Plasma Xanthine Oxidoreductase Activity Is Associated with a High Risk of Cardiovascular Disease in a General Japanese Population
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Cohort Data Collection
2.3. Measurements of XOR Activity
2.4. Framingham Risk Score
2.5. Statistical Analysis
3. Results
3.1. Baseline Characteristics of the Study Population
3.2. Concentration and Distribution of XOR Activity
3.3. Association between XOR Activity and Baseline Characteristics
3.4. Comparisons of FRS among Participants with Hypertension, Diabetes, or Dyslipidemia and Those without Them
3.5. Comparisons between XOR Activity and Baseline Characteristics
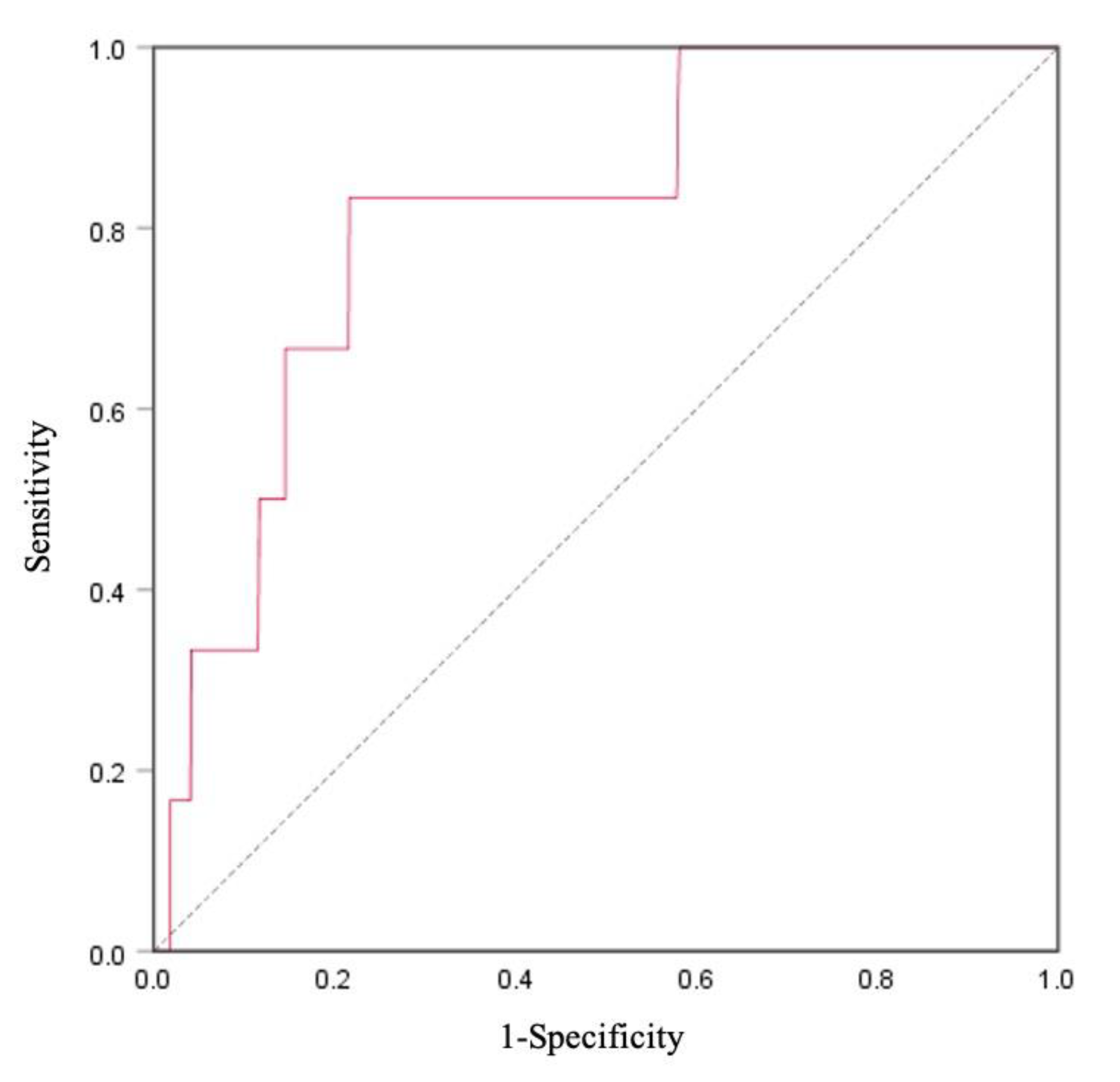
3.6. XOR Activity for the Identification of High Risk for CVD
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nishino, T. The conversion of xanthine dehydrogenase to xanthine oxidase and the role of the enzyme in reperfusion injury. J. Biochem. 1994, 116, 1–6. [Google Scholar] [CrossRef]
- Battelli, M.G.; Bolognesi, A.; Polito, L. Pathophysiology of circulating xanthine oxidoreductase: New emerging roles for a multi-tasking enzyme. Biochim. Biophys. Acta 2014, 1842, 1502–1517. [Google Scholar] [CrossRef] [PubMed]
- Battelli, M.G.; Polito, L.; Bolognesi, A. Xanthine oxidoreductase in atherosclerosis pathogenesis: Not only oxidative stress. Atherosclerosis 2014, 237, 562–567. [Google Scholar] [CrossRef]
- Amaya, Y.; Yamazaki, K.; Sato, M.; Noda, K.; Nishino, T.; Nishino, T. Proteolytic conversion of xanthine dehydrogenase from the NAD-dependent type to the O2-dependent type. Amino acid sequence of rat liver xanthine dehydrogenase and identification of the cleavage sites of the enzyme protein during irreversible conversion by trypsin. J. Biol. Chem. 1990, 265, 14170–14175. [Google Scholar] [PubMed]
- Meneshian, A.; Bulkley, G.B. The physiology of endothelial xanthine oxidase: From urate catabolism to reperfusion injury to inflammatory signal transduction. Microcirculation 2002, 9, 161–175. [Google Scholar] [CrossRef] [PubMed]
