Access to Daylight at Home Improves Circadian Alignment, Sleep, and Mental Health in Healthy Adults: A Crossover Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Experimental Conditions
2.3. Study Outcomes
2.3.1. Environmental Conditions
2.3.2. Objective Sleep and Rest-Activity Patterns
2.3.3. Biospecimen Collection for Dim Light Melatonin Onset (DLMO) Analysis
2.3.4. Surveys
- Sleep disturbance 4a (SD4a), which measures perceptions of sleep quality, depth, and restoration (i.e., Sleep quality rating, with higher score as poorer quality) using four items;
- Sleep-related impairment 8a (SRI8a), which measures perceptions of alertness, sleepiness during waking hours, and associated functional impairments (e.g., “I had a hard time getting things done today because I was sleepy”) using eight items;
- Anxiety 4a (A4a), which measures self-reported fear, panic, anxious misery, and tension (e.g., “My worries overwhelmed me”) using four items;
- Psychological stress 4a (PS4a), which measures feelings of being overwhelmed or lacking control (e.g., “I felt stressed”) using four items;
- Depression 4a (D4a), which measures negative mood and views of self-worth (e.g., ”I felt depressed”) using four items; and
- Positive affect 15a (PA15a), which measures momentary experiences such as pleasure, joy, pride and engagement (e.g., “I felt attentive”) using 15 items.
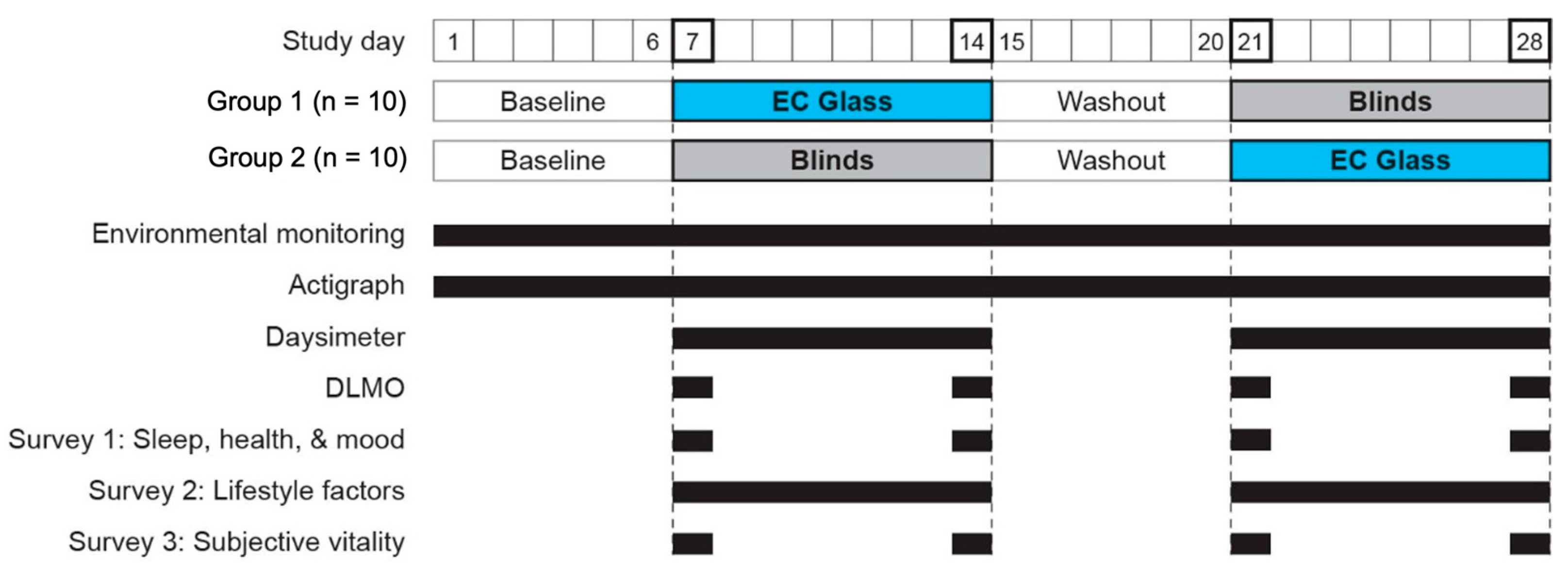
2.4. Study Protocol
2.5. Statistical Analyses
3. Results
3.1. Study Population
3.2. Environmental Conditions
