The Quality of Life of Children with Myelomeningocele: A Cross-Sectional Preliminary Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Data Analysis
- Qualitative variables (e.g., child’s sex) were cross-tabulated as numbers (n) and percentages (%), and associations between pairs of variables were evaluated using Pearson’s chi-squared test;
- For quantitative variables (e.g., age), means (M), standard deviations (SD), and the median (Me), lower quartile (Q1), upper quartile (Q3), and extreme (Min and Max) values were calculated;
- For all quantitative variables, distribution normality was verified using the Shapiro–Wilk test;
- The significance of differences between mean values of quantitative variables with a normal distribution and homogeneous variances in two independent groups (e.g., comorbidity: present or absent) was verified using Student’s t-test. For larger numbers of groups (e.g., areas of functioning), single-factor analysis of variance (ANOVA) was used;
- The strength and direction of linear correlations between two continuous variables was determined by regression analysis. Pearson’s linear correlation coefficient (r) was calculated and, if significant (p < 0.05; r ≠ 0), i.e., if the regression coefficient significantly differed from 0, regression equation coefficients (bi) were estimated. Regression coefficients were estimated using the least square method;
- Calculations were performed using Statistica v. 12.5 software and Excel spreadsheets.
3. Results
3.1. Baseline Characteristics
3.2. Analysis of QoL Measured Using the PedsQLTM 4.0 Generic Core Questionnaire
3.3. The Impact of Demographic Characteristics on QoL
3.4. QoL in Children with MMC
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liptak, G.S.; Samra, A. Optimizing health care for children with spina bifida. Dev. Disabil. Res. Rev. 2010, 16, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Schroder, H.K.; Nunes, J.C.; Madiera, L.; Moritz, J.L.; Wulz, R.; Linhares, M.N. Postsurgical infection after myelomeningocoele repair: A multivariate analysis of 60 consecutive cases. Clin. Neurol. Neurosurg. 2012, 114, 981–985. [Google Scholar] [CrossRef]
- Sawn, K.J.; Thompson, N.P. The experience of finding an effective bowel management program for children with spina bifida: The parent’s perspective. J. Ped. Nurs. 2009, 24, 280–291. [Google Scholar] [CrossRef] [PubMed]
- Alatise, O.I.; Adeolu, A.A.; Komolase, E.O.; Adejuyigbe, O.; Sowande, O.A. Pattern and factors affecting management outcome of spina bifida cystica in Iie-Ife Nigeria. Pediatr. Neurosurg. 2006, 42, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Halaby, E.; Ismail, M.T. Delayed hydrocephalus after re-pairing un-rupture myelomeningocele. Egypt. J. Neurosurg. 2016, 31, 167–170. [Google Scholar]
