Association of Self-Reported Physical Fitness during Late Pregnancy with Birth Outcomes and Oxytocin Administration during Labour—The GESTAFIT Project
Abstract
:1. Introduction
2. Materials and Methods
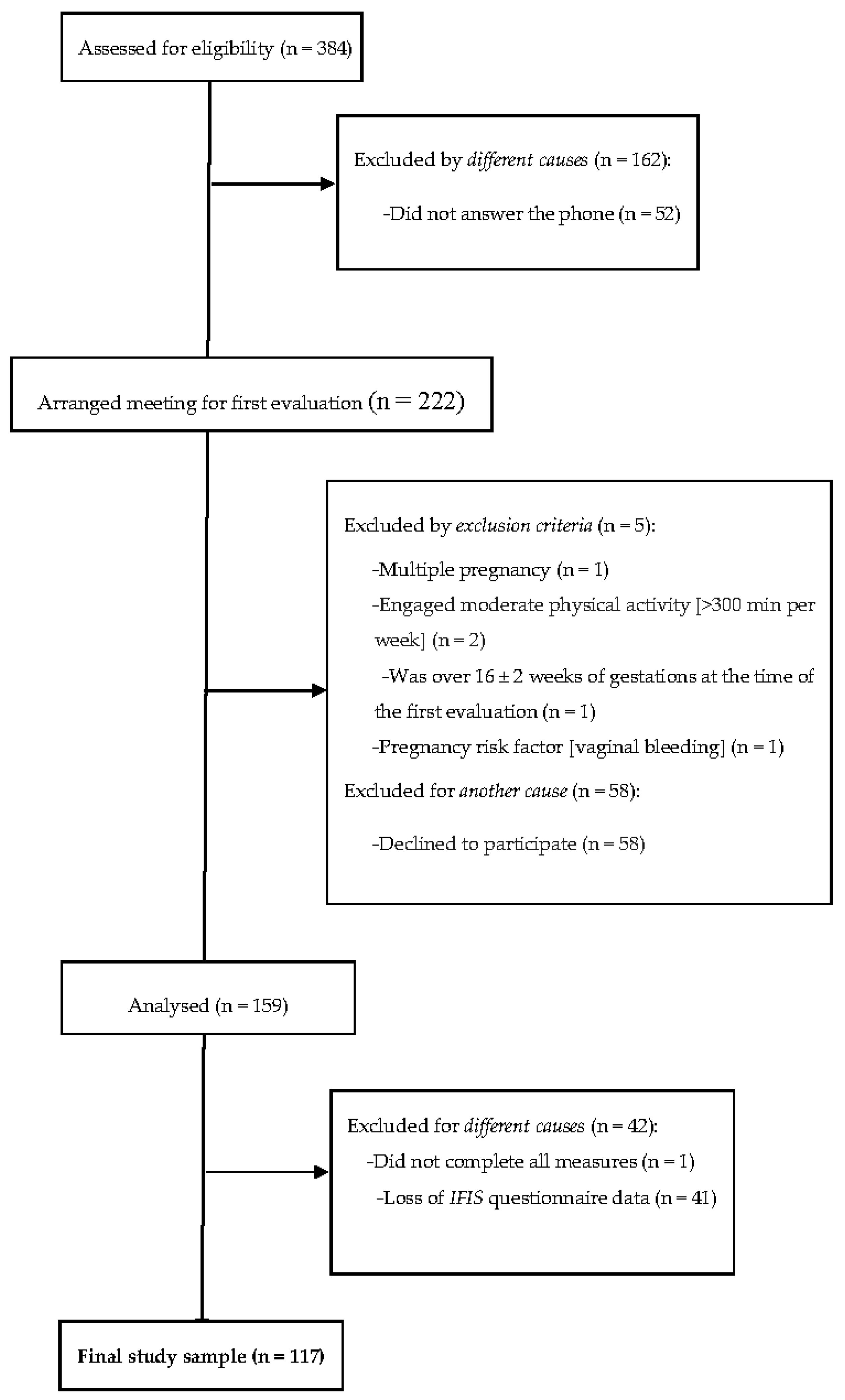
2.1. Study Design and Participants
2.2. Procedures
2.3. Sociodemographic and Clinical Data
2.4. Anthropometry and Body Composition
2.5. Obstetric History
2.6. Birth Outcomes
2.6.1. Umbilical Cord Blood Gas Analysis
2.6.2. Oxytocin Administration before or during Labour
2.7. Physical Fitness
2.8. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Roescher, A.M.; Timmer, A.; Erwich, J.J.H.M.; Bos, A.F. Placental Pathology, Perinatal Death, Neonatal Outcome, and Neurological Development: A Systematic Review. PLoS ONE 2014, 9, e89419. [Google Scholar] [CrossRef] [Green Version]
- Khaskheli, M.-N.; Baloch, S.; Sheeba, A. Iatrogenic Risks and Maternal Health: Issues and Outcomes. Pak. J. Med. Sci. 1969, 30, 111–115. [Google Scholar] [CrossRef]
- Reyes, L.M.; Davenport, M.H. Exercise as a therapeutic intervention to optimize fetal weight. Pharmacol. Res. 2018, 132, 160–167. [Google Scholar] [CrossRef]
- Perales, M.; Calabria, I.; Lopez, C.; Franco, E.; Coteron, J.; Barakat, R. Regular Exercise Throughout Pregnancy is Associated with a Shorter First Stage of Labor. Am. J. Health Promot. 2016, 30, 149–157. [Google Scholar] [CrossRef]
- Żelaźniewicz, A.; Pawłowski, B. Maternal hand grip strength in pregnancy, newborn sex and birth weight. Early Hum. Dev. 2018, 119, 51–55. [Google Scholar] [CrossRef]
- Engberg, E.; Tikkanen, H.O.; Koponen, A.; Hägglund, H.; Kukkonen-Harjula, K.; Tiitinen, A.; Peltonen, J.E.; Pöyhönen-Alho, M. Cardiorespiratory fitness and health-related quality of life in women at risk for gestational diabetes. Scand. J. Med. Sci. Sports 2017, 28, 203–211. [Google Scholar] [CrossRef]
- García, L.B.; Coll-Risco, I.; Ocón-Hernández, O.; Romero-Gallardo, L.; Acosta-Manzano, P.; May, L.; Aparicio, A.V. Association of objectively measured physical fitness during pregnancy with maternal and neonatal outcomes. The Gestafit Project. PLoS ONE 2020, 15, e0229079. [Google Scholar] [CrossRef] [Green Version]
- Lothian, J.A. Healthy Birth Practice #4: Avoid Interventions Unless They Are Medically Necessary. J. Périnat. Educ. 2014, 23, 198–206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Page, K.; McCool, W.F.; Guidera, M. Examination of the Pharmacology of Oxytocin and Clinical Guidelines for Use in Labor. J. Midwifery Women’s Health 2017, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Rousseau, A.; Burguet, A. Oxytocin Administration during Spontaneous Labor: Guidelines for Clinical Practice. Chapter 5: Maternal Risk and Adverse Effects of Using Oxytocin Augmentation during Spontaneous Labor. La Rev. Sage Femme 2017, 16, 63–82. [Google Scholar] [CrossRef]