- Berry, C.E.; Hare, J.M. Xanthine oxidoreductase and cardiovascular disease: Molecular mechanisms and pathophysiological implications. J. Physiol. 2004, 555, 589–606. [Google Scholar] [CrossRef] [PubMed]
- Paravicini, T.M.; Touyz, R.M. NADPH oxidases, reactive oxygen species, and hypertension: Clinical implications and therapeutic possibilities. Diabetes Care 2008, 31, S170–S180. [Google Scholar] [CrossRef]
- Drummond, G.R.; Selemidis, S.; Griendling, K.K.; Sobey, C.G. Combating oxidative stress in vascular disease: NADPH oxidases as therapeutic targets. Nat. Rev. Drug Discov. 2011, 10, 453–471. [Google Scholar] [CrossRef] [PubMed]
- Fujimura, Y.; Yamauchi, Y.; Murase, T.; Nakamura, T.; Fujita, S.I.; Fujisaka, T.; Ito, T.; Sohmiya, K.; Hoshiga, M.; Ishizaka, N. Relationship between plasma xanthine oxidoreductase activity and left ventricular ejection fraction and hypertrophy among cardiac patients. PLoS ONE 2017, 12, e0182699. [Google Scholar]
- Nakatani, A.; Nakatani, S.; Ishimura, E.; Murase, T.; Nakamura, T.; Sakura, M.; Tateishi, Y.; Tsuda, A.; Kurajoh, M.; Moru, K.; et al. Xanthine oxidoreductase activity is associated with serum uric acid and glycemic control in hemodialysis patients. Sci. Rep. 2017, 7, 15416. [Google Scholar] [CrossRef] [PubMed]
- Otaki, Y.; Watanabe, T.; Kinoshita, D.; Yokoyama, M.; Takahashi, T.; Toshima, T.; Sugai, T.; Murase, T.; Nakamura, T.; Nishiyama, S.; et al. Association of plasma xanthine oxidoreductase activity with severity and clinical outcome in patients with chronic heart failure. Int. J. Cardiol. 2017, 228, 151–157. [Google Scholar] [CrossRef]
- Furuhashi, M.; Matsumoto, M.; Tanaka, M.; Moniwa, N.; Murase, T.; Nakamura, T.; Ohnishi, H.; Saitoh, S.; Shimamoto, K.; Miura, T. Plasma xanthine oxidoreductase activity as a novel biomarker of metabolic disorders in a general population. Circ. J. 2018, 82, 1892–1899. [Google Scholar] [CrossRef] [PubMed]
- Kushiyama, A.; Okubo, H.; Sakoda, H.; Kikuchi, T.; Fujishiro, M.; Sato, H.; Kushiyama, S.; Iwashita, M.; Nishimura, F.; Fukushima, T.; et al. Xanthine oxidoreductase is involved in macrophage foam cell formation and atherosclerosis development. Arterioscler. Thromb. Vasc. Biol. 2012, 2, 291–298. [Google Scholar] [CrossRef]
