An Analysis of Maternal, Social and Household Factors Associated with Childhood Anemia
Abstract
1. An Analysis of Maternal, Social and Household Factors Associated with Childhood Anemia
Background
2. Methodology
2.1. Dependent and Independent Variables
2.2. Analyses
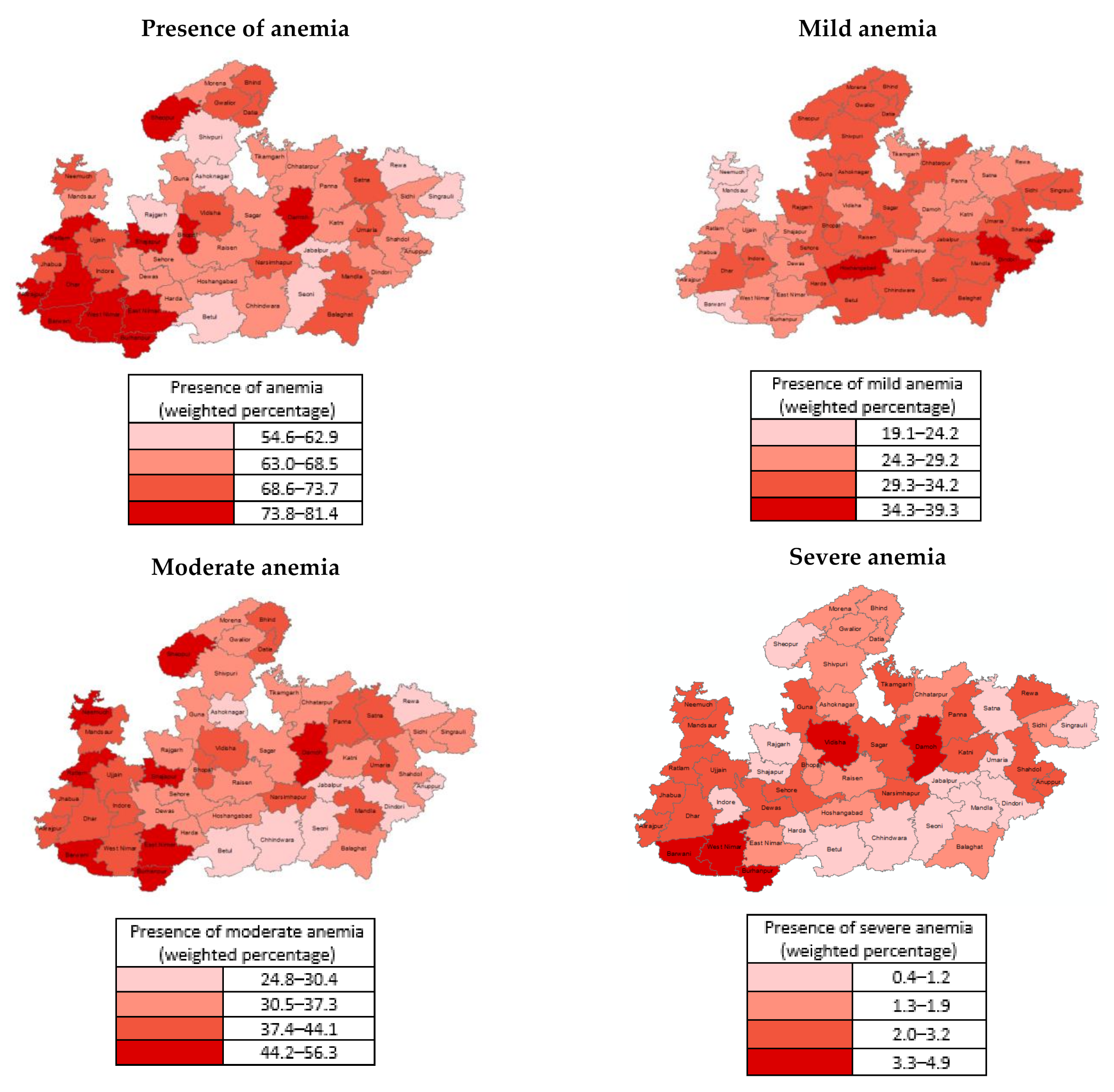
3. Results
Regression Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| NFHS | National Family Health Survey; |
| DHS | Demographic and Health Survey; |
| RKSK | Rashtriya Kishore Swasthya Karyakram; |
| NRC | Nutritional Rehabilitation Centers, |
| AWC | Anganwadi Centers; |
| ST | Scheduled Tribe; |
| LBW | Low Birth Weight; |
| VLBW | Very Low Birth Weight |
References
- Measham, A.R.; Chatterjee, M. Wasting Away: The Crisis of Malnutrition in India; The World Bank: Washington, DC, USA, 1999. [Google Scholar]
- Thakur, N.; Chandra, J.; Pemde, H.; Singh, V. Anemia in severe acute malnutrition. Nutrition 2014, 30, 440–442. [Google Scholar] [CrossRef]
- Varghese, J.S.; Stein, A.D. Malnutrition among women and children in India: Limited evidence of clustering of underweight, anemia, overweight, and stunting within individuals and households at both state and district levels. Am. J. Clin. Nutr. 2019, 109, 1207–1215. [Google Scholar] [CrossRef]
- World Health Organization. Haemoglobin Concentrations for the Diagnosis of Anaemia and Assessment of Severity. Vitamin and Mineral Nutrition Information System. World Health Organization: Geneva, Switzerland, (WHO/NMH/NHD/MNM/11.1). 2011. Available online: http://www.who.int/vmnis/indicators/haemoglobin.pdf (accessed on 22 December 2020).
