution in taiwan. Inhal. Toxicol. 2008, 20, 31–36. [Google Scholar] [CrossRef]
- Liu, C.C.; Chen, C.C.; Wu, T.N.; Yang, C.Y. Association of brain cancer with residential exposure to petrochemical air pollution in Taiwan. J. Toxicol. Environ. Health Part A 2008, 71, 310–314. [Google Scholar] [CrossRef]
- Yu, C.L.; Wang, S.F.; Pan, P.C.; Wu, M.T.; Ho, C.K.; Smith, T.J.; Li, Y.; Pothier, L.J.; Christiani, D.C. No association between residential exposure to petrochemicals and brain tumor risk. Cancer Epidemiol. Biomark. Prev. 2005, 14, 3007–3009. [Google Scholar] [CrossRef][Green Version]
- Micheli, A.; Meneghini, E.; Mariottini, M.; Baldini, M.; Baili, P.; Di Salvo, F.; Sant, M. Risk of death for hematological malignancies for residents close to an Italian petrochemical refinery: A population-based case-control study. Cancer Causes Control 2014, 25, 1635–1644. [Google Scholar] [CrossRef][Green Version]
- McKenzie, L.M.; Allshouse, W.B.; Byers, T.E.; Bedrick, E.J.; Serdar, B.; Adgate, J.L. Childhood hematologic cancer and residential proximity to oil and gas development. PLoS ONE 2017, 12, e0170423. [Google Scholar] [CrossRef] [PubMed]
- Wong, O.; Raabe, G.K. A critical review of cancer epidemiology in the petroleum industry, with a meta-analysis of a combined database of more than 350,000 workers. Regul. Toxicol. Pharmacol. 2000, 32, 78–98. [Google Scholar] [CrossRef] [PubMed]
- IARC. Asbestos (Chrysotile, Amosite, Crocidolite, Tremolite, Actinolite and Anthophyllite) IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. In Arsenic, Metals, Fibres and Dusts. Lyon (FR): International Agency for Research on Cancer (IARC Monographs on the Evaluation of Carcinogenic Risks to Humans); IARC: Lyon, France, 2012; Volume 100C. [Google Scholar]
- Schüz, J.; Bukhtiyarov, I.; Olsson, A.; Moissonnier, M.; Ostroumova, E.; Feletto, E.; Schonfeld, S.J.; Byrnes, G.; Tskhomariia, I.; McCormack, V.; et al. Occupational cohort study of current and former workers exposed to chrysotile in mine and processing facilities in Asbest, the Russian Federation: Cohort profile of the Asbest Chrysotile Cohort study. PLoS ONE 2020, 15, e0236475. [Google Scholar] [CrossRef] [PubMed]
- Røe, O.D.; Stella, G.M. Malignant pleural mesothelioma: History, controversy and future of a manmade epidemic. Eur. Respir. Rev. 2015, 24, 115. [Google Scholar] [CrossRef]
- Gennaro, V.; Finkelstein, M.M.; Ceppi, M.; Fontana, V.; Montanaro, F.; Perrotta, A.; Puntoni, R.; Silvano, S. Mesothelioma and lung tumors attributable to asbestos among petroleum workers. Am. J. Ind. Med. 2000, 37, 275–282. [Google Scholar] [CrossRef]
- Redondo, P.; Ribeiro, M.; Lopes, M.; Borges, M.; Gonçalves, F.R. Holistic view of patients with melanoma of the skin: How can health systems create value and achieve better clinical outcomes? Ecancermedicalscience 2019, 13, 959. [Google Scholar] [CrossRef]
