Are Orienteers Protected Enough against Tick Bites? Estimating Human Exposure to Tick Bites through a Participative Science Survey during an Orienteering Competition
Abstract
1. Introduction
2. Materials and Methods
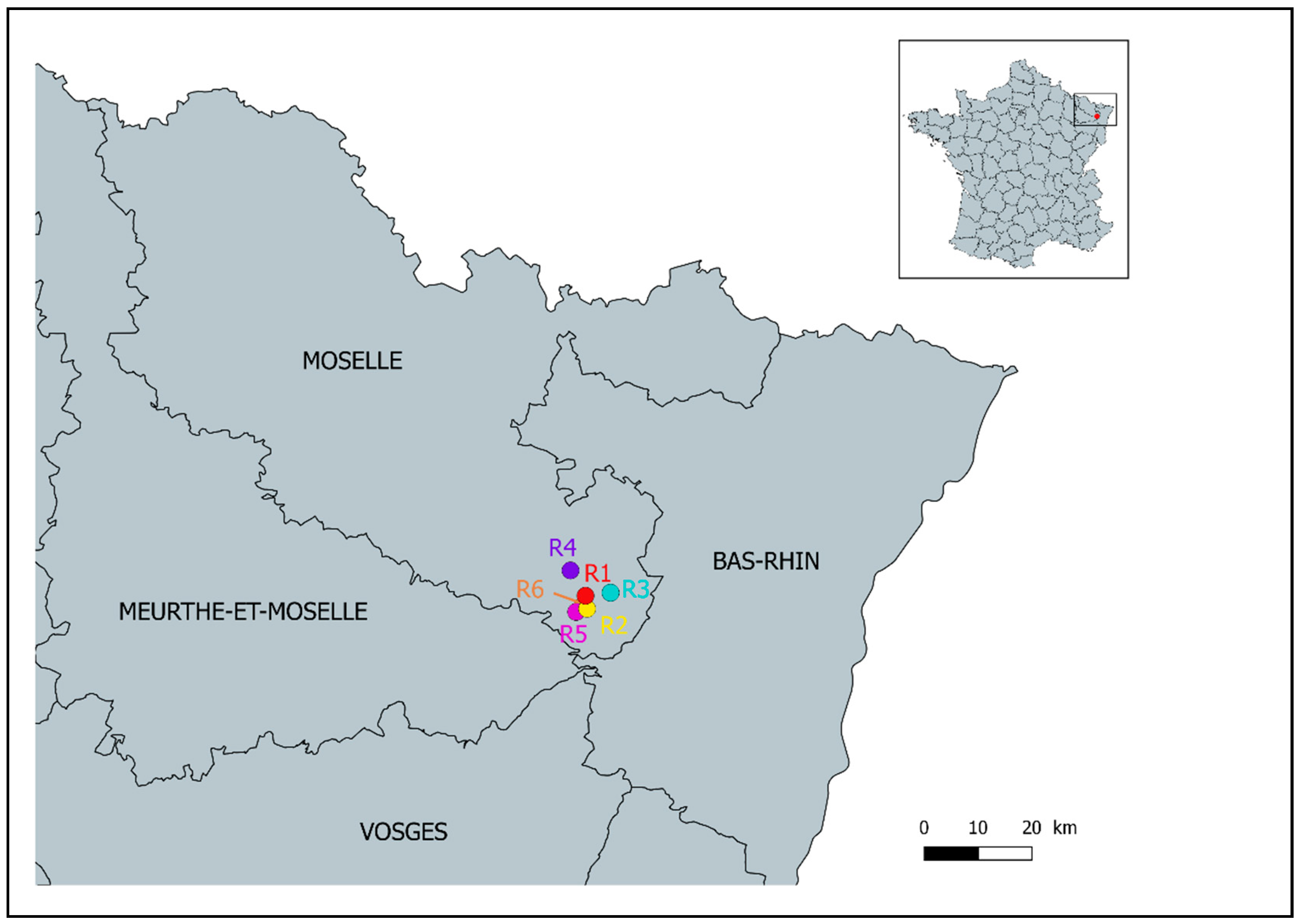
2.1. Sample Collection
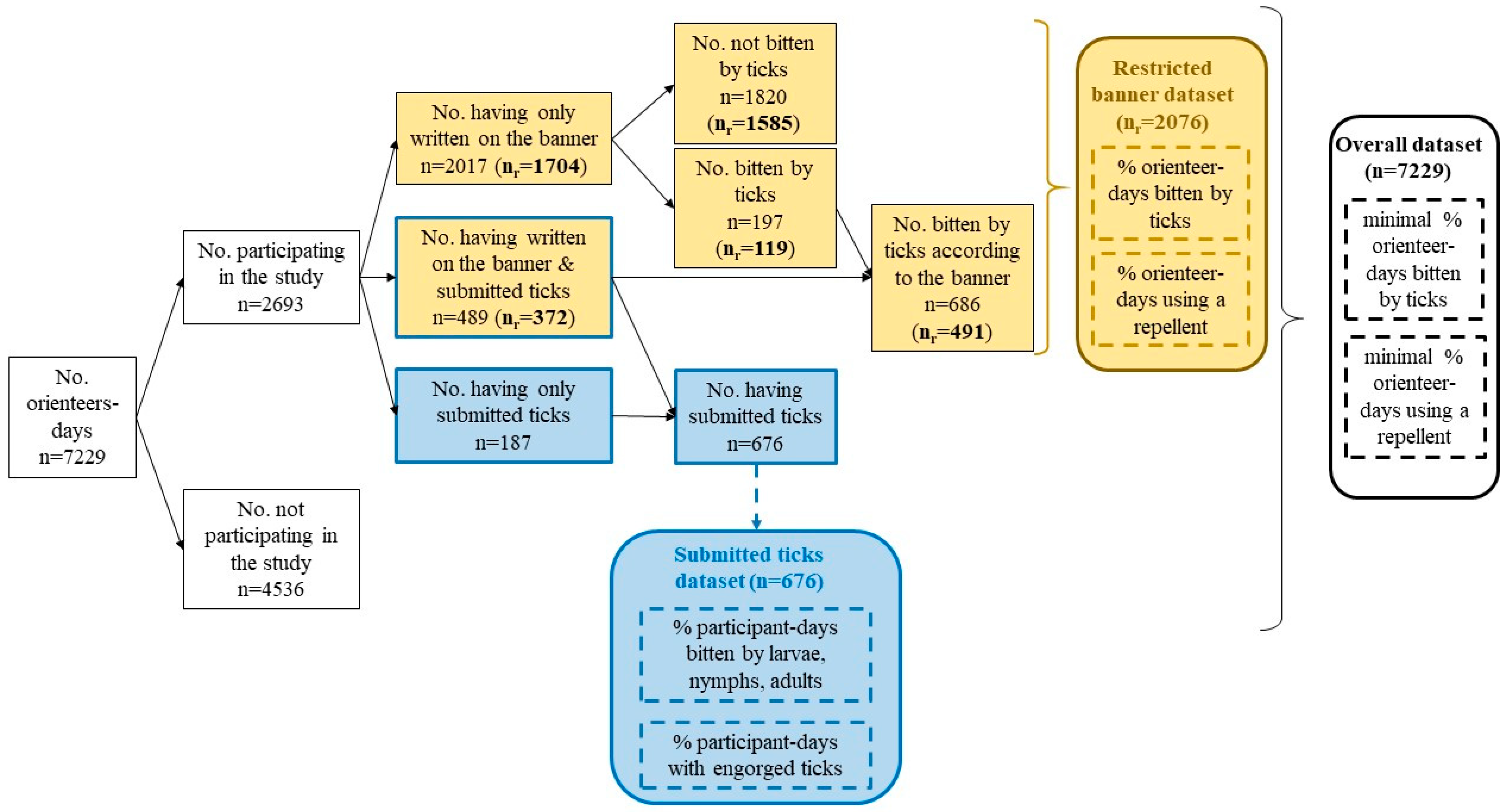
2.2. Data Cleaning
2.3. Participation in the Study
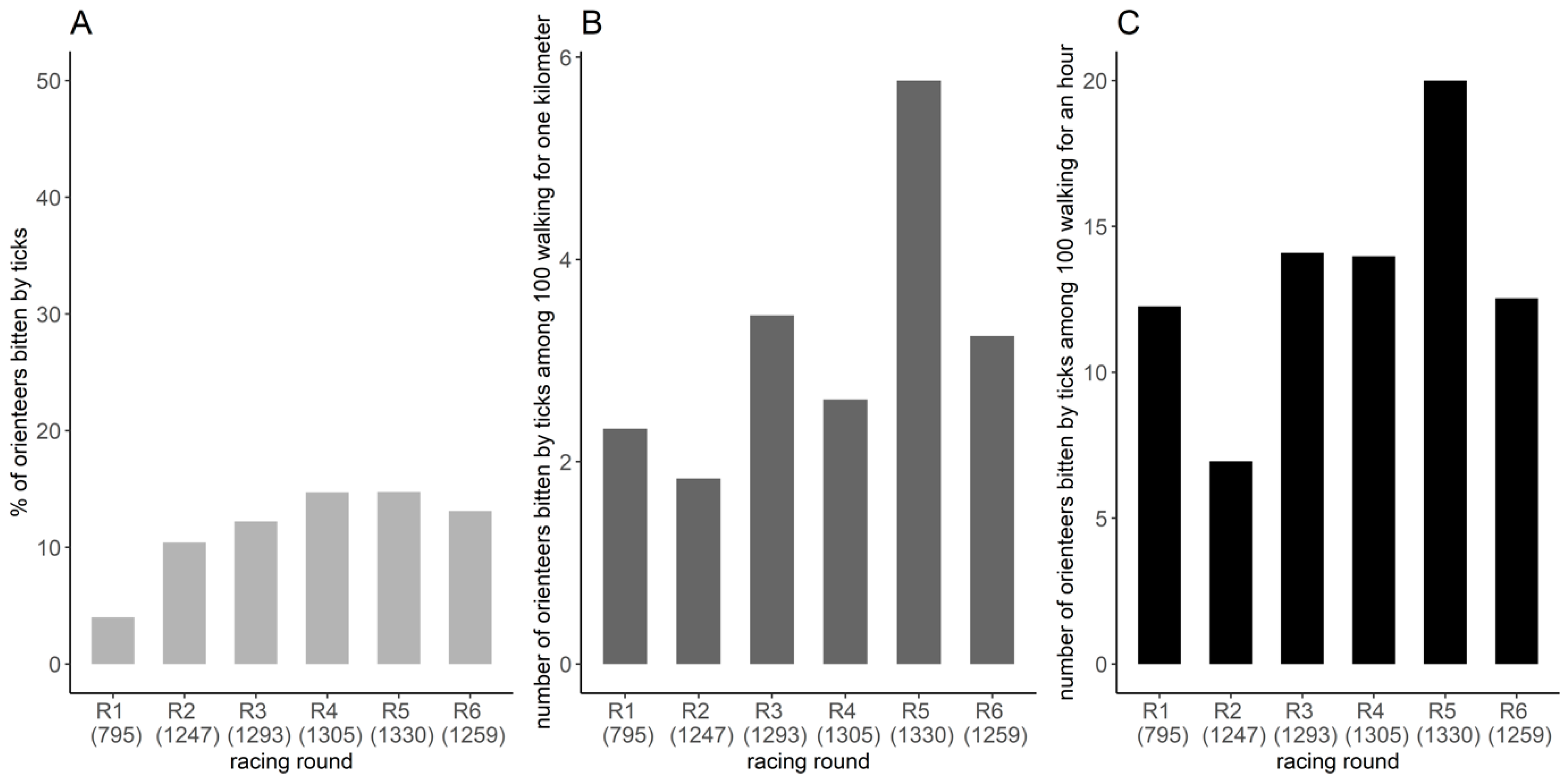
2.4. Frequency of Tick Bites among Orienteers
2.5. Analysis of Submitted Ticks: Frequency of Engorged Ticks and of Larvae, Nymphs, and Adults
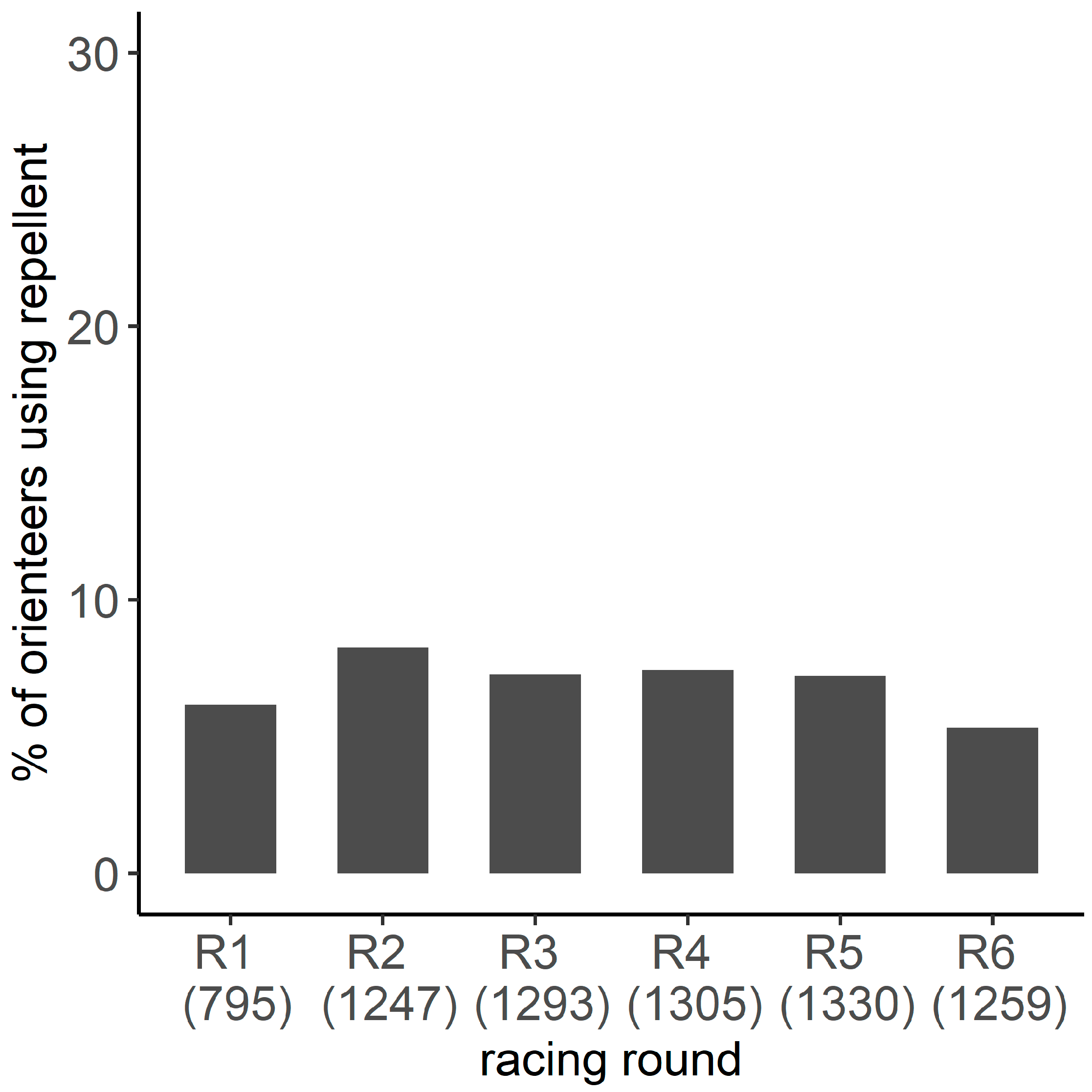
2.6. Frequency of Repellent Use and Effect on the Prevalence of Tick Infestation
3. Results
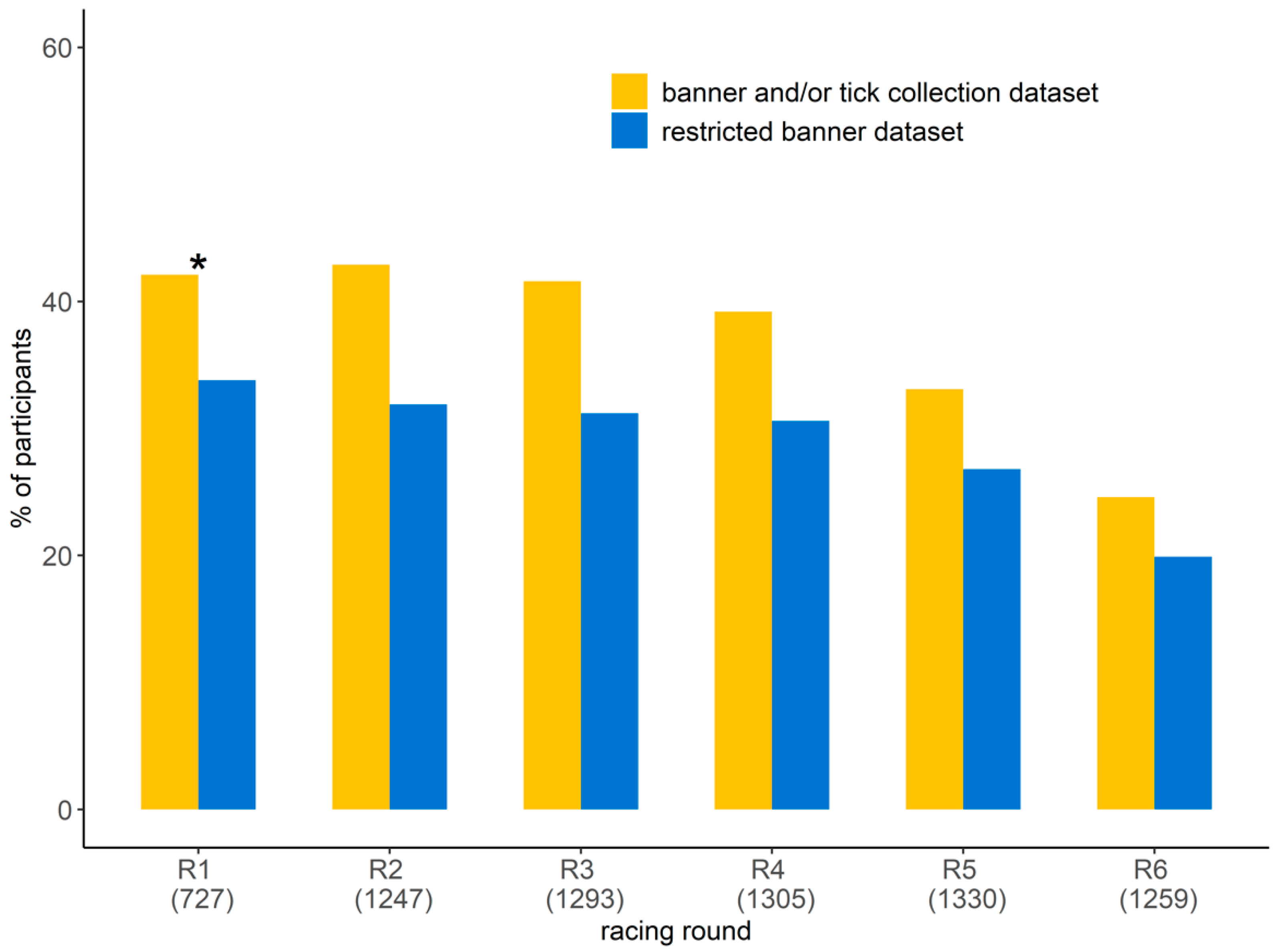
3.1. Participation in the Study
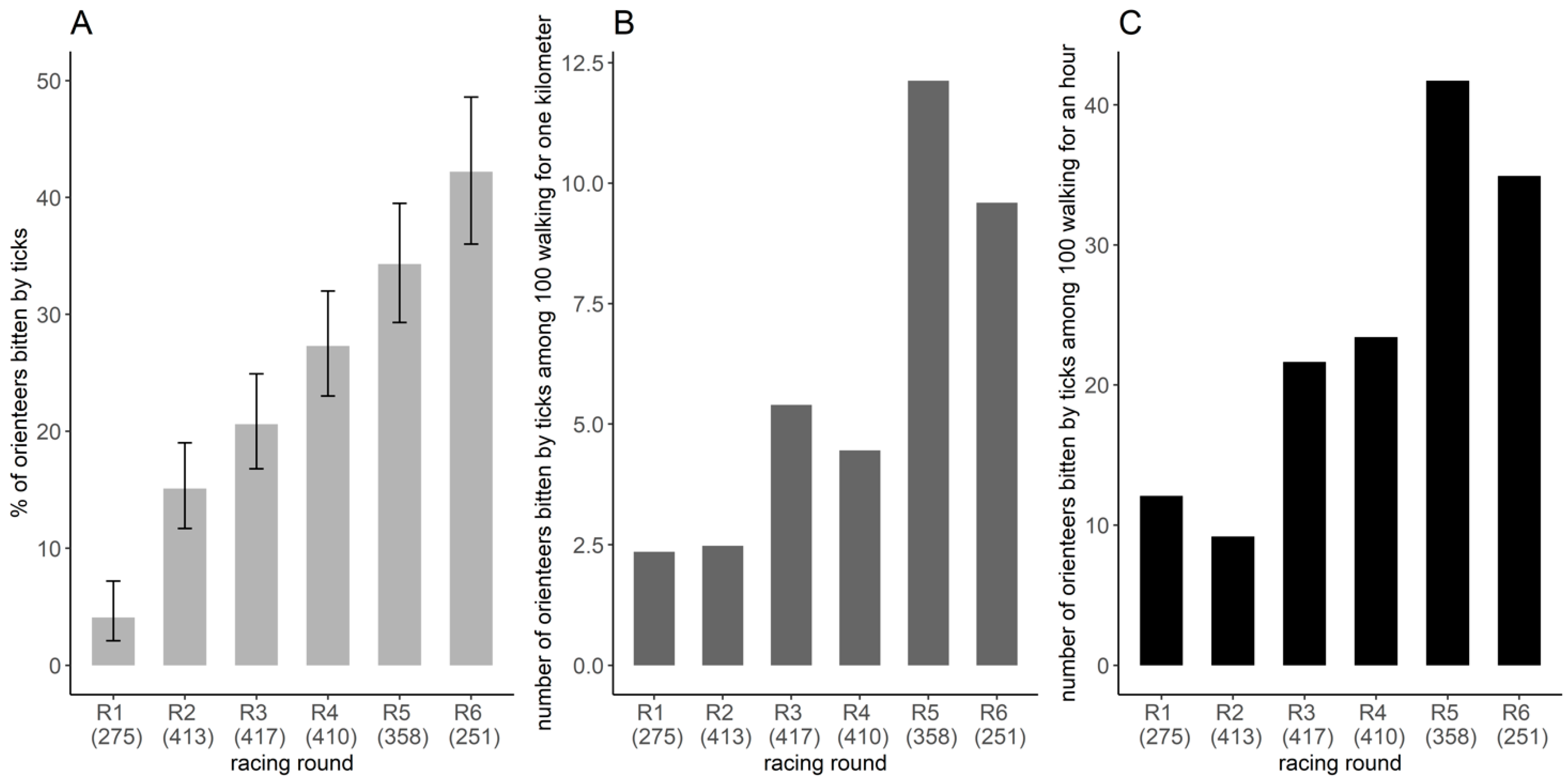
3.2. Frequency of Tick Bites among Orienteers
3.3. Analysis of Submitted Ticks: Frequency of Engorged Ticks and of Larvae, Nymphs, and Adults
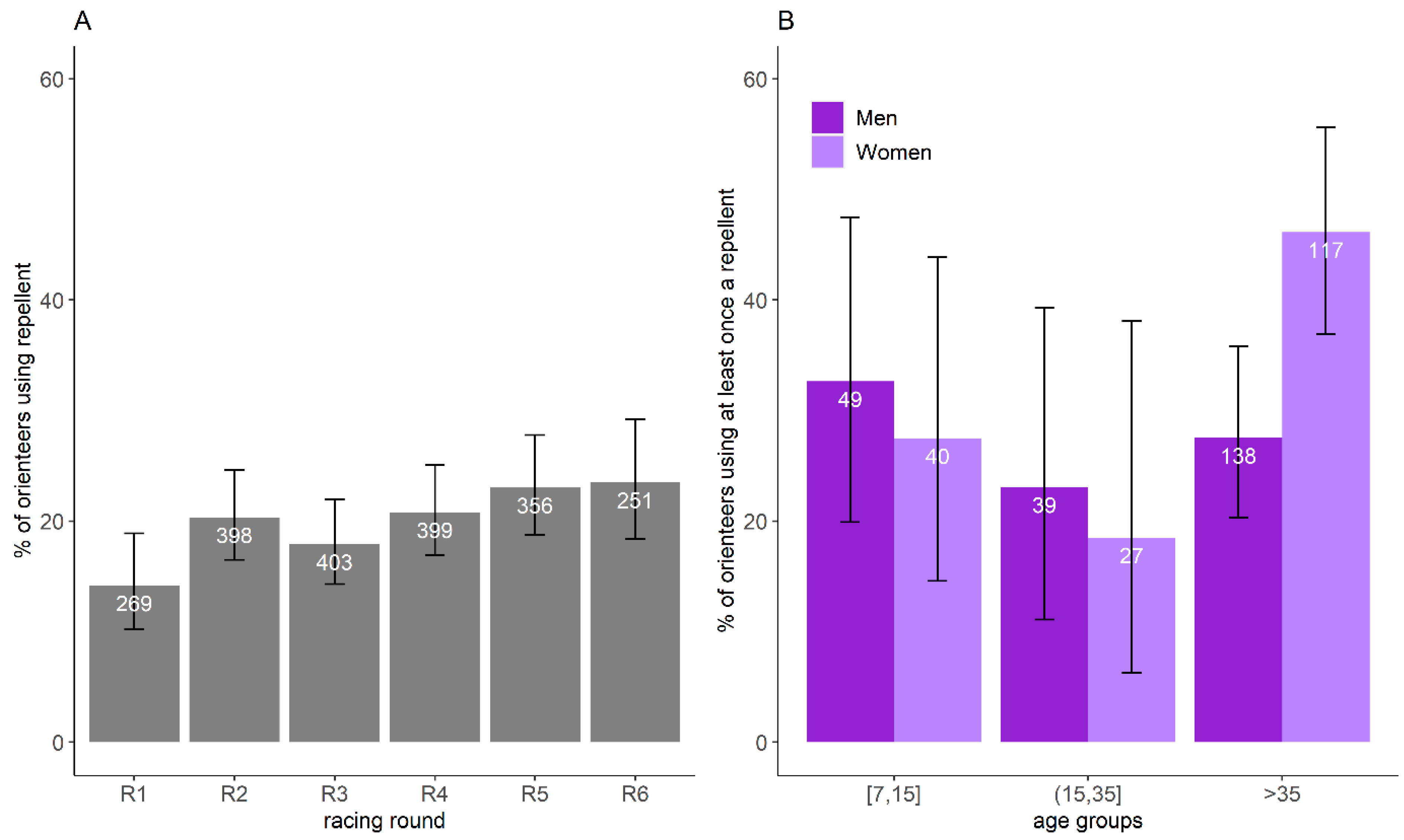
3.4. Frequency of Repellent Use
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
Appendix A.1. Materiel & Methods
Appendix A.1.1. Frequency of Tick Bites among Orienteers
Appendix A.1.2. Frequency of Repellent Use
Appendix A.2. Results
Appendix A.2.1. Frequency of Tick Bites among Orienteers
Appendix A.2.2. Frequency of Repellent Use
References
- Dantas-Torres, F.; Chomel, B.B.; Otranto, D. Ticks and Tick-Borne Diseases: A One Health Perspective. Trends Parasitol. 2012, 28, 437–446. [Google Scholar] [CrossRef]
- Heyman, P.; Cochez, C.; Hofhuis, A.; van der Giessen, J.; Sprong, H.; Porter, S.R.; Losson, B.; Saegerman, C.; Donoso-Mantke, O.; Niedrig, M.; et al. A Clear and Present Danger: Tick-Borne Diseases in Europe. Expert Rev. Anti-Infect. Ther. 2010, 8, 33–50. [Google Scholar] [CrossRef] [PubMed]
- Zanzani, S.A.; Rimoldi, S.G.; Manfredi, M.; Grande, R.; Gazzonis, A.L.; Merli, S.; Olivieri, E.; Giacomet, V.; Antinori, S.; Cislaghi, G.; et al. Lyme Borreliosis Incidence in Lombardy, Italy (2000–2015): Spatiotemporal Analysis and Environmental Risk Factors. Ticks Tick-Borne Dis. 2019, 10, 101257. [Google Scholar] [CrossRef]
- Zeman, P.; Benes, C. Spatial Distribution of a Population at Risk: An Important Factor for Understanding the Recent Rise in Tick-Borne Diseases (Lyme Borreliosis and Tick-Borne Encephalitis in the Czech Republic). Ticks Tick-Borne Dis. 2013, 4, 522–530. [Google Scholar] [CrossRef]
