Are the Effects of DES Over? A Tragic Lesson from the Past
Abstract
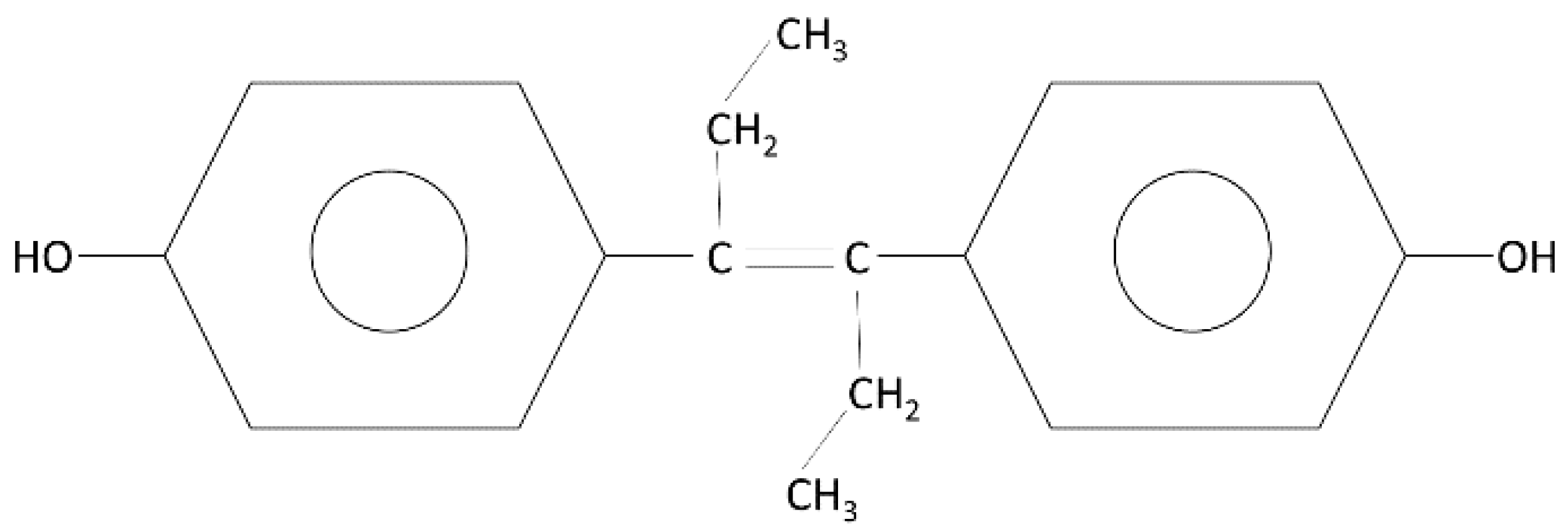
1. Introduction
2. Relevant Health Effects Induced by DES
2.1. DES and Abnormalities in the Reproductive Tract
2.2. DES and Female Lower Genital Tract Cancer
2.3. DES and Breast Cancer
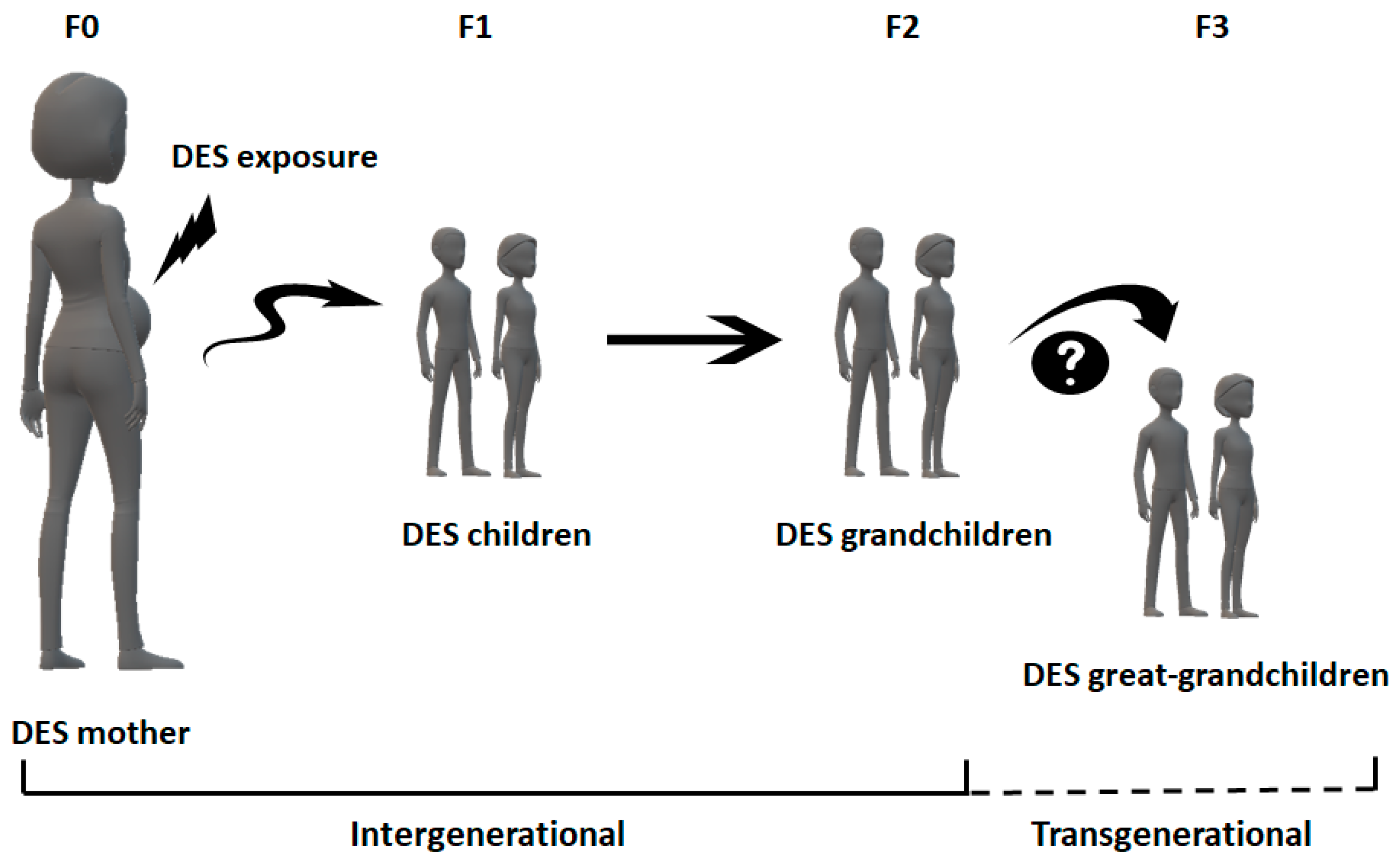
2.4. DES and the Third Generation
3. Discussion
4. Conclusions
Funding
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Dodds, E.; Goldberg, L.; Lawson, W.; Robinson, R. OEstrogenic Activity of Certain Synthetic Compounds. Nature 1938, 141, 247–248. [Google Scholar] [CrossRef]
- Abboud, A. Diethylstilbestrol (DES) in the US. In Embryo Project Encyclopedia; 23 3 2015; ISSN 1940-5030. Available online: http://embryo.asu.edu/handle/10776/8305 (accessed on 3 May 2021).