- IARC. Occupational exposures in petroleum refining; crude oil and major petroleum fuels. IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. IARC Monogr. Eval. Carcinog. Risks Hum. 1989, 45, 1–322. [Google Scholar]
- De Roos, A.J.; Spinelli, J.; Brown, E.B.; Atanackovic, D.; Baris, D.; Bernstein, L.; Bhatti, P.; Camp, N.J.; Chiu, B.C.; Clavel, J.; et al. Pooled study of occupational exposure to aromatic hydrocarbon solvents and risk of multiple myeloma. Occup. Environ. Med. 2018, 75, 798–806. [Google Scholar] [CrossRef] [PubMed]
- Espina, C.; Straif, K.; Friis, S.; Kogevinas, M.; Saracci, R.; Vainio, H.; Schüz, J. European Code against Cancer 4th Edition: Environment, occupation and cancer. Cancer Epidemiol. 2015, 39, S84–S92. [Google Scholar] [CrossRef] [PubMed]
- IARC. Outdoor Air Pollution. In IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 2016; Volume 109. [Google Scholar]
- IARC. Beryllium, Cadmium, Mercury, and Exposures in the Glass Manufacturing Industry. In IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 1993; Volume 58, pp. 1–417. [Google Scholar]
- Buhmeida, A.; Pyrhönen, S.; Laato, M.; Collan, Y. Prognostic factors in prostate cancer. Diagn. Pathol. 2006, 1, 4. [Google Scholar] [CrossRef]
- Lideikaitė, A.; Mozūraitienė, J.; Letautienė, S. Analysis of prognostic factors for melanoma patients. Acta Med. Litu. 2017, 24, 25–34. [Google Scholar] [CrossRef]
- Vahedian-Ardakani, H.-A.; Moghimi, M.; Shayestehpour, M.; Doosti, M.; Alimohammadi, F. Survival Rate of Patients with Bladder Cancer in Yazd, Central Province of Iran. Int J. Cancer Manag. 2018, 11, e61436. [Google Scholar] [CrossRef]
- Mosallanejad, Z.; Fakhri, Y.; Ferrante, M.; Zandsalimi, Y.; Amirhajeloo, L.R.; Amanidaz, N.; Moradi, B.; Keramati, H. Association between benzene exposure and childhood leukemia: A systematic review and meta-analysis. Int. J. Pharm. Technol. 2016, 8, 4640–4652. [Google Scholar]
- Boonhat, H.; Lin, R.-T. Association between leukemia incidence and mortality and residential petrochemical exposure: A systematic review and meta-analysis. Environ. Int. 2020, 145, 106090. [Google Scholar] [CrossRef] [PubMed]
- Schüz, J.; Espina, C.; Villain, P.; Herrero, R.; Leon, M.E.; Minozzi, S.; Romieu, I.; Segnan, N.; Wardle, J.; Wiseman, M.; et al. European Code against Cancer 4th Edition: 12 ways to reduce your cancer risk. Cancer Epidemiol. 2015, 39, S1–S10. [Google Scholar] [CrossRef] [PubMed]
- IARC. World Cancer Report: Cancer Research for Cancer Prevention; Wild, C.P., Weiderpass, E., Stewart, B.W., Eds.; IARC: Lyon, France, 2020. [Google Scholar]
- Shah, D. Healthy worker effect phenomenon. Indian J. Occup. Environ. Med. 2009, 13, 77–79. [Google Scholar] [CrossRef] [PubMed]
- Dienye, P.O.; Akani, A.B.; Itimi, K. Uses of crude oil as traditional medicine: A survey of mothers in a rural clinic in South-south Nigeria. Rural Remote Health 2012, 12, 1858. [Google Scholar] [PubMed]