- Hofhuis, A.; Harms, M.; van den Wijngaard, C.; Sprong, H.; van Pelt, W. Continuing Increase of Tick Bites and Lyme Disease between 1994 and 2009. Ticks Tick-Borne Dis. 2015, 6, 69–74. [Google Scholar] [CrossRef]
- Guglielmone, A.A.; Robbins, R.G. Hard Ticks (Acari: Ixodida: Ixodidae) Parasitizing Humans: A Global Overview; Springer International Publishing: Cham, Switzerland, 2018; ISBN 978-3-319-95551-3. [Google Scholar]
- Estrada-Peña, A.; Mihalca, A.D.; Petney, T.N. Ticks of Europe and North Africa: A Guide to Species Identification; Springer: Berlin/Heidelberg, Germany, 2018; ISBN 3-319-63760-6. [Google Scholar]
- Cull, B.; Pietzsch, M.E.; Gillingham, E.L.; McGinley, L.; Medlock, J.M.; Hansford, K.M. Seasonality and Anatomical Location of Human Tick Bites in the United Kingdom. Zoonoses Public Health 2020, 67, 112–121. [Google Scholar] [CrossRef]
- Cull, B.; Pietzsch, M.E.; Hansford, K.M.; Gillingham, E.L.; Medlock, J.M. Surveillance of British Ticks: An Overview of Species Records, Host Associations, and New Records of Ixodes Ricinus Distribution. Ticks Tick-Borne Dis. 2018, 9, 605–614. [Google Scholar] [CrossRef]
- Battisti, E.; Zanet, S.; Boraso, F.; Minniti, D.; Giacometti, M.; Duscher, G.G.; Ferroglio, E. Survey on Tick-Borne Pathogens in Ticks Removed from Humans in Northwestern Italy. Vet. Parasitol. Reg. Stud. Rep. 2019, 18, 100352. [Google Scholar] [CrossRef]
- Robertson, J.N.; Gray, J.S.; Stewart, P. Tick Bite and Lyme Borreliosis Risk at a Recreational Site in England. Eur. J. Epidemiol. 2000, 16, 647–652. [Google Scholar] [CrossRef] [PubMed]
- Wilhelmsson, P.; Lindblom, P.; Fryland, L.; Ernerudh, J.; Forsberg, P.; Lindgren, P.-E. Prevalence, Diversity, and Load of Borrelia Species in Ticks That Have Fed on Humans in Regions of Sweden and Åland Islands, Finland with Different Lyme Borreliosis Incidences. PLoS ONE 2013, 8, e81433. [Google Scholar] [CrossRef] [PubMed]
- Faulde, M.K.; Rutenfranz, M.; Hepke, J.; Rogge, M.; Görner, A.; Keth, A. Human Tick Infestation Pattern, Tick-Bite Rate, and Associated Borrelia Burgdorferi s.l. Infection Risk during Occupational Tick Exposure at the Seedorf Military Training Area, Northwestern Germany. Ticks Tick-Borne Dis. 2014, 5, 594–599. [Google Scholar] [CrossRef] [PubMed]
