Associations of Residential Brownness and Greenness with Fasting Glucose in Young Healthy Adults Living in the Desert
Abstract
1. Introduction
2. Methods
2.1. Study Population
2.2. Study Assessments
2.3. Statistical Analyses
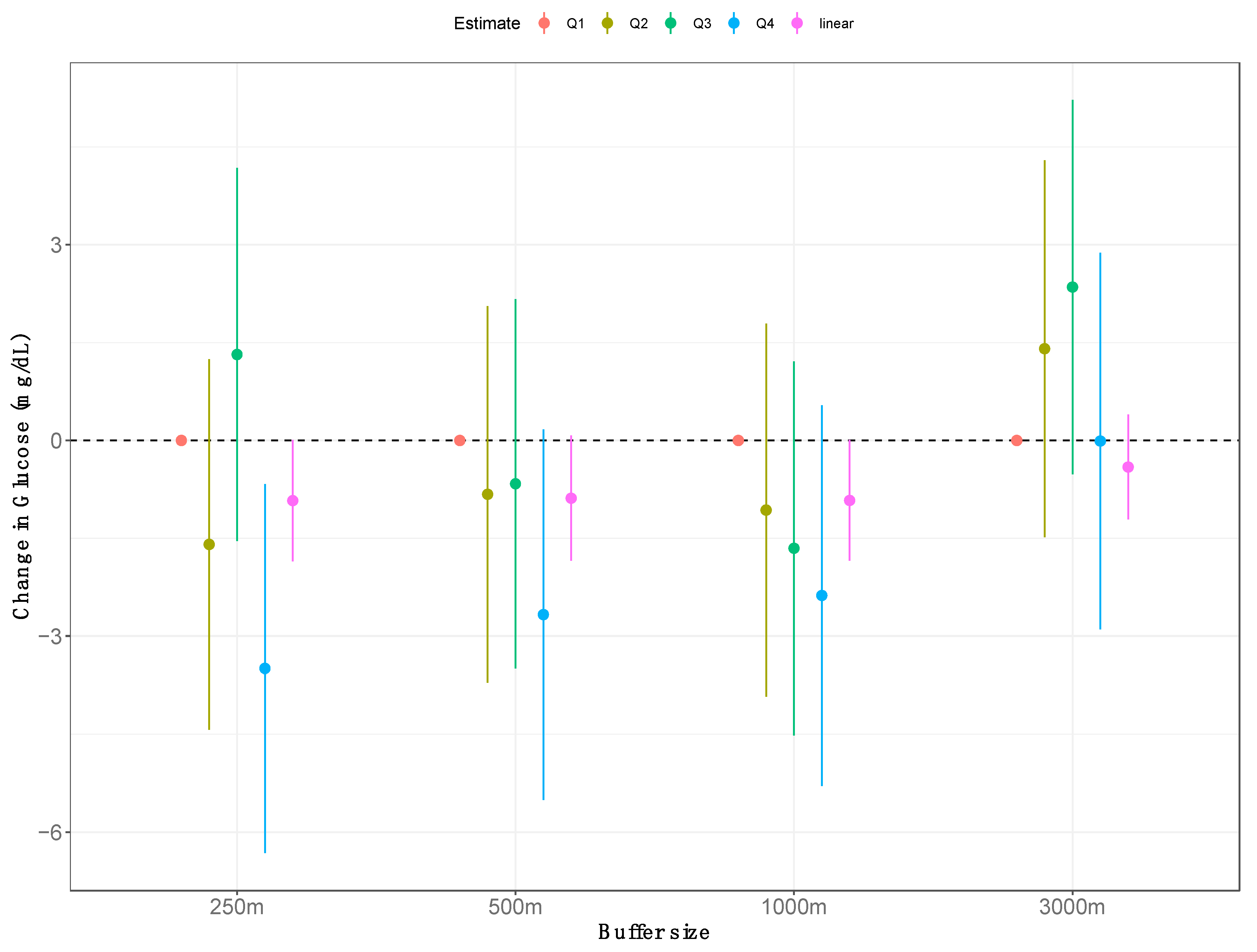
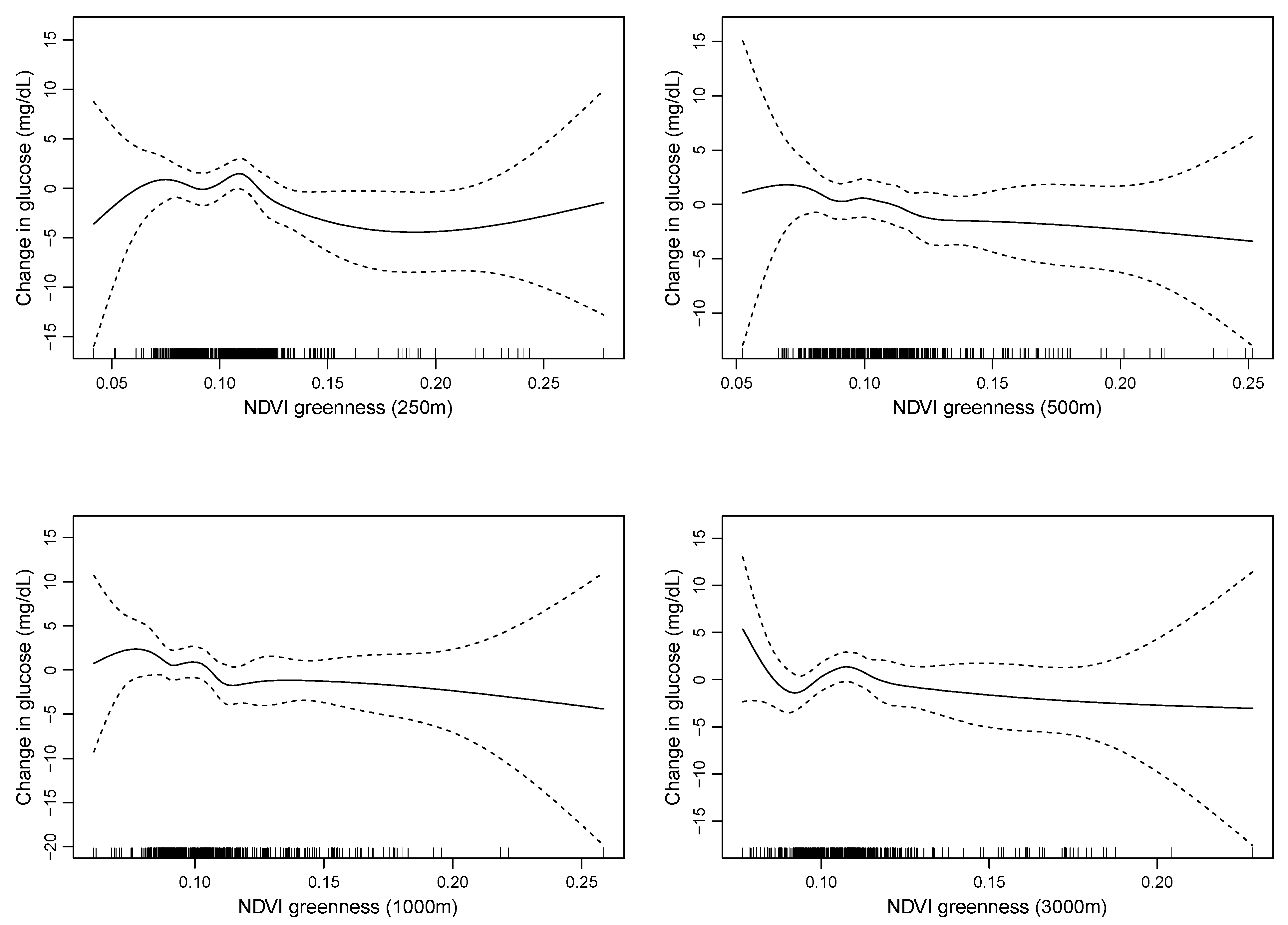
3. Results
4. Discussion
4.1. Implications for Theory
4.2. Implications for Practice
4.3. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kaplan, S. The restorative benefits of nature: Toward an integrative framework. J. Environ. Psychol. 1995, 15, 169–182. [Google Scholar] [CrossRef]
- Kellert, S.R.; Wilson, E.O. The Biophilia Hypothesis; Island Press: Washington, DC, USA, 1993. [Google Scholar]
- Moreno Brito Moura, J.; Soares Ferreira Júnior, W.; Cristina da Silva, T.; Paulino Albuquerque, U. Landscapes preferences in the human species: Insights for ethnobiology from evolutionary psychology. Ethnobiol. Conserv. 2017, 6, 1–7. [Google Scholar] [CrossRef]
- Ulrich, R.S. Aesthetic and Affective Response to Natural Environment. In Behavior and the Natural Environment; Springer: Boston, MA, USA, 1983; pp. 85–125. ISBN 978-1-4613-3539-9. [Google Scholar]
- Bratman, G.N.; Anderson, C.B.; Berman, M.G.; Cochran, B.; De Vries, S.; Flanders, J.; Folke, C.; Frumkin, H.; Gross, J.J.; Hartig, T.; et al. Nature and mental health: An ecosystem service perspective. Sci. Adv. 2019, 5, eaax0903. [Google Scholar] [CrossRef] [PubMed]
- Frumkin, H.; Bratman, G.N.; Breslow, S.J.; Cochran, B.; Kahn, P.H., Jr.; Lawler, J.J.; Levin, P.S.; Tandon, P.S.; Varanasi, U.; Wolf, K.L.; et al. Nature Contact and Human Health: A Research Agenda. Environ. Health Perspect. 2017, 125, 075001. [Google Scholar] [CrossRef] [PubMed]
- Hartig, T.; van den Berg, A.E.; Hagerhall, C.M.; Tomalak, M.; Bauer, N.; Hansmann, R.; Ojala, A.; Syngollitou, E.; Carrus, G.; van Herzele, A.; et al. Health Benefits of Nature Experience: Psychological, Social and Cultural Processes. In Forests, Trees and Human Health; Springer: Dordrecht, The Netherlands, 2011; pp. 127–168. ISBN 978-90-481-9806-1. [Google Scholar]
- James, P.; Banay, R.F.; Hart, J.E.; Laden, F. A Review of the Health Benefits of Greenness. Curr. Epidemiol. Rep. 2015, 2, 131–142. [Google Scholar] [CrossRef]
- Astell-Burt, T.; Feng, X.; Kolt, G.S. Does access to neighbourhood green space promote a healthy duration of sleep? Novel findings from a cross-sectional study of 259 319 Australians. BMJ Open 2013, 3. [Google Scholar] [CrossRef]
- Grigsby-Toussaint, D.S.; Turi, K.N.; Krupa, M.; Williams, N.J.; Pandi-Perumal, S.R.; Jean-Louis, G. Sleep insufficiency and the natural environment: Results from the US Behavioral Risk Factor Surveillance System survey. Prev. Med. 2015, 78, 78–84. [Google Scholar] [CrossRef]
