Case Fatality as an Indicator for the Human Toxicity of Pesticides—A Systematic Scoping Review on the Availability and Variability of Severity Indicators of Pesticide Poisoning
Abstract
1. Introduction
- For which active ingredients in pesticides or for which group of pesticides have human case- fatality-ratios been published?
- What is the geographical distribution and the variability of the reported case-fatality ratios?
- What is the relationship between the human case-fatality and WHO hazard classes?
- Which factors influence the case-fatality?
- Which severity scores are used with respect to pesticide poisonings?
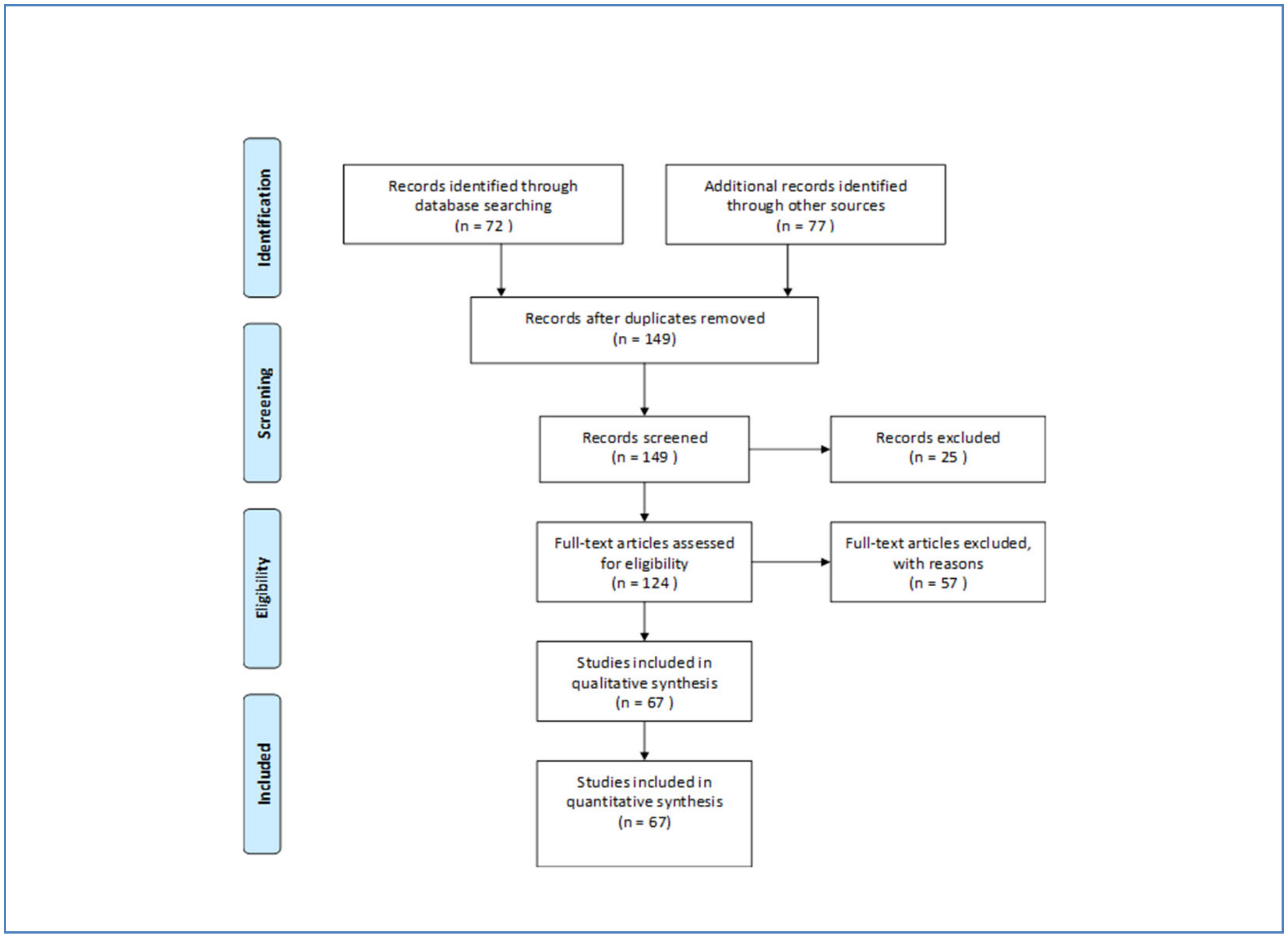
2. Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO—World Health Organization. Public Health Impacts of Pesticides Used in Agriculture. Available online: http://apps.who.int/iris/bitstream/10665/39772/1/9241561394.pdf (accessed on 26 May 2021).
- Boedeker, W.; Watts, M.; Clausing, P.; Marquez, E. The global distribution of acute unintentional pesticide poisoning: Estimations based on a systematic review. BMC Public Health 2020, 20, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Mew, E.J.; Padmanathan, P.; Konradsen, F.; Eddleston, M.; Chang, S.-S.; Phillips, M.R.; Gunnell, D. The global burden of fatal self-poisoning with pesticides 2006–2015: Systematic review. J. Affect. Disord. 2017, 219, 93–104. [Google Scholar] [CrossRef]
- Dinham, B. Communities in Peril: Global Report on Health Impacts of Pesticide Use in Agriculture; Pesticide Action Network Asia Pacific: Penang, Malaysia, 2021; Available online: http://www.pan-germany.org/download/PAN-I_CBM-Global-Report_1006-final.pdf (accessed on 25 May 2021).
- WHO & FAO—World Health Organization and Food and Agriculture Organization. International Code of Conduct on Pesticide Management. Guidelines on Highly Hazardous Pesticides. Available online: http://apps.who.int/iris/bitstream/10665/205561/1/9789241510417_eng.pdf (accessed on 25 May 2021).
- WHO—World Health Organization. WHO Recommended Classification of Pesticides by Hazard and Guideline to Classification. Available online: http://www.who.int/ipcs/publications/pesticides_hazard/en/ (accessed on 25 May 2021).
