Advances and Applications of Water Phytoremediation: A Potential Biotechnological Approach for the Treatment of Heavy Metals from Contaminated Water
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
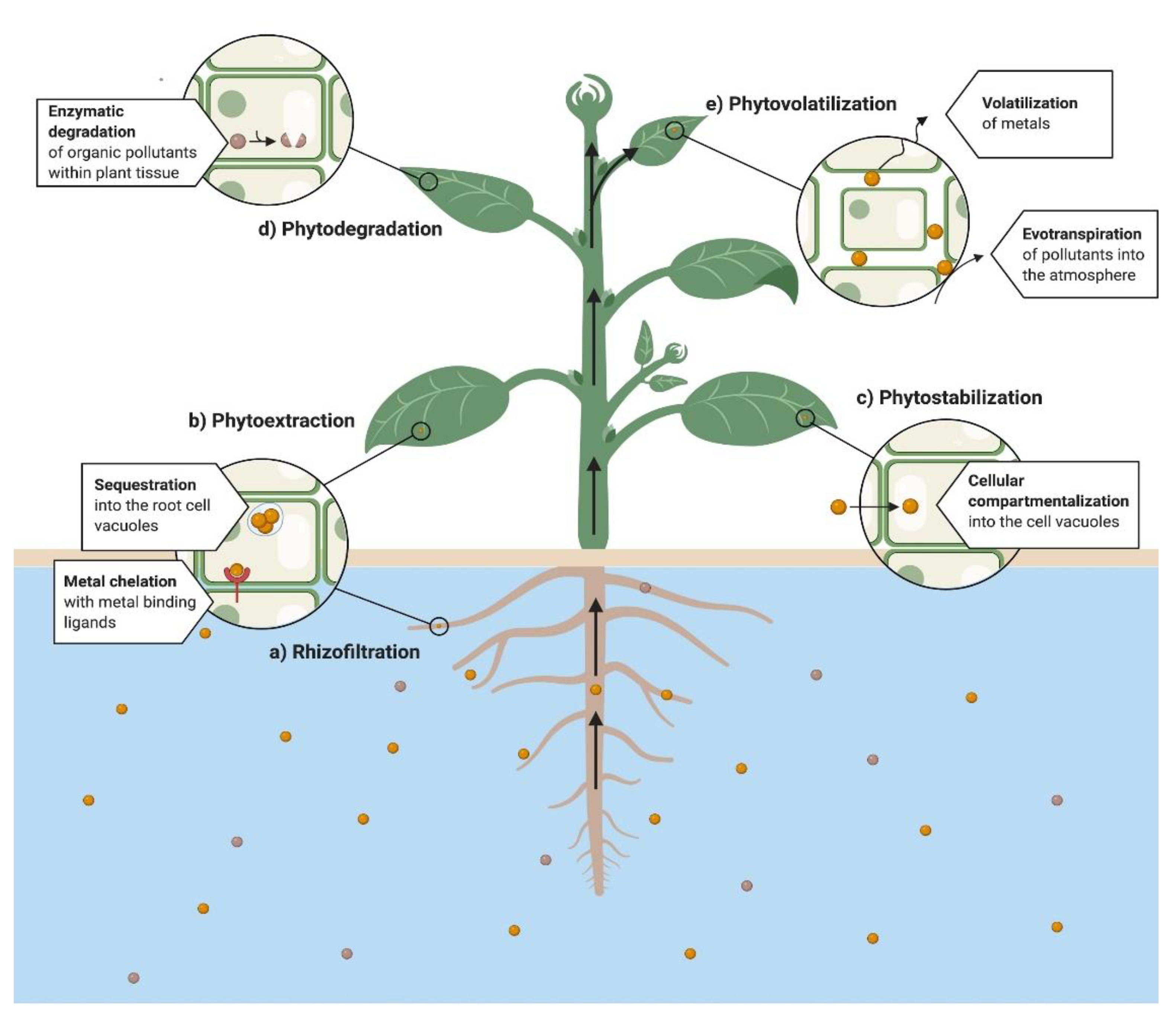
4. Mechanistic Approach
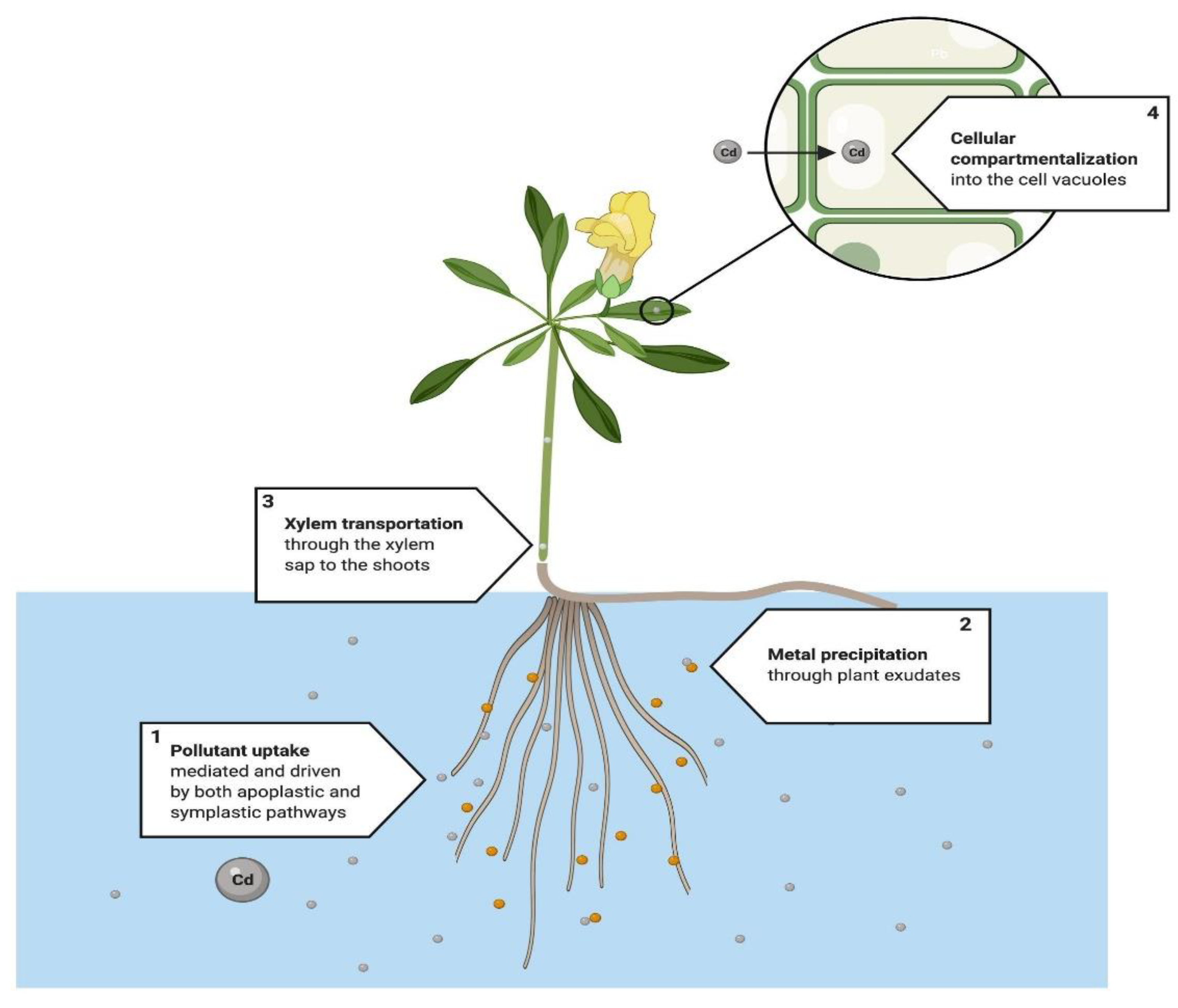
4.1. Phytoextraction
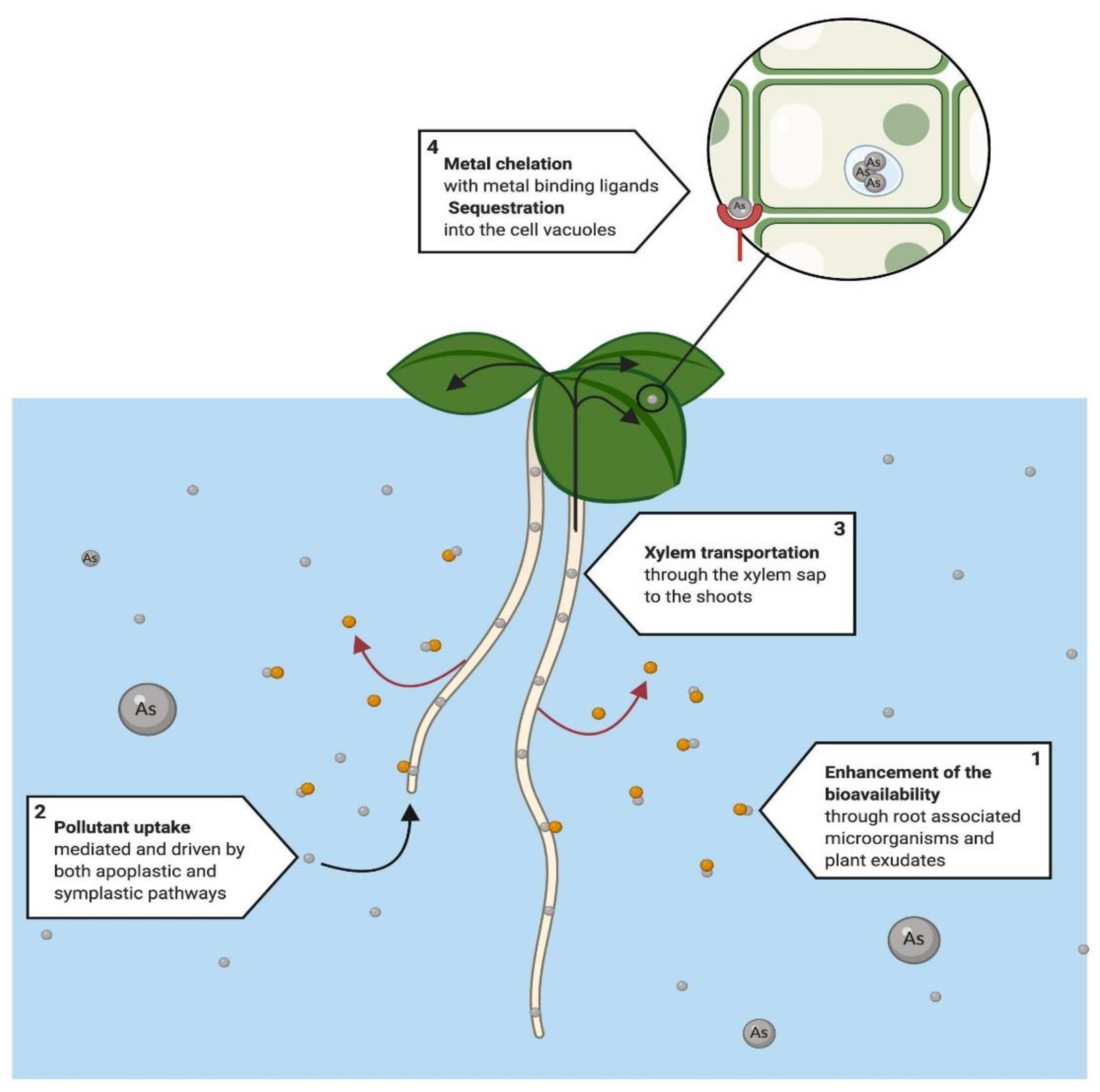
