Implementation of Person-Centered Care: A Feasibility Study Using the WE-CARE Roadmap
Abstract
1. Introduction
- To what extent can the three PCC routines previously tested in Sweden and England be implemented in a rehabilitation hospital for children in Poland?
- To what extent are the enablers described in the WE-CARE roadmap helpful in this PCC implementation process?
2. Materials and Methods
2.1. Design, Setting, and Participants
- The recommendations for scoliosis treatment are, to great extent, standardized, and thus the treatment process and adherence could be consistently monitored. The International Scientific Society on Scoliosis Orthopaedic and Rehabilitation Treatment (SOSORT) recommendations indicate that bracing (primarily the Cheneau corset) must be worn for as much of any 24 h period as possible [18].
- There is a lack of understanding among the general population about scoliosis and its treatment. Upon diagnosis, young people and their parents need support and psycho-education. In addition, it is often very difficult for young patients to follow the treatment recommendations, especially in the group at high risk of progression that requires Full Time Rigid Bracing, i.e., wearing a corset for a minimum 20 h daily and performing special exercises [18]. However, even for patients with a lower rate of scoliosis progression and less than full-time bracing recommendations, treatment adherence is challenging [19]. Healthcare professionals at the rehabilitation hospital have validated this finding in their own patients. Lastly, scoliosis is often diagnosed at a young age, which means that treatment success depends on the child or adolescent adhering to the treatment plan [20].
- The number of new patients diagnosed with scoliosis at the hospital was considered to be large enough, but also feasible enough, to effectively start a PCC intervention.
2.2. Interventions
2.2.1. PCC Intervention Approach
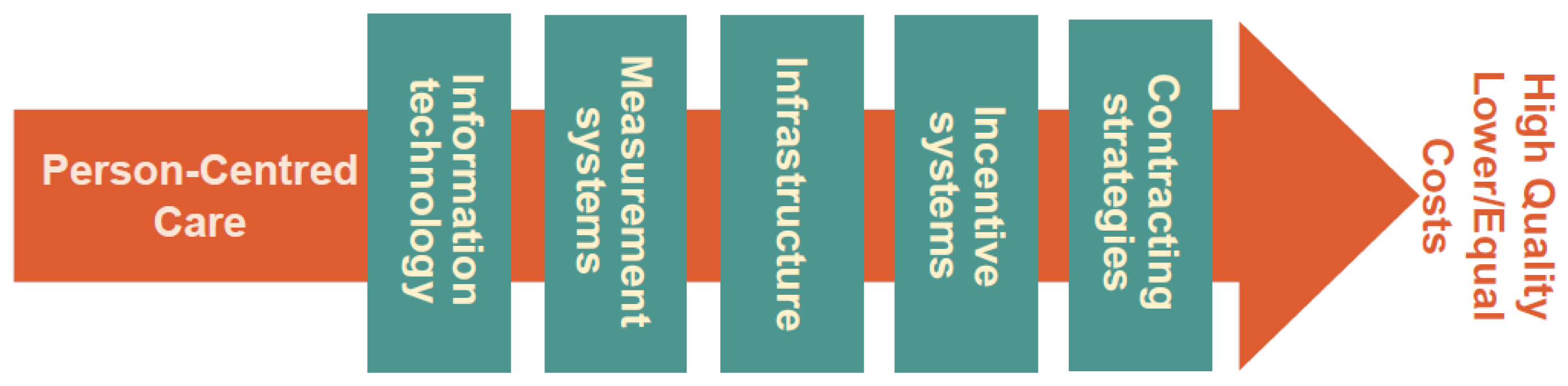
2.2.2. We-CARE Roadmap
- Information Technology (IT) might include IT solutions for communication or wearable devices for monitoring.
- Infrastructure relates to the necessary resources and structures empowering the delivery of PCC (e.g., hospital organization, home care and their interaction).
- Incentive systems include all conceivable means which could motivate both patients and professionals to engage in behaviors congruent with PCC.
- Contracting strategies includes financial solutions and mutual agreements regulating commitments and deliverables.
- Measurement systems to capture all necessary aspects of the implementation process and its outcome.
