Characterization of IL-2 Stimulation and TRPM7 Pharmacomodulation in NK Cell Cytotoxicity and Channel Co-Localization with PIP2 in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Patients
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Peripheral Blood Mononuclear Cell Isolation and Natural Killer Cell Purification
2.3. Interleukin-2 Stimulation and Drug Treatment
2.4. Natural Killer Cell Cytotoxic Activity Assay
2.5. Co-Localization of Transient Receptor Potential Melastatin 7 with Actin and Phosphoinositol 4,5-Bisphosphate in Natural Killer Cells
2.6. Statistical Analysis
3. Results
3.1. Participant Demographics
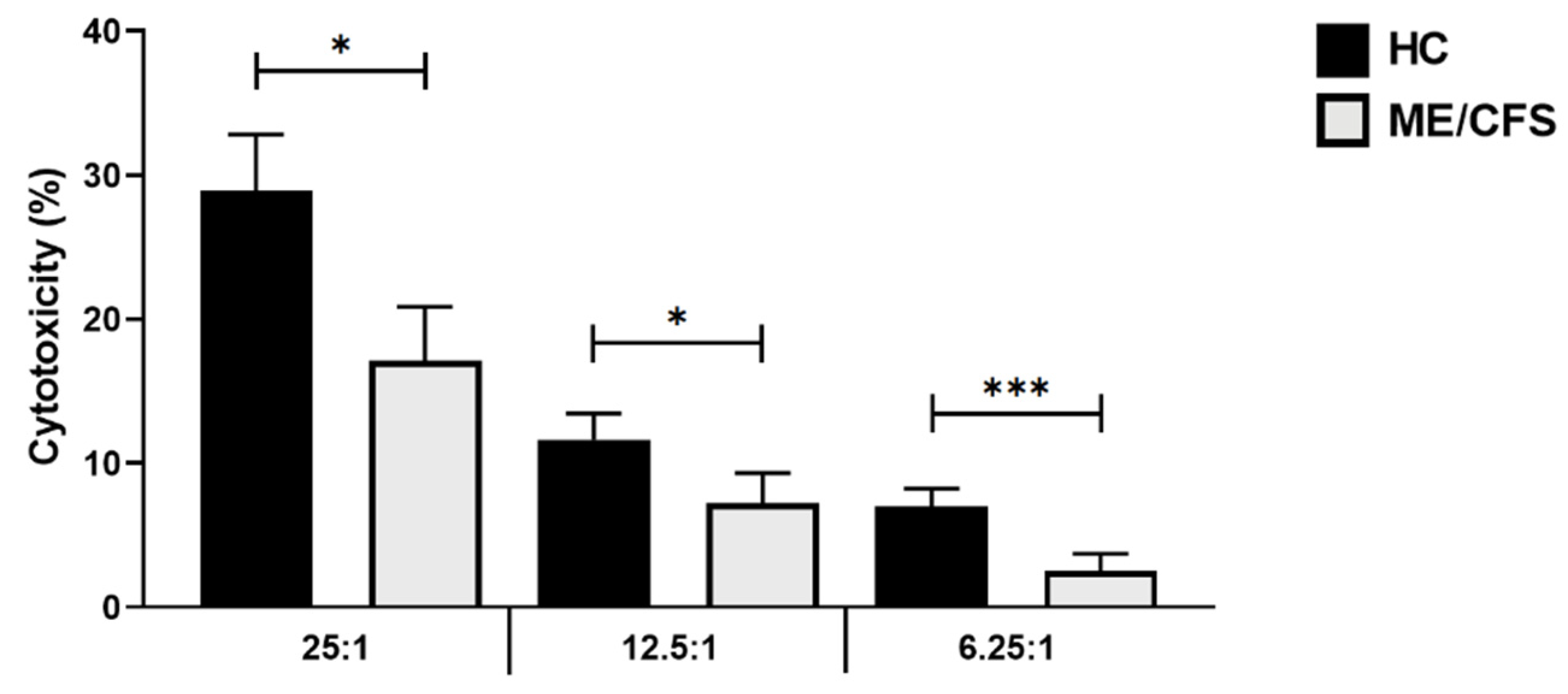
3.2. Baseline Natural Killer Cell Cytotoxic Activity
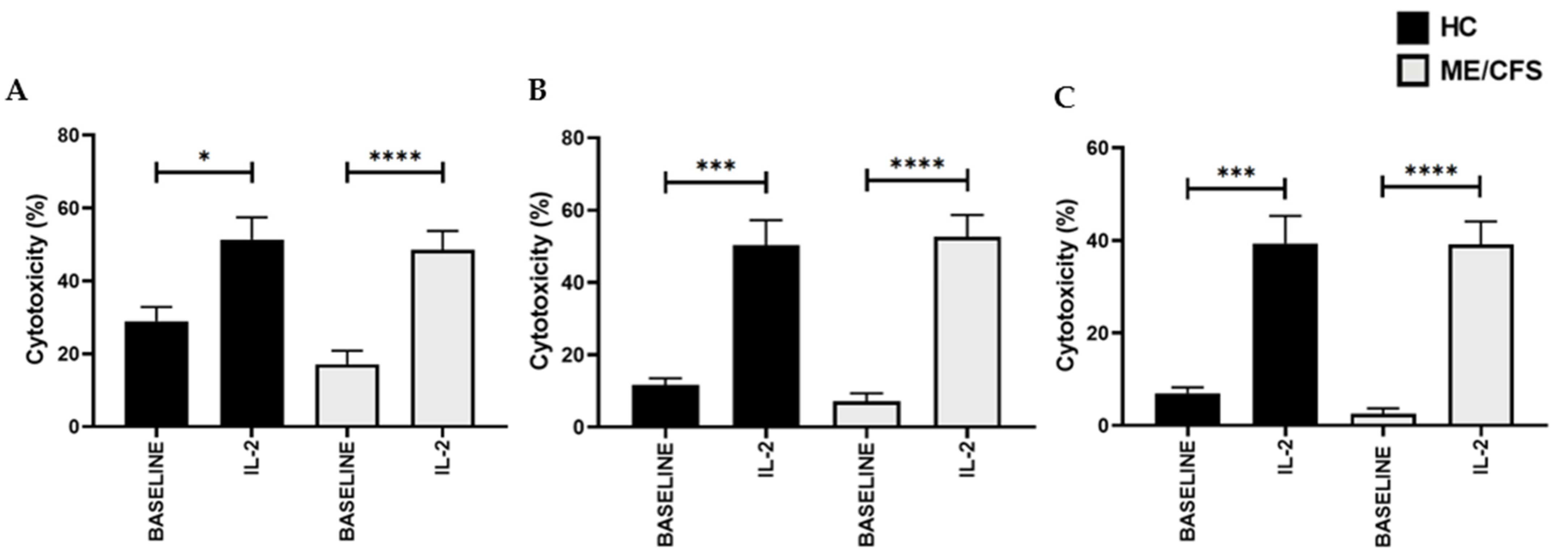
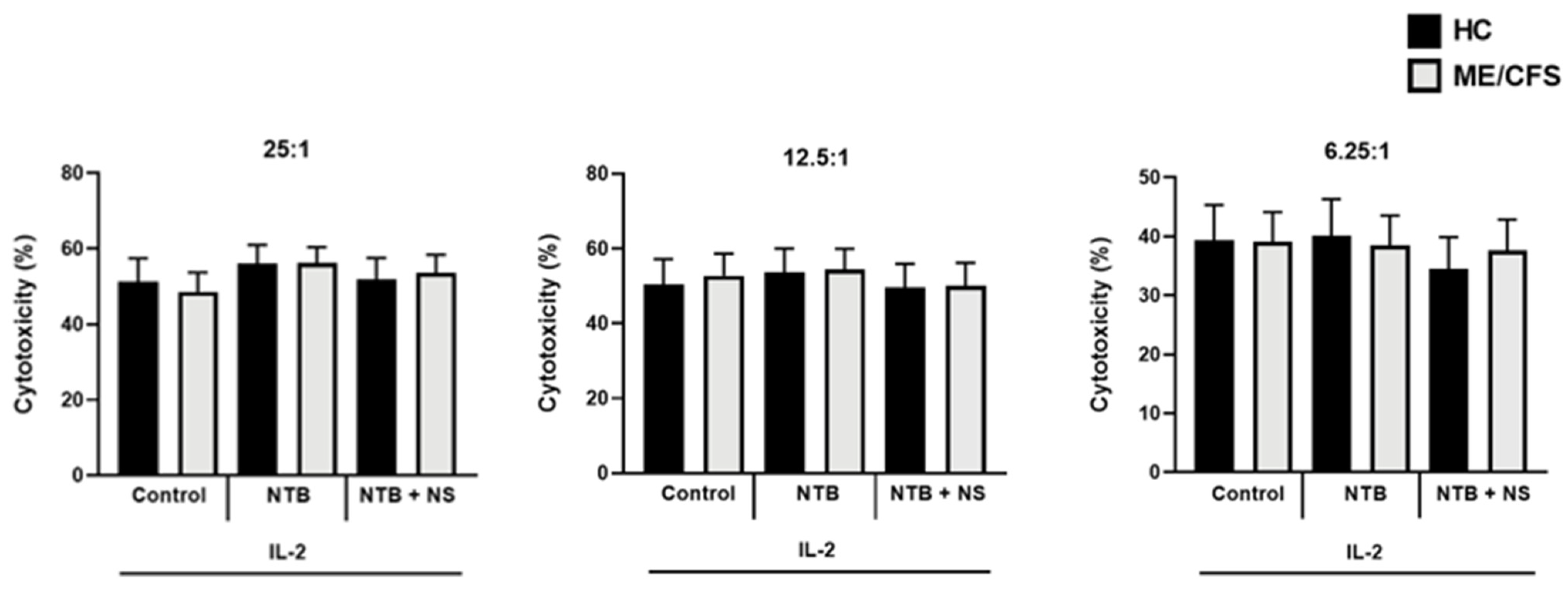
3.3. Natural Killer Cell Cytotoxic Activity Post-Interleukin-2 Stimulation and Drug Treatment
3.4. Co-Localization of Transient Receptor Potential Melastatin 7 with Actin
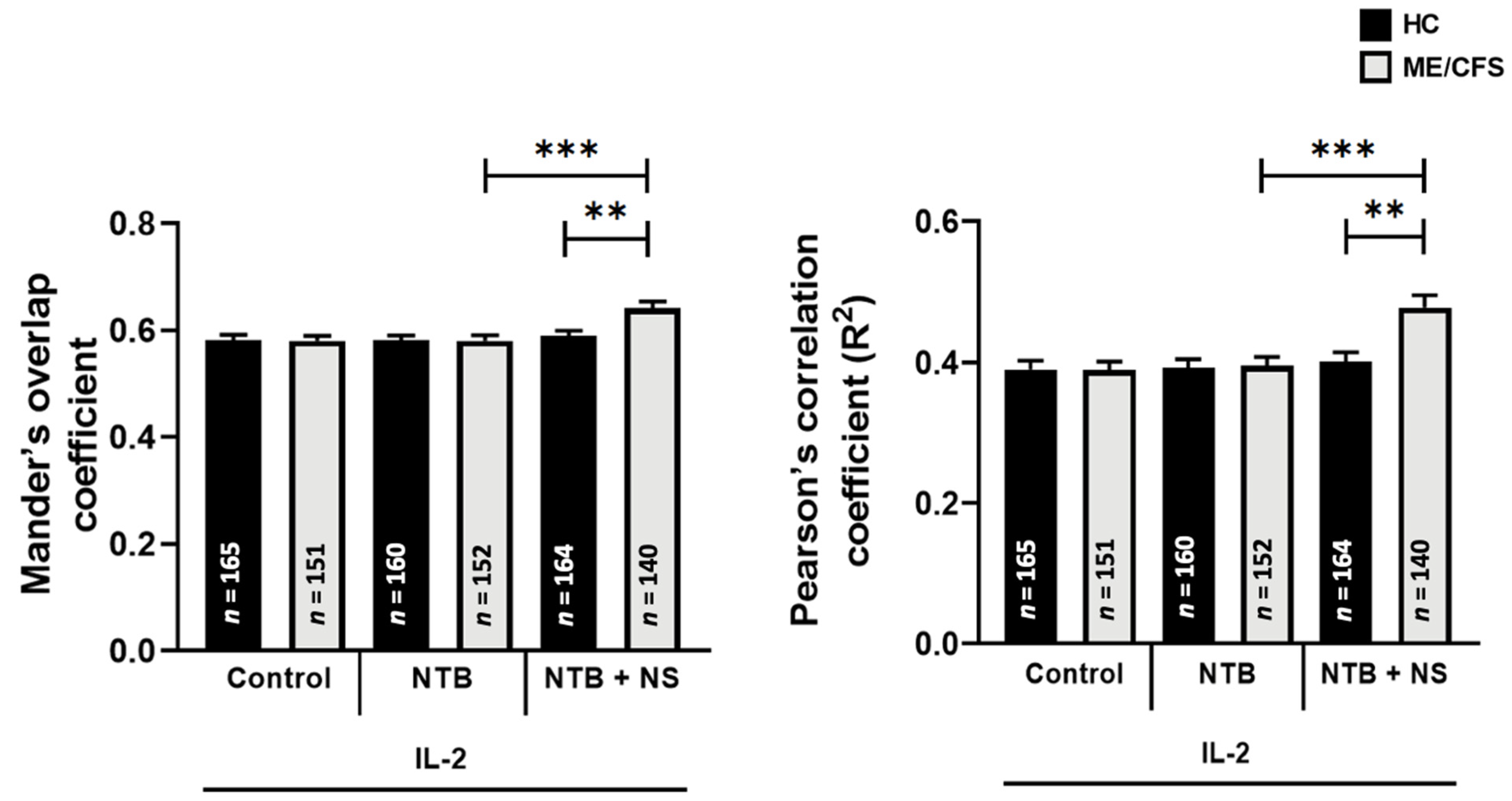
3.5. Co-Localization of Transient Receptor Potential Melastatin 7 with Phosphatidylinositol 4,5-Bisphosphate
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Appendix A
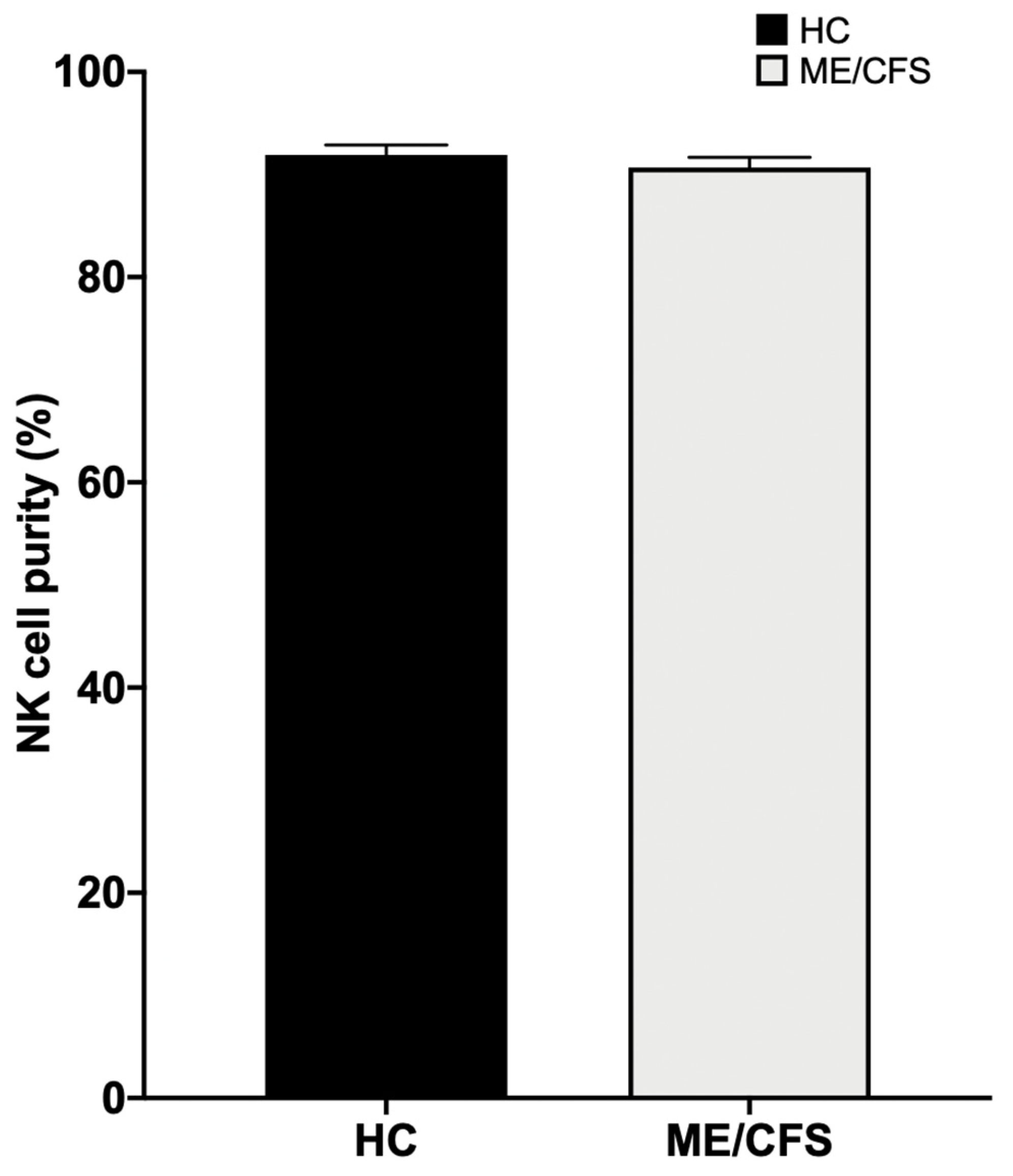
Appendix B
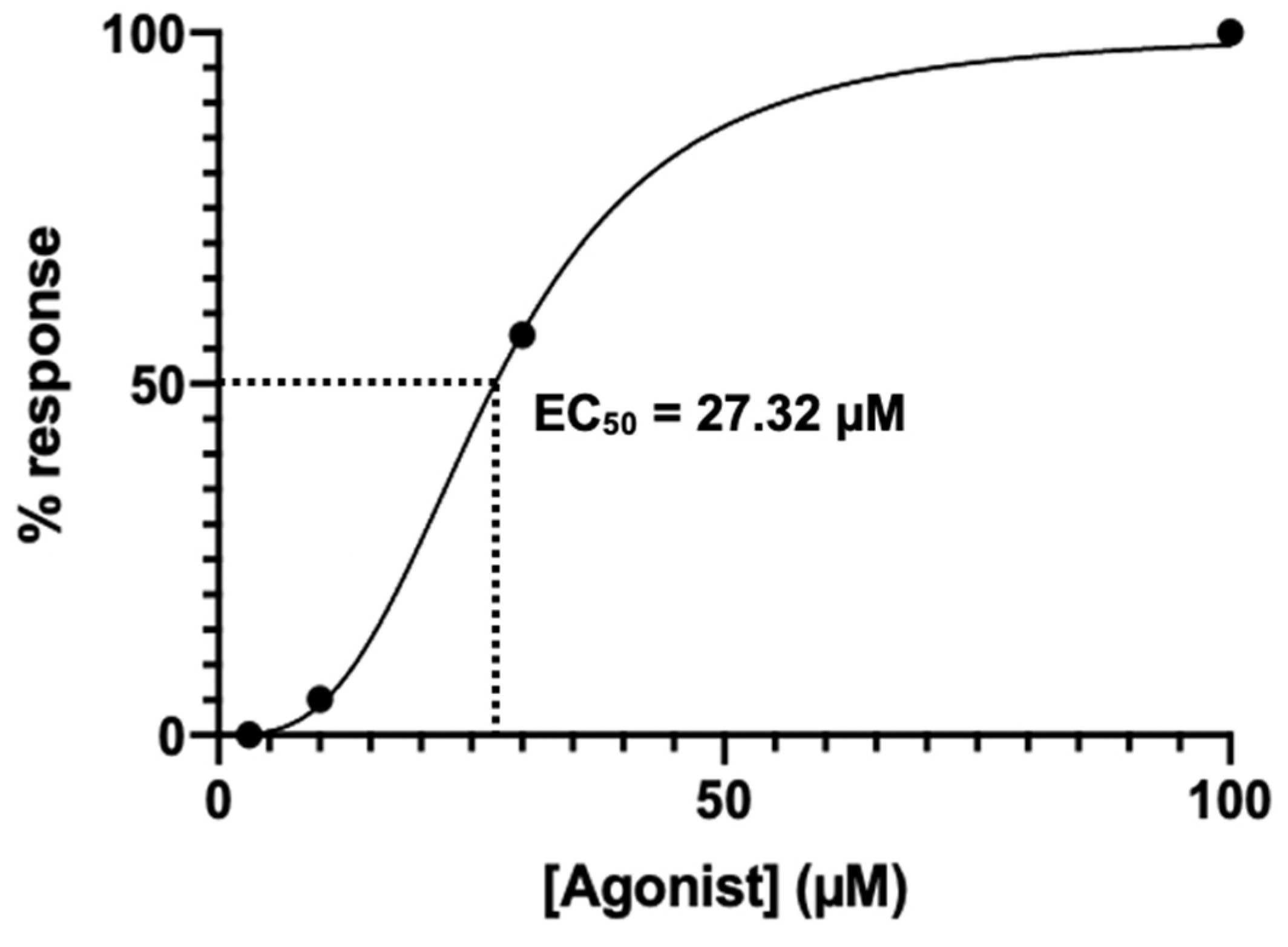
Appendix C
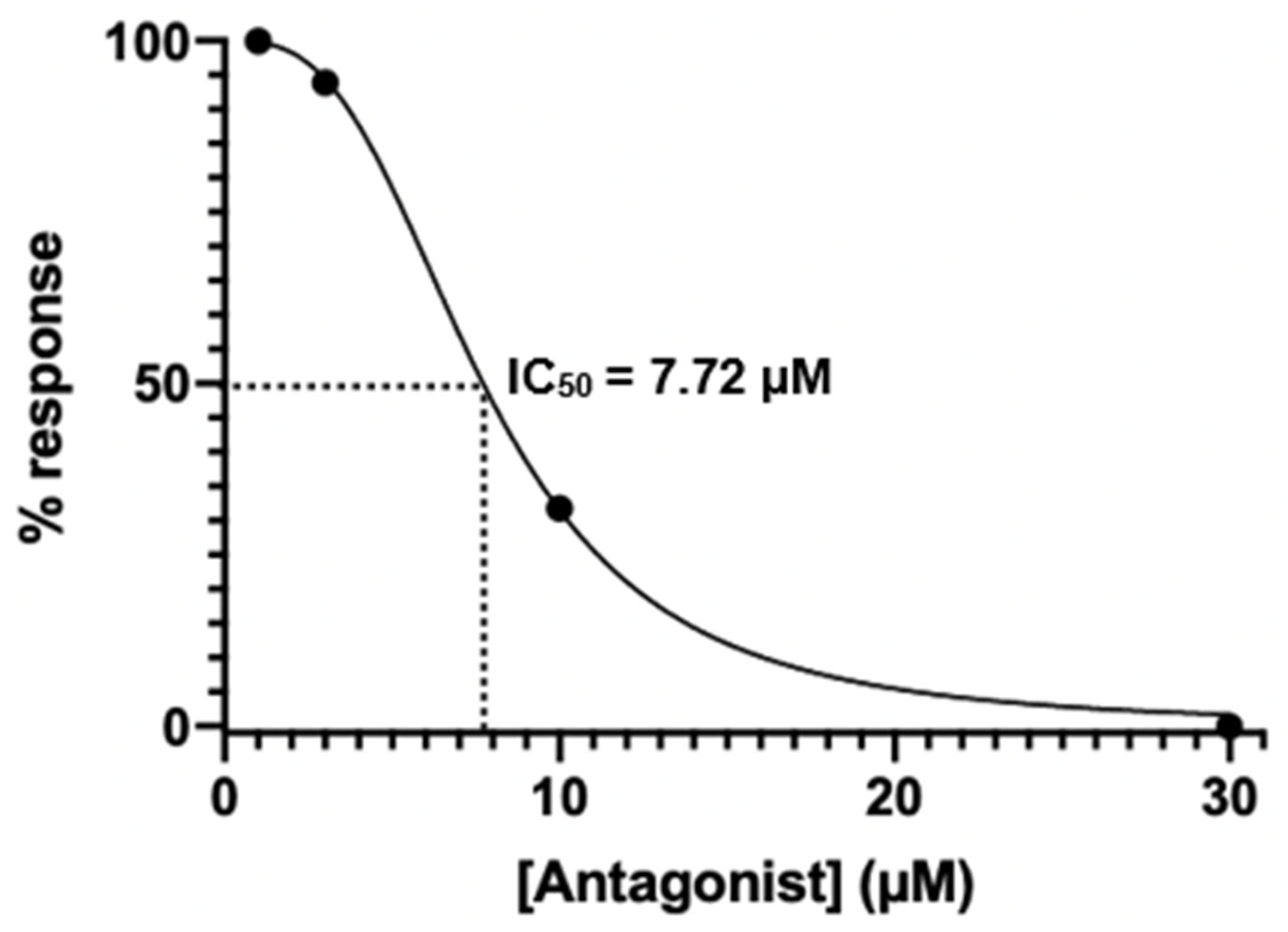
Appendix D
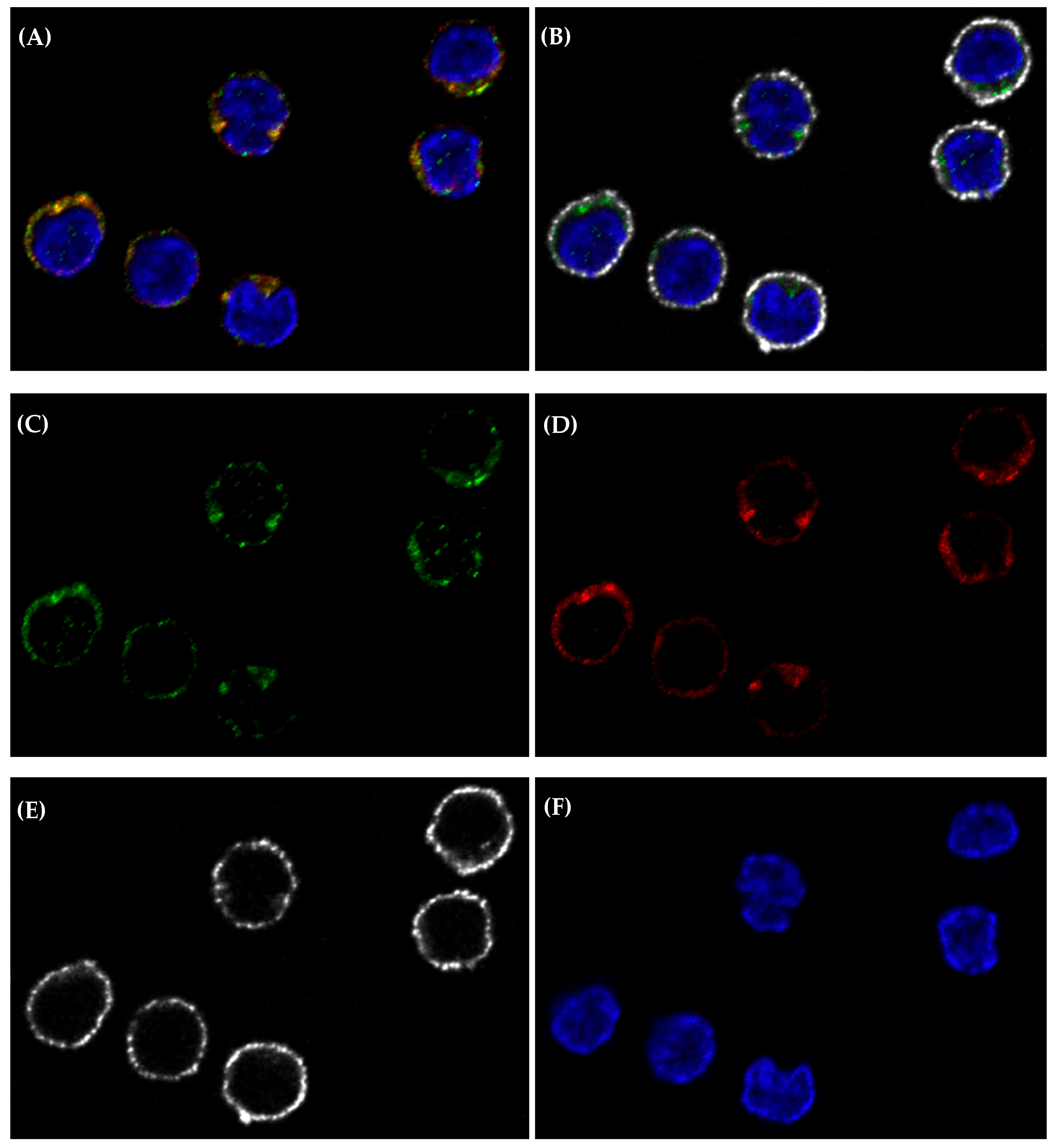
Appendix E

References
- Vivier, E.; Tomasello, E.; Baratin, M.; Walzer, T.; Ugolini, S. Functions of natural killer cells. Nat. Immunol. 2008, 9, 503–510. [Google Scholar] [CrossRef]
- Fehniger, T.; Cooper, M.; Nuovo, G.J.; Cella, M.; Facchetti, F.; Colonna, M.; Caligiuri, M.A. CD56bright natural killer cells are present in human lymph nodes and are activated by T cell-derived IL-2: A potential new link between adaptive and innate immunity. Blood 2003, 101, 3052–3057. [Google Scholar] [CrossRef] [Green Version]
- Caligiuri, M.A. Human natural killer cells. Blood 2008, 112, 461–469. [Google Scholar] [CrossRef]
- Poli, A.; Michel, T.; Thérésine, M.; Andrès, E.; Hentges, F.; Zimmer, J. CD56brightnatural killer (NK) cells: An important NK cell subset. Immunology 2009, 126, 458–465. [Google Scholar] [CrossRef]
- Lopez, J.A.; Brennan, A.J.; Whisstock, J.C.; Voskoboinik, I.; Trapani, J.A. Protecting a serial killer: Pathways for perforin trafficking and self-defence ensure sequential target cell death. Trends Immunol. 2012, 33, 406–412. [Google Scholar] [CrossRef]
- Voskoboinik, I.; Whisstock, J.; Trapani, J. Perforin and granzymes: Function, dysfunction and human pathology. Nat. Rev. Immunol. 2015, 15, 388–400. [Google Scholar] [CrossRef]
- Bolitho, P.; Voskoboinik, I.; Trapani, J.; Smyth, M.J. Apoptosis induced by the lymphocyte effector molecule perforin. Curr. Opin. Immunol. 2007, 19, 339–347. [Google Scholar] [CrossRef]