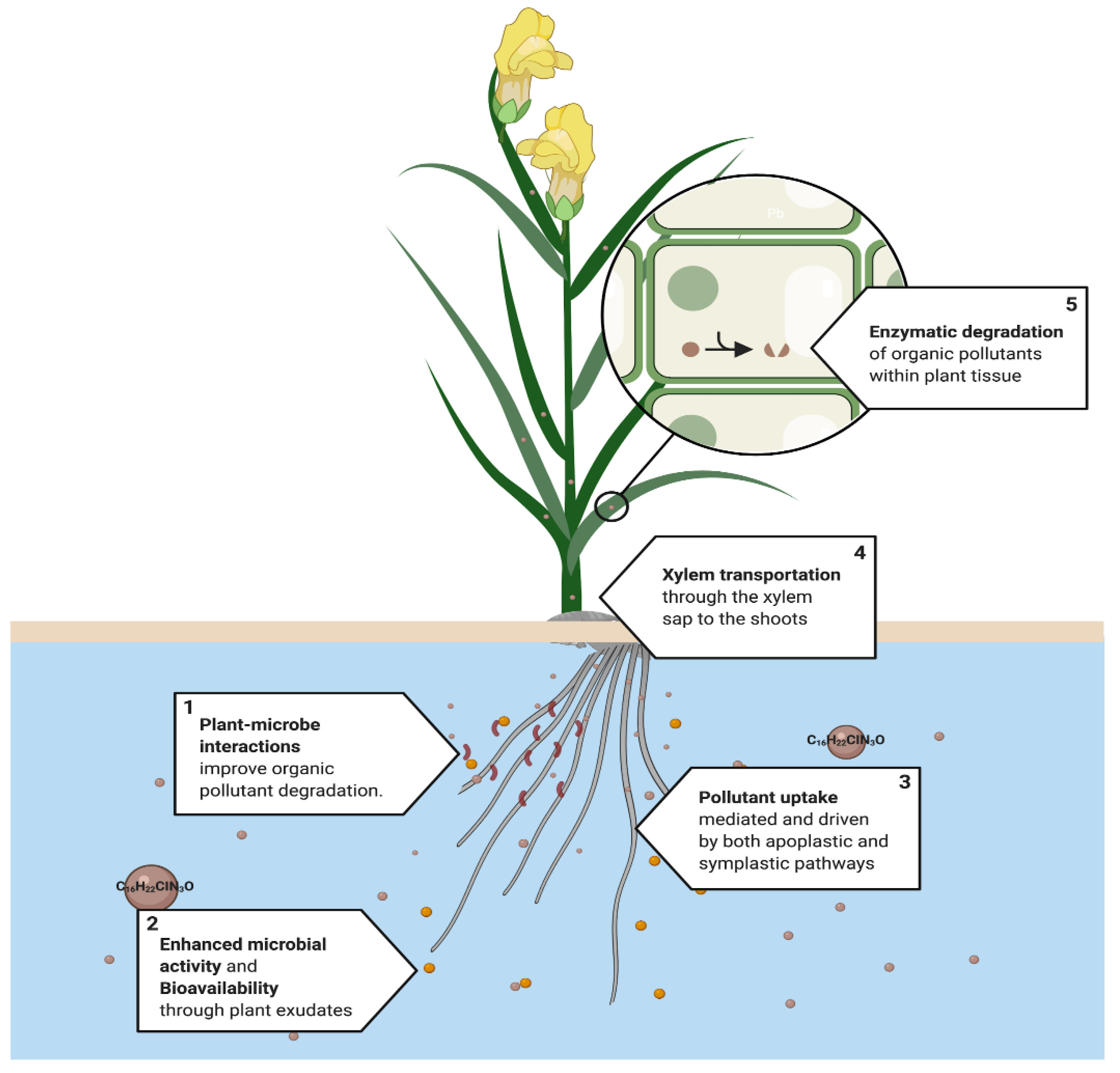
4.2. Rhizofiltration
4.3. Phytodegradation
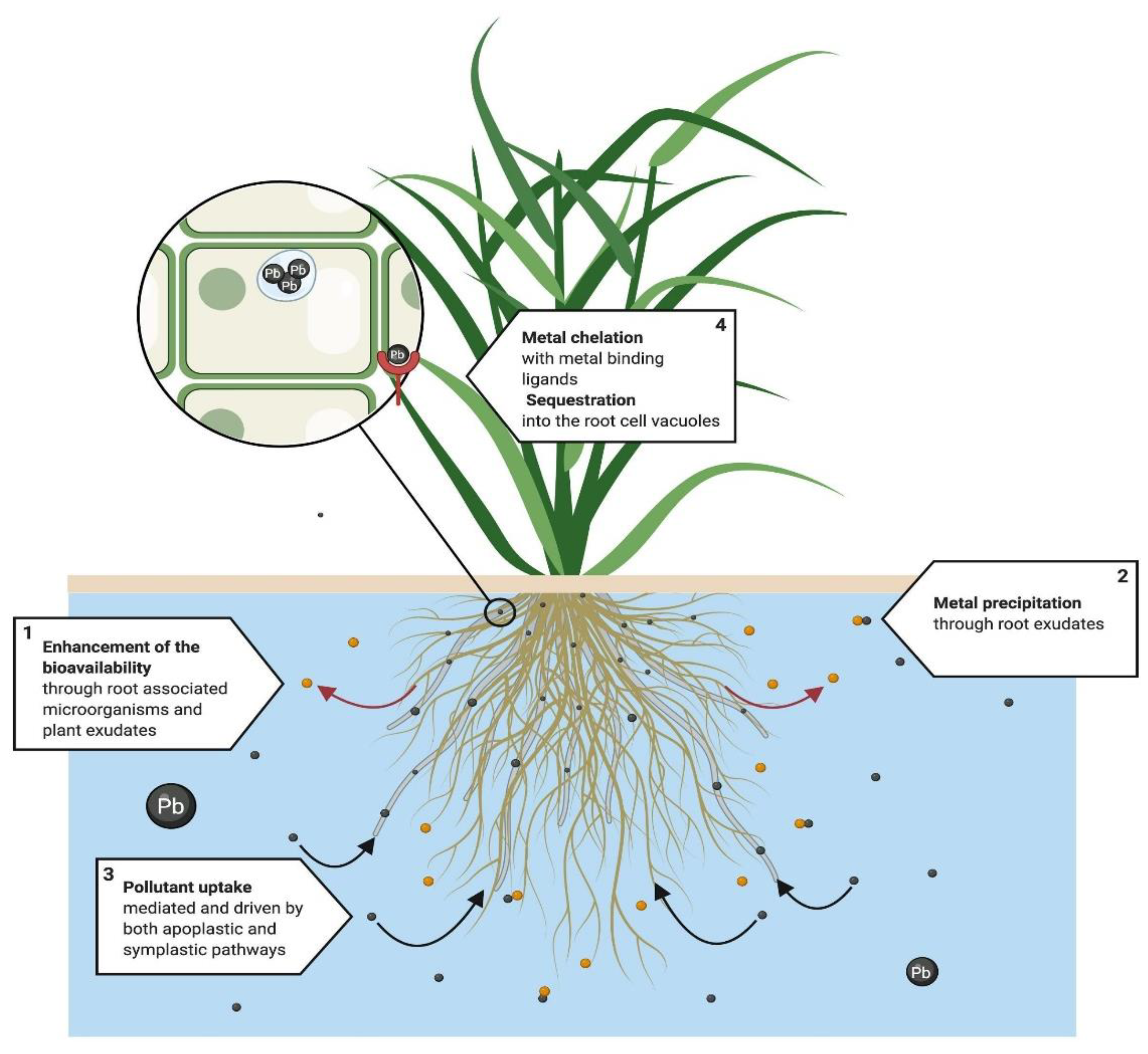
4.4. Phytostabilization
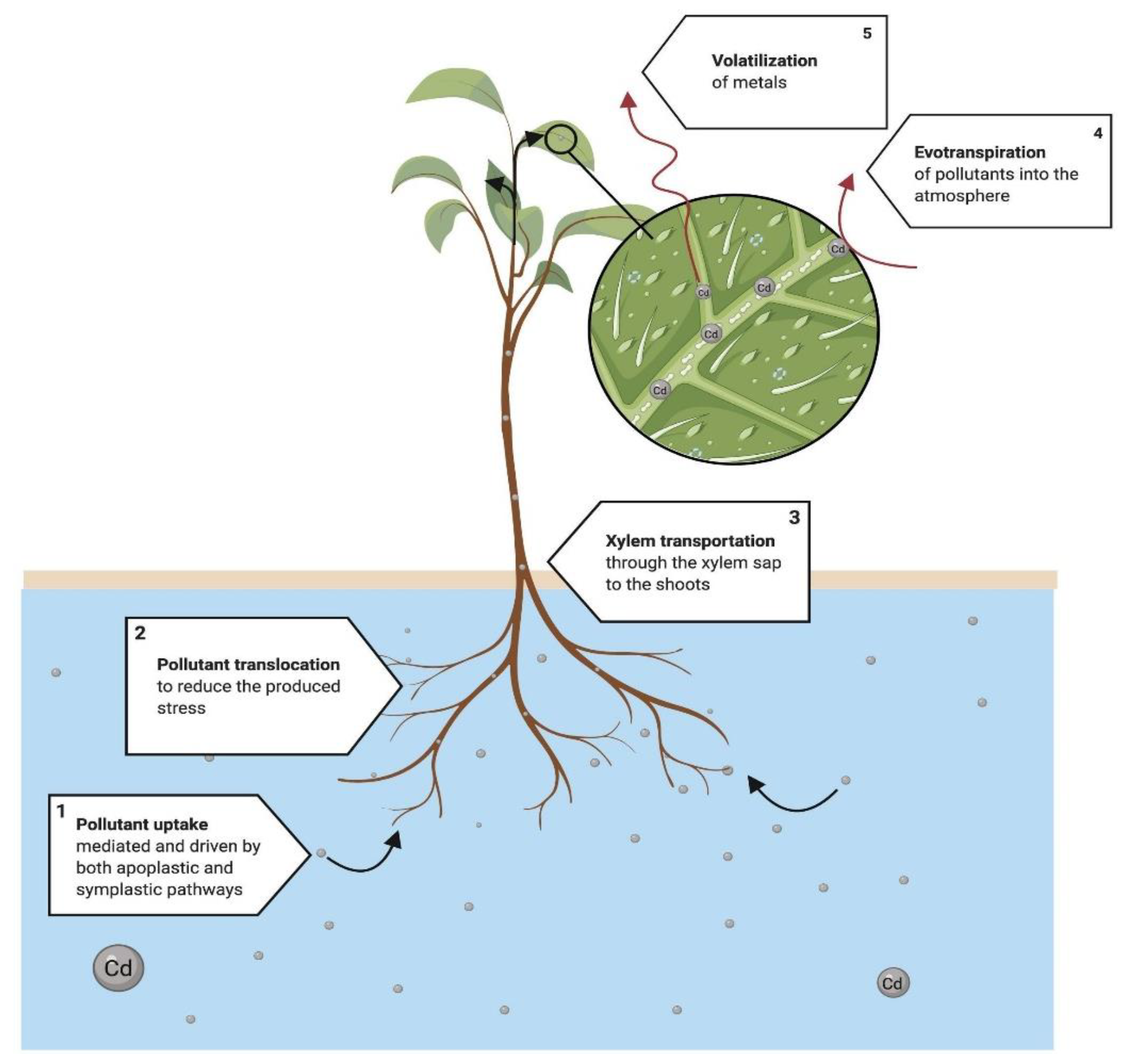