- Kase, B.A.; Northrup, H.; Morrison, A.C.; Davidson, C.M.; Goiffon, A.M.; Fletcher, J.M.; Ostermaier, K.K.; Tyerman, G.H.; Au, K.S. Association of copper-zinc superoxide dismutase (SOD1) and manganese superoxide dismutase (SOD2) genes with nonsyndromic myelomeningocele. Birth Defects Research. Part, A. Clin. Mol. Teratol. 2012, 94, 762–769. [Google Scholar] [CrossRef] [Green Version]
- Dlouhy, B.J.; Menezes, A.H. Hydrocephalus in Chiari Malformations and Other Craniovertebral Junction Abnormalities. In Pediatric Hydrocephalus; Cinalli, G., Ozek, M., Sainte-Rose, C., Eds.; Springer: Cham, Switzerland, 2018. [Google Scholar] [CrossRef]
- McLone, D.G.; Dias, M.S. The Chiari II malformation: Cause and impact. Childs Nerv. Syst. 2003, 19, 540–550. [Google Scholar] [CrossRef]
- Liptak, G.S.; Dosa, N.P. Myelomeningocele. Pediatr. Rev. 2010, 31, 443–450. [Google Scholar] [CrossRef]
- Oncel, M.Y.; Ozdemir, R.; Kahilogulları, G.; Yurttutan, S.; Erdeve, O.; Dilmen, U. The effect of surgery time on prognosis in newborns with meningomyelocele. J. Korean Neurosurg. Soc. 2012, 51, 359–362. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Publications of the World Health Organization; WHO: Geneva, Switzerland, 1958; pp. 101–102. [Google Scholar]
- Waters, E.; Davis, E.; Ronen, G.M.; Rosenbaum, P.; Livingston, M.; Saigal, S. Quality of life instruments for children and adolescents with neurodisabilities: How to choose the appropriate instrument. Dev. Med. Child. Neurol. 2009, 51, 660–669. [Google Scholar] [CrossRef]
- Varni, J.W.; Burwinkle, T.M.; Seid, M. The PedsQL 4.0 as a pediatric population health measure: Feasibility, reliability, and validity. Ambul. Pediatrics Off. J. Ambul. Pediatric Assoc. 2003, 3, 329–341. [Google Scholar] [CrossRef]
- Varni, J.W.; Limbers, C.A.; Burwinkle, T.M. Impaired health-related quality of life in children and adolescents with chronic conditions: A comparative analysis of 10 disease clusters and 33 disease categories/severities utilizing the PedsQLTM 4.0 Generic Core Scales. Health Qual. Life Outcomes 2012, 5, 43. [Google Scholar] [CrossRef] [Green Version]
- Varni, J.W.; Limbers, C.A.; Burwinkle, T.M. How young can children reliably and validly self-report their health-related quality of life? An analysis of 8591 children across age subgroups with the PedsQL 4.0 Generic Core Scales. Health Qual. Life Outcomes 2007, 5, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Varni, J.W.; Limbers, C.A.; Burwinkle, T.M. Parent proxy-report of their children’s health-related quality of life: An analysis of 13,878 parents’ reliability and validity across age subgroups using the PedsQL 4.0 Generic Core Scales. Health Qual. Life Outcomes 2007, 5, 