- Balgir, R.S. Spectrum of Haemoglobinopathies in the state of Orissa, India: A ten years cohort study. J. Assoc. Physicians India 2005, 53, 1021–1026. [Google Scholar]
- De, M.; Halder, A.; Podder, S.; Sen, R.; Chakrabarty, S.; Sengupta, B.; Chakraborty, T.; Das, U.; Talukder, G. Anemia and hemoglobinopathies in tribal population of Eastern and North-eastern India. Hematology 2006, 11, 371–373. [Google Scholar] [CrossRef] [PubMed]
- Iyer, S.; Sakhare, S.; Sengupta, C.; Velumani, A. Hemoglobinopathy in India. Clin. Chim. Acta 2015, 444, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Subramaniam, G.; Girish, M. Iron deficiency anemia in children. Indian J. Pediatrics 2015, 82, 558–564. [Google Scholar] [CrossRef]
- Corrêa, G.; Das, M.; Kovelamudi, R.; Jaladi, N.; Pignon, C.; Vysyaraju, K.; Yedla, U.; Laxmi, V.; Vemula, P.; Gowthami, V.; et al. High burden of malaria and anemia among tribal pregnant women in a chronic conflict corridor in India. Confl. Health 2017, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Sekhar, D.L.; Murray-Kolb, L.E.; Kunselman, A.R.; Weisman, C.S.; Paul, I.M. Association between menarche and iron deficiency in non-anemic young women. PLoS ONE 2017, 12, e0177183. [Google Scholar] [CrossRef]
- Dairo, M.D.; Lawoyin, T.O. Socio-demographic determinants of anaemia in pregnancy at primary care level: A study in urban and rural Oyo State, Nigeria. Afr. J. Med. Med. Sci. 2004, 33, 213–217. [Google Scholar]
- Elmardi, K.A.; Adam, I.; Malik, E.M.; Abdelrahim, T.A.; Elhag, M.S.; Ibrahim, A.A.; Babiker, M.A.; Elhassan, A.H.; Kafy, H.T.; Elshafie, A.T.; et al. Prevalence and determinants of anaemia in women of reproductive age in Sudan: Analysis of a cross-sectional household survey. BMC Public Health 2020, 20, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Skolmowska, D.; Głąbska, D. Analysis of heme and non-heme iron intake and iron dietary sources in adolescent menstruating females in a national polish sample. Nutrients 2019, 11, 1049. [Google Scholar] [CrossRef]
- Bentley, M.E.; Griffiths, P.L. The burden of anemia among women in India. Eur. J. Clin. Nutr. 2003, 57, 52–60. [Google Scholar] [CrossRef]
- Kedar, P.S.; Harigae, H.; Ito, E.; Muramatsu, H.; Kojima, S.; Okuno, Y.; Fujiwara, T.; Dongerdiye, R.; Warang, P.P.; Madkaikar, M.R. Study of pathophysiology and molecular characterization of congenital anemia in India using targeted next-generation sequencing approach. Int. J. Hematol. 2019, 110, 618–626. [Google Scholar] [CrossRef]
- Mahajan, S.D.; Singh, S.; Shah, P.; Gupta, N.; Kochupillai, N. Effect of maternal malnutrition and anemia on the endocrine regulation of fetal growth. Endocr. Res. 2004, 30, 189–203. [Google Scholar] [CrossRef]
- Mudur, G. India launches national rural health mission. BMJ 2005, 330, 920. [Google Scholar] [CrossRef][Green Version]
- Singh, K.; Badgaiyan, N.; Ranjan, A.; Dixit, H.O.; Kaushik, A.; Kushwaha, K.P.; Aguayo, V.M. Management of children with severe acute malnutrition: Experience of Nutrition Rehabilitation Centers in Uttar Pradesh, India. Indian Pediatr. 2014, 51, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, V.; Sahoo, D.P.; Parida, S.P. India steps ahead to curb anemia: Anemia Mukt Bharat. Indian J. Community Health 2018, 30, 312–316. [Google Scholar]
- Deb, S. Implementation of national iron plus initiative for child health: Challenges ahead. Indian J. Public Health 2015, 59, 1. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Weekly Iron-Folic Acid Supplementation (WIFS) in Women of Reproductive age: Its Role in Promoting Optimal Maternal and Child Health (No.WHO/NMH/NHD/MNM/09.2); World Health Organization: Geneva, Switzerland, 2009. [Google Scholar]
- Kumar, A. National nutritional anaemia control programme in India. Indian J. Public Health 1999, 43, 3. [Google Scholar]
- Anemia Mukt Bharat; Ministry of Health and Family Welfare, Government of India. Intensified National Iron Plus Initiative (I-NIPI); Operational guidelines for program managers; Ministry of Health and Family Welfare, Government of India: New Delhi, India, 2018.
