Spontaneous Pneumothorax in COVID-19 Patients Treated with High-Flow Nasal Cannula outside the ICU: A Case Series
Abstract
1. Introduction
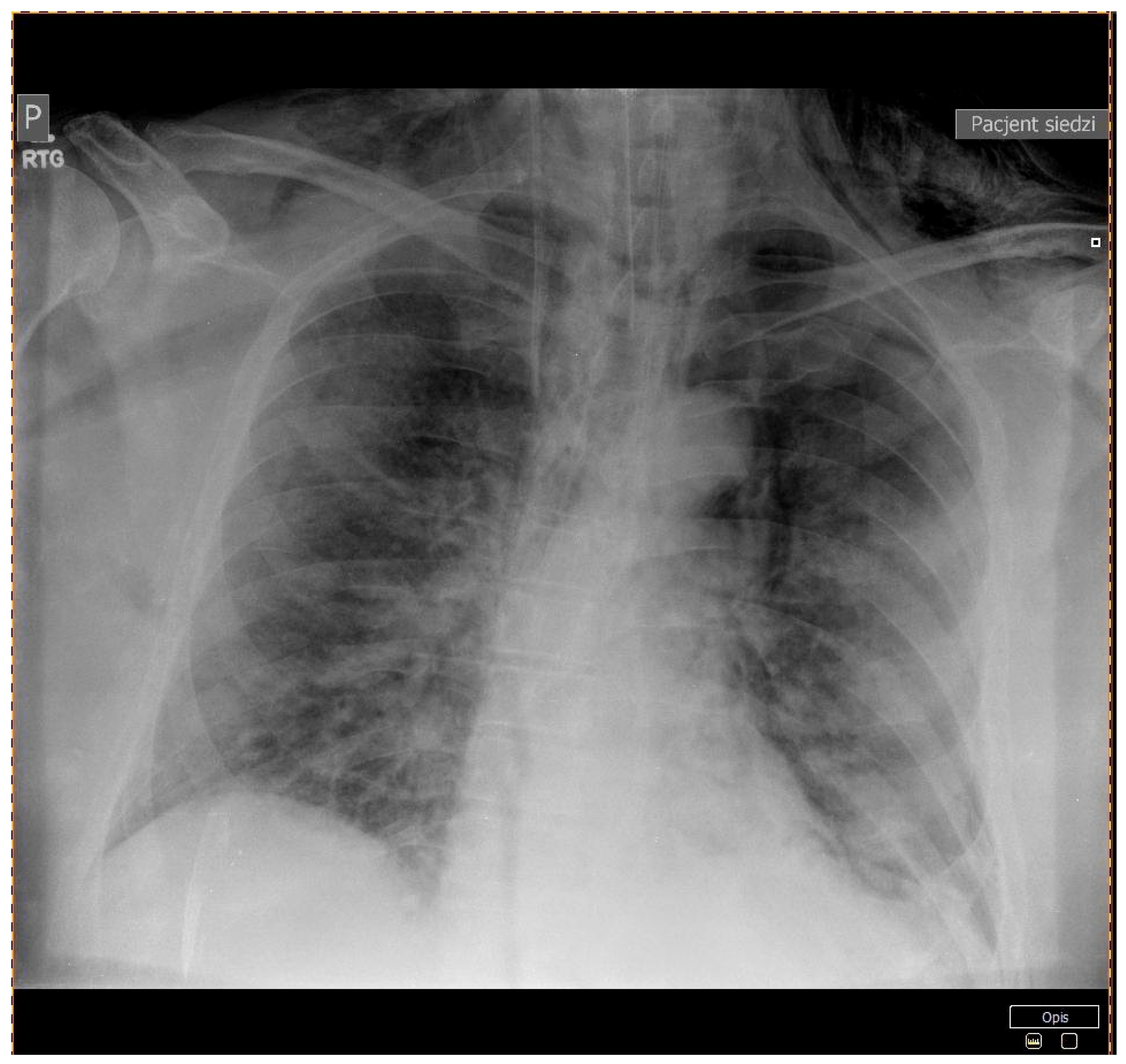
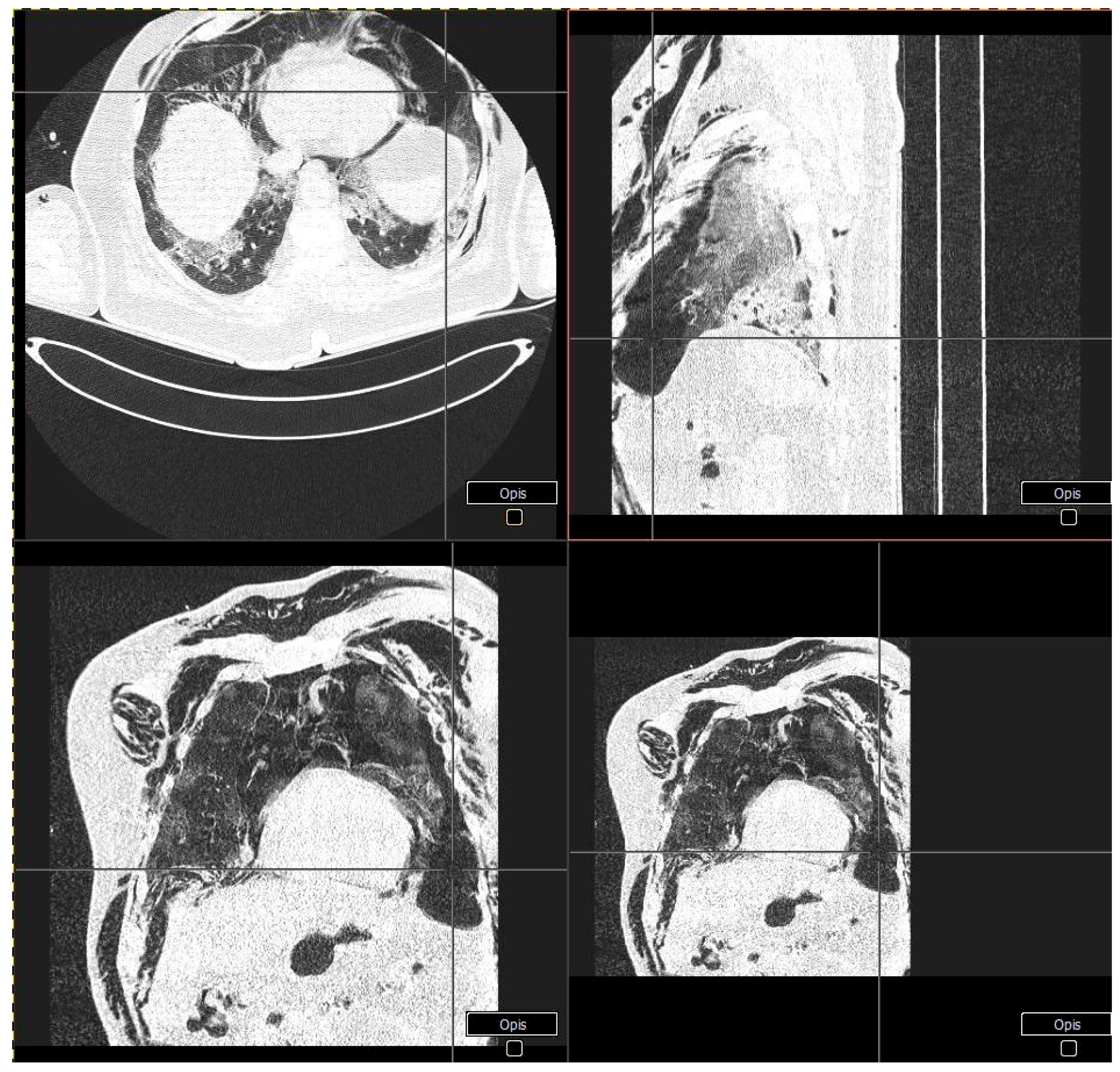
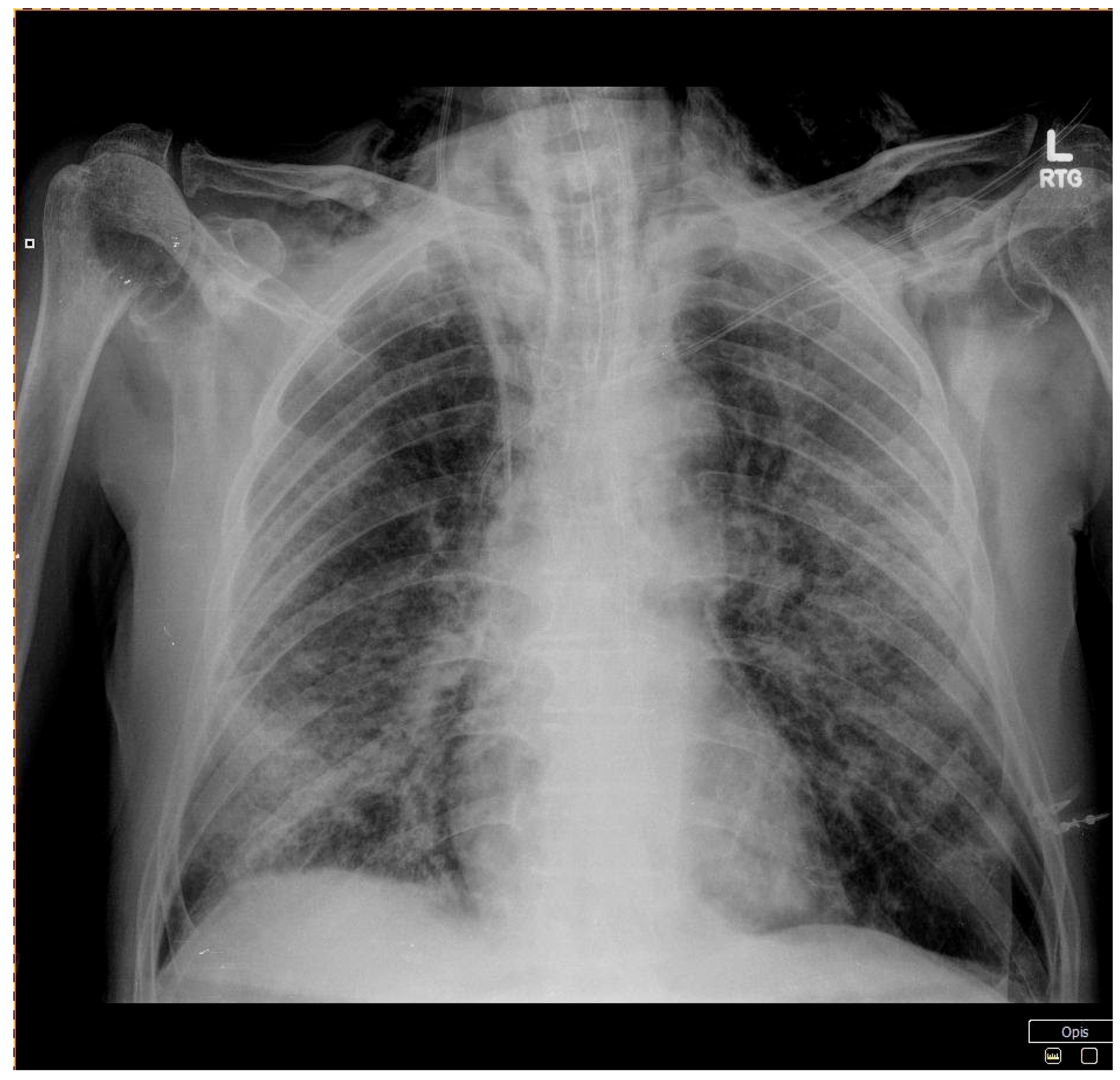
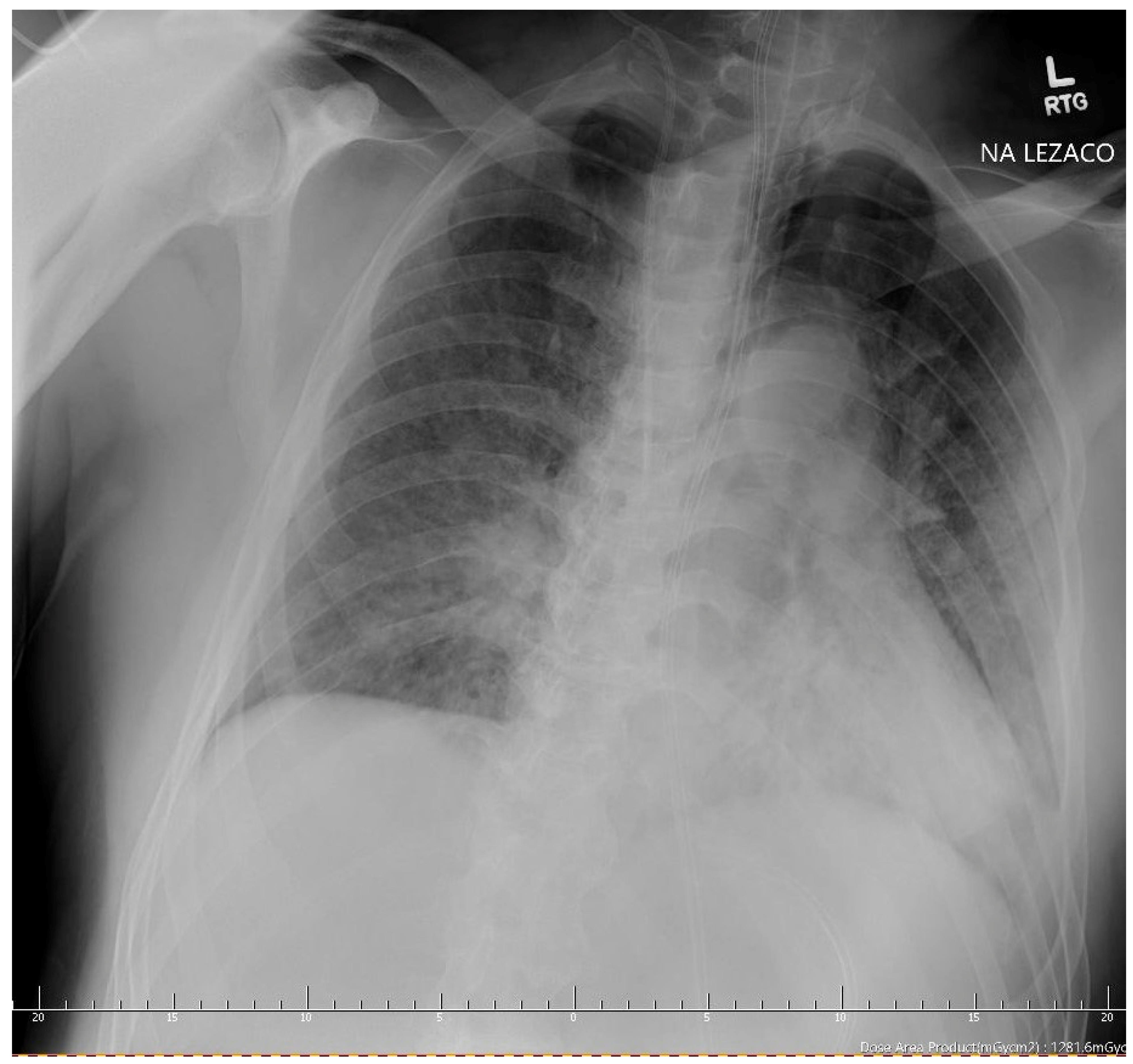
2. Case Reports
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- World Health Organization. COVID-19 Weekly Epidemiological Update 22. World Health Organization, 7 February 2021; pp. 1–3. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/weekly_epidemiological_update_22.pdf (accessed on 9 February 2021).
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Who, W. Clinical management of severe acute respiratory infection (SARI) when COVID-19 disease is suspected. Interim Guid. Pediatr. Med. Rodz. 2020, 16, 9–26. [Google Scholar]
- Spagnolo, P.; Balestro, E.; Aliberti, S.; Cocconcelli, E.; Biondini, D.; Casa, G.D.; Sverzellati, N.; Maher, T.M. Pulmonary fibrosis secondary to COVID-19: A call to arms? Lancet Respir. Med. 2020, 8, 750–752. [Google Scholar] [CrossRef]
- Lechowicz, K.; Drożdżal, S.; Machaj, F.; Rosik, J.; Szostak, B.; Zegan-Barańska, M.; Biernawska, J.; Dabrowski, W.; Rotter, I.; Kotfis, K. COVID-19: The Potential Treatment of Pulmonary Fibrosis Associated with SARS-CoV-2 Infection. J. Clin. Med. 2020, 9, 1917. [Google Scholar] [CrossRef] [PubMed]