- Rao, C.S.; Venkateswarlu, V.; Surender, T.; Eddleston, M.; Buckley, N.A. Pesticide poisoning in south India: Opportunities for prevention and improved medical management. Trop. Med. Int. Health 2005, 10, 581–588. [Google Scholar]
- Dawson, A.H.; Eddleston, M.; Senarathna, L.; Mohamed, F.; Gawarammana, I.; Bowe, S.J.; Manuweera, G.; Buckley, N.A. Acute Human Lethal Toxicity of Agricultural Pesticides: A Prospective Cohort Study. PLoS Med. 2010, 7, e1000357. [Google Scholar] [CrossRef]
- Van der Hoek, W.; Konradsen, F. Analysis of 8000 hospital admissions for acute poisoning in a rural area of Sri Lanka. Clin. Toxicol. 2006, 44, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Buckley, N.A.; Fahim, M.; Raubenheimer, J.; Gawarammana, I.B.; Eddleston, M.; Roberts, M.S.; Dawson, A.H. Case fatality of agricultural pesticides after self-poisoning in Sri Lanka: A prospective cohort study. Lancet Glob. Health 2021, 9, e854–e862. [Google Scholar] [CrossRef]
- Oprita, B.; Aignatoaie, B.; Gabor-Postole, D.A. Scores and scales used in emergency medicine. Pract. Toxicol. J. Med. Life 2014, 7, 4–7. [Google Scholar]
- Persson, H.E.; Sjoberg, G.K.; Haines, J.A.; De Pronczuk, G. Poisoning severity score. Grading of acute poisoning. J. Toxicol. Clin. Toxicol. 1998, 36, 205–213. [Google Scholar] [CrossRef]
- Martin, J.; David, L.N. Gray Measurement of Relative Variation: Sociological Examples. Am. Sociol. Rev. 1971, 36, 496. [Google Scholar] [CrossRef]
- Agarwal, S. A Clinical, Biochemical, Neurobehavioral, and Sociopsychological Study of 190 Patients Admitted to Hospital as a Result of Acute Organophosphorus Poisoning. Environ. Res. 1993, 62, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Akdur, O.; Durukan, P.; Özkan, S.; Avsarogullari, L.; Vardar, A.; Kavalci, C.; Ikizceli, I. Poisoning severity score, Glasgow coma scale, corrected QT interval in acute organophosphate poisoning. Hum. Exp. Toxicol. 2010, 29, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Bilgin, T.E.; Camdeviren, H.; Yapici, D.; Doruk, N.; Altunkan, A.A.; Altunkan, Z.; Oral, U. The comparison of the efficacy of scoring systems in organophosphate poisoning. Toxicol. Ind. Health 2005, 21, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Caldas, E.D.; Rebelo, F.M.; Heliodoro, V.O.; Magalhaes, A.F.A.; Rebelo, R.M. Poisonings with pesticides in the Federal District of Brazil. Clin. Toxicol. 2008, 46, 1058–1063. [Google Scholar] [CrossRef]
- Carroll, R.; Metcalfe, C.; Gunnell, D.; Mohamed, F.; Eddleston, M. Diurnal variation in probability of death following self-poisoning in Sri Lanka-evidence for chronotoxicity in humans. Int. J. Epidemiol. 2012, 41, 1821–1828. [Google Scholar] [CrossRef] [PubMed]
- Chaou, C.H.; Lin, C.C.; Chen, H.Y.; Lee, C.H.; Chen, T.H.H. Chlorpyrifos is associated with slower serum cholinesterase recovery in acute organophosphate-poisoned patients. Clin. Toxicol. 2013, 51, 402–408. [Google Scholar] [CrossRef]
- Chen, H.-H.; Lin, J.-L.; Huang, W.-H.; Weng, C.-H.; Lee, S.-Y.; Hsu, C.-W.; Chen, K.-H.; Wang, I.-K.; Liang, C.-C.; Chang, C.-T. Spectrum of corrosive esophageal injury after intentional paraquat or glyphosate-surfactant herbicide ingestion. Int. J. Gen. Med. 2013, 6, 677–683. [Google Scholar] [PubMed]
- Chen, Y.J.; Wu, M.L.; Deng, J.F.; Yang, C.C. The epidemiology of glyphosate-surfactant herbicide poisoning in Taiwan, 1986–2007: A poison center study. Clin. Toxicol. 2009, 47, 670–677. [Google Scholar] [CrossRef] [PubMed]
- Churi, S.; Bhakta, K.; Madhan, R. Organophosphate Poisoning: Prediction of Severity and Outcome by Glasgow Coma Scale, Poisoning Severity Score, Acute Physiology and Chronic Health Evaluation Ii Score, and Simplified Acute Physiology Score Ii. J. Emerg. Nurs. 2012, 38, 493–495. [Google Scholar] [CrossRef]
- Davies, J.O.J.; Eddleston, M.; Buckley, N.A. Predicting outcome in acute organophosphorus poisoning with a poison severity score or the Glasgow coma scale. Qjm. Int. J. Med. 2008, 101, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Deguigne, M.B.; Lagarce, L.; Boels, D.; Harry, P. Metam sodium intoxication: The specific role of degradation products—Methyl isothiocyanate and carbon disulphide—As a function of exposure. Clin. Toxicol. 2011, 49, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Dippenaar, R.; Diedericks, R.J. Paediatric organophosphate poisoning—A rural hospital experience. Samj. S. Afr. Med. J. 2005, 95, 678–681. [Google Scholar]
