Producers of Engineered Nanomaterials—What Motivates Company and Worker Participation in Biomonitoring Programs?
Abstract
1. Introduction
2. Materials and Methods
2.1. Survey Design
2.2. Questionnaire Creation and Translation
2.2.1. Questionnaire Aimed at Managers
2.2.2. Questionnaire Aimed at Workers
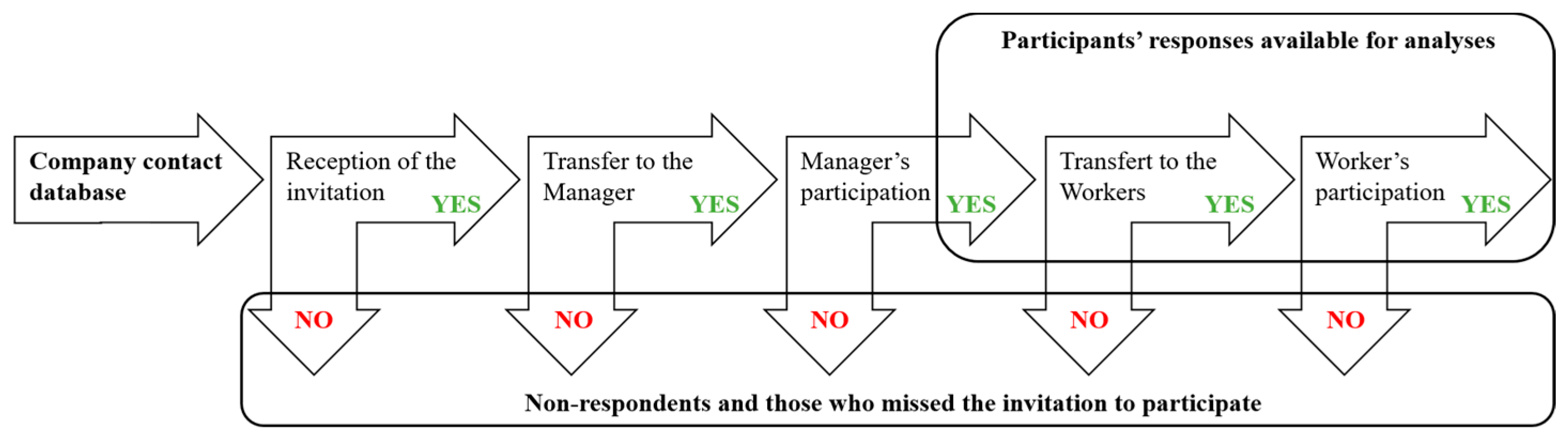
2.3. Survey Administration and Data Management
3. Results
3.1. General Company Characteristics
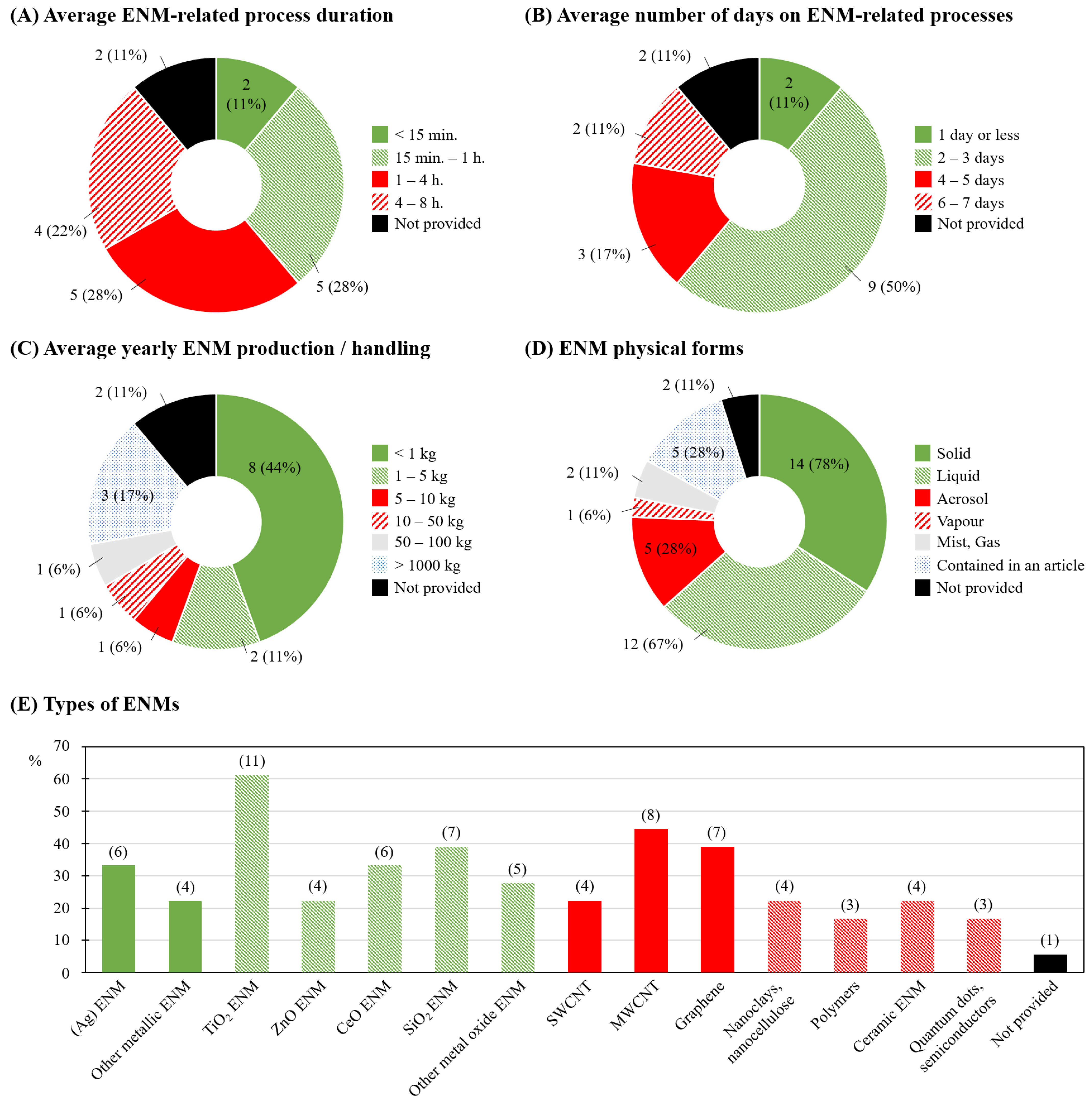
3.2. ENM Characteristics and Handling Specificities
3.3. Health and Safety Plan and Practice
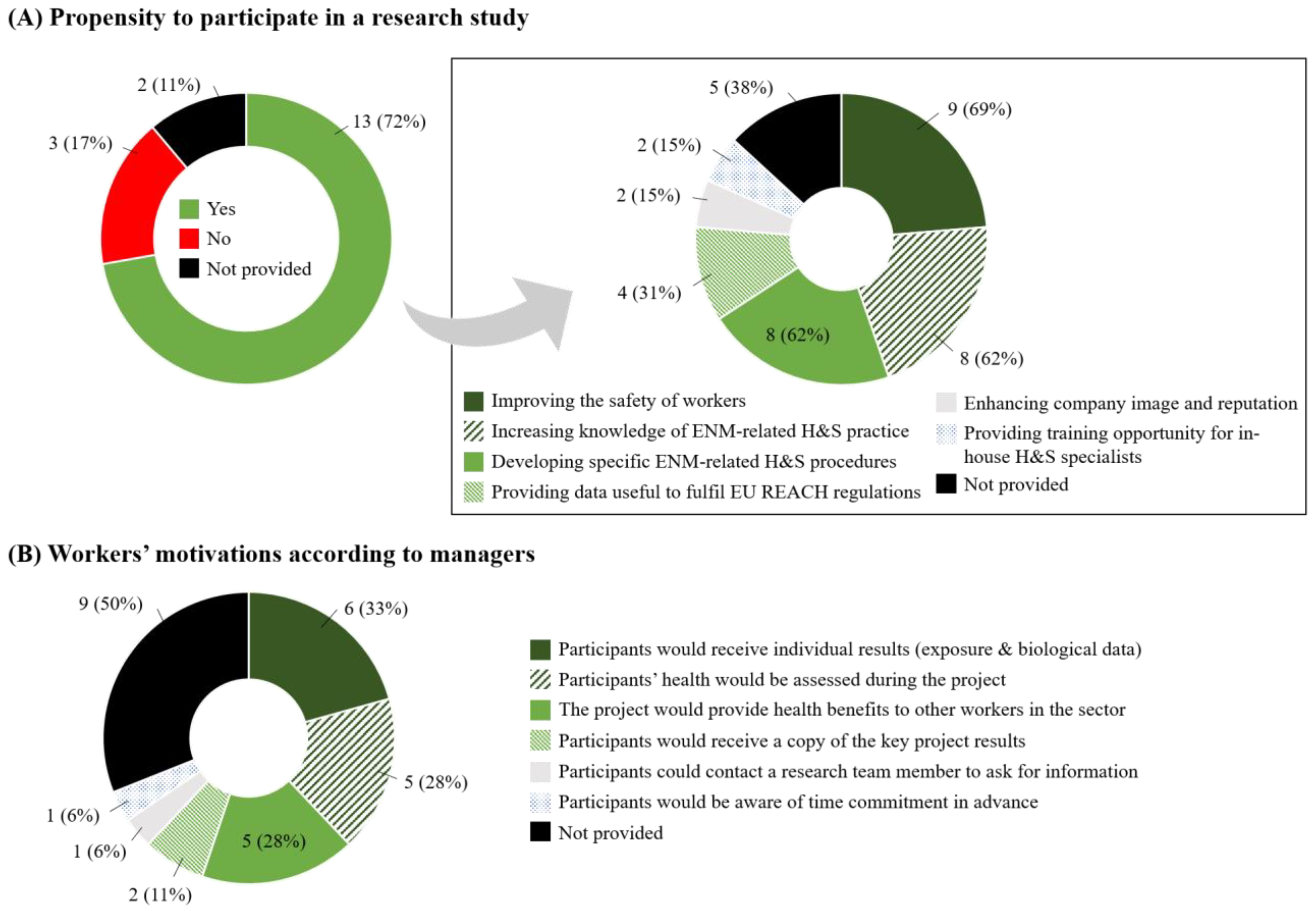
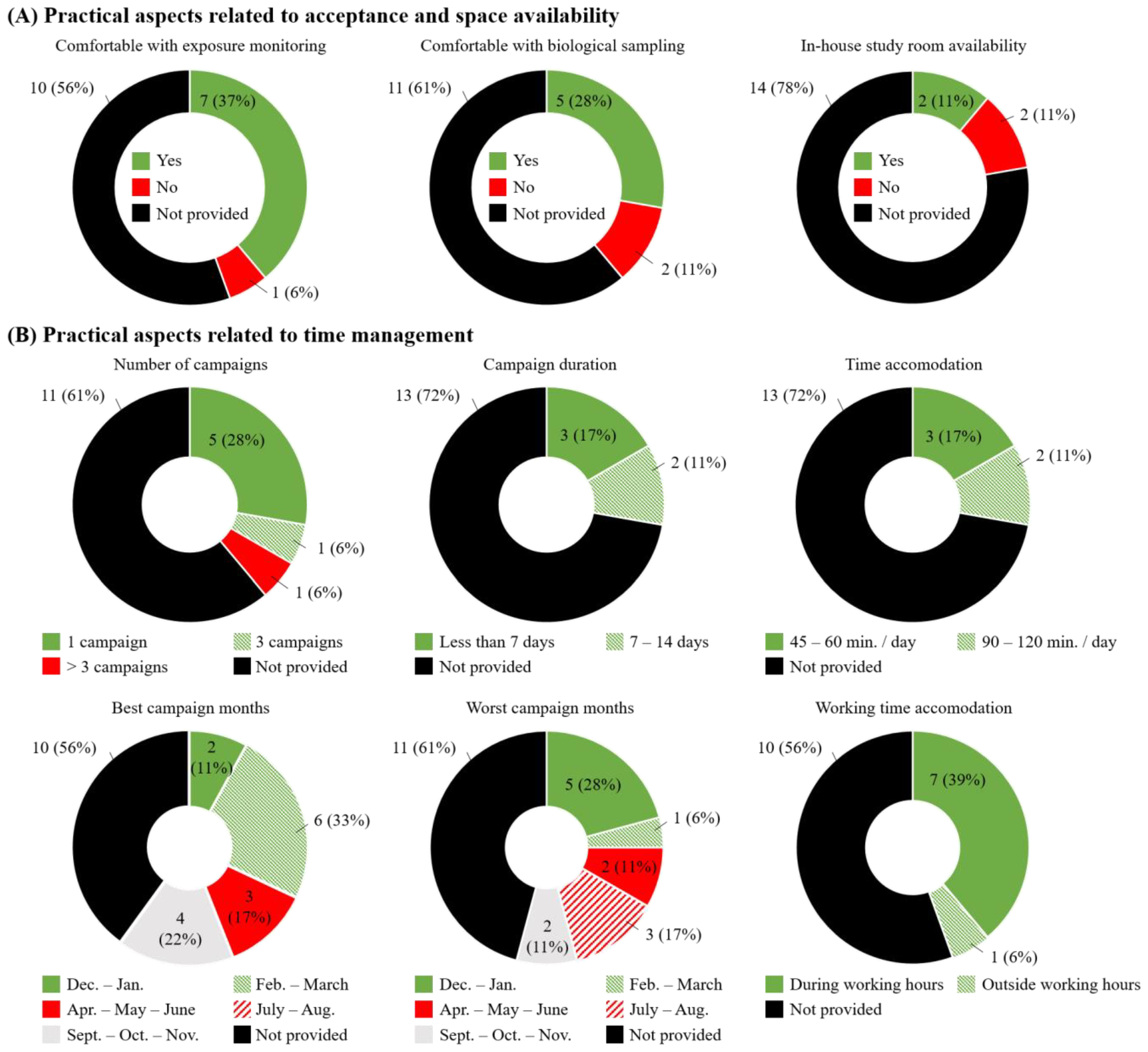
3.4. Managers’ Participation Acceptance and Study Practical Feasibility
3.5. Workers’ Participation Acceptance and Study Practical Feasibility
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- EC. Communication from the Commission to the European Parliament, the Council and the European Economic and Social Committee: Second Regulatory Review on Nanomaterials; European Commission: Brussels, Belgium, 2012. [Google Scholar]
- Boogaard, P.J.; Hays, S.M.; Aylward, L.L. Human biomonitoring as a pragmatic tool to support health risk management of chemicals—Examples under the EU REACH programme. Regul. Toxicol. Pharmacol. 2011, 59, 125–132. [Google Scholar] [CrossRef]
- Louro, H.; Heinälä, M.; Bessems, J.; Buekers, J.; Vermeire, T.; Woutersen, M.; van Engelen, J.; Borges, T.; Rousselle, C.; Ougier, E.; et al. Human biomonitoring in health risk assessment in Europe: Current practices and recommendations for the future. Int. J. Hyg. Environ. Health 2019, 222, 727–737. [Google Scholar] [CrossRef] [PubMed]
