Modeling the Potential Future Distribution of Anthrax Outbreaks under Multiple Climate Change Scenarios for Kenya
Abstract
1. Introduction
2. Materials and Methods
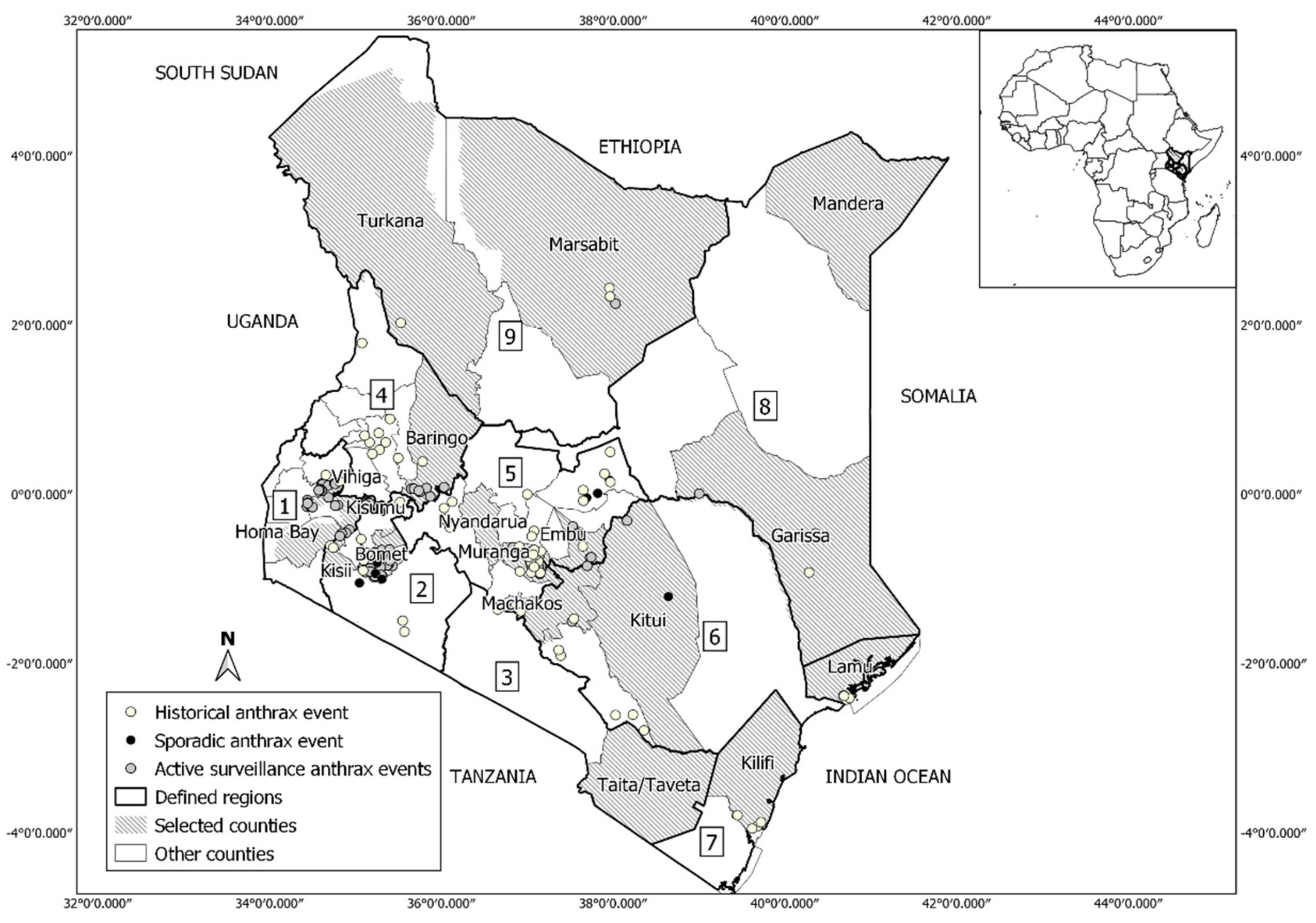
2.1. Study Area
2.2. Anthrax Occurrence Data
2.3. Predictive Data and Variable Selection
2.4. Model Building and Evaluation
3. Results
3.1. Model Variables
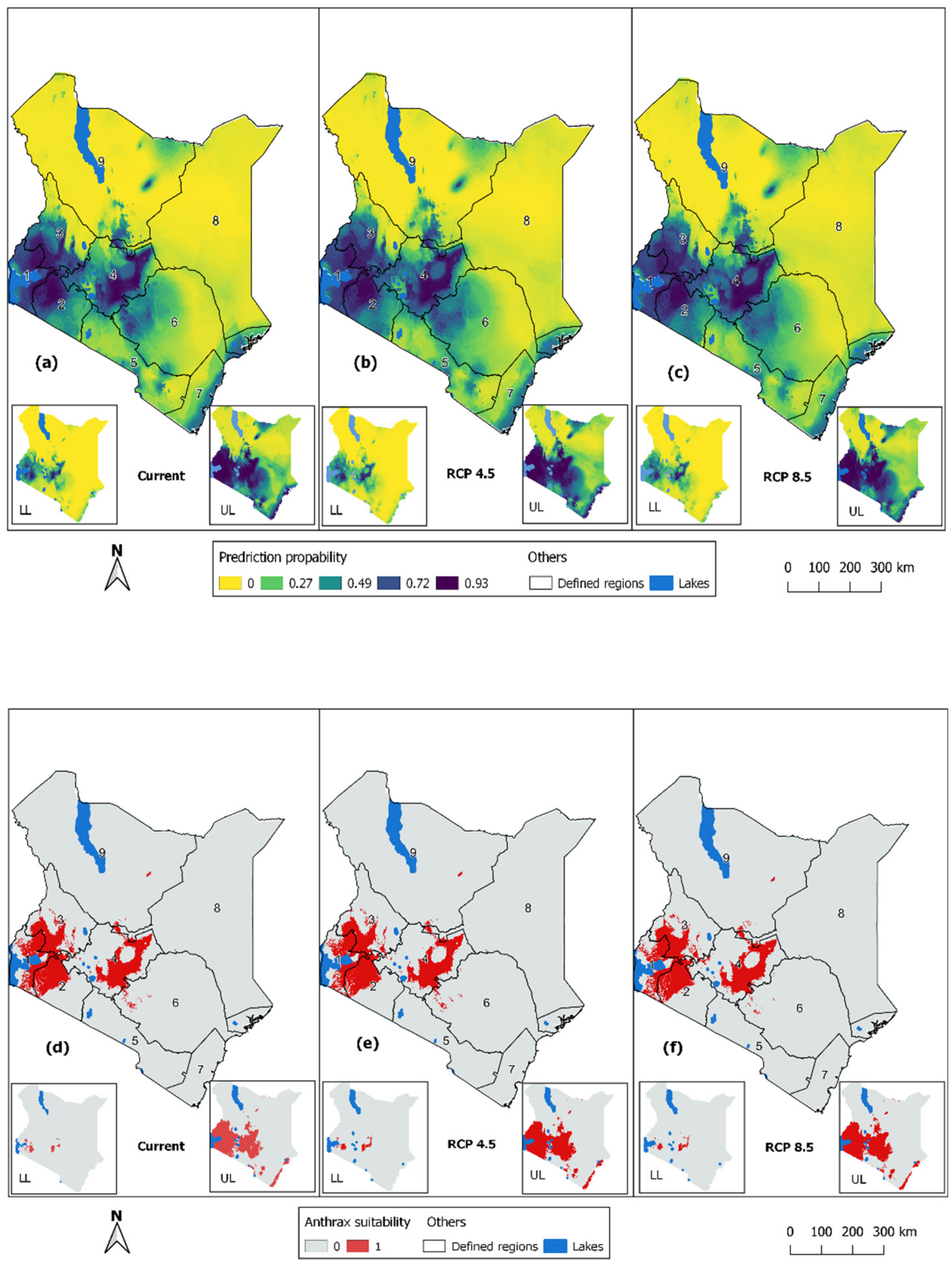
3.2. Prediction of Potential Anthrax Distribution Due to Climate Changes
3.3. Variable Contribution
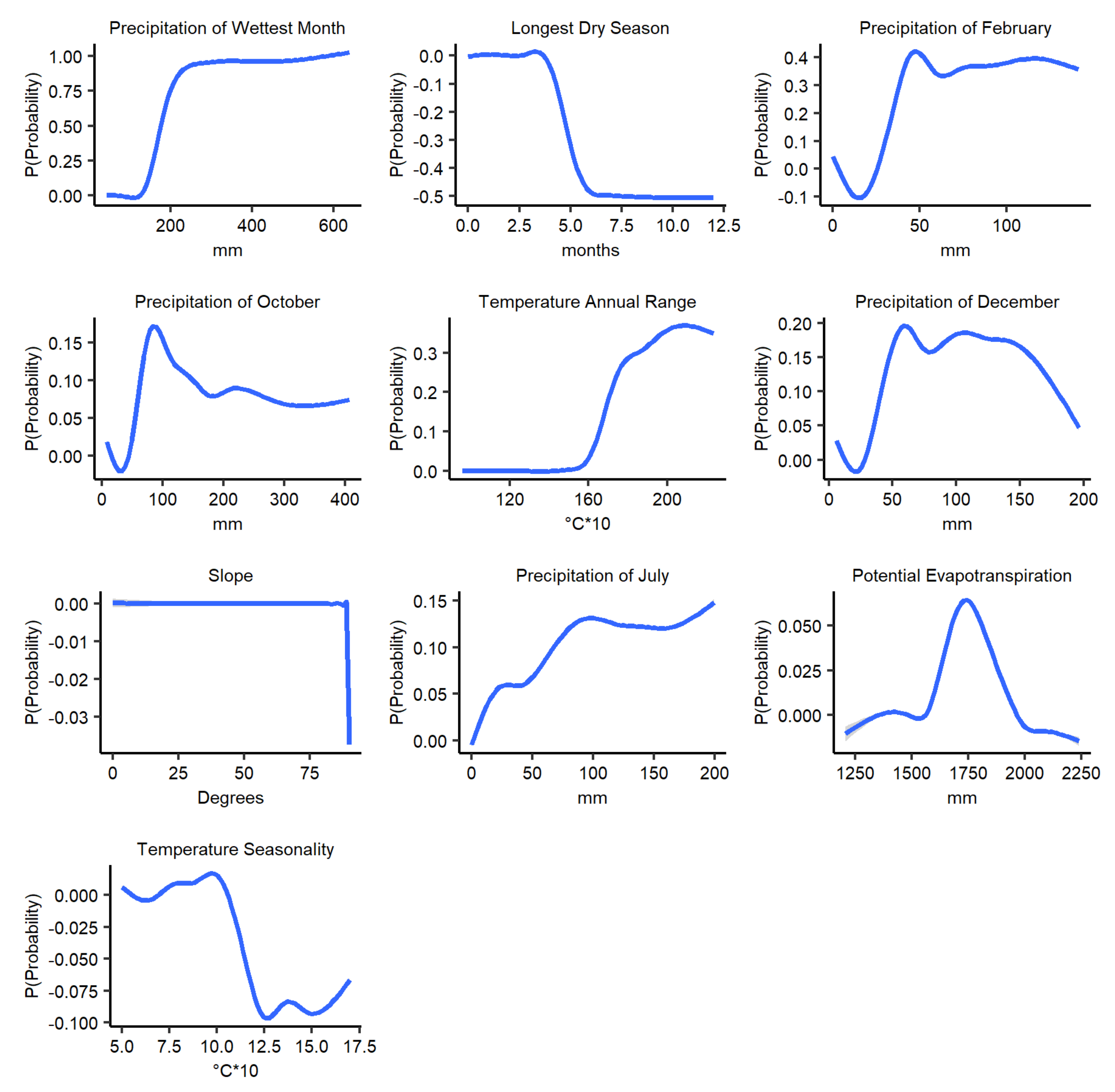
3.4. Marginal Effect of the Climatic Variables on Anthrax Distribution Predictions
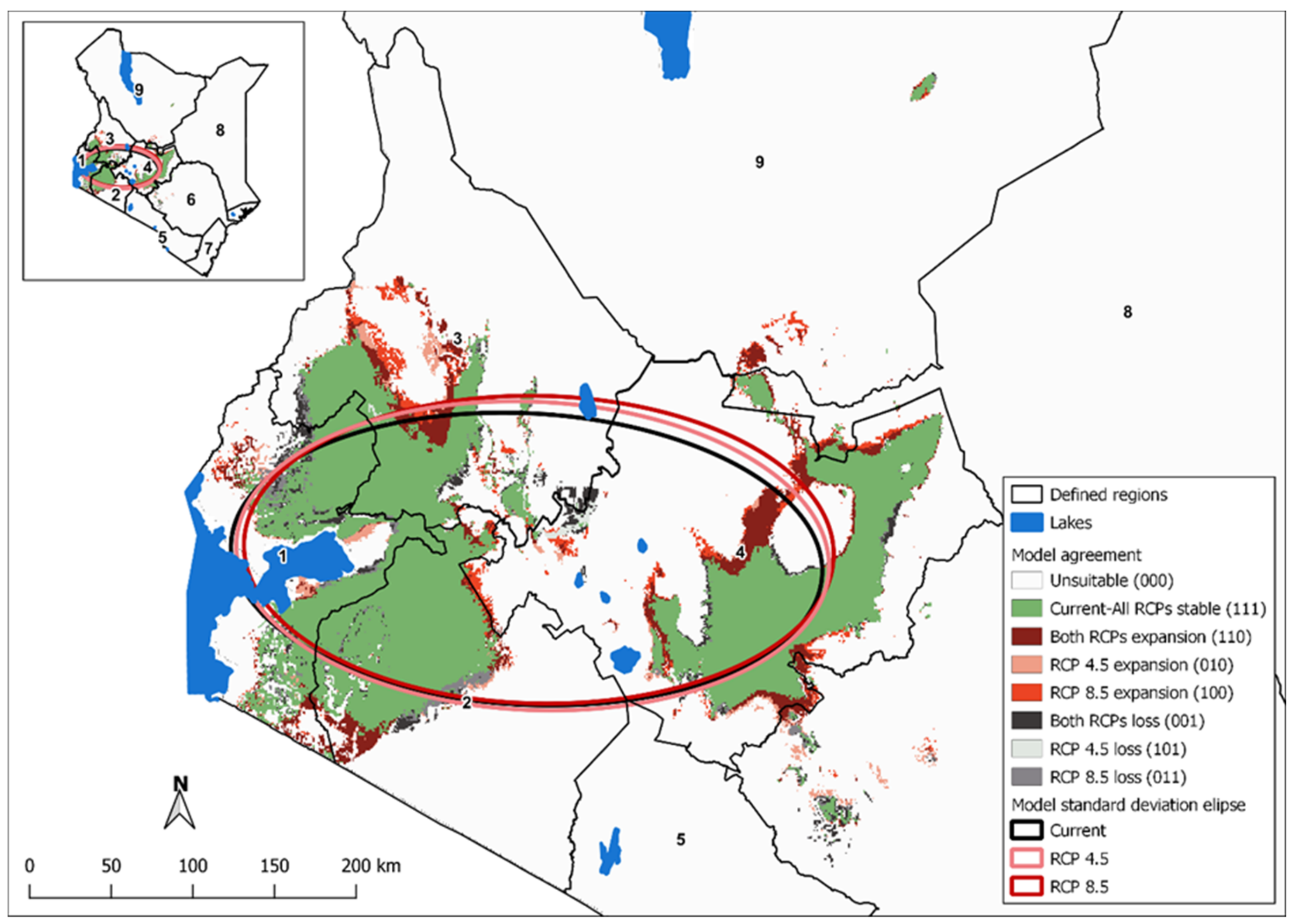
3.5. Change Detection
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Turnbull, P.C.B.; WHO. Anthrax in Humans and Animals; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Hugh-Jones, M.; Blackburn, J. The ecology of Bacillus anthracis. Mol. Asp. Med. 2009, 30, 356–367. [Google Scholar] [CrossRef]
- Carlson, C.J.; Kracalik, I.T.; Ross, N.; Alexander, K.A.; Hugh-Jones, M.E.; Fegan, M.; Elkin, B.T.; Epp, T.; Shury, T.K.; Zhang, W. The global distribution of Bacillus anthracis and associated anthrax risk to humans, livestock and wildlife. Nat. Microbiol. 2019, 4, 1337–1343. [Google Scholar] [CrossRef]
- Driciru, M.; Rwego, I.B.; Asiimwe, B.; Travis, D.A.; Alvarez, J.; VanderWaal, K.; Pelican, K. Spatio-temporal epidemiology of anthrax in Hippopotamus amphibious in Queen Elizabeth protected area, Uganda. PLoS ONE 2018, 13, e0206922. [Google Scholar] [CrossRef]
- Mwakapeje, E.R.; Høgset, S.; Fyumagwa, R.; Nonga, H.E.; Mdegela, R.H.; Skjerve, E. Anthrax outbreaks in the humans-livestock and wildlife interface areas of Northern Tanzania: A retrospective record review 2006–2016. BMC Public Health 2018, 18, 106. [Google Scholar] [CrossRef]
- Assefa, A.; Bihon, A.; Tibebu, A. Anthrax in the Amhara regional state of Ethiopia; spatiotemporal analysis and environmental suitability modeling with an ensemble approach. Prev. Vet. Med. 2020, 184, 105155. [Google Scholar] [CrossRef]
- Munyua, P.; Bitek, A.; Osoro, E.; Pieracci, E.G.; Muema, J.; Mwatondo, A.; Kungu, M.; Nanyingi, M.; Gharpure, R.; Njenga, K. Prioritization of zoonotic diseases in Kenya, 2015. PLoS ONE 2016, 11, e0161576. [Google Scholar] [CrossRef] [PubMed]
