Development of Machine Learning Models for Prediction of Smoking Cessation Outcome
Abstract
1. Introduction
2. Materials and Methods
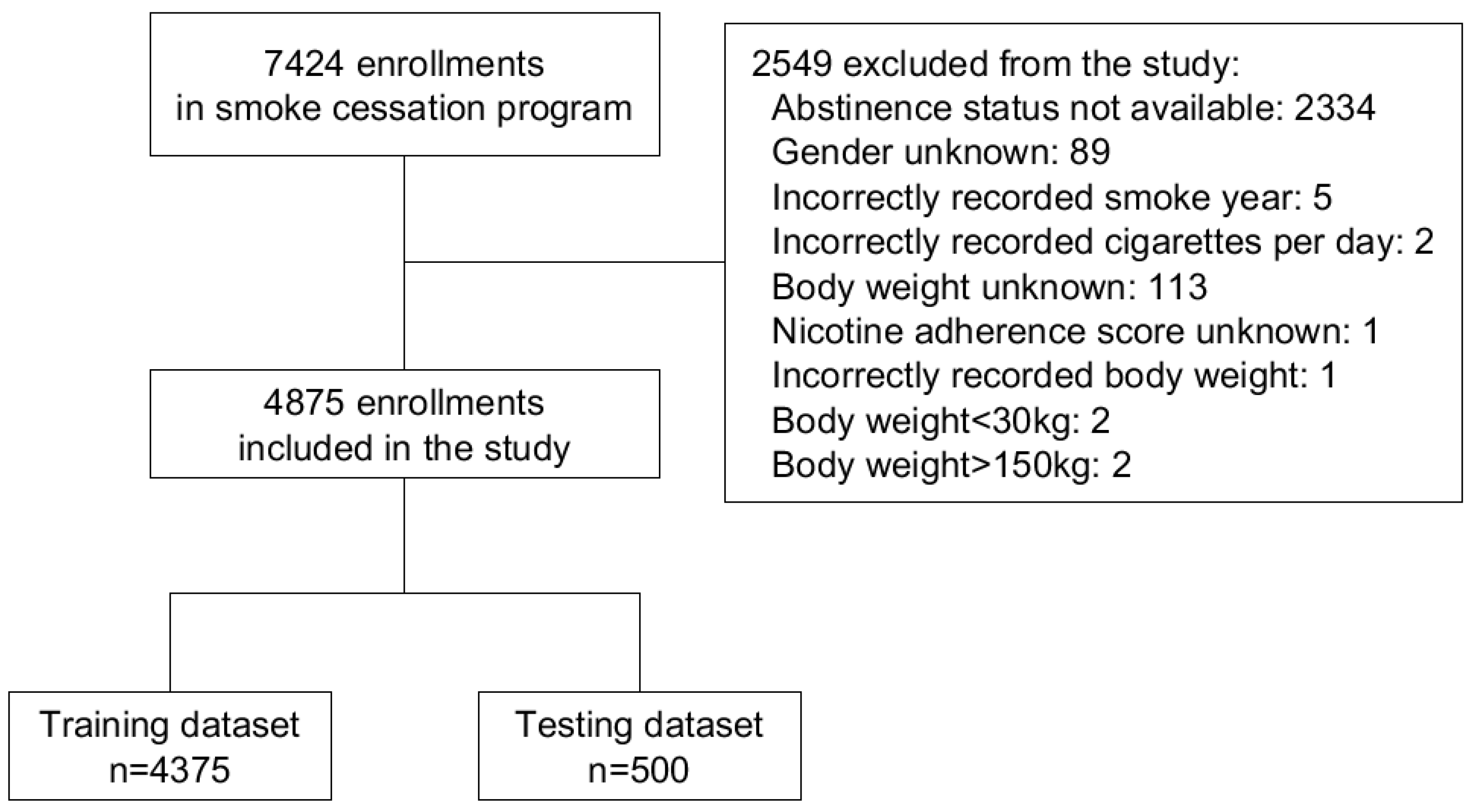
2.1. Data Acquirement
2.2. Feature Selection and Data Preprocessing
2.3. Machine Learning Model Development
2.4. Statistical Analysis
2.5. Application of the Machine Learning Model
3. Results
3.1. Characteristics of Enrolled Data
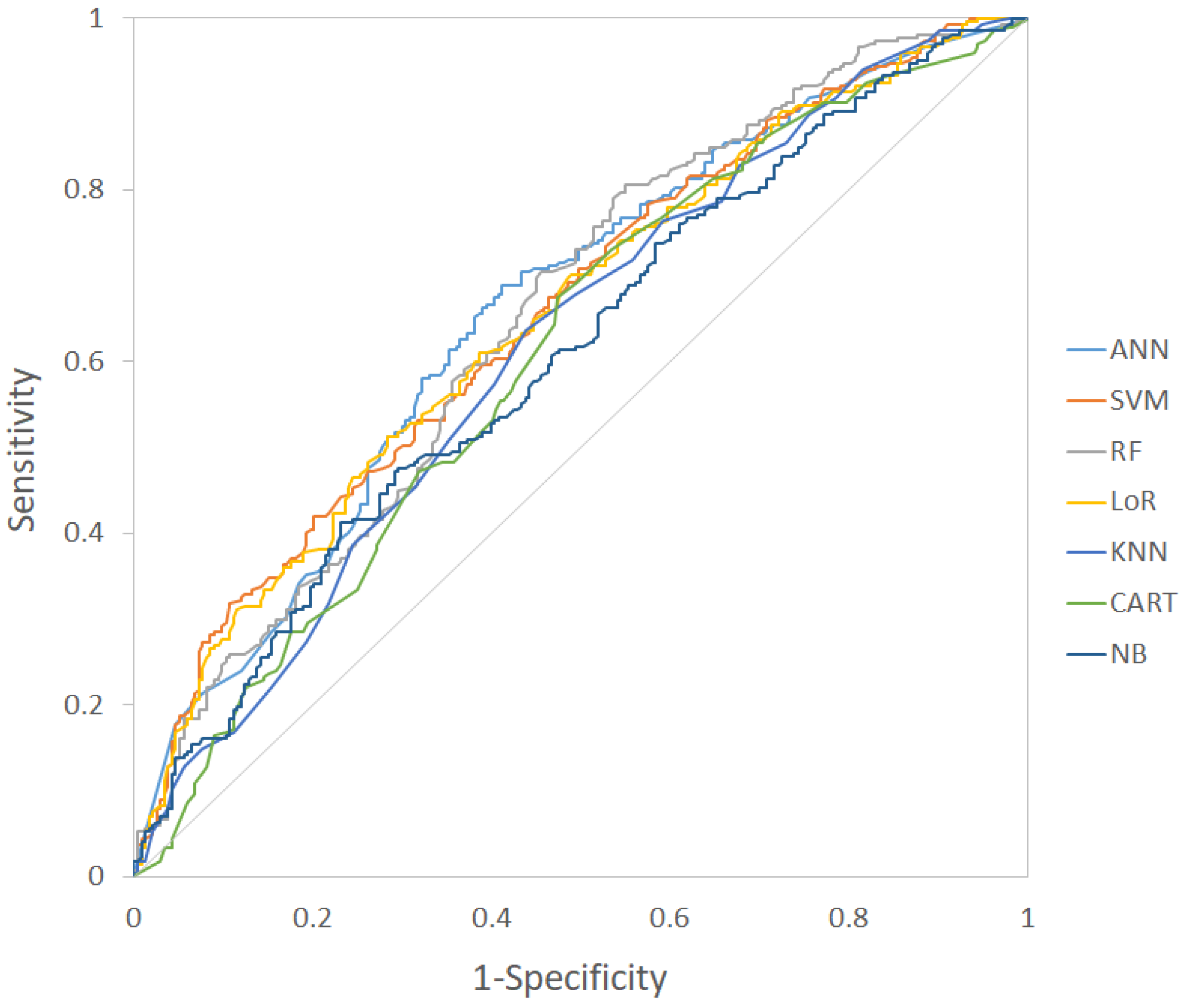