4.5. Phytovolatilization
4.6. Relationship Root-Microorganisms
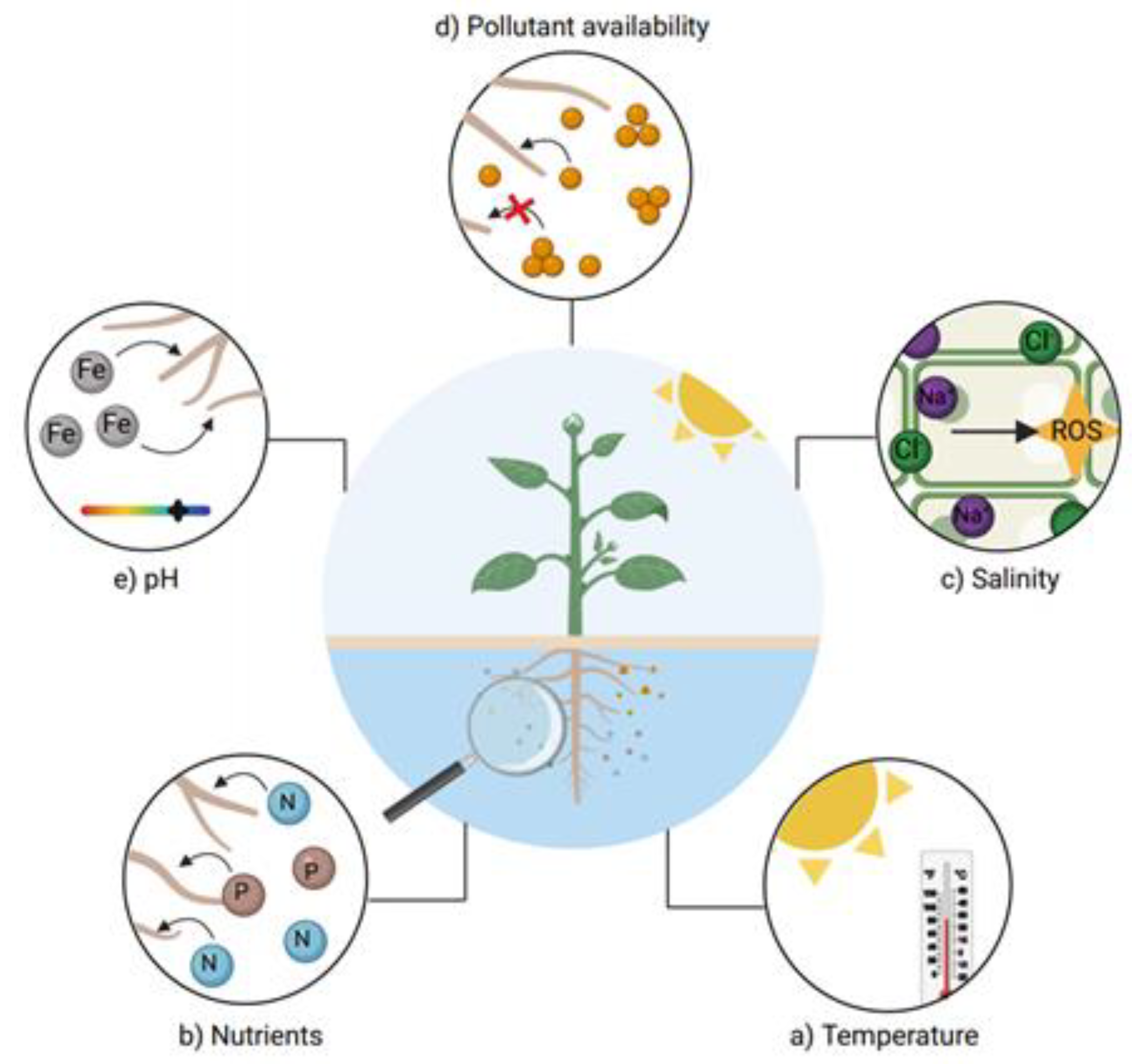
4.7. Environmental Characteristics That Influence Water Phytoremediation
5. Obtained Patents in the Field of Water Phytoremediation
6. Current Challenges and Literature Gaps
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Drinking Water. 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/drinking-water#:~:text=Contaminated%20water%20and%20poor%20sanitation,A%2C%20typhoid%2C%20and%20polio (accessed on 16 November 2020).