2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Varni, J.W.; Sherman, S.A.; Burwinkle, T.M. The PedsQLTM Family Impact Module: Preliminary reliability and validity. Health Qual. Life Outcomes 2004, 2, 55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oddson, B.E.; Clancy, C.A.; McGrath, P.J. The role of pain in reduced quality of life and depressive symptomology in children with spina bifida. Clin. J. Pain 2006, 22, 784–789. [Google Scholar] [CrossRef]
- Abresch, R.T.; McDonald, D.A.; Widman, L.M.; McGinnis, K.; Hickey, K.J. Impact of spinal cord dysfunction and obesity on the health-related quality of life of children and adolescents. J. Spinal Cord Med. 2007, 30, S112–S118. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, A.; Gorzkowski, M.; Altiok, H.; Hassani, S.; Ahn, K.W. Activity level, functional health, and quality of life of children with myelomeningocele as perceived by parents. Clin. Orthop. Relat. Res. 2011, 469, 1230–1235. [Google Scholar] [CrossRef] [Green Version]
- Johansen, H.; Dammann, B.; Andresen, I.L.; Fagerland, M.W. Health-related quality of life for children with rare diagnoses, their parents’ satisfaction with life and the association between the two. Health Qual. Life Outcomes 2013, 11, 152. [Google Scholar] [CrossRef] [Green Version]
- Freeman, K.A.; Smith, K.; Adams, E.; Mizokawa, S.; Neville-Jan, A. Is continence status associated with quality of life in young children with spina bifida? J. Pediatric Rehabil. Med. 2013, 6, 215–223. [Google Scholar]
- Ramachandra, P.; Palazzi, K.L.; Skalsky, A.J.; Marietti, S.; Chiang, G. Shunted hydrocephalus has a significant impact on quality of life in children with spina bifida. PM&R 2013, 5, 825–831. [Google Scholar]
- Murray, C.B.; Holmbeck, G.N.; Ros, A.M.; Flores, D.M.; Mir, S.A.; Varni, J.W. A longitudinal examination of health-related quality of life in children and adolescents with spina bifida. J. Pediatric Psychol. 2015, 40, 419–430. [Google Scholar] [CrossRef] [Green Version]
- Buffart, L.M.; van den Berg-Emons, R.J.; van Meeteren, J.; Stam, H.J.; Roebroeck, M.E. Lifestyle, participation, and health-related quality of life in adolescents and young adults with myelomeningocele. Dev. Med. Child Neurol. 2009, 51, 886–894. [Google Scholar] [CrossRef] [PubMed]
- Sawin, K.J.; Bellin, M.H. Quality of life in individuals with spina bifida: A research update. Dev. Disabil. Res. Rev. 2010, 16, 47–59. [Google Scholar] [CrossRef] [PubMed]
- MacNeily, A.E.; Jafari, S.; Scott, H.; Dalgetty, A.; Afshar, K. Health related quality of life in patients with spina bifida: A prospective assessment before and after lower urinary tract reconstruction. J. Urology 2009, 182, 1984–1991. [Google Scholar] [CrossRef]