- Brimdyr, K.; Cadwell, K.; Widström, A.; Svensson, K.; Neumann, M.; Hart, E.A.; Harrington, S.; Phillips, R. The Association Between Common Labor Drugs and Suckling When Skin-to-Skin During the First Hour After Birth. Birth 2015, 42, 319–328. [Google Scholar] [CrossRef] [Green Version]
- Mottola, M.F.; Davenport, M.H.; Brun, C.R.; Inglis, S.D.; Charlesworth, S.; Sopper, M.M. VO2peak Prediction and Exercise Prescription for Pregnant Women. Med. Sci. Sports Exerc. 2006, 38, 1389–1395. [Google Scholar] [CrossRef]
- Bohannon, R.W. Muscle strength: Clinical and prognostic value of hand-grip dynamometry. Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 465–470. [Google Scholar] [CrossRef]
- Irving, G.; Neves, A.L.; Dambha-Miller, H.; Oishi, A.; Tagashira, H.; Verho, A.; Holden, J. International Variations in Primary Care Physician Consultation Time: A Systematic Review of 67 Countries. BMJ Open 2017, 7, e017902. [Google Scholar] [CrossRef]
- Romero-Gallardo, L.; Soriano-Maldonado, A.; Ocón-Hernández, O.; Acosta-Manzano, P.; Coll-Risco, I.; Borges-Cosic, M.; Ortega, F.B.; Aparicio, V.A. International Fitness Scale—IFIS: Validity and association with health-related quality of life in pregnant women. Scand. J. Med. Sci. Sports 2019, 30, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Aparicio, V.A.; Ocón, O.; Padilla-Vinuesa, C.; Soriano-Maldonado, A.; Romero-Gallardo, L.; Borges-Cósic, M.; Coll-Risco, I.; Ruiz-Cabello, P.; Acosta-Manzano, P.; Estévez-López, F.; et al. Effects of supervised aerobic and strength training in overweight and grade I obese pregnant women on maternal and foetal health markers: The GESTAFIT randomized controlled trial. BMC Pregnancy Childbirth 2016, 16, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Committee Opinion No 611. Obstet. Gynecol. 2014, 124, 863–866. [CrossRef]
- Ortega, F.B.; Ruiz, J.; Romero, V.E.; Vicente-Rodriguez, G.; Martínez-Gómez, D.; Manios, Y.; Béghin, L.; Molnar, D.; Widhalm, K.; A Moreno, L.; et al. The International Fitness Scale (IFIS): Usefulness of self-reported fitness in youth. Int. J. Epidemiol. 2011, 40, 701–711. [Google Scholar] [CrossRef] [Green Version]
