Willingness to Contribute to Bio-Larviciding in the Fight against Malaria: A Contingent Valuation Study among Rice Farmers in Rwanda
Abstract
1. Introduction
1.1. Purpose of the Study
1.2. Linking Malaria and Rice Cultivation
1.3. Potential of Biological Larviciding
1.4. Willingness to Contribute to Larviciding
2. Materials and Methods
2.1. Study Setting
2.2. Sample
2.3. Data Collection
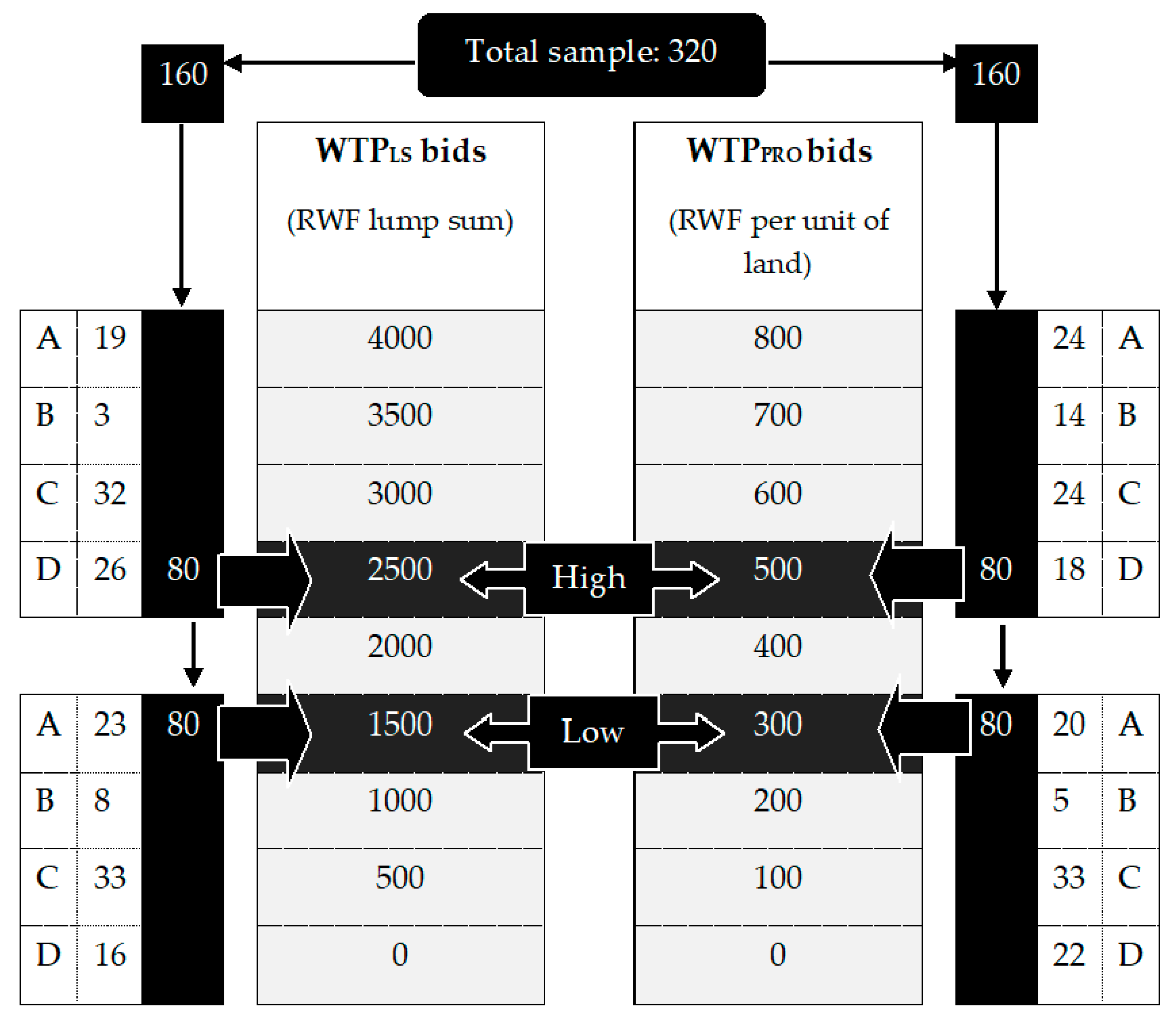
2.4. Bidding Game Procedure
2.5. Data Analysis
3. Results
3.1. Descriptive Statistics
3.1.1. Demographics
3.1.2. Livelihood of Farmer Households
3.1.3. Acceptability of Bti
3.1.4. Willingness-to-Pay (WTP) for Bti
3.2. Factors Associated with the Willingness to Contribute
3.2.1. Individual and Household Factors
3.2.2. The Cooperative Factor
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- African Leaders Malaria Alliance; Asia Pacific Leaders Malaria Alliance; Malaria No More UK; RBM Partnership to End Malaria; World Health Organization. The Commonwealth Malaria Report 2021 1. 2021. Available online: https://malarianomore.org.uk/sites/default/files/Commonwealth%20Malaria%20Report%202021.pdf (accessed on 15 September 2021).
