Health Risk Assessment of Mercury Exposure from Fish Consumption in Munduruku Indigenous Communities in the Brazilian Amazon
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Fish Capture
2.3. Mercury Analysis
2.4. Data Collection: Participants’ Weight, Family Composition and Fish Consumption
2.5. Potential Fish Consumption Estimative from Catch Effort
2.6. Health Risk Assessment
3. Results
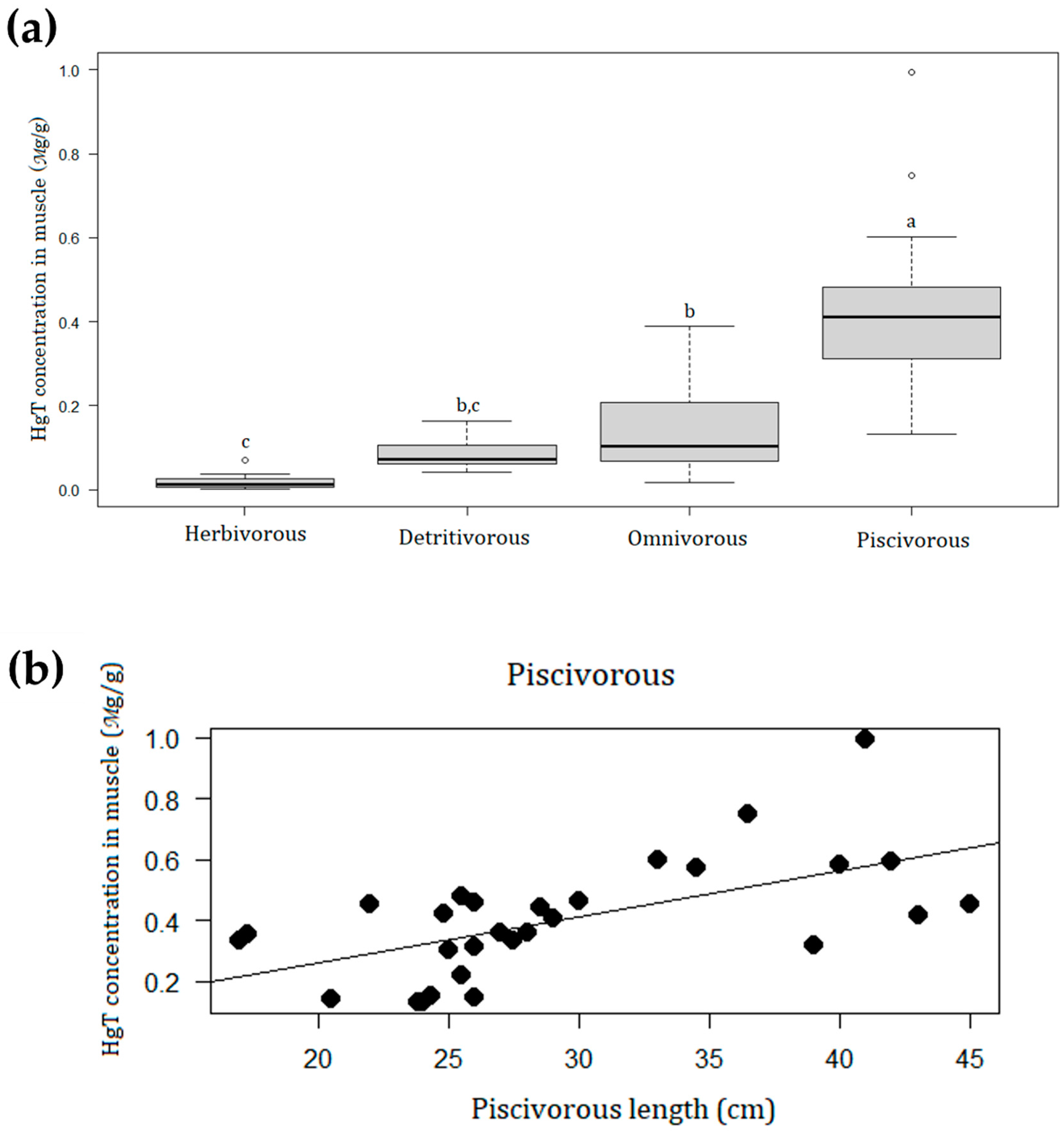
3.1. Fish Catch and Mercury Contamination
3.2. Weight Measurement, Family Composition and Fish Consumption
3.3. Health Risk Assessment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. The State of World Fisheries and Aquaculture; FAO: Rome, Italy, 2020; ISBN 978-92-5-132692-3. [Google Scholar]
- Kawarazuka, N.; Béné, C. Linking small-scale fisheries and aquaculture to household nutritional security: A review of the literature. Food Secur. 2010, 2, 343–357. [Google Scholar] [CrossRef]
- Hicks, C.C.; Cohen, P.J.; Graham, N.A.J.; Nash, K.L.; Allison, E.H.; D’Lima, C.; Mills, D.J.; Roscher, M.; Thilsted, S.H.; Thorne-Lyman, A.L.; et al. Harnessing global fisheries to tackle micronutrient deficiencies. Nature 2019, 566, 378–382. [Google Scholar] [CrossRef]
- Funge-Smith, S.; Bennett, A.A. Fresh look at inland fisheries and their role in food security and livelihoods. Fish Fish. 2019, 20, 1176–1195. [Google Scholar] [CrossRef]
- Fluet-Chouinard, E.; Funge-Smith, S.; McIntyre, P.B. Global hidden harvest of freshwater fish revealed by household surveys. Proc. Natl. Acad. Sci. USA 2018, 115, 7623–7628. [Google Scholar] [CrossRef] [PubMed]
- Béné, C.; Steel, E.; Kambala, L.B.; Gordon, A. Fish as the “bank in the water”: Evidence from chronic-poor communities in Congo. Food Policy 2009, 34, 108–118. [Google Scholar] [CrossRef]
- McIntyre, P.B.; Liermann, C.A.R.; Revenga, C. Linking freshwater fishery management to global good security and biodiversity conservation. Proc. Natl. Acad. Sci. USA 2016, 113, 12880–12885. [Google Scholar] [CrossRef] [PubMed]
- Welcomme, R.L. River Fisheries; FAO: Rome, Italy, 1985; ISBN 978-92-5-102299-3. [Google Scholar]
- Jézéquel, C.; Tedesco, P.A.; Bigorne, R.; Maldonado-Ocampo, J.A.; Ortega, H.; Hidalgo, M.; Martens, K.; Torrente-Vilara, G.; Zuanon, J.; Acosta, A.; et al. A Database of freshwater fish species of the Amazon basin. Sci. Data 2020, 7, 96. [Google Scholar] [CrossRef] [PubMed]
- Isaac, V.J.; Almeida, M.C. El Consumo de Pescado en la Amazonía Brasileña; FAO: Rome, Italy, 2011; ISBN 978-92-5-307029-9. [Google Scholar]