- Gomez, J.P.; Nekorchuk, D.M.; Mao, L.; Ryan, S.J.; Ponciano, J.M.; Blackburn, J.K. Decoupling environmental effects and host population dynamics for anthrax, a classic reservoir-driven disease. PLoS ONE 2018, 13, e0208621. [Google Scholar] [CrossRef] [PubMed]
- Blackburn, J.K.; Goodin, D.G. Differentiation of springtime vegetation indices associated with summer anthrax epizootics in west Texas, USA, deer. J. Wildl. Dis. 2013, 49, 699–703. [Google Scholar] [CrossRef]
- Fasanella, A.; Galante, D.; Garofolo, G.; Jones, M.H. Anthrax undervalued zoonosis. Vet. Microbiol. 2010, 140, 318–331. [Google Scholar] [CrossRef] [PubMed]
- Barro, A.S.; Fegan, M.; Moloney, B.; Porter, K.; Muller, J.; Warner, S.; Blackburn, J.K. Redefining the Australian anthrax belt: Modeling the ecological niche and predicting the geographic distribution of Bacillus anthracis. PLoS Negl. Trop. Dis. 2016, 10, e0004689. [Google Scholar] [CrossRef]
- Otieno, F.T.; Gachohi, J.; Gikuma-Njuru, P.; Kariuki, P.; Oyas, H.; Canfield, S.A.; Blackburn, J.K.; Njenga, K.; Bett, B. Modeling the spatial distribution of anthrax in southern Kenya. PLoS Negl. Trop. Dis. 2021, 5, e0009301. [Google Scholar] [CrossRef]
- Muturi, M.; Gachohi, J.; Mwatondo, A.; Lekolool, I.; Gakuya, F.; Bett, A.; Osoro, E.; Bitek, A.; Thumbi, S.M.; Munyua, P. Recurrent Anthrax Outbreaks in Humans, Livestock, and Wildlife in the Same Locality, Kenya, 2014–2017. Am. J. Trop. Med. Hyg. 2018, 99, 833–839. [Google Scholar] [CrossRef] [PubMed]
- Gachohi, J.; Bett, B.; Karanja, S.; Kikuvi, G.; Nyamai, M.; Mwangi, T.; Njenga, K. A systematic mapping protocol of methods and practices employed in ecological niche modelling of anthrax. Glob. Epidemiol. 2019, 1, 100014. [Google Scholar] [CrossRef]
- McMichael, A.J.; Campbell-Lendrum, D.; Kovats, S.; Edwards, S.; Wilkinson, P.; Wilson, T.; Nicholls, R.; Hales, S.; Tanser, F.; Le Sueur, D. Global Climate Change; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Stocker, T.F.; Qin, D.; Plattner, G.-K.; Tignor, M.M.B.; Allen, S.K.; Boschung, J.; Nauels, A.; Xia, Y.; Bex, V.; Midgley, P.M. Climate Change 2013: The physical science basis. In Contribution of Working Group I to the Fifth Assessment Report of IPCC the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2014. [Google Scholar]