- Karema, C.; Wen, S.; Sidibe, A.; Smith, J.L.; Gosling, R.; Hakizimana, E.; Tanner, M.; Noor, A.M.; Tatarsky, A. History of malaria control in Rwanda: Implications for future elimination in Rwanda and other malaria-endemic countries. Malar. J. 2020, 19, 1–12. [Google Scholar] [CrossRef]
- World Health Organization. Global Vector Control Response 2017–2030; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Mapua, S.A.; Finda, M.F.; Nambunga, I.H.; Msugupakulya, B.J.; Ukio, K.; Chaki, P.P.; Tripet, F.; Kelly, A.H.; Christofides, N.; Lezaun, J.; et al. Addressing key gaps in implementation of mosquito larviciding to accelerate malaria vector control in southern Tanzania: Results of a stakeholder engagement process in local district councils. Malar. J. 2021, 20, 1–14. [Google Scholar] [CrossRef]
- Gowelo, S.; McCann, R.; Koenraadt, C.J.M.; Takken, W.; Berg, H.V.D.; Manda-Taylor, L. Community factors affecting participation in larval source management for malaria control in Chikwawa District, Southern Malawi. Malar. J. 2020, 19, 195. [Google Scholar] [CrossRef] [PubMed]
- Onwujekwe, O.; Malik, E.-F.M.; Mustafa, S.H.; Mnzava, A. Socio-economic inequity in demand for insecticide-treated nets, in-door residual house spraying, larviciding and fogging in Sudan. Malar. J. 2005, 4, 62. [Google Scholar] [CrossRef] [PubMed]
- Mboera, L.E.G.; Kramer, R.A.; Miranda, M.L.; Kilima, S.P.; Shayo, E.H.; Lesser, A. Community Knowledge and Acceptance of Larviciding for Malaria Control in a Rural District of East-Central Tanzania. Int. J. Environ. Res. Public Health 2014, 11, 5137–5154. [Google Scholar] [CrossRef] [PubMed]
- Diiro, G.; Kassie, M.; Muriithi, B.; Gathogo, N.; Kidoido, M.; Marubu, R.; Ochola, J.B.; Mutero, C. Are Individuals Willing to Pay for Community-Based Eco-Friendly Malaria Vector Control Strategies? A Case of Mosquito Larviciding Using Plant-Based Biopesticides in Kenya. Sustainability 2020, 12, 8552. [Google Scholar] [CrossRef]
- Ijumba, J.N.; Lindsay, S.W. Impact of irrigation on malaria in Africa: Paddies paradox. Med. Veter- Èntomol. 2001, 15, 1–11. [Google Scholar] [CrossRef]
- Chan, J.K.; Tusting, L.S.; Bottomley, C.; Saito, K.; Djouaka, R.; Lines, J. Rice and Malaria in Africa: A Systematic Review and Meta-Analysis. Lancet 2021. preprint. [Google Scholar] [CrossRef]
- President’s Malaria Initiative. Rwanda Malaria Operational Plan FY 2016; CreateSpace: Scotts Valley, CA, USA, 2016. [Google Scholar]
- Rulisa, S.; Kateera, F.; Bizimana, J.P.; Agaba, S.; Dukuzumuremyi, J.; Baas, L.; Harelimana, J.D.D.; Mens, P.F.; Boer, K.R.; De Vries, P.J. Malaria Prevalence, Spatial Clustering and Risk Factors in a Low Endemic Area of Eastern Rwanda: A Cross Sectional Study. PLoS ONE 2013, 8, e69443. [Google Scholar] [CrossRef]