- Černý, J.; Lynn, G.; Hrnková, J.; Golovchenko, M.; Rudenko, N.; Grubhoffer, L. Management Options for Ixodes Ricinus-Associated Pathogens: A Review of Prevention Strategies. Int. J. Environ. Res. Public. Health 2020, 17, 1830. [Google Scholar] [CrossRef] [PubMed]
- Bellet-Edimo, R.; Betschart, B.; Gern, L. Frequency and Efficiency of Transovarial and Subsequent Transstadial Transmission of Borrelia Burgdorferi Sensu Lato in Ixodes Ricinus Ticks. Bull Société Neuchâtel Sci. Nat. 2005, 128, 117–125. [Google Scholar]
- Richter, D.; Debski, A.; Hubalek, Z.; Matuschka, F.-R. Absence of Lyme Disease Spirochetes in Larval Ixodes Ricinus Ticks. Vector-Borne Zoonotic Dis. 2012, 12, 21–27. [Google Scholar] [CrossRef]
- Rollend, L.; Fish, D.; Childs, J.E. Transovarial Transmission of Borrelia Spirochetes by Ixodes Scapularis: A Summary of the Literature and Recent Observations. Ticks Tick-Borne Dis. 2013, 4, 46–51. [Google Scholar] [CrossRef]
- Richards, S.L.; Langley, R.; Apperson, C.S.; Watson, E. Do Tick Attachment Times Vary between Different Tick-Pathogen Systems? Environments 2017, 4, 37. [Google Scholar] [CrossRef]
- Stamm, L.V. Tick-Borne Diseases on the Rise: An Ounce of Prevention Is Worth a Pound of Cure. Future Microbiol. 2019, 14, 833–835. [Google Scholar] [CrossRef]
- Van der Heijden, A.; Mulder, B.C.; Poortvliet, P.M.; van Vliet, A.J.H. Social-Cognitive Determinants of the Tick Check: A Cross-Sectional Study on Self-Protective Behavior in Combatting Lyme Disease. BMC Public Health 2017, 17. [Google Scholar] [CrossRef]
- Tahir, D.; Meyer, L.; Fourie, J.; Jongejan, F.; Mather, T.; Choumet, V.; Blagburn, B.; Straubinger, R.K.; Varloud, M. Interrupted Blood Feeding in Ticks: Causes and Consequences. Microorganisms 2020, 8, 910. [Google Scholar] [CrossRef]
- Aenishaenslin, C.; Michel, P.; Ravel, A.; Gern, L.; Milord, F.; Waaub, J.-P.; Bélanger, D. Factors Associated with Preventive Behaviors Regarding Lyme Disease in Canada and Switzerland: A Comparative Study. BMC Public Health 2015, 15, 185. [Google Scholar] [CrossRef] [PubMed]
- Bartosik, K.; Kubrak, T.; Olszewski, T.; Jung, M.; Buczek, A. Prevention of Tick bites and protection against tick-borne diseases in south-eastern poland. Ann. Agric. Environ. Med. 2008, 15, 181–185. [Google Scholar]