- Morita, E.; Imai, M.; Okawa, M.; Miyaura, T.; Miyazaki, S. A before and after comparison of the effects of forest walking on the sleep of a community-based sample of people with sleep complaints. Biopsychosoc. Med. 2011, 5, 13. [Google Scholar] [CrossRef]
- Shin, J.C.; Parab, K.V.; An, R.; Grigsby-Toussaint, D.S. Greenspace exposure and sleep: A systematic review. Environ. Res. 2020, 182, 109081. [Google Scholar] [CrossRef]
- Triguero-Mas, M.; Donaire-Gonzalez, D.; Seto, E.; Valentin, A.; Martinez, D.; Smith, G.; Hurst, G.; Carrasco-Turigas, G.; Masterson, D.; van den Berg, M.; et al. Natural outdoor environments and mental health: Stress as a possible mechanism. Environ. Res. 2017, 159, 629–638. [Google Scholar] [CrossRef]
- Berman, M.G.; Jonides, J.; Kaplan, S. The Cognitive Benefits of Interacting With Nature. Psychol. Sci. 2008, 19, 1207–1212. [Google Scholar] [CrossRef] [PubMed]
- Bratman, G.N.; Daily, G.C.; Levy, B.J.; Gross, J.J. The benefits of nature experience: Improved affect and cognition. Landsc. Urban Plan. 2015, 138, 41–50. [Google Scholar] [CrossRef]
- Wells, N.M. At Home with Nature. Environ. Behav. 2000, 32, 775–795. [Google Scholar] [CrossRef]
- Kuo, M. How might contact with nature promote human health? Promising mechanisms and a possible central pathway. Front. Psychol. 2015, 6, 1093. [Google Scholar] [CrossRef] [PubMed]
- Li, Q. Effect of forest bathing trips on human immune function. Environ. Health Prev. Med. 2010, 15, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Morimoto, K.; Kobayashi, M.; Inagaki, H.; Katsumata, M.; Hirata, Y.; Hirata, K.; Suzuki, H.; Li, Y.J.; Wakayama, Y.; et al. Visiting a Forest, but Not a City, Increases Human Natural Killer Activity and Expression of Anti-Cancer Proteins. Int. J. Immunopathol. Pharmacol. 2008, 21, 117–127. [Google Scholar] [CrossRef]
- Mygind, L.; Kjeldsted, E.; Hartmeyer, R.; Mygind, E.; Stevenson, M.P.; Quintana, D.S.; Bentsen, P. Effects of Public Green Space on Acute Psychophysiological Stress Response: A Systematic Review and Meta-Analysis of the Experimental and Quasi-Experimental Evidence. Environ. Behav. 2019. [Google Scholar] [CrossRef]
- Roe, J.J.; Thompson, C.W.; Aspinall, P.A.; Brewer, M.J.; Duff, E.I.; Miller, D.; Mitchell, R.; Clow, A. Green space and stress: Evidence from cortisol measures in deprived urban communities. Int. J. Environ. Res. Public Health 2013, 10, 4086–4103. [Google Scholar] [CrossRef]
- Ulrich, R.S.; Simons, R.F.; Losito, B.D.; Fiorito, E.; Miles, M.A.; Zelson, M. Stress recovery during exposure to natural and urban environments. J. Environ. Psychol. 1991, 11, 201–230. [Google Scholar] [CrossRef]
- Van den Berg, A.E.; Maas, J.; Verheij, R.A.; Groenewegen, P.P. Green space as a buffer between stressful life events and health. Soc. Sci. Med. 2010, 70, 1203–1210. [Google Scholar] [CrossRef]