- Isaac, V.J.; Almeida, M.C.; Giarrizzo, T.; Deus, C.P.; Vale, R.; Klein, G.; Begossi, A. Food consumption as an indicator of the conservation of natural resources in riverine communities of the Brazilian Amazon. An. Acad. Bras. Ciências 2015, 87, 2229–2242. [Google Scholar] [CrossRef] [PubMed]
- Begossi, A.; Salivonchyk, S.; Hallwass, G.; Hanazaki, N.; Lopes, P.; Silvano, R.A.M.; Dumaresq, D.; Pittock, J. Fish consumption on the Amazon: A review of biodiversity, hydropower and food security issues. Braz. J. Biol. 2019, 79, 345–357. [Google Scholar] [CrossRef] [PubMed]
- Prestes-Carneiro, G.; Béarez, P.; Bailon, S.; Py-Daniel, A.R.; Neves, E.G. Subsistence fishery at Hatahara (750-1230 CE), a pre-Columbian central Amazonian village. J. Archaeol. Sci. Rep. 2015, 8, 454–462. [Google Scholar] [CrossRef]
- Prestes-Carneiro, G.; Béarez, P.; Shock, M.P.; Prümers, H.; Betancourt, C.J. Pre-Hispanic fishing practices in interfluvial Amazonia: Zooarchaeological evidence from managed landscapes on the Llanos de Mojos savanna. PLoS ONE 2019, 14, e0214638. [Google Scholar] [CrossRef]
- Fearnside, P.M. Social impacts of Brazil’s Tucuruí Dam. Environ. Manag. 1999, 24, 483–495. [Google Scholar] [CrossRef]
- Nevado, J.B.; Martín-Doimeadios, R.R.; Bernardo, F.G.; Moreno, M.J.; Herculano, A.M.; Do Nascimento, J.L.M.; Crespo-López, M.E. Mercury in the Tapajós River basin, Brazilian Amazon: A review. Environ. Int. 2010, 36, 593–608. [Google Scholar] [CrossRef]
- Hallwass, G.; Lopes, P.F.; Juras, A.A.; Silvano, R.A.M. Fishers’ knowledge identifies environmental changes and fish abundance trends in impounded tropical rivers. Ecol. Appl. 2013, 23, 392–407. [Google Scholar] [CrossRef] [PubMed]
- Arrifano, G.P.; Martín-Doimeadios, R.C.R.; Jiménez-Moreno, M.; Ramírez-Mateos, V.; Da Silva, N.F.; Souza-Monteiro, J.R.; Augusto-Oliveira, M.; Paraense, R.S.O.; Macchid, B.M.; Nascimento, J.L.M.; et al. Large-scale projects in the amazon and human exposure to mercury: The case-study of the Tucuruí Dam. Ecotoxicol. Environ. Saf. 2018, 147, 299–305. [Google Scholar] [CrossRef] [PubMed]
- RAISG. 2018. Available online: https://www.amazoniasocioambiental.org/pt-br/radar/mapa-inedito-indica-epidemia-de-garimpo-ilegal-na-panamazonia/ (accessed on 3 March 2021).
- Silva-Junior, C.H.L.; Pessôa, A.C.M.; Carvalho, N.S.; Reis, J.B.C.; Anderson, L.O.; Aragão, L.E.O.C. The Brazilian Amazon deforestation rate in 2020 is the greatest of the decade. Nat. Ecol. Evol. 2021, 5, 144–145. [Google Scholar] [CrossRef] [PubMed]