- Lafferty, K.D. The ecology of climate change and infectious diseases. Ecology 2009, 90, 888–900. [Google Scholar] [CrossRef]
- Lippi, C.A.; Stewart-Ibarra, A.M.; Franklin Bajaña Loor, M.E.; Dueñas Zambrano, J.E.; Espinoza Lopez, N.A.; Blackburn, J.K.; Ryan, S.J. Geographic shifts in Aedes aegypti habitat suitability in Ecuador using larval surveillance data and ecological niche modeling: Implications of climate change for public health vector control. PLoS Negl. Trop. Dis. 2019, 13, e0007322. [Google Scholar] [CrossRef]
- De Oliveira, S.V.; Romero-Alvarez, D.; Martins, T.F.; dos Santos, J.P.; Labruna, M.B.; Gazeta, G.S.; Escobar, L.E.; Gurgel-Gonçalves, R. Amblyomma ticks and future climate: Range contraction due to climate warming. Acta Trop. 2017, 176, 340–348. [Google Scholar] [CrossRef]
- de La Rocque, S.; Rioux, J.-A.; Slingenbergh, J. Climate change: Effects on animal disease systems and implications for surveillance and control. Rev. Sci. Tech. 2008, 27, 339. [Google Scholar] [PubMed]
- Epstein, P. The ecology of climate change and infectious diseases: Comment. Ecology 2010, 91, 925–928. [Google Scholar] [CrossRef]
- Blackburn, J.K. Integrating geographic information systems and ecological niche modeling into disease ecology: A case study of Bacillus anthracis in the United States and Mexico. In Emerging and Endemic Pathogens; Springer: Berlin/Heidelberg, Germany, 2010; pp. 59–88. [Google Scholar]
- Garrett, K. The Effect of Climate Change on Risk of Anthrax Infection in the Kobuk Valley, Alaska. Ph.D. Thesis, University of Pittsburgh, Pittsburgh, PA, USA, 2017. [Google Scholar]
- Gale, P.; Drew, T.; Phipps, L.P.; David, G.; Wooldridge, M. The effect of climate change on the occurrence and prevalence of livestock diseases in Great Britain. J. Appl. Microbiol. 2009, 106, 1409–1423. [Google Scholar] [CrossRef] [PubMed]
- Mbow, H.-O.P.; Reisinger, A.; Canadell, J.; O’Brien, P. Special Report on Climate Change, Desertification, Land Degradation, Sustainable Land Management, Food Security, and Greenhouse Gas Fluxes in Terrestrial Ecosystems (SR2); IPCC: Geneva, Switzerland, 2017. [Google Scholar]