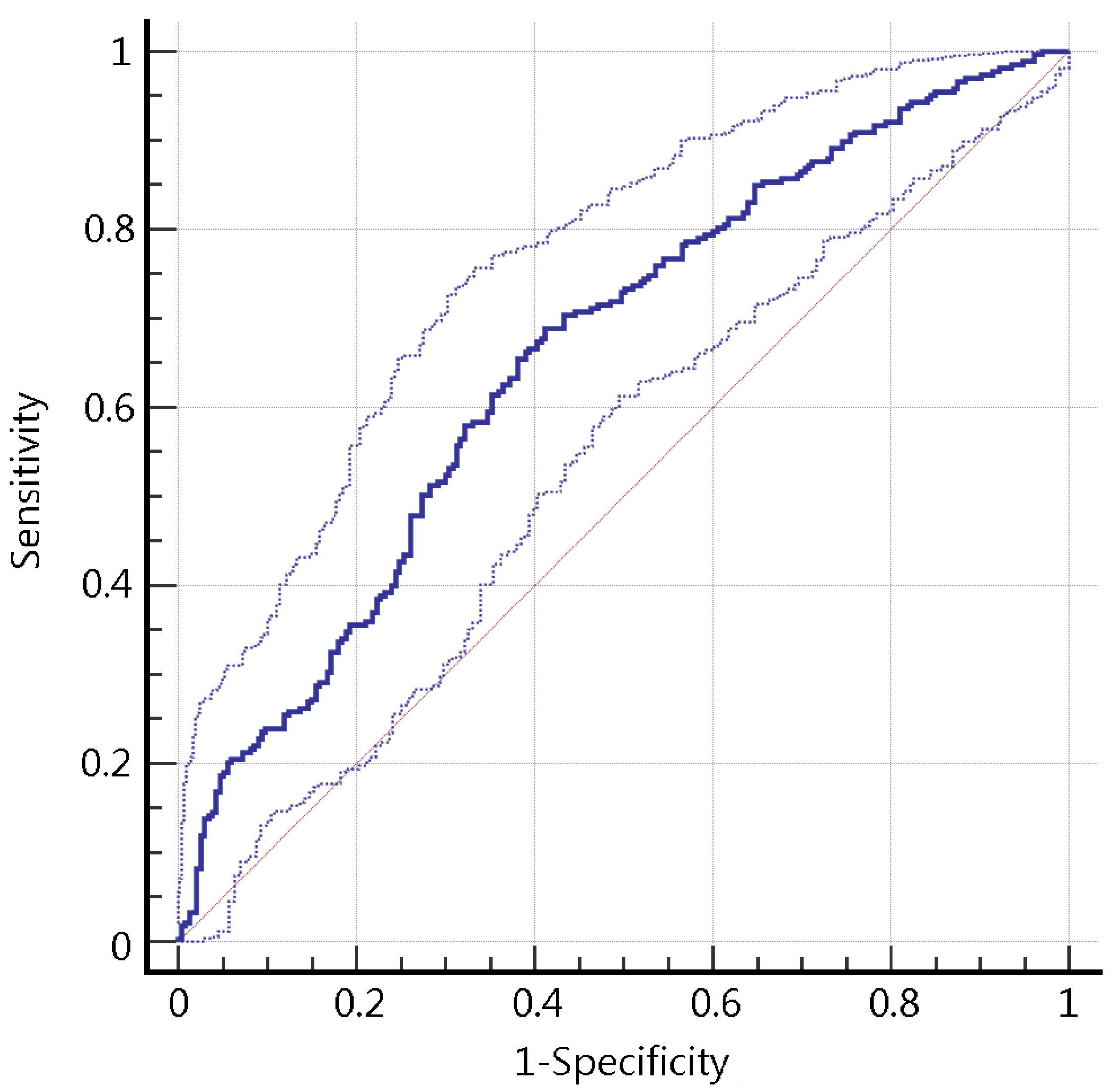
3.2. Model Performance
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Peacock, A.; Leung, J.; Larney, S.; Colledge, S.; Hickman, M.; Rehm, J.; Giovino, G.A.; West, R.; Hall, W.; Griffiths, P. Global statistics on alcohol, tobacco and illicit drug use: 2017 status report. Addiction 2018, 113, 1905–1926. [Google Scholar] [CrossRef] [PubMed]
- Onor, I.O.; Stirling, D.L.; Williams, S.R.; Bediako, D.; Borghol, A.; Harris, M.B.; Darensburg, T.B.; Clay, S.D.; Okpechi, S.C.; Sarpong, D.F. Clinical effects of cigarette smoking: Epidemiologic impact and review of pharmacotherapy options. Int. J. Environ. Res. Public Health 2017, 14, 1147. [Google Scholar] [CrossRef] [PubMed]
- Unified Development Ordinance. The Health Consequences of Smoking—50 Years of Progress: A Report of the Surgeon General; US Department of Health and Human Services: Atlanta, GA, USA, 2014.