- Programme, UNEP United Nations Environment Programme. Environmental Assessment of Ogoniland: Site Factsheets, Executive Summary and Full Report; United Nations Environment Programme: Nairobi, Kenya, 2011. [Google Scholar]
- Rajabi, H.; Mosleh, M.H.; Mandal, P.; Lea-Langton, A.; Sedighi, M. Emissions of volatile organic compounds from crude oil processing—Global emission inventory and environmental release. Sci. Total Environ. 2020, 727, 138654. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, H.; Alkhateeb, T.T.Y.; Furqan, M. Oil sector and CO2 emissions in Saudi Arabia: Asymmetry analysis. Palgrave Commun. 2020, 6, 88. [Google Scholar] [CrossRef]
| First Author, Year [Reference] | Cancer Site/Type | Country | Minimum Employment Time | Follow-Up (Years) | Exposed Population | |
|---|---|---|---|---|---|---|
| Cohort studies: | ||||||
| Christie, 1991 [19] | AML, Brain, Kidney, Leukemia, Lung, Melanoma, MM, NHL, Skin and Urinary bladder | Australia | 5 Years | 9 | >1500 | |
| Gun, 2004 [20] | ALL, AML, Brain, CML, Colon, Rectum, Gall bladder, Kidney, Liver, Lung, Mesothelioma, MM, NHL, Esophagus, Pancreas, Prostate, Skin melanoma, Skin, Stomach, Testis and Urinary bladder | Australia | 5 Years | 16 | 17,163 | |
| Gun, 2006a [9] | Mesothelioma | Australia | 5 Years | 22 | 16,543 | |
| Gun, 2006b * [21] | ALL, AML, Brain, CML, Colon, Gall bladder, Kidney, Leukemia, Liver, Lung, MM, NHL, Esophagus, Pancreas, Prostate, Skin melanoma, Skin, Stomach, Testis and Urinary bladder | Australia | 5 Years | 22 | 17,903 | |
| Schnatter, 1993 [22] | Brain | Canada | 1 Year | 20 | 6672 | |
| Lewis, 2003 [23] | Gall bladder, Kidney, Leukemia, NHL, Prostate and Testis | Canada | 1 Month | 23 | 25,292 | |
| Schnatter, 2012 [24] | Lung (Incidence studies) | Canada | 1 Year | 31 | 17,216 | |
| Schnatter, 2019 [25] | AML, Breast, Colon, HL, Kidney, Leukemia, Liver, Lung, Melanoma, Mesothelioma, NHL, Esophagus, Pancreas, Prostate, Rectum, Stomach and Urinary bladder | Canada | 1 Year | 43 | 29,379 | |
| Gennaro, 1994 [26] | Mesothelioma | Italy | 1 Year | 77 | 2300 | |
| Consonni, 1999 [27] | Brain, HL, Kidney, Leukemia, Lung, NHL, Skin melanoma, Urinary bladder | Italy | 1 Day | 43 | 1583 | |
| Pasetto, 2012 [28] | HL, Leukaemia, Lung, Mesothelioma, MM, NHL and Skin melanoma, | Italy | 10 Years | 43 | 5627 | |
| Bonzini, 2019 [29] | Colon, HL, Liver, Lung, Mesothelioma, MM, NHL, Pancreas, Prostate, Rectum, Skin, Stomach and Urinary bladder | Italy | 1 Year | 63 | 5112 | |
| Koh, 2011 [30] | Prostate, Skin and Urinary bladder | Korea | 1 Day | 16 | 8866 | |
| Koh, 2014 [31] | Brain, Colon, Leukaemia, Liver, Lung, NHL, Esophagus, Pancreas, Prostate and Stomach | Korea | NR | 6 | 14,698 | |
| Aas, 2009 [32] | AML, Leukaemia, Lung, Mesothelioma, Prostate and Skin melanoma | Norway | 20 Days | 41 | 28,000 | |