- Wang, S.; El-Deiry, W.S. TRAIL and apoptosis induction by TNF-family death receptors. Oncogene 2003, 22, 8628–8633. [Google Scholar] [CrossRef] [Green Version]
- Screpanti, V.; Wallin, R.P.; Grandien, A.; Ljunggren, H.-G. Impact of FASL-induced apoptosis in the elimination of tumor cells by NK cells. Mol. Immunol. 2005, 42, 495–499. [Google Scholar] [CrossRef]
- Olejniczak, K.; Kasprzak, A. Biological properties of interleukin 2 and its role in pathogenesis of selected diseases—A review. Med. Sci. Monit. 2008, 14, RA179–RA189. [Google Scholar]
- Henney, C.S.; Kuribayashi, K.; Kern, D.E.; Gillis, S. Interleukin-2 augments natural killer cell activity. Nature 1981, 291, 335–338. [Google Scholar] [CrossRef]
- Carson, W.E.; Parihar, R.; Lindemann, M.J.; Personeni, N.; Dierksheide, J.; Meropol, N.J.; Baselga, J.; Caligiuri, M.A. Interleukin-2 enhances the natural killer cell response to Herceptin-coated Her2/neu-positive breast cancer cells. Eur. J. Immunol. 2001, 31, 3016–3025. [Google Scholar] [CrossRef]
- Mirandola, P.; Ponti, C.; Gobbi, G.; Sponzilli, I.; Melloni, E.; Vitale, M. The response of human natural killer cells to interleukin-2. J. Endocrinol. Invest. 2004, 27, 146–150. [Google Scholar]
- Cooper, M.; Fehniger, T.; Caligiuri, M.A. The biology of human natural killer-cell subsets. Trends Immunol. 2001, 22, 633–640. [Google Scholar] [CrossRef]
- Becknell, B.; Caligiuri, M.A. Interleukin-2, Interleukin-15, and Their Roles in Human Natural Killer Cells. Adv. Immunol. 2005, 86, 209–239. [Google Scholar] [CrossRef]
- Hartzell, C.A.; Jankowska, K.I.; Burkhardt, J.; Lewis, R.S. Calcium influx through CRAC channels controls actin organization and dynamics at the immune synapse. eLife 2016, 5, e14850. [Google Scholar] [CrossRef]
- Clapham, D.E. Calcium Signaling. Cell 2007, 131, 1047–1058. [Google Scholar] [CrossRef] [Green Version]
- Krzewski, K.; Coligan, J.E. Human NK cell lytic granules and regulation of their exocytosis. Front. Immunol. 2012, 3, 335. [Google Scholar] [CrossRef] [Green Version]
- Masi, G.; Baldari, C.T. Signaling at the immune synapse: Vesicular trafficking takes the stage. Cell. Mol. Immunol. 2013, 10, 459–462. [Google Scholar] [CrossRef] [Green Version]
- Smyth, M.J.; Cretney, E.; Kelly, J.M.; Westwood, J.A.; Street, S.E.; Yagita, H.; Takeda, K.; van Dommelen, S.L.; Degli-Esposti, M.A.; Hayakawa, Y. Activation of NK cell cytotoxicity. Mol. Immunol. 2005, 42, 501–510. [Google Scholar] [CrossRef]
- Nicotera, P.; Orrenius, S. The role of calcium in apoptosis. Cell Calcium 1998, 23, 173–180. [Google Scholar] [CrossRef]