- Bjorkman, S.; Taylor, H.S. Diethylstilbestrol (DES). Encycl. Reprod. 2018, 2, 760–766. [Google Scholar] [CrossRef]
- Eve, L.; Fervers, B.; Le Romancer, M.; Etienne-Selloum, N. Exposure to endocrine disrupting chemicals and risk of breast cancer. Int. J. Mol. Sci. 2020, 21, 9139. [Google Scholar] [CrossRef] [PubMed]
- IARC Monographs on the Evaluation of Carcinogenic to Humans: Diethylstilbestrol. Int. Agency Res. Cancer 2012, 100, 175–218.
- Dieckmann, W.; Davis, M.; Rynkiewicz, L.; Pottinger, R. Does the administration of diethylstilbestrol during pregnancy have therapeutic value? Am. J. Obstet. Gynecol. 1953, 66, 1062–1081. [Google Scholar] [CrossRef]
- Ibarreta, D.; Swan, S.H. The DES story: Long-term consequences of prenatal exposure. In Precautionary Princ. 20th Century Late Lessons from Early Warnings, 1st ed.; Harremoes, P., Gee, D., MacGarvin, M., Stirling, A., Keys, J., Wynne, B., Guedes Vaz, S., Eds.; Earthscan Publications Ltd.: London, UK, 2002; pp. 90–99. [Google Scholar]
- Reed, C.E.; Fenton, S.E. Exposure to diethylstilbestrol during sensitive life stages: A legacy of heritable health effects. Birth Defects Res. Part C Embryo Today Rev. 2013, 99, 134–146. [Google Scholar] [CrossRef]
- Giusti, R.M.; Iwamoto, K.; Hatch, E.E. Diethylstilbestrol revisited: A review of the long-term health effects. Ann. Intern. Med. 1995, 122, 778–788. [Google Scholar] [CrossRef] [PubMed]
- Peethambaram, P.P.; Ingle, J.N.; Suman, V.J.; Hartmann, L.C.; Loprinzi, C.L. Randomized trial of diethylstilbestrol vs. tamoxifen in postmenopausal women with metastatic breast cancer. An updated analysis. Breast Cancer Res. Treat. 1999, 54, 117–122. [Google Scholar] [CrossRef]
- Ali, A.; Khalil, M.A.I.; Khan, N.; Abu Bakar, M.; Amjad, A.; Ahmed, I.; Mir, K. Diethylstilbestrol in the Treatment of Castration-resistant Prostate Cancer: A Lower-middle-income Country Experience. Cureus 2019, 11, 4–10. [Google Scholar] [CrossRef]
- Noller, K.L.; Fish, C.R. Diethylstilbestrol usage: Its interesting past, important present, and questionable future. Med. Clin. N. Am. 1974, 58, 793–810. [Google Scholar] [CrossRef]
- Camp, A.A.; Couch, J.R.; Quisenberry, J.H. Response to Diethylstilbestrol Injections of Growing Chicks Fed Various Protein Levels. Poult. Sci. 1957, 36, 171–178. [Google Scholar] [CrossRef]
- Giblin, B.T. Synthetic Hormone Use In Beef And The Us Regulatory Dilemma. Law Sch. Stud. Scholarsh. 2013, 476, 1–26. [Google Scholar]
- Raun, A.; Preston, R. History of diethylstilbestrol use in cattle. J. Anim. Sci. 2002, 305, 1–7. [Google Scholar]
- Wahlstrom, R.C. A note on the effect of tylosin and a combination of diethylstilbestrol and methyltestosterone on performance and carcass characteristics of finishing pigs. Anim. Prod. 1970, 12, 181–183. [Google Scholar] [CrossRef]
- Zervos, C.; Rodricks, J.V. FDA’s Ban of DES in Meat Production. Am. Stat. 1982, 36, 278–283. [Google Scholar]
- Loizzo, A.; Gatti, G.L.; Macri, A.; Moretti, G.; Ortolani, E.; Palazzesi, S. The case of diethylstilbestrol treated veal contained in homogenized baby-foods in Italy. Methodological and toxicological aspects. Ann. Ist. Super. Sanita 1984, 20, 215–220. [Google Scholar] [PubMed]
- Bravo, J.C.; Garcinuño, R.M.; Fernández, P.; Durand, J.S. A new molecularly imprinted polymer for the on-column solid-phase extraction of diethylstilbestrol from aqueous samples. Anal. Bioanal. Chem. 2007, 388, 1039–1045. [Google Scholar] [CrossRef] [PubMed]
- Yin, K.; Yu, F.; Liu, D.; Xie, Z.; Chen, L. Cyanine-based colorimetric and fluorescent probe for the selective detection of diethylstilbestrol in seawater, shrimp and fish samples. Sens. Actuators B Chem. 2016, 223, 799–805. [Google Scholar] [CrossRef]
- Solé, M.; De Alda, M.J.L.; Castillo, M.; Porte, C.; Ladegaard-Pedersen, K.; Barceló, D. Estrogenicity determination in sewage treatment plants and surface waters from the catalonian area (NE Spain). Environ. Sci. Technol. 2000, 34, 5076–5083. [Google Scholar] [CrossRef]
- Chen, T.S.; Chen, T.C.; Yeh, K.J.C.; Chao, H.R.; Liaw, E.T.; Hsieh, C.Y.; Chen, K.C.; Hsieh, L.T.; Yeh, Y.L. High estrogen concentrations in receiving river discharge from a concentrated livestock feedlot. Sci. Total Environ. 2010, 408, 3223–3230. [Google Scholar] [CrossRef]
- Lucci, P.; Núñez, O.; Galceran, M.T. Solid-phase extraction using molecularly imprinted polymer for selective extraction of natural and synthetic estrogens from aqueous samples. J. Chromatogr. A 2011, 1218, 4828–4833. [Google Scholar] [CrossRef]