Infrastructure
Information Technologies
Incentive Systems
Measurement Systems
2.3. Methods
3. Results and Discussion
3.1. Quantitative Data on Feasibility of PCC Implementation
3.2. Qualitative Data on Implementation of PCC Routines
3.2.1. From ‘Talking to’, to ‘Talking with’. Creating a Partnership (PCC Routine 1)
“We have always had the feeling that the patient is standing somehow nearby. That means, the patient stands in the center and we all bend over him as if we impose our will on him. And now (…) after such a change of approach, it seems that the patient joined our interdisciplinary team and the relationship is closer.”(HCP1)
“We let (the patient) speak, we listen to children, what they think about it (their disease and treatment plan), more than before, it requires more time, … and parents who have some expectations, read something on the internet, so we together verify this.”.(HCP4)
“When it turned out that something was wrong with our daughter’s spine, we read on the Internet and talked to various specialists. After examination, the X-ray, everyone tried to convince us of the “right” therapy. We were confused. But no one really gave us enough time, or explained the mechanism of scoliosis, or the advantages and disadvantages of each therapy. Only in Ameryka (city in which Voivodeship Rehabilitation Hospital for Children is located). Previously, (in other facilities) nobody asked about our feelings or wanted to agree on an approach with us. Nowhere else we were able to discuss our concerns with the team of specialists. Knowing more about scoliosis, it was easier for us to talk to specialists and make decisions (about the treatment plan).”(P1)
3.2.2. Jointly Created Care Plans. Working the Partnership (PCC Routine 2)
“We discuss with patients, we want to know what they think about it (proposed treatment) and how they assess it. If even there are tears at the beginning, that there is a corset on, then the discussion goes on, so that the patient begins to be interested in what’s next, what’s the outcome. That there are not only dark thoughts... that there is light in the tunnel, and that it will be better. (We discuss together with the child and the parents) what path to take for this child, so that (the treatment) meets their expectations and resolves the health problem.”(HCP8)
“I like this (PCC) approach, that medics are talking a lot with us, are really listening to and considering our suggestions. We really feel important … and that we have an influence.”(P2)
“I like this control. I forgot to do something according to the treatment plan … actually, I procrastinated, but they called me, asked me how we are doing. Well, I was ashamed … but they motivated me. I did it quickly. Actually, I am happy about that.”(P3)
3.2.3. Safeguarding the Partnership (PCC Routine 3)
“Now (after implementation of PCC), one needs to pay more attention to talking with the patient about whether he/she accepts this method. Earlier ... yes, I might have asked, but in general, I approached the patient in such a way that I said ‘listen, we will practice with this method’ and that’s it. The patient did what I expected of him. However, now there is a conversation with the patient. If the patient does not accept it (the physiotherapeutic method), then I just change the method or ask a colleague (who is expert in other methods) for help, to show a patient how to work by another method (that is more acceptable to him/her).”(HCP9)
3.3. Qualitative Data on Feasibility of Use of the Enablers in the WE-CARE Roadmap
“What motivates me (to implement PCC) are the outcomes of treatment. … that a patient adheres more to the treatment plan. That they participate, this (co-creation of treatment plan) is not next to them. Well, (earlier) I had a feeling, that I am saying something (about the treatment), the parent goes out of their way (be driven round the bend), but the patient (behaves) as if he was outside (that the situation does not relate to them).”(HCP5)
“I see how my daughter is now engaged in the following of the therapy. She very scrupulously wears the corset, and logs her hours (in the ‘Activity Diary’). She is doing this for herself, but she can also show this to me and the doctor.”(P2)