- Chen, L.; Zhou, S.; Shi, Y.; Wang, C.; Li, B.; Li, Y.; Wu, S. Heavy metals in food crops, soil, and water in the Lihe River Watershed of the Taihu Region and their potential health risks when ingested. Sci. Total Environ. 2018, 615, 141–149. [Google Scholar] [CrossRef]
- Bamuwamye, M.; Ogwok, P.; Tumuhairwe, V.; Eragu, R.; Nakisozi, H.; Ogwang, P.E. Human health risk assessment of heavy metals in Kampala (Uganda) drinking water. J. Food Res. 2017, 6, 6–16. [Google Scholar] [CrossRef]
- Ashraf, N.; Glaeser, E.; Holland, A.; Steinberg, B.M. Water, Health and Wealth; Working Paper 23807; National Bureau of Economic Research: Cambridge, MA, USA, 2017. [Google Scholar]
- Su, F.; Wu, J.; He, S. Set pair analysis-Markov chain model for groundwater quality assessment and prediction: A case study of Xi’an city, China. Hum. Ecol. Risk Assess. Int. J. 2019, 25, 158–175. [Google Scholar] [CrossRef]
- Li, P.; Wu, J. Drinking Water Quality and Public Health. Expo. Health 2019, 11, 73–79. [Google Scholar] [CrossRef]
- Scheili, A.; Rodriguez, M.; Sadiq, R. Seasonal and spatial variations of source and drinking water quality in small municipal systems of two Canadian regions. Sci. Total Environ. 2015, 508, 514–524. [Google Scholar] [CrossRef] [PubMed]
- Karthigadevi, G.; Manikandan, S.; Karmegam, N.; Subbaiya, R.; Chozhavendhan, S.; Balasubramani, R.; Chang, S.W.; Awasthi, M.K. Chemico-nanotreatment methods for the removal of persistent organic pollutants and xenobiotics in water—A review. Bioresour. Technol. 2021, 324, 124678. [Google Scholar] [CrossRef] [PubMed]
- Pi, Y.; Li, X.; Xia, Q.; Wu, J.; Li, Y.; Xiao, J.; Li, Z. Adsorptive and photocatalytic removal of Persistent Organic Pollutants (POPs) in water by metal-organic frameworks (MOFs). Chem. Eng. J. 2018, 337, 351–371. [Google Scholar] [CrossRef]
- Abdel-Satar, A.M.; Ali, M.H.; Goher, M.E. Indices of water quality and metal pollution of Nile River, Egypt. Egypt. J. Aquat. Res. 2017, 43, 21–29. [Google Scholar] [CrossRef]
- Anh, H.Q.; Le, T.P.Q.; Da Le, N.; Lu, X.X.; Duong, T.T.; Garnier, J.; Rochelle-Newall, E.; Zhang, S.; Oh, N.-H.; Oeurng, C.; et al. Antibiotics in surface water of East and Southeast Asian countries: A focused review on contamination status, pollution sources, potential risks, and future perspectives. Sci. Total Environ. 2021, 764, 142865. [Google Scholar] [CrossRef]
- Chowdhary, P.; Raj, A.; Bharagava, R.N. Environmental pollution and health hazards from distillery wastewater and treatment approaches to combat the environmental threats: A review. Chemosphere 2018, 194, 229–246. [Google Scholar] [CrossRef]
- Dwivedi, S.; Mishra, S.; Tripathi, R.D. Ganga water pollution: A potential health threat to inhabitants of Ganga basin. Environ. Int. 2018, 117, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Parihar, R.D.; Sharma, A.; Bakshi, P.; Sidhu, G.P.S.; Bali, A.S.; Karaouzas, I.; Bhardwaj, R.; Thukral, A.K.; Gyasi-Agyei, Y.; et al. Global evaluation of heavy metal content in surface water bodies: A meta-analysis using heavy metal pollution indices and multivariate statistical analyses. Chemosphere 2019, 236, 124364. [Google Scholar] [CrossRef]
- Li, X.; Wang, B.; Cao, Y.; Zhao, S.; Wang, H.; Feng, X.; Zhou, J.; Ma, X. Water Contaminant Elimination Based on Metal–Organic Frameworks and Perspective on Their Industrial Applications. ACS Sustain. Chem. Eng. 2019, 7, 4548–4563. [Google Scholar] [CrossRef]
- Mohiuddin, A.K. Chemical Contaminants and Pollutants in the Measurable Life of Dhaka City. Eur. J. Sustain. Dev. Res. 2019, 3, em0083. [Google Scholar] [CrossRef]
- Ustaoğlu, F.; Tepe, Y. Water quality and sediment contamination assessment of Pazarsuyu Stream, Turkey using multivariate statistical methods and pollution indicators. Int. Soil Water Conserv. Res. 2019, 7, 47–56. [Google Scholar] [CrossRef]
- Al Osman, M.; Yang, F.; Massey, I.Y. Exposure routes and health effects of heavy metals on children. BioMetals 2019, 32, 563–573. [Google Scholar] [CrossRef]
- Fu, Z.; Xi, S. The effects of heavy metals on human metabolism. Toxicol. Mech. Methods 2019, 30, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Sankhla, M.S.; Kumar, R. Contaminant of Heavy Metals in Groundwater & its Toxic Effects on Human Health & Environment. SSRN Electron. J. 2019. [Google Scholar] [CrossRef]
- Tangahu, B.V.; Abdullah, S.R.S.; Basri, H.; Idris, M.; Anuar, N.; Mukhlisin, M. A Review on Heavy Metals (As, Pb, and Hg) Uptake by Plants through Phytoremediation. Int. J. Chem. Eng. 2011, 2011, 939161. [Google Scholar] [CrossRef]
- EPA (United States Environmental Protection Agency). EnviroAtlas. Heavy Metals. 2020. Available online: https://cfpub.epa.gov/ncea/iris2/chemicalLanding.cfm?substance_nmbr=278&layout=option1 (accessed on 16 November 2020).