- Shimamoto, K.; Ando, K.; Fujita, T.; Hasebe, N.; Higaki, J.; Horiuchi, M.; Imai, Y.; Imaizumi, T.; Ishimitsu, T.; Ito, M.; et al. Japanese Society of Hypertension Committee for Guidelines for the Management of Hypertension. The Japanese society of hypertension guidelines for the management of hypertension (JSH 2014). Hypertens. Res. 2014, 37, 253–390. [Google Scholar]
- Haneda, M.; Noda, M.; Origasa, H.; Noto, H.; Yabe, D.; Fujita, Y.; Goto, A.; Kondo, T.; Araki, E. Japanese clinical practice guideline for diabetes 2016. Diabetol. Int. 2018, 9, 1–45. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, M.; Yokote, K.; Arai, H.; Iida, M.; Ishigaki, Y.; Ishibashi, S.; Umemoto, S.; Egusa, G.; Ohmura, H.; Okamura, T.; et al. Japan atherosclerosis society (JAS) guidelines for prevention of atherosclerotic cardiovascular diseases 2017. J. Atheroscler. Thromb. 2018, 25, 846–984. [Google Scholar] [CrossRef]
- Murase, T.; Nampei, M.; Oka, M.; Miyachi, A.; Nakamura, T. A highly sensitive assay of human plasma xanthine oxidoreductase activity using stable isotope-labeled xanthine and LC/TQMS. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2016, 1039, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Murase, T.; Oka, M.; Nampei, M.; Miyachi, A.; Nakamura, T. A highly sensitive assay for xanthine oxidoreductase activity using a combination of [(13) C2, (15) N2]xanthine and liquid chromatography/triple quadrupole mass spectrometry. J. Label. Comp. Radiopharm. 2016, 59, 214–220. [Google Scholar] [CrossRef]
- Wilson, P.W.; D’Agostino, R.B.; Levy, D.; Belanger, A.M.; Silbershatz, H.; Kannel, W.B. Prediction of coronary heart disease using risk factor categories. Circulation 1998, 97, 1837–1847. [Google Scholar] [CrossRef]
- SPRINT Research Group; Wright, J.T., Jr.; Williamson, J.D.; Whelton, P.K.; Snyder, J.K.; Sink, K.M.; Rocco, M.V.; Reboussin, D.M.; Rahman, M.; Oparil, S.; et al. A randomized trial of intensive versus standard blood-pressure control. N. Engl. J. Med. 2015, 373, 2103–2116. [Google Scholar]