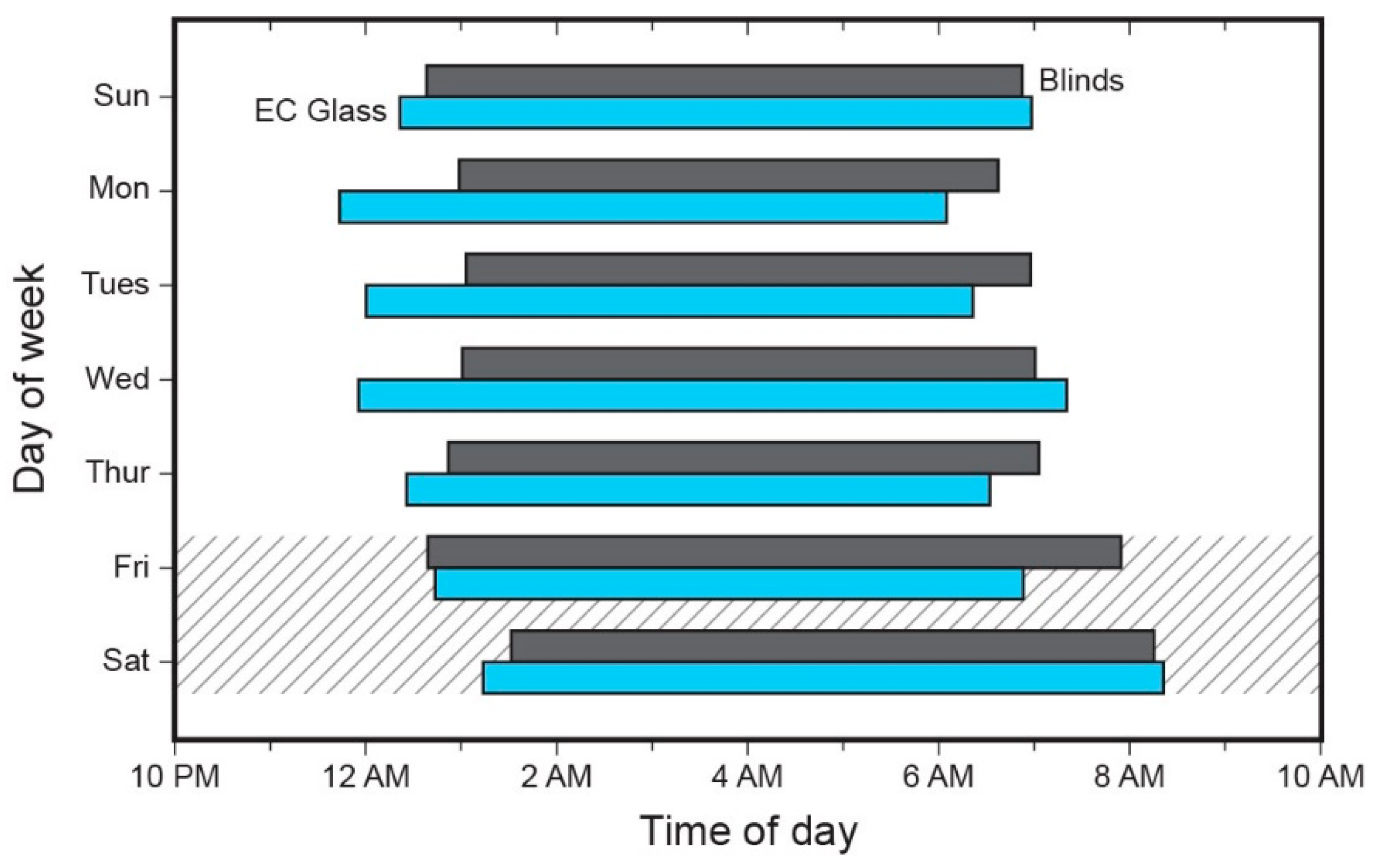
3.3. Sleep Timing
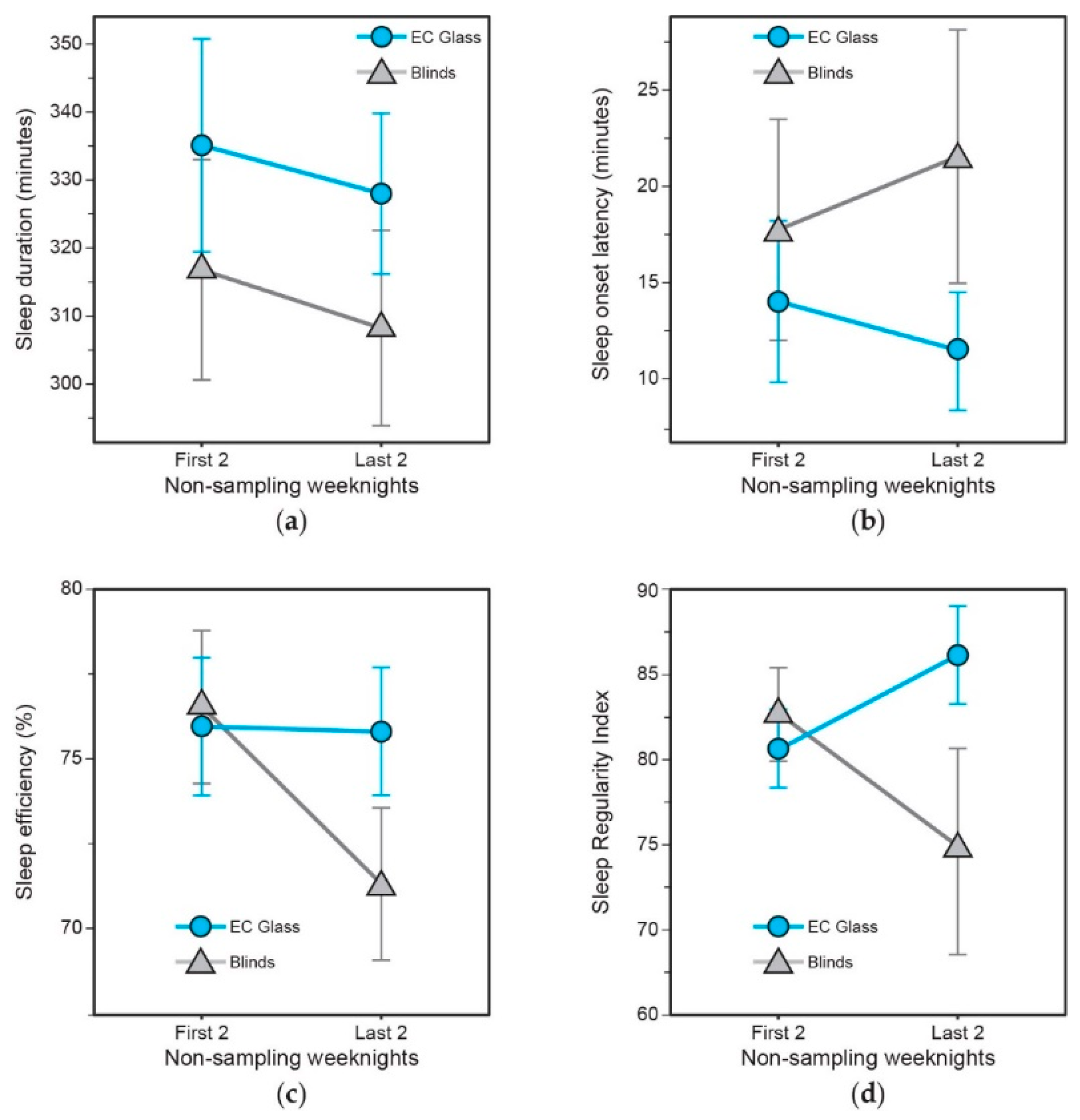
3.4. Objective Sleep Quality and Sleep Regularity
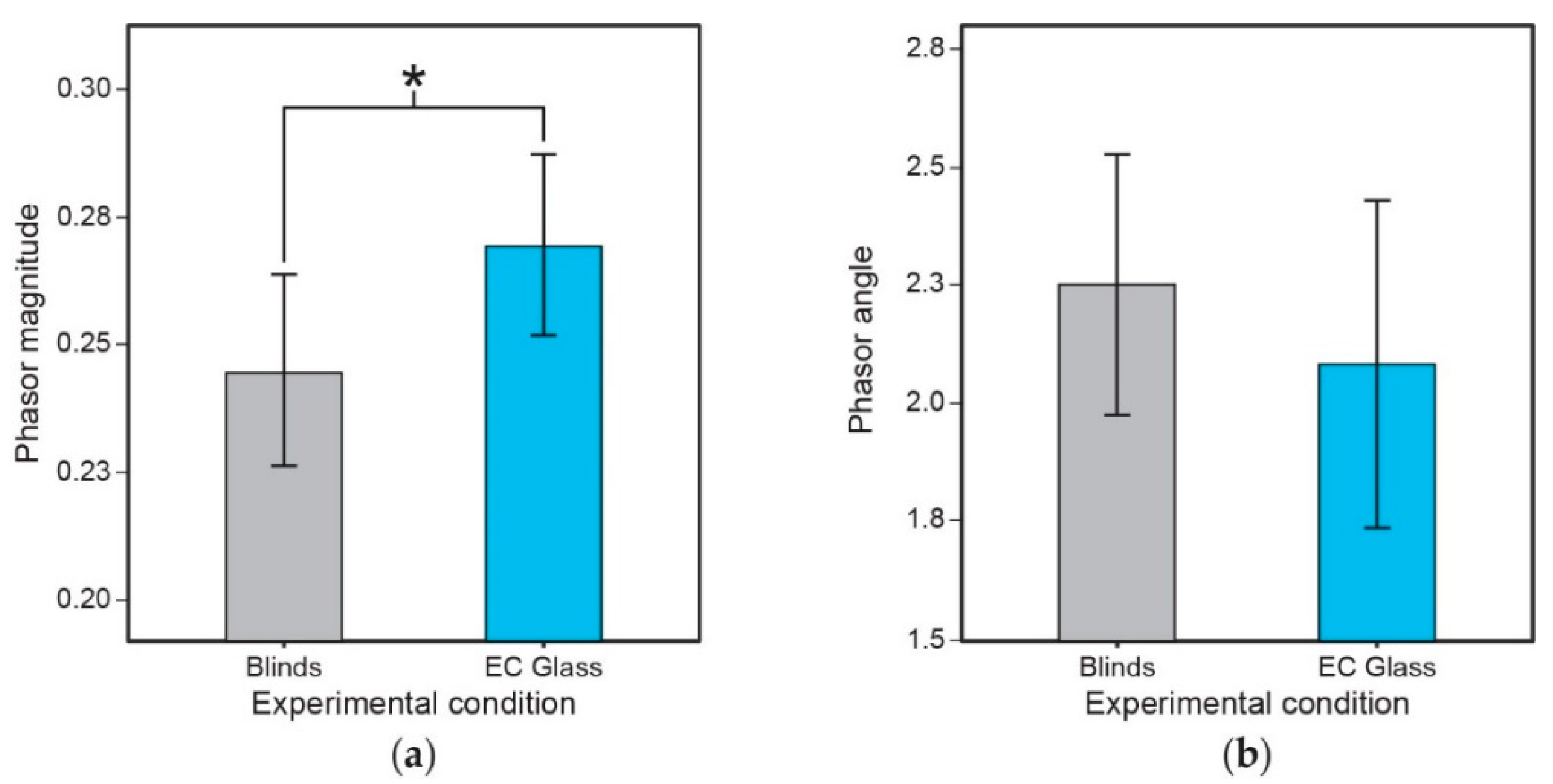
3.5. Phase Relationship between Light-Dark and Activity-Rest (Phasor Analyses)
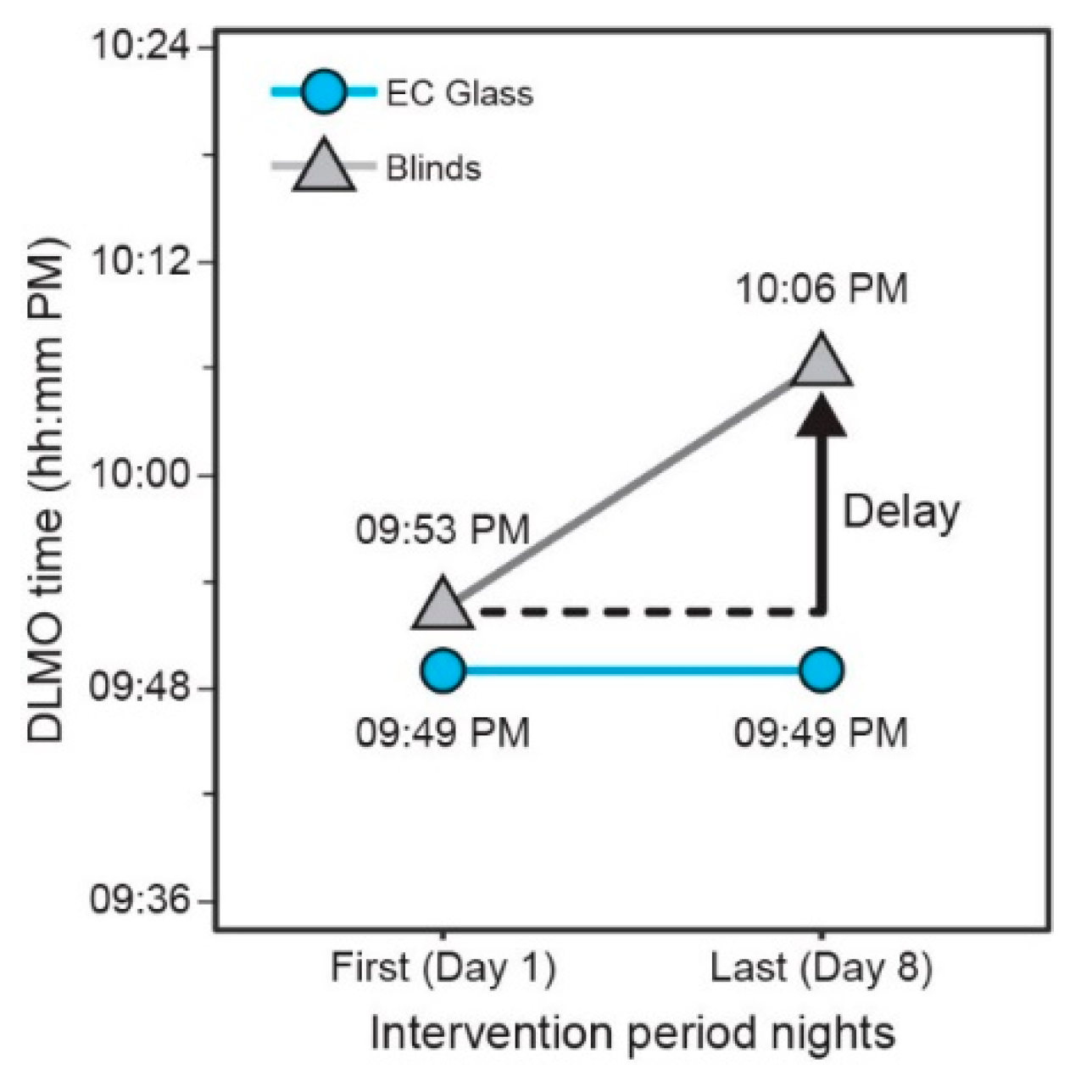
3.6. Dim Light Melatonin Onset (DLMO)
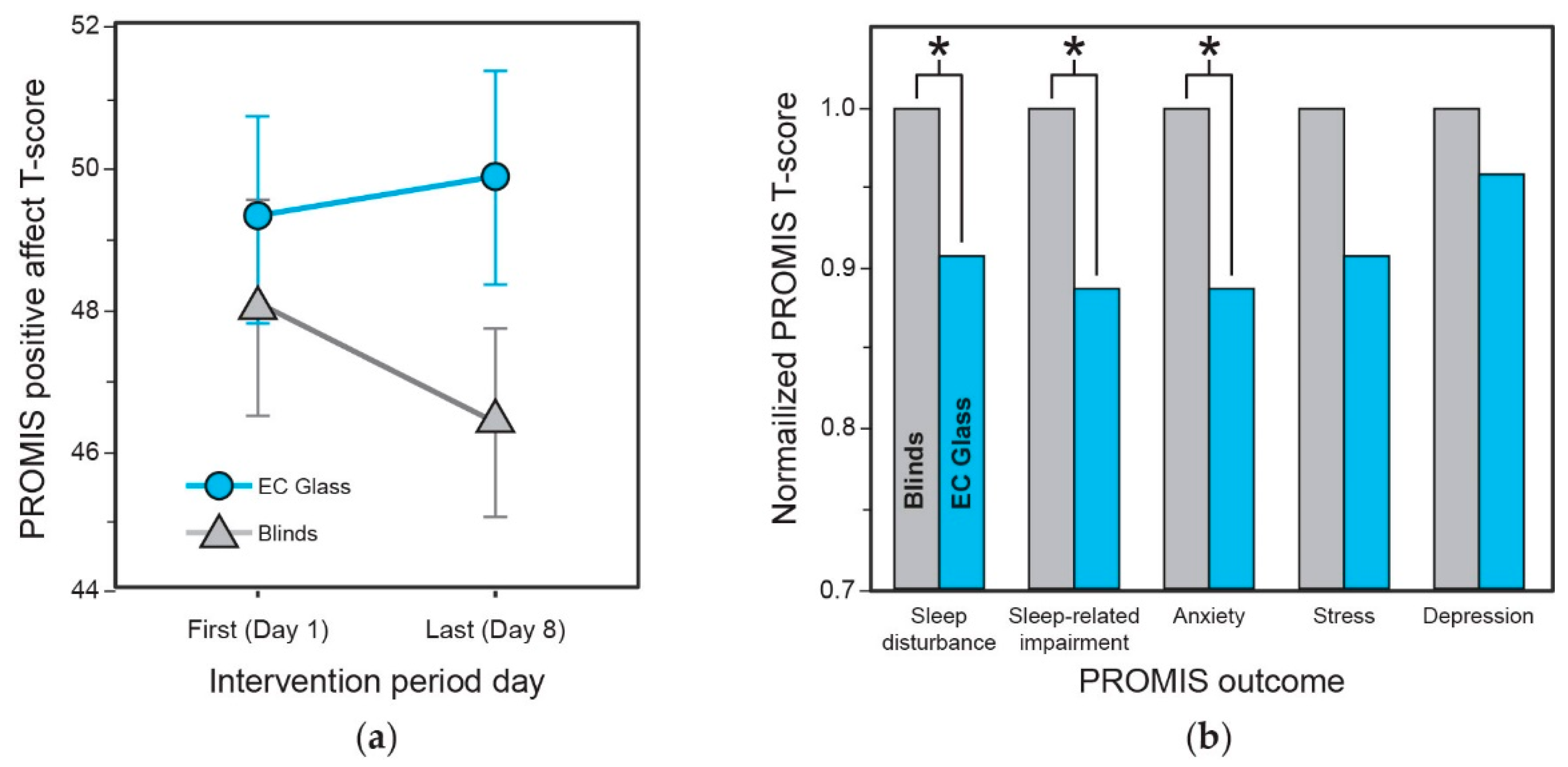
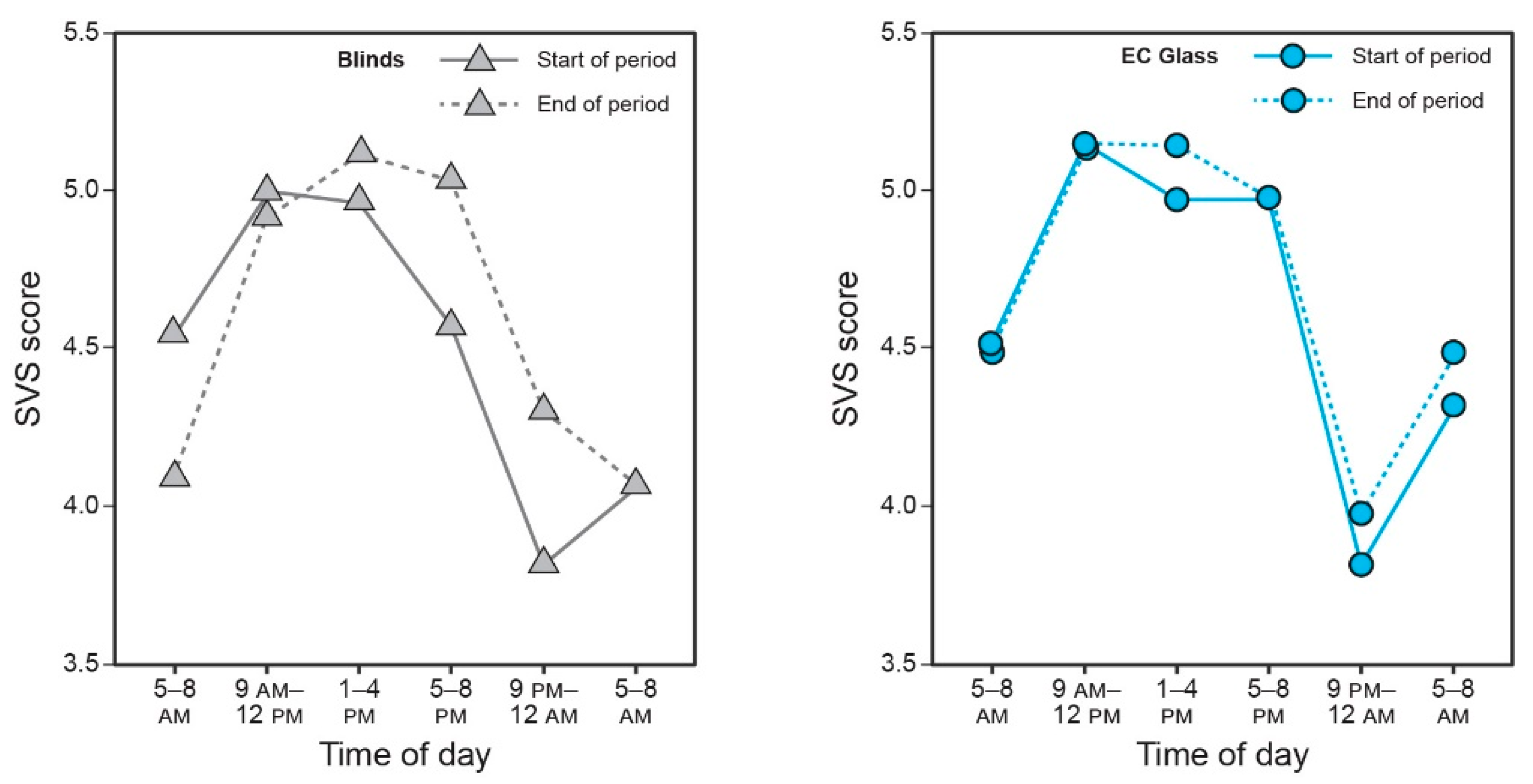
3.7. Subjective Mental Health and Vitality
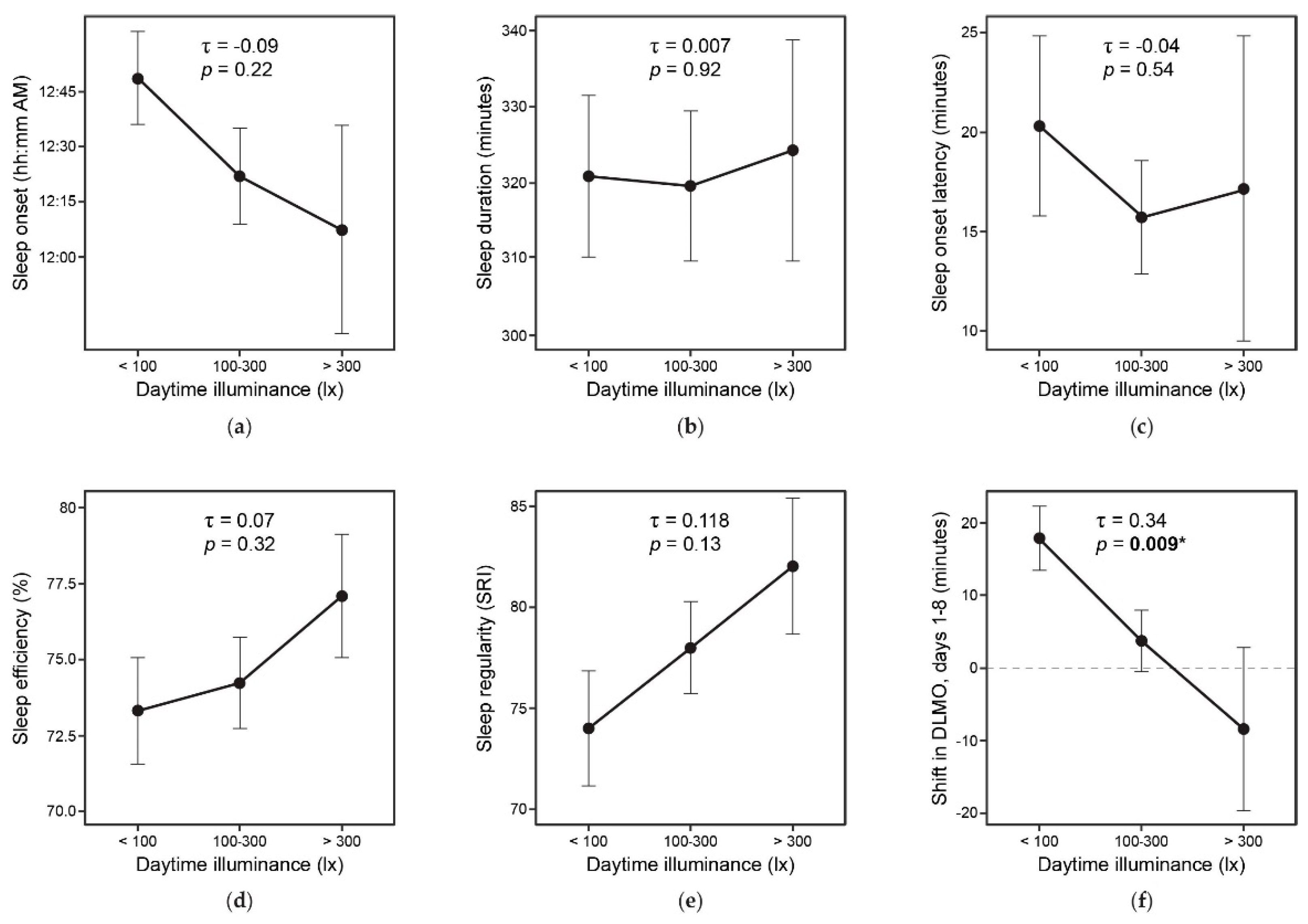
3.8. Daylight and Study Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Potter, G.D.; Skene, D.J.; Arendt, J.; Cade, J.E.; Grant, P.J.; Hardie, L.J. Circadian rhythm and sleep disruption: Causes, metabolic consequences, and countermeasures. Endocr. Rev. 2016, 37, 584–608. [Google Scholar] [CrossRef] [Green Version]
