Association between Influenza Vaccine Administration and Primary Care Consultations for Respiratory Infections: Sentinel Network Study of Five Seasons (2014/2015–2018/2019) in the UK
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Source and Study Design
2.2. Study Period and Study Population
2.3. Statistical Analyses
3. Results
3.1. Population Characteristics
3.2. Vaccine Type
3.3. Primary Care Consultations
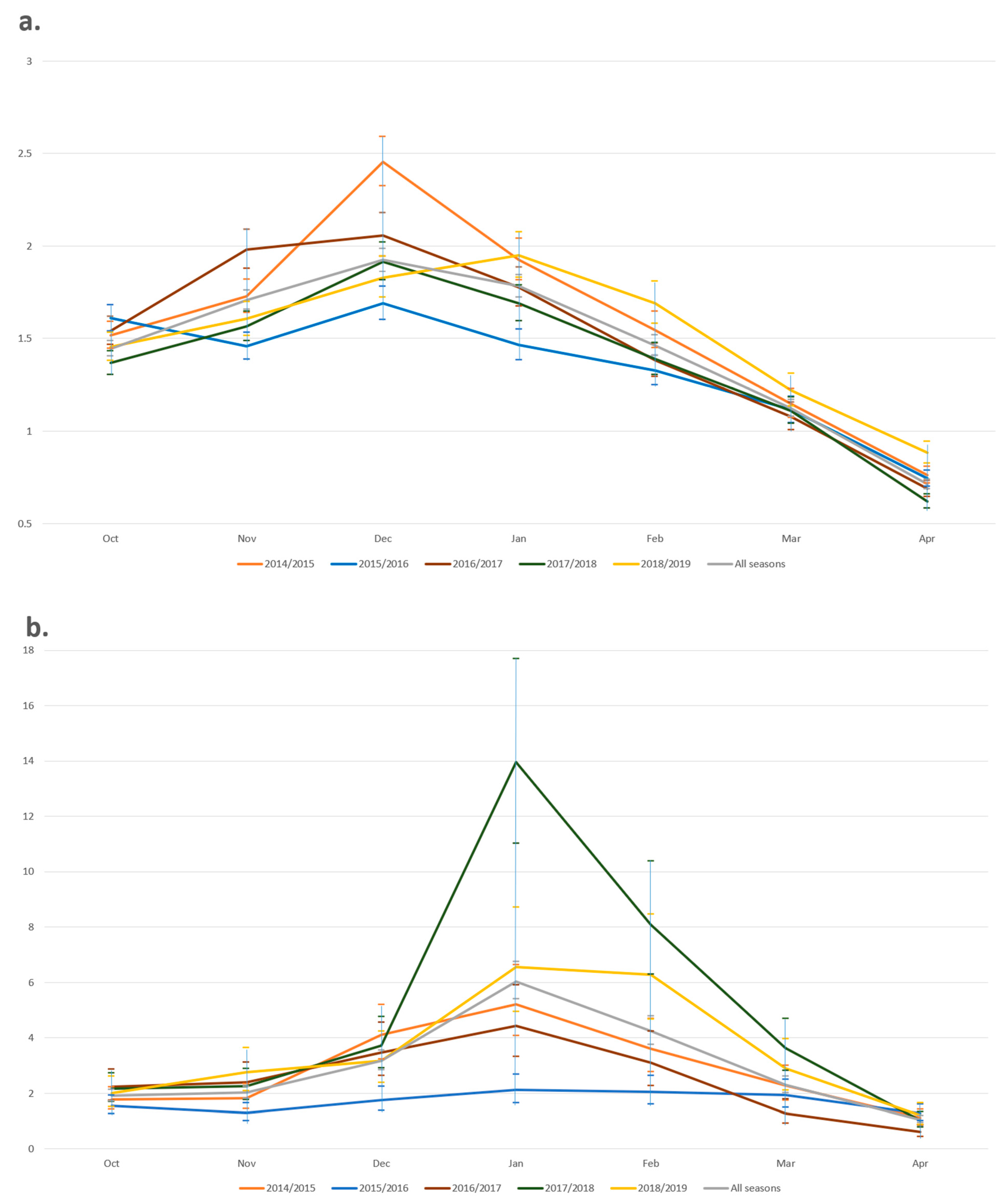
3.4. Timing of Consultations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fleming, D.M.; Taylor, R.J.; Haguinet, F.; Schuck-Paim, C.; Logie, J.; Webb, D.J.; Lustig, R.L.; Matias, G. Influenza-attributable burden in United Kingdom primary care. Epidemiol. Infect. 2016, 144, 537–547. [Google Scholar] [CrossRef]
- World Health Organization. Influenza (Seasonal). Available online: https://www.who.int/en/news-room/fact-sheets/detail/influenza-(seasonal) (accessed on 1 July 2020).