- LaMontagne, A.D.; Herrick, R.F.; Van Dyke, M.V.; Martyny, J.W.; Ruttenber, A.J. Exposure databases and exposure surveillance: Promise and practice. Am. Ind. Hyg. Assoc. J. 2002, 62, 205–212. [Google Scholar] [CrossRef]
- Brook, D.; Cowley, S.; Else, D.; Leggett, S. International Review of Surveillance and Control of Workplace Exposures; NOHSAC: Wellington, New Zealand, 2006; p. 99. [Google Scholar]
- Canu, I.G.; Boutou-Kempf, O.; Delabre, L.; Ducamp, S.; Iwatsubo, Y.; Marchand, J.L.; Imbernon, E. French registry of workers handling engineered nanomaterials as an instrument of integrated system for surveillance and research. J. Phys. Conf. Ser. 2013, 429, 012066. [Google Scholar] [CrossRef]
- Canu, I.G.; Burstyn, I.; Richardson, D.B. Commentary. Epidemiology 2016, 27, 765–768. [Google Scholar] [CrossRef]
- Debia, M.; Bakhiyi, B.; Ostiguy, C.; Verbeek, J.H.; Brouwer, D.H.; Murashov, V. A Systematic Review of Reported Exposure to Engineered Nanomaterials. Ann. Occup. Hyg. 2016, 60, 916–935. [Google Scholar] [CrossRef]
- Canu, I.G.; Bateson, T.F.; Bouvard, V.; Debia, M.; Dion, C.; Savolainen, K.; Yu, I.-J. Human exposure to carbon-based fibrous nanomaterials: A review. Int. J. Hyg. Environ. Health 2016, 219, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Boccuni, F.; Gagliardi, D.; Ferrante, R.; Rondinone, B.M.; Iavicoli, S. Measurement techniques of exposure to nanomaterials in the workplace for low- and medium-income countries: A systematic review. Int. J. Hyg. Environ. Health 2017, 220, 1089–1097. [Google Scholar] [CrossRef]