- Nakagawa, S.; Cuthill, I.C. Effect size, confidence interval and statistical significance: A practical guide for biologists. Biol. Rev. 2007, 82, 591–605. [Google Scholar] [CrossRef]
- Kardel, K.R.; Johansen, B.; Voldner, N.; Iversen, P.O.; Henriksen, T. Association between aerobic fitness in late pregnancy and duration of labor in nulliparous women. Acta Obstet. Gynecol. Scand. 2009, 88, 948–952. [Google Scholar] [CrossRef] [PubMed]
- Bisson, M.; Alméras, N.; Plaisance, J.; Rhéaume, C.; Bujold, E.; Tremblay, A.; Marc, I. Maternal fitness at the onset of the second trimester of pregnancy: Correlates and relationship with infant birth weight. Pediatr. Obes. 2013, 8, 464–474. [Google Scholar] [CrossRef] [PubMed]
- Malin, G.L.; Morris, K.; Khan, K.S. Strength of association between umbilical cord pH and perinatal and long term outcomes: Systematic review and meta-analysis. BMJ 2010, 340, c1471. [Google Scholar] [CrossRef] [Green Version]
- Tzeng, Y.L.; Kuo, S.Y.; Tsai, S.H. Childbirth-Related Fatigue during Labor: An Important but Overlooked Symptom. J. Nurs. 2013, 60, 16–21. [Google Scholar] [CrossRef]
- Coats, T.; Shakur, H. Antifibrinolytic drugs for acute traumatic injury. Cochrane Database Syst. Rev. 2004. [Google Scholar] [CrossRef] [Green Version]
- De Jonge, A.; Teunissen, D.A.M.; Van Diem, M.T.; Scheepers, P.L.H.; Lagro-Janssen, A.L.M. Woman’s Position during Second Stage of Labour: Views of primary care midwives. Cochrane Database Syst. Rev. 2008, 68, 347–356. [Google Scholar]
- Caspersen, C.J.; Powell, K.E.; Christenson, G.M. Physical activity, exercise, and physical fitness: Definitions and distinctions for health-related research. Public Health Rep. 1985, 100, 126–131. [Google Scholar]
- Irianti, S.; Ginandjar, A.B.; Krisnadi, S.R.; Effendi, J.S.; Nataprawira, D.; Gandamihardja, S. Aerobic Exercise and Its Effect on Oxytocin Level and Labor Progression. In IOP Conference Series: Materials Science and Engineering; IOP Science: Bristol, UK, 2017. [Google Scholar]
- Ebrahimzadeh, S.; Golmakani, N.; Kabirian, M.; Shakeri, M.T. Study of correlation between maternal fatigue and uterine contraction pattern in the active phase of labour. J. Clin. Nurs. 2012, 21, 1563–1569. [Google Scholar] [CrossRef]
- Dupont, C.; Carayol, M.; Le Ray, C.; Barasinski, C.; Beranger, R.; Burguet, A.; Chantry, A.; Chiesa, C.; Coulm, B.; Evrard, A.; et al. Recommandations pour l’administration d’oxytocine au cours du travail spontané. Texte court des recommandations. La Rev. Sage femme 2017, 16, 111–118. [Google Scholar] [CrossRef]