- Iuliano, A.D.; Roguski, K.M.; Chang, H.H.; Muscatello, D.J.; Palekar, R.; Tempia, S.; Cohen, C.; Gran, J.M.; Schanzer, D.; Cowling, B.J.; et al. Estimates of global seasonal influenza-associated respiratory mortality: A modelling study. Lancet 2018, 391, 1285–1300. [Google Scholar] [CrossRef]
- Thompson, W.W.; Shay, D.K.; Weintraub, E.; Brammer, L.; Cox, N.; Anderson, L.J.; Fukuda, K. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA 2003, 289, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, Y.; O’Brien, K.L.; Madhi, S.A.; Widdowson, M.A.; Byass, P.; Omer, S.B.; Abbas, Q.; Ali, A.; Amu, A.; et al. Global burden of respiratory infections associated with seasonal influenza in children under 5 years in 2018: A systematic review and modelling study. Lancet Glob. Health 2020, 8, e497–e510. [Google Scholar] [CrossRef]
- Vasileiou, E.; Sheikh, A.; Butler, C.; Von Wissmann, B.; McMenamin, J.; Ritchie, L.; Tian, L.; Simpson, C. Effectiveness of influenza vaccination for preventing influenza-related complications in people with asthma: A systematic review protocol. BMJ Open 2016, 6, e010133. [Google Scholar] [CrossRef] [PubMed]
- Ainslie, K.E.; Haber, M.; Orenstein, W.A. Challenges in estimating influenza vaccine effectiveness. Expert Rev. Vaccines 2019, 18, 615–628. [Google Scholar] [CrossRef]
- World Health Organization. Resolutions and Decisions of Regional Interest Adopted by the Fifty-Sixth World Health Assembly and by the Executive Board at Its 111th and 112th Sessions. 2003. Available online: https://apps.who.int/iris/handle/10665/122144 (accessed on 1 July 2020).
- Peasah, S.K.; Azziz-Baumgartner, E.; Breese, J.; Meltzer, M.I.; Widdowson, M.A. Influenza cost and cost-effectiveness studies globally–A review. Vaccine 2013, 31, 5339–5348. [Google Scholar] [CrossRef]
- Pitman, R.J.; Melegaro, A.; Gelb, D.; Siddiqui, M.R.; Gay, N.J.; Edmunds, W.J. Assessing the burden of influenza and other respiratory infections in England and Wales. J. Infect. 2007, 54, 530–538. [Google Scholar] [CrossRef]
- Paget, W.J.; Balderston, C.; Casas, I.; Donker, G.; Edelman, L.; Fleming, D.; Larrauri, A.; Meijer, A.; Puzelli, S.; Rizzo, C.; et al. Assessing the burden of paediatric influenza in Europe: The European Paediatric Influenza Analysis (EPIA) project. Eur. J. Pediatr. 2010, 169, 997–1008. [Google Scholar] [CrossRef]
- Pockett, R.D.; Watkins, J.; McEwan, P.; Meier, G. Burden of illness in UK subjects with reported respiratory infections vaccinated or unvaccinated against influenza: A retrospective observational study. PLoS ONE 2015, 10, e0134928. [Google Scholar] [CrossRef][Green Version]
- Hauge, S.H.; Bakken, I.J.; de Blasio, B.F.; Håberg, S.E. Burden of medically attended influenza in Norway 2008–2017. Influenza Respir. Viruses 2019, 13, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Putri, W.C.; Muscatello, D.J.; Stockwell, M.S.; Newall, A.T. Economic burden of seasonal influenza in the United States. Vaccine 2018, 36, 3960–3966. [Google Scholar] [CrossRef] [PubMed]
- de Lusignan, S.; Correa, A.; Ellis, J.; Pebody, R. Influenza vaccination: In the UK and across Europe. Br. J. Gen. Pract. 2016, 66, 452–453. [Google Scholar] [CrossRef] [PubMed]
- Public Health England. Seasonal Influenza Vaccine Uptake in GP Patients: Winter Season 2018 to 2019-Final Data for 1 September 2018 to 28 February 2019. Available online: https://www.gov.uk/government/statistics/seasonal-flu-vaccine-uptake-in-gp-patients-winter-2018-to-2019 (accessed on 30 May 2019).