- Baird, I.G.; Silvano, R.A.M.; Parlee, B.; Poesch, M.; Maclean, B.; Napoleon, A.; Lepine, M.; Hallwass, G. The downstream impacts of hydropower dams and indigenous and local knowledge: Examples from the Peace–Athabasca, Mekong, and Amazon. Environ. Manag. 2021, 67, 682–696. [Google Scholar] [CrossRef] [PubMed]
- Ferrante, L.; Andrade, M.B.T.; Leite, L.; Silva-Junior, C.A.; Lima, M.; Coelho-Junior, M.G.; Da Silva Neto, E.C.; Campolina, D.; Carolino, K.; Diele-Viegas, L.M.; et al. Brazils Highway BR-319: The road to the collapse of the Amazon and the violation of indigenous rights. J. Geogr. Soc. Berl. 2021, 152, 65–70. [Google Scholar] [CrossRef]
- Malm, O. Gold mining as a source of mercury exposure in the Brazilian Amazon. Environ. Res. 1998, 77, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Kalamandeen, M.; Gloor, E.; Johnson, I.; Agard, S.; Katow, M.; Vanbrooke, A.; Ashley, D.; Batterman, S.A.; Ziv, G.; Holder-Collins, K.; et al. Limited biomass recovery from gold mining in Amazonian forests. J. Appl. Ecol. 2020, 57, 1730–1740. [Google Scholar] [CrossRef]
- Ferrante, L.; Fearnside, P.M. Brazil’s new president and “ruralists” threaten Amazonia’s environment, traditional peoples and the global climate. Environ. Conserv. 2019, 46, 261–263. [Google Scholar] [CrossRef]
- Siqueira-Gay, J.; Soares-Filho, B.; Sanchez, L.E.; Oviedo, A.; Sonter, L.J. Proposed legislation to mine Brazil’s indigenous lands will threaten Amazon forests and their valuable ecosystem services. One Earth 2020, 3, 356–362. [Google Scholar] [CrossRef]
- Crespo-Lopez, M.E.; Augusto-Oliveira, M.; Lopes-Araújo, A.; Santos-Sacramento, L.; Yuki Takeda, P.; Macchi, B.M.; Do Nascimento, J.L.M.; Maia, C.S.F.; Lima, R.R.; Arrifano, G.P. Mercury: What can we learn from the Amazon? Environ. Int. 2021, 146, 106223. [Google Scholar] [CrossRef]
- Lacerda, E.M.D.C.B.; Souza, G.D.S.; Cortes, M.I.T.; Rodrigues, A.R.; Pinheiro, M.C.N.; Silveira, L.C.D.L.; Ventura, D.F. Comparison of visual functions of two Amazonian populations: Possible consequences of different mercury exposure. Front. Neurosci. 2020, 13, 1428. [Google Scholar] [CrossRef] [PubMed]
- Salonen, J.T.; Seppänen, K.; Nyyssönen, K.; Korpela, H.; Kauhanen, J.; Kantola, M.; Tuomilehto, J.; Esterbauer, H.; Tatzber, F.; Salonen, R. Intake of mercury from fish, lipid peroxidation, and the risk of myocardial infarction and coronary, cardiovascular, and any death in eastern Finnish men. Circulation 1995, 91, 645–655. [Google Scholar] [CrossRef] [PubMed]