- Kim, K.-H.; Kabir, E.; Jahan, S.A. Exposure to pesticides and the associated human health effects. Sci. Total Environ. 2017, 575, 525–535. [Google Scholar] [CrossRef]
- Hua, M.; Zhang, S.; Pan, B.; Zhang, W.; Lv, L.; Zhang, Q. Heavy metal removal from water/wastewater by nanosized metal oxides: A review. J. Hazard. Mat. 2012, 211, 317–331. [Google Scholar] [CrossRef] [PubMed]
- Heltshe, J.F.; Forrester, N.E. Estimating species richness using the jackknife procedure. Biometrics 1983, 39, 1–11. [Google Scholar] [CrossRef]
- Zeng, X.; Xia, Y.; Tong, H. Jackknife approach to the estimation of mutual information. Proc. Natl. Acad. Sci. USA 2018, 115, 9956–9961. [Google Scholar] [CrossRef] [PubMed]
- Bokhari, S.H.; Ahmad, I.; Mahmood-Ul-Hassan, M.; Mohammad, A. Phytoremediation potential of Lemna minor L. for heavy metals. Int. J. Phytoremediat. 2015, 18, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Tauqeer, H.M.; Ali, S.; Rizwan, M.; Ali, Q.; Saeed, R.; Iftikhar, U.; Ahmad, R.; Farid, M.; Abbasi, G.H. Phytoremediation of heavy metals by Alternanthera bettzickiana: Growth and physiological response. Ecotoxicol. Environ. Saf. 2016, 126, 138–146. [Google Scholar] [CrossRef]
- Nayak, A.K.; Panda, S.S.; Basu, A.; Dhal, N.K. Enhancement of toxic Cr (VI), Fe, and other heavy metals phytoremediation by the synergistic combination of native Bacillus cereus strain and Vetiveria zizanioides L. Int. J. Phytoremediat. 2018, 20, 682–691. [Google Scholar] [CrossRef]
- Zhang, X.; Li, M.; Yang, H.; Li, X.; Cui, Z. Physiological responses of Suaeda glauca and Arabidopsis thaliana in phytoremediation of heavy metals. J. Environ. Manag. 2018, 223, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Adler, P.R.; Summerfelt, S.T.; Glenn, D.; Takeda, F. Mechanistic approach to phytoremediation of water. Ecol. Eng. 2003, 20, 251–264. [Google Scholar] [CrossRef]
- Bolisetty, S.; Peydayesh, M.; Mezzenga, R. Sustainable technologies for water purification from heavy metals: Review and analysis. Chem. Soc. Rev. 2019, 48, 463–487. [Google Scholar] [CrossRef] [PubMed]
- Joseph, L.; Jun, B.M.; Flora, J.R.; Park, C.M.; Yoon, Y. Removal of heavy metals from water sources in the developing world using low-cost materials: A review. Chemosphere 2019, 229, 142–159. [Google Scholar] [CrossRef]
- Muthusaravanan, S.; Sivarajasekar, N.; Vivek, J.S.; Paramasivan, T.; Naushad, M.; Prakashmaran, J.; Gayathri, V.; Al-Duaij, O.K. Phytoremediation of heavy metals: Mechanisms, methods and enhancements. Environ. Chem. Lett. 2018, 16, 1339–1359. [Google Scholar] [CrossRef]
- Mirza, N.; Mahmood, Q.; Pervez, A.; Ahmad, R.; Farooq, R.; Shah, M.M.; Azim, M.R. Phytoremediation potential of Arundo donax in arsenic-contaminated synthetic wastewater. Bioresour. Technol. 2010, 101, 5815–5819. [Google Scholar] [CrossRef] [PubMed]
- Pandey, V.C. Phytoremediation of heavy metals from fly ash pond by Azolla caroliniana. Ecotoxicol. Environ. Saf. 2012, 82, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Benaroya, R.O.; Tzin, V.; Tel-Or, E.; Zamski, E. Lead accumulation in the aquatic fern Azolla filiculoides. Plant Physiol. Biochem. 2004, 42, 639–645. [Google Scholar] [CrossRef] [PubMed]
- Valderrama, A.; Tapia, J.; Peñailillo, P.; Carvajal, D.E. Water phytoremediation of cadmium and copper using Azolla filiculoides Lam. in a hydroponic system. Water Environ. J. 2012, 27, 293–300. [Google Scholar] [CrossRef]
- Rai, P.K. Technical Note: Phytoremediation of Hg and Cd from Industrial Effluents using an Aquatic Free Floating Macrophyte Azolla pinnata. Int. J. Phytoremediat. 2008, 10, 430–439. [Google Scholar] [CrossRef]
- Khandare, R.V.; Watharkar, A.D.; Pawar, P.K.; Jagtap, A.A.; Desai, N.S. Hydrophytic plants Canna indica, Epipremnum aureum, Cyperus alternifolius and Cyperus rotundus for phytoremediation of fluoride from water. Environ. Technol. Innov. 2021, 21, 101234. [Google Scholar] [CrossRef]
- Abdallah, M.A.M. Phytoremediation of heavy metals from aqueous