- Kapil, U.; Kapil, R.; Gupta, A. Prevention and control of anemia amongst children and adolescents: Theory and practice in India. Indian J. Pediatr. 2019, 86, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Onyeneho, N.G.; Ozumba, B.C.; Subramanian, S.V. Determinants of childhood anemia in India. Sci. Rep. 2019, 9, 1–7. [Google Scholar] [CrossRef]
- Pasricha, S.R.; Black, J.; Muthayya, S.; Shet, A.; Bhat, V.; Nagaraj, S.; Prashanth, N.S.; Sudarshan, H.; Biggs, B.-A. Determinants of anemia among young children in rural India. Pediatrics 2010, 126, e140–e149. [Google Scholar] [CrossRef] [PubMed]
- Kanjilal, B.; Mazumdar, P.G.; Mukherjee, M.; Rahman, M.H. Nutritional status of children in India: Household socio-economic condition as the contextual determinant. Int. J. Equity Health 2010, 9, 19. [Google Scholar] [CrossRef]
- Sharma, H.; Singh, S.K.; Srivastava, S. Socio-economic inequality and spatial heterogeneity in anaemia among children in India: Evidence from NFHS-4 (2015–16). Clin. Epidemiol. Glob. Health 2020, 8, 1158–1171. [Google Scholar] [CrossRef]
- Kotecha, P.V. Nutritional anemia in young children with focus on Asia and India. Indian J. Community Med. Off. Publ. Indian Assoc. Prev. Soc. Med. 2011, 36, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Pollitt, E.; Viteri, F.; Saco-Pollitt, C.; Leibel, R.L. Behavioral Effects of Iron Deficiency Anemia in Children; Raven Press: New York, NY, USA, 1982. [Google Scholar]
- Zavaleta, N.; Astete-Robilliard, L. Effect of anemia on child development: Long-term consequences. Rev. Peru. Med. Exp. Salud Publica 2017, 34, 716–722. [Google Scholar] [CrossRef]
- Wagstaff, A. Socioeconomic inequalities in child mortality: Comparisons across nine developing countries. Bull. World Health Organ. 2000, 78, 19–29. [Google Scholar]
- Bharati, P.; Som, S.; Chakrabarty, S.; Bharati, S.; Pal, M. Prevalence of anemia and its determinants among nonpregnant and pregnant women in India. Asia Pac. J. Public Health 2008, 20, 347–359. [Google Scholar] [CrossRef]
- Kaur, K. Anaemia ‘a silent killer’ among women in India: Present scenario. Eur. J. Zool. Res. 2014, 3, 32–36. [Google Scholar]
- Toteja, G.S.; Singh, P.; Dhillon, B.S.; Saxena, B.N.; Ahmed, F.U.; Singh, R.P.; Prakash, B.; Vijayaraghavan, K.; Singh, Y.; Rauf, A.; et al. Prevalence of anemia among pregnant women and adolescent girls in 16 districts of India. Food Nutr. Bull. 2006, 27, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Self, S.; Grabowski, R. Female autonomy and health care in developing countries. Rev. Dev. Econ. 2012, 16, 185–198. [Google Scholar] [CrossRef]
- Adhikari, T.; Vir, S.C.; Pandey, A.; Yadav, R.J. Undernutrition in Children under Two Years (U2) in India: An Analysis of Determinants. Stat. Appl. 2014, 13, 25–36. [Google Scholar]
- Kalaivani, K. Prevalence & consequences of anaemia in pregnancy. Indian J. Med. Res. 2009, 130, 627–633. [Google Scholar]
- Kapur, D.; Agarwal, K.N.; Sharma, S.; Kela, K.; Kaur, I. Iron status of children aged 9–36 months in an urban slum Integrated Child Development Services project in Delhi. Indian Pediatr. 2002, 39, 136–144. [Google Scholar]
- Kumar, K.J.; Asha, N.; Murthy, D.S.; Sujatha, M.S.; Manjunath, V.G. Maternal anemia in various trimesters and its effect on newborn weight and maturity: An observational study. Int. J. Prev. Med. 2013, 4, 193. [Google Scholar] [PubMed]
- Ray, R. Mother’s autonomy and child anemia: A case study from India. Child. Youth Serv. Rev. 2020, 112, 104537. [Google Scholar] [CrossRef]
- Kamble, Á.; Chaturvedi, P. Epidemiology of sickle cell disease in a rural hospital of central India. Indian Pediatr. 2000, 37, 391–396. [Google Scholar]
- Caulfield, L.E.; de Onis, M.; Blössner, M.; Black, R.E. Undernutrition as an underlying cause of child deaths associated with diarrhea, pneumonia, malaria, and measles. Am. J. Clin. Nutr. 2004, 80, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Nandy, S.; Irving, M.; Gordon, D.; Subramanian, S.V.; Smith, G.D. Poverty, child undernutrition and morbidity: New evidence from India. Bull. World Health Organ. 2005, 83, 210–216. [Google Scholar]
- Benson, T.; Shekar, M. Trends and Issues in Child Undernutrition. In Disease and Mortality in Sub-Saharan Africa, 2nd ed.; World Bank Publications: Washington, DC, USA, 2006. [Google Scholar]
- International Institute for Population Sciences (IIPS). National Family Health Survey (NFHS-4), 2015–2016; IIPS: Mumbai, India, 2017. [Google Scholar]
- International Institute for Population Sciences—IIPS/India; ICF. IIndia National Family Health Survey NFHS-4 2015-16. Mumbai, India: IIPS and ICF. 2017. Available online: http://dhsprogram.com/pubs/pdf/FR339/FR339.pdf (accessed on 15 March 2021).