- Banay, R.F.; James, P.; Hart, J.E.; Kubzansky, L.D.; Spiegelman, D.; Okereke, O.I.; Spengler, J.D.; Laden, F. Greenness and Depression Incidence among Older Women. Environ. Health Perspect. 2019, 127, 27001. [Google Scholar] [CrossRef] [PubMed]
- Beyer, K.M.; Kaltenbach, A.; Szabo, A.; Bogar, S.; Nieto, F.J.; Malecki, K.M. Exposure to neighborhood green space and mental health: Evidence from the survey of the health of Wisconsin. Int. J. Environ. Res. Public Health 2014, 11, 3453–3472. [Google Scholar] [CrossRef] [PubMed]
- Maas, J.; Verheij, R.A.; de Vries, S.; Spreeuwenberg, P.; Schellevis, F.G.; Groenewegen, P.P. Morbidity is related to a green living environment. J. Epidemiol. Community Health 2009, 63, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Roberts, H.; van Lissa, C.; Hagedoorn, P.; Kellar, I.; Helbich, M. The effect of short-term exposure to the natural environment on depressive mood: A systematic review and meta-analysis. Environ. Res. 2019, 177. [Google Scholar] [CrossRef]
- Cracknell, D. By the Sea: The Therapeutic Benefits of Being in, on and by the Water; Hachette UK: London, UK, 2019. [Google Scholar]
- Gascon, M.; Triguero-Mas, M.; Martinez, D.; Dadvand, P.; Forns, J.; Plasencia, A.; Nieuwenhuijsen, M.J. Mental health benefits of long-term exposure to residential green and blue spaces: A systematic review. Int. J. Environ. Res. Public Health 2015, 12, 4354–4379. [Google Scholar] [CrossRef]
- Gascon, M.; Zijlema, W.; Vert, C.; White, M.P.; Nieuwenhuijsen, M.J. Outdoor blue spaces, human health and well-being: A systematic review of quantitative studies. Int. J. Hyg. Environ. Health 2017, 220, 1207–1221. [Google Scholar] [CrossRef] [PubMed]
- Völker, S.; Kistemann, T. The impact of blue space on human health and well-being-Salutogenetic health effects of inland surface waters: A review. Int. J. Hyg. Environ. Health 2011, 214, 449–460. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, B.W.; Lovell, R.; Higgins, S.L.; White, M.P.; Alcock, I.; Osborne, N.J.; Husk, K.; Sabel, C.E.; Depledge, M.H. Beyond greenspace: An ecological study of population general health and indicators of natural environment type and quality. Int. J. Health Geogr. 2015, 14, 17. [Google Scholar] [CrossRef]
- Wheeler, B.W.; White, M.; Stahl-Timmins, W.; Depledge, M.H. Does living by the coast improve health and wellbeing? Health Place 2012, 18, 1198–1201. [Google Scholar] [CrossRef]
- White, M.P.; Alcock, I.; Wheeler, B.W.; Depledge, M.H. Coastal proximity, health and well-being: Results from a longitudinal panel survey. Health Place 2013, 23, 97–103. [Google Scholar] [CrossRef]
- Tchakerian, V.; Pease, P. The Critical Zone in Desert Environments. In Developments in Earth Surface Processes; Giardino, J.R., Houser, C., Eds.; Elsevier: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Appleton, J. The Experience of Landscape; John Wiley: New York, NY, USA, 1975. [Google Scholar]