- Eddleston, M.; Eyer, P.; Worek, F.; Mohamed, F.; Senarathna, L.; von Meyer, L.; Juszczak, E.; Hittarage, A.; Azhar, S.; Dissanayake, W.; et al. Differences between organophosphorus insecticides in human self-poisoning: A prospective cohort study. Lancet 2005, 366, 1452–1459. [Google Scholar] [CrossRef]
- Eddleston, M.; Rajapakshe, M.; Roberts, D.; Reginald, K.; Sheriff, M.H.R.; Dissanayake, W.; Buckley, N. Severe Propanil [N-(3,4-Dichlorophenyl) Propanamide] Pesticide Self-Poisoning. J. Toxicol. Clin. Toxicol. 2002, 40, 847–854. [Google Scholar] [CrossRef] [PubMed]
- Eddleston, M.; Sudarshan, K.; Senthilkumaran, M.; Reginald, K.; Karalliedde, L.; Senarathna, L.; de Silva, D.; Sheriff, M.H.R.; Buckley, N.A.; Gunnell, D. Patterns of hospital transfer for self-poisoned patients in rural Sri Lanka: Implications for estimating the incidence of self-poisoning in the developing world. Bull. World Health Organ. 2006, 84, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Eddleston, M.; Eyer, P.; Worek, F.; Sheriff, M.H.R.; Buckley, N.A. Predicting outcome using butyryl-cholinesterase activity in organophosphorus pesticide self-poisoning. Qjm. Int. J. Med. 2008, 101, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Emerson, G.M.; Gray, M.N.; Jelinek, G.A.; Mountain, D.; Mead, H.J. Organophosphate poisoning in Perth, Western Australia, 1987. J. Emerg. Med. 1999, 17, 273–277. [Google Scholar] [CrossRef]
- Goel, A.; Joseph, S.; Dutta, T.K. Organophosphate poisoning: Predicting the need for ventilatory support. J. Assoc. Physicians India 1998, 46, 786–790. [Google Scholar]
- Grmec, S.; Mally, S.; Klemen, P. Glasgow coma scale score and QTc interval in the prognosis of organophosphate poisoning. Acad. Emerg. Med. 2004, 11, 925–930. [Google Scholar] [CrossRef]
- Hrabetz, H.; Thiermann, H.; Felgenhauer, N.; Zilker, T.; Haller, B.; Nahrig, J.; Saugel, B.; Eyer, F. Organophosphate poisoning in the developed world—A single centre experience from here to the millennium. Chem. Biol. Interact. 2013, 206, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Huang, N.C.; Lin, S.L.; Hung, Y.M.; Hung, S.Y.; Chung, H.M. Severity assessment in acute paraquat poisoning by analysis of APACHE II score. J. Formos. Med. Assoc. 2003, 102, 782–787. [Google Scholar]
- Ikebuchi, J.; Proudfoot, A.T.; Matsubara, K.; Hampson, E.C.G.M.; Tomita, M.; Suzuki, K.; Fuke, C.; Ijiri, I.; Tsu-nerari, T.; Yuasa, I.; et al. Toxicological Index of Paraquat—A New Strategy for Assessment of Severity of Paraquat Poisoning in 128 Patients. Forensic Sci. Int. 1993, 59, 85–87. [Google Scholar] [CrossRef]
- Kumar, S.V.; Venkateswarlu, V.; Sasikala, M.; Kumar, G.V. A study on poisoning cases in a tertiary care hospital. J. Nat. Sci. Biol. Med. 2010, 1, 35–39. [Google Scholar] [CrossRef] [PubMed]
- Lauterbach, M.; Solak, E.; Kaes, J.; Wiechelt, J.; Von Mach, M.A.; Weilemann, L.S. Epidemiology of hydrogen phosphide exposures in humans reported to the poison center in Mainz, Germany, 1983. Clin. Toxicol. 2005, 43, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Shih, C.P.; Hsu, K.H.; Hung, D.Z.; Lin, C.C. The early prognostic factors of glypho-sate-surfactant intoxication. Am. J. Emerg. Med. 2008, 26, 275–281. [Google Scholar] [CrossRef]
- Lee, H.L.; Chen, K.W.; Chi, C.H.; Huang, J.J.; Tsai, L.M. Clinical presentations and prognostic factors of a glyphosate-surfactant herbicide intoxication: A review of 131 cases. Acad. Emerg. Med. 2000, 7, 906–910. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.; Tai, D.Y.H. Clinical features of patients with acute organophosphate poisoning requiring intensive care. Intensive Care Med. 2001, 27, 694–699. [Google Scholar] [CrossRef]
- Lee, Y.; Lee, J.H.; Seong, A.J.; Hong, C.K.; Lee, H.J.; Shin, D.H.; Hwang, S.Y. Arterial lactate as a predictor of mortality in emergency department patients with paraquat intoxication. Clin. Toxicol. 2012, 50, 52–56. [Google Scholar] [CrossRef]