- Kateera, F.; Mens, P.F.; Hakizimana, E.; Ingabire, C.M.; Muragijemariya, L.; Karinda, P.; Grobusch, M.P.; Mutesa, L.; Van Vugt, M. Malaria parasite carriage and risk determinants in a rural population: A malariometric survey in Rwanda. Malar. J. 2015, 14, 1–11. [Google Scholar] [CrossRef][Green Version]
- Tusting, L.S.; Thwing, J.; Sinclair, D.; Fillinger, U.; Gimnig, J.; E Bonner, K.; Bottomley, C.; Lindsay, S.W. Mosquito larval source management for controlling malaria. Cochrane Database Syst. Rev. 2013, 8, CD008923. [Google Scholar] [CrossRef] [PubMed]
- Klinkenberg, E.; Takken, W.; Huibers, F.; Touré, Y. The phenology of malaria mosquitoes in irrigated rice fields in Mali. Acta Trop. 2002, 85, 71–82. [Google Scholar] [CrossRef]
- Bukhari, T.; Takken, W.; Githeko, A.K.; Koenraadt, C. Efficacy of Aquatain, a Monomolecular Film, for the Control of Malaria Vectors in Rice Paddies. PLoS ONE 2011, 6, e21713. [Google Scholar] [CrossRef] [PubMed]
- Raghavendra, K.; Barik, T.K.; Reddy, B.P.N.; Sharma, P.; Dash, A.P. Malaria vector control: From past to future. Parasitol. Res. 2011, 108, 757–779. [Google Scholar] [CrossRef]
- Walker, K. A Review of Control Methods for African Malaria Vectors; Activity Report 108; Environmental Health Project, Bureau for Global Health, US Agency for International Development: Washington, DC, USA, 2002. [Google Scholar]
- Chandra, G.; Bhattacharjee, I.; Chatterjee, S.N.; Ghosh, A. Mosquito control by larvivorous fish. Indian J. Med. Res. 2008, 127, 13–27. [Google Scholar]
- Choi, L.; Majambere, S.; Wilson, A.L. Larviciding to prevent malaria transmission. Cochrane Database Syst. Rev. 2019, 8, CD012736. [Google Scholar] [CrossRef]
- Land, M.; Miljand, M. Biological Control of Mosquitoes Using Bacillus thuringiensis israelensis: A Pilot Study of Effects on Target Organisms, Non-Target Organisms and Humans; Mistra EviEM: Stockholm, Sweden, 2014. [Google Scholar]
- Dambach, P.; Louis, V.R.; Kaiser, A.; Ouedraogo, S.; Sié, A.; Sauerborn, R.; Becker, N. Efficacy of Bacillus thuringiensis var. israelensis against malaria mosquitoes in northwestern Burkina Faso. Parasites Vectors 2014, 7, 1–8. [Google Scholar] [CrossRef]
- Teng, H.-J.; Lu, L.-C.; Wu, Y.-L.; Fang, J.-G. Evaluation of various control agents against mosquito larvae in rice paddies in Taiwan. J. Vector Ecol. 2005, 30, 126–132. [Google Scholar]
- Ramírez-Lepe, M.; Ramírez-Suero, M. Biological Control of Mosquito Larvae by Bacillus Thuringiensis Subsp. Israelensis. In Insecticides–Pest Engineering; Perveen, F., Ed.; IntechOpen: London, UK, 2012. [Google Scholar] [CrossRef]
- Glare, T.R.; Callaghan, M.O. Environmental and Health Impacts of Bacillus Thuringiensis Israelensis; Report for the Ministry of Health: Wellington, New Zealand, 1998.