- Fillion, M.; Mergler, D.; Passos, C.J.S.; Larribe, F.; Lemire, M.; Guimarães, J.R.D. A preliminary study of mercury exposure and blood pressure in the Brazilian Amazon. Environ. Health 2006, 5, 1–9. [Google Scholar] [CrossRef]
- Vasconcellos, A.C.S.; Barrocas, P.R.G.; Ruiz, C.M.V.; Mourão, D.D.S.; Hacon, S.D.S. Burden of Mild Mental Retardation attributed to prenatal methylmercury exposure in Amazon: Local and regional estimates. Cienc. Saude Coletiva 2018, 23, 3535–3545. [Google Scholar] [CrossRef] [PubMed]
- Poulin, J.; Gibb, H.; Prüss-Üstün, A.; World Health Organization. Mercury: Assessing the Environmental Burden of Disease at National and Local Levels; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Bose-O’Reilly, S.; McCarty, K.M.; Steckling, N.; Lettmeier, B. Mercury exposure and children’s health. Curr. Probl. Pediatric Adolesc. Health Care 2010, 40, 186–215. [Google Scholar] [CrossRef]
- Basta, P.C.; Viana, P.V.S.; Vasconcellos, A.; Périssé, A.; Hofer, C.; Paiva, N.S.; Kempton, J.W.; Ciampi de Andrade, D.; Oliveira, R.A.A.; Achatz, R.W.; et al. Mercury exposure in Munduruku indigenous communities from Brazilian Amazon: Methodological background and an overview of the principal results. Int. J. Environ. Res. Public Health 2021, 18. in press. [Google Scholar]
- Santos, G.M.; De Mérona, B.; Juras, A.A.; Jégu, M. Peixes do Baixo Rio Tocantins: 20 Anos Depois da Usina Hidrelétrica Tucuruí; Eletronorte: Brasília, Brasil, 2004; p. 216. [Google Scholar]
- Santos, G.M.; Ferreira, E.J.G.; Zuanon, J.A.S. Peixes Comerciais de Manaus; Ibama/PróVárzea: Manaus, Brazil, 2006. [Google Scholar]
- Akagi, H.; Suzuki, T.; Arimura, K.; Ando, T.; Sakamoto, M.; Satoh, H.; Matsuyama, A. Mercury Analysis Manual, 105th ed.; Ministry of the Environmental: Tokyo, Japan, 2004. Available online: http://nimd.env.go.jp/kenkyu/docs/march_mercury_analysis_manual(e).pdf (accessed on 21 November 2019).
- da Silva, S.F.; de Oliveira Lima, M. Mercury in fish marketed in the Amazon triple frontier and health risk assessment. Chemosphere 2000, 248, 125989. [Google Scholar] [CrossRef]
- World Health Organization. Guidance for Identifying Populations at Risk from Mercury Exposure; Mercury Publications: Geneva, Switzerland, 2008. [Google Scholar]
- U.S.-EPA. Reference Dose for Methylmercury; U.S. Environmental Protection Agency: Washington, DC, USA, 2000.