- Novello, A.C. Surgeon General’s report on the health benefits of smoking cessation. Public Health Rep. 1990, 105, 545. [Google Scholar] [PubMed]
- Kenfield, S.A.; Stampfer, M.J.; Rosner, B.A.; Colditz, G.A. Smoking and smoking cessation in relation to mortality in women. JAMA 2008, 299, 2037–2047. [Google Scholar] [CrossRef]
- Goren, A.; Annunziata, K.; Schnoll, R.A.; Suaya, J.A. Smoking cessation and attempted cessation among adults in the United States. PLoS ONE 2014, 9, e93014. [Google Scholar] [CrossRef]
- Fiore, M.C.; Jaén, C.R.; Baker, T.B.; Bailey, W.C.; Benowitz, N.L.; Curry, S.J.; Dorfman, S.F.; Froelicher, E.S.; Goldstein, M.G.; Healton, C.G. Treating Tobacco Use and Dependence: 2008 Update; US Department of Health and Human Services: Rockville, MD, USA, 2008.
- Anthenelli, R.M.; Benowitz, N.L.; West, R.; St Aubin, L.; McRae, T.; Lawrence, D.; Ascher, J.; Russ, C.; Krishen, A.; Evins, A.E. Neuropsychiatric safety and efficacy of varenicline, bupropion, and nicotine patch in smokers with and without psychiatric disorders (EAGLES): A double-blind, randomised, placebo-controlled clinical trial. Lancet 2016, 387, 2507–2520. [Google Scholar] [CrossRef]
- Vogt, F.; Hall, S.; Marteau, T.M. General practitioners’ and family physicians’ negative beliefs and attitudes towards discussing smoking cessation with patients: A systematic review. Addiction 2005, 100, 1423–1431. [Google Scholar] [CrossRef]
- Huang, W.-H.; Hsu, H.-Y.; Chang, B.C.-C.; Chang, F.-C. Factors correlated with success rate of outpatient smoking cessation services in Taiwan. Int. J. Environ. Res. Public Health 2018, 15, 1218. [Google Scholar] [CrossRef]
- Caponnetto, P.; Polosa, R. Common predictors of smoking cessation in clinical practice. Respir. Med. 2008, 102, 1182–1192. [Google Scholar] [CrossRef] [PubMed]
- Vangeli, E.; Stapleton, J.; Smit, E.S.; Borland, R.; West, R. Predictors of attempts to stop smoking and their success in adult general population samples: A systematic review. Addiction 2011, 106, 2110–2121. [Google Scholar] [CrossRef] [PubMed]
- López-Torrecillas, F.; Perales, J.C.; Nieto-Ruiz, A.; Verdejo-García, A. Temperament and impulsivity predictors of smoking cessation outcomes. PLoS ONE 2014, 9, e112440. [Google Scholar] [CrossRef]
- Krittanawong, C.; Zhang, H.; Wang, Z.; Aydar, M.; Kitai, T. Artificial intelligence in precision cardiovascular medicine. J. Am. Coll. Cardiol. 2017, 69, 2657–2664. [Google Scholar] [CrossRef]
- Adam, N.R.; Wieder, R.; Ghosh, D. Data science, learning, and applications to biomedical and health sciences. Ann. N. Y. Acad. Sci. 2017, 1387, 5–11. [Google Scholar] [CrossRef]
- Lee, C.K.; Hofer, I.; Gabel, E.; Baldi, P.; Cannesson, M. Development and validation of a deep neural network model for prediction of postoperative in-hospital mortality. Anesthesiology 2018, 129, 649. [Google Scholar] [CrossRef]