- Petersen, L.K.; Vogel, I.; Agger, A.O.; Westergård, J.; Nils, M.; Uldbjerg, N. Variations in serum relaxin (hRLX-2) concentrations during human pregnancy. Acta Obstet. Gynecol. Scand. 1995, 74, 251–256. [Google Scholar] [CrossRef]
- Vodstrcil, L.A.; Tare, M.; Novak, J.; Dragomir, N.; Ramirez, R.J.; Wlodek, M.E.; Conrad, K.P.; Parry, L.J. Relaxin mediates uterine artery compliance during pregnancy and increases uterine blood flow. FASEB J. 2012, 26, 4035–4044. [Google Scholar] [CrossRef] [Green Version]
- Pupula, M.; MacLennan, A.H. Effect of porcine relaxin on spontaneous, oxytocin-driven and prostaglandin-driven pig myometrial activity in vitro. J. Reprod. Med. 1989, 34, 819–823. [Google Scholar] [PubMed]
- Pantelis, A.; Sotiriadis, A.; Chatzistamatiou, K.; Pratilas, G.; Dinas, K. Serum relaxin and cervical length for prediction of spontaneous preterm birth in second-trimester symptomatic women. Ultrasound Obstet. Gynecol. 2018, 52, 763–768. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marshall, S.A.; Senadheera, S.N.; Parry, L.J.; Girling, J.E. The Role of Relaxin in Normal and Abnormal Uterine Function during the Menstrual Cycle and Early Pregnancy. Reprod. Sci. 2017, 24, 342–354. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health and Social Policy and Equality. Report on Attention to Delivery and Birth in the National Health System; Ministry of Health and Social Policy and Equality: Madrid, Spain, 2012; Volume 103. (In Spanish) [Google Scholar]
- Penfield, C.A.; Wing, D.A. Labor Induction Techniques: Which Is the Best? Obstet. Gynecol. Clin. 2017, 46, 509–521. [Google Scholar]
- Fernández, I.O.; Gabriel, M.M.; Martínez, A.M.; Morillo, A.F.-C.; Sánchez, F.L.; Costarelli, V. Newborn feeding behaviour depressed by intrapartum oxytocin: A pilot study. Acta Paediatr. 2012, 101, 749–754. [Google Scholar] [CrossRef] [PubMed]
- Burguet, A.; Rousseau, A. Oxytocin Administration during Spontaneous Labor: Guidelines for Clinical Practice. Chapter 6: Fetal, Neonatal and Pediatric Risks and Adverse Effects of Using Oxytocin Augmentation during Spontaneous Labor. Rev. Sage Femme 2017, 46, 523–530. [Google Scholar] [CrossRef]
| Maternal Outcomes | n | Mean (SD) |
|---|---|---|
| Age, years | 117 | 33.1 (4.5) |
| Body mass index at 16th gestational week, Kg/m2 | 117 | 24.6 (4.1) |
| n (%) | ||
| Living with a partner | 115 (98.3) | |
| Educational status | 117 | |
| Primary or high-school | 24 (20.6) | |
| Specialized training | 19 (16.2) | |
| University degree | 74 (63.2) | |
| Working status | 117 | |
| Homework/unemployed | 34 (29.1) | |