- FAO/WHO. Evaluation of certain food additives and contaminants: Sixty-first report of the Joint FAO/WHO Expert Committee on Food Additives. In Proceedings of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), Rome, Italy, 10–19 June 2003. [Google Scholar]
- Domingo, J.L. Omega-3 fatty acids and the benefits of fish consumption: Is all that glitters gold? Environ. Int. 2007, 33, 993–998. [Google Scholar] [CrossRef]
- Sidhu, K.S. Health benefits and potential risks related to consumption of fish or fish oil. Regul. Toxicol. Pharmacol. 2003, 38, 336–344. [Google Scholar] [CrossRef] [PubMed]
- Arruda, M.C.F. Avaliação Dos Indicadores da Política de Pesca do Programa Zona Franca Verde: Perspectivas Econômicas e Ambientais; Universidade Federal do Amazonas: Manaus, Brasil, 2017. [Google Scholar]
- Cerdeira, R.G.P.; Ruffino, M.I.; Isaac, V.J. Consumo de pescado e outros alimentos pela população ribeirinha do lago grande de Monte Alegre, PA-Brasil. Acta Amaz. 1997, 27, 213–227. [Google Scholar] [CrossRef]
- Batista, V.D.; Isaac, V.J.; Viana, J.P. Exploração e Manejo Dos Recursos Pesqueiros da Amazônia. A Pesca e os Recursos Pesqueiros na Amazônia Brasileira; Ibama/ProVárzea: Manaus, Brasil, 2004; pp. 63–151. [Google Scholar]
- Hu, X.F.; Singh, K.; Chan, H.M. Mercury exposure, blood pressure, and hypertension: A systematic review and dose–response meta-analysis. Environ. Health Perspect. 2018, 126, 076002. [Google Scholar] [CrossRef] [PubMed]
- Weihe, P.; Grandjean, P.; Jørgensen, P.J. Application of hair-mercury analysis to determine the impact of a seafood advisory. Environ. Res. 2005, 97, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Crump, K.S.; Kjellström, T.; Shipp, A.M.; Silvers, A.; Stewart, A. Influence of prenatal mercury exposure upon scholastic and psychological test performance: Benchmark analysis of a New Zealand cohort. Risk Anal. 1998, 18, 701–713. [Google Scholar] [CrossRef]
- Santos Serrão de Castro, N.; de Oliveira Lima, M. Hair as a biomarker of long-term mercury exposure in Brazilian Amazon: A systematic review. Int. J. Environ. Res. Public Health 2018, 15, 500. [Google Scholar] [CrossRef] [PubMed]
- Reuben, A.; Frischtak, H.; Berky, A.; Ortiz, E.J.; Morales, A.M.; Hsu-Kim, H.; Pendergast, L.L.; Pan, W.K. Elevated hair mercury levels are associated with neurodevelopmental deficits in children living near artisanal and small-scale gold mining in Peru. Geo. Health 2020, 4, e2019GH000222. [Google Scholar]
- Costa Junior, J.M.F.; Lima, A.A.D.S.; Rodrigues Junior, D.; Khoury, E.D.T.; Souza, G.D.S.; Silveira, L.C.D.L.; Pinheiro, M.D.C.N. Emotional and motor symptoms in riverside dwellers exposed to mercury in the Amazon. Rev. Bras. De Epidemiol. 2017, 20, 212–224. [Google Scholar] [CrossRef][Green Version]
- Marques, R.C.; Bernardi, J.V.; Abreu, L.; Dórea, J.G. Neurodevelopment outcomes in children exposed to organic mercury from multiple sources in a tin-ore mine environment in Brazil. Arch. Environ. Contam. Toxicol. 2015, 68, 432–441. [Google Scholar] [CrossRef] [PubMed]
- Marques, R.C.; Bernardi, J.V.; Cunha, M.P.; Dórea, J.G. Impact of organic mercury exposure and home delivery on neurodevelopment of Amazonian children. Int. J. Hygen Environ. Health 2016, 219, 498–502. [Google Scholar] [CrossRef] [PubMed]
- Malm, O.; Castro, M.B.; Bastos, W.R.; Branches, F.J.; Guimarães, J.R.; Zuffo, C.E.; Pfeiffer, W.C. An assessment of Hg pollution in different goldmining areas, Amazon Brazil. Sci. Total Environ. 1995, 175, 127–140. [Google Scholar] [CrossRef]