- Shalev, I. Pictorial and mental arid landscape images reduce the motivation to change negative habits. J. Environ. Psychol. 2016, 45, 30–39. [Google Scholar] [CrossRef]
- Han, K.-T. Responses to Six Major Terrestrial Biomes in Terms of Scenic Beauty, Preference, and Restorativeness. Environ. Behav. 2007, 39, 529–556. [Google Scholar] [CrossRef]
- Andrews, P. Palaeoecology of Laetoli. J. Hum. Evol. 1989, 18, 173–181. [Google Scholar] [CrossRef]
- Balling, J.D.; Falk, J.H. Development of Visual Preference for Natural Environments. Environ. Behav. 1982, 14, 5–28. [Google Scholar] [CrossRef]
- Orians, G.H. Habitat selection: General theory and applications to human behavior. In The Evolution of Human Social Behavior; Elsevier: Amsterdam, The Netherlands, 1980; pp. 49–66. [Google Scholar]
- Millennim Ecosystem Assessment. Ecosystems and Human Well-Being: Biodiversity Synthesis; World Resources Institute: Washington, DC, USA, 2005. [Google Scholar]
- Huang, J.; Yu, H.; Guan, X.; Wang, G.; Guo, R. Accelerated dryland expansion under climate change. Nat. Clim. Chang. 2015, 6, 166–171. [Google Scholar] [CrossRef]
- Dendup, T.; Feng, X.; Clingan, S.; Astell-Burt, T. Environmental Risk Factors for Developing Type 2 Diabetes Mellitus: A Systematic Review. Int. J. Environ. Res. Public Health 2018, 15, 78. [Google Scholar] [CrossRef]
- Butler, A.M. Social Determinants of Health and Racial/Ethnic Disparities in Type 2 Diabetes in Youth. Curr. Diabetes Rep. 2017, 17, 60. [Google Scholar] [CrossRef]
- Astell-Burt, T.; Feng, X.; Kolt, G.S. Is neighborhood green space associated with a lower risk of type 2 diabetes? Evidence from 267,072 Australians. Diabetes Care 2014, 37, 197–201. [Google Scholar] [CrossRef]
- Dadvand, P.; Poursafa, P.; Heshmat, R.; Motlagh, M.E.; Qorbani, M.; Basagana, X.; Kelishadi, R. Use of green spaces and blood glucose in children; a population-based CASPIAN-V study. Environ. Pollut. 2018, 243, 1134–1140. [Google Scholar] [CrossRef]
- Liao, J.; Chen, X.; Xu, S.; Li, Y.; Zhang, B.; Cao, Z.; Zhang, Y.; Liang, S.; Hu, K.; Xia, W. Effect of residential exposure to green space on maternal blood glucose levels, impaired glucose tolerance, and gestational diabetes mellitus. Environ. Res. 2019, 176. [Google Scholar] [CrossRef]
- Yang, B.Y.; Markevych, I.; Heinrich, J.; Bowatte, G.; Bloom, M.S.; Guo, Y.; Dharmage, S.C.; Jalaludin, B.; Knibbs, L.D.; Morawska, L.; et al. Associations of greenness with diabetes mellitus and glucose-homeostasis markers: The 33 Communities Chinese Health Study. Int. J. Hyg. Environ. Health 2019, 222, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Dalton, A.M.; Jones, A.P.; Sharp, S.J.; Cooper, A.J.; Griffin, S.; Wareham, N.J. Residential neighbourhood greenspace is associated with reduced risk of incident diabetes in older people: A prospective cohort study. BMC Public Health 2016, 16, 1171. [Google Scholar] [CrossRef] [PubMed]