- Xiong, P.; Gan, N.; Cui, H.; Zhou, J.; Cao, Y.; Hu, F.; Li, T. Incubation-free electrochemical immunoassay for diethylstilbestrol in milk using gold nanoparticle-antibody conjugates for signal amplification. Microchim. Acta 2014, 181, 453–462. [Google Scholar] [CrossRef]
- Nazli, B.; Çolak, H.; Aydin, A.; Hampikyan, H. The presence of some anabolic residues in meat and meat products sold in İstanbul. Turkish J. Vet. Anim. Sci. 2005, 29, 691–699. [Google Scholar]
- Metzler, M. The metabolism of diethylstilbestrol. CRC Crit. Rev. Biochem. 1981, 10, 171–212. [Google Scholar] [CrossRef] [PubMed]
- Lagiou, P. Intrauterine exposures, pregnancy estrogens and breast cancer risk: Where do we currently stand? Breast Cancer Res. 2006, 8, 7–8. [Google Scholar] [CrossRef] [PubMed]
- Malone, K.E. Diethylstilbestrol ( DES ) and Breast Cancer. Epidemiol. Rev. 1993, 15, 108–109. [Google Scholar] [CrossRef] [PubMed]
- Ma, L. Endocrine disruptors in female reproductive tract development and carcinogenesis. Trends Endocrinol. Metab. 2009, 20, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, R.H.; Adam, E. Findings in female offspring of women exposed in utero to diethylstilbestrol. Obstet. Gynecol. 2002, 99, 197–200. [Google Scholar] [CrossRef]
- Titus-Ernstoff, L.; Troisi, R.; Hatch, E.E.; Wise, L.A.; Palmer, J.; Hyer, M.; Kaufman, R.; Adam, E.; Strohsnitter, W.; Noller, K.; et al. Menstrual and reproductive characteristics of women whose mothers were exposed in utero to diethylstilbestrol (DES). Int. J. Epidemiol. 2006, 35, 862–868. [Google Scholar] [CrossRef]
- Hoover, R.H.; Hyer, M.; Pfeiffer, R.M.; Adam, E.; Bond, B.; Cheville, A.L.; Colton, T.; Hartge, P.; Hatch, E.E.; Herbst, A.L.; et al. Adverse Health Outcomes in Women Exposed In Utero to Diethylstilbestrol. Obstet. Gynecol. Surv. 2011, 67, 94–96. [Google Scholar] [CrossRef]
- Conlon, J.L. Diethylstilbestrol: Potential health risks for women exposed in utero and their offspring. J. Am. Acad. Physician Assist. 2017, 30, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Guzmán-Arriaga, C.; Zambrano, E. Compuestos disruptores endocrinos y su participación en la programación del eje reproductivo. Rev. Investig. Clin. 2007, 59, 73–81. [Google Scholar]
- Palmer, J.R.; Herbst, A.L.; Noller, K.L.; Boggs, D.A.; Troisi, R.; Titus-Ernstoff, L.; Hatch, E.E.; Wise, L.A.; Strohsnitter, W.C.; Hoover, R.N. Urogenital abnormalities in men exposed to diethylstilbestrol in utero: A cohort study. Environ. Health A Glob. Access Sci. Source 2009, 8, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Tournaire, M.; Devouche, E.; Epelboin, S.; Cabau, A.; Dunbavand, A.; Levadou, A. Birth defects in children of men exposed in utero to diethylstilbestrol (DES). Therapie 2018, 73, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Kilcoyne, K.R.; Mitchell, R.T. Effect of environmental and pharmaceutical exposures on fetal testis development and function: A systematic review of human experimental data. Hum. Reprod. Update 2019, 25, 397–421. [Google Scholar] [CrossRef] [PubMed]
- Goyal, H.O.; Robateau, A.; Braden, T.D.; Williams, C.S.; Srivastava, K.K.; Ali, K. Neonatal estrogen exposure of male rats alters reproductive functions at adulthood. Biol. Reprod. 2003, 68, 2081–2091. [Google Scholar] [CrossRef]
- Rahman, M.M.; Wie, J.; Cho, J.H.; Tae, H.J.; Ahn, D.; Lee, S.W.; Kim, I.S.; Park, B.Y. Diethylstilbestrol induces morphological changes in the spermatogonia, Sertoli cells and Leydig cells of adult rat. Res. Vet. Sci. 2019, 124, 433–438. [Google Scholar] [CrossRef]
- Strohsnitter, W.C.; Noller, K.L.; Hoover, R.N.; Robboy, S.J.; Palmer, J.R.; Titus-Ernstoff, L.; Kaufman, R.H.; Adam, E.; Herbst, A.L.; Hatch, E.E. Cancer risk in men exposed in utero to diethylstilbestrol. J. Natl. Cancer Inst. 2001, 93, 545–551. [Google Scholar] [CrossRef]
- Hom, M.; Sriprasert, I.; Ihenacho, U.; Castelao, J.E.; Siegmund, K.; Bernstein, L.; Cortessis, V.K. Systematic Review and Meta-analysis of Testicular Germ Cell Tumors Following In Utero Exposure to Diethylstilbestrol. JNCI Cancer Spectr. 2019, 3, pkz045. [Google Scholar] [CrossRef]
- Strohsnitter, W.C.; Hyer, M.; Bertrand, K.A.; Cheville, A.L.; Palmer, J.R.; Hatch, E.E.; Aagaard, K.M.; Titus, L.; Romero, I.L.; Huo, D.; et al. Prenatal Diethylstilbestrol Exposure and Cancer Risk in Males. Cancer Epidemiol. Biomark. Prev. 2021. epub ahead. [Google Scholar] [CrossRef]
- Herbst, A.L.; Ulfelder, H.; Poskanzer, D.C. Adenocarcinoma of the vagina: Association of maternal stilbestrol therapy with tumor appearance in young women. N. Engl. J. Med. 1971, 284, 878–881. [Google Scholar] [CrossRef]