- Eisen, L.; Dolan, M.C. Evidence for Personal Protective Measures to Reduce Human Contact with Blacklegged Ticks and for Environmentally Based Control Methods to Suppress Host-Seeking Blacklegged Ticks and Reduce Infection with Lyme Disease Spirochetes in Tick Vectors and Rodent Reservoirs. J. Med. Entomol. 2016, 53, 1063–1092. [Google Scholar] [CrossRef]
- Eisen, L.; Eisen, R.J. Critical Evaluation of the Linkage Between Tick-Based Risk Measures and the Occurrence of Lyme Disease Cases. J. Med. Entomol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Lane, R.S.; Steinlein, D.B.; Mun, J. Human Behaviors Elevating Exposure to Ixodes Pacificus (Acari: Ixodidae) Nymphs and Their Associated Bacterial Zoonotic Agents in a Hardwood Forest. J. Med. Entomol. 2004, 41, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Porter, W.T.; Motyka, P.J.; Wachara, J.; Barrand, Z.A.; Hmood, Z.; McLaughlin, M.; Pemberton, K.; Nieto, N.C. Citizen Science Informs Human-Tick Exposure in the Northeastern United States. Int. J. Health Geogr. 2019, 18, 9. [Google Scholar] [CrossRef]
- Randolph, S.E. Human Activities Predominate in Determining Changing Incidence of Tick-Borne Encephalitis in Europe. Eurosurveillance 2010, 15, 19606. [Google Scholar] [CrossRef]
- Hall, J.L.; Alpers, K.; Bown, K.J.; Martin, S.J.; Birtles, R.J. Use of Mass-Participation Outdoor Events to Assess Human Exposure to Tickborne Pathogens. Emerg. Infect. Dis. 2017, 23, 463–467. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Fons, F.; Gilbert, L. The Role of Deer as Vehicles to Move Ticks, Ixodes Ricinus, between Contrasting Habitats. Int. J. Parasitol. 2010, 40, 1013–1020. [Google Scholar] [CrossRef] [PubMed]
- Tack, W.; Madder, M.; Baeten, L.; Vanhellemont, M.; Verheyen, K. Shrub Clearing Adversely Affects the Abundance of Ixodes Ricinus Ticks. Exp. Appl. Acarol. 2013, 60, 411–420. [Google Scholar] [CrossRef]
- Knight, R.L.; Gutzwiller, K.L. Wildlife and Recreationist; Island Press: Washington, DC, USA, 1995; ISBN 1-61091-120-2. [Google Scholar]
- Holland, W.H.; Powell, R.B.; Thomsen, J.M.; Monz, C.A. A Systematic Review of the Psychological, Social, and Educational Outcomes Associated with Participation in Wildland Recreational Activities. J. Outdoor Recreat. Educ. Leadersh. 2018, 10. [Google Scholar] [CrossRef]
- De Keukeleire, M.; Vanwambeke, S.O.; Somassè, E.; Kabamba, B.; Luyasu, V.; Robert, A. Scouts, Forests, and Ticks: Impact of Landscapes on Human-Tick Contacts. Ticks Tick-borne Dis. 2015, 6, 636–644. [Google Scholar] [CrossRef]