| Stenehjem, 2014 [33] | Lung, Mesothelioma, Skin melanoma and Urinary bladder | Norway | 20 Days | 11 | 41,140 | |
| Kirkeleit, 2008 [34] | ALL, AML, CLL, CML and MM | Norway | <1 Year | 22 | 27,919 | |
| Jarvholm, 1997 [35] | Brain, Kidney, Leukaemia, Lung, Prostate, and Skin | Sweden | 1 Year | 34 | 4319 | |
| Rushton,1993 [36] | Brain, HL, Kidney, Lung, Leukaemia and MM | UK | 1 Year | 40 | 23,306 | |
| Sorahan, 2007 [37] | AML, Breast, Colon, Gall bladder, HL, Kidney, Leukaemia, Liver, Lung, Mesothelioma, MM, Esophagus, Pancreas, Prostate, Rectum, Skin, Stomach, Testis and Urinary bladder | UK | 1 Year | 53 | 45,032 | |
| Satin, 1996 [38] | AML, Brain, Breast, HL, Kidney, Leukaemia, Lung and Skin melanoma, | USA | 1 Day | 51 | 17,844 | |
| Divine,1999a [39] | Brain, Lung and Mesothelioma | USA | 1 Year | 47 | 28,840 | |
| Gamble, 2000 [40] | Kidney, Leukaemia, Lung, Mesothelioma, Prostate and Skin melanoma | USA | 1 Month | 23 | 6238 | |
| Huebner, 2000 [41] | Leukaemia, MM and NHL | USA | 1 Day | 12 | 8942 | |
| Sathiakumar, 2001 [42] | Brain, Colon, Kidney, Lung, NHL, Prostate, Skin melanoma, Testis and Urinary bladder | USA | NR | 12 | 5641 | |
| Wong, 2001a [43] | Brain, Breast, Colon, HL kidney, Leukaemia, Lung, Esophagus, Prostate, Skin melanoma | USA | 1 Year | 39 | 3328 | |
| Satin, 2002 [44] | ALL, AML, CLL, CML, Colon, HL, Kidney, Leukaemia, Liver, Lung, MM, NHL, Esophagus, Pancreas, Rectum, Skin melanoma and Stomach | USA | 1 Year | 46 | 18,512 | |
| Tsai 2003 [45] | Kidney, Leukaemia, Liver, Lung, Prostate and Rectum | USA | 6 Months | 31 | 4221 | |
| Huebner, 2004 [46] | Brain and Skin melanoma | USA | 1 Month | 28 | 14,644 | |
| Buffler, 2004 [47] | Brain | USA | 6 Months | 32 | 3779 | |
| Tsai, 2004 [48] | Brain, Colon, Kidney, Leukaemia, Lung, MM, NHL, Pancreas, Prostate, Skin melanoma, Stomach and Urinary bladder | USA | >6 Months | 12 | 4639 | |
| Tsai, 2007 [49] | Brain, Colon, HL, Kidney, Leukaemia, Liver, Lung, NHL, Esophagus, Pancreas, Prostate, Rectum, Skin melanoma and Stomach | USA | 3 Months | 56 | 10,621 | |
| Huebner2009 [50] | ALL, AML, Breast, CLL, CML, Colon, HL, Kidney, Leukaemia, Liver, Lung, Mesothelioma, MM, NHL, Esophagus, Pancreas, Prostate, Rectum, Skin melanoma, Skin, Stomach and Urinary bladder | USA | 1 Day | 22 | 127,266 | |
| Divine, 1999b [51] | HL, Kidney, Leukaemia, Pancreas, Prostate, Skin, Stomach and Urinary bladder | USA | 1 Year | 47 | 28,480 | |
| Wong, 2001b [52] | Brain, Colon, HL, Kidney, Leukaemia, Lung, Pancreas, Rectum, Skin melanoma and Stomach | USA | 1 Year | 43 | 7543 | |
| Nested Case-control studies | Exposure | Cases (n) | Controls (n) | |||
| Finkelstein, 1996 [53] | Mesothelioma | Asbestos | Canada | NR | 17 | 46 |
| Schnatter,1996 [54] | Leukaemia | Benzene | Canada | NR | 14 | 55 |
| Anttila, 2015 [55] | Kidney | Hydrocarbons | Finland | 3 Months | 30 | 81 |
| Rushton, 2014 [56] | AML, CLL | Benzene | Intercontinental | I Year | 140 | 568 |