- Schulte, P.; Leso, V.; Niang, M.; Iavicoli, I. Current state of knowledge on the health effects of engineered nanomaterials in workers: A systematic review of human studies and epidemiological investigations. Scand. J. Work Environ. Health 2019, 45, 217–238. [Google Scholar] [CrossRef] [PubMed]
- Bergamaschi, E.; Poland, C.; Canu, I.G.; Prina-Mello, A. The role of biological monitoring in nano-safety. Nano Today 2015, 10, 274–277. [Google Scholar] [CrossRef]
- Schulte, P.; Leso, V.; Niang, M.; Iavicoli, I. Biological monitoring of workers exposed to engineered nanomaterials. Toxicol. Lett. 2018, 298, 112–124. [Google Scholar] [CrossRef] [PubMed]
- Schulte, P.A.; Iavicoli, I.; Rantanen, J.H.; Dahmann, D.; Iavicoli, S.; Pipke, R.; Canu, I.G.; Boccuni, F.; Ricci, M.; Polci, M.L.; et al. Assessing the protection of the nanomaterial workforce. Nanotoxicology 2016, 10, 1013–1019. [Google Scholar] [CrossRef] [PubMed]
- Gulumian, M.; Verbeek, J.; Andraos, C.; Sanabria, N.; De Jager, P. Systematic Review of Screening and Surveillance Programs to Protect Workers from Nanomaterials. PLoS ONE 2016, 11, e0166071. [Google Scholar] [CrossRef]
- Canu, I.G.; Schulte, P.; Riediker, M.; Fatkhutdinova, L.; Bergamaschi, E. Methodological, political and legal issues in the assessment of the effects of nanotechnology on human health. J. Epidemiol. Community Health 2018, 72, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Schmid, K.; Danuser, B.; Riediker, M. Nanoparticle Usage and Protection Measures in the Manufacturing Industry—A Representative Survey. J. Occup. Environ. Hyg. 2010, 7, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Jacquet, F. Repérage des salariés potentiellement exposés aux nanoparticules. Ref. Santé Trav. 2012, 132, 33–38. [Google Scholar]