- Bett, B.K.; Otieno, F.T.; Murithi, F. Climate Change and Disease Dynamics: Predicted Changes in Ecological Niches for Rift Valley Fever in East Africa; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Linthicum, K.J.; Anyamba, A.; Tucker, C.J.; Kelley, P.W.; Myers, M.F.; Peters, C.J. Climate and satellite indicators to forecast Rift Valley fever epidemics in Kenya. Science 1999, 285, 397–400. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, S.W.; Bødker, R.; Malima, R.; Msangeni, H.A.; Kisinza, W. Effect of 1997–98 EI Niño on highland malaria in Tanzania. Lancet 2000, 355, 989–990. [Google Scholar] [CrossRef]
- Caminade, C.; McIntyre, K.M.; Jones, A.E. Impact of recent and future climate change on vector-borne diseases. Ann. N. Y. Acad. Sci. 2019, 1436, 157. [Google Scholar] [CrossRef]
- Joyner, T.A.; Lukhnova, L.; Pazilov, Y.; Temiralyeva, G.; Hugh-Jones, M.E.; Aikimbayev, A.; Blackburn, J.K. Modeling the potential distribution of Bacillus anthracis under multiple climate change scenarios for Kazakhstan. PLoS ONE 2010, 5, e9596. [Google Scholar] [CrossRef]
- Maksimović, Z.; Cornwell, M.S.; Semren, O.; Rifatbegović, M. The apparent role of climate change in a recent anthrax outbreak in cattle. Rev. Sci. Tech. Off. Int. Epiz. 2017, 36, 959–963. [Google Scholar] [CrossRef] [PubMed]
- Walsh, M.G.; de Smalen, A.W.; Mor, S.M. Climatic influence on anthrax suitability in warming northern latitudes. Sci. Rep. 2018, 8, 9269. [Google Scholar] [CrossRef]
- Njoka, J.T.; Yanda, P.; Maganga, F.; Liwenga, E.; Kateka, A.; Henku, A.; Mabhuye, E.; Malik, N.; Bavo, C. Kenya: Country situation assessment. In Pathways to Resilience in Semi-Arid Economies; International Development Research Centre: Ottawa, ON, Canada, 2016. [Google Scholar]
- Blackburn, J.K.; McNyset, K.M.; Curtis, A.; Hugh-Jones, M.E. Modeling the geographic distribution of Bacillus anthracis, the causative agent of anthrax disease, for the contiguous United States using predictive ecologic niche modeling. Am. J. Trop. Med. Hyg. 2007, 77, 1103–1110. [Google Scholar] [CrossRef]