- MacDonald, N.E. Vaccine hesitancy: Definition, scope and determinants. Vaccine 2015, 33, 4161–4164. [Google Scholar] [CrossRef] [PubMed]
- Hollmeyer, H.G.; Hayden, F.; Poland, G.; Buchholz, U. Influenza vaccination of health care workers in hospitals—A review of studies on attitudes and predictors. Vaccine 2009, 27, 3935–3944. [Google Scholar] [CrossRef]
- Haviari, S.; Bénet, T.; Saadatian-Elahi, M.; André, P.; Loulergue, P.; Vanhems, P. Vaccination of healthcare workers: A review. Hum. Vaccin. Immunother. 2015, 11, 2522–2537. [Google Scholar] [CrossRef]
- Smedley, J.; Poole, J.; Waclawski, E.; Stevens, A.; Harrison, J.; Watson, J.; Hayward, A.; Coggon, D. Influenza immunisation: Attitudes and beliefs of UK healthcare workers. Occup. Environ. Med. 2007, 64, 223–227. [Google Scholar] [CrossRef]
- Gazibara, T.; Kovacevic, N.; Kisic-Tepavcevic, D.; Nurkovic, S.; Kurtagic, I.; Gazibara, T.; Pekmezovic, T. Flu vaccination among older persons: Study of knowledge and practices. J. Health Popul. Nutr. 2019, 38, 1–9. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Misconceptions about Seasonal Flu and Flu Vaccines. Available online: https://www.cdc.gov/flu/prevent/misconceptions.htm (accessed on 1 September 2020).
- Stuart, M.J. Review of strategies to enhance the uptake of seasonal influenza vaccination by Australian healthcare workers. Commun. Dis. Intell. Q. Rep. 2012, 36, E268. [Google Scholar]
- Morbey, R.A.; Elliot, A.J.; Harcourt, S.; Smith, S.; de Lusignan, S.; Pebody, R.; Yeates, A.; Zambon, M.; Smith, G.E. Estimating the burden on general practitioner services in England from increases in respiratory disease associated with seasonal respiratory pathogen activity. Epidemiol. Infect. 2018, 146, 1389–1396. [Google Scholar] [CrossRef]
- Cheshire, A.; Ridge, D.; Hughes, J.; Peters, D.; Panagioti, M.; Simon, C.; Lewith, G. Influences on GP coping and resilience: A qualitative study in primary care. Br. J. Gen. Pract. 2017, 67, e428–e436. [Google Scholar] [CrossRef] [PubMed]
- Hall, L.H.; Johnson, J.; Heyhoe, J.; Watt, I.; Anderson, K.; O’Connor, D.B. Strategies to improve general practitioner well-being: Findings from a focus group study. Fam. Pract. 2018, 35, 511–516. [Google Scholar] [CrossRef] [PubMed]
- de Lusignan, S. Codes, classifications, terminologies and nomenclatures: Definition, development and application in practice. J. Innov. Health Inform. 2005, 13, 65–69. [Google Scholar] [CrossRef] [PubMed]
- de Lusignan, S.; Correa, A.; Smith, G.E.; Yonova, I.; Pebody, R.; Ferreira, F.; Elliot, A.J.; Fleming, D. RCGP Research and Surveillance Centre: 50 years’ surveillance of influenza, infections, and respiratory conditions. Br. J. Gen. Prac. 2017, 67, 440. [Google Scholar] [CrossRef]
- Smith, S.; Morbey, R.; de Lusignan, S.; Pebody, R.G.; Smith, G.E.; Elliot, A.J. Investigating regional variation of respiratory infections in a general practice syndromic surveillance system. J. Public Health (Oxf.) 2020. [Google Scholar] [CrossRef]
- Smith, S.; Morbey, R.; Pebody, R.G.; Hughes, T.C.; de Lusignan, S.; Yeates, F.A.; Thomas, H.; O’Brien, S.J.; Smith, G.E.; Elliot, A.J. Retrospective Observational Study of Atypical Winter Respiratory Illness Season Using Real-Time Syndromic Surveillance, England, 2014–2015. Emerg Infect. Dis. 2017, 23, 1834. [Google Scholar] [CrossRef]
- Public Health England. Weekly National Flu Reports: 2019 to 2020 Season 2020. Available online: https://www.gov.uk/government/statistics/weekly-national-flu-reports-2019-to-2020-season (accessed on 1 October 2020).