- Pinton, P.; Giorgi, C.; Siviero, R.; Zecchini, E.; Rizzuto, R. Calcium and apoptosis: ER-mitochondria Ca2+ transfer in the control of apoptosis. Oncogene 2008, 27, 6407–6418. [Google Scholar] [CrossRef] [Green Version]
- Enandagopal, N.; Ali, A.K.; Komal, A.K.; Elee, S.-H. The Critical Role of IL-15-PI3K-mTOR Pathway in Natural Killer Cell Effector Functions. Front. Immunol. 2014, 5, 187. [Google Scholar] [CrossRef]
- Tassi, I.; Klesney-Tait, J.; Colonna, M. Dissecting natural killer cell activation pathways through analysis of genetic mutations in human and mouse. Immunol. Rev. 2006, 214, 92–105. [Google Scholar] [CrossRef]
- Broad, L.M.; Braun, F.-J.; Lievremont, J.-P.; Bird, G.S.J.; Kurosaki, T., Jr.; Putney, J.W. Role of the Phospholipase C-Inositol 1,4,5-Trisphosphate Pathway in Calcium Release-activated Calcium Current and Capacitative Calcium Entry. J. Biol. Chem. 2001, 276, 15945–15952. [Google Scholar] [CrossRef] [Green Version]
- Putney, J.W.; Steinckwich-Besançon, N.; Numaga-Tomita, T.; Davis, F.M.; Desai, P.N.; D’Agostin, D.M.; Wu, S.; Bird, G.S. The functions of store-operated calcium channels. Biochim. Biophys. Acta Bioenerg. 2016, 1864, 900–906. [Google Scholar] [CrossRef]
- Nilius, B.; Owsianik, G. The transient receptor potential family of ion channels. Genome Biol. 2011, 12, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Nadler, M.J.S.; Hermosura, M.C.; Inabe, K.; Perraud, A.-L.; Zhu, Q.; Stokes, A.; Kurosaki, T.; Kinet, J.-P.; Penner, R.; Scharenberg, A.M.; et al. LTRPC7 is a Mg·ATP-regulated divalent cation channel required for cell viability. Nat. Cell Biol. 2001, 411, 590–595. [Google Scholar] [CrossRef]
- Schmitz, C.; Perraud, A.-L.; Johnson, C.O.; Inabe, K.; Smith, M.K.; Penner, R.; Kurosaki, T.; Fleig, A.; Scharenberg, A.M. Regulation of Vertebrate Cellular Mg2+ Homeostasis by TRPM7. Cell 2003, 114, 191–200. [Google Scholar] [CrossRef] [Green Version]
- Monteilh-Zoller, M.K.; Hermosura, M.C.; Nadler, M.J.; Scharenberg, A.M.; Penner, R.; Fleig, A. TRPM7 Provides an Ion Channel Mechanism for Cellular Entry of Trace Metal Ions. J. Gen. Physiol. 2002, 121, 49–60. [Google Scholar] [CrossRef] [Green Version]
- Smyth, J.T.; Hwang, S.-Y.; Tomita, T.; DeHaven, W.I.; Mercer, J.C.; Putney, J.W. Activation and regulation of store-operated calcium entry. J. Cell. Mol. Med. 2010, 14, 2337–2349. [Google Scholar] [CrossRef]
- Deason-Towne, F.; Perraud, A.-L.; Schmitz, C. Identification of Ser/Thr phosphorylation sites in the C2-domain of phospholipase C γ2 (PLCγ2) using TRPM7-kinase. Cell. Signal. 2012, 24, 2070–2075. [Google Scholar] [CrossRef] [Green Version]
- Zou, Z.-G.; Rios, F.J.; Montezano, A.C.; Touyz, R.M. TRPM7, Magnesium, and Signaling. Int. J. Mol. Sci. 2019, 20, 1877. [Google Scholar] [CrossRef] [Green Version]