- Warren, D.L.; Seifert, S.N. Ecological niche modeling in Maxent: The importance of model complexity and the performance of model selection criteria. Ecol. Appl. 2011, 21, 335–342. [Google Scholar] [CrossRef]
- Crisp, M.D.; Trewick, S.A.; Cook, L.G. Hypothesis testing in biogeography. Trends Ecol. Evol. 2011, 26, 66–72. [Google Scholar] [CrossRef]
- Kracalik, I.T.; Kenu, E.; Ayamdooh, E.N.; Allegye-Cudjoe, E.; Polkuu, P.N.; Frimpong, J.A.; Nyarko, K.M.; Bower, W.A.; Traxler, R.; Blackburn, J.K. Modeling the environmental suitability of anthrax in Ghana and estimating populations at risk: Implications for vaccination and control. PLoS Negl. Trop. Dis. 2017, 11, e0005885. [Google Scholar] [CrossRef]
- Steenkamp, P.J. Ecological Suitability Modelling for Anthrax in the Kruger National Park, South Africa. Ph.D. Thesis, University of Pretoria, Pretoria, South Africa, 2013. [Google Scholar]
- Chen, W.-J.; Lai, S.-J.; Yang, Y.; Liu, K.; Li, X.-L.; Yao, H.-W.; Li, Y.; Zhou, H.; Wang, L.-P.; Mu, D. Mapping the distribution of anthrax in mainland China, 2005–2013. PLoS Negl. Trop. Dis. 2016, 10, e0004637. [Google Scholar] [CrossRef]
- Mullins, J.C.; Garofolo, G.; Van Ert, M.; Fasanella, A.; Lukhnova, L.; Hugh-Jones, M.E.; Blackburn, J.K. Ecological niche modeling of bacillus anthracis on three continents: Evidence for genetic-ecological divergence? PLoS ONE 2013, 8, e72451. [Google Scholar] [CrossRef] [PubMed]
- Abdrakhmanov, S.K.; Mukhanbetkaliyev, Y.Y.; Korennoy, F.I.; Sultanov, A.A.; Kadyrov, A.S.; Kushubaev, D.B.; Bakishev, T.G. Maximum entropy modeling risk of anthrax in the Republic of Kazakhstan. Prev. Vet. Med. 2017, 144, 149–157. [Google Scholar] [CrossRef]
- Blackburn, J.K.; Matakarimov, S.; Kozhokeeva, S.; Tagaeva, Z.; Bell, L.K.; Kracalik, I.T.; Zhunushov, A. Modeling the ecological niche of Bacillus anthracis to map anthrax risk in Kyrgyzstan. Am. J. Trop. Med. Hyg. 2017, 96, 550–556. [Google Scholar] [CrossRef]
- Blackburn, J.K.; Odugbo, M.O.; Van Ert, M.; O’Shea, B.; Mullins, J.; Perrenten, V.; Maho, A.; Hugh-Jones, M.; Hadfield, T. Bacillus anthracis diversity and geographic potential across Nigeria, Cameroon and chad: Further support of a Novel West African Lineage. PLoS Negl. Trop. Dis. 2015, 9, e0003931. [Google Scholar]