- Kuppusamy, U.R.; Indran, M.; Rokiah, P. Glycaemic control in relation to xanthine oxidase and antioxidant indices in Malaysian Type 2 diabetes patients. Diabet. Med. 2005, 22, 1343–1346. [Google Scholar] [CrossRef]
- Suvorava, T.; Kojda, G. Reactive oxygen species as cardiovascular mediators: Lessons from endothelial-specific protein overexpression mouse models. Biochim. Biophys. Acta 2009, 1787, 802–810. [Google Scholar] [CrossRef] [PubMed]
- Förstermann, U.; Xia, N.; Li, H. Roles of vascular oxidative stress and nitric oxide in the pathogenesis of atherosclerosis. Circ. Res. 2017, 120, 713–735. [Google Scholar] [CrossRef] [PubMed]
- Kelley, E.E. A new paradigm for XOR-catalyzed reactive species generation in the endothelium. Pharmacol. Rep. 2015, 67, 669–674. [Google Scholar] [CrossRef] [PubMed]
- Tsushima, Y.; Nishizawa, H.; Tochino, Y.; Nakatsuji, H.; Sekimoto, R.; Nagao, H.; Shirakura, T.; Kato, K.; Imaizumi, K.; Takahashi, H.; et al. Uric acid secretion from mouse adipose tissue uric acid secretion from adipose tissue and its increase in obesity. J. Biol. Chem. 2013, 288, 27138–27149. [Google Scholar] [CrossRef] [PubMed]
- Battelli, M.G.; Polito, L.; Bortolotti, M.; Bolognesi, A. Xanthine oxidoreductase-derived reactive species: Physiological and pathological effects. Oxid. Med. Cell. Longev. 2016, 2016, 3527579. [Google Scholar] [CrossRef]
- Matsushita, M.; Murase, T.; Nakamura, T.; Takayasu, T.; Asano, M.; Okajima, F.; Kobayashi, N.; Hata, N.; Asai, K.; Shimizu, W.; et al. Plasma xanthine oxidoreductase (XOR) activity in cardiovascular disease outpatients. Circ. Rep. 2020, 2, 104–112. [Google Scholar] [CrossRef]
- Yudkin, J.S.; Stehouwer, C.D.; Emeis, J.J.; Coppack, S.W. C-reactive protein in healthy subjects: Associations with obesity; insulin resistance; and endothelial dysfunction: A potential role for cytokines originating from adipose tissue? Arterioscler. Thromb. Vasc. Biol. 1999, 19, 972–978. [Google Scholar] [CrossRef] [PubMed]