- Gillette, M.U.; Tischkau, S.A. Suprachiasmatic nucleus: The brain’s circadian clock. Recent Prog. Horm. Res. 1999, 54, 33–59. [Google Scholar]
- Deboer, T. Sleep homeostasis and the circadian clock: Do the circadian pacemaker and the sleep homeostat influence each other’ s functioning? Neurobiol. Sleep Circadian Rhythm. 2018, 5, 68–77. [Google Scholar] [CrossRef]
- Gharaveis, A.; Yekita, H.; Shamloo, G. The perceptions of nurses about the behavioral needs for daylighting and view to the outside in inpatient facilities. HERD 2020, 13, 191–205. [Google Scholar] [CrossRef]
- Gharaveis, A.; Shepley, M.M.; Gaines, K. The role of daylighting in skilled nursing short-term rehabilitation facilities. HERD 2016, 9, 105–118. [Google Scholar] [CrossRef]
- Figueiro, M.G.; Steverson, B.; Heerwagen, J.; Kampschroer, K.; Hunter, C.M.; Gonzales, K.; Plitnick, B.; Rea, M.S. The impact of daytime light exposures on sleep and mood in office workers. Sleep Health 2017, 3, 204–215. [Google Scholar] [CrossRef] [PubMed]
- Boubekri, M.; Cheung, I.N.; Reid, K.J.; Wang, C.H.; Zee, P.C. Impact of windows and daylight exposure on overall health and sleep quality of office workers: A case-control pilot study. J. Clin. Sleep Med. 2014, 10, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Jarboe, C.; Figueiro, M. More Daytime Light = Better Nighttime Sleep. Available online: https://issuu.com/designinglighting/docs/designing_lighting_inaugural_edition/s/10913761 (accessed on 6 July 2021).