- Brabo, E.D.S.; Santos, E.D.O.; Jesus, I.M.D.; Mascarenhas, A.F.; Faial, K.F. Mercury levels in fish consumed by the Sai Cinza indigenous community, Munduruku Reservation, Jacareacanga County, State of Para, Brazil. Cad. Saúde Pública 1999, 15, 325–332. [Google Scholar] [CrossRef]
- Roulet, M.; Lucotte, M.; Canuel, R.; Farella, N.; Courcelles, M.; Guimaraes, J.R.; Amorim, M. Increase in mercury contamination recorded in lacustrine sediments following deforestation in the central Amazon. Chem. Geol. 2000, 165, 243–266. [Google Scholar] [CrossRef]
- de Oliveira Santos, E.C.; de Jesus, I.M.; da Silva Brabo, E.; Loureiro, E.C.B.; da Silva Mascarenhas, A.F.; Weirich, J.; Cleary, D. Mercury exposures in riverside Amazon communities in Para, Brazil. Environ. Res. 2000, 84, 100–107. [Google Scholar] [CrossRef][Green Version]
- Dórea, J.G.; Barbosa, A.C.; Ferrari, Í.; De Souza, J.R. Fish consumption (Hair Mercury) and nutritional status of Amazonian Amer-Indian Children. Am. J. Hum. Biol. Off. J. Hum. Biol. Assoc. 2005, 17, 507–514. [Google Scholar] [CrossRef]
- Telmer, K.; Costa, M.; Angélica, R.S.; Araujo, E.S.; Maurice, Y. The source and fate of sediment and mercury in the Tapajós River, Pará, Brazilian Amazon: Ground-and space-based evidence. J. Environ. Manag. 2006, 81, 101–113. [Google Scholar] [CrossRef]
- Hacon, S.; Barrocas, P.R.; Vasconcellos, A.C.S.D.; Barcellos, C.; Wasserman, J.C.; Campos, R.C.; Azevedo-Carloni, F.B. An overview of mercury contamination research in the Amazon basin with an emphasis on Brazil. Cad. Saúde Pública 2008, 24, 1479–1492. [Google Scholar] [CrossRef] [PubMed]
- Passos, C.J.S.; Da Silva, D.S.; Lemire, M.; Fillion, M.; Guimaraes, J.R.D.; Lucotte, M.; Mergler, D. Daily mercury intake in fish-eating populations in the Brazilian Amazon. J. Expo. Sci. Environ. Epidemiol. 2008, 18, 76–87. [Google Scholar] [CrossRef]
- Faial, K.; Deus, R.; Deus, S.; Neves, R.; Jesus, I.; Santos, E.; Brasil, D. Mercury levels assessment in hair of riverside inhabitants of the Tapajós River, Pará State, Amazon, Brazil: Fish consumption as a possible route of exposure. J. Trace Elem. Med. Biol. 2015, 30, 66–76. [Google Scholar] [CrossRef]
- Freitas, J.S.; Lacerda, E.; Maria, C.; Rodrigues, D., Jr.; Corvelo, T.C.O.; Silveira, L.C.L.; Souza, G.S. Mercury exposure of children living in Amazonian villages: Influence of geographical location where they lived during prenatal and postnatal development. An. Acad. Bras. Ciências 2019, 91. [Google Scholar] [CrossRef] [PubMed]
- Lino, A.S.; Kasper, D.; Guida, Y.S.; Thomaz, J.R.; Malm, O. Total and methyl mercury distribution in water, sediment, plankton, and fish along the Tapajós River basin in the Brazilian Amazon. Chemosphere 2019, 235, 690–700. [Google Scholar] [CrossRef] [PubMed]
- Brasil. Resolução RDC nº42. Dispõe Sobre o Regulamento Técnico MERCOSUL sobre Limites Máximos de Contaminantes Inorgânicos em Alimentos; Diário Oficial da União da República Federativa do Brasil: Brasília, Brasil, 2013.
- FAO/WHO. Joint FAO/WHO Expert Committee on Food Additives (JECFA), Report of the Tenth Section, Rotterdam, The Netherlands 4 to 8 April 2016. Available online: http://www.fao.org/fao-who-codexalimentarius/sh-proxy/en/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FMeetings%252FCX-735-10%252FReport%252FREP16_CFe.pdf (accessed on 22 June 2020).