- Olsson, I.; Dahl, M.; Mattsson, S.; Wendelius, M.; Astrom, E.; Westbom, L. Medical problems in adolescents with myelomeningocele (MMC): An inventory of the Swedish MMC. Acta Paediatr. 2007, 96, 446–449. [Google Scholar] [CrossRef]
- Hampton, L.E.; Fletcher, J.M.; Cirino, P.T.; Blaser, S.; Kramer, L.A.; Drake, J. Hydrocephalus status in spina bifida: An evaluation of variations in neuropsychological outcomes. Clinical article. J. Neurosurg. Pediatr. 2011, 8, 289–298. [Google Scholar] [CrossRef]
- Karmur, B.S.; Kulkarni, A.V. Medical and socioeconomic predictors of quality of life in myelomeningocele patients with shunted hydrocephalus. Childs Nerv. Syst. 2018, 34, 741–747. [Google Scholar] [CrossRef]
- Sawin, K.J.; Brei, T.J.; Buran, C.F.; Fastenau, P.S. Factors associated with quality of life in adolescents with spina bifida. J. Holist Nurs. 2001, 20, 279–304. [Google Scholar] [CrossRef]
- Tew, B. The effects of spina bifida and hydrocephalus upon learning and behaviour. In Current concepts in Spina Bifida & Hydrocephalus. Clinics in Developmental Medicine; Bannister, C.M., Tew, B., Eds.; Mac Keith Press: London, UK, 1991; pp. 158–179. [Google Scholar]
- Kulkarni, A.V.; Rabin, D.; Drake, J.M. An instrument to measure the health status in children with hydrocephalus: The Hydrocephalus Outcome Questionnaire. J. Neurosurg. 2004, 101, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Malakounides, G.; Lee, F.; Murphy, F.; Boddy, S.A. Single centre experience: Long term outcomes in spina bifida patients. J. Pediatr. Urol. 2013, 9, 585–589. [Google Scholar] [CrossRef]
- Clayton, D.B.; Brock, J.W. The urologist’s role in the management of spina bifida: A continuum of care. Urology 2010, 76, 32–38. [Google Scholar] [CrossRef]
- Wallander, J.L.; Varni, J.W. Effects of pediatric chronic physical disorders on child and family adjustment. J. Child Psychol. Psychiatry 1998, 39, 29–46. [Google Scholar] [CrossRef]
- Eggenberger, S.K.; Nelms, T.P. Being family: Family experiences when an adult member is hospitalized with a critical illness. J. Clin. Nurs. 2007, 16, 1618–1628. [Google Scholar] [CrossRef] [PubMed]
- Solomon, M.; Pistrung, N.; Barker, C. The benefits of mutual support groups for parents of children with disabilities. Am. J. Commun. Psychol. 2001, 29, 113–132. [Google Scholar] [CrossRef] [PubMed]
- Okurowska-Zawada, B.; Kulak, W.; Otapowicz, D.; Sienkiewicz, D.; Paszko-Patej, G.; Wojtkowski, J. Quality of life in children and adolescents with cerebral palsy and myelomeningocele. Pediatric Neurol. 2011, 45, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Raina, P. The Health and Well-Being of Caregivers of Children With Cerebral Palsy. Pediatrics 2005, 115, e626–e636. [Google Scholar] [CrossRef] [Green Version]