- Troisi, R.; Hatch, E.E.; Titus-ernstoff, L.; Hyer, M.; Palmer, J.R.; Robboy, S.J.; Strohsnitter, W.C.; Kaufman, R.; Herbst, A.L.; Hoover, R.N. Cancer risk in women prenatally exposed to diethylstilbestrol. Int. J. Cancer 2007, 121, 356–360. [Google Scholar] [CrossRef] [PubMed]
- Verloop, J.; Van Leeuwen, F.E.; Helmerhorst, T.J.M.; Van Boven, H.H.; Rookus, M.A. Cancer risk in DES daughters. Cancer Causes Control 2010, 21, 999–1007. [Google Scholar] [CrossRef]
- Herbst, A.L. Behavior of estrogen-associated female genital tract cancer and its relation to neoplasia following intrauterine exposure to diethylstilbestrol (DES). Gynecol. Oncol. 2000, 76, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Titus-Ernstoff, L.; Hatch, E.E.; Hoover, R.N.; Palmer, J.; Greenberg, E.R.; Ricker, W.; Kaufman, R.; Noller, K.; Herbst, A.L.; Colton, T.; et al. Long-term cancer risk in women given diethylstilbestrol (DES) during pregnancy. Br. J. Cancer 2001, 84, 126–133. [Google Scholar] [CrossRef]
- Troisi, R.; Hatch, E.E.; Titus, L.; Strohsnitter, W.; Gail, M.H.; Huo, D.; Adam, E.; Robboy, S.J.; Hyer, M.; Hoover, R.N.; et al. Prenatal diethylstilbestrol exposure and cancer risk in women. Environ. Mol. Mutagen. 2019, 60, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Palmer, J.R.; Anderson, D.; Helmrich, S.P.; Herbst, A.L. Risk factors for diethylstilbestrol-associated clear cell adenocarcinoma. Obstet. Gynecol. 2000, 95, 814–820. [Google Scholar] [CrossRef] [PubMed]
- Bromer, J.G.; Wu, J.; Zhou, Y.; Taylor, H.S. Hypermethylation of Homeobox A10 by in Utero Diethylstilbestrol Exposure: An Epigenetic Mechanism for Altered Developmental Programming. Endocrinology 2009, 150, 3376–3382. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Barclay, D.L. Congenital diethylstilbestrol-associated vaginal/cervical adenosis (DES babies). J. Ark. Med. Soc. 1979, 75, 451–452. [Google Scholar]
- Titus-Ernstoff, L.; Troisi, R.; Hatch, E.; Hyer, M.; Wise, L.A.; Palmer, J.R.; Kaufman, R.; Adam, E.; Noller, K.; Herbst, A.L.; et al. Offspring of women exposed in utero to diethylstilbestrol (DES): A preliminary report of benign and malignant pathology in the third generation. Epidemiology 2008, 19, 251–257. [Google Scholar] [CrossRef]
- Greenberg, E.R.; Barnes, A.B.; Resseguie, L.; Barrett, J.A.; Burnside, S.; Lanza, L.L.; Neff, R.K.; Stevens, M.; Young, R.H.; Colton, T. Breast cancer in mothers given diethylstilbestrol in pregnancy. N. Engl. J. Med. 1984, 311, 1393–1398. [Google Scholar] [CrossRef] [PubMed]
- Calle, E.E.; Mervis, C.A.; Thun, M.J.; Rodriguez, C.; Wingo, P.A.; Heath, C.W. Diethylstilbestrol and risk of fatal breast cancer in a prospective cohort of US women. Am. J. Epidemiol. 1996, 144, 645–652. [Google Scholar] [CrossRef]
- Colton, T.; Greenberg, E.R.; Noller, K.; Resseguie, L.; Van Bennekom, C.; Heeren, T.; Zhang, Y. Breast cancer in mothers prescribed diethylstilbestrol in pregnancy: Further follow-up. Obstet. Gynecol. Surv. 1994, 49, 35–37. [Google Scholar] [CrossRef]
- Palmer, J.R.; Hatch, E.E.; Rosenberg, C.L.; Hartge, P.; Kaufman, R.H.; Titus-Ernstoff, L.; Noller, K.L.; Herbst, A.L.; Rao, R.S.; Troisi, R.; et al. Risk of breast cancer in women exposed to diethylstilbestrol in utero: Preliminary results (United States). Cancer Causes Control 2002, 13, 753–758. [Google Scholar] [CrossRef]
- Palmer, J.R.; Wise, L.A.; Hatch, E.E.; Troisi, R.; Titus-Ernstoff, L.; Strohsnitter, W.; Kaufman, R.; Herbst, A.L.; Noller, K.L.; Hyer, M.; et al. Prenatal diethylstilbestrol exposure and risk of breast cancer. Cancer Epidemiol. Biomarkers Prev. 2006, 15, 1509–1514. [Google Scholar] [CrossRef]
- Tournaire, M.; Devouche, E.; Espié, M.; Asselain, B.; Levadou, A.; Cabau, A.; Dunbavand, A.; Grosclaude, P.; Epelboin, S. Cancer Risk in Women Exposed to Diethylstilbestrol in Utero. Therapie 2015, 70, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Strohsnitter, W.C.; Bertrand, K.A.; Troisi, R.; Scott, C.G.; Cheville, A.L.; Hoover, R.N.; Palmer, J.R.; Vachon, C.M. Prenatal diethylstilbestrol exposure and mammographic density. Int. J. Cancer 2018, 143, 1374–1378. [Google Scholar] [CrossRef]
- Kawaguchi, H.; Miyoshi, N.; Miyamoto, Y.; Souda, M.; Umekita, Y.; Yasuda, N.; Yoshida, H. Effects of fetal exposure to diethylstilbestrol on mammary tumorigenesis in rats. J. Vet. Med. Sci. 2009, 71, 1599–1608. [Google Scholar] [CrossRef]
- Rothschild, T.; Boylan, E.S.; Calhoon, R.E.; Vonderhaar, B.K. Transplacental Effects of Diethylstilbestrol on Mammary Development and Tumorigenesis in Female ACI Rats. Cancer Res. 1987, 47, 4508–4516. [Google Scholar]