- Clark, C.; Sbihi, H.; Tamburic, L.; Brauer, M.; Frank, L.D.; Davies, H.W. Association of Long-Term Exposure to Transportation Noise and Traffic-Related Air Pollution with the Incidence of Diabetes: A Prospective Cohort Study. Environ. Health Perspect. 2017, 125. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.-C.; Yen, Y.-T.; Lao, X.Q.; Chen, Y.-H.; Chan, T.-C. Association between neighborhood greenspace and fasting plasma glucose from a large cohort study in Taiwan. Urban For. Urban Green. 2019, 44. [Google Scholar] [CrossRef]
- Olvera Alvarez, H.A.; Provencio-Vasquez, E.; Slavich, G.M.; Laurent, J.G.C.; Browning, M.; McKee-Lopez, G.; Robbins, L.; Spengler, J.D. Stress and Health in Nursing Students. Nurs. Res. 2019, 68, 453–463. [Google Scholar] [CrossRef]
- Shemesh, T.; Rowley, K.G.; Shephard, M.; Piers, L.S.; O’Dea, K. Agreement between laboratory results and on-site pathology testing using Bayer DCA2000+ and Cholestech LDX point-of-care methods in remote Australian Aboriginal communities. Clin. Chim. Acta 2006, 367, 69–76. [Google Scholar] [CrossRef]
- Browning, M.; Lee, K. Within What Distance Does “Greenness” Best Predict Physical Health? A Systematic Review of Articles with GIS Buffer Analyses across the Lifespan. Int. J. Environ. Res. Public Health 2017, 14, 675. [Google Scholar] [CrossRef]
- Vermote, E.; Justice, C.; Claverie, M.; Franch, B. Preliminary analysis of the performance of the Landsat 8/OLI land surface reflectance product. Remote Sens. Environ. 2016, 185, 46–56. [Google Scholar] [CrossRef]
- Bohannon, R.W. Comfortable and maximum walking speed of adults aged 20—79 years: Reference values and determinants. Age Ageing 1997, 26, 15–19. [Google Scholar] [CrossRef]
- Wickham, J.D.; Stehman, S.V.; Gass, L.; Dewitz, J.; Fry, J.A.; Wade, T.G. Accuracy assessment of NLCD 2006 land cover and impervious surface. Remote Sens. Environ. 2013, 130, 294–304. [Google Scholar] [CrossRef]
- Ainsworth, B.E.; Haskell, W.L.; Herrmann, S.D.; Meckes, N.; Bassett, D.R., Jr.; Tudor-Locke, C.; Greer, J.L.; Vezina, J.; Whitt-Glover, M.C.; Leon, A.S. Compendium of Physical Activities: A second update of codes and MET values. Med. Sci. Sports Exerc. 2011, 43, 1575–1581. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.W.; Chung, R.; Juarez, D.T. Prevalence of comorbid conditions with aging among patients with diabetes and cardiovascular disease. Hawaii Med. J. 2011, 70, 209–213. [Google Scholar] [PubMed]
- Pescador Jimenez, M.I.; Oken, E.; Gold, D.R.; Luttmann-Gibson, H.; Requia, W.J.; Gingras, V.; Hivert, M.-F.; Rimm, E.B.; James, P. Abstract 026: Early Life Exposure to Green Areas and Diabetes Risk, an Assessment From Infancy to Early Teens. Circulation 2019, 139. [Google Scholar] [CrossRef]