- Levy-Khademi, F.; Tenenbaum, A.N.; Wexler, I.D.; Amitai, Y. Unintentional organophosphate in-toxication in children. Pediatric Emerg. Care 2007, 23, 716–718. [Google Scholar] [CrossRef]
- Lifshitz, M.; Shahak, E.; Bolotin, A.; Sofer, S. Carbamate poisoning in early childhood and in adults. J. Toxicol. Clin. Toxicol. 1997, 35, 25–27. [Google Scholar] [CrossRef]
- Lifshitz, M.; Shahak, E.; Sofer, S. Carbamate and organophosphate poisoning in young children. Pediatr. Emerg. Care 1999, 15, 102–103. [Google Scholar] [CrossRef]
- Lima, J.S.; Reis, C.A.G. Poisoning Due to Illegal Use of Carbamates as a Rodenticide in Rio-De-Janeiro. J. Toxicol. Clin. Toxicol. 1995, 33, 687–690. [Google Scholar] [CrossRef]
- Lo, Y.C.; Yang, C.C.; Deng, J.F. Acute alachlor and butachlor herbicide poisoning. Clin. Toxicol. 2008, 46, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Min, Y.G.; Ahn, J.H.; Chan, Y.C.; Ng, S.H.; Tse, M.L.; Lau, F.L.; Chan, C.K. Prediction of prognosis in acute paraquat poisoning using severity scoring system in emergency department. Clin. Toxicol. 2011, 49, 840–845. [Google Scholar] [CrossRef]
- Mowry, J.B.; Spyker, D.A.; Cantilena, L.R.; Bailey, J.E.; Ford, M. ABSTRACTS 2012 Annual Report of the American Association of Poison Control Centers’ National Poison Data System (NPDS): 30th Annual Report Abstracts. Clin. Toxicol. 2013, 51, 949–1229. [Google Scholar] [CrossRef] [PubMed]
- Murali, R.; Bhalla, A.; Singh, D.; Singh, S. Acute pesticide poisoning: 15 years experience of a large North-West Indian hospital. Clin. Toxicol. 2009, 47, 35–38. [Google Scholar] [CrossRef] [PubMed]
- Nagami, H.; Nishigaki, Y.; Matsushima, S.; Matsushita, T.; Asanuma, S.; Yajima, N.; Usuda, M.; Hirosawa, M. Hospital-based survey of pesticide poisoning in Japan, 1998. Int. J. Occup. Environ. Health 2005, 11, 180–184. [Google Scholar] [CrossRef] [PubMed]
- Nelson, L.S.; Perrone, J.; DeRoos, F.; Stork, C.; Hoffman, R.S. Aldicarb poisoning bay an illicit rodenticide imported into the United States: Tres Pasitos. J. Toxicol. Clin. Toxicol. 2001, 39, 447–452. [Google Scholar] [CrossRef]
- Nisse, P.; Deveaux, M.; Tellart, A.S.; Dherbecourt, V.; Peucelle, D.; Mathieu-Nolf, M. Aldicarb poisoning: Review of the intoxication cases in north of France 1998. Acta Clin. Belg. Supp. 2002, 1, 12–15. [Google Scholar] [CrossRef] [PubMed]
- Peter, J.V.; Thomas, L.; Graham, P.L.; Moran, J.L.; Abhilash, K.P.P.; Jasmine, S.; Iyyadurai, R. Performance of clinical scoring systems in acute organophosphate poisoning. Clin. Toxicol. 2013, 51, 850–854. [Google Scholar] [CrossRef]
- Recena, M.C.P.; Pires, D.X.; Caldas, E.D. Acute poisoning with pesticides in the state of Mato Grosso do Sul, Brazil. Sci. Total Environ. 2006, 357, 88–95. [Google Scholar] [CrossRef]
- Roberts, D.M.; Buckley, N.A.; Mohamed, F.; Eddleston, M.; Goldstein, D.A.; Mehrsheikh, A.; Bleeke, M.S.; Dawson, A.H. A prospective observational study of the clinical toxicology of glyphosate-containing herbicides in adults with acute self-poisoning. Clin. Toxicol. 2010, 48, 129–136. [Google Scholar] [CrossRef]
- Saadeh, A.M.; Alali, M.K.; Farsakh, N.A.; Ghani, M.A. Clinical and sociodemographic features of acute carbamate and organophosphate poisoning: A study of 70 adult patients in North Jordan. J. Toxicol. Clin. Toxicol. 1996, 34, 45–51. [Google Scholar] [CrossRef]
- Senanayake, N.; Desilva, H.J.; Karalliedde, L. A Scale to Assess Severity in Organophosphorus Intoxication—Pop Scale. Hum. Exp. Toxicol. 1993, 12, 297–299. [Google Scholar] [CrossRef] [PubMed]
- Seok, S.J.; Park, J.S.; Hong, J.R.; Gil, H.W.; Yang, J.O.; Lee, E.Y.; Song, H.Y.; Hong, S.Y. Surfactant volume is an essential element in human toxicity in acute glyphosate herbicide intoxication. Clin. Toxicol. 2011, 49, 892–899. [Google Scholar] [CrossRef]
- Seok, S.J.; Choi, S.C.; Gil, H.W.; Yang, J.O.; Lee, E.Y.; Song, H.Y.; Hong, S.Y. Acute Oral Poisoning Due to Chloracetanilide Herbicides. J. Korean Med. Sci. 2012, 27, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Shadnia, S.; Darabi, D.; Pajoumand, A.; Salimi, A.; Abdollahi, M. A simplified acute physiology score in the prediction of acute organophosphate poisoning outcome in an intensive care unit. Hum. Exp. Imental Toxicol. 2007, 26, 623–627. [Google Scholar] [CrossRef] [PubMed]