- Mwakapeje, E.R.; Ndimuligo, S.A.; Mosomtai, G.; Ayebare, S.; Nyakarahuka, L.; Nonga, H.E.; Mdegela, R.H.; Skjerve, E. Ecological niche modeling as a tool for prediction of the potential geographic distribution of Bacillus anthracis spores in Tanzania. Int. J. Infect. Dis. 2019, 79, 142–151. [Google Scholar] [CrossRef]
- Chikerema, S.M.; Murwira, A.; Matope, G.; Pfukenyi, D.M. Spatial modelling of Bacillus anthracis ecological niche in Zimbabwe. Prev. Vet. Med. 2013, 111, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Acharya, B.K.; Cao, C.; Xu, M.; Khanal, L.; Naeem, S.; Pandit, S. Present and future of dengue fever in Nepal: Mapping climatic suitability by ecological niche model. Int. J. Environ. Res. Public Health 2018, 15, 187. [Google Scholar] [CrossRef]
- Carvalho, M.C.; Gomide, L.R.; dos Santos, R.M.; Scolforo, J.R.S.; de Carvalho, L.M.T.; de Mello, J.M. Modeling Ecological Niche of Tree Species in Brazilian Tropical Area. CERNE 2017, 23, 229–240. [Google Scholar] [CrossRef]
- Pearson, R.G.; Thuiller, W.; Araújo, M.B.; Martinez-Meyer, E.; Brotons, L.; McClean, C.; Miles, L.; Segurado, P.; Dawson, T.P.; Lees, D.C. Model-based uncertainty in species range prediction. J. Biogeogr. 2006, 33, 1704–1711. [Google Scholar] [CrossRef]
- Van Vuuren, D.P.; Edmonds, J.; Kainuma, M.; Riahi, K.; Thomson, A.; Hibbard, K.; Hurtt, G.C.; Kram, T.; Krey, V.; Lamarque, J.-F. The representative concentration pathways: An overview. Clim. Chang. 2011, 109, 5. [Google Scholar] [CrossRef]
- McSweeney, C.; New, M.; Lizcano, G. UNDP Climate Change Country Profiles: Kenya; UNDP: Nairobi, Kenya, 2008. [Google Scholar]
- Quantum GIS Development Team. Quantum GIS Geographic Information System. Open Source Geospatial Foundation Project. QGIS Development Team. 2018. Available online: http://qgis.osgeo.org (accessed on 20 January 2018).
- Platts, P.J.; Omeny, P.A.; Marchant, R. AFRICLIM: High-Resolution Climate Projections for Ecological Applications in Africa. The University of York. 2015. Available online: https://webfiles.york.ac.uk/KITE/AfriClim (accessed on 5 December 2020).