- Mauri, T.; Turrini, C.; Eronia, N.; Grasselli, G.; Volta, C.A.; Bellani, G.; Pesenti, A. Physiologic effects of high-flow nasal cannula in acute hypoxemic respiratory failure. Am. J. Respir. Crit. Care Med. 2017, 195, 1207–1215. [Google Scholar] [CrossRef] [PubMed]
- Geng, S.; Mei, Q.; Zhu, C.; Yang, T.; Yang, Y.; Fang, X.; Pan, A. High flow nasal cannula is a good treatment option for COVID-19. Heart Lung 2020, 49, 444–445. [Google Scholar] [CrossRef] [PubMed]
- Gürün Kaya, A.; Öz, M.; Erol, S.; Çiftçi, F.; Çiledağ, A.; Kaya, A. High flow nasal cannula in COVID-19: A literature review. Tuberk. Toraks 2020, 68, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Zarogoulidis, P.; Kioumis, I.; Pitsiou, G.; Porpodis, K.; Lampaki, S.; Papaiwannou, A.; Katsikogiannis, N.; Zaric, B.; Branislav, P.; Secen, N.; et al. Pneumothorax: From definition to diagnosis and treatment. J. Thorac. Dis. 2014, 6 (Suppl. 4), S372–S376. [Google Scholar] [CrossRef] [PubMed]
- Zantah, M.; Dominguez Castillo, E.; Townsend, R.; Dikengil, F.; Criner, G.J. Pneumothorax in COVID-19 disease- incidence and clinical characteristics. Respir. Res. 2020, 21. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet (Lond. Engl.) 2020, 395, 507–513. [Google Scholar] [CrossRef]
- Martinelli, A.W.; Ingle, T.; Newman, J.; Nadeem, I.; Jackson, K.; Lane, N.D.; Melhorn, J.; Davies, H.E.; Rostron, A.J.; Adeni, A.; et al. COVID-19 and pneumothorax: A multicentre retrospective case series. Eur. Respir. J. 2020, 56. [Google Scholar] [CrossRef] [PubMed]