- Shadnia, S.; Mehrpour, O.; Soltaninejad, K. A simplified acute physiology score in the prediction of acute aluminum phosphide poisoning. Indian J. Med. Sci 2010, 64, 532–539. [Google Scholar]
- Shadnia, S.; Sasanian, G.; Allami, P.; Hosseini, A.; Ranjbar, A.; Amini-Shirazi, N.; Abdollahi, M. A retrospective 7-years study of aluminum phosphide poisoning in Tehran: Opportunities for prevention. Hum. Exp. Toxicol. 2009, 28, 209–213. [Google Scholar] [CrossRef]
- Suh, J.H.; Oh, B.J.; Roh, H.K. Clinical outcomes after suicidal ingestion of glyphosate surfactant herbicide: Severity of intoxication according to amount ingested. Clininal Toxicol. 2007, 45, 641. [Google Scholar]
- Sungur, M.; Guven, M. Intensive care management of organophosphate insecticide poisoning. Crit. Care 2001, 5, 211–215. [Google Scholar] [CrossRef]
- Sungurtekin, H.; Gurses, E.; Balci, C. Evaluation of several clinical scoring tools in organophos-phate poisoned patients. Clin. Toxicol. 2006, 44, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Tagwireyi, D.; Ball, D.E.; Nhachi, C.F.B. Toxicoepidemiology in Zimbabwe: Pesticide poisoning admissions to major hospitals. Clin. Toxicol. 2006, 44, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Talbot, A.R.; Shiaw, M.H.; Huang, J.S.; Yang, S.F.; Goo, T.S.; Wang, S.H.; Chen, C.L.; Sanford, T.R. Acute-Poisoning with A Glyphosate-Surfactant Herbicide (Round-Up)—A Review of 93 Cases. Hum. Exp. Toxicol. 1991, 10, 1–8. [Google Scholar] [CrossRef]
- Tominack, R.L.; Yang, G.Y.; Tsai, W.J.; Chung, H.M.; Deng, J.F. Taiwan-National-Poison-Center Survey of Glyphosate Surfactant Herbicide Ingestions. J. Toxicol. Clin. Toxicol. 1991, 29, 91–109. [Google Scholar] [CrossRef] [PubMed]
- Tsao, T.C.Y.; Juang, Y.C.; Lan, R.S.; Shieh, W.B.; Lee, C.H. Respiratory-Failure of Acute Organophos-phate and Carbamate Poisoning. Chest 1990, 98, 631–636. [Google Scholar] [CrossRef]
- Verhulst, L.; Waggie, Z.; Hatherill, M.; Reynolds, L.; Argent, A. Presentation and outcome of severe anticholinesterase insecticide poisoning. Arch. Dis. Child. 2002, 86, 352–355. [Google Scholar] [CrossRef][Green Version]
- Vucinic, S.; Jovanovic, D.; Antonijevic, B.; Vucinic, Z.; Djordjevic, D.; Potrebic, O.; Rezic, T. Acute carbamate poisoning treated in the National Poison Control Centre during ten years period. Clin. Toxicol. 2009, 47, 506–507. [Google Scholar]
- Weissmann-Brenner, A.; David, A.; Vidan, A.; Hourvitz, A. Organophosphate poisoning: A multi-hospital survey. Isr. Med. Assoc. J. 2002, 4, 573–576. [Google Scholar]
- Weng, C.H.; Hu, C.C.; Lin, J.L.; Lin-Tan, D.T.; Huang, W.H.; Hsu, C.W.; Yen, T.H. Sequential Organ Failure Assessment Score Can Predict Mortality in Patients with Paraquat Intoxication. PLoS ONE 2012, 7, e51743. [Google Scholar] [CrossRef]
- Xia, C.Y.; Wang, M.; Liang, Q.; Yun, L.A.; Kang, H.S.; Fan, L.; Wang, D.S.; Zhang, G.Y. Changes in monoclonal HLA-DR antigen expression in acute organophosphorus pesticide-poisoned patients. Exp. Ther. Med. 2014, 7, 137–140. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, M.; Yamashita, M.; Tanaka, J.; Ando, Y. Human mortality in organophosphate poisonings. Vet. Hum. Toxicol. 1997, 39, 84–85. [Google Scholar] [PubMed]
- Yang, C.C.; Wu, J.F.; Ong, H.C.; Hung, S.C.; Kuo, Y.P.; Sa, C.H.; Chen, S.S.; Deng, J.F. Taiwan National Poison Center: Epidemiologic data 1985. J. Toxicol. Clin. Toxicol. 1996, 34, 651–663. [Google Scholar] [CrossRef] [PubMed]
- Zaheer, M.S.; Aslam, M.; Gupta, V.; Sharma, V.; Khan, S.A. Profile of poisoning cases at A North Indian tertiary care hospital. Health Popul. Perspect. Issues 2009, 32, 176–183. [Google Scholar]
- Clemedson, C.; Crafoord, B.; Ekwall, B.; Hallander, S.; Walum, E.; Bondesson, I. MEIC Evaluation of Acute Systemic Toxicity: Part, V. Rodent and Human Toxicity Data for the 50 Reference Chemicals. Altern. Lab. Anim. 1998, 26, 571–616. [Google Scholar] [CrossRef]
- Dharmani, C.; Jaga, K. Epidemiology of acute organophosphate poisoning in hospital emergency room patients. Rev. Environ. Health 2005, 20, 215–232. [Google Scholar] [CrossRef] [PubMed]