- Uchmanowicz, I.; Jankowska-Polańska, B.; De Walden-Gałuszko, K. Wybrane zagadnienia psychoonkologii. In Jakość Życia W Chorobach Wewnętrznych–Wybrane Kwestionariusze Badawcze; Europejskie Centrum Kształcenia Podyplomowego: Wrocław, Poland, 2013. [Google Scholar]
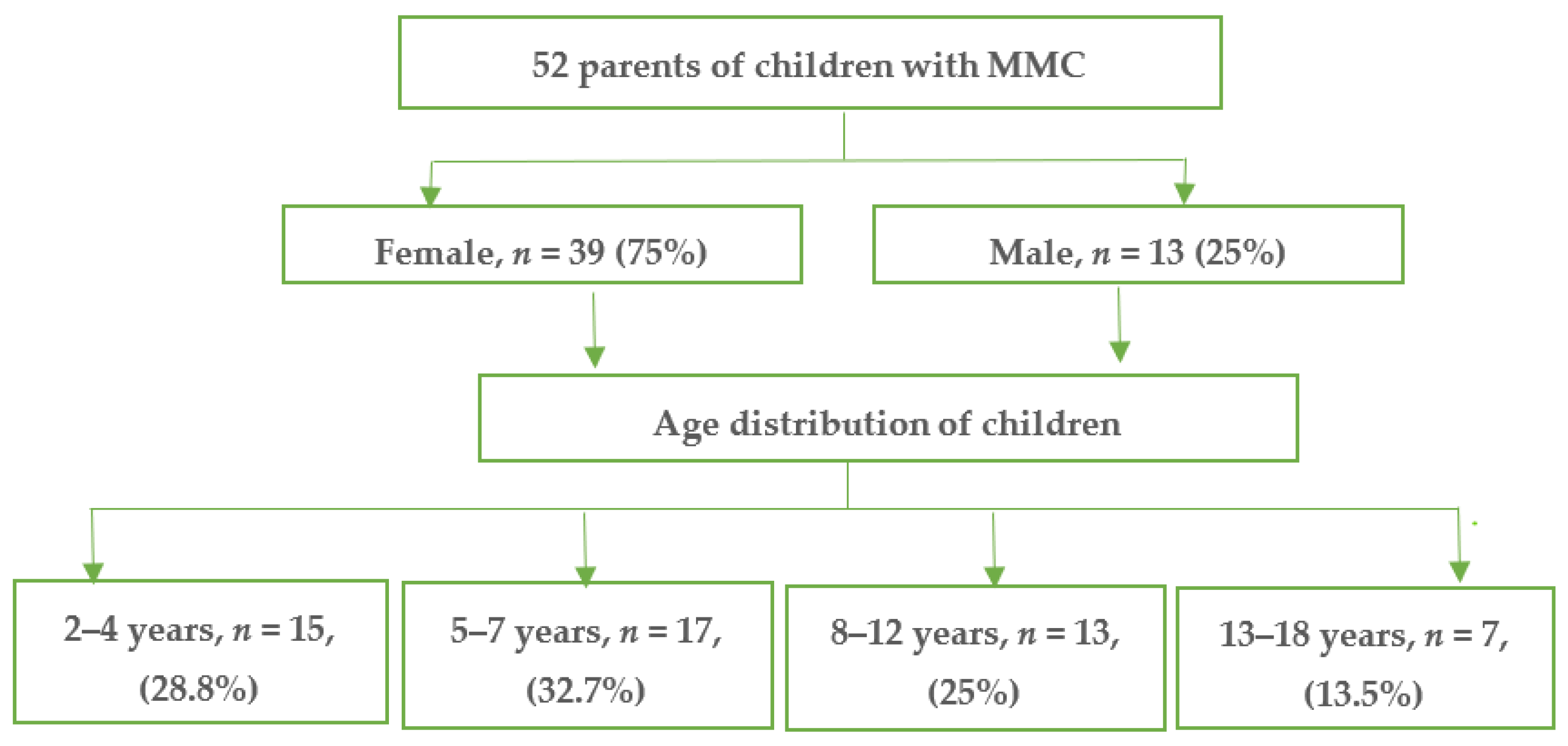
| Characteristic (Variable) | Statistics | |
|---|---|---|
| Parent’s age (years) | ||
| M ± SD | 35.5 ± 7.2 | |
| Me [Q1; Q3] | 35 [30; 40] | |
| Min–Max | 22–58 | |
| Child’s age (years) | ||
| M ± SD | 7.5 ± 10.0 | |
| Me [Q1; Q3] | 7 [4; 10] | |
| Min–Max | 1–17 | |
| Child’s sex (n and %) | ||
| Girl | 41 | 78.8 |
| Boy | 11 | 21.2 |
| Residence (n and %) | ||
| Rural | 25 | 48.1 |
| Urban | 27 | 51.9 |
| Gestational age at birth (n and %) | ||
| ≤37 weeks | 30 | 57.7 |
| >37 weeks | 22 | 42.3 |
| Children diagnosed with associated birth defects (n and %) | 49 | 94.2 |
| Associated comorbidities (n and %) | ||
| Hydrocephalus | 22 | 42.3 |
| Neurogenic bladder | 42 | 80.7 |
| Neurogenic bowel | 38 | 73.1 |
| Superficial and deep sensation loss | 13 | 25.0 |
| Lower extremity paralysis or paresis | 23 | 44.2 |
| Spinal deformity | 21 | 40.4 |
| Foot deformities (n and %) | ||
| Talipes equinovarus | 24 | 46.2 |
| Talipes calcaneus | 2 | 3.8 |
| Hip dysplasia | 9 | 17.3 |
| Other | 4 | 7.7 |
| Has the child’s illness forced the parent to quit their job (n and %) | 32 | 61.5 |
| Which parent has quit their job? (n and %) | ||
| Mother | 28 | 53.8 |
| Father | 4 | 7.7 |
| Questionnaire | Descriptive Statistics | ||
|---|---|---|---|
| M ± SD | Me [Q1; Q3] | Min–Max | |
| Physical functioning (PF) | 41.8 ± 20.1 | 42 [25; 53] | 9–100 |
| Emotional functioning (EF) | 63.9 ± 14.5 | 65 [50; 73] | 35–100 |
| Social functioning (SF) | 58.6 ± 19.9 | 63 [48; 70] | 10–100 |
| School/preschool/nursery (role) functioning (RF) | 72.4 ± 22.9 | 75 [58; 92] | 8–100 |
| Overall functioning score | 56.4 ± 14.7 | 57 [49; 64] | 27–92 |
| Area of Functioning | PF | EF | SF | RF |
|---|---|---|---|---|
| M = 41.6 | M = 63.9 | M = 58.6 | M = 72.4 | |
| PF | × | p < 0.001 | p < 0.001 | p < 0.001 |
| EF | p < 0.001 | × | p = 0.489 | p = 0.120 |
| SF | p < 0.001 | p = 0.489 | × | p = 0.002 |
| RF | p < 0.001 | p = 0.120 | p = 0.002 | × |
| Child’s Area of Functioning | Total Score | |||
|---|---|---|---|---|
| PF | EF | SF | RF | |
| r = −0.141 | r = −0.445 | r = −0.019 | r = −0.220 | r = −0.211 |
| p = 0.320 | p = 0.001 | p = 0.896 | p = 0.117 | p = 0.133 |
| Child’s Area of Functioning | Hydrocephalus | p-Value | |
|---|---|---|---|
| Present n = 49 | None n = 3 | ||
| Physical functioning (PF) | 32.2 ± 17.1 | 48.9 ± 19.4 | 0.002 |
| Emotional functioning (EF) | 60.7 ± 15.2 | 66.3 ± 13.7 | 0.167 |
| Social functioning (SF) | 50.0 ± 22.0 | 64.8 ± 15.8 | 0.007 |