- Maclure, M.; Fireman, B.; Nelson, J.C.; Hua, W.; Shoaibi, A.; Paredes, A.; Madigan, D. When should case-only designs be used for safety monitoring of medical products? Pharmacoepidemiol. Drug Saf. 2012, 21, 50–61. [Google Scholar] [CrossRef]
- Takeuchi, Y.; Shinozaki, T.; Matsuyama, Y. A comparison of estimators from self-controlled case series, case-crossover design, and sequence symmetry analysis for pharmacoepidemiological studies. BMC Med. Res. Methodol. 2018, 18, 1–5. [Google Scholar] [CrossRef]
- Campos, L.F.; Şentürk, D.; Chen, Y.; Nguyen, D.V. Bias and estimation under misspecification of the risk period in self-controlled case series studies. Stat 2017, 6, 373–389. [Google Scholar] [CrossRef]
- Salisbury, D.; Ramsay, M. Influenza. In Immunisation against Infectious Disease (The Green Book); Public Health England: London, UK, 2013; Chapter 19; p. 18. [Google Scholar]
- Ferdinands, J.M.; Fry, A.M.; Reynolds, S.; Petrie, J.G.; Flannery, B.; Jackson, M.L.; Belongia, E.A. Intraseason waning of influenza vaccine protection: Evidence from the US influenza vaccine effectiveness network, 2011–2012 through 2014–2015. Clin. Infect. Dis. 2017, 64, 544–550. [Google Scholar] [CrossRef]
- Song, J.Y.; Cheong, H.J.; Hwang, I.S.; Choi, W.S.; Jo, Y.M.; Park, D.W.; Cho, G.J.; Hwang, T.G.; Kim, W.J. Long-term immunogenicity of influenza vaccine among the elderly: Risk factors for poor immune response and persistence. Vaccine 2010, 28, 3929–3935. [Google Scholar] [CrossRef] [PubMed]
- Radin, J.M.; Hawksworth, A.W.; Myers, C.A.; Ricketts, M.N.; Hansen, E.A.; Brice, G.T. Influenza vaccine effectiveness: Maintained protection throughout the duration of influenza seasons 2010–2011 through 2013–2014. Vaccine 2016, 34, 3907–3912. [Google Scholar] [CrossRef] [PubMed]
- Kissling, E.; Nunes, B.; Robertson, C.; Valenciano, M.; Reuss, A.; Larrauri, A.; Cohen, J.M.; Oroszi, B.; Rizzo, C.; Machado, A.; et al. I-MOVE multicentre case–control study 2010/11 to 2014/15: Is there within-season waning of influenza type/subtype vaccine effectiveness with increasing time since vaccination? Eurosurveillance 2016, 21, 30201. [Google Scholar] [CrossRef] [PubMed]
- Rambhia, K.J.; Rambhia, M.T. Early bird gets the flu: What should be done about waning intraseasonal immunity against seasonal influenza? Clin. Infect. Dis. 2019, 68, 1235–1240. [Google Scholar] [CrossRef]
- Young, B.; Sadarangani, S.; Jiang, L.; Wilder-Smith, A.; Chen, M.I. Duration of influenza vaccine effectiveness: A systematic review, meta-analysis, and meta-regression of test-negative design case-control studies. J. Infect. Dis 2018, 217, 731–741. [Google Scholar] [CrossRef]
- Bullock, W.; Smith, R.; Heligman, C. Managing the “Worried Well” during a large-scale incident. J. Emerg. Manag. 2018, 16, 61–66. [Google Scholar] [CrossRef]
- Spence, D. Bad Medicine: The worried hell. Br. J. Gen. Pract. 2016, 66, 526. [Google Scholar] [CrossRef]
- Rikin, S.; Jia, H.; Vargas, C.Y.; de Belliard, Y.C.; Reed, C.; LaRussa, P.; Larson, E.L.; Saiman, L.; Stockwell, M.S. Assessment of temporally-related acute respiratory illness following influenza vaccination. Vaccine 2018, 36, 1958–1964. [Google Scholar] [CrossRef]
- Cowling, B.J.; Fang, V.J.; Nishiura, H.; Chan, K.H.; Ng, S.; Ip, D.K.; Chiu, S.S.; Leung, G.M.; Peiris, J.M. Increased risk of noninfluenza respiratory virus infections associated with receipt of inactivated influenza vaccine. Clin. Infect. Dis. 2012, 54, 1778–1783. [Google Scholar] [CrossRef]
- Dierig, A.; Heron, L.G.; Lambert, S.B.; Yin, J.K.; Leask, J.; Chow, M.Y.K.; Sloots, T.P.; Nissen, M.D.; Ridda, I.; Booy, R. Epidemiology of respiratory viral infections in children enrolled in a study of influenza vaccine effectiveness. Influenza Respir. Viruses 2014, 8, 293–301. [Google Scholar] [CrossRef]
- Correa, A.; Hinton, W.; McGovern, A.; van Vlymen, J.; Yonova, I.; Jones, S.; de Lusignan, S. Royal College of General Practitioners Research and Surveillance Centre (RCGP RSC) sentinel network: A cohort profile. BMJ Open 2016, 6, 4. [Google Scholar] [CrossRef] [PubMed]
- Petersen, I.; Douglas, I.; Whitaker, H. Self controlled case series methods: An alternative to standard epidemiological study designs. BMJ 2016, 354, i4515. [Google Scholar] [CrossRef] [PubMed]
- Nichol, K.L.; Mendelman, P.M.; Mallon, K.P.; Jackson, L.A.; Gorse, G.J.; Belshe, R.B.; Glezen, W.P.; Wittes, J. Effectiveness of live, attenuated intranasal influenza virus vaccine in healthy, working adults: A randomized controlled trial. JAMA 1999, 282, 137–144. [Google Scholar] [CrossRef] [PubMed]
- de Lusignan, S.; Ferreira, F.; Damaso, S.; Byford, R.; Pathirannehelage, S.; Yeakey, A.; Yonova, I.; Schuind, A.; Dos Santos, G. Enhanced passive surveillance of influenza vaccination in England, 2016−2017–an observational study using an adverse events reporting card. Hum. Vaccin. Immunother. 2019, 15, 1048–1059. [Google Scholar] [CrossRef]
- Cross, J.W.; Joy, M.; McGee, C.; Akinyemi, O.; Gatenby, P.; de Lusignan, S. Adverse events of interest vary by influenza vaccine type and brand: Sentinel network study of eight seasons (2010–2018). Vaccine 2020, 38, 3869–3880. [Google Scholar] [CrossRef]
- Nakafero, G.; Grainge, M.J.; Myles, P.R.; Mallen, C.D.; Zhang, W.; Doherty, M.; Nguyen-Van-Tam, J.S.; Abhishek, A. Association between inactivated influenza vaccine and primary care consultations for autoimmune rheumatic disease flares: A self-controlled case series study using data from the Clinical Practice Research Datalink. Ann. Rheum. Dis. 2019, 78, 1122–1126. [Google Scholar] [CrossRef]
- Public Health England. Surveillance of Influenza and Other Respiratory Viruses in the United Kingdom: Winter 2014 to 2015. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/429617/Annualreport_March2015_ver4.pdf (accessed on 5 August 2020).