- Tucker, L.; Patel, S.; Vatsis, C.; Poma, A.; Ammar, A.; Nasser, W.; Mukkera, S.; Vo, M.; Khan, R.; Carlan, S. Pneumothorax and Pneumomediastinum Secondary to COVID-19 Disease Unrelated to Mechanical Ventilation. Case Rep. Crit. Care 2020, 2020, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Shi, L.; Wang, Y.; Zhang, J.; Huang, L.; Zhang, C.; Liu, S.; Zhao, P.; Liu, H.; Zhu, L.; et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020, 8, 420–422. [Google Scholar] [CrossRef]
- McGuinness, G.; Zhan, C.; Rosenberg, N.; Azour, L.; Wickstrom, M.; Mason, D.M.; Thomas, K.M.; Moore, W.H. Increased incidence of barotrauma in patients with COVID-19 on invasive mechanical ventilation. Radiology 2020, 297, E252–E262. [Google Scholar] [CrossRef] [PubMed]
- Woodside, K.J.; van Sonnenberg, E.; Chon, K.S.; Loran, D.B.; Tocino, I.M.; Zwischenberger, J.B. Pneumothorax in patients with acute respiratory distress syndrome: Pathophysiology, detection, and treatment. J. Intensive Care Med. 2003, 18, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Akdogan, R.E.; Mohammed, T.; Syeda, A.; Jiwa, N.; Ibrahim, O.; Mutneja, R. Pneumothorax in Mechanically Ventilated Patients with COVID-19 Infection. Case Rep. Crit. Care 2021, 2021, 1–8. [Google Scholar] [CrossRef] [PubMed]
- McCool, F.D. Global physiology and pathophysiology of cough: ACCP evidence-based clinical practice guidelines. Chest 2006, 129, 48S–53S. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Danckers, M.; Sanghavi, D.; Chakraborty, R.K. StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar] [PubMed]