- Pullum, T.W.; Collison, D.K.; Namaste, S.; Garrett, D. Hemoglobin Data in DHS Surveys: Intrinsic Variation and Measurement Error; International Health and Development: Rockville, MD, USA, 2017. [Google Scholar]
- Taneja, G.; Sridhar, V.S.; Mohanty, J.S.; Joshi, A.; Bhushan, P.; Jain, M.; Gupta, S.; Khera, A.; Kumar, R.; Gera, R. India’s RMNCH+A Strategy: Approach, learnings and limitations. BMJ Glob. Health 2019, 4, e001162. [Google Scholar] [CrossRef]
- Vart, P.; Jaglan, A.; Shafique, K. Caste-based social inequalities and childhood anemia in India: Results from the National Family Health Survey (NFHS) 2005–2006. BMC Public Health 2015, 15, 537. [Google Scholar] [CrossRef] [PubMed]
- Sachdev, H.P.S.; Gera, T. Preventing childhood anemia in India: Iron supplementation and beyond. Eur. J. Clin. Nutr. 2013, 67, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Bhujade, R.; Mishra, B.N.; Ibrahim, T.; Sinha, A.; Chouhan, D.S. Can Sever Acute Malnourished children be effectively rehabilitated physically, biochemically and developmentally at nutritional rehabilitation centers: A follow up study from Ujjain. J. Fam. Med. Prim. Care 2021, 10, 343. [Google Scholar] [CrossRef]
- Dasgupta, R.; Ahuja, S.; Yumnam, V. Can nutrition rehabilitation centers address severe malnutrition in India? Indian Pediatrics 2014, 51, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Singh, R. Monitoring Development through GIS Visualisation. Soc. Chang. 2016, 46, 27–45. [Google Scholar] [CrossRef]
- Yadav, R.; Lazarus, M.; Ghanghoria, P.; Singh, M.P.S.S.; Gupta, R.B.; Kumar, S.; Sharma, R.K.; Shanmugam, R. Sickle cell disease in Madhya Pradesh, Central India: A comparison of clinical profile of sickle cell homozygote vs. sickle-beta thalassaemia individuals. Hematology 2016, 21, 558–563. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, D.; Mukherjee, M.B. Sickle cell disease in India. Curr. Opin. Hematol. 2002, 9, 117–122. [Google Scholar] [CrossRef]
| Characteristic | Total (n = 20,102) | Anemic (n = 14,015) | Non Anemic (n = 6087) | |||
|---|---|---|---|---|---|---|
| n | Weighted % | n | Weighted % | n | Weighted % | |
| Current age of the mother | ||||||
| 15–19 years | 368 | 1.8 | 293 | 2.0 | 75 | 1.2 |
| 20–24 years | 7116 | 35.6 | 5304 | 38.3 | 1812 | 29.4 |
| 25–29 years | 7954 | 39.3 | 5302 | 37.5 | 2652 | 43.3 |
| 30–34 years | 3272 | 16.4 | 2214 | 15.7 | 1058 | 17.9 |
| 35–39 years | 1035 | 5.2 | 662 | 4.8 | 373 | 6.1 |
| 40–44 years | 267 | 1.3 | 189 | 1.3 | 78 | 1.3 |
| 45–49 years | 90 | 0.4 | 51 | 0.3 | 39 | 0.7 |
| Mother’s education | ||||||
| No education | 7276 | 34.3 | 5271 | 35.6 | 2005 | 31.4 |
| Primary | 3794 | 18.6 | 2687 | 18.8 | 1107 | 17.9 |
| Secondary | 7840 | 40.9 | 5340 | 40.2 | 2500 | 42.4 |
| Higher | 1192 | 6.3 | 717 | 5.4 | 475 | 8.3 |
| Mother’s anemia status | ||||||
| Not anemic | 8225 | 42.4 | 5202 | 38.7 | 3023 | 50.8 |
| Mild | 8395 | 41.6 | 6079 | 43.2 | 2316 | 37.9 |
| Moderate | 3181 | 14.9 | 2502 | 16.9 | 679 | 10.5 |
| Severe | 231 | 1.1 | 179 | 1.2 | 52 | 0.8 |
| Missing: 0.35% (n = 70) | ||||||
| Place of residence | ||||||
| Rural | 15,187 | 74.1 | 10,729 | 75.2 | 4458 | 71.7 |
| Urban | 4915 | 25.9 | 3286 | 24.8 | 1629 | 28.3 |
| Sanitation facility $ | ||||||
| Improved sanitation facility | 6630 | 35.9 | 4398 | 34.3 | 2232 | 39.4 |
| Not improved sanitation facility | 331 | 1.8 | 231 | 1.7 | 100 | 1.8 |
| No toilet facility-Bush/fields | 12,180 | 62.4 | 8712 | 64.0 | 3468 | 58.9 |
| Missing: 5.02% (n = 961) | ||||||
| Drinking water source # | ||||||