- Wormsbaecher, C.; Hindman, A.R.; Avendano, A.; Cortes, M.; Bushman, A.; Onua, L.; Kovalchin, C.E.; Murphy, A.R.; Helber, H.L.; Shapiro, A.; et al. In utero estrogenic endocrine disruption alters the stroma to increase extracellular matrix density and mammary gland stiffness. Breast Cancer Res. 2020, 22, 41–53. [Google Scholar] [CrossRef]
- Bern, H.A.; Mills, K.T.; Hatch, D.; Ostrander, P.L.; Iguchi, T. Altered mammary responsiveness to estradiol and progesterone in mice exposed neonatally to diethylstibestrol. Cancer Lett. 1992, 63, 117–124. [Google Scholar] [CrossRef]
- Bern, H.A.; Edery, M.; Mills, K.T.; Kohrman, A.H.; Mori, T.; Larson, L. Long-Term Alterations in Histology and Steroid Receptor Levels of the Genital Tract and Mammary Gland following Neonatal Exposure of Female BALB/cCrgl Mice to Various Doses of Diethylstilbestrol. Cancer Res. 1987, 47, 4165–4172. [Google Scholar]
- Umekita, Y.; Souda, M.; Hatanaka, K.; Hamada, T.; Yoshioka, T.; Kawaguchi, H.; Tanimoto, A. Gene expression profile of terminal end buds in rat mammary glands exposed to diethylstilbestrol in neonatal period. Toxicol. Lett. 2011, 205, 15–25. [Google Scholar] [CrossRef]
- Hilakivi-Clarke, L. Maternal exposure to diethylstilbestrol during pregnancy and increased breast cancer risk in daughters. Breast Cancer Res. 2014, 16, 208. [Google Scholar] [CrossRef] [PubMed]
- Hsu, P.-Y.; Deatherage, D.E.; Rodriguez, B.A.T.; Liyanarachchi, S.; Weng, Y.-I.; Zuo, T.; Liu, J.; Cheng, A.S.L.; Huang, T.H.-M. Xenoestrogen-Induced Epigenetic Repression of microRNA-9-3 in Breast Epithelial Cells. Cancer Res. 2009, 69, 5936–5945. [Google Scholar] [CrossRef]
- Doherty, L.F.; Bromer, J.G.; Zhou, Y.; Aldad, T.S.; Taylor, H.S. In utero exposure to diethylstilbestrol (DES) or bisphenol-A (BPA) increases EZH2 expression in the mammary gland: An epigenetic mechanism linking endocrine disruptors to breast cancer. Horm. Cancer 2010, 1, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Bhan, A.; Hussain, I.; Ansari, K.I.; Bobzean, S.A.M.; Perrotti, L.I.; Mandal, S.S. Bisphenol-A and diethylstilbestrol exposure induces the expression of breast cancer associated long noncoding RNA HOTAIR in vitro and in vivo. J. Steroid Biochem. Mol. Biol. 2014, 141, 160–170. [Google Scholar] [CrossRef]
- Newbold, R.R.; Padilla-Banks, E.; Jefferson, W.N. Adverse effects of the model environmental estrogen diethylstilbestrol are transmitted to subsequent generations. Endocrinology 2006, 147, 11–17. [Google Scholar] [CrossRef]
- Newbold, R.R.; Hanson, R.B.; Jefferson, W.N.; Bullock, B.C.; Haseman, J.; McLachlan, J.A. Increased tumors but uncompromised fertility in the female descendants of mice exposed developmentally to diethylstilbestrol. Carcinogenesis 1998, 19, 1655–1663. [Google Scholar] [CrossRef]
- Walker, B.E. Tumors of female offspring of mice exposed prenatally to diethylstilbestrol. J. Natl. Cancer Inst. 1984, 73, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Newbold, R.R.; Hanson, R.B.; Jefferson, W.N.; Bullock, B.C.; Haseman, J.; McLachlan, J.A. Proliferative lesions and reproductive tract tumors in male descendants of mice exposed developmentally to diethylstilbestrol. Carcinogenesis 2000, 21, 1355–1363. [Google Scholar] [CrossRef]
- Mc Lachlan, J.A. Commentary: Prenatal exposure to diethylstilbestrol (DES): A continuing story. Int. J. Epidemiol. 2006, 35, 868–870. [Google Scholar] [CrossRef] [PubMed]
- Veurink, M.; Koster, M.; Berg, T.W. The history of DES, lessons to be learned. Pharm. World Sci. 2005, 27, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Tournaire, M.; Epelboin, S.; Devouche, E.; Viot, G.; Le Bidois, J.; Cabau, A.; Dunbavand, A.; Levadou, A. Adverse health effects in children of women exposed in utero to diethylstilbestrol (DES). Therapie 2016, 71, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Titus, L.; Hatch, E.E.; Drake, K.M.; Parker, S.E.; Hyer, M.; Palmer, J.R.; Strohsnitter, W.C.; Adam, E.; Herbst, A.L.; Huo, D.; et al. Reproductive and hormone-related outcomes in women whose mothers were exposed in utero to diethylstilbestrol (DES): A report from the US National Cancer Institute DES Third Generation Study. Reprod. Toxicol. 2019, 84, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Gaspari, L.; Cassel-knipping, N.; Villeret, J.; Verschuur, A.; Carcopino-tusoli, X.; Hamamah, S.; Kalfa, N.; Sultan, C. Diethylstilbestrol exposure during pregnancy with primary clear cell carcinoma of the cervix in an 8-year-old granddaughter: A multigenerational effect of endocrine disruptors? Hum. Reprod. 2020, 36, 82–86. [Google Scholar] [CrossRef]