- Faouzi, M.; Kilch, T.; Horgen, F.D.; Fleig, A.; Penner, R. The TRPM7 channel kinase regulates store-operated calcium entry. J. Physiol. 2017, 595, 3165–3180. [Google Scholar] [CrossRef] [Green Version]
- Beesetty, P.; Wieczerzak, K.B.; Gibson, J.N.; Kaitsuka, T.; Luu, C.T.; Matsushita, M.; Kozak, J.A. Inactivation of TRPM7 kinase in mice results in enlarged spleens, reduced T-cell proliferation and diminished store-operated calcium entry. Sci. Rep. 2018, 8, 1–22. [Google Scholar] [CrossRef]
- Hofmann, T.; Schäfer, S.; Linseisen, M.; Sytik, L.; Gudermann, T.; Chubanov, V. Activation of TRPM7 channels by small molecules under physiological conditions. Eur. J. Physiol. 2014, 466, 2177–2189. [Google Scholar] [CrossRef]
- Chubanov, V.; Schäfer, S.; Ferioli, S.; Gudermann, T. Natural and Synthetic Modulators of the TRPM7 Channel. Cells 2014, 3, 1089–1101. [Google Scholar] [CrossRef]
- Chubanov, V.; Gudermann, T. Mapping TRPM7 Function by NS8593. Int. J. Mol. Sci. 2020, 21, 7017. [Google Scholar] [CrossRef]
- Carruthers, B.M.; van de Sande, M.I.; de Meirleir, K.L.; Klimas, N.G.; Broderick, G.; Mitchell, T.; Staines, D.; Powles, A.C.P.; Speight, N.; Vallings, R.; et al. Myalgic encephalomyelitis: International Consensus Criteria. J. Intern. Med. 2011, 270, 327–338. [Google Scholar] [CrossRef] [Green Version]
- Chacko, A.; Staines, D.R.; Johnston, S.C.; Marshall-Gradisnik, S.M. Dysregulation of Protein Kinase Gene Expression in NK Cells from Chronic Fatigue Syndrome/Myalgic Encephalomyelitis Patients. Gene Regul. Syst. Biol. 2016, 10, 85–93. [Google Scholar] [CrossRef]
- Nguyen, T.; Staines, D.; Nilius, B.; Smith, P.; Marshall-Gradisnik, S. Novel identification and characterisation of Transient receptor potential melastatin 3 ion channels on Natural Killer cells and B lymphocytes: Effects on cell signalling in Chronic fatigue syndrome/Myalgic encephalomyelitis patients. Biol. Res. 2016, 49, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, T.; Johnston, S.; Clarke, L.; Smith, P.; Staines, D.; Marshall-Gradisnik, S. Impaired calcium mobilization in natural killer cells from chronic fatigue syndrome/myalgic encephalomyelitis patients is associated with transient receptor potential melastatin 3 ion channels. Clin. Exp. Immunol. 2016, 187, 284–293. [Google Scholar] [CrossRef] [Green Version]
- Huth, T.K.; Staines, D.; Marshall-Gradisnik, S. ERK1/2, MEK1/2 and p38 downstream signalling molecules impaired in CD56 dim CD16+ and CD56 bright CD16 dim/- natural killer cells in Chronic Fatigue Syndrome/Myalgic Encephalomyelitis patients. J. Transl. Med. 2016, 14, 97. [Google Scholar] [CrossRef] [Green Version]
- Cabanas, H.; Muraki, K.; Eaton, N.; Balinas, C.; Staines, D.; Marshall-Gradisnik, S. Loss of Transient Receptor Potential Melastatin 3 ion channel function in natural killer cells from Chronic Fatigue Syndrome/Myalgic Encephalomyelitis patients. Mol. Med. 2018, 24, 1–10. [Google Scholar] [CrossRef]