“We prescribe a corset, and it is made. The patient goes somewhere, someone says that this method is not so necessary. The patient is not motivated enough, so they listen to what suits them and reject the corset. (Public) money is spent on this corset, for this treatment, there are, so, … financial losses for this orthopedic equipment. Actually double losses, because he went somewhere else to another clinic. And as a consequence, … well, they come to us, let’s say after a year, ... We have such a patient now. Her health has deteriorated, she has had a significant progression of scoliosis. The corset was made and thrown out. A new one needs to be made, and possibly even a scoliosis surgery would need to be performed because the scoliosis is already so advanced that it requires much more money than the corset itself.”(R3)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ekman, I.; Busse, R.; van Ginneken, E.; Van Hoof, C.; van Ittersum, L.; Klink, A.; Kremer, J.A.; Miraldo, M.; Olauson, A.; De Raedt, W.; et al. Health-care improvements in a financially constrained environment. Lancet 2016, 387, 646–647. [Google Scholar] [CrossRef]
- Hansson, E.; Ekman, I.; Swedberg, K.; Wolf, A.; Dudas, K.; Ehlers, L.; Olsson, L.E. Person-centred care for patients with chronic heart failure—A cost-utility analysis. Eur. J. Cardiovasc. Nurs. 2016, 15, 276–284. [Google Scholar] [CrossRef]
- Pirhonen, L.; Bolin, K.; Olofsson, E.H.; Fors, A.; Ekman, I.; Swedberg, K.; Gyllensten, H. Person-Centred Care in Patients with Acute Coronary Syndrome: Cost-Effectiveness Analysis Alongside a Randomised Controlled Trial. Pharm. Open 2019, 3, 495–504. [Google Scholar] [CrossRef]
- Lloyd, H.M.; Pearson, M.; Sheaff, R.; Asthana, S.; Wheat, H.; Sugavanam, T.P.; Britten, N.; Valderas, J.; Bainbridge, M.; Witts, L.; et al. Collaborative action for person-centred coordinated care (P3C): An approach to support the development of a comprehensive system-wide solution to fragmented care. Health Res. Policy Syst. 2017, 15, 98. [Google Scholar] [CrossRef] [PubMed]
- Close, J.; Fosh, B.; Wheat, H.; Horrell, J.; Lee, W.; Byng, R.; Bainbridge, M.; Blackwell, R.; Witts, L.; Hall, L.; et al. Longitudinal evaluation of a countywide alternative to the Quality and Outcomes Framework in UK General Practice aimed at improving Person Centred Coordinated Care. BMJ Open 2019, 9, e029721. [Google Scholar] [CrossRef] [PubMed]
- Britten, N.; Moore, L.; Lydahl, D.; Naldemirci, O.; Elam, M.; Wolf, A. Elaboration of the Gothenburg model of person-centred care. Health Expect 2017, 20, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, H.M.; Ekman, I.; Rogers, H.L.; Raposo, V.; Melo, P.; Marinkovic, V.D.; Buttigieg, S.C.; Srulovici, E.; Lewandowski, R.A.; Britten, N. Supporting Innovative Person-Centred Care in Financially Constrained Environments: The WE CARE Exploratory Health Laboratory Evaluation Strategy. Int. J. Environ. Res. Public Health 2020, 17, 3050. [Google Scholar] [CrossRef] [PubMed]
- Sackett, D.L.; Rosenberg, W.M.; Gray, J.A.; Haynes, R.B.; Richardson, W.S. Evidence based medicine: What it is and what it isn’t. BMJ 1996, 312, 71–72. [Google Scholar] [CrossRef]
- Hoffmann, T.C.; Montori, V.M.; Del Mar, C. The connection between evidence-based medicine and shared decision making. JAMA 2014, 312, 1295–1296. [Google Scholar] [CrossRef] [PubMed]
- Ekman, I.; Swedberg, K.; Taft, C.; Lindseth, A.; Norberg, A.; Brink, E.; Carlsson, J.; Dahlin-Ivanoff, S.; Johansson, I.L.; Kjellgren, K.; et al. Person-centered care—Ready for prime time. Eur. J. Cardiovasc. Nurs. 2011, 10, 248–251. [Google Scholar] [CrossRef] [PubMed]
- Ekman, I.; Wolf, A.; Olsson, L.E.; Taft, C.; Dudas, K.; Schaufelberger, M.; Swedberg, K. Effects of person-centred care in patients with chronic heart failure: The PCC-HF study. Eur. Heart J. 2012, 33, 1112–1119. [Google Scholar] [CrossRef] [PubMed]
- Fors, A.; Ekman, I.; Taft, C.; Bjorkelund, C.; Frid, K.; Larsson, M.E.; Thorn, J.; Ulin, K.; Wolf, A.; Swedberg, K. Person-centred care after acute coronary syndrome, from hospital to primary care—A randomised controlled trial. Int. J. Cardiol. 2015, 187, 693–699. [Google Scholar] [CrossRef] [PubMed]
- Fors, A.; Swedberg, K.; Ulin, K.; Wolf, A.; Ekman, I. Effects of person-centred care after an event of acute coronary syndrome: Two-year follow-up of a randomised controlled trial. Int. J. Cardiol. 2017, 249, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Porter, M.E. What is value in health care? N. Engl. J. Med. 2010, 363, 2477–2481. [Google Scholar] [CrossRef] [PubMed]
- Olsson, L.E.; Hansson, E.; Ekman, I. Evaluation of person-centred care after hip replacement-a controlled before and after study on the effects of fear of movement and self-efficacy compared to standard care. BMC Nurs. 2016, 15, 53. [Google Scholar] [CrossRef] [PubMed]
- Personcentrerad Vård i Sverige [Person-centred care in Sweden]. Swedish Association of Local Authorities and Regions. Available online: https://webbutik.skr.se/bilder/artiklar/pdf/7585-631-5.pdf?issuusl=ignore (accessed on 12 February 2021).
- CEN/TC450. Patient Involvement in Person-Centred Care. Available online: https://standards.cen.eu/dyn/www/f?p=204:110:0::::FSP_PROJECT,FSP_LANG_ID:65031,25&cs=170DF874D1EFAE86E8A8205451C28A49D (accessed on 12 February 2021).