- Broekhuizen, K.; De Vries, S.; Pierik, F. Healthy Aging in a Green Living Environment: A Systematic Review of the Literature; TNO: Leiden, The Netherlands, 2013. [Google Scholar]
- Hartig, T.; Mitchell, R.; de Vries, S.; Frumkin, H. Nature and health. Annu. Rev. Public Health 2014, 35, 207–228. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, R.; Kaplan, S. The Experience of Nature: A Psychological Perspective; Cambridge University Press: New York, NY, USA, 1989. [Google Scholar]
- Wilson, E.O. Biophilia; Harvard University Press: Cambridge, MA, USA, 1984. [Google Scholar]
- Houlden, V.; Weich, S.; Porto de Albuquerque, J.; Jarvis, S.; Rees, K. The relationship between greenspace and the mental wellbeing of adults: A systematic review. PLoS ONE 2018, 13, e0203000. [Google Scholar] [CrossRef]
- James, P.; Hart, J.E.; Banay, R.F.; Laden, F. Exposure to Greenness and Mortality in a Nationwide Prospective Cohort Study of Women. Environ. Health Perspect. 2016, 124, 1344–1352. [Google Scholar] [CrossRef]
- DeMiglio, L.; Williams, A. A sense of place, a sense of well-being. In Sense of Place, Health and Quality of Life; Routledge: London, UK, 2008; pp. 35–50. [Google Scholar]
- Hausmann, A.; Slotow, R.O.B.; Burns, J.K.; Di Minin, E. The ecosystem service of sense of place: Benefits for human well-being and biodiversity conservation. Environ. Conserv. 2015, 43, 117–127. [Google Scholar] [CrossRef]
- Wellen, K.E.; Hotamisligil., G.S. Inflammation, stress, and diabetes. J. Clin. Investig. 2005, 115, 1111–1119. [Google Scholar] [CrossRef]
- Dismukes, A.R.; Shirtcliff, E.A.; Hanson, J.L.; Pollak, S.D. Context influences the interplay of endocrine axes across the day. Dev. Psychobiol. 2015, 57, 731–741. [Google Scholar] [CrossRef]
- Ahmad, M.H.; Fatima, M.; Mondal, A.C. Role of Hypothalamic-Pituitary-Adrenal Axis, Hypothalamic-Pituitary-Gonadal Axis and Insulin Signaling in the Pathophysiology of Alzheimer’s Disease. Neuropsychobiology 2019, 77, 197–205. [Google Scholar] [CrossRef]
- Rigolon, A.; Browning, M.; Jennings, V. Inequities in the quality of urban park systems: An environmental justice investigation of cities in the United States. Landsc. Urban Plan. 2018, 178, 156–169. [Google Scholar] [CrossRef]
- Brown, S.C.; Lombard, J.; Wang, K.; Byrne, M.M.; Toro, M.; Plater-Zyberk, E.; Feaster, D.J.; Kardys, J.; Nardi, M.I.; Perez-Gomez, G.; et al. Neighborhood Greenness and Chronic Health Conditions in Medicare Beneficiaries. Am. J. Prev. Med. 2016, 51, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Diabetes—United States, 2006 and 2010. Mmwr. Surveill. Summ. 2013, 62, 99–104. [Google Scholar]
- U.S. Census Bureau. Fast Growth in the Desert Southwest Continues. Available online: https://www.census.gov/library/stories/2019/02/fast-growth-in-desert-southwest-continues.html (accessed on 17 November 2020).