- Public Health England. Surveillance of Influenza and Other Respiratory Viruses in the United Kingdom: Winter 2015 to 2016. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/526405/Flu_Annual_Report_2015_2016.pdf (accessed on 5 August 2020).
- Public Health England. Surveillance of Influenza and Other Respiratory Viruses in the UK: Winter 2016 to 2017. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/613493/Surveillance_of_influenza_and_other_respiratory_viruses_in_the_UK_2016_to_2017.pdf (accessed on 5 August 2020).
- Public Health England. Surveillance of Influenza and Other Respiratory Viruses in the UK: Winter 2017 to 2018. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/740606/Surveillance_of_influenza_and_other_respiratory_viruses_in_the_UK_2017_to_2018.pdf (accessed on 5 August 2020).
- Pebody, R.; Djennad, A.; Ellis, J.; Andrews, N.; Marques, D.F.; Cottrell, S.; Reynolds, A.J.; Gunson, R.; Galiano, M.; Hoschler, K.; et al. End of season influenza vaccine effectiveness in adults and children in the United Kingdom in 2017/18. Eurosurveillance 2019, 24, 1800488. [Google Scholar] [CrossRef]
- de Lusignan, S.; Correa, A.; Pathirannehelage, S.; Byford, R.; Yonova, I.; Elliot, A.J.; Lamagni, T.; Amirthalingam, G.; Pebody, R.; Smith, G.; et al. RCGP Research and Surveillance Centre Annual Report 2014-2015: Disparities in presentations to primary care. Br. J. Gen. Pract. 2017, 67, e29–e40. [Google Scholar] [CrossRef]
- Chen, F.; Stevens, R. Applying lessons from behavioral economics to increase flu vaccination rates. Health Promot. Int. 2017, 32, 1067–1073. [Google Scholar] [CrossRef]
- Is My Study Research? Available online: http://www.hra-decisiontools.org.uk/research/question1.html (accessed on 15 February 2020).
| 2014/2015 | 2015/2016 | 2016/2017 | 2017/2018 | 2018/2019 | All Years | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Participants | 702,402 | 750,819 | 783,157 | 802,001 | 803,321 | 3,841,700 | ||||||
| Sex (%) | ||||||||||||
| Female | 385,186 | (54.8) | 409,582 | (54.6) | 424,536 | (54.2) | 435,135 | (54.3) | 434,291 | (54.1) | 2,088,730 | (54.4) |
| Male | 317,216 | (45.2) | 341,237 | (45.4) | 358,621 | (45.8) | 366,866 | (45.7) | 369,030 | (45.9) | 1,752,970 | (45.6) |
| Age Band (%) | ||||||||||||
| <1 | 566 | (0.1) | 348 | (0) | 0 | (0) | 0 | (0) | 0 | (0) | 914 | (0) |