- Cabanas, H.; Muraki, K.; Balinas, C.; Eaton-Fitch, N.; Staines, D.; Marshall-Gradisnik, S. Validation of impaired Transient Receptor Potential Melastatin 3 ion channel activity in natural killer cells from Chronic Fatigue Syndrome/Myalgic Encephalomyelitis patients. Mol. Med. 2019, 25, 14. [Google Scholar] [CrossRef] [Green Version]
- Balinas, C.; Cabanas, H.; Staines, D.; Marshall-Gradisnik, S. Transient receptor potential melastatin 2 channels are overexpressed in myalgic encephalomyelitis/chronic fatigue syndrome patients. J. Transl. Med. 2019, 17, 1–11. [Google Scholar] [CrossRef]
- Eaton-Fitch, N.; Cabanas, H.; du Preez, S.; Staines, D.; Marshall-Gradisnik, S. The effect of IL-2 stimulation and treatment of TRPM3 on channel co-localisation with PIP2 and NK cell function in myalgic encephalomyelitis/chronic fatigue syndrome patients. J. Transl. Med. 2021, 19, 1–19. [Google Scholar] [CrossRef]
- Panda, S.K.; Ravindran, B. Isolation of Human PBMCs. BioProtoc. 2013, 3, e323. [Google Scholar] [CrossRef]
- Eaton-Fitch, N.; Du Preez, S.; Cabanas, H.; Staines, D.; Marshall-Gradisnik, S. A systematic review of natural killer cells profile and cytotoxic function in myalgic encephalomyelitis/chronic fatigue syndrome. Syst. Rev. 2019, 8, 1–13. [Google Scholar] [CrossRef]
- Klimas, N.G.; Salvato, F.R.; Morgan, R.; Fletcher, M.A. Immunologic abnormalities in chronic fatigue syndrome. J. Clin. Microbiol. 1990, 28, 1403–1410. [Google Scholar] [CrossRef] [Green Version]
- Maher, K.J.; Klimas, N.G.; Fletcher, M.A. Chronic fatigue syndrome is associated with diminished intracellular perforin. Clin. Exp. Immunol. 2005, 142, 505–511. [Google Scholar] [CrossRef]
- Fletcher, M.A.; Zeng, X.R.; Maher, K.; Levis, S.; Hurwitz, B.; Antoni, M.; Broderick, G.; Klimas, N.G. Biomarkers in Chronic Fatigue Syndrome: Evaluation of Natural Killer Cell Function and Dipeptidyl Peptidase IV/CD26. PLoS ONE 2010, 5, e10817. [Google Scholar] [CrossRef]
- Brenu, E.W.; van Driel, M.L.; Staines, D.R.; Ashton, K.J.; Ramos, S.B.; Keane, J.; Klimas, N.G.; Marshall-Gradisnik, S.M. Immunological abnormalities as potential biomarkers in Chronic Fatigue Syndrome/Myalgic Encephalomyelitis. J. Transl. Med. 2011, 9, 81. [Google Scholar] [CrossRef] [Green Version]
- Brenu, E.W.; Van Driel, M.L.; Staines, D.R.; Ashton, K.J.; Hardcastle, S.L.; Keane, J.; Tajouri, L.; Peterson, D.; Ramos, S.B.; Marshall-Gradisnik, S.M. Longitudinal investigation of natural killer cells and cytokines in chronic fatigue syndrome/myalgic encephalomyelitis. J. Transl. Med. 2012, 10, 88. [Google Scholar] [CrossRef] [Green Version]