| Glass, 2014 [57] | CML | Benzene | Intercontinental | NR | 28 | 122 |
| Stenehjem, 2015 [58] | AML, CLL and MM | Benzene | Norway | 20 Days | 91 | 415 |
| Rushton,1997 [59] | Leukaemia | Benzene | UK | I Year | 91 | 364 |
| Poole, 1993 [60] | Kidney | Hydrocarbons | USA | 6 Months | 100 | 406 |
| Author and Year | Cancer Site/Type | Type of Study | Country | Data Source | Follow-Up/Years of Exposure | Exposed Population |
|---|---|---|---|---|---|---|
| Cohort studies | ||||||
| Hurtig, 2002 [61] | Brain, HL, Lung and Skin melanoma | Cohort | Ecuador | National records | 4 | ~280,000 |
| Hurtig, 2004 [62] | Leukaemia | Cohort | Ecuador | National records | 20 | ~356,406 |
| Kelsh, 2009 [63] | Brain, Lung, Leukaemia, MM and NHL and Skin melanoma | Cohort | Ecuador | National records | 6 | 15335 |
| Ramis, 2012 [64] | NHL | Cohort | Spain | National records | 10 | 1,744,988 |
| Barregard, 2009 [65] | Leukaemia | Cohort | Sweden | National records | 30 | 15,000 |
| Sans, 1995 [66] | HL, Leukaemia, Lung, MM and NHL | Cohort | UK | Research Group Population Censuses and Welsh cancer registry | 18 | 115,721 |
| Cross sectional studies | ||||||
| Choi, 2018 [67] | Prostate | Cross sectional | Korea | Divisional and National records | 63,042 | |
| Lyons, 1995 [68] | Leukaemia | Cross sectional | UK | Wales and National records | 2632 | |
| Case control studies | Cases (n) | Controls (n) | ||||
| Tsai, 2009[69] | Urinary bladder | Case control | China/Taiwan | Bureau of Vital Statistics of the Taiwan | 821 | 821 |
| Weng, 2008 [70] | Leukaemia | Case control | China/Taiwan | Bureau of Vital Statistics of the Taiwan | 405 | 405 |
| Liu, 2008 [71] | Brain | Case control | China/Taiwan | Bureau of Vital Statistics of the Taiwan | 340 | 340 |
| Yu, 2005 [72] | Brian | Case control | China/Taiwan | Taiwanese population registry data | 143 | 364 |
| Micheli, 2014 [73] | Hematological malignancies | Case control | Italy | Italian National Institute of Statistics | 177 | 349 |
| McKenzie, 2017 [74] | Leukaemia | Case control | USA | Colorado cancer registry | 138 | 528 |
| Cancer Site/Type | n | Refinery OR 95% CI | n | Petroleum OR 95% CI | n | Petrochemical OR 95% CI | n | Refinery and Petrochemical OR 95% CI | n | Offshore OR 95% CI |
|---|---|---|---|---|---|---|---|---|---|---|
| Respiratory system | ||||||||||
| Lung | 8 | 0.83, 0.76–0.91 | 7 | 0.73, 0.63–0.85 | 2 | 0.57, 0.32–1.02 | 7 | 0.80, 0.67–0.96 | 3 | 1.20, 1.03–1.39 |
| Mesothelioma | 4 | 1.94, 1.00–3.76 | 5 | 1.58, 1.30–1.93 | x | 2 | 2.86, 2.16–3.77 | 2 | 2.47, 1.66–3.67 | |
| Skin | ||||||||||
| Skin melanoma | 7 | 1.15, 0.88–1.50 | 6 | 1.28, 1.10–1.50 | 2 | 0.85, 0.24–3.06 | 7 | 0.84, 0.55–1.27 | 2 | 1.44, 0.68–3.02 |
| Skin cancer (Non-specific) | 2 | 1.18, 0.71–1.95 | 5 | 0.89, 0.75–1.06 | x | 2 | 1.12, 0.72–1.74 | x | ||
| Urinary tract | ||||||||||