- Blatt, J.; Van Le, L.; Weiner, T.; Sailer, S. Ovarian carcinoma in an adolescent with transgenerational exposure to diethylstilbestrol. J. Pediatr. Hematol. Oncol. 2003, 25, 635–636. [Google Scholar] [CrossRef]
- Wilcox, A.J.; Umbach, D.M.; Hornsby, P.P.; Herbst, A.L. Age at menarche among diethylstilbestrol granddaughters. Am. J. Obstet. Gynecol. 1995, 173, 835–836. [Google Scholar] [CrossRef]
- Kalfa, N.; Paris, F.; Soyer-Gobillard, M.O.; Daures, J.P.; Sultan, C. Prevalence of hypospadias in grandsons of women exposed to diethylstilbestrol during pregnancy: A multigenerational national cohort study. Fertil. Steril. 2011, 95, 2574–2577. [Google Scholar] [CrossRef]
- Klip, H.; Verloop, J.; Van Gool, J.D.; Koster, M.E.T.A.; Burger, C.W.; Van Leeuwen, F.E. Hypospadias in sons of women exposed to diethylstilbestrol in utero: A cohort study. Lancet 2002, 359, 1102–1107. [Google Scholar] [CrossRef]
- Brouwers, M.M.; Feitz, W.F.J.; Roelofs, L.A.J.; Kiemeney, L.A.L.M.; de Gier, R.P.E.; Roeleveld, N. Hypospadias: A transgenerational effect of diethylstilbestrol? Hum. Reprod. 2006, 21, 666–669. [Google Scholar] [CrossRef]
- Ferguson, L.; Agoulnik, A.I. Testicular cancer and cryptorchidism. Front. Endocrinol. (Lausanne) 2013, 4, 1–9. [Google Scholar] [CrossRef]
- Mahawong, P.; Sinclair, A.; Li, Y.; Schlomer, B.; Rodriguez, E.; Ferretti, M.M.; Liu, B.; Baskin, L.S.; Cunha, G.R. Prenatal diethylstilbestrol induces malformation of the external genitalia of male and female mice and persistent second-generation developmental abnormalities of the external genitalia in two mouse strains. Differentiation 2014, 88, 51–69. [Google Scholar] [CrossRef] [PubMed]
- Gaspari, L.; Paris, F.; Hamamah, S.; Kalfa, N.; Sultan, C. “Idiopathic” partial androgen insensitivity syndrome in 11 grandsons of women treated by diethylstilbestrol during gestation: A multi—Generational impact of endocrine disruptor contamination? J. Endocrinol. Investig. 2021, 44, 379–381. [Google Scholar] [CrossRef]
- Kioumourtzoglou, M.A.; Coull, B.A.; O’Reilly, É.J.; Ascherio, A.; Weisskopf, M.G. Association of exposure to diethylstilbestrol during pregnancy with multigenerational neurodevelopmental deficits. JAMA Pediatr. 2018, 172, 670–677. [Google Scholar] [CrossRef]
- Titus-Ernstoff, L.; Troisi, R.; Hatch, E.E.; Palmer, J.R.; Hyer, M.; Kaufman, R.; Adam, E.; Noller, K.; Hoover, R.N. Birth defects in the sons and daughters of women who were exposed in utero to diethylstilbestrol (DES). Int. J. Androl. 2010, 33, 377–384. [Google Scholar] [CrossRef]
- Harris, R.M.; Waring, R.H. Diethylstilboestrol—A long-term legacy. Maturitas 2012, 72, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Al Jishi, T.; Sergi, C. Current perspective of diethylstilbestrol (DES) exposure in mothers and offspring. Reprod. Toxicol. 2017, 71, 71–77. [Google Scholar] [CrossRef]
- Zamora-León, S.P.; Delgado-López, F. Polycyclic aromatic hydrocarbons and their association with breast cancer. Bangladesh J. Med. Sci. 2020, 19, 194–199. [Google Scholar] [CrossRef]
- Delgado-López, F.; Zamora-León, S.P. Breast cancer and environmental contamination: A real connection? Cogent Med. 2018, 5, 1520470. [Google Scholar] [CrossRef]
- De Felici, M.; Sala, G. La Epigenetic Reprogramming in the Mammalian Germ Line: Possible Effects by Endocrine Disruptors on Primordial Germ Cells. Open Biotechnol. J. 2016, 10, 36–41. [Google Scholar] [CrossRef]
- Ben Maamar, M.; Nilsson, E.E.; Skinner, M.K. Epigenetic transgenerational inheritance, gametogenesis and germline development. Biol. Reprod. 2021, ioab085, 1–23. [Google Scholar] [CrossRef]
- Horsthemke, B. A critical view on transgenerational epigenetic inheritance in humans. Nat. Commun. 2018, 9, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Van Cauwenbergh, O.; Di Serafino, A.; Tytgat, J.; Soubry, A. Transgenerational epigenetic effects from male exposure to endocrine-disrupting compounds: A systematic review on research in mammals. Clin. Epigenetics 2020, 12, 1–23. [Google Scholar] [CrossRef]
- Tantitamit, T.; Hamontri, S.; Rangsiratanakul, L. Clear cell adenocarcinoma of the cervix in second generation young women who are without maternal exposure to diethylstilbestrol: A case report. Gynecol. Oncol. Rep. 2017, 20, 34–36. [Google Scholar] [CrossRef] [PubMed]