- Endo, C.-A.; Ostiguy, C.; Dossa, N.I.; Emond, C. Portrait de la Nanotechnologie au Québec dans Les Milieux Industriels et de la Recherche Universitaire et Publique; Institut de recherche Robert-Sauvé en Santé et en Sécurité au Travail (IRSST): Montréal, QC, Canada, 2014. [Google Scholar]
- Iavicoli, I.; Leso, V.; Piacci, M.; Cioffi, D.L.; Canu, I.G.; Schulte, P.A. An Exploratory Assessment of Applying Risk Management Practices to Engineered Nanomaterials. Int. J. Environ. Res. Public Health 2019, 16, 3290. [Google Scholar] [CrossRef] [PubMed]
- Canu, I.; Ducros, C.; Ducamp, S.; Delabre, L.; Audignon-Durand, S.; Durand, C.; Iwatsubo, Y.; Jezewski-Serra, D.; Le Bihan, O.; Malard, S.; et al. A standardized non-instrumental tool for characterizing workstations concerned with exposure to engineered nanomaterials. J. Phys. Conf. Ser. 2015, 617, 012036. [Google Scholar] [CrossRef]
- Canu, I.G.; Jezewski-Serra, D.; Delabre, L.; Ducamp, S.; Iwatsubo, Y.; Audignon-Durand, S.; Ducros, C.; Radauceanu, A.; Durand, C.; Witschger, O.; et al. Qualitative and Semiquantitative Assessment of Exposure to Engineered Nanomaterials within the French EpiNano Program: Inter- and Intramethod Reliability Study. Ann. Work Expo. Health 2017, 61, 87–97. [Google Scholar] [CrossRef]
- Teschke, K.; Marino, S.; Chu, R.; Tsui, J.K.C.; Harris, M.A.; Marion, S.A. Public Opinions about Participating in Health Research. Can. J. Public Health 2010, 101, 159–164. [Google Scholar] [CrossRef]