| Urinary bladder | 3 | 1.00, 0.88–1.14 | 7 | 0.93, 0.69–1.25 | x | 4 | 1.08, 0.78–1.49 | x | ||
| Kidney | 8 | 1.10, 0.98–1.24 | 7 | 0.91, 0.77–1.06 | x | 6 | 1.00, 0.79–1.25 | x | ||
| Reproductive system | ||||||||||
| Prostate | 4 | 1.01, 0.89–1.15 | 8 | 0.97, 0.83–1.146 | x | 7 | 1.01, 0.93–1.11 | x | ||
| Testis | 2 | 0.63, 0.28–1.42 | 4 | 1.00, 0.68–1.48 | x | x | - | |||
| Breast | 5 | 1.00, 0.78–1.27 | x | x | x | - | ||||
| Digestive and accessory digestive organ | ||||||||||
| Esophagus | 5 | 0.91, 0.81–1.03 | 3 | 0.67, 0.56–0.80 | x | 2 | 0.88, 0.59–1.30 | - | ||
| Stomach | 5 | 0.96, 0.83–1.10 | 4 | 0.79, 0.68–0.92 | x | 4 | 0.76, 0.60–0.95 | - | ||
| Colo-rectal | 12 | 0.87, 0.80–0.94 | 7 | 0.81, 0.74–0.88 | x | 6 | 0.87, 0.76–0.98 | - | ||
| Liver | 4 | 0.63, 0.36–1.08 | 4 | 0.73, 0.62–0.86 | - | 3 | 0.76, 0.56–1.02 | - | ||
| Gall bladder | 2 | 1.08, 0.45–2.56 | 3 | 1.83, 0.69–4.82 | - | - | - | |||
| Pancreas | 5 | 0.88, 0.72–1.07 | 4 | 0.84, 0.75–0.95 | - | 0.94, 0.78–1.13 | - | |||
| Central Nervous System | ||||||||||
| Brain | 7 | 1.00, 0.88–1.13 | 7 | 1.02, 0.89–1.16 | x | 6 | 1.05, 0.78–1.42 | - | ||
| Hematological | ||||||||||
| Hodgkin’s lymphoma | 8 | 1.07, 0.85–1.34 | 2 | 1.11, 0.75–1.63 | x | 3 | 0.86, 0.43–1.71 | - | ||
| Non-Hodgkin’s lymphoma | 2 | 0.96, 0.74–1.23 | 8 | 0.97, 0.87–1.09 | 2 | 1.09, 0.35–3.32 | 5 | 1.04, 0.63–1.72 | - | |
| Multiple Myeloma | 5 | 1.03, 0.82–1.30 | 7 | 1.26, 0.94–1.68 | x | x | x | |||
| Leukaemia | 16 | 1.07, 0.98–1.17 | 1.07, 0.91–1.26 | x | 8 | 1.02, 0.80–1.29 | 6 | 1.47, 1.12–1.92 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Onyije, F.M.; Hosseini, B.; Togawa, K.; Schüz, J.; Olsson, A. Cancer Incidence and Mortality among Petroleum Industry Workers and Residents Living in Oil Producing Communities: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2021, 18, 4343. https://doi.org/10.3390/ijerph18084343
Onyije FM, Hosseini B, Togawa K, Schüz J, Olsson A. Cancer Incidence and Mortality among Petroleum Industry Workers and Residents Living in Oil Producing Communities: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health. 2021; 18(8):4343. https://doi.org/10.3390/ijerph18084343
Chicago/Turabian StyleOnyije, Felix M., Bayan Hosseini, Kayo Togawa, Joachim Schüz, and Ann Olsson. 2021. "Cancer Incidence and Mortality among Petroleum Industry Workers and Residents Living in Oil Producing Communities: A Systematic Review and Meta-Analysis" International Journal of Environmental Research and Public Health 18, no. 8: 4343. https://doi.org/10.3390/ijerph18084343
APA StyleOnyije, F. M., Hosseini, B., Togawa, K., Schüz, J., & Olsson, A. (2021). Cancer Incidence and Mortality among Petroleum Industry Workers and Residents Living in Oil Producing Communities: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health, 18(8), 4343. https://doi.org/10.3390/ijerph18084343