- Rodriguez-Romero, V.; Bergstrom, R.F.; Decker, B.S.; Lahu, G.; Vakilynejad, M.; Bies, R.R. Prediction of Nephropathy in Type 2 Diabetes: An Analysis of the ACCORD Trial Applying Machine Learning Techniques. Clin. Transl. Sci. 2019, 12, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Mezzatesta, S.; Torino, C.; De Meo, P.; Fiumara, G.; Vilasi, A. A machine learning-based approach for predicting the outbreak of cardiovascular diseases in patients on dialysis. Comput. Methods Programs Biomed. 2019, 177, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Coughlin, L.N.; Tegge, A.N.; Sheffer, C.E.; Bickel, W.K. A machine-learning approach to predicting smoking cessation treatment outcomes. Nicotine Tob. Res. 2020, 22, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Heatherton, T.F.; Kozlowski, L.T.; Frecker, R.C.; FAGERSTROM, K.O. The Fagerström test for nicotine dependence: A revision of the Fagerstrom Tolerance Questionnaire. Br. J. Addict. 1991, 86, 1119–1127. [Google Scholar] [CrossRef]
- Kim, J.S.; Merrill, R.K.; Arvind, V.; Kaji, D.; Pasik, S.D.; Nwachukwu, C.C.; Vargas, L.; Osman, N.S.; Oermann, E.K.; Caridi, J.M.; et al. Examining the Ability of Artificial Neural Networks Machine Learning Models to Accurately Predict Complications Following Posterior Lumbar Spine Fusion. Spine 2018, 43, 853–860. [Google Scholar] [CrossRef] [PubMed]
- Hamori, S.; Kawai, M.; Kume, T.; Murakami, Y.; Watanabe, C. Ensemble learning or deep learning? Application to default risk analysis. J. Risk Financ. Manag. 2018, 11, 12. [Google Scholar] [CrossRef]
- Pedregosa, F.; Varoquaux, G.; Gramfort, A.; Michel, V.; Thirion, B.; Grisel, O. Scikit-learn: Machine Learning in Python. J. Mach. Learn. Res. 2012, 12, 2825–2830. [Google Scholar]
- DeLong, E.R.; DeLong, D.M.; Clarke-Pearson, D.L. Comparing the areas under two or more correlated receiver operating characteristic curves: A nonparametric approach. Biometrics 1988, 44, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Dreiseitl, S.; Ohno-Machado, L. Logistic regression and artificial neural network classification models: A methodology review. J. Biomed. Inform. 2002, 35, 352–359. [Google Scholar] [CrossRef]
- Witten, I.H.; Frank, E.; Hall, M.A. Chapter 5—Credibility: Evaluating What’s Been Learned. In Data Mining: Practical Machine Learning Tools and Techniques, 3rd ed.; Witten, I.H., Frank, E., Hall, M.A., Eds.; Morgan Kaufmann: Boston, MA, USA, 2011; pp. 147–187. [Google Scholar] [CrossRef]
- SRNT Subcommittee on Biochemical Verification. Biochemical verification of tobacco use and cessation. Nicotine Tob. Res. 2002, 4, 149–159. [Google Scholar] [CrossRef]
- Benowitz, N.L.; Bernert, J.T.; Foulds, J.; Hecht, S.S.; Jacob, P.; Jarvis, M.J.; Joseph, A.; Oncken, C.; Piper, M.E. Biochemical Verification of Tobacco Use and Abstinence: 2019 Update. Nicotine Tob. Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Hukkanen, J.; Jacob, P.; Benowitz, N.L. Metabolism and disposition kinetics of nicotine. Pharmacol. Rev. 2005, 57, 79–115. [Google Scholar] [CrossRef] [PubMed]
| Total (n = 4875) | Training Dataset (n = 4375) | Testing Dataset (n = 500) | p-Value | ||
|---|---|---|---|---|---|
| Gender (n, %) | Female | 1040 (21.3) | 938 (21.4) | 102 (20.4) | 0.5907 |
| Male | 3835 (78.7) | 3437 (78.6) | 398 (79.6) | ||
| Age (years) | 46.7 ± 12.7 | 46.7 ± 12.7 | 46.9 ± 13.0 | 0.8325 | |
| Body weight (kg) | 71.0 ± 14.9 | 71.1 ± 15.0 | 70.6 ± 14.3 | 0.5200 | |
| Duration of smoking (years) | 25.0 ± 12.2 | 25.1 ± 12.2 | 24.7 ± 12.2 | 0.5294 | |
| Number of cigarettes smoked per day at baseline (stick) | 20.1 ± 12.3 | 20.1 ± 12.4 | 20.2 ± 11.9 | 0.8987 | |