- Baloch, R.M.M.; Maesano, C.N.; Christoffersen, J.; Mandin, C.; Csobod, E.; Fernandes, E.d.O.; Annesi-Maesano, I.; Consortium, O.B.O.T.S. Daylight and School Performance in European Schoolchildren. Int. J. Environ. Res. Public Health 2020, 18, 258. [Google Scholar] [CrossRef]
- Van Den Wymelenberg, K. Patterns of occupant interaction with window blinds: A literature review. Energ. Build. 2012, 51, 165–176. [Google Scholar] [CrossRef]
- Rea, M.S. Window blind occlusion: A pilot study. Build. Environ. 1984, 19, 133–137. [Google Scholar] [CrossRef] [Green Version]
- Spitschan, M.; Aguirre, G.K.; Brainard, D.H.; Sweeney, A.M. Variation of outdoor illumination as a function of solar elevation and light pollution. Sci. Rep. 2016, 6, 26756. [Google Scholar] [CrossRef] [PubMed]
- Jamrozik, A.; Clements, N.; Hasan, S.S.; Zhao, J.; Zhang, R.; Campanella, C.; Loftness, V.; Porter, P.; Ly, S.; Wang, S.; et al. Access to daylight and view in an office improves cognitive performance and satisfaction and reduces eyestrain: A controlled crossover study. Build. Environ. 2019, 165, 106379. [Google Scholar] [CrossRef]
- Ware, J.E., Jr.; Sherbourne, C.D. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med. Care 1992, 30, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Roenneberg, T.; Wirz-Justice, A.; Merrow, M. Life between clocks: Daily temporal patterns of human chronotypes. J. Biol. Rhythm. 2003, 18, 80–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2000, 284, 3043–3045. [Google Scholar] [CrossRef]
- Portaluppi, F.; Touitou, Y.; Smolensky, M.H. Ethical and methodological standards for laboratory and medical biological rhythm research. Chronobiol. Int. 2008, 25, 999–1016. [Google Scholar] [CrossRef]
- Bierman, A.; Klein, T.R.; Rea, M.S. The Daysimeter: A device for measuring optical radiation as a stimulus for the human circadian system. Meas. Sci. Technol. 2005, 16, 2292–2299. [Google Scholar] [CrossRef]
- Phillips, A.J.K.; Clerx, W.M.; O’Brien, C.S.; Sano, A.; Barger, L.K.; Picard, R.W.; Lockley, S.W.; Klerman, E.B.; Czeisler, C.A. Irregular sleep/wake patterns are associated with poorer academic performance and delayed circadian and sleep/wake timing. Sci. Rep. 2017, 7, 3216. [Google Scholar] [CrossRef]
- Miller, D.; Figueiro, M.G.; Bierman, A.; Schernhammer, E.; Rea, M.S. Ecological measurements of light exposure, activity and circadian disruption. Lighting Res. Technol. 2010, 42, 271–284. [Google Scholar] [CrossRef] [Green Version]
- Rea, M.S.; Figueiro, M.G.; Bullough, J.D.; Bierman, A. A model of phototransduction by the human circadian system. Brain Res. Rev. 2005, 50, 213–228. [Google Scholar] [CrossRef]
- Rea, M.S.; Figueiro, M.G.; Bierman, A.; Hamner, R. Modelling the spectral sensitivity of the human circadian system. Lighting Res. Technol. 2012, 44, 386–396. [Google Scholar] [CrossRef]
- Rea, M.S.; Figueiro, M.G.; Bierman, A.; Bullough, J.D. Circadian light. J. Circadian Rhythm. 2010, 8, 2. [Google Scholar] [CrossRef] [Green Version]
- Smith, M.R.; Revell, V.L.; Eastman, C.I. Phase advancing the human circadian clock with blue-enriched polychromatic light. Sleep Med. 2009, 10, 287–294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cella, D.; Riley, W.; Stone, A.; Rothrock, N.; Reeve, B.; Yount, S.; Amtmann, D.; Bode, R.; Buysse, D.; Choi, S. The Patient-Reported Outcomes Measurement Information System (PROMIS) developed and tested its first wave of adult self-reported health outcome item banks: 2005–2008. J. Clin. Epidemiol. 2010, 63, 1179–1194. [Google Scholar] [CrossRef] [Green Version]
- Quach, C.W.; Langer, M.M.; Chen, R.C.; Thissen, D.; Usinger, D.S.; Emerson, M.A.; Reeve, B.B. Reliability and validity of PROMIS measures administered by telephone interview in a longitudinal localized prostate cancer study. Qual. Life. Res. 2016, 25, 2811–2823. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, S.J.; Orbai, A.; Duncan, T.; DeLeon, E.; Ruffing, V.; Clegg-Smith, K.; Bingham, C.O. Reliability and validity of selected PROMIS measures in people with rheumatoid arthritis. PLoS ONE. 2015, 10, e0138543. [Google Scholar] [CrossRef] [PubMed]
- Riley, W.T.; Rothrock, N.; Bruce, B.; Christodolou, C.; Cook, K.; Hahn, E.A.; Cella, D. Patient-reported outcomes measurement information system (PROMIS) domain names and definition revisions: Further evaluation of content validity in IRT-derived item banks. Qual. Life. Res. 2010, 19, 1311–1321. [Google Scholar] [CrossRef] [Green Version]
- DeWalt, D.A.; Rothrock, N.; Yount, S.; Stone, A.A. Evaluation of item candidates: The PROMIS Qualitative Item Review. Med. Care. 2007, 45, S12–S21. [Google Scholar] [CrossRef] [Green Version]
- Northwestern University. PROMIS. Available online: https://www.healthmeasures.net/explore-measurement-systems/promis (accessed on 1 April 2021).
- Ryan, R.M.; Frederick, C. On energy, personality, and health: Subjective vitality as a dynamic reflection of well-being. J. Pers. 1997, 65, 529–565. [Google Scholar] [CrossRef]
- Figueiro, M.G.; Kalsher, M.; Steverson, B.C.; Heerwagen, J.; Kampschroer, K.; Rea, M.S. Circadian-effective light and its impact on alertness in office workers. Lighting Res. Technol. 2019, 51, 171–183. [Google Scholar] [CrossRef]
- Bostic, T.J.; Rubio, D.M.; Hood, M. A validation of the Subjective Vitality Scale using structural equation modeling. Soc. Indic. Res. 2000, 52, 313–324. [Google Scholar] [CrossRef]
- Blume, C.; Garbazza, C.; Spitschan, M. Effects of light on human circadian rhythms, sleep and mood. Somnologie 2019, 23, 147–156. [Google Scholar] [CrossRef] [Green Version]
- Figueiro, M.G.; Nagare, R.; Price, L.L.A. Non-visual effects of light: How to use light to promote circadian entrainment and elicit alertness. Lighting Res. Technol. 2018, 50, 38–62. [Google Scholar] [CrossRef] [PubMed]
- Mishima, K.; Okawa, M.; Shimizu, T.; Hishikawa, Y. Diminished melatonin secretion in the elderly caused by insufficient environmental illumination. J. Clin. Endocrinol. Metab. 2001, 86, 129–134. [Google Scholar] [CrossRef]
- Viola, A.U.; James, L.M.; Schlangen, L.J.; Dijk, D.J. Blue-enriched white light in the workplace improves self-reported alertness, performance and sleep quality. Scand. J. Work Environ. Health 2008, 34, 297–306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Figueiro, M.G.; Rea, M.S. Office lighting and personal light exposures in two seasons: Impact on sleep and mood. Lighting Res. Technol. 2016, 48, 352–364. [Google Scholar] [CrossRef]
- Roenneberg, T.; Merrow, M. Entrainment of the human circadian clock. Cold Spring Harb. Symp. Quant. Biol. 2007, 72, 293–299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- National Institute of Neurological Disorders and Stroke. Brain Basics: Understanding Sleep. Available online: https://www.ninds.nih.gov/Disorders/Patient-Caregiver-Education/Understanding-Sleep (accessed on 26 May 2021).