- Earth Resources Observation and Science Center; U.S. Geological Survey; U.S. Department of the Interior. USGS 30 ARC-second Global Elevation Data, GTOPO30; Research Data Archive at the National Center for Atmospheric Research, Computational and Information Systems Laboratory: Boulder, CO, USA, 1997. Available online: https://doi.org/10.5065/A1Z4-EE71 (accessed on 5 December 2020).
- Thompson, C.G.; Kim, R.S.; Aloe, A.M.; Becker, B.J. Extracting the Variance Inflation Factor and Other Multicollinearity Diagnostics from Typical Regression Results. Basic Appl. Soc. Psychol. 2017, 39, 81–90. [Google Scholar] [CrossRef]
- O’brien, R.M. A caution regarding rules of thumb for variance inflation factors. Qual. Quant. 2007, 41, 673–690. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2018. [Google Scholar]
- Elith, J.; Leathwick, J.R.; Hastie, T. A working guide to boosted regression trees. J. Anim. Ecol. 2008, 77, 802–813. [Google Scholar] [CrossRef]
- Elith, J.; Leathwick, J. Boosted Regression Trees for Ecological Modeling. 2017. Available online: https://mran.microsoft.com/snapshot/2016-11-19/web/packages/dismo/vignettes/brt.pdf (accessed on 3 January 2020).
- Ridgeway, G.; Southworth, M.H.; RUnit, S. Package ‘gbm’. Viitattu 2013, 10, 40. Available online: http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.398.7110&rep=rep1&type=pdf (accessed on 26 March 2021).
- Lobo, J.M.; Jiménez-Valverde, A.; Real, R. AUC: A misleading measure of the performance of predictive distribution models. Glob. Ecol. Biogeogr. 2008, 17, 145–151. [Google Scholar] [CrossRef]
- Yackulic, C.B.; Chandler, R.; Zipkin, E.F.; Royle, J.A.; Nichols, J.D.; Campbell Grant, E.H.; Veran, S. Presence-only modelling using MAXENT: When can we trust the inferences? Methods Ecol. Evol. 2013, 4, 236–243. [Google Scholar] [CrossRef]
- Greenwell, B.M. pdp: An R package for constructing partial dependence plots. R J. 2017, 9, 421. [Google Scholar] [CrossRef]
- Habibzadeh, F.; Habibzadeh, P.; Yadollahie, M. On determining the most appropriate test cut-off value: The case of tests with continuous results. Biochem. Med. 2016, 26, 297–307. [Google Scholar] [CrossRef]
- Bui, R.; Buliung, R.N.; Remmel, T.K.; Buliung, M.R.N. Package ‘aspace.’ 2012. Available online: http://cran.ma.imperial.ac.uk/web/packages/aspace/aspace.pdf (accessed on 10 January 2021).