| Partial-time employed/student | 34 (29.1) | |
| Full-time employed | 49 (41.8) | |
| Self-reported Physical Fitness (0–5) | ||
| 34th gestational week | 117 | |
| Overall physical fitness | 3.3 (0.7) | |
| Cardiorespiratory fitness | 2.6 (0.8) | |
| Muscular strength | 3.3 (0.7) | |
| Speed-agility | 3.0 (0.7) | |
| Flexibility | 3.1 (1.0) | |
| Type of birth | 110 | |
| Miscarriages | 117 | 0 (0) |
| Spontaneous Instrumental vacuum/forceps | 65 (59.1) 20 (18.2) | |
| Caesarean section | 25 (22.7) | |
| Oxytocin administered during labor, n % | 30 (27.8) | |
| Birth place | 115 | |
| Public Hospital | 108 (93.9) | |
| Private Hospital | 6 (5.2) | |
| Home | 1 (0.9) | |
| Parity | 117 | |
| Nulliparous | 68 (58.1) | |
| Multiparous | 49 (41.9) | |
| Neonatal outcomes | 110 | |
| Sex (female, n (%)) | 55 (50) | |
| Gestational age at birth, wk | 39.5 (1.2) | |
| Birthweight, g | 3338.5 (428.7) | |
| Apgar Test 1 min | 8.6 (1.1) | |
| Apgar Test 5 min | 9.6 (0.7) | |
| Umbilical Cord blood Gas | ||
| Arterial pH | 80 | 7.2 (0.07) |
| Arterial Partial Pressure CO2, mmHg | 75 | 51.6 (10.7) |
| Arterial Partial Pressure O2, mmHg | 72 | 19.5 (9.4) |
| Arterial O2 saturation, % | 69 | 34.6 (22.8) |
| Venous pH | 93 | 7.3 (0.1) |
| Venous Partial Pressure CO2, mmHg | 89 | 39.7 (7.7) |
| Venous Partial Pressure O2, mmHg | 80 | 26.2 (8.5) |
| Venous O2 saturation, % | 77 | 54.7 (18.0) |
| Overall Fitness | Cardiorespiratory Fitness | Muscular Strength | Speed-Agility | Flexibility | |
|---|---|---|---|---|---|
| Maternal outcomes | |||||
| Week of gestation (at birth) (n = 101) | 0.018 | 0.083 | 0.087 | 0.070 | 0.014 |
| Duration of first stage of labour a (n = 63) | −0.014 | −0.063 | 0.013 | 0.061 | 0.085 |
| Duration of second stage of labour a (n = 69) | −0.021 | −0.069 | −0.040 | 0.103 | 0.152 |
| Neonatal outcomes | |||||
| Birthweight (n = 101) | −0.009 | −0.112 | 0.053 | 0.070 | 0.003 |
| Apgar Test 1 min a (n = 95) | 0.101 | 0.075 | −0.072 | 0.034 | −0.048 |
| Apgar Test 5 min a (n = 95) | −0.081 | −0.042 | 0.002 | −0.107 | −0.177 |
| Cord blood arterial pH a (n = 69) | 0.109 | 0.056 | 0.154 | 0.232 * | −0.017 |
| Cord blood arterial partial pressure of CO2 a (n = 65) | −0.044 | 0.005 | −0.094 | −0.165 | 0.049 |
| Cord blood arterial partial pressure of O2 a (n = 62) | −0.006 | −0.156 | 0.042 | −0.020 | −0.102 |
| Cord blood arterial oxygen saturation a (n = 59) | −0.051 | −0.110 | 0.045 | −0.023 | −0.130 |
| Cord blood venous pH a (n = 81) | 0.207 | 0.091 | 0.214 | 0.239 * | −0.028 |