- Slegers, C.; Zion, D.; Glass, D.; Kelsall, H.; Fritschi, L.; Brown, N.; Loff, B. Why Do People Participate in Epidemiological Research? J. Bioethical Inq. 2015, 12, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Foth, H.; Hayes, A. Concept of REACH and impact on evaluation of chemicals. Hum. Exp. Toxicol. 2008, 27, 5–21. [Google Scholar] [CrossRef] [PubMed]
- Beard, J.D.; Erdely, A.; Dahm, M.M.; de Perio, M.A.; Birch, M.E.; Evans, D.E.; Fernback, J.E.; Eye, T.; Kodali, V.; Mercer, R.R.; et al. Carbon nanotube and nanofiber exposure and sputum and blood biomarkers of early effect among U.S. workers. Environ. Int. 2018, 116, 214–228. [Google Scholar] [CrossRef]
- Canu, I.G.; Jezewski-Serra, D.; Laurene, D.; Ducamp, S.; Lemaitre, A.; Chami, K.; Jaegle, I.; Iwatsubo, Y. O33-2 Epinano, the french program for epidemiological surveillance of workers potentially exposed to engineered nanomaterials: The state of play after first 18 months of operation. Oral Sess. 33 Dis. Surveill. 2016, 73, A61. [Google Scholar] [CrossRef]

| Geographical Location | Number (%) of Companies | |
| Europe | Spain | 3 (17%) |
| Italy | 2 (11%) | |
| Germany | 2 (11%) | |
| Portugal | 1 (6%) | |
| Norway | 1 (6%) | |
| Outside Europe | USA | 2 (11%) |
| Brazil | 2 (11%) | |
| Not provided | 5 (28%) | |
| Sector for ENM-Related Activities | ||
| Production | 6 (33%) | |
| Use | 4 (22%) | |
| Storage, packaging, commercialization, distribution | 3 (17%) | |
| R&D, lab. use or characterization, scale-up | 12 (67%) | |
| Nano-safety, hygienist-related tasks | 6 (33%) | |
| Not provided | 2 (11%) | |
| Number of Employees Manipulating ENMs | ||
| <10 employees | 9 (50%) | |
| 10–49 employees | 3 (17%) | |
| 50–250 employees | 1 (6%) | |
| >250 employees | 3 (17%) | |