- Washio, K.W.; Kusunoki, Y.; Murase, T.; Nakamura, T.; Osugi, K.; Ohigashi, M.; Sukenaga, T.; Ochi, F.; Matsuo, T.; Katsuno, T.; et al. Xanthine oxidoreductase activity is correlated with insulin resistance and subclinical inflammation in young humans. Metabolism 2017, 70, 51–56. [Google Scholar] [CrossRef]
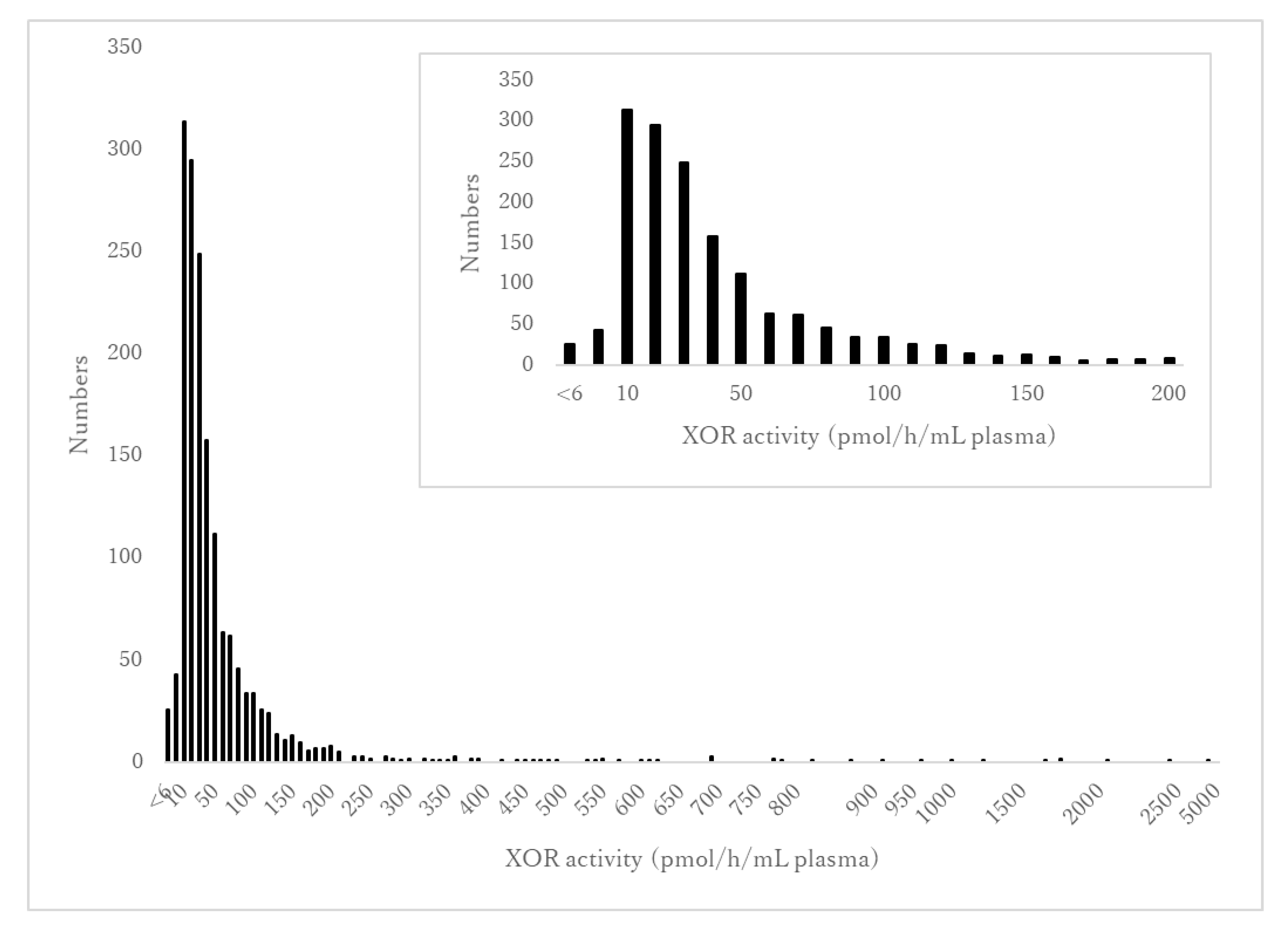
| Total | Males | Females | p Value | |
|---|---|---|---|---|
| No. of participants | 1631 | 510 | 1121 | |
| Age (years old) | 66.1 ± 9.6 | 69.1 ± 7.4 | 64.7 ± 10.1 | <0.001 |
| BMI (kg/m2) | 23.7 ± 9.4 | 23.9 ± 2.9 | 23.1 ± 3.6 | <0.001 |
| Current smoking (%) | 8.9 | 20.1 | 4.0 | <0.001 |
| Current drinking (%) | 40.7 | 71.1 | 27.3 | <0.001 |
| Alcohol consumption (%) | 1.3 | 4.3 | 0 | <0.001 |
| Medical history | ||||
| Hypertension (%) | 46.2 | 53.1 | 43.1 | <0.001 |
| Diabetes (%) | 8.9 | 13.5 | 6.8 | <0.001 |
| Dyslipidemia (%) | 41.8 | 39.4 | 42.8 | 0.196 |
| Framingham Risk Score ≥ 15 (%) | 3.5 | 9.2 | 0.9 | <0.001 |
| SBP (mmHg) | 133.7 ± 18.8 | 136.0 ± 17.6 | 132.6 ± 19.2 | 0.001 |
| DBP (mmHg) | 74.3 ± 10.8 | 76.3 ± 10.9 | 73.4 ± 10.6 | <0.001 |
| XOR (pmol/h/mL plasma) | 34.8 (20.8–62.7) | 43.7 (25.3–77.3) | 31.6 (19.5–58.4) | <0.001 |
| UA (mg/mL) | 5.0 ± 4.9 | 5.9 ± 1.2 | 4.6 ± 1.0 | <0.001 |
| AST (IU/L) | 23.0 (20.0–27.0) | 24.0 (21.0–30.0) | 24.2 (20.0–26.0) | <0.001 |
| ALT (IU/L) | 18.0 (14.0–23.0) | 20.0 (16.0–26.0) | 19.6 (14.0–22.0) | <0.001 |
| LDL-cholesterol (mg/dL) | 114.6 ± 28.1 | 107.9 ± 26.2 | 117.6 ± 28.5 | <0.001 |
| HDL-cholesterol (mg/dL) | 61.7 ± 14.5 | 56.4 ± 14.1 | 64.1 ± 14.1 | <0.001 |
| Total-choresterol (mg/dL) | 208.8 ± 34.5 | 196.5 ± 31.3 | 214.4 ± 34.5 | <0.001 |
| HbA1c (%) | 5.6 ± 0.6 | 5.7 ± 0.7 | 5.6 ± 0.5 | 0.005 |
| r | p Value | |
|---|---|---|
| Age | 0.10 | <0.001 |
| Gender | −0.11 | <0.001 |
| BMI | 0.33 | <0.001 |
| SBP | 0.15 | <0.001 |
| DBP | 0.14 | <0.001 |
| Log10UA | 0.22 | <0.001 |
| Log10AST | 0.58 | <0.001 |
| Log10ALT | 0.68 | <0.001 |
| LDL-cholesterol | 0.03 | 0.304 |
| HDL-cholesterol | −0.19 | <0.001 |
| Total-cholesterol | −0.03 | 0.315 |