- Gustafson, R.; Forsgren, M.; Gardulf, A.; Granström, M.; Svenungsson, B. Antibody Prevalence and Clinical Manifestations of Lyme Borreliosis and Tick-Borne Encephalitis in Swedish Orienteers. Scand. J. Infect. Dis. 1993, 25, 605–611. [Google Scholar] [CrossRef]
- McGill, S.; Wesslen, L.; Hjelm, E.; Holmberg, M.; Rolf, C.; Friman, G. Serological and Epidemiological Analysis of the Prevalence of Bartonella Spp. Antibodies in Swedish Elite Orienteers 1992–93. Scand. J. Infect. Dis. 2001, 33, 423–428. [Google Scholar] [CrossRef]
- Fahrer, H.; Sauvain, M.J.; Zhioua, E.; Van Hoecke, C.; Gern, L.E. Longterm Survey (7 Years) in a Population at Risk for Lyme Borreliosis: What Happens to the Seropositive Individuals? Eur. J. Epidemiol. 1998, 14, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Fahrer, H.; van der Linden, S.M.; Sauvain, M.-J.; Gern, L.; Zhioua, E.; Aeschlimann, A. The Prevalence and Incidence of Clinical and Asymptomatic Lyme Borreliosis in a Population at Risk. J. Infect. Dis. 1991, 163, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Zhioua, E.; Gern, L.; Aeschlimann, A.; Sauvain, M.J.; Van der Linden, S.; Fahrer, H. Longitudinal Study of Lyme Borreliosis in a High Risk Population in Switzerland. Parasite 1998, 5, 383–386. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.; Boudreau, C.R.; Patterson, J.W.; Bradet-Legris, J.; Lloyd, V.K. Citizen Science and Community Engagement in Tick Surveillance—A Canadian Case Study. Healthcare 2018, 6, 22. [Google Scholar] [CrossRef] [PubMed]
- Pearson, G.A. Alternative Facts and Entomological Engagement. Ann. Entomol. Soc. Am. 2019, 112, 85–88. [Google Scholar] [CrossRef]
- Sakamoto, J.M. Progress, Challenges, and the Role of Public Engagement to Improve Tick-Borne Disease Literacy. Curr. Opin. Insect Sci. 2018, 28, 81–89. [Google Scholar] [CrossRef]
- Gray, J.; Stanek, G.; Kundi, M.; Kocianova, E. Dimensions of Engorging Ixodes Ricinus as a Measure of Feeding Duration. Int. J. Med. Microbiol. 2005, 295, 567–572. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Slunge, D.; Boman, A. Learning to Live with Ticks? The Role of Exposure and Risk Perceptions in Protective Behaviour against Tick-Borne Diseases. PLoS ONE 2018, 13, e0198286. [Google Scholar] [CrossRef] [PubMed]