- Yitshak-Sade, M.; Kloog, I.; Novack, V. Do air pollution and neighborhood greenness exposures improve the predicted cardiovascular risk? Environ. Int. 2017, 107, 147–153. [Google Scholar] [CrossRef] [PubMed]

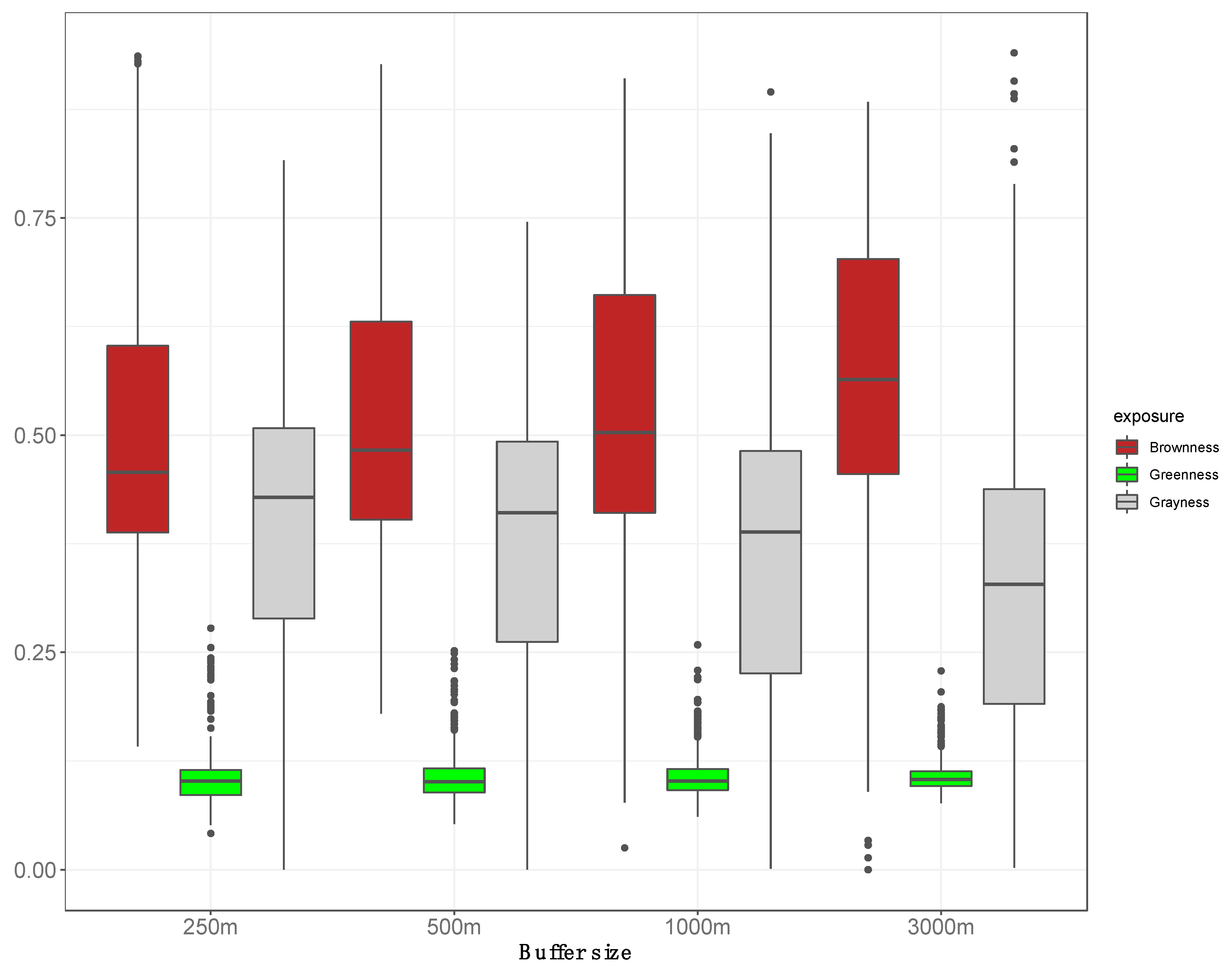
| Characteristic | Overall | Greenness (Q1) ** | Greenness (Q2) ** | Greenness (Q3) ** | Greenness (Q4) ** | ANOVA F-Test p-Value |
|---|---|---|---|---|---|---|
| Age (years), mean ± SD | 24.20 ± 4.53 | 25.08 ± 4.97 | 23.67 ± 4.10 | 24.54 ± 4.94 | 23.51 ± 3.88 | 0.03 |
| BMI (kg/m2), mean ± SD | 25.38 ± 5.36 | 25.01 ± 5.53 | 24.60 ± 4.74 | 26.33 ± 6.04 | 25.58 ± 4.92 | 0.08 |
| Male, n (%) | 92 (20.2) | 24 (21.1) | 20 (17.5) | 28 (24.6) | 20 (17.5) | 0.50 |
| Race, n (%) | 0.17 | |||||
| American Indian or Alaska Native | 10 (2.2) | 4 (3.5) | 1 (0.9) | 2 (1.8) | 3 (2.6) | |
| Asian | 9 (2.0) | 3 (2.6) | 2 (1.8) | 2 (1.8) | 2 (1.8) | |
| Black or African American | 11 (2.4) | 6 (5.3) | 4 (3.5) | 0 (0) | 1 (0.9) | |
| Native Hawaiian or Other Pacific Islander | 1 (0.2) | 1 (0.9) | 0 (0) | 0 (0) | 0 (0) | |
| White | 425 (93.2) | 100 (87.7) | 107 (93.9) | 110 (96.5) | 108 (94.7) | |
| Hispanic or Latino, n (%) | 413 (90.6) | 98 (86.0) | 105 (92.1) | 104 (91.2) | 106 (93.0) | 0.27 |
| Maternal Education, n (%) | 0.08 | |||||
| No High School | 49 (10.7) | 19 (16.7) | 9 (7.8) | 7 (6.1) | 14 (12.2) | |
| Some High School | 35 (7.7) | 8 (7.0) | 3 (2.6) | 11 (9.6) | 13 (11.4) | |
| High School Graduate | 97 (21.0) | 19 (16.7) | 22 (19.3) | 26 (22.8) | 30 (26.3) | |
| Some College | 129 (28.2) | 29 (25.4) | 41 (36.0) | 36 (31.6) | 23 (20.2) | |
| College graduate | 142 (31.1) | 38 (33.3) | 38 (33.3) | 32 (28.1) | 34 (29.8) | |
| Income/Poverty *, mean ± SD | 2.27 ± 1.83 | 2.26 ± 1.94 | 2.11 ± 1.64 | 2.43 ± 1.91 | 2.30 ± 1.84 | 0.93 |
| Physical activity * (MET hours/week), mean ± SD | 63.6 ± 73.9 | 59.2 ± 57.9 | 59.3 ± 73.4 | 69.0 ± 91.3 | 67.7 ± 69.9 | 0.26 |
| Sleep (hours) *, mean ± SD | 6.39 ± 1.08 | 6.42 ± 1.06 | 6.32 ± 1.09 | 6.37 ± 1.12 | 6.46 ± 1.07 | 0.70 |
| Model | Exposure(s) | Change in Fasting Glucose, mg/dL (95% CI) | ||||