- Fillinger, U.; Lindsay, S.W. Suppression of exposure to malaria vectors by an order of magnitude using microbial larvicides in rural Kenya. Trop. Med. Int. Health 2006, 11, 1629–1642. [Google Scholar] [CrossRef]
- Derua, Y.A.; Kweka, E.J.; Kisinza, W.N.; Githeko, A.K.; Mosha, F.W. Bacterial larvicides used for malaria vector control in sub-Saharan Africa: Review of their effectiveness and operational feasibility. Parasites Vectors 2019, 12, 1–18. [Google Scholar] [CrossRef]
- Mutero, C.M.; Okoyo, C.; Girma, M.; Mwangangi, J.; Kibe, L.; Ng’Ang’A, P.; Kussa, D.; Diiro, G.; Affognon, H.; Mbogo, C.M. Evaluating the impact of larviciding with Bti and community education and mobilization as supplementary integrated vector management interventions for malaria control in Kenya and Ethiopia. Malar. J. 2020, 19, 1–17. [Google Scholar] [CrossRef]
- Worrall, E.; Fillinger, U. Large-scale use of mosquito larval source management for malaria control in Africa: A cost analysis. Malar. J. 2011, 10, 338. [Google Scholar] [CrossRef] [PubMed]
- Fillinger, U.; Kannady, K.; William, G.; Vanek, M.J.; Dongus, S.; Nyika, D.; Geissbühler, Y.; Chaki, P.P.; Govella, N.J.; Mathenge, E.M.; et al. A tool box for operational mosquito larval control: Preliminary results and early lessons from the Urban Malaria Control Programme in Dar es Salaam, Tanzania. Malar. J. 2008, 7, 20. [Google Scholar] [CrossRef]
- Trapero-Bertran, M.; Mistry, H.; Shen, J.; Fox-Rushby, J. A systematic review and meta-analysis of willingness-to-pay values: The case of malaria control interventions. Health Econ. 2012, 22, 428–450. [Google Scholar] [CrossRef] [PubMed]
- Matindo, A.Y.; Kapalata, S.N.; Katalambula, L.K.; Meshi, E.B.; Munisi, D.Z. Biolarviciding for malaria vector control: Acceptance and associated factors in southern Tanzania. Curr. Res. Parasitol. Vector-Borne Dis. 2021, 1, 100038. [Google Scholar] [CrossRef]
- Berg, H.V.D.; Knols, B.G. The Farmer Field School: A method for enhancing the role of rural communities in malaria control ? Malar. J. 2006, 5, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Mazigo, H.D.; Massawe, I.S.; Rumisha, S.F.; Kweka, E.J.; Mboera, L.E.G. Rice farmers’ perceptions and acceptability in the use of a combination of biolarvicide (Bacillus thuringiensis var. israeliensis) and fertilizers application for malaria control and increase rice productivity in a rural district of central Tanzania. Malar. J. 2019, 18, 71. [Google Scholar] [CrossRef]
- Klose, T. The contingent valuation method in health care. Health Policy 1999, 47, 97–123. [Google Scholar] [CrossRef]