- Septfons, A.; Paty, M.-C.; De Valk, H.; Couturier, E.; Gauthier, A. Pratiques de prévention et connaissance de la borréliose de Lyme: Baromètre santé 2016. Bull. Epidémiologique Hebd. 2018, 19–20, 419–427. [Google Scholar]
- Jore, S.; Vanwambeke, S.O.; Slunge, D.; Boman, A.; Krogfelt, K.A.; Jepsen, M.T.; Vold, L. Spatial Tick Bite Exposure and Associated Risk Factors in Scandinavia. Infect. Ecol. Epidemiol. 2020, 10, 1764693. [Google Scholar] [CrossRef]
- Millet, I.; Ragionieri, M.; Tomassone, L.; Trentin, C.; Mannelli, A. Assessment of the Exposure of People to Questing Ticks Carrying Agents of Zoonoses in Aosta Valley, Italy. Vet. Sci. 2019, 6, 28. [Google Scholar] [CrossRef] [PubMed]
- Lauterbach, R.; Wells, K.; O’Hara, R.B.; Kalko, E.K.V.; Renner, S.C. Variable Strength of Forest Stand Attributes and Weather Conditions on the Questing Activity of Ixodes Ricinus Ticks over Years in Managed Forests. PLoS ONE 2013, 8, e55365. [Google Scholar] [CrossRef]
- Li, S.; Heyman, P.; Cochez, C.; Simons, L.; Vanwambeke, S.O. A Multi-Level Analysis of the Relationship between Environmental Factors and Questing Ixodes Ricinus Dynamics in Belgium. Parasites Vectors 2012, 5, 149. [Google Scholar] [CrossRef] [PubMed]
- Medlock, J.M.; Hansford, K.M.; Bormane, A.; Derdakova, M.; Estrada-Peña, A.; George, J.-C.; Golovljova, I.; Jaenson, T.G.T.; Jensen, J.-K.; Jensen, P.M.; et al. Driving Forces for Changes in Geographical Distribution of Ixodes Ricinus Ticks in Europe. Parasites Vectors 2013, 6, 1. [Google Scholar] [CrossRef]
- Vourc’h, G.; Abrial, D.; Bord, S.; Jacquot, M.; Masséglia, S.; Poux, V.; Pisanu, B.; Bailly, X.; Chapuis, J.-L. Mapping Human Risk of Infection with Borrelia Burgdorferi Sensu Lato, the Agent of Lyme Borreliosis, in a Periurban Forest in France. Ticks Tick-Borne Dis. 2016, 7, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Bournez, L.; Umhang, G.; Moinet, M.; Boucher, J.-M.; Demerson, J.-M.; Caillot, C.; Legras, L.; Devillers, E.; Hansmann, Y.; Velay, A.; et al. Disappearance of TBEV Circulation among Rodents in a Natural Focus in Alsace, Eastern France. Pathogens 2020, 9, 930. [Google Scholar] [CrossRef] [PubMed]
- CNR Borrelia Densité Moyenne En Nymphes Ixodes Ricinus En Alsace Depuis 2013. Available online: www.chru-strasbourg.fr/Les-centres-de-reference/Borrelia (accessed on 17 December 2020).