- PAN International List of Highly Hazardous Pesticides. Available online: http://www.pan-germany.org/download/PAN_HHP_List_161212_F.pdf (accessed on 25 May 2021).
- Lee, H.L.; Lin, H.J.; Yeh, S.T.Y.; Chi, C.H.; Guo, H.R. Presentations of patients of poisoning and predictors of poisoning-related fatality: Findings from a hospital-based prospective study. BMC Public Health 2008, 8, 7. [Google Scholar] [CrossRef] [PubMed]
- Moon, J.M.; Chun, J.M. Acute endosulfan poisonings: A retrospective study. Hum. Exp. Ment. Toxicol. 2009, 28, 309–316. [Google Scholar] [CrossRef]
- Eddleston, M. Patterns and problems of deliberate self-poisoning in the developing world. Q J. Med. Int. J. Med. 2000, 93, 1–20. [Google Scholar] [CrossRef]
- Chugh, S.N.; Dushyant Ram, S.; Arora, B.; Malhotra, K.C. Incidence & outcome of aluminium phosphide poisoning in a hospital study. Indian J. Med. Res. 1991, 94, 232–235. [Google Scholar]
- Jones, G.M.; Vale, J.A. Mechanisms of Toxicity, Clinical Features, and Management of Diquat Poisoning: A Review. J. Toxicol. Clin. Toxicol. 2000, 38, 123–128. [Google Scholar] [CrossRef]
- Mohamed, F.; Senarathna, L.; Percy, A.; Abeyewardene, M.; Eaglesham, G.; Cheng, R.; Azher, S.; Hittarage, A.; Dissanayake, W.; Sheriff, R.; et al. Acute Human Self-Poisoning with the N-Phenylpyrazole Insecticide Fipronil—A GABAA-Gated Chloride Channel Blocker. J. Toxicol. Clin. Toxicol. 2004, 42, 955–963. [Google Scholar] [CrossRef] [PubMed]
- Kasiotis, K.M.; Baira, E.; Manea-Karga, E.; Nikolopoulou, D.; Ganas, K.; Machera, K. Investigating a human pesticide intoxication incident: The importance of robust analytical approaches. Open Chem. 2021, 19, 107–118. [Google Scholar] [CrossRef]
- Moebus, S.; Bödeker, W. Mortality of intentional and unintentional pesticide poisonings in Germany from 1980. J. Public Health Policy 2015, 36, 170–180. [Google Scholar] [CrossRef] [PubMed]
- Moebus, S.; Boedeker, W. Frequency and trends of hospital treated pesticide poisonings in Germany 2000. Ger. Med. Sci. 2017, 15, 28860960. [Google Scholar] [CrossRef]
| Group of Pesticide | Publications | Countries | Case-Fatality-Ratio (%) | Cases (n) | Severity Indicator 1 | Country | Reference | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| N | n | Median | Min | Max | Median | Min | Max | ||||
| carbamates | 9 | 6 | 5.1 | 0.0 | 14.2 | 60 | 6 | 1433 | CFR | Brazil, India, Israel, Serbia, Sri Lanka, Taiwan | [7,8,18,28,43,45,69,71,76] |
| carbamates/OP 2 | 1 | 1 | 5.0 | 280 | PSS, CFR | Brazil | [17] | ||||
| chloracetanilide | 1 | 1 | 3.6 | 28 | CFR, PSS | Korea | [58] | ||||
| coumarin | 1 | 1 | 0.0 | 82 | PSS, CFR | Brazil | [17] | ||||
| cyanide | 1 | 1 | 24.1 | CFR | Taiwan | [76] | |||||
| diethyl-OP | 1 | 1 | 38.0 | 8 | CFR, PSS, APACHE, SOFA, GCS | Germany | [33] | ||||
| dimethyl-OP | 1 | 1 | 11.0 | 19 | CFR, PSS, APACHE, SOFA, GCS | Germany | [33] | ||||
| fungicides | 1 | 1 | 6.1 | 49 | CFR | Sri Lanka | [8] | ||||
| herbicides | 1 | 1 | 12.4 | 2783 | CFR | Sri Lanka | [8] | ||||
| organochlorines | 2 | 2 | 18.4 | 16.7 | 20.0 | 112 | 12 | 212 | CFR | India, Sri Lanka | [7,8] |
| organophosphate | 31 | 14 | 11.1 | 2.9 | 73.0 | 94 | 16 | 5226 | CFR, APACHE, PSS, SOFA, GCSCFR, SAPS, CFRCFR, SAPSII | Australia, China, Germany, India, Iran, Israel, Japan, Jordan, Slovenia, SouthAfrica, Sri Lanka, Taiwan, Turkey, Zimbabwe | [7,8,14,15,16,18,19,22,23,25,30,31,32,33,36,42,44,53,56,57,60,64,65,66,69,70,72,74,75,76,77] |
| pyrethrins | 1 | 1 | 0.0 | 5522 | CFR | USA | [48] | ||||
| pyrethroids | 3 | 3 | 0.7 | 0.0 | 1.0 | 203 | 140 | 23,853 | PSS, CFR | Brazil, Sri Lanka, USA | [8,17,48] |
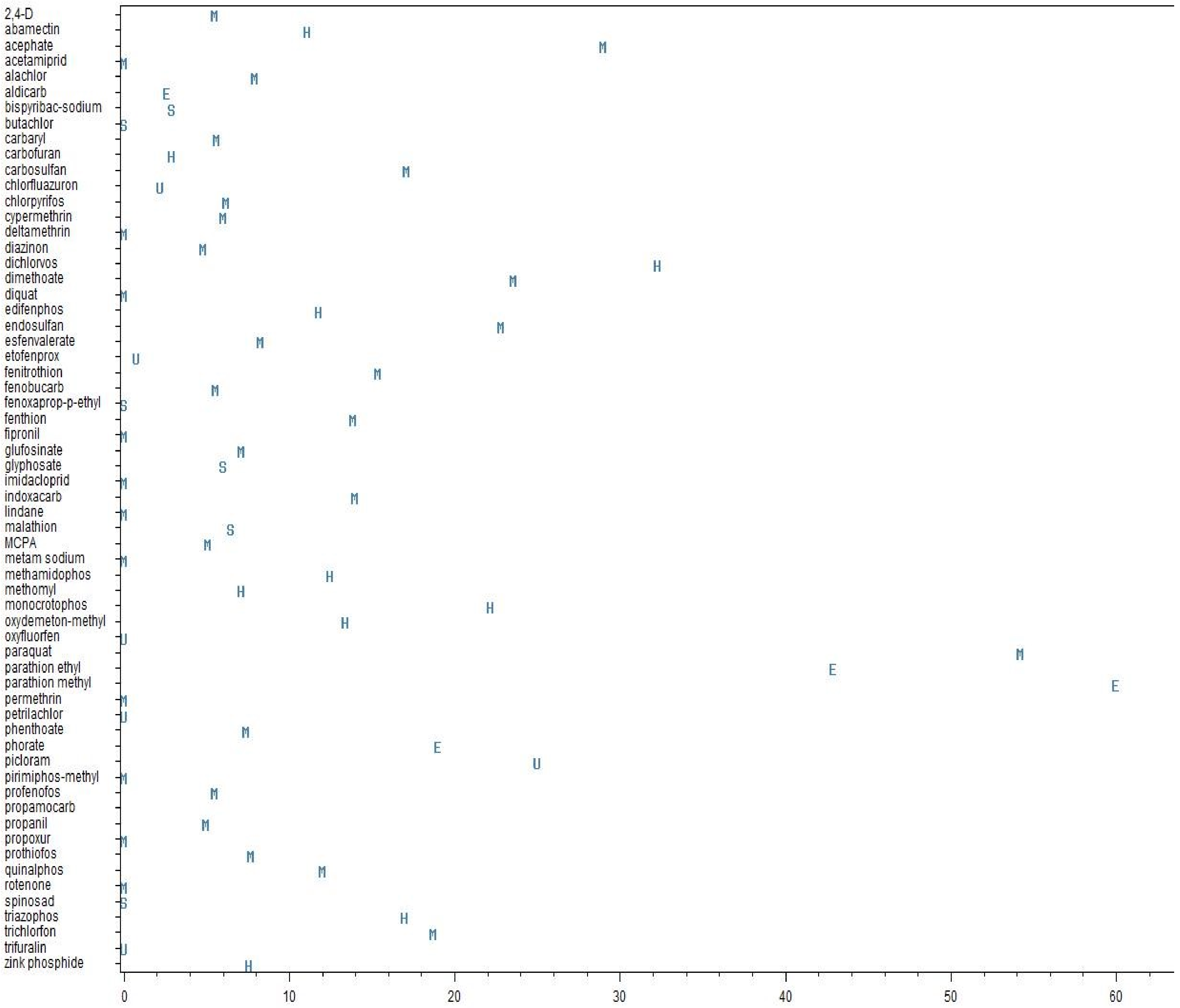
| Active Ingredients | |||||||||||
| 2,4-D | 1 | 1 | 5.5 | 20 | CFR | Brazil | [54] | ||||
| abamectin | 1 | 1 | 11.1 | 18 | CFR | Sri Lanka | [8] | ||||
| acephate | 1 | 1 | 29.0 | 14 | CFR | India | [7] | ||||
| acetamiprid | 1 | 1 | 0.0 | 11 | CFR | Sri Lanka | [8] | ||||
| alachlor | 2 | 2 | 8.0 | 4.8 | 11.1 | 36 | 9 | 63 | CFR | Sri Lanka, Taiwan | [8,46] |
| aldicarb | 2 | 2 | 2.6 | 0.0 | 5.2 | 37 | 35 | 39 | CFR, PSS | France, USA | [51,52] |