- Ives, B.; Eastman, C. Bidding Games for Valuation of Aesthetic Environmental public goods. J. Environ. Econ. Manag. 1974, 1, 132–149. [Google Scholar]
- Frew, E.J.; Wolstenholme, J.L.; Whynes, D.K. Comparing willingness-to-pay: Bidding game format versus open-ended and payment scale formats. Health Policy 2004, 68, 289–298. [Google Scholar] [CrossRef]
- Mitchell, R.C.; Carson, R.T. Using Surveys to Value Public Goods: The Contingent Valuation Method; RFF Press: New York, NY, USA, 2013. [Google Scholar] [CrossRef]
- O’Brien, B.; Viramontes, J.L. Willingness to Pay: A Valid and Reliable Measure of Health State Preference? Med. Decis. Mak. 1997, 14, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Ingabire, C.M.; Hakizimana, E.; Rulisa, A.; Kateera, F.; Borne, B.V.D.; Muvunyi, C.M.; Mutesa, L.; Van Vugt, M.; Koenraadt, C.J.M.; Takken, W.; et al. Community-based biological control of malaria mosquitoes using Bacillus thuringiensis var. israelensis (Bti) in Rwanda: Community awareness, acceptance and participation. Malar. J. 2017, 16, 1–13. [Google Scholar] [CrossRef] [PubMed]
| Frequency | Percentage (%) | Cumulative (%) | |
|---|---|---|---|
| Location | |||
| Bihari | 21 | 6.6 | |
| Gatanga | 22 | 6.9 | |
| Gikundamvura | 47 | 14.7 | |
| Kindama | 79 | 24.7 | |
| Ruhuha | 25 | 7.8 | |
| Outside Ruhuha sector | 126 | 39.4 | |
| Gender | |||
| Female | 145 | 45.3 | |
| Marital status | |||
| Never Married | 4 | 1.3 | |
| Married | 236 | 73.8 | |
| Living together | 35 | 10.9 | |
| Separated/Divorced | 10 | 3.1 | |
| Widowed | 35 | 10.9 | |
| Age group (in years) | |||
| <35 | 74 | 23.1 | 23.1 |
| 35–44 | 104 | 32.5 | 55.6 |
| 45–54 | 74 | 23.1 | 78.8 |
| ≥55 | 68 | 21.3 | 100.0 |
| Education level | |||
| None | 78 | 24.4 | 24.4 |
| Primary | 222 | 69.4 | 93.8 |
| Post-primary/vocational | 6 | 1.9 | 95.7 |
| Secondary school or higher | 14 | 4.4 | 100.0 |
| Variable | Mean | SD | Min–Max |
|---|---|---|---|
| Number of HH members | 6 | 2.0 | 1–16 |
| Number of children below 18 | 3 | 1.8 | 0–10 |
| Number of children below five | 1 | 0.8 | 0–4 |
| Number of years in rice farming | 11.9 | 5.7 | 0–36 |
| Number of years in cooperative | 7.8 | 2.9 | 0–23 |
| HH members in rice farming | 2 | 1.0 | 1–9 |
| HH members of rice cooperative | 1 | 0.5 | 1–4 |
| Rice (%) kept by the HH | 20 | 0 | 20 |
| Share (%) of rice eaten (from part kept by HH) | 89.2 | 27.5 | 0–100 |
| Share (%) of rice sold (from part kept by HH) | 5.4 | 19.7 | 0–100 |
| Size of land (in are = 100 m2) | 5.5 | 3.7 | 1–30 |
| (1) log(WTPLS) | (2) log(WTPPRO) | (3) log(WTPLS / [WTPPRO*Land Size]) | |
|---|---|---|---|