- Cai, D.J.; Mednick, S.A.; Harrison, E.M.; Kanady, J.C.; Mednick, S.C. REM, not incubation, improves creativity by priming associative networks. Proc. Natl. Acad. Sci. USA 2009, 106, 10130–10134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boubekri, M.; Lee, J.; MacNaughton, P.; Woo, M.; Schuyler, L.; Tinianov, B.; Satish, U. The Impact of Optimized Daylight and Views on the Sleep Duration and Cognitive Performance of Office Workers. Int. J. Env. Res. Public Health 2020, 17, 3219. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.A.; Flynn-Evans, E.E.; Aeschbach, D.; Brainard, G.C.; Czeisler, C.A.; Lockley, S.W. Diurnal spectral sensitivity of the acute alerting effects of light. Sleep 2014, 37, 271–281. [Google Scholar] [CrossRef] [Green Version]
- Mullington, J.M.; Haack, M.; Toth, M.; Serrador, J.M.; Meier-Ewert, H.K. Cardiovascular, inflammatory, and metabolic consequences of sleep deprivation. Prog. Cardiovasc. Dis. 2009, 51, 294–302. [Google Scholar] [CrossRef] [Green Version]
- Cincin, A.; Sari, I.; Sunbul, M.; Kepez, A.; Oguz, M.; Sert, S.; Sahin, A.; Ozben, B.; Tigen, K.; Basaran, Y. Effect of acute sleep deprivation on left atrial mechanics assessed by three-dimensional echocardiography. Sleep Breath. 2016, 20, 227–235. [Google Scholar] [CrossRef]
- Qian, J.; Dalla Man, C.; Morris, C.J.; Cobelli, C.; Scheer, F. Differential effects of the circadian system and circadian misalignment on insulin sensitivity and insulin secretion in humans. Diabetes Obes. Metab. 2018, 20, 2481–2485. [Google Scholar] [CrossRef] [PubMed]
- Davies, S.K.; Ang, J.E.; Revell, V.L.; Holmes, B.; Mann, A.; Robertson, F.P.; Cui, N.; Middleton, B.; Ackermann, K.; Kayser, M.; et al. Effect of sleep deprivation on the human metabolome. Proc. Natl. Acad. Sci. USA 2014, 111, 10761–10766. [Google Scholar] [CrossRef] [Green Version]
- Arble, D.M.; Bass, J.; Behn, C.D.; Butler, M.P.; Challet, E.; Czeisler, C.; Depner, C.M.; Elmquist, J.; Franken, P.; Grandner, M.A.; et al. Impact of Sleep and Circadian Disruption on Energy Balance and Diabetes: A summary of workshop discussions. Sleep 2015, 38, 1849–1860. [Google Scholar] [CrossRef]
- Alhola, P.; Polo-Kantola, P. Sleep deprivation: Impact on cognitive performance. Neuropsychiatr. Dis. Treat. 2007, 3, 553–567. [Google Scholar]
- Walker, W.H.; Walton, J.C.; DeVries, A.C.; Nelson, R.J. Circadian rhythm disruption and mental health. Transl. Psychiatry 2020, 10, 28. [Google Scholar] [CrossRef] [Green Version]
- Bedrosian, T.A.; Nelson, R.J. Timing of light exposure affects mood and brain circuits. Transl. Psychiatry 2017, 7, e1017. [Google Scholar] [CrossRef]
- Frank, E.; Sidor, M.M.; Gamble, K.L.; Cirelli, C.; Sharkey, K.M.; Hoyle, N.; Tikotzky, L.; Talbot, L.S.; McCarthy, M.J.; Hasler, B.P. Circadian clocks, brain function, and development. Ann. N. Y. Acad. Sci. 2013, 1306, 43–67. [Google Scholar] [CrossRef]
- Fang, Y.; Forger, D.B.; Frank, E.; Sen, S.; Goldstein, C. Day-to-day variability in sleep parameters and depression risk: A prospective cohort study of training physicians. NPJ Digit. Med. 2021, 4, 28. [Google Scholar] [CrossRef]
- Huang, T.; Redline, S. Cross-sectional and prospective associations of actigraphy-assessed sleep regularity with metabolic abnormalities: The multi-ethnic study of atherosclerosis. Diabetes Care 2019, 42, 1422–1429. [Google Scholar] [CrossRef]
- Huang, T.; Mariani, S.; Redline, S. Sleep irregularity and risk of cardiovascular events. J. Am. Coll. Cardiol. 2020, 75, 991–999. [Google Scholar] [CrossRef] [PubMed]
- Savvidis, C.; Koutsilieris, M. Circadian rhythm disruption in cancer biology. Mol. Med. 2012, 18, 1249–1260. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). International Agency for Research on Cancer (IARC). Preamble. IARC Monogr. Identif. Carcinog. Hazards Hum. 2019, 7, 1–41. [Google Scholar]
- Figueiro, M.G. Disruption of circadian rhythms by light during day and night. Curr. Sleep Med. Rep. 2017, 3, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Donehower, L.A.; Herron, A.J.; Moore, D.D.; Fu, L. Disrupting circadian homeostasis of sympathetic signaling promotes tumor development in mice. PLoS ONE 2010, 5, e10995. [Google Scholar] [CrossRef] [Green Version]
- De Bundel, D.; Gangarossa, G.; Biever, A.; Bonnefont, X.; Valjent, E. Cognitive dysfunction, elevated anxiety, and reduced cocaine response in circadian clock-deficient cryptochrome knockout mice. Front. Behav. Neurosci. 2013, 7, 152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aschoff, J. Circadian rhythms in man. Science 1965, 148, 1427–1432. [Google Scholar] [CrossRef] [Green Version]
- Aschoff, J. Human circadian rhythms in activity, body temperature and other functions. Life Sci. Space Res. 1967, 5, 159–173. [Google Scholar]
- Figueiro, M.G.; Plitnick, B.; Roohan, C.; Sahin, L.; Kalsher, M.; Rea, M.S. Effects of a tailored lighting intervention on sleep quality, rest–activity, mood, and behavior in older adults with Alzheimer’s disease and related dementias: A randomized clinical trial. J. Clin. Sleep Med. 2019, 15, 1757–1767. [Google Scholar] [CrossRef] [Green Version]