- Jung, C.; Wernly, B.; Muessig, J.M.; Kelm, M.; Boumendil, A.; Morandi, A.; Andersen, F.H.; Artigas, A.; Bertolini, G.; Cecconi, M.; et al. VIP1 study group. A comparison of very old patients admitted to intensive care unit after acute versus elective surgery or intervention. J. Crit. Care. 2019, 52, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Fried, M.W.; Crawford, J.M.; Mospan, A.R.; Watkins, S.E.; Munoz Hernandez, B.; Zink, R.C.; Elliott, S.; Burleson, K.; Landis, C.; Reddy, K.R.; et al. Patient Characteristics and Outcomes of 11,721 Patients with COVID19 Hospitalized Across the United States. Clin Infect Dis. 2020, 28, 1268. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
| Patient Characteristics (Sex, Age) | Patient 1 (Male, 64) | Patient 2 (Male, 83) | Patient 3 (Male, 56) | ||||
|---|---|---|---|---|---|---|---|
| At Admission to the Hospital | 1 Day After Conversion of Respiratory Support | At Admission to The Hospital | 1 Day After Conversion of Respiratory Support | At Admission to the Hospital | 1 Day After Conversion of Respiratory Support | ||
| Co-morbidities | Arterial hypertension, diabetes mellitus type 2 | Rheumatoid arthritis, arterial hypertension, benign prostate hyperplasia, ischemic heart disease | Diabetes mellitus type 2, arterial hypertension | ||||
| BMI [kg/m2] | 37 | 21.6 | 29.22 | ||||
| Obesity class | Class 2 Obesity | Normal | Overweight | ||||