| aldrin | 1 | 1 | 13.3 | 49 | CFR | Brazil | [54] | ||||
| aluminium phosphide | 2 | 1 | 48.9 | 31.0 | 66.7 | 255 | 39 | 471 | CFR, APACHE, SAPS, GCS | Iran | [61,62] |
| bispyribac-sodium | 1 | 1 | 2.9 | 103 | CFR | Sri Lanka | [8] | ||||
| butachlor | 1 | 1 | 0.0 | 70 | CFR | Taiwan | [46] | ||||
| carbaryl | 1 | 1 | 5.6 | 18 | CFR | Sri Lanka | [8] | ||||
| carbofuran | 3 | 2 | 2.9 | 1.0 | 4.1 | 209 | 100 | 479 | CFR | Brazil, Sri Lanka | [8,9,54] |
| carbosulfan | 2 | 1 | 17.1 | 10.7 | 23.5 | 198 | 51 | 345 | CFR | Sri Lanka | [8,9] |
| chlorfluazuron | 1 | 1 | 2.2 | 45 | CFR | Sri Lanka | [8] | ||||
| chlorpyrifos | 7 | 3 | 6.2 | 5.2 | 8.0 | 208 | 34 | 1376 | CFR, GCS, PSS | Brazil, India, Sri Lanka | [7,8,9,23,26,29,54] |
| cypermethrin | 2 | 2 | 6.1 | 5.1 | 7.0 | 50 | 41 | 58 | CFR | Brazil, India | [7,54] |
| deltamethrin | 1 | 1 | 0.0 | 11 | CFR | Sri Lanka | [8] | ||||
| diazinon | 1 | 1 | 4.8 | 84 | CFR | Sri Lanka | [8] | ||||
| dichlorvos | 2 | 1 | 32.3 | 31.3 | 33.3 | 13 | 9 | 16 | CFR | Japan | [50,75] |
| dimethoate | 6 | 2 | 23.6 | 5.5 | 30.8 | 268 | 17 | 833 | CFR, GCS, PSS | Brazil, Sri Lanka | [8,9,23,26,29,54] |
| diquat | 1 | 1 | 0.0 | 312 | CFR | USA | [48] | ||||
| edifenphos | 1 | 1 | 11.8 | 17 | CFR | Sri Lanka | [8] | ||||
| endosulfan | 6 | 3 | 22.9 | 20.2 | 29.3 | 86 | 9 | 400 | CFR | Brazil, India, Sri Lanka | [7,8,9,36,49,54] |
| endrin | 1 | 1 | 5.0 | 74 | CFR | India | [7] | ||||
| esfenvalerate | 1 | 1 | 8.3 | 12 | CFR | Sri Lanka | [8] | ||||
| etofenprox | 1 | 1 | 0.8 | 121 | CFR | Sri Lanka | [8] | ||||
| fenitrothion | 2 | 1 | 15.4 | 9.4 | 21.3 | 40 | 32 | 47 | CFR | Japan | [50,75] |
| fenobucarb | 2 | 1 | 5.6 | 5.3 | 5.8 | 71 | 38 | 104 | CFR | Sri Lanka | [8,9] |
| fenoxaprop-p-ethyl | 1 | 1 | 0.0 | 74 | CFR | Sri Lanka | [8] | ||||
| fenthion | 4 | 1 | 13.9 | 4.3 | 16.2 | 111 | 23 | 237 | CFR, GCS, PSS | Sri Lanka | [8,9,23,26] |
| fipronil | 1 | 1 | 0.0 | 26 | CFR | Sri Lanka | [8] | ||||
| glufosinate | 1 | 1 | 7.1 | 14 | CFR | Japan | [50] | ||||
| glyphosate | 16 | 6 | 6.1 | 0.1 | 29.3 | 102 | 15 | 3464 | CFR, PSS | Brazil, Japan, Korea, Sri Lanka, Taiwan, USA | [8,9,18,20,21,38,39,48,50,54,55,59,63,67,68,76] |
| hydrogen phosphide | 1 | 1 | 2.6 | 152 | CFR, PSS | Germany | [37] | ||||
| imidacloprid | 2 | 2 | 0.0 | 0.0 | 0.0 | 39 | 8 | 70 | CFR | India, Sri Lanka | [7,8] |
| indoxacarb | 1 | 1 | 14.0 | 7 | CFR | India | [7] | ||||
| lindane | 1 | 1 | 0.0 | 3 | CFR | Sri Lanka | [8] | ||||
| malathion | 7 | 5 | 6.5 | 0.0 | 25.0 | 23 | 5 | 209 | CFR, APACHE | Brazil, India, Japan, Singapore, Sri Lanka | [7,8,9,40,50,54,75] |
| MCPA | 2 | 1 | 5.1 | 4.8 | 5.4 | 387 | 93 | 681 | CFR | Sri Lanka | [8,9] |
| metam sodium | 1 | 1 | 0.0 | 102 | CFR | France | [24] | ||||
| methamidophos | 3 | 2 | 12.5 | 11.5 | 15.4 | 26 | 8 | 191 | CFR | Brazil, Sri Lanka | [8,9,54] |
| methomyl | 2 | 1 | 7.2 | 0.0 | 14.3 | 31 | 7 | 54 | CFR | Sri Lanka | [8,9] |
| monocrotophos | 3 | 3 | 22.2 | 20.4 | 35.0 | 99 | 54 | 257 | CFR | Brazil, India, Sri Lanka | [7,9,54] |
| oxydemeton-methyl | 2 | 2 | 13.4 | 12.5 | 14.3 | 11 | 8 | 14 | CFR, PSS, APACHE, SOFA, GCSCFR | Germany, Sri Lanka | [8,33] |
| oxyfluorfen | 1 | 1 | 0.0 | 15 | CFR | Sri Lanka | [8] | ||||
| paraquat | 14 | 6 | 54.2 | 1.4 | 83.6 | 115 | 7 | 1046 | CFR | Brazil, Japan, Korea, Sri Lanka, Taiwan, USA | [8,9,18,20,28,34,35,41,47,48,50,54,73,76] |
| parathion ethyl | 1 | 1 | 42.9 | 7 | CFR, PSS, APACHE, SOFA, GCS | Germany | [33] | ||||
| parathion methyl | 1 | 1 | 60.0 | 5 | CFR | India | [7] | ||||
| permethrin | 1 | 1 | 0.0 | 13 | CFR | Sri Lanka | [8] | ||||
| petrilachlor | 1 | 1 | 0.0 | 11 | CFR | Sri Lanka | [8] | ||||
| phenthoate | 2 | 1 | 7.4 | 6.5 | 8.3 | 96 | 24 | 168 | CFR | Sri Lanka | [8,9] |
| phorate | 1 | 1 | 19.0 | 21 | CFR | India | [7] | ||||
| picloram | 1 | 1 | 25.0 | 5 | CFR | Brazil | [54] | ||||
| pirimiphos-methyl | 1 | 1 | 0.0 | 12 | CFR | Sri Lanka | [8] | ||||
| profenofos | 2 | 1 | 5.5 | 0.0 | 11.0 | 84 | 22 | 146 | CFR | Sri Lanka | [8,9] |
| propamocarb | 1 | 1 | 100.0 | 1 | CFR | Sri Lanka | [8] | ||||
| propanil | 3 | 1 | 5.0 | 1.6 | 10.9 | 150 | 64 | 412 | CFR | Sri Lanka | [8,9,27] |