| 1–4 | 51,980 | (7.4) | 63,324 | (8.4) | 61,750 | (7.9) | 35,120 | (4.4) | 15,480 | (1.9) | 227,654 | (5.9) |
| 5–9 | 10,953 | (1.6) | 22,096 | (2.9) | 29,722 | (3.8) | 35,178 | (4.4) | 44,403 | (5.5) | 142,352 | (3.7) |
| 10–14 | 10,965 | (1.6) | 9084 | (1.2) | 9569 | (1.2) | 10,518 | (1.3) | 10,117 | (1.3) | 50,253 | (1.3) |
| 15–19 | 5782 | (0.8) | 6117 | (0.8) | 6633 | (0.8) | 7708 | (1) | 7529 | (0.9) | 33,769 | (0.9) |
| 20–24 | 7628 | (1.1) | 7478 | (1) | 7170 | (0.9) | 7356 | (0.9) | 6595 | (0.8) | 36,227 | (0.9) |
| 25–29 | 11,289 | (1.6) | 11,060 | (1.5) | 10,682 | (1.4) | 10,669 | (1.3) | 8,971 | (1.1) | 52,671 | (1.4) |
| 30–34 | 15,079 | (2.1) | 14,592 | (1.9) | 14,123 | (1.8) | 14,277 | (1.8) | 12,162 | (1.5) | 70,233 | (1.8) |
| 35–39 | 15,926 | (2.3) | 15,930 | (2.1) | 15,937 | (2) | 16,534 | (2.1) | 14,930 | (1.9) | 79,257 | (2.1) |
| 40–44 | 21,759 | (3.1) | 21,568 | (2.9) | 21,038 | (2.7) | 21,446 | (2.7) | 18,901 | (2.4) | 104,712 | (2.7) |
| 45–49 | 28,825 | (4.1) | 29,130 | (3.9) | 29,380 | (3.8) | 31,333 | (3.9) | 29,072 | (3.6) | 147,740 | (3.9) |
| 50–54 | 35,914 | (5.1) | 37,061 | (4.9) | 38,065 | (4.9) | 40,962 | (5.1) | 38,991 | (4.9) | 190,993 | (5) |
| 55–59 | 42,264 | (6) | 43,520 | (5.8) | 45,624 | (5.8) | 48,899 | (6.1) | 47,963 | (6) | 228,270 | (5.9) |
| 60–64 | 69,334 | (9.9) | 67,642 | (9) | 67,858 | (8.7) | 71,451 | (8.9) | 69,384 | (8.6) | 345,669 | (9) |
| 65–69 | 130,530 | (18.6) | 131,239 | (17.5) | 132,257 | (16.9) | 132,361 | (16.5) | 124,932 | (15.6) | 651,319 | (17) |
| 70–74 | 100,488 | (14.3) | 107,254 | (14.3) | 112,618 | (14.4) | 121,626 | (15.2) | 134,622 | (16.8) | 576,608 | (15) |
| 75–79 | 77,286 | (11) | 84,202 | (11.2) | 88,337 | (11.3) | 90,357 | (11.3) | 95,964 | (11.9) | 436,146 | (11.4) |
| 80–84 | 44,375 | (6.3) | 51,215 | (6.8) | 57,330 | (7.3) | 62,973 | (7.9) | 69,811 | (8.7) | 285,704 | (7.4) |
| 85–89 | 17,170 | (2.4) | 21,677 | (2.9) | 26,373 | (3.4) | 31,553 | (3.9) | 37,780 | (4.7) | 134,553 | (3.5) |
| 90–94 | 3996 | (0.6) | 5772 | (0.8) | 7659 | (1) | 9991 | (1.2) | 13,069 | (1.6) | 40,487 | (1.1) |
| 95+ | 293 | (0) | 510 | (0.1) | 1032 | (0.1) | 1689 | (0.2) | 2645 | (0.3) | 6169 | (0.2) |
| 2014/2015 | 2015/2016 | 2016/2017 | 2017/2018 | 2018/2019 | All Years | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Valency, Route (%) | ||||||||||||
| TRI, LAIV | 720 | (0.1) | 43 | (0) | 0 | (0) | 0 | (0) | 0 | (0) | 763 | (0) |