- Escher, J.; Robotti, S. Pregnancy drugs, fetal germline epigenome, and risks for next-generation pathology: A call to action. Environ. Mol. Mutagen. 2019, 60, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Troisi, R.; Titus, L.; Hatch, E.E.; Palmer, J.R.; Huo, D.; Strohsnitter, W.C.; Adam, E.; Ricker, W.; Hyer, M.; Hoover, R.N. A prospective cohort study of prenatal diethylstilbestrol exposure and cardiovascular disease risk. J. Clin. Endocrinol. Metab. 2018, 103, 206–212. [Google Scholar] [CrossRef]
- Troisi, R.; Hyer, M.; Titus, L.; Palmer, J.R.; Hatch, E.E.; Huo, D.; Aagaard, K.M.; Strohsnitter, W.C.; Hoover, R.N. Prenatal diethylstilbestrol exposure and risk of diabetes, gallbladder disease, and pancreatic disorders and malignancies. J. Dev. Orig. Health Dis. 2021, 12, 619–626. [Google Scholar] [CrossRef]
- National Academies of Sciences, Engineering, and Medicine. Gulf War and Health; The National Academies Press: Washington, DC, USA, 2018; Volume 11, Chapter 3; ISBN 9780309478236. [Google Scholar]
- Skinner, M.K. Endocrine Disruptor Induction of Epigenetic Transgenerational Inheritance of Disease. Mol. Cell Endocrinol. 2014, 398, 4–12. [Google Scholar] [CrossRef]
- Rissman, E.F.; Adli, M. Minireview: Transgenerational Epigenetic Inheritance: Focus on Endocrine Disrupting Compounds. Endocrinology 2014, 155, 2770–2780. [Google Scholar] [CrossRef] [PubMed]
- Walker, B.E.; Haven, M.I. Intensity of multigenerational carcinogenesis from Diethylstilbestrol in mice. Carcinogenesis 1997, 18, 791–793. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Xavier, M.; Roman, S.D.; Aitken, R.J.; Nixon, B. Transgenerational inheritance: How impacts to the epigenetic and genetic information of parents affect offspring health. Hum. Reprod. Update 2019, 25, 518–540. [Google Scholar] [CrossRef] [PubMed]
| Generation Exposed To Des | Human Clinical Effects | Authors | |
|---|---|---|---|
| Female abnormalities in the reproductive tract | DES daughters (F1) and DES granddaughters (germ cells; F2) | 28 DES granddaughters did not show abnormalities in the lower genital tract contrary to their DES mothers (high frequency). | [30] Kaufman et al. |
| DES granddaughters (germ cells; F2) | DES granddaughters with irregular menstrual periods and amenorrhea; no risk of reproductive dysfunction. | [31] Titus-Ernstoff et al. | |
| DES granddaughters (germ cells; F2) | Increased risk of irregular menstrual periods (more common in DES granddaughters of DES mothers with vaginal epithelial changes) and amenorrhea. Possible increased risk of ectopic pregnancy. | [77] Titus et al. | |
| DES granddaughters (F2) | No differences in age at menarche between DES granddaughters and DES daughters. | [80] Wilcox et al. | |
| Female lower genital tract and breast cancers | DES mothers (F0) | Modest increased risk of breast cancer (not aggravated by a family history of breast cancer, oral contraceptives, or hormone replacement therapy). No evidence of a risk for ovarian, endometrial, or other cancers. | [47] Titus-Ernstoff et al. |
| DES mothers (F0) | Moderate increase in the risk of breast cancer (risk rises over time). | [53] Greenberg et al. | |
| DES mothers (F0) | Increase in fatal breast cancers (did not increase over time). | [54] Calle et al. | |
| DES mothers (F0) | Modest increased risk of breast cancer (statistically significant); it did not increase over time. | [55] Colton et al. | |
| DES daughters (F1) | High risk of cervical intraepithelial neoplasia and breast cancer (at 40 years or older). Also, early menopause, infertility, abortion, premature delivery, ectopic pregnancy, stillbirth. The risk was higher in women with vaginal epithelial changes. | [32] Hoover et al. | |
| DES daughters (F1) | Increase in adenocarcinoma of the vagina in young women (cluster of 15–22-year-old women). | [43] Herbst et al. | |
| DES daughters (F1) | Increased risk of CCA of the vagina and cervix, and breast cancer. | [44] Troisi et al. | |
| DES daughters (F1) | Increased risk of CCA of the vagina and cervix; marginally increased risk of melanoma (before age 40). No increased risk of breast cancer (cohort relatively young). | [45] Verloop et al. | |
| DES daughters (F1) | Excess risk of breast cancer, increased risk of lower genital tract malignancies (relatively small absolute risk), and pancreatic cancer. No increased risk of overall cancer. | [48] Troisi et al. | |
| DES daughters (F1) | No postnatal cofactors were identified in association with the risk of developing CAA. | [49] Palmer et al. | |
| DES daughters (F1) | Elevated risk of breast cancer only in women 40 years of age or older. | [56] Palmer et al. | |
| DES daughters (F1) | Increased risk of breast cancer after 40 years old. | [57] Palmer et al. | |