| Cord blood venous partial pressure of CO2 a (n = 78) | −0.074 | 0.024 | −0.183 | −0.130 | 0.040 |
| Cord blood venous partial pressure of O2 a (n = 69) | 0.094 | −0.062 | 0.130 | 0.133 | 0.034 |
| Cord blood venous O2 saturation a (n = 66) | 0.127 | 0.011 | 0.108 | 0.033 | 0.109 |
| Oxytocin Was Not Administered (n = 78) | Oxytocin Was Administered (n = 30) | p | p * | Effect Size d-Cohen (95% CI) | |
|---|---|---|---|---|---|
| Overall fitness | 3.38 (0.8) | 3.25 (0.7) | 0.067 | 0.348 | 0.16 (−0.22, 0.55) |
| Cardiorespiratory fitness | 2.73 (0.7) | 2.32 (0.8) | 0.044 | 0.013 | 0.55 (0.14, 0.93) |
| Muscular strength | 3.27 (0.7) | 3.29 (0.7) | 0.966 | 0.982 | −0.014 (−0.37, 0.35) |
| Speed-agility | 2.95 (0.7) | 2.96 (0.6) | 0.967 | 0.728 | 0.0 (−0.51, 0.37) |
| Flexibility | 3.27 (1.1) | 2.75 (0.9) | 0.013 | 0.040 | 0.51 (0.09, 0.89) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baena-García, L.; Marín-Jiménez, N.; Romero-Gallardo, L.; Borges-Cosic, M.; Ocón-Hernández, O.; Flor-Alemany, M.; Aparicio, V.A. Association of Self-Reported Physical Fitness during Late Pregnancy with Birth Outcomes and Oxytocin Administration during Labour—The GESTAFIT Project. Int. J. Environ. Res. Public Health 2021, 18, 8201. https://doi.org/10.3390/ijerph18158201
Baena-García L, Marín-Jiménez N, Romero-Gallardo L, Borges-Cosic M, Ocón-Hernández O, Flor-Alemany M, Aparicio VA. Association of Self-Reported Physical Fitness during Late Pregnancy with Birth Outcomes and Oxytocin Administration during Labour—The GESTAFIT Project. International Journal of Environmental Research and Public Health. 2021; 18(15):8201. https://doi.org/10.3390/ijerph18158201
Chicago/Turabian StyleBaena-García, Laura, Nuria Marín-Jiménez, Lidia Romero-Gallardo, Milkana Borges-Cosic, Olga Ocón-Hernández, Marta Flor-Alemany, and Virginia A. Aparicio. 2021. "Association of Self-Reported Physical Fitness during Late Pregnancy with Birth Outcomes and Oxytocin Administration during Labour—The GESTAFIT Project" International Journal of Environmental Research and Public Health 18, no. 15: 8201. https://doi.org/10.3390/ijerph18158201
APA StyleBaena-García, L., Marín-Jiménez, N., Romero-Gallardo, L., Borges-Cosic, M., Ocón-Hernández, O., Flor-Alemany, M., & Aparicio, V. A. (2021). Association of Self-Reported Physical Fitness during Late Pregnancy with Birth Outcomes and Oxytocin Administration during Labour—The GESTAFIT Project. International Journal of Environmental Research and Public Health, 18(15), 8201. https://doi.org/10.3390/ijerph18158201