| TRI, IIV | 627,720 | (89.4) | 646,975 | (86.2) | 65,2191 | (83) | 466,050 | (58.1) | 1655 | (0.2) | 2,394,591 | (62.3) |
| QUAD, LAIV | 66,605 | (9.5) | 88,809 | (11.8) | 96,617 | (12) | 77,192 | (9.6) | 68,783 | (8.6) | 398,006 | (10.4) |
| QUAD, IIV | 7357 | (1) | 14,991 | (2) | 34,349 | (4) | 258,759 | (32.3) | 235,125 | (29.3) | 550,581 | (14.3) |
| aTIV, IIV | 0 | (0) | 1 | (0) | 0 | (0) | 0 | (0) | 497,758 | (62) | 497,759 | (13) |
| Total | 702,402 | 750,819 | 783,157 | 802,001 | 803,321 | 3,841,700 |
| ARTI Consultations | ILI Consultations | No Consultations | Total | ||||
|---|---|---|---|---|---|---|---|
| Vaccine Valency, Route (%) | |||||||
| TRI, LAIV | 79 | (10.3) | 2 | (2.6) | 682 | (89.4) | 763 |
| TRI, IIV | 106,850 | (4.5) | 9946 | (0.4) | 2,277,795 | (95.1) | 2,394,591 |
| QUAD, LAIV | 73,042 | (18.4) | 649 | (0.2) | 324,315 | (81.5) | 398,006 |
| QUAD, IIV | 28,912 | (5.3) | 2947 | (0.5) | 518,722 | (94.2) | 550,581 |
| aTIV, IIV | 14,099 | (2.8) | 1488 | (0.3) | 482,172 | (96.9) | 497,759 |
| All vaccinations | 222,982 | (5.8) | 15,032 | (0.4) | 3,603,686 | (93.8) | 3,841,700 |
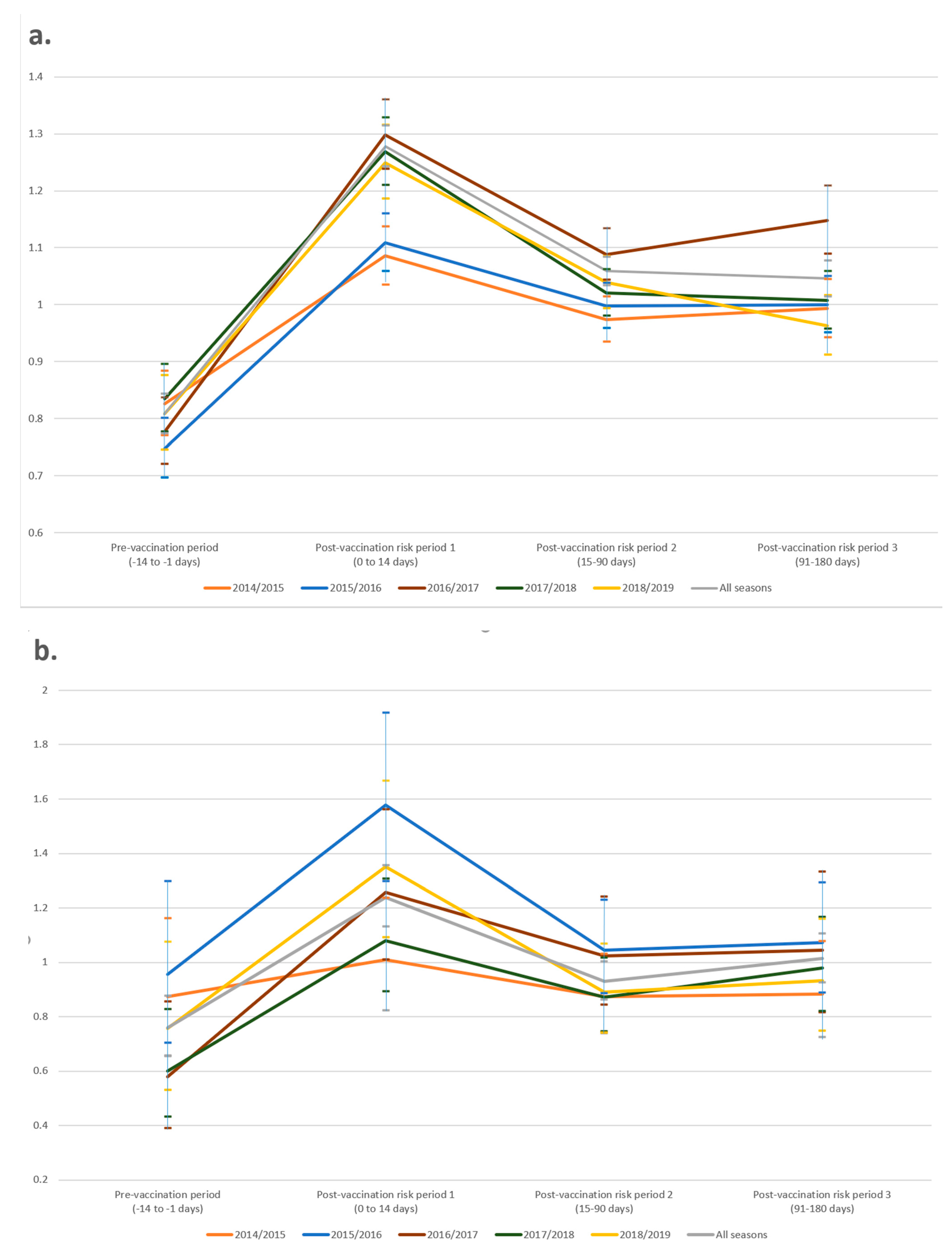
| Year | Exposure Risk Period | ARTI | ILI | ||||
|---|---|---|---|---|---|---|---|
| RI | CI (95%) | p-Value | RI | CI (95%) | p-Value | ||
| Pre-vaccination −14 to −1 days | |||||||
| 2014/2015 | 0.83 | 0.77–0.88 | <0.001 | 0.87 | 0.66–1.16 | 0.36 | |
| 2015/2016 | 0.75 | 0.70–0.80 | <0.001 | 0.96 | 0.70–1.30 | 0.77 | |
| 2016/2017 | 0.78 | 0.72–0.84 | <0.001 | 0.58 | 0.39–0.86 | 0.0062 | |
| 2017/2018 | 0.83 | 0.78–0.90 | <0.001 | 0.60 | 0.43–0.83 | 0.0019 | |
| 2018/2019 | 0.81 | 0.75–0.88 | <0.001 | 0.76 | 0.53–1.08 | 0.12 | |
| All years (2014–2019) | 0.81 | 0.77–0.84 | <0.001 | 0.76 | 0.66–0.88 | <0.001 | |
| Post-vaccination 0–14 days | |||||||
| 2014/2015 | 1.09 | 1.04–1.14 | <0.001 | 1.01 | 0.82–1.24 | 0.93 | |
| 2015/2016 | 1.11 | 1.06–1.16 | <0.001 | 1.58 | 1.30–1.92 | <0.001 | |
| 2016/2017 | 1.30 | 1.24–1.36 | <0.001 | 1.26 | 1.01–1.56 | 0.039 | |
| 2017/2018 | 1.27 | 1.21–1.33 | <0.001 | 1.08 | 0.89–1.31 | 0.43 | |
| 2018/2019 | 1.25 | 1.10–1.32 | <0.001 | 1.35 | 1.09–1.67 | 0.0054 | |
| All years (2014–2019) | 1.28 | 1.24–1.31 | <0.001 | 1.24 | 1.13–1.36 | <0.001 | |
| Post-vaccination 15–90 days | |||||||
| 2014/2015 | 0.97 | 0.93–1.01 | 0.20 | 0.87 | 0.74–1.03 | 0.11 | |