| DES daughters (F1) | Significant increase of breast cancer in women (younger than 40 years) and CCA of the cervix or vagina. No significant increase in overall cancer. | [58] Tournaire et al. | |
| DES daughters (F1) | No association between prenatal exposure to low doses of DES and increased mammographic density in premenopausal or postmenopausal women (did not discard the possibility of an association with higher doses of DES exposure). | [59] Strohsnitter et al. | |
| DES granddaughters (and DES grandsons) (germ cells; F2) | Moderate increase in the risk of breast cancer (risk rises over time). Increased risk of CCA of the vagina and cervix, and higher than expected incidence of ovarian cancer (3 cases). No overall increase of cancer risk in DES grandchildren. | [52] Titus-Ernstoff et al. | |
| DES granddaughter (germ cells; (F2) | Case report of CCA of the vagina and cervix of an 8-year-old girl (with a history of severe vaginal bleeding). DES mother had a hysterectomy. | [78] Gaspari et al. | |
| DES granddaughter (germ cells; F2) | Case report of a 15-year-old girl with small-cell carcinoma of the ovary. | [79] Blatt et al. | |
| Male abnormalities and cancers of the reproductive tract | DES sons (F1) | DES sons showed an increased risk of urogenital abnormalities (strongest association with early gestational exposure). | [35] Palmer et al. |
| DES sons (F1) | No increased risk of overall cancer in DES sons; testicular cancer may be increased in DES sons. | [40] Strohsnitter et al. | |
| DES sons (F1) | Threefold increase in testicular cancer. | [41] Hom et al. | |
| DES sons (F1) | Increased risk of hypospadias (few cases). | [82] Klip et al. | |
| DES sons (F1) | No increase in overall or prostate cancer. Unexpected reduction in the risk of cancers of the urinary system. | [42] Strohsnitter et al. | |
| DES grandsons (and DES granddaughters) (germ cells; F2) | Increased incidence of cryptorchidism and hypoplasia of the penis; no increased incidence of hypospadias. No increase of genital anomalies in girls. All grandchildren were born to DES sons. | [36] Tournaire et al. | |
| DES grandsons (germ cells; F2) | Increase in hypospadias in DES grandsons (born to DES daughters), even though the absolute risk is low; no mutations and no polymorphisms of the AR and MAMLD1 genes were found. Results based on few cases. | [81] Kalfa et al. | |
| DES grandsons (germ cells; F2) | Increased risk of hypospadias when DES grandsons were born to DES daughters but not to DES sons. | [83] Brouwers et al. | |
| DES grandsons (germ cells; F2) | 11 DES grandsons with “idiopathic partial androgen insensitivity-like syndrome”. | [86] Gaspari et al. | |
| Other alterations | DES daughters (F1) | Associations with coronary artery disease and myocardial infarction, but not with stroke. | [99] Troisi et al. |
| DES children (F1) | Increased risk of pancreatic cancer in DES daughters but not in DES sons. | [100] Troisi et al. | |
| DES grandchildren (germ cells; F2) | Increased cerebral palsy, increased defects in lip/palate, esophagus, musculoskeletal and circulatory systems. Also, increased male genital tract anomalies. No significant abnormalities in female genital tract and no increase of breast, uterus, and ovary cancers. | [76] Tournaire et al. | |
| DES grandchildren (germ cells; F2) | Increased ADHD risk (only first trimester of DES exposure during pregnancy). | [87] Kioumourtzoglou et al. | |
| DES grandchildren (germ cells; F2) | Overall birth defects were elevated in grandchildren. Granddaughters appeared to have an increased risk of heart defects. | [88] Titus-Ernstoff et al. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zamora-León, P. Are the Effects of DES Over? A Tragic Lesson from the Past. Int. J. Environ. Res. Public Health 2021, 18, 10309. https://doi.org/10.3390/ijerph181910309
Zamora-León P. Are the Effects of DES Over? A Tragic Lesson from the Past. International Journal of Environmental Research and Public Health. 2021; 18(19):10309. https://doi.org/10.3390/ijerph181910309
Chicago/Turabian StyleZamora-León, Pilar. 2021. "Are the Effects of DES Over? A Tragic Lesson from the Past" International Journal of Environmental Research and Public Health 18, no. 19: 10309. https://doi.org/10.3390/ijerph181910309
APA StyleZamora-León, P. (2021). Are the Effects of DES Over? A Tragic Lesson from the Past. International Journal of Environmental Research and Public Health, 18(19), 10309. https://doi.org/10.3390/ijerph181910309