- Negrini, S.; Donzelli, S.; Aulisa, A.G.; Czaprowski, D.; Schreiber, S.; de Mauroy, J.C.; Diers, H.; Grivas, T.B.; Knott, P.; Kotwicki, T.; et al. 2016 SOSORT guidelines: Orthopaedic and rehabilitation treatment of idiopathic scoliosis during growth. Scoliosis Spinal Disord. 2018, 13, 3. [Google Scholar] [CrossRef]
- Karol, L.A.; Virostek, D.; Felton, K.; Wheeler, L. Effect of Compliance Counseling on Brace Use and Success in Patients with Adolescent Idiopathic Scoliosis. J. Bone Jt. Surg. Am. 2016, 98, 9–14. [Google Scholar] [CrossRef]
- Weinstein, S.L.; Dolan, L.A.; Wright, J.G.; Dobbs, M.B. Effects of bracing in adolescents with idiopathic scoliosis. N. Engl. J. Med. 2013, 369, 1512–1521. [Google Scholar] [CrossRef] [PubMed]
- Dolan, L.A.; Donzelli, S.; Zaina, F.; Weinstein, S.L.; Negrini, S. Adolescent Idiopathic Scoliosis Bracing Success Is Influenced by Time in Brace: Comparative Effectiveness Analysis of BrAIST and ISICO Cohorts. Spine 2020, 45, 1193–1199. [Google Scholar] [CrossRef]
- Britten, N.; Ekman, I.; Naldemirci, O.; Javinger, M.; Hedman, H.; Wolf, A. Learning from Gothenburg model of person centred healthcare. BMJ 2020, 370, m2738. [Google Scholar] [CrossRef]
- Lewandowski, R. The WE-CARE Roadmap: A Framework for Implementation of Person-Centred Care and Health Promotion in Medical Organi-zations. J. Appl. Manag. Investig. 2020, 9, 120–132. [Google Scholar]
- Mutual Meetings. A Training Programme in Person-Centred Care for Everyone within Health and Social Care. Centre for Person-Centred Care at the University of Gothenburg. Available online: https://medmanniska.org/?lang=en (accessed on 12 February 2021).
- Malina, M.A.S.; Selto, F.H. Communicating and Controlling Strategy: An Empirical Study of the Effectiveness of the Balanced Scorecard. J. Manag. Account. Res. 2001, 13, 47–90. [Google Scholar] [CrossRef]
- Goncharuk, A.G.; Lewandowski, R.; Cirella, G.T. Motivators for medical staff with a high gap in healthcare efficiency: Comparative research from Poland and Ukraine. Int. J. Health Plan. Manag. 2020, 35, 1314–1334. [Google Scholar] [CrossRef]
- Hsieh, H.F.; Shannon, S.E. Three approaches to qualitative content analysis. Qual. Health Res. 2005, 15, 1277–1288. [Google Scholar] [CrossRef] [PubMed]
- Flamholtz, E. Effective Organizational Control: A Framework, Applications, and Implications. Eur. Manag. J. 1996, 14, 596–611. [Google Scholar] [CrossRef]
- Pirhonen, L.; Olofsson, E.H.; Fors, A.; Ekman, I.; Bolin, K. Effects of person-centred care on health outcomes—A randomized controlled trial in patients with acute coronary syndrome. Health Policy 2017, 121, 169–179. [Google Scholar] [CrossRef]
| Number of PCC Patients | Percent of Patients with a Narrative Summary in the EMR by Type of Health Care Professional | Percent with a Documented Treatment Plan | Percent of Patients Using the Activity Diary | |||
|---|---|---|---|---|---|---|
| 51 | Physician | Physiotherapist | Nurse | Psychologist | 86% (n = 44) | 100% (n = 51) |
| 92% (n = 47) | 100% (n = 51) | 98% (n = 50) | 98% (n = 50) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lewandowski, R.A.; Lewandowski, J.B.; Ekman, I.; Swedberg, K.; Törnell, J.; Rogers, H.L. Implementation of Person-Centered Care: A Feasibility Study Using the WE-CARE Roadmap. Int. J. Environ. Res. Public Health 2021, 18, 2205. https://doi.org/10.3390/ijerph18052205
Lewandowski RA, Lewandowski JB, Ekman I, Swedberg K, Törnell J, Rogers HL. Implementation of Person-Centered Care: A Feasibility Study Using the WE-CARE Roadmap. International Journal of Environmental Research and Public Health. 2021; 18(5):2205. https://doi.org/10.3390/ijerph18052205
Chicago/Turabian StyleLewandowski, Roman A., Jędrzej B. Lewandowski, Inger Ekman, Karl Swedberg, Jan Törnell, and Heather L. Rogers. 2021. "Implementation of Person-Centered Care: A Feasibility Study Using the WE-CARE Roadmap" International Journal of Environmental Research and Public Health 18, no. 5: 2205. https://doi.org/10.3390/ijerph18052205
APA StyleLewandowski, R. A., Lewandowski, J. B., Ekman, I., Swedberg, K., Törnell, J., & Rogers, H. L. (2021). Implementation of Person-Centered Care: A Feasibility Study Using the WE-CARE Roadmap. International Journal of Environmental Research and Public Health, 18(5), 2205. https://doi.org/10.3390/ijerph18052205