| Symptoms | Dyspnea, fever, weakness | Fever, headache, dyspnea, taste and smell disturbances | Fatigue, dyspnea, fever, loss of appetite | ||||
| CT scan | Patchy and partially coalescing geographic ground-glass opacities | Diffuse bilateral ground-glass opacities and fibrotic changes In the form of bronchiectasis in lower lobes | Bilateral ground-glass opacities covering less than 50% of the lungs | ||||
| Respiratory support | Type | Non-rebreathing facemask | High-flow nasal cannula | Non-rebreathing facemask | High-flow nasal cannula | Simple nasal cannula | High-flow nasal cannula |
| Flow [L/min] | 14 | 50 | 15 | 60 | 3 | 50 | |
| FiO2 (%) | >90 | 75 | >90 | 80 | >90 | 82 | |
| Temperature | ~24 °C (room temp.) | 31 °C | ~24 °C (room temp.) | 31 °C | ~24 °C (room temp.) | 31 °C | |
| Saturation (%) | 94 | 90 | 95 | 98 | 94 | 71 | |
| pH | 7.40 | 7.49 | 7.49 | 7.53 | 7.57 | 7.51 | |
| pO2 (mmHg) | 73 | 54 | 78 | 96 | 66 | 33 | |
| pCO2 (mmHg) | 41 | 33 | 30 | 33 | 28 | 28 | |
| Lactate (mmol/L) | 1.1 | 1.4 | 1.3 | 1.6 | 1.6 | 1.9 | |
| WBC (G/L) | 9.9 | 16.3 | 5.3 | 16.3 | 8.33 | 13.5 | |
| Hgb (mmol/L) | 8.9 | 8.3 | 8.1 | 7.8 | 9.9 | 8.5 | |
| IL-6 (pg/mL) | 10.2 | 202.0 | 40.8 | 439.0 | 110 | 128 | |
| D-dimer (ng/mL) | 1026 | 1566 | 4820 | >7650 | 700 | 2206 | |
| LDH (U/L) | 523.0 | 523.0 | 592.0 | - | - | 771 | |
| CRP (mg/L) | 81.7 | 94.6 | 64.5 | 40.9 | 29 | 161 | |
| Serum creatinine (mg/dL) | 1.0 | 0.8 | 0.8 | 0.7 | 1.18 | 0.78 | |
| Serum sodium (mmol/L) | 139 | 139 | 136 | 139 | 129 | 128 | |