| Ambition (urge to quit) | Yes | 2692 (55.2) | 2430 (55.5) | 262 (52.4) | 0.1806 |
| No | 2183 (44.8) | 1945 (44.5) | 238 (47.6) | ||
| Physician clinics visit | Yes | 4393 (90.1) | 3937 (90.0) | 456 (91.2) | 0.3899 |
| No | 482 (9.9) | 438 (10.0) | 44 (8.8) | ||
| Educator clinics visit | Yes | 1426 (29.3) | 1289 (29.5) | 137 (27.4) | 0.3368 |
| No | 3449 (70.7) | 3086 (70.5) | 363 (72.6) | ||
| FTND score (point) | 6.4 ± 2.3 | 6.4 ± 2.3 | 6.2 ± 2.4 | 0.1400 | |
| Exhaled CO level tested at baseline | Yes | 3987 (81.8) | 3576 (81.7) | 411 (82.2) | 0.7995 |
| No | 888 (18.2) | 799 (18.3) | 89 (17.8) | ||
| Exhaled CO levels (ppm) | 15.9 ± 10.1 | 15.9 ± 10.1 | 16.0 ± 10.3 | 0.7581 | |
| Smoking cessation drugs prescribed at the 1st visit | Nil | 441 (9.1) | 399 (9.1) | 42 (8.4) | 0.1406 |
| NRT | 943 (19.3) | 846 (19.3) | 97 (19.4) | ||
| Bupropion | 9 (0.2) | 6 (0.1) | 3 (0.6) | ||
| Varenicline | 3482 (71.4) | 3124 (71.4) | 358 (71.6) | ||
| Use varenicline during treatment | No | 1338 (27.4) | 1204 (27.5) | 134 (26.8) | 0.7325 |
| Yes | 3537 (72.6) | 3171 (72.5) | 366 (73.2) | ||
| Point prevalence abstinence (n, %) | Success | 2615(53.6) | 2348 (53.7) | 267 (53.4) | 0.9092 |
| Fail | 2260(46.4) | 2027 (46.3) | 233 (46.6) |
| Sensitivity | Specificity | Accuracy | ROC Value (95% CI) | |
|---|---|---|---|---|
| ANN | 0.704 | 0.567 | 0.640 | 0.660 (0.617–0.702) |
| SVM | 0.768 | 0.433 | 0.612 | 0.658 (0.614–0.699) |
| RF | 0.757 | 0.485 | 0.626 | 0.654 (0.610–0.695) |
| LoR | 0.742 | 0.459 | 0.608 | 0.653 (0.609–0.694) |
| KNN | 0.764 | 0.408 | 0.598 | 0.618 (0.573–0.660) |
| CART | 0.674 | 0.528 | 0.606 | 0.612 (0.568–0.655) |
| NB | 0.614 | 0.524 | 0.568 | 0.608 (0.564–0.651) |
| ANN | SVM | RF | LoR | KNN | CART | NB | |
|---|---|---|---|---|---|---|---|
| ANN | 1.0000 | ||||||
| SVM | 0.7997 | 1.0000 | |||||
| RF | 0.6882 | 0.8158 | 1.0000 | ||||
| LoR | 0.4873 | 0.2595 | 0.9518 | 1.0000 | |||
| KNN | 0.0491 | 0.0601 | 0.1308 | 0.0945 | 1.0000 | ||
| CART | 0.0505 | 0.058 | 0.0615 | 0.0944 | 0.8391 | 1.0000 | |
| NB | 0.0068 | 0.0009 | 0.0335 | 0.0031 | 0.6769 | 0.8865 | 1.0000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lai, C.-C.; Huang, W.-H.; Chang, B.C.-C.; Hwang, L.-C. Development of Machine Learning Models for Prediction of Smoking Cessation Outcome. Int. J. Environ. Res. Public Health 2021, 18, 2584. https://doi.org/10.3390/ijerph18052584
Lai C-C, Huang W-H, Chang BC-C, Hwang L-C. Development of Machine Learning Models for Prediction of Smoking Cessation Outcome. International Journal of Environmental Research and Public Health. 2021; 18(5):2584. https://doi.org/10.3390/ijerph18052584
Chicago/Turabian StyleLai, Cheng-Chien, Wei-Hsin Huang, Betty Chia-Chen Chang, and Lee-Ching Hwang. 2021. "Development of Machine Learning Models for Prediction of Smoking Cessation Outcome" International Journal of Environmental Research and Public Health 18, no. 5: 2584. https://doi.org/10.3390/ijerph18052584
APA StyleLai, C.-C., Huang, W.-H., Chang, B. C.-C., & Hwang, L.-C. (2021). Development of Machine Learning Models for Prediction of Smoking Cessation Outcome. International Journal of Environmental Research and Public Health, 18(5), 2584. https://doi.org/10.3390/ijerph18052584