| Improved water source | 15,915 | 83.1 | 11,056 | 82.9 | 4859 | 83.6 |
| No improved water source | 3226 | 16.9 | 2285 | 17.1 | 941 | 16.4 |
| Missing: 5.02% (n = 961) | ||||||
| Religion and caste | ||||||
| Hindu high caste | 1876 | 10.0 | 1220 | 9.4 | 656 | 11.3 |
| Hindu lower caste | 10,928 | 57.2 | 7372 | 55.6 | 3556 | 60.7 |
| Hindu ST a | 5203 | 23.2 | 3973 | 25.5 | 1230 | 18.0 |
| Hindu others | 351 | 1.7 | 246 | 1.8 | 105 | 1.5 |
| Muslim | 1597 | 7.1 | 1111 | 6.9 | 486 | 7.5 |
| Others | 147 | 0.8 | 93 | 0.7 | 54 | 0.9 |
| Wealth quintile | ||||||
| Poorest | 7174 | 34.7 | 5217 | 36.3 | 1957 | 31.1 |
| Poorer | 4733 | 23.6 | 3351 | 23.8 | 1382 | 22.9 |
| Middle | 3133 | 15.7 | 2188 | 15.6 | 945 | 15.9 |
| Richer | 2706 | 13.7 | 1780 | 13.0 | 926 | 15.2 |
| Richest | 2356 | 12.4 | 1479 | 11.3 | 877 | 15.0 |
| Gender of the child | ||||||
| Female | 9592 | 47.9 | 6663 | 47.7 | 2929 | 48.2 |
| Male | 10,510 | 52.1 | 7352 | 52.3 | 3158 | 51.8 |
| Age of the child | ||||||
| Less than 1 year old | 2222 | 11.1 | 1667 | 12.0 | 555 | 8.9 |
| 1–2 years old | 4413 | 21.9 | 3598 | 25.8 | 815 | 13.1 |
| 2–3 years old | 4469 | 22.3 | 3326 | 23.7 | 1143 | 19.1 |
| 3–4 years old | 4550 | 22.7 | 2920 | 20.9 | 1630 | 26.6 |
| 4–5 years old | 4448 | 22.1 | 2504 | 17.6 | 1944 | 32.2 |
| Birthweight of the child | ||||||
| VLBW * | 752 | 3.8 | 576 | 4.3 | 176 | 2.8 |
| LBW * | 6889 | 34.5 | 4857 | 34.9 | 2032 | 33.5 |
| Normal weight * | 8436 | 42.6 | 5713 | 41.3 | 2723 | 45.6 |
| Overweight * | 234 | 1.2 | 143 | 1.1 | 91 | 1.6 |
| Not weighed at birth | 3145 | 14.5 | 2266 | 15.0 | 879 | 13.2 |
| Don’t know | 646 | 3.4 | 460 | 3.4 | 186 | 3.3 |
| Taking iron pills, sprinkles or syrup | ||||||
| Yes | 5367 | 26.0 | 3749 | 25.9 | 1618 | 26.3 |
| No | 14,579 | 73.2 | 10,158 | 73.3 | 4421 | 73.0 |
| Don’t know | 156 | 0.8 | 108 | 0.8 | 48 | 0.7 |
| Drugs for intestinal parasites in last 6 months | ||||||
| Yes | 6051 | 29.7 | 4212 | 29.5 | 1839 | 30.1 |
| No | 13,889 | 69.4 | 9693 | 69.6 | 4196 | 69.0 |
| Don’t know | 162 | 0.9 | 110 | 0.9 | 52 | 0.9 |
| Presence of stunting | ||||||
| Not stunted | 10,651 | 53.3 | 7020 | 50.0 | 3631 | 60.5 |
| Flagged for height for age | 916 | 4.4 | 688 | 4.8 | 228 | 3.3 |
| Moderately or severely stunted | 4711 | 23.6 | 3322 | 23.9 | 1389 | 22.7 |
| Severely stunted | 3824 | 18.8 | 2985 | 21.2 | 839 | 13.5 |
| Presence of wasting | ||||||
| Normal weight/no wasting | 14,106 | 70.7 | 9697 | 69.4 | 4409 | 73.6 |
| Flagged | 916 | 4.4 | 688 | 4.8 | 228 | 3.3 |
| Moderately | 3151 | 15.5 | 2229 | 15.8 | 922 | 14.8 |
| Severely wasted | 1676 | 8.1 | 1214 | 8.5 | 462 | 7.3 |
| Overweight | 253 | 1.4 | 187 | 1.5 | 66 | 1.1 |
| Characteristic | Mild Anemic (n = 5898) Hb 10–10.9 g/dL | Moderately Anemic (n = 7701) Hb 7.1–9.9 g/dL | Severely Anemic (n = 416) Hb < 7.1 g/dL | |||
|---|---|---|---|---|---|---|
| n | % | n | % | n | % | |
| Current age of the mother | ||||||
| 15–19 years | 103 | 1.7 | 172 | 2.2 | 18 | 4.4 |
| 20–24 years | 2087 | 35.8 | 3056 | 40.1 | 161 | 40.2 |
| 25–29 years | 2300 | 38.6 | 2859 | 36.9 | 143 | 33.6 |
| 30–34 years | 997 | 16.8 | 1147 | 14.9 | 70 | 15.8 |
| 35–39 years | 306 | 5.4 | 338 | 4.3 | 18 | 4.6 |
| 40–44 years | 83 | 1.4 | 101 | 1.3 | 5 | 1.3 |
| 45–49 years | 22 | 0.3 | 28 | 0.3 | 1 | 0.1 |
| Mother’s education | ||||||
| No education | 2084 | 33.5 | 3004 | 36.8 | 183 | 43.9 |
| Primary | 1152 | 19.2 | 1466 | 18.7 | 69 | 16.4 |
| Secondary | 2306 | 41.2 | 2887 | 39.6 | 147 | 35.6 |
| Higher | 356 | 6.1 | 344 | 4.9 | 17 | 4.2 |
| Mother’s anemia status | ||||||