- Fan, M.Y.; Turka, L.A. Immunometabolism and PI(3)K Signaling As a Link between IL-2, Foxp3 Expression, and Suppressor Function in Regulatory T Cells. Front. Immunol. 2018, 9, 69. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, T.; Mattarollo, S.R. Natural killer cell metabolism. Mol. Immunol. 2019, 115, 3–11. [Google Scholar] [CrossRef]
- Goldsmith, Z.G.; Dhanasekaran, D.N. G Protein regulation of MAPK networks. Oncogene 2007, 26, 3122–3142. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Allan, D.S.J.; Krzewski, K.; Ge, B.; Kopcow, H.; Strominger, J.L. CD28-stimulated ERK2 phosphorylation is required for polarization of the microtubule organizing center and granules in YTS NK cells. Proc. Natl. Acad. Sci. USA 2006, 103, 10346–10351. [Google Scholar] [CrossRef] [Green Version]
- Chuderland, D.; Seger, R. Calcium regulates ERK signaling by modulating its protein-protein interactions. Commun. Integr. Biol. 2008, 1, 4–5. [Google Scholar] [CrossRef] [Green Version]
- Lanier, L.L. Up on the tightrope: Natural killer cell activation and inhibition. Nat. Immunol. 2008, 9, 495–502. [Google Scholar] [CrossRef]
- Schwarz, E.C.; Qu, B.; Hoth, M. Calcium, cancer and killing: The role of calcium in killing cancer cells by cytotoxic T lymphocytes and natural killer cells. Biochim. Biophys. Acta Bioenerg. 2013, 1833, 1603–1611. [Google Scholar] [CrossRef] [Green Version]
- Clark, K.; Langeslag, M.; Van Leeuwen, B.; Ran, L.; Ryazanov, A.G.; Figdor, C.; Moolenaar, W.H.; Jalink, K.; van Leeuwen, F.N. TRPM7, a novel regulator of actomyosin contractility and cell adhesion. EMBO J. 2006, 25, 290–301. [Google Scholar] [CrossRef]
- Runnels, L.; Yue, L.; Clapham, D. The TRPM7 channel is inactivated by PIP2 hydrolysis. Nat. Cell Biol. 2002, 4, 329–336. [Google Scholar] [CrossRef]
- Langeslag, M.; Clark, K.; Moolenaar, W.H.; van Leeuwen, F.N.; Jalink, K. Activation of TRPM7 Channels by Phospholipase C-coupled Receptor Agonists. J. Biol. Chem. 2007, 282, 232–239. [Google Scholar] [CrossRef] [Green Version]
- Macianskiene, R.; Gwanyanya, A.; Vereecke, J.; Mubagwa, K. Inhibition of the Magnesium-Sensitive TRPM7-like Channel in Cardiac Myocytes by Nonhydrolysable GTP Analogs: Involvement of Phosphoinositide Metabolism. Cell. Physiol. Biochem. 2008, 22, 109–118. [Google Scholar] [CrossRef]
- Callera, G.E.; He, Y.; Yogi, A.; Montezano, A.; Paravicini, T.; Yao, G.; Touyz, R.M. Regulation of the novel Mg2+ transporter transient receptor potential melastatin 7 (TRPM7) cation channel by bradykinin in vascular smooth muscle cells. J. Hypertens. 2009, 27, 155–166. [Google Scholar] [CrossRef]
- Yogi, A.; Callera, G.E.; Tostes, R.; Touyz, R.M. Bradykinin regulates calpain and proinflammatory signaling through TRPM7-sensitive pathways in vascular smooth muscle cells. Am. J. Physiol. Integr. Comp. Physiol. 2009, 296, R201–R207. [Google Scholar] [CrossRef] [Green Version]