- Sonenshine, D.E.; Roe, R.M. (Eds.) Biology of Ticks, 2nd ed.; Oxford University Press: New York, NY, USA, 2014; ISBN 978-0-19-974405-3. [Google Scholar]
- Brunner, J.L.; Ostfeld, R.S. Multiple Causes of Variable Tick Burdens on Small-Mammal Hosts. Ecology 2008, 89, 2259–2272. [Google Scholar] [CrossRef]
- Perkins, S.E.; Cattadori, I.M.; Tagliapietra, V.; Rizzoli, A.P.; Hudson, P.J. Empirical Evidence for Key Hosts in Persistence of a Tick-Borne Disease. Int. J. Parasitol. 2003, 33, 909–917. [Google Scholar] [CrossRef]
- Randolph, S.E.; Miklisova, D.; Lysy, J.; Rogers, D.J.; Labuda, M. Incidence from Coincidence: Patterns of Tick Infestations on Rodents Facilitate Transmission of Tick-Borne Encephalitis Virus. Parasitology 1999, 118, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Woolhouse, M.E.; Dye, C.; Etard, J.-F.; Smith, T.; Charlwood, J.D.; Garnett, G.P.; Hagan, P.; Hii, J.L.K.; Ndhlovu, P.D.; Quinnell, R.J. Heterogeneities in the Transmission of Infectious Agents: Implications for the Design of Control Programs. Proc. Natl. Acad. Sci. USA 1997, 94, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Telford, S.R., 3rd; Urioste, S.S.; Spielman, A. Clustering of Host-Seeking Nymphal Deer Ticks (Ixodes Dammini) Infected by Lyme Disease Spirochetes (Borrelia Burgdorferi). Am. J. Trop. Med. Hyg. 1992, 47, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Burke, G.; Wikel, S.K.; Spielman, A.; Telford, S.R.; McKay, K.; Krause, P.J. Hypersensitivity to Ticks and Lyme Disease Risk. Emerg. Infect. Dis. 2005, 11, 36–41. [Google Scholar] [CrossRef]
- Dautel, H.; Dippel, C.; Werkhausen, A.; Diller, R. Efficacy Testing of Several Ixodes Ricinus Tick Repellents: Different Results with Different Assays. Ticks Tick-Borne Dis. 2013, 4, 256–263. [Google Scholar] [CrossRef]
- Pages, F.; Dautel, H.; Duvallet, G.; Kahl, O.; de Gentile, L.; Boulanger, N. Tick Repellents for Human Use: Prevention of Tick Bites and Tick-Borne Diseases. Vector-Borne Zoonotic Dis. 2014, 14, 85–93. [Google Scholar] [CrossRef]
- Faulde, M.; Scharninghausen, J.; Tisch, M. Preventive Effect of Permethrin-Impregnated Clothing to Ixodes Ricinus Ticks and Associated Borrelia Burgdorferi s.l. in Germany. Int. J. Med. Microbiol. 2008, 298, 321–324. [Google Scholar] [CrossRef]
- Miller, N.J.; Rainone, E.E.; Dyer, M.C.; González, M.L.; Mather, T.N. Tick Bite Protection With Permethrin-Treated Summer-Weight Clothing. J. Med. Entomol. 2011, 48, 327–333. [Google Scholar] [CrossRef]
- Staub, D.; Debrunner, M.; Amsler, L.; Steffen, R. Effectiveness of a Repellent Containing DEET and EBAAP for Preventing Tick Bites. Wilderness Environ. Med. 2002, 13, 12–20. [Google Scholar] [CrossRef]
- Lupi, E.; Hatz, C.; Schlagenhauf, P. The Efficacy of Repellents against Aedes, Anopheles, Culex and Ixodes Spp.—A Literature Review. Travel Med. Infect. Dis. 2013, 11, 374–411. [Google Scholar] [CrossRef]
- Semmler, M.; Abdel-Ghaffar, F.; Al-Rasheid, K.A.S.; Mehlhorn, H. Comparison of the Tick Repellent Efficacy of Chemical and Biological Products Originating from Europe and the USA. Parasitol. Res. 2011, 108, 899–904. [Google Scholar] [CrossRef] [PubMed]
- Mitschler, A.; Grange, F.; Lipsker, D.; Jaulhac, B.; Piemont, Y.; Belanger, P.; Pagnon, X.; Mayer, O.; Guillaume, J.-C. Connaissance et prévention des borrélioses par piqûres de tiques: Enquête dans la population d’une région endémique européenne, l’Alsace. Annales de Dermatologie et de Vénéréologie 2004, 131, 547–553. [Google Scholar] [CrossRef]
- Bron, G.M.; Fernandez, M.d.P.; Larson, S.R.; Maus, A.; Gustafson, D.; Tsao, J.I.; Diuk-Wasser, M.A.; Bartholomay, L.C.; Paskewitz, S.M. Context Matters: Contrasting Behavioral and Residential Risk Factors for Lyme Disease between High-Incidence States in the Northeastern and Midwestern United States. Ticks Tick-Borne Dis. 2020, 11, 101515. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Eggers, P.; Arana, A.; Kresse, B.; Rios, K.; Brown, L.; Sampson, L.; Kploanyi, M. Knowledge and Preventive Behaviors towards Tick-Borne Diseases in Delaware. Ticks Tick-Borne Dis. 2018, 9, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Pańczuk, A.; Tokarska-Rodak, M.; Mikuľáková, W.; Kendrová, L.; Magurová, D. Exposure to Ticks and Undertaking Lyme Borreliosis Prevention Activities among Students from Poland and Slovakia. Ann. Agric. Environ. Med. 2018, 26, 217–221. [Google Scholar] [CrossRef] [PubMed]