| Not anemic | 49 | 0.9 | 119 | 1.4 | 11 | 2.1 |
| Mild | 863 | 14.0 | 1515 | 18.6 | 124 | 27.6 |
| Moderate | 2555 | 43.1 | 3362 | 43.5 | 162 | 40.0 |
| Severe | 2405 | 42.0 | 2680 | 36.5 | 117 | 30.2 |
| Missing: 0.35% (n = 70) | ||||||
| Place of residence | ||||||
| Rural | 4517 | 75.3 | 5909 | 75.2 | 303 | 71.9 |
| Urban | 1381 | 24.7 | 1792 | 24.8 | 113 | 28.1 |
| Sanitation facility | ||||||
| Improved sanitation facility | 1943 | 35.9 | 2320 | 33.1 | 135 | 33.4 |
| Not improved sanitation facility | 88 | 1.5 | 136 | 1.9 | 7 | 1.8 |
| No toilet facility-Bush/fields | 3589 | 62.6 | 4867 | 65.0 | 256 | 64.9 |
| Missing: 5.02% (n = 961) | ||||||
| Drinking water source | ||||||
| Improved drinking water source | 4633 | 82.6 | 6091 | 83.1 | 332 | 82.9 |
| No improved drinking water source | 987 | 17.4 | 1232 | 16.9 | 66 | 17.1 |
| Missing: 5.02% (n = 961) | ||||||
| Religion and caste | ||||||
| Hindu high caste | 542 | 10.1 | 651 | 9.0 | 27 | 7.2 |
| Hindu lower caste | 3205 | 56.3 | 3958 | 55.1 | 209 | 54.1 |
| Hindu ST | 1588 | 24.5 | 2256 | 26.2 | 129 | 28.8 |
| Hindu others | 108 | 1.8 | 132 | 1.7 | 6 | 1.6 |
| Muslim | 409 | 6.4 | 659 | 7.3 | 43 | 8.0 |
| Others | 46 | 0.8 | 45 | 0.7 | 2 | 0.3 |
| Wealth quintile | ||||||
| Poorest | 2125 | 35.2 | 2941 | 37.2 | 151 | 35.5 |
| Poorer | 1385 | 23.3 | 1861 | 24.2 | 105 | 25.5 |
| Middle | 907 | 15.3 | 1205 | 15.6 | 76 | 19.2 |
| Richer | 765 | 13.5 | 970 | 12.8 | 45 | 9.6 |
| Richest | 716 | 12.6 | 724 | 10.3 | 39 | 10.3 |
| Gender of the child | ||||||
| Female | 3001 | 50.4 | 4094 | 53.2 | 257 | 60.9 |
| Male | 2897 | 49.6 | 3607 | 46.8 | 159 | 39.1 |
| Age of the child | 5898 | |||||
| Less than 1 year old | 1176 | 19.9 | 2262 | 29.8 | 160 | 37.9 |
| 1–2 years old | 1299 | 22.4 | 1919 | 24.6 | 108 | 25.2 |
| 2–3 years old | 1405 | 23.8 | 1460 | 19.1 | 55 | 14.0 |
| 3–4 years old | 1381 | 23.1 | 1083 | 13.7 | 40 | 9.4 |
| 4–5 years old | 637 | 10.8 | 977 | 12.9 | 53 | 13.5 |
| Birthweight of the child | ||||||
| VLBW * | 197 | 3.6 | 338 | 4.5 | 41 | 10.4 |
| LBW * | 2010 | 34.1 | 2703 | 35.7 | 144 | 33.4 |
| Normal weight * | 2544 | 43.6 | 3036 | 39.9 | 133 | 32.7 |
| Overweight * | 54 | 1.0 | 82 | 1.1 | 7 | 2.0 |
| Not weighed at birth | 903 | 14.5 | 1289 | 15.3 | 74 | 17.8 |
| Don’t know | 190 | 3.3 | 253 | 3.5 | 17 | 3.6 |
| Taking iron pills, sprinkles or syrup | ||||||
| Yes | 1580 | 25.9 | 2056 | 25.9 | 113 | 25.5 |
| No | 4278 | 73.4 | 5582 | 73.3 | 298 | 73.2 |
| Don’t know | 40 | 0.7 | 63 | 0.8 | 5 | 1.3 |
| Drugs for intestinal parasites in last 6 months | ||||||
| Yes | 1777 | 29.6 | 2296 | 29.3 | 139 | 32.6 |
| No | 4065 | 69.2 | 5353 | 70.0 | 275 | 66.9 |
| Don’t know | 56 | 1.1 | 52 | 0.7 | 2 | 0.5 |
| Presence of stunting | ||||||
| Not stunted | 3227 | 54.7 | 3648 | 47.1 | 145 | 36.8 |
| Flagged for height for age | 268 | 4.5 | 385 | 4.9 | 35 | 8.6 |
| Moderately or severely stunted | 987 | 16.6 | 1858 | 24.3 | 140 | 30.9 |
| Severely stunted | 1416 | 24.2 | 1810 | 23.7 | 96 | 23.7 |
| Presence of wasting | ||||||
| Normal weight/no wasting | 4087 | 69.8 | 5334 | 69.4 | 276 | 65.5 |
| Flagged | 268 | 4.5 | 385 | 4.9 | 35 | 8.6 |
| Moderately | 933 | 15.8 | 1237 | 15.8 | 59 | 13.8 |
| Severely wasted | 534 | 8.6 | 637 | 8.3 | 43 | 11.2 |
| Overweight | 76 | 1.3 | 108 | 1.6 | 3 | 1.0 |
| Sociodemographic Characteristics | Model 1 (Anemia vs. No Anemia) | Model 2 (Levels of Anemia vs. No Anemia) | ||
|---|---|---|---|---|
| Anemic (Hb < 11 g/dL) aOR (95% CI) | Mild Anemic (Hb 10–10.9 g/dL) aOR (95% CI) | Moderately Anemic (Hb 7.1–9.9 g/dL) aOR (95% CI) | Severely Anemic (Hb < 7.1 g/dL) aOR (95% CI) | |