| Serum potassium (mmol/L) | 4.8 | 5.3 | 3.9 | 4.3 | 4.24 | 5.0 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nalewajska, M.; Feret, W.; Wojczyński, Ł.; Witkiewicz, W.; Wiśniewska, M.; Kotfis, K. Spontaneous Pneumothorax in COVID-19 Patients Treated with High-Flow Nasal Cannula outside the ICU: A Case Series. Int. J. Environ. Res. Public Health 2021, 18, 2191. https://doi.org/10.3390/ijerph18042191
Nalewajska M, Feret W, Wojczyński Ł, Witkiewicz W, Wiśniewska M, Kotfis K. Spontaneous Pneumothorax in COVID-19 Patients Treated with High-Flow Nasal Cannula outside the ICU: A Case Series. International Journal of Environmental Research and Public Health. 2021; 18(4):2191. https://doi.org/10.3390/ijerph18042191
Chicago/Turabian StyleNalewajska, Magdalena, Wiktoria Feret, Łukasz Wojczyński, Wojciech Witkiewicz, Magda Wiśniewska, and Katarzyna Kotfis. 2021. "Spontaneous Pneumothorax in COVID-19 Patients Treated with High-Flow Nasal Cannula outside the ICU: A Case Series" International Journal of Environmental Research and Public Health 18, no. 4: 2191. https://doi.org/10.3390/ijerph18042191
APA StyleNalewajska, M., Feret, W., Wojczyński, Ł., Witkiewicz, W., Wiśniewska, M., & Kotfis, K. (2021). Spontaneous Pneumothorax in COVID-19 Patients Treated with High-Flow Nasal Cannula outside the ICU: A Case Series. International Journal of Environmental Research and Public Health, 18(4), 2191. https://doi.org/10.3390/ijerph18042191