| propoxur | 1 | 1 | 0.0 | 16 | CFR | Sri Lanka | [8] | ||||
| prothiofos | 1 | 1 | 7.7 | 13 | CFR | Sri Lanka | [8] | ||||
| quinalphos | 2 | 2 | 12.1 | 12.0 | 12.1 | 101 | 78 | 124 | CFR | India, Sri Lanka | [7,8] |
| rotenone | 1 | 1 | 0.0 | 54 | CFR | USA | [48] | ||||
| spinosad | 1 | 1 | 0.0 | 4 | CFR | India | [7] | ||||
| triazophos | 1 | 1 | 17.0 | 6 | CFR | India | [7] | ||||
| trichlorfon | 2 | 1 | 18.8 | 0.0 | 37.5 | 8 | 7 | 8 | CFR | Japan | [50,75] |
| trifuralin | 1 | 1 | 0.0 | 17 | CFR | Brazil | [54] | ||||
| zink phosphide | 2 | 1 | 7.6 | 4.2 | 11.0 | 30 | 24 | 35 | CFR | India | [49,77] |
| Country | No. of Paper | No. of Papers Providing Case-Fatality on | |
|---|---|---|---|
| Group Level | Active Ingredient Level | ||
| Australia | 1 | 1 | - |
| Brazil | 3 | 2 | 1 |
| China | 1 | 1 | - |
| France | 2 | - | 2 |
| Germany | 2 | 1 | 2 |
| India | 8 | 7 | 4 |
| Iran | 3 | 1 | 2 |
| Israel | 4 | 4 | - |
| Japan | 3 | 1 | 3 |
| Jordan | 1 | 1 | - |
| Korea | 5 | 1 | 4 |
| Serbia | 1 | 1 | - |
| Singapore | 1 | - | 1 |
| Slovenia | 1 | 1 | - |
| South Africa | 2 | 2 | - |
| Sri Lanka | 10 | 5 | 9 |
| Taiwan | 12 | 3 | 10 |
| Turkey | 4 | 4 | - |
| USA | 2 | 1 | 2 |
| Zimbabwe | 1 | 1 | - |
| All | 67 | 38 | 40 |
| WHO Class * | Median Case-Fatality-Ratio | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| <1 | 1 – <10 | 10 – <20 | ≥20% | All | ||||||
| N | % | N | % | N | % | N | % | N | % | |
| Ia | 1 | 4 | 1 | 8 | 2 | 18 | 4 | 6 | ||
| Ib | 3 | 13 | 5 | 38 | 2 | 18 | 10 | 15 | ||
| II | 11 | 61 | 14 | 58 | 6 | 46 | 4 | 36 | 35 | 53 |
| III | 3 | 17 | 3 | 13 | 6 | 9 | ||||
| O | 1 | 4 | 1 | 8 | 2 | 3 | ||||
| U | 4 | 22 | 1 | 4 | 2 | 18 | 7 | 11 | ||
| VF | 1 | 4 | 1 | 9 | 2 | 3 | ||||
| All | 18 | 100 | 24 | 100 | 13 | 100 | 11 | 100 | 66 | 100 |
| Name | WHO Class ** | Publications | Cases | Case Fatality Ratio | |||||
|---|---|---|---|---|---|---|---|---|---|
| n | Median | Min | Mean | Median | Max | CV | CV Norm | ||
| carbamates | 9 | 60 | 0 | 5 | 5 | 14 | 98 | 35 | |
| organophosphate | 31 | 94 | 3 | 15 | 11 | 73 | 92 | 17 | |
| chlorpyrifos | II | 7 | 208 | 5 | 7 | 6 | 8 | 19 | 8 |
| dimethoate | II | 6 | 268 | 6 | 22 | 24 | 31 | 40 | 18 |
| endosulfan | II | 6 | 86 | 20 | 24 | 23 | 29 | 16 | 7 |
| fenthion | II | 4 | 111 | 4 | 12 | 14 | 16 | 44 | 26 |
| glyphosate | III | 16 | 102 | 0 | 7 | 6 | 29 | 100 | 26 |
| malathion | III | 7 | 23 | 0 | 10 | 7 | 25 | 93 | 38 |
| paraquat | II | 14 | 115 | 1 | 49 | 54 | 84 | 56 | 16 |
| Indicator | Pesticide Groups | Active Ingredients |
|---|---|---|
| APACHE—Acute Physiology and Chronic Health Evaluation Score | 8 | 4 |
| GCS—Glasgow Coma Score | 10 | 11 |
| PSS—Poisoning Severity Score | 11 | 8 |
| SAPS—Simplified Acute Physiology Score | 4 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moebus, S.; Boedeker, W. Case Fatality as an Indicator for the Human Toxicity of Pesticides—A Systematic Scoping Review on the Availability and Variability of Severity Indicators of Pesticide Poisoning. Int. J. Environ. Res. Public Health 2021, 18, 8307. https://doi.org/10.3390/ijerph18168307
Moebus S, Boedeker W. Case Fatality as an Indicator for the Human Toxicity of Pesticides—A Systematic Scoping Review on the Availability and Variability of Severity Indicators of Pesticide Poisoning. International Journal of Environmental Research and Public Health. 2021; 18(16):8307. https://doi.org/10.3390/ijerph18168307
Chicago/Turabian StyleMoebus, Susanne, and Wolfgang Boedeker. 2021. "Case Fatality as an Indicator for the Human Toxicity of Pesticides—A Systematic Scoping Review on the Availability and Variability of Severity Indicators of Pesticide Poisoning" International Journal of Environmental Research and Public Health 18, no. 16: 8307. https://doi.org/10.3390/ijerph18168307
APA StyleMoebus, S., & Boedeker, W. (2021). Case Fatality as an Indicator for the Human Toxicity of Pesticides—A Systematic Scoping Review on the Availability and Variability of Severity Indicators of Pesticide Poisoning. International Journal of Environmental Research and Public Health, 18(16), 8307. https://doi.org/10.3390/ijerph18168307