| HbA1c | 0.25 | <0.001 |
| β | p Value | |
|---|---|---|
| Age | 0.04 | 0.081 |
| Gender | −0.01 | 0.772 |
| BMI | 0.26 | <0.001 |
| Hypertension | 0.03 | 0.188 |
| Diabetes | 0.09 | <0.001 |
| Dyslipidemia | 0.08 | 0.001 |
| Alcohol consumption | 0.01 | 0.811 |
| Log10UA | 0.13 | <0.001 |
| No of Subjects | FRS | p Value | |
|---|---|---|---|
| Hypertension | |||
| + | 754 | 7.0 ± 4.2 | <0.001 |
| − | 877 | 4.1 ± 2.9 | |
| Diabetes | |||
| + | 145 | 11.9 ± 6.6 | <0.001 |
| − | 1486 | 4.8 ± 2.8 | |
| Dyslipidemia | |||
| + | 681 | 6.4 ± 4.4 | <0.001 |
| − | 950 | 4.7 ± 3.3 |
| XOR Activity Quartile (pmol/h/mL Plasma) | Q1 (–20.80) | Q2 (20.81–34.80) | Q3 (34.81–62.70) | Q4 (62.71–) | p Value |
|---|---|---|---|---|---|
| No. of participants | 411 | 405 | 408 | 407 | |
| Age (years old) | 64.2 ± 11.5 | 65.9 ± 9.7 | 67.1 ± 8.6 | 67.3 ± 7.8 | <0.001 |
| Gender (female, %) | 76.6 | 73.8 | 62.3 | 62.2 | <0.001 |
| BMI (kg/m2) | 22.1 ± 3.0 | 22.8 ± 2.9 | 23.6 ± 3.3 | 24.9 ± 3.8 | <0.001 |
| Current smoking (%) | 8.3 | 10.1 | 8.6 | 8.7 | 0.906 |
| Current drinkng (%) | 34.1 | 40.2 | 44.1 | 43.5 | 0.134 |
| Alcohol consumption (%) | 0.7 | 0.7 | 2.9 | 1.0 | 0.015 |
| Current drinking (%) | 34.1 | 40.2 | 44.1 | 43.5 | 0.134 |
| Medical history | |||||
| Hypertension (%) | 37.0 | 44.7 | 49.8 | 53.6 | <0.001 |
| Diabetes (%) | 4.9 | 6.2 | 8.6 | 16.0 | <0.001 |
| Dyslipidemia (%) | 32.6 | 40.7 | 41.9 | 51.8 | <0.001 |
| Framingham Risk Score ≥ 15 (%) | 1.5 | 2.5 | 3.4 | 6.6 | <0.001 |
| SBP (mmHg) | 130.0 ± 19.7 | 132.7 ± 18.9 | 135.2 ± 17.3 | 136.8 ± 18.5 | <0.001 |
| DBP (mmHg) | 72.4 ± 11.0 | 73.9 ± 10.8 | 74.9 ± 10.2 | 75.8 ± 11.0 | <0.001 |
| UA (mg/mL) | 4.7 ± 1.2 | 4.8 ± 1.1 | 5.3 ± 1.2 | 5.3 ± 1.2 | <0.001 |
| AST (IU/L) | 21.0 (18.0–23.0) | 22.0 (19.0–25.0) | 24.0 (21.0–27.0) | 28.0 (24.0–34.0) | <0.001 |
| ALT (IU/L) | 14.0 (12.0–17.0) | 16.0 (14.0–19.0) | 19.0 (16.0–23.0) | 26.0 (20.0–35.0) | <0.001 |
| LDL-cholesterol (mg/dL) | 113.8 ± 26.3 | 114.8 ± 27.0 | 114.8 ± 29.4 | 114.9 ± 29.7 | 0.932 |