| Fixed effects variables | Coeff. (st.dev) ^ | Coeff. (st.dev) ^ | Coeff. (st.dev) ^ |
| Progressive treatment presented first (ref.= lump sum first) | 0.000 (0.041) | 0.015 (0.039) | −0.008 (0.027) |
| High starting bid (ref.= low starting bid) | 0.087 (0.041) ** | 0.046 (0.039) | 0.030 (0.027) |
| Male respondent | 0.005 (0.043) | 0.037 (0.041) | −0.046 (0.028) |
| Age respondent | −0.002 (0.002) | −0.001 (0.002) | −0.001 (0.001) |
| Number of HH members | 0.026 (0.018) | 0.035 (0.017) ** | −0.009 (0.012) |
| Number of children under 5 | 0.000 (0.032) | −0.016 (0.030) | 0.011 (0.021) |
| Number of children under 18 | −0.030 (0.021) | −0.022 (0.020) | −0.011 (0.013) |
| Respondent did not complete primary education (dummy) | 0.081 (0.051) | 0.083 (0.049) * | 0.025 (0.033) |
| Rice income per capita below sample average (dummy) | −0.097 (0.055) * | −0.085 (0.052) | −0.019 (0.036) |
| Share of rice income out of total HH income (%) | −0.001 (0.001) | −0.003 (0.001) *** | 0.003 (0.001) *** |
| Rising trend of income from rice over past 3 seasons | 0.086 (0.039) ** | 0.073 (0.057) | 0.025 (0.026) |
| Rice farming considered unprofitable (dummy) | −0.119 (0.100) | −0.143 (0.095) | 0.014 (0.065) |
| Rice is primary income-generating crop (dummy) | 0.037 (0.059) | 0.073 (0.057) | −0.063 (0.039) |
| Number of years in rice farming | 0.003 (0.005) | −0.001 (0.005) | 0.002 (0.003) |
| Years of membership in rice cooperative | −0.013 (0.008) | −0.011 (0.008) | −0.001 (0.006) |
| Land size (in are) | 0.084 (0.028) *** | 0.033 (0.027) | 0.098 (0.018) *** |
| Relative land size (# of st.dev. from mean land size in own cooperative) | −0.248 (0.094) *** | −0.167 (0.090) * | −0.423 (0.062) *** |
| Log of share capital invested in cooperative | −0.081 (0.085) | −0.062 (0.081) | 0.004 (0.056) |
| Log of total contributions paid to cooperative (for running costs, fertilizer, and insecticide), past year | −0.018 (0.094) | −0.043 (0.090) | −0.031 (0.061) |
| Any malaria case in HH in past year (dummy) | 0.079 (0.045) * | 0.050 (0.043) | 0.048 (0.030) |
| Loss of any family member due to malaria (dummy) | −0.014 (0.101) | 0.092 (0.096) | −0.096 (0.066) |
| Experience of (very) frequent mosquito bites in rice field (dummy) | −0.094 (0.097) | −0.070 (0.092) | −0.002 (0.063) |
| Rice field considered important breeding site for mosquito (dummy) | −0.153 (0.101) | −0.054 (0.097) | −0.122 (0.066) * |
| Confident that Bti will reduce mosquito population (dummy) | −0.104 (0.127) | −0.162 (0.121) | 0.036 (0.083) |
| Trust in safety of Bti for animals (dummy) | 0.086 (0.028) *** | 0.097 (0.027) *** | −0.002 (0.018) |