- Klepeis, N.E.; Nelson, W.C.; Ott, W.R.; Robinson, J.P.; Tsang, A.M.; Switzer, P.; Behar, J.V.; Hern, S.C.; Engelmann, W.H. The National Human Activity Pattern Survey (NHAPS): A resource for assessing exposure to environmental pollutants. J. Expo. Sci. Environ. Epidemiol. 2001, 11, 231–252. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Attribute | Variable | n |
|---|---|---|
| Sex | Female | 11 (55%) |
| Male | 8 (40%) | |
| Non-binary | 1 (5%) | |
| Age | Mean (SD) | 35 (13) |
| Median (Min, Max) | 33 (21, 77) | |
| Race | Asian | 2 (10%) |
| Black | 3 (15%) | |
| Hispanic/Latino/Spanish | 3 (15%) | |
| Multiracial | 3 (15%) | |
| White | 9 (45%) | |
| Education | High school graduate | 2 (10%) |
| Some college | 1 (5%) | |
| College degree | 9 (45%) | |
| Graduate degree | 8 (40%) | |
| Income | $25 K–$75 K | 2 (10%) |
| $75–$100 K | 4 (20%) | |
| $100–$125 K | 4 (20%) | |
| $125–$150 K | 2 (10%) | |
| >$150 K | 5 (25%) | |
| Prefer not to answer | 3 (15%) | |
| Employment | No | 5 (25%) |
| Yes | 15 (75%) | |
| General health | Poor | — |
| Fair | 1 (5%) | |
| Good | 4 (20%) | |
| Very good | 11 (55%) | |
| Excellent | 4 (20%) | |
| Medication use | Allergy | 4 (20%) |
| Anxiety | — | |
| Melatonin | — | |
| Sleep | — | |
| MCTQ a sleep duration (workday) | Mean hours (SD) | 8.2 (0.9) |
| Median hours (Min, Max) | 8.3 (5.8, 9.5) | |
| MCTQ a sleep duration (non-workday) | Mean hours (SD) | 8.1 (1.3) |
| Median hours (Min, Max) | 8.2 h (6.0, 12) | |
| MCTQ score b | Mean (SD) | 4.1 (1.0) |
| Median (Min, Max) | 4.0 (2.0, 5.0) | |
| Slightly early | 2 (10%) | |
| Normal | 3 (15%) | |
| Slightly late | 7 (35%) | |
| Moderately late | 8 (40%) |
| Condition | Parameter (Unit) | Daytime Mean (7 AM to 5 PM) | Daytime Median (7 AM to 5 PM) | ||
|---|---|---|---|---|---|
| Blinds | EC Glass | Blinds | EC Glass | ||
| Light | Illuminance (lx) | 148.7 | 185.3 | 66.2 | 115.7 |
| Circadian stimulus | 0.138 | 0.156 | 0.105 | 0.127 | |
| Melanopic lux | 177.2 | 202.4 | 129.8 | 253.1 | |
| Air Quality | CO2 (ppm) | 795.1 | 776.4 | 695.2 | 670.4 |
| PM2.5 (ug/m3) | 18.4 | 17.5 | 2.9 | 3.3 | |
| Thermal | Temperature (°F) | 70.6 | 71.1 | 70.2 | 71.0 |
| Relative humidity (%) | 37.9 | 39.3 | 37. | 38.6 | |
| Noise | Noise (dB) | 55.6 | 55.3 | 54.6 | 54.1 |
| Predictor | Difference in Sleep Onset Time (Minutes) | |
| Estimate | p | |
| Intercept | 12:33 AM | 0.215 |
| Condition (EC Glass) | −22.2 | 0.050 |
| Caffeine after 12 PM (yes) | −4.8 | 0.790 |
| Exercise after 5 PM (hours) | −7.2 | 0.788 |
| Alcoholic drinks (number) | 6.0 | 0.344 |
| Medication after 5 PM (yes) | 6.6 | 0.774 |
| Screen duration after 5 PM (hours) | 1.8 | 0.718 |
| Predictor | Duration (min) | Efficiency (%) | Latency (min) | Regularity (SRI) | ||||
|---|---|---|---|---|---|---|---|---|
| Estimate | p-Value | Estimate | p-Value | Estimate | p-Value | Estimate | p-Value | |
| Intercept | 318.18 | <0.001 | 72.47 | <0.001 | 18.86 | 0.003 | 81.63 | <0.001 |
| Condition (EC Glass) | 16.11 | 0.169 | 0.42 | 0.821 | −5.12 | 0.319 | 0.88 | 0.001 |
| Caffeine after 12 PM (yes) | −12.50 | 0.450 | −0.44 | 0.868 | −1.48 | 0.806 | 1.54 | <0.001 |
| Exercise after 5 PM (hours) | 42.71 | 0.113 | −0.36 | 0.933 | 13.25 | 0.239 | −6.09 | <0.001 |
| Alcoholic drinks (n) | −5.57 | 0.324 | 0.47 | 0.606 | −1.70 | 0.402 | −0.04 | 0.722 |
| Medication after 5 PM (yes) | −4.20 | 0.823 | −1.12 | 0.717 | 1.65 | 0.792 | −4.23 | <0.001 |
| Screen duration after 5 PM (hours) | −0.40 | 0.934 | 0.52 | 0.499 | 0.51 | 0.745 | −0.59 | <0.001 |
| DLMO Onset Shift (Minutes) | ||
|---|---|---|
| Predictor | Estimate (95% CI) | p |
| Intercept | −16.63 | <0.001 |
| Condition (EC Glass) | −15.07 | 0.027 |
| Caffeine after 12 PM (yes) | −3.95 | 0.317 |
| Exercise after 5 PM (hours) | 2.27 | 0.860 |
| Alcoholic drinks (n) | 2.13 | 0.675 |
| Medication after 5 PM (yes) | 4.04 | 0.614 |
| Screen duration after 5 PM (hours) | 1.43 | 0.486 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagare, R.; Woo, M.; MacNaughton, P.; Plitnick, B.; Tinianov, B.; Figueiro, M. Access to Daylight at Home Improves Circadian Alignment, Sleep, and Mental Health in Healthy Adults: A Crossover Study. Int. J. Environ. Res. Public Health 2021, 18, 9980. https://doi.org/10.3390/ijerph18199980
Nagare R, Woo M, MacNaughton P, Plitnick B, Tinianov B, Figueiro M. Access to Daylight at Home Improves Circadian Alignment, Sleep, and Mental Health in Healthy Adults: A Crossover Study. International Journal of Environmental Research and Public Health. 2021; 18(19):9980. https://doi.org/10.3390/ijerph18199980
Chicago/Turabian StyleNagare, Rohan, May Woo, Piers MacNaughton, Barbara Plitnick, Brandon Tinianov, and Mariana Figueiro. 2021. "Access to Daylight at Home Improves Circadian Alignment, Sleep, and Mental Health in Healthy Adults: A Crossover Study" International Journal of Environmental Research and Public Health 18, no. 19: 9980. https://doi.org/10.3390/ijerph18199980
APA StyleNagare, R., Woo, M., MacNaughton, P., Plitnick, B., Tinianov, B., & Figueiro, M. (2021). Access to Daylight at Home Improves Circadian Alignment, Sleep, and Mental Health in Healthy Adults: A Crossover Study. International Journal of Environmental Research and Public Health, 18(19), 9980. https://doi.org/10.3390/ijerph18199980