- Bett, B.K.; Gachohi, J.M.; Gachohi, J.M.; Gakuya, F.; Lekolool, I.; Osoro, E.; Nderitu, L.; Munyua, P.; Ngere, I.; Kemunto, N. Insights from Kenya: Why Anthrax Outbreaks Recur in the Same Areas; International Livestock Research Institute (ILRI): Nairobi, Kenya, 2018. [Google Scholar]
- Evengård, B.; Sauerborn, R. Climate change influences infectious diseases both in the Arctic and the tropics: Joining the dots. Glob. Health Action 2009, 2, 2106. [Google Scholar] [CrossRef] [PubMed]
- Mweya, C.N.; Mboera, L.E.G.; Kimera, S.I. Climate influence on emerging risk areas for Rift Valley fever epidemics in Tanzania. Am. J. Trop. Med. Hyg. 2017, 97, 109–114. [Google Scholar] [CrossRef]
- Njenga, M.K.; Bett, B. Rift Valley fever virus—how and where virus is maintained during inter-epidemic periods. Curr. Clin. Microbiol. Rep. 2019, 6, 18–24. [Google Scholar] [CrossRef]
- Kangbai, J.; Momoh, E. Anthropogenic climatic change risks a global anthrax outbreak: A short communication. J. Trop. Dis. 2017, 5, 2. [Google Scholar] [CrossRef]
- Dragon, D.C.; Rennie, R.P. The ecology of anthrax spores: Tough but not invincible. Can. Vet. J. 1995, 36, 295. [Google Scholar] [PubMed]
- Walsh, M.G.; Mor, S.M.; Hossain, S. The elephant–livestock interface modulates anthrax suitability in India. Proc. R. Soc. B 2019, 286, 20190179. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.L. Epidemiology of Anthrax in the Kruger National Park, South Africa: Genetic Diversity and Environment; Louisiana State University: Baton Rouge, LA, USA, 1999. [Google Scholar]
- Pearce, J.L.; Boyce, M.S. Modelling distribution and abundance with presence-only data. J. Appl. Ecol. 2006, 43, 405–412. [Google Scholar] [CrossRef]
- Thuiller, W.; Guéguen, M.; Renaud, J.; Karger, D.N.; Zimmermann, N.E. Uncertainty in ensembles of global biodiversity scenarios. Nat. Commun. 2019, 10, 1446. [Google Scholar] [CrossRef] [PubMed]

| Variable | Unit |
|---|---|
| Precipitation of wettest month | mm |
| Temperature Seasonality | °C*10 |
| Annual temperature range | °C*10 |
| Length of longest dry season | months |
| Potential evapotranspiration | mm |
| Mean precipitation of October | mm |
| Mean precipitation of December | mm |
| Mean precipitation of February | mm |
| Mean precipitation of July | mm |
| Slope | degrees |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Otieno, F.T.; Gachohi, J.; Gikuma-Njuru, P.; Kariuki, P.; Oyas, H.; Canfield, S.A.; Bett, B.; Njenga, M.K.; Blackburn, J.K. Modeling the Potential Future Distribution of Anthrax Outbreaks under Multiple Climate Change Scenarios for Kenya. Int. J. Environ. Res. Public Health 2021, 18, 4176. https://doi.org/10.3390/ijerph18084176
Otieno FT, Gachohi J, Gikuma-Njuru P, Kariuki P, Oyas H, Canfield SA, Bett B, Njenga MK, Blackburn JK. Modeling the Potential Future Distribution of Anthrax Outbreaks under Multiple Climate Change Scenarios for Kenya. International Journal of Environmental Research and Public Health. 2021; 18(8):4176. https://doi.org/10.3390/ijerph18084176
Chicago/Turabian StyleOtieno, Fredrick Tom, John Gachohi, Peter Gikuma-Njuru, Patrick Kariuki, Harry Oyas, Samuel A. Canfield, Bernard Bett, Moses Kariuki Njenga, and Jason K. Blackburn. 2021. "Modeling the Potential Future Distribution of Anthrax Outbreaks under Multiple Climate Change Scenarios for Kenya" International Journal of Environmental Research and Public Health 18, no. 8: 4176. https://doi.org/10.3390/ijerph18084176
APA StyleOtieno, F. T., Gachohi, J., Gikuma-Njuru, P., Kariuki, P., Oyas, H., Canfield, S. A., Bett, B., Njenga, M. K., & Blackburn, J. K. (2021). Modeling the Potential Future Distribution of Anthrax Outbreaks under Multiple Climate Change Scenarios for Kenya. International Journal of Environmental Research and Public Health, 18(8), 4176. https://doi.org/10.3390/ijerph18084176