|---|---|---|---|---|---|---|
| Brownness | Greenness | Grayness | ||||
| 1 | ● | −0.01 (−0.07, 0.05) | ||||
| 2 | ● | ◆ | −0.01 (−0.07, 0.05) | −0.32 (−0.63, −0.01) | ||
| 3 | ● | ■ | 0.31 (−0.02, 0.63) | 0.32 (0.01, 0.63) | ||
| 4 | ◆ | −0.32 (−0.63, −0.01) | ||||
| 5 | ◆ | ■ | −0.31 (−0.63, 0.02) | 0.01 (−0.05, 0.07) | ||
| 6 | ■ | 0.02 (−0.04, 0.08) | ||||
| Potential Mediator | Association with An IQR Increase in Greenness | |
|---|---|---|
| Change (95% CI) | OR (95% CI) | |
| BMI (kg/m3) | 0.49 (0.00, 0.97) | 1.21 (0.96, 1.52) |
| Physical Activity (percent change) ** | 5.1 (−5.5, 16.8) | 1.42 (0.92, 2.19) |
| Sleep Duration (hours) | −0.04 (−0.14, 0.05) | |
| Sleep Quality | 1.16 (0.95, 1.40) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olvera-Alvarez, H.A.; Browning, M.H.E.M.; Neophytou, A.M.; Bratman, G.N. Associations of Residential Brownness and Greenness with Fasting Glucose in Young Healthy Adults Living in the Desert. Int. J. Environ. Res. Public Health 2021, 18, 520. https://doi.org/10.3390/ijerph18020520
Olvera-Alvarez HA, Browning MHEM, Neophytou AM, Bratman GN. Associations of Residential Brownness and Greenness with Fasting Glucose in Young Healthy Adults Living in the Desert. International Journal of Environmental Research and Public Health. 2021; 18(2):520. https://doi.org/10.3390/ijerph18020520
Chicago/Turabian StyleOlvera-Alvarez, Hector A., Matthew H. E. M. Browning, Andreas M. Neophytou, and Gregory N. Bratman. 2021. "Associations of Residential Brownness and Greenness with Fasting Glucose in Young Healthy Adults Living in the Desert" International Journal of Environmental Research and Public Health 18, no. 2: 520. https://doi.org/10.3390/ijerph18020520
APA StyleOlvera-Alvarez, H. A., Browning, M. H. E. M., Neophytou, A. M., & Bratman, G. N. (2021). Associations of Residential Brownness and Greenness with Fasting Glucose in Young Healthy Adults Living in the Desert. International Journal of Environmental Research and Public Health, 18(2), 520. https://doi.org/10.3390/ijerph18020520