| Not provided | 2 (11%) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crézé, C.; François, M.; Hopf, N.B.; Dorribo, V.; Sauvain, J.-J.; Bergamaschi, E.; Garzaro, G.; Domat, M.; Friesl, J.; Penssler, E.; et al. Producers of Engineered Nanomaterials—What Motivates Company and Worker Participation in Biomonitoring Programs? Int. J. Environ. Res. Public Health 2021, 18, 3851. https://doi.org/10.3390/ijerph18083851
Crézé C, François M, Hopf NB, Dorribo V, Sauvain J-J, Bergamaschi E, Garzaro G, Domat M, Friesl J, Penssler E, et al. Producers of Engineered Nanomaterials—What Motivates Company and Worker Participation in Biomonitoring Programs? International Journal of Environmental Research and Public Health. 2021; 18(8):3851. https://doi.org/10.3390/ijerph18083851
Chicago/Turabian StyleCrézé, Camille, Marjorie François, Nancy B. Hopf, Victor Dorribo, Jean-Jacques Sauvain, Enrico Bergamaschi, Giacomo Garzaro, Maida Domat, Judith Friesl, Eva Penssler, and et al. 2021. "Producers of Engineered Nanomaterials—What Motivates Company and Worker Participation in Biomonitoring Programs?" International Journal of Environmental Research and Public Health 18, no. 8: 3851. https://doi.org/10.3390/ijerph18083851
APA StyleCrézé, C., François, M., Hopf, N. B., Dorribo, V., Sauvain, J.-J., Bergamaschi, E., Garzaro, G., Domat, M., Friesl, J., Penssler, E., Progiou, A., & Guseva Canu, I. (2021). Producers of Engineered Nanomaterials—What Motivates Company and Worker Participation in Biomonitoring Programs? International Journal of Environmental Research and Public Health, 18(8), 3851. https://doi.org/10.3390/ijerph18083851