| School/preschool/nursery (role) functioning (RF) | 64.8 ± 27.2 | 78.1 ± 17.6 | 0.037 |
| Total score | 49.2 ± 13.8 | 61.6 ± 13.2 | 0.002 |
| Child’s Area of Functioning | Neurogenic Bowel | p-Value | ||
|---|---|---|---|---|
| None n = 10 | Constipation n = 10 | Constant Passing of Stool n = 32 | ||
| Physical functioning (PF) | 53.1 ± 17.8 | 38.1 ± 20.0 | 32.3 ± 14.4 | 0.040 |
| Emotional functioning (EF) | 65.7 ± 19.6 | 62.6 ± 12.6 | 71.7 ± 5.8 | 0.512 |
| Social functioning (SF) | 59.3 ± 19.9 | 58.7 ± 18.7 | 53.3 ± 88.8 | 0.896 |
| School/preschool (role) functioning (RF) | 71.4 ± 23.5 | 71.2 ± 23.0 | 91.7 ± 14.4 | 0.332 |
| Total score | 60.3 ± 17.5 | 54.9 ± 13.6 | 55.5 ± 15.1 | 0.508 |
| Child’s Area of Functioning | Lower Extremity Paralysis or Paresis | p-Value | |
|---|---|---|---|
| Present N = 23 | None N = 29 | ||
| Physical functioning (PF) | 32.3 ± 15.8 | 49.4 ± 20.2 | 0.002 |
| Emotional functioning (EF) | 60.7 ± 13.8 | 66.6 ± 14.7 | 0.147 |
| Social functioning (SF) | 52.8 ± 22.4 | 63.1 ± 16.7 | 0.064 |
| School/preschool/nursery (role) functioning (RF) | 65.9 ± 25.6 | 77.6 ± 19.4 | 0.068 |
| Total score | 50.1 ± 12.7 | 61.3 ± 14.4 | 0.005 |
| Child’s Area of Functioning | Foot Deformities | p-Value | |
|---|---|---|---|
| Present N = 30 | None N = 22 | ||
| Physical functioning (PF) | 36.8 ± 14.4 | 48.7 ± 24.7 | 0.033 |
| Emotional functioning (EF) | 62.2 ± 12.4 | 66.4 ± 16.9 | 0.307 |
| Social functioning (SF) | 58.8 ± 16.2 | 58.2 ± 24.4 | 0.908 |
| School/preschool/nursery (role) functioning (RF) | 70.3 ± 24.0 | 75.4 ± 21.4 | 0.433 |
| Total score | 54.0 ± 10.7 | 59.6 ± 18.6 | 0.178 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rozensztrauch, A.; Iwańska, M.; Bagłaj, M. The Quality of Life of Children with Myelomeningocele: A Cross-Sectional Preliminary Study. Int. J. Environ. Res. Public Health 2021, 18, 10756. https://doi.org/10.3390/ijerph182010756
Rozensztrauch A, Iwańska M, Bagłaj M. The Quality of Life of Children with Myelomeningocele: A Cross-Sectional Preliminary Study. International Journal of Environmental Research and Public Health. 2021; 18(20):10756. https://doi.org/10.3390/ijerph182010756
Chicago/Turabian StyleRozensztrauch, Anna, Magdalena Iwańska, and Maciej Bagłaj. 2021. "The Quality of Life of Children with Myelomeningocele: A Cross-Sectional Preliminary Study" International Journal of Environmental Research and Public Health 18, no. 20: 10756. https://doi.org/10.3390/ijerph182010756
APA StyleRozensztrauch, A., Iwańska, M., & Bagłaj, M. (2021). The Quality of Life of Children with Myelomeningocele: A Cross-Sectional Preliminary Study. International Journal of Environmental Research and Public Health, 18(20), 10756. https://doi.org/10.3390/ijerph182010756