| Orienteers (n = 1491) | Participation by Writing on the Banner or by Submitting Ticks (n = 710) | Participation by Writing on the Banner Only ≥ 4 Times (n = 410) | ||||
|---|---|---|---|---|---|---|
| n | % | n | % | n | % | |
| Gender | ||||||
| Men | 772 | 59.6 | 395 | 55.6 | 226 | 55.1 |
| Women | 524 | 40.3 | 315 | 44.4 | 184 | 44.9 |
| Unknown | 195 | |||||
| Age | ||||||
| (7,15] | 268 | 20.4 | 166 | 23.4 | 89 | 21.7 |
| (15,20] | 137 | 10.5 | 73 | 10.3 | 32 | 7.8 |
| (20,35] | 86 | 6.6 | 53 | 7.5 | 34 | 8.3 |
| (35,50] | 340 | 26.0 | 186 | 26.2 | 105 | 25.6 |
| (50,65] | 336 | 25.7 | 173 | 24.4 | 110 | 26.8 |
| (65,82] | 141 | 10.8 | 59 | 8.3 | 40 | 9.8 |
| Unknown | 183 | 0 | 0 | |||
| No. racing rounds run | ||||||
| 1 | 81 | 5.4 | 3 | 0.4 | 0 | 0 |
| 2 | 55 | 3.7 | 14 | 2.0 | 0 | 0 |
| 3 | 87 | 5.8 | 20 | 2.8 | 0 | 0 |
| 4 | 64 | 4.3 | 19 | 2.7 | 6 | 1.5 |
| 5 | 456 | 30.6 | 251 | 35.4 | 135 | 32.9 |
| 6 | 748 | 50.2 | 403 | 56.8 | 269 | 65.6 |
| No. racing rounds with a participation in the study | ||||||
| 1 | - | - | 122 | 17.2 | 0 | 0 |
| 2 | - | - | 87 | 12.3 | 0 | 0 |
| 3 | - | - | 72 | 10.1 | 0 | 0 |
| 4 | - | - | 125 | 17.6 | 122 | 29.8 |
| 5 | - | - | 143 | 20.1 | 140 | 34.1 |
| 6 | - | - | 161 | 22.7 | 148 | 36.1 |
| Racing Round | No. of Orienteers with Ticks | No. of Ticks Collected | ||||||
|---|---|---|---|---|---|---|---|---|
| Tot. | Eng. | Larvae | Nymphs | Females | ||||
| Tot. | Eng. (%) | Tot. | Eng. (%) | Tot. | Eng. (%) | |||
| 2 | 85 | 8 (9.5%) | 34 | 0 | 93 | 9 (9.7%) | 5 | 0 |
| 3 | 112 | 13 (11.6%) | 55 | 0 | 125 | 12 (9.6%) | 3 | 1 (33.3%) |
| 4 | 154 | 14 (9.1%) | 39 | 0 | 229 | 15 (6.6%) | 7 | 0 |
| 5 | 176 | 24 (13.6%) | 228 | 1 (0.4%) | 329 | 28 (8.5%) | 8 | 0 |
| 6 | 149 | 16 (10.7%) | 208 | 1 (0.5%) | 286 | 16 (5.6%) | 1 | 0 |
| Total runners-days | 676 | 75 (11.1%) | 564 | 2 (0.4%) | 1062 | 80 (7.5%) | 24 | 1 (4.2%) |
| Racing Round | No. of Orienteers with Non-Engorged Ticks | No (%) of Orienteers with Non-Engorged | Mean Number of Non-Engorged (L,N,F) per Orienteer Bitten by Ticks | ||||
|---|---|---|---|---|---|---|---|
| Larvae | Nymphs | Female | Larvae | Nymphs | Female | ||
| 2 | 79 | 13 (16.4%) | 67 (84.8%) | 3 (3.8%) | 0.43 | 1.06 | 0.06 |
| 3 | 102 | 24 (23.5%) | 86 (84.3%) | 2 (2.0%) | 0.54 | 1.11 | 0.02 |
| 4 | 144 | 24 (16.7%) | 132 (91.7%) | 7 (4.9 %) | 0.27 | 1.49 | 0.05 |
| 5 | 161 | 47 (29.2%) | 144 (89.4%) | 8 (5.0%) | 1.41 | 1.87 | 0.05 |
| 6 | 138 | 48 (34.8%) | 125 (90.6%) | 1 (0.7%) | 1.50 | 1.96 | 0.01 |
| Total orienteer-days | 624 | 156 (25.0%) | 554 (88.8%) | 21 (3.4%) | 0.90 | 1.57 | 0.04 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Durand, J.; Bournez, L.; Marchand, J.; Schmid, C.; Carravieri, I.; Palin, B.; Galley, C.; Godard, V.; Brun-Jacob, A.; Cosson, J.-F.; et al. Are Orienteers Protected Enough against Tick Bites? Estimating Human Exposure to Tick Bites through a Participative Science Survey during an Orienteering Competition. Int. J. Environ. Res. Public Health 2021, 18, 3161. https://doi.org/10.3390/ijerph18063161
Durand J, Bournez L, Marchand J, Schmid C, Carravieri I, Palin B, Galley C, Godard V, Brun-Jacob A, Cosson J-F, et al. Are Orienteers Protected Enough against Tick Bites? Estimating Human Exposure to Tick Bites through a Participative Science Survey during an Orienteering Competition. International Journal of Environmental Research and Public Health. 2021; 18(6):3161. https://doi.org/10.3390/ijerph18063161
Chicago/Turabian StyleDurand, Jonas, Laure Bournez, Julien Marchand, Claire Schmid, Irene Carravieri, Béatrice Palin, Cyril Galley, Vincent Godard, Annick Brun-Jacob, Jean-François Cosson, and et al. 2021. "Are Orienteers Protected Enough against Tick Bites? Estimating Human Exposure to Tick Bites through a Participative Science Survey during an Orienteering Competition" International Journal of Environmental Research and Public Health 18, no. 6: 3161. https://doi.org/10.3390/ijerph18063161
APA StyleDurand, J., Bournez, L., Marchand, J., Schmid, C., Carravieri, I., Palin, B., Galley, C., Godard, V., Brun-Jacob, A., Cosson, J.-F., & Frey-Klett, P. (2021). Are Orienteers Protected Enough against Tick Bites? Estimating Human Exposure to Tick Bites through a Participative Science Survey during an Orienteering Competition. International Journal of Environmental Research and Public Health, 18(6), 3161. https://doi.org/10.3390/ijerph18063161