- Davis, F.; Azimi, I.; Faville, R.A.; Peters, A.A.; Jalink, K.; Putney, J.; Goodhill, G.; Thompson, E.W.; Roberts-Thomson, S.; Monteith, G. Induction of epithelial–mesenchymal transition (EMT) in breast cancer cells is calcium signal dependent. Oncogene 2013, 33, 2307–2316. [Google Scholar] [CrossRef] [Green Version]
- Abiria, S.A.; Krapivinsky, G.; Sah, R.; Santa-Cruz, A.G.; Chaudhuri, D.; Zhang, J.; Adstamongkonkul, P.; DeCaen, P.G.; Clapham, D.E. TRPM7 senses oxidative stress to release Zn2+ from unique intracellular vesicles. Proc. Natl. Acad. Sci. USA 2017, 114, E6079–E6088. [Google Scholar] [CrossRef] [Green Version]
- Chokshi, R.; Matsushita, M.; Kozak, J.A. Detailed examination of Mg2+ and pH sensitivity of human TRPM7 channels. Am. J. Physiol. Physiol. 2012, 302, C1004–C1011. [Google Scholar] [CrossRef] [Green Version]
- Jahn, R.; Scheller, R.H. SNAREs—Engines for membrane fusion. Nat. Rev. Mol. Cell Biol. 2006, 7, 631–643. [Google Scholar] [CrossRef]
- Marshall-Gradisnik, S.; Huth, T.; Chacko, A.; Smith, P.; Staines, D.; Johnston, S. Natural killer cells and single nucleotide polymorphisms of specific ion channels and receptor genes in myalgic encephalomyelitis/chronic fatigue syndrome. Appl. Clin. Genet. 2016, 9, 39–47. [Google Scholar] [CrossRef] [Green Version]
- Gavériaux-Ruff, C.; Filliol, D.; Simonin, F.; Matthes, H.W.D.; Kieffer, B.L. Immunosuppression by δ-Opioid Antagonist Naltrindole: δ- and Triple μ/δ/κ-Opioid Receptor Knockout Mice Reveal a Nonopioid Activity. J. Pharmacol. Exp. Ther. 2001, 298, 1193–1198. [Google Scholar]
- House, R.V.; Thomas, P.T.; Kozak, J.T.; Bhargava, H.N. Suppression of immune function by non-peptidic delta opioid receptor antagonists. Neurosci. Lett. 1995, 198, 119–122. [Google Scholar] [CrossRef]
- Mundra, J.J.; Terskiy, A.; Howells, R.D. Naltrindole inhibits human multiple myeloma cell proliferation in vitro and in a murine xenograft model in vivo. J. Pharmacol. Exp. Ther. 2012, 342, 273–287. [Google Scholar] [CrossRef] [Green Version]

| Category | Item | HC | ME/CFS | p-Value |
|---|---|---|---|---|
| General demographics | Age (years) | 48.1 ± 2.1 | 48.5 ± 2.0 | 0.7264 |
| Gender | ||||
| Male (%, n) | 23.5, 5 | 23.5, 5 | ||
| Female (%, n) | 76.5, 13 | 76.5, 13 | ||
| BMI (kg/m2) | 23.9 ± 0.9 | 25.2 ± 1.0 | 0.3435 | |
| WHODAS | Understanding and communication | 7.1 ± 2.0 | 41.6 ± 3.1 | **** |
| Mobility | 2.4 ± 1.4 | 38.2 ± 6.2 | **** | |
| Self-care | 0.4 ± 0.4 | 18.4 ± 4.9 | *** | |
| Interpersonal relationships | 3.3 ± 1.7 | 33.5 ± 5.0 | **** | |
| Life activities | 7.4 ± 3.5 | 62.5 ± 6.2 | **** | |