| 2015/2016 | 1.00 | 0.96–1.04 | 0.92 | 1.04 | 0.89–1.23 | 0.60 | |
| 2016/2017 | 1.09 | 1.04–1.13 | <0.001 | 1.02 | 0.84–1.24 | 0.81 | |
| 2017/2018 | 1.02 | 0.98–1.06 | 0.31 | 0.87 | 0.75–1.02 | 0.08 | |
| 2018/2019 | 1.04 | 0.99–1.09 | 0.089 | 0.89 | 0.74–1.07 | 0.21 | |
| All years (2014–2019) | 1.06 | 1.03–1.08 | <0.001 | 0.93 | 0.86–1.00 | 0.06 | |
| Post-vaccination 91–180 days | |||||||
| 2014/2015 | 0.99 | 0.94–1.05 | 0.79 | 0.88 | 0.72–1.08 | 0.22 | |
| 2015/2016 | 1.00 | 0.95–1.05 | 0.99 | 1.07 | 0.89–1.29 | 0.47 | |
| 2016/2017 | 1.15 | 1.09–1.21 | <0.001 | 1.04 | 0.82–1.33 | 0.73 | |
| 2017/2018 | 1.01 | 0.96–1.06 | 0.77 | 0.98 | 0.82–1.17 | 0.81 | |
| 2018/2019 | 0.96 | 0.91–1.02 | 0.18 | 0.93 | 0.75–1.16 | 0.53 | |
| All years (2014–2019) | 1.05 | 1.02–1.08 | 0.0034 | 1.01 | 0.93–1.11 | 0.77 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parimalanathan, V.; Joy, M.; Van Dam, P.J.; Fan, X.; de Lusignan, S. Association between Influenza Vaccine Administration and Primary Care Consultations for Respiratory Infections: Sentinel Network Study of Five Seasons (2014/2015–2018/2019) in the UK. Int. J. Environ. Res. Public Health 2021, 18, 523. https://doi.org/10.3390/ijerph18020523
Parimalanathan V, Joy M, Van Dam PJ, Fan X, de Lusignan S. Association between Influenza Vaccine Administration and Primary Care Consultations for Respiratory Infections: Sentinel Network Study of Five Seasons (2014/2015–2018/2019) in the UK. International Journal of Environmental Research and Public Health. 2021; 18(2):523. https://doi.org/10.3390/ijerph18020523
Chicago/Turabian StyleParimalanathan, Vaishnavi, Mark Joy, Pieter Jan Van Dam, Xuejuan Fan, and Simon de Lusignan. 2021. "Association between Influenza Vaccine Administration and Primary Care Consultations for Respiratory Infections: Sentinel Network Study of Five Seasons (2014/2015–2018/2019) in the UK" International Journal of Environmental Research and Public Health 18, no. 2: 523. https://doi.org/10.3390/ijerph18020523
APA StyleParimalanathan, V., Joy, M., Van Dam, P. J., Fan, X., & de Lusignan, S. (2021). Association between Influenza Vaccine Administration and Primary Care Consultations for Respiratory Infections: Sentinel Network Study of Five Seasons (2014/2015–2018/2019) in the UK. International Journal of Environmental Research and Public Health, 18(2), 523. https://doi.org/10.3390/ijerph18020523