| Random effect variable | |||
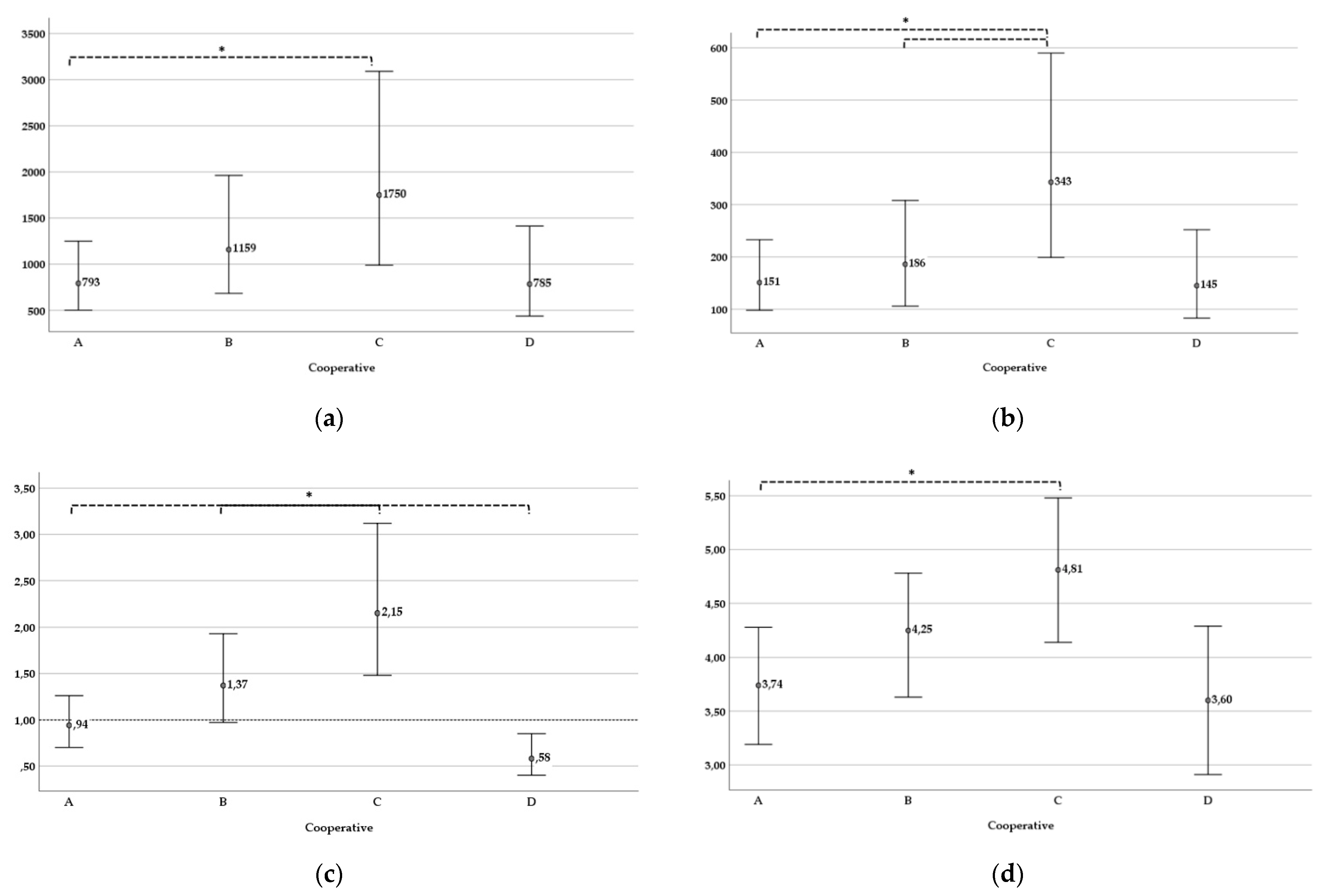
| Membership in cooperative A, B, C or D | F = 4.44 *** | F = 5.44 *** | F = 10.128 *** |
| Estimated marginal means [confidence interval] | |||
| Cooperative A (n = 85) | 2.899 [2.701, 3.097] | 2.179 [1.990, 2.368] | −0.028 [−0.157, 0.101] |
| Cooperative B (n = 30) | 3.064 [2.835, 3.293] | 2.270 [2.025, 2.488] | 0.136 [−0.013, 0.286] |
| Cooperative C (n = 122) | 3.243 [2.995, 3.490] | 2.535 [2.299, 2.771] | 0.332 [0.171, 0.494] |
| Cooperative D (n = 81) | 2.895 [2.641, 3.150] | 2.160 [1.917, 2.402] | −0.236 [−0.402, −0.070] |
| Number of observations | 318 | 318 | 318 |
| Levene’s test of equal variances | F= 0.989 (p > 0.05) | F= 1.061 (p > 0.05) | F= 1.135 (p > 0.05) |
| Breusch-Pagan test for heteroskedasticity | Χ2 = 0.263 (p > 0.05) | Χ2 = 0.869 (p > 0.05) | Χ2 = 1.301 (p > 0.05) |
| Normality tests | see Supplementary Materials | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rulisa, A.; van Kempen, L.; Mutesa, L.; Hakizimana, E.; Ingabire, C.M.; Kateera, F.; Koenraadt, C.J.M.; van Vugt, M.; van den Borne, B. Willingness to Contribute to Bio-Larviciding in the Fight against Malaria: A Contingent Valuation Study among Rice Farmers in Rwanda. Int. J. Environ. Res. Public Health 2021, 18, 11575. https://doi.org/10.3390/ijerph182111575
Rulisa A, van Kempen L, Mutesa L, Hakizimana E, Ingabire CM, Kateera F, Koenraadt CJM, van Vugt M, van den Borne B. Willingness to Contribute to Bio-Larviciding in the Fight against Malaria: A Contingent Valuation Study among Rice Farmers in Rwanda. International Journal of Environmental Research and Public Health. 2021; 18(21):11575. https://doi.org/10.3390/ijerph182111575
Chicago/Turabian StyleRulisa, Alexis, Luuk van Kempen, Leon Mutesa, Emmanuel Hakizimana, Chantal M. Ingabire, Fredrick Kateera, Constantianus J. M. Koenraadt, Michèle van Vugt, and Bart van den Borne. 2021. "Willingness to Contribute to Bio-Larviciding in the Fight against Malaria: A Contingent Valuation Study among Rice Farmers in Rwanda" International Journal of Environmental Research and Public Health 18, no. 21: 11575. https://doi.org/10.3390/ijerph182111575
APA StyleRulisa, A., van Kempen, L., Mutesa, L., Hakizimana, E., Ingabire, C. M., Kateera, F., Koenraadt, C. J. M., van Vugt, M., & van den Borne, B. (2021). Willingness to Contribute to Bio-Larviciding in the Fight against Malaria: A Contingent Valuation Study among Rice Farmers in Rwanda. International Journal of Environmental Research and Public Health, 18(21), 11575. https://doi.org/10.3390/ijerph182111575