- Holmes, R. Non-dietary modifiers of nutritional status in tropical forest populations of Venezuela. Interciencia 1984, 9, 386–390. [Google Scholar]
- Dufour, D.L. Diet and nutritional status of Ameridians: A review of the literature. Cad. Saúde Pública 1991, 7, 481–502. [Google Scholar] [CrossRef] [PubMed]
| Fish Species | Popular Name | N | Size (cm) | Weight (g) | Trophic Level | Hg (µg/g) (SD) | Min–Max (Hg) |
|---|---|---|---|---|---|---|---|
| Serrasalmus rhombeus | Piranha Preta | 6 | 17–34.5 | 140–1305 | Piscivorous | 0.71 (±0.61) | 0.33–1.95 |
| Pseudoplatystoma fasciatum | Surubim | 6 | 23.8–45 | 141–907 | Piscivorous | 0.24 (±0.15) | 0.13–0.45 |
| Pinirampus pirinampu | Barbado | 8 | 17.3–42 | 109–961 | Piscivorous | 0.49 (±0.14) | 0.31–0.75 |
| Cichla ocellaris | Tucunaré | 6 | 25–29 | 347–571 | Piscivorous | 0.33 (±0.06) | 0.22–0.41 |
| Rhaphiodon vulpinus | Peixe Cachorro | 2 | 39–41 | 328–469 | Piscivorous | 0.66 (±0.48) | 0.32–1.00 |
| Ageneiosus inermis | Mandubé | 1 | 33 | 550 | Piscivorous | 0.6 | - |
| Pachyurus junki | Corvina | 1 | 20.5 | 148 | Piscivorous | 0.14 | - |
| Geophagus proximus | Caratinga | 10 | 10.5–18.5 | 36–171 | Omnivorous | 0.07 (±0.03) | 0.03–0.10 |
| Pimelodus blochii | Mandii | 7 | 14.5–17.3 | 60–84 | Omnivorous | 0.20 (±0.05) | 0.13–0.28 |
| Leporinus fasciatus | Aracu Flamengo | 5 | 17.7–23.3 | 102–244 | Omnivorous | 0.09 (±0.02) | 0.05–0.11 |
| Caenotropus labyrinthicus | João Duro | 6 | 13.7–14.8 | 63–73 | Omnivorous | 0.28 (±0.07) | 0.17–0.39 |
| Hemiodus unimaculatus | Charuto | 1 | 17.5 | 95 | Omnivorous | 0.02 | - |
| Schizodon vittatus | Aracu | 4 | 21.3–27.3 | 163–351 | Herbivorous | 0.03 (±0.01) | 0.02–0.04 |
| Myloplus rubripinnis | Pacu Branco | 7 | 12.5–20.5 | 89–390 | Herbivorous | 0.02 (±0.03) | 0.01–0.07 |
| Semaprochilodus insignis | Jaraqui Escama Grossa | 6 | 21–23.5 | 223–329 | Detritivorous | 0.11 (±0.05) | 0.05–0.16 |
| Prochilodus nigricans | Curimatá | 6 | 20.5–24 | 253–369 | Detritivorous | 0.07 (±0.02) | 0.04–0.10 |
| Curimata sp. | Branquinha | 6 | 12.7–14 | 64–85 | Detritivorous | 0.09 (±0.03) | 0.06–0.13 |
| Fish Catch | ||
| Total fish caught (n°) | 88 | |
| Catch period (days) | 7 | |
| Fish caught per day (n°) | 12.6 | |
| Average weight of fish (grams) | 268.2 | |
| Amount of fish per family (grams) | 3371.7 | |
| Family composition and weight measurements (Kg) | ||
| Average number of individuals per family | 4 | |
| Average number of adults | 2 | |
| Average number of children | 2 | |
| Average weight of childbearing age women (n = 53) | 49.89 | |
| Average weight of adult men (≥12 years) (n = 58) | 56.45 | |
| Average weight of children (from 5|−12 years old) (n = 24) | 24.45 | |
| Average weight of children (2|−5 years) (n = 42) | 14.07 | |
| Fish consumption estimative (grams) | ||
| Weekly Intake | Daily Intake | |
| Adult men (45%) | 1517.2 | 216.75 |
| Childbearing age women (35%) | 1180.1 | 168.58 |