| HDL-cholesterol (mg/dL) | 64.2 ± 14.9 | 64.0 ± 14.8 | 61.7 ± 13.9 | 56.9 ± 13.3 | <0.001 |
| Total-cholesterol (mg/dL) | 209.0 ± 34.0 | 210.8 ± 32.5 | 210.0 ± 35.3 | 205.7 ± 36.1 | 0.169 |
| HbA1c (%) | 5.5 ± 0.4 | 5.6 ± 0.5 | 5.6 ± 0.5 | 5.8 ± 0.7 | <0.001 |
| XOR Quartiles | OR | 95% CI | p Value |
|---|---|---|---|
| Q1 | Reference | ||
| Q2 | 1.56 | 0.56–4.35 | 0.396 |
| Q3 | 1.68 | 0.63–4.50 | 0.298 |
| Q4 | 2.93 | 1.16–7.40 | 0.023 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kotozaki, Y.; Satoh, M.; Tanno, K.; Ohmomo, H.; Otomo, R.; Tanaka, F.; Nasu, T.; Taguchi, S.; Kikuchi, H.; Kobayashi, T.; et al. Plasma Xanthine Oxidoreductase Activity Is Associated with a High Risk of Cardiovascular Disease in a General Japanese Population. Int. J. Environ. Res. Public Health 2021, 18, 1894. https://doi.org/10.3390/ijerph18041894
Kotozaki Y, Satoh M, Tanno K, Ohmomo H, Otomo R, Tanaka F, Nasu T, Taguchi S, Kikuchi H, Kobayashi T, et al. Plasma Xanthine Oxidoreductase Activity Is Associated with a High Risk of Cardiovascular Disease in a General Japanese Population. International Journal of Environmental Research and Public Health. 2021; 18(4):1894. https://doi.org/10.3390/ijerph18041894
Chicago/Turabian StyleKotozaki, Yuka, Mamoru Satoh, Kozo Tanno, Hideki Ohmomo, Ryo Otomo, Fumitaka Tanaka, Takahito Nasu, Satoru Taguchi, Hiroto Kikuchi, Takamasa Kobayashi, and et al. 2021. "Plasma Xanthine Oxidoreductase Activity Is Associated with a High Risk of Cardiovascular Disease in a General Japanese Population" International Journal of Environmental Research and Public Health 18, no. 4: 1894. https://doi.org/10.3390/ijerph18041894
APA StyleKotozaki, Y., Satoh, M., Tanno, K., Ohmomo, H., Otomo, R., Tanaka, F., Nasu, T., Taguchi, S., Kikuchi, H., Kobayashi, T., Shimizu, A., Sakata, K., Hitomi, J., Sobue, K., & Sasaki, M. (2021). Plasma Xanthine Oxidoreductase Activity Is Associated with a High Risk of Cardiovascular Disease in a General Japanese Population. International Journal of Environmental Research and Public Health, 18(4), 1894. https://doi.org/10.3390/ijerph18041894