| Current age of the mother | ||||
| 15–19 years | 1.19 (0.88, 1.60) | 1.19 (0.84, 1.70) | 1.14 (0.84, 1.56) | 2.08 (1.06, 4.06) |
| 20–24 years | 1.23 (1.13, 1.34) | 1.20 (1.08, 1.32) | 1.26 (1.14, 1.39) | 1.28 (0.97, 1.69) |
| 25–29 years | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) |
| 30–34 years | 1.10 (0.99, 1.22) | 1.10 (0.98, 1.24) | 1.09 (0.97, 1.23) | 1.31 (0.90, 1.89) |
| 35–39 years | 0.98 (0.83, 1.16) | 1.04 (0.86, 1.25) | 0.92 (0.75, 1.13) | 1.05 (0.58, 1.90) |
| 40–44 years | 1.22 (0.89, 1.67) | 1.19 (0.85, 1.65) | 1.26 (0.86, 1.85) | 1.31 (0.47, 3.65) |
| 45–49 years | 0.71 (0.46, 1.10) | 0.66 (0.38, 1.16) | 0.78 (0.46, 1.32) | 0.31 (0.04, 2.55) |
| Mother’s education | ||||
| No education | 1.54 (1.27, 1.87) | 1.36 (1.10, 1.69) | 1.69 (1.33, 2.15) | 2.25 (1.13, 4.48) |
| Primary | 1.53 (1.26, 1.86) | 1.45 (1.17, 1.80) | 1.61 (1.27, 2.05) | 1.58 (0.78, 3.22) |
| Secondary | 1.32 (1.11, 1.58) | 1.27 (1.04, 1.55) | 1.38 (1.11, 1.72) | 1.27 (0.66, 2.45) |
| Higher | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) |
| Mother’s anemia status | ||||
| Not anemic | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) |
| Mild | 1.43 (1.33, 1.55) | 1.33 (1.21, 1.45) | 1.53 (1.40, 1.67) | 1.65 (1.22, 2.23) |
| Moderate | 1.98 (1.76, 2.22) | 1.56 (1.37, 1.78) | 2.32 (2.04, 2.64) | 4.18 (3.00, 5.82) |
| Severe | 1.85 (1.28, 2.67) | 1.25 (0.82, 1.92) | 2.37 (1.58, 3.56) | 4.98 (2.29, 10.79) |
| Place of residence | ||||
| Rural | 0.98 (0.87, 1.11) | 1.06 (0.93, 1.21) | 0.93 (0.81, 1.07) | 0.73 (0.46, 1.16) |
| Urban | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) |
| Sanitation facility | ||||
| Improved sanitation facility | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) |
| Not improved sanitation facility | 0.92 (0.69, 1.22) | 0.82 (0.61, 1.11) | 1.01 (0.72, 1.41) | 0.87 (0.34, 2.21) |
| No toilet facility-Bush/fields | 0.92 (0.81, 1.04) | 0.91 (0.79, 1.06) | 0.93 (0.81, 1.07) | 0.84 (0.56, 1.27) |
| Drinking water source | ||||
| Improved drinking water source | 1.06 (0.96, 1.18) | 1.03 (0.92, 1.15) | 1.09 (0.96, 1.23) | 1.05 (0.76, 1.46) |
| No improved drinking water source | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) |
| Religion and caste | ||||
| Hindu high caste | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) |
| Hindu lower caste | 0.92 (0.80, 1.05) | 0.93 (0.80, 1.08) | 0.90 (0.76, 1.06) | 1.19 (0.73, 1.92) |
| Hindu ST | 1.31 (1.11, 1.54) | 1.31 (1.10, 1.56) | 1.29 (1.06, 1.56) | 1.88 (1.07, 3.29) |
| Hindu others | 1.22 (0.89, 1.68) | 1.26 (0.87, 1.82) | 1.18 (0.83, 1.68) | 1.38 (0.50, 3.83) |
| Muslim | 0.94 (0.77, 1.14) | 0.86 (0.69, 1.07) | 1.00 (0.79, 1.26) | 1.27 (0.66, 2.44) |
| Others | 0.96 (0.62, 1.49) | 1.04 (0.65, 1.67) | 0.89 (0.51, 1.54) | 0.71 (0.15, 3.37) |
| Wealth quintile | ||||
| Middle | 1.07 (0.90, 1.28) | 0.97 (0.80, 1.19) | 1.17 (0.94, 1.44) | 1.29 (0.71, 2.34) |
| Poorer | 1.08 (0.89, 1.31) | 1.00 (0.80, 1.24) | 1.17 (0.93, 1.47) | 1.05 (0.55, 2.00) |
| Poorest | 1.1 (0.90, 1.35) | 1.01 (0.80, 1.27) | 1.22 (0.95, 1.55) | 0.86 (0.43, 1.72) |
| Richer | 0.97 (0.83, 1.13) | 0.93 (0.78, 1.11) | 1.02 (0.84, 1.24) | 0.68 (0.39, 1.18) |
| Richest | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) |
| Gender of the child | ||||
| Female | 1 (0.93, 1.08) | 1.07 (0.99, 1.16) | 0.96 (0.89, 1.04) | 0.71 (0.56, 0.90) |
| Male | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) |
| Age of the child | ||||