| Participation in work/school | 7.4 ± 3.7 | 12.6 ± 5.1 | 0.4555 | |
| Participation in society | 4.4 ± 1.9 | 52.2 ± 5.1 | **** | |
| Illness demographic (SF-36) | Pain (%) | 90.7 ± 3.2 | 37.5 ± 5.1 | **** |
| Physical functioning (%) | 92.7 ± 4.7 | 47.6 ± 6.4 | **** | |
| Role physical (%) | 93.8 ± 2.7 | 25.0 ± 5.5 | **** | |
| General health (%) | 80.6 ± 2.7 | 27.2 ± 3.3 | **** | |
| Social functioning (%) | 89.7 ± 6.2 | 27.2 ± 4.7 | **** | |
| Role emotional (%) | 95.1 ± 2.4 | 63.7 ± 6.9 | *** | |
| Wellbeing | 74.8 ± 3.5 | 43.9 ± 2.8 | **** | |
| Full blood count | White blood cells | 6.2 ± 0.5 | 5.7 ± 0.3 | 0.5454 |
| Lymphocytes | 1.70 ± 0.15 | 1.85 ± 0.09 | 0.1243 | |
| Neutrophils | 3.77 ± 0.39 | 3.26 ± 0.27 | 0.3524 | |
| Monocytes | 0.49 ± 0.05 | 0.42 ± 0.02 | 0.2778 | |
| Eosinophils | 0.13 ± 0.02 | 0.14 ± 0.02 | 0.8048 | |
| Basophils | 0.04 ± 0.00 | 0.04 ± 0.00 | 0.3378 | |
| Platelets | 265 ± 19 | 242 ± 9 | 0.2085 | |
| Red blood cells | 4.51 ± 0.09 | 4.57 ± 0.12 | 0.6645 | |
| Hematocrit | 0.40 ± 0.01 | 0.41 ± 0.01 | 0.3416 | |
| Hemoglobin | 131 ± 3 | 137 ± 3 | 0.2628 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Du Preez, S.; Eaton-Fitch, N.; Cabanas, H.; Staines, D.; Marshall-Gradisnik, S. Characterization of IL-2 Stimulation and TRPM7 Pharmacomodulation in NK Cell Cytotoxicity and Channel Co-Localization with PIP2 in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Patients. Int. J. Environ. Res. Public Health 2021, 18, 11879. https://doi.org/10.3390/ijerph182211879
Du Preez S, Eaton-Fitch N, Cabanas H, Staines D, Marshall-Gradisnik S. Characterization of IL-2 Stimulation and TRPM7 Pharmacomodulation in NK Cell Cytotoxicity and Channel Co-Localization with PIP2 in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Patients. International Journal of Environmental Research and Public Health. 2021; 18(22):11879. https://doi.org/10.3390/ijerph182211879
Chicago/Turabian StyleDu Preez, Stanley, Natalie Eaton-Fitch, Helene Cabanas, Donald Staines, and Sonya Marshall-Gradisnik. 2021. "Characterization of IL-2 Stimulation and TRPM7 Pharmacomodulation in NK Cell Cytotoxicity and Channel Co-Localization with PIP2 in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Patients" International Journal of Environmental Research and Public Health 18, no. 22: 11879. https://doi.org/10.3390/ijerph182211879
APA StyleDu Preez, S., Eaton-Fitch, N., Cabanas, H., Staines, D., & Marshall-Gradisnik, S. (2021). Characterization of IL-2 Stimulation and TRPM7 Pharmacomodulation in NK Cell Cytotoxicity and Channel Co-Localization with PIP2 in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Patients. International Journal of Environmental Research and Public Health, 18(22), 11879. https://doi.org/10.3390/ijerph182211879