| Children 5|−12 years old (15%) | 505.7 | 72.25 |
| Children aged 2|−5 years (5%) | 168.6 | 24.08 |
| Scenarios Constructed | Hg-intake Dose µg/kg bw/day | Risk Ratio | ||
|---|---|---|---|---|
| U.S. EPA | FAO/WHO (Women and Children) | FAO/WHO (Adults in General) | ||
| Scenario 1—Rainy Season | ||||
| Women of childbearing age | 0.78 | 7.84 | 3.41 | N.A. |
| Adult Men | 0.89 | 8.91 | N.A. | 1.98 |
| Children 5|−12 years old | 0.69 | 6.86 | 2.98 | N.A. |
| Children 2|−5 years | 0.40 | 3.97 | 1.73 | N.A. |
| Scenario 2—Dry Season | ||||
| Women of childbearing age | 0.59 | 5.95 | 2.59 | N.A. |
| Adult Men | 0.68 | 6.76 | N.A. | 1.50 |
| Children 5|−12 years old | 0.52 | 5.20 | 2.26 | N.A. |
| Children 2|−5 years | 0.30 | 3.01 | 1.31 | N.A. |
| Scenario 3—Current | ||||
| Women of childbearing age | 0.73 | 7.29 | 3.17 | N.A. |
| Adult Men | 0.83 | 8.28 | N.A. | 1.84 |
| Children 5|−12 years old | 0.64 | 6.37 | 2.77 | N.A. |
| Children 2|−5 years | 0.37 | 3.69 | 1.60 | N.A. |
| Scenario 4—Critical | ||||
| Women of childbearing age | 2.28 | 22.76 | 9.90 | N.A. |
| Adult Men | 2.59 | 25.86 | N.A. | 5.75 |
| Children 5|−12 years old | 1.99 | 19.90 | 8.65 | N.A. |
| Children 2|−5 years | 1.15 | 11.53 | 5.01 | N.A. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vasconcellos, A.C.S.d.; Hallwass, G.; Bezerra, J.G.; Aciole, A.N.S.; Meneses, H.N.d.M.; Lima, M.d.O.; Jesus, I.M.d.; Hacon, S.d.S.; Basta, P.C. Health Risk Assessment of Mercury Exposure from Fish Consumption in Munduruku Indigenous Communities in the Brazilian Amazon. Int. J. Environ. Res. Public Health 2021, 18, 7940. https://doi.org/10.3390/ijerph18157940
Vasconcellos ACSd, Hallwass G, Bezerra JG, Aciole ANS, Meneses HNdM, Lima MdO, Jesus IMd, Hacon SdS, Basta PC. Health Risk Assessment of Mercury Exposure from Fish Consumption in Munduruku Indigenous Communities in the Brazilian Amazon. International Journal of Environmental Research and Public Health. 2021; 18(15):7940. https://doi.org/10.3390/ijerph18157940
Chicago/Turabian StyleVasconcellos, Ana Claudia Santiago de, Gustavo Hallwass, Jaqueline Gato Bezerra, Angélico Nonato Serrão Aciole, Heloisa Nascimento de Moura Meneses, Marcelo de Oliveira Lima, Iracina Maura de Jesus, Sandra de Souza Hacon, and Paulo Cesar Basta. 2021. "Health Risk Assessment of Mercury Exposure from Fish Consumption in Munduruku Indigenous Communities in the Brazilian Amazon" International Journal of Environmental Research and Public Health 18, no. 15: 7940. https://doi.org/10.3390/ijerph18157940
APA StyleVasconcellos, A. C. S. d., Hallwass, G., Bezerra, J. G., Aciole, A. N. S., Meneses, H. N. d. M., Lima, M. d. O., Jesus, I. M. d., Hacon, S. d. S., & Basta, P. C. (2021). Health Risk Assessment of Mercury Exposure from Fish Consumption in Munduruku Indigenous Communities in the Brazilian Amazon. International Journal of Environmental Research and Public Health, 18(15), 7940. https://doi.org/10.3390/ijerph18157940