| Less than 1 year old | 2.61 (2.27, 3.00) | 1.75 (1.48, 2.06) | 3.73 (3.17, 4.38) | 5.86 (3.63, 9.48) |
| 1–2 years old | 3.59 (3.19, 4.05) | 2.13 (1.85, 2.45) | 5.45 (4.76, 6.24) | 9.71 (6.39, 14.74) |
| 2–3 years old | 2.25 (2.02, 2.49) | 1.63 (1.44, 1.84) | 3.01 (2.66, 3.42) | 4.50 (2.96, 6.85) |
| 3–4 years old | 1.44 (1.30, 1.60) | 1.26 (1.12, 1.43) | 1.67 (1.46, 1.90) | 1.73 (1.12, 2.70) |
| 4–5 years old | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) |
| Birthweight of the child | ||||
| VLBW (Less than 1500 g) | 1.48 (1.20, 1.83) | 1.22 (0.95, 1.56) | 1.64 (1.30, 2.06) | 4.28 (2.67, 6.87) |
| LBW (1500–2500 g) | 1.07 (0.99, 1.17) | 1.01 (0.92, 1.11) | 1.13 (1.02, 1.24) | 1.31 (1.00, 1.73) |
| Normal weight (2500–4000 g) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) |
| Overweight (4000+ grams) | 0.8 (0.58, 1.11) | 0.70 (0.46, 1.06) | 0.86 (0.59, 1.23) | 2.37 (0.94, 6.00) |
| Not weighed at birth | 1.06 (0.94, 1.19) | 1.00 (0.88, 1.14) | 1.10 (0.96, 1.27) | 1.51 (1.03, 2.22) |
| Don’t know | 1.13 (0.92, 1.40) | 1.01 (0.79, 1.30) | 1.24 (0.97, 1.59) | 1.67 (0.92, 3.04) |
| Taking iron pills, sprinkles or syrup | ||||
| Don’t know | 1.06 (0.69, 1.61) | 0.91 (0.56, 1.48) | 1.17 (0.70, 1.96) | 2.55 (0.93, 7.02) |
| Yes | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) |
| No | 1.04 (0.94, 1.15) | 1.06 (0.94, 1.18) | 1.02 (0.91, 1.15) | 1.10 (0.81, 1.51) |
| Drugs for intestinal parasites in last 6 months | ||||
| Don’t know | 1.04 (0.70, 1.53) | 1.44 (0.93, 2.22) | 0.75 (0.45, 1.24) | 0.33 (0.08, 1.32) |
| Yes | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) |
| No | 1.01 (0.92, 1.11) | 1.00 (0.90, 1.12) | 1.02 (0.92, 1.13) | 0.86 (0.65, 1.15) |
| Presence of stunting | ||||
| Not stunted | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) |
| Flagged for height for age | 1.66 (0.87, 3.16) | 1.28 (0.62, 2.64) | 2.19 (1.03, 4.66) | 0.67 (0.10, 4.61) |
| Moderately or severely stunted | 1.27 (1.16, 1.39) | 1.17 (1.05, 1.29) | 1.36 (1.23, 1.51) | 1.66 (1.22, 2.27) |
| Severely stunted | 1.73 (1.55, 1.94) | 1.28 (1.12, 1.46) | 2.14 (1.89, 2.43) | 3.19 (2.36, 4.31) |
| Presence of wasting | ||||
| Normal weight/no wasting | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) | 1.00 (ref) |
| Flagged | 0.92 (0.49, 1.75) | 1.13 (0.55, 2.31) | 0.71 (0.34, 1.50) | 4.86 (0.71, 33.48) |
| Moderately or severely wasted | 1.03 (0.93, 1.14) | 1.07 (0.95, 1.21) | 1.00 (0.89, 1.12) | 0.90 (0.63, 1.28) |
| Overweight | 1.36 (0.93, 2.00) | 1.37 (0.92, 2.04) | 1.39 (0.86, 2.25) | 0.85 (0.25, 2.87) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chandran, V.; Kirby, R.S. An Analysis of Maternal, Social and Household Factors Associated with Childhood Anemia. Int. J. Environ. Res. Public Health 2021, 18, 3105. https://doi.org/10.3390/ijerph18063105
Chandran V, Kirby RS. An Analysis of Maternal, Social and Household Factors Associated with Childhood Anemia. International Journal of Environmental Research and Public Health. 2021; 18(6):3105. https://doi.org/10.3390/ijerph18063105
Chicago/Turabian StyleChandran, Vidya, and Russell S. Kirby. 2021. "An Analysis of Maternal, Social and Household Factors Associated with Childhood Anemia" International Journal of Environmental Research and Public Health 18, no. 6: 3105. https://doi.org/10.3390/ijerph18063105
APA StyleChandran, V., & Kirby, R. S. (2021). An Analysis of Maternal, Social and Household Factors Associated with Childhood Anemia. International Journal of Environmental Research and Public Health, 18(6), 3105. https://doi.org/10.3390/ijerph18063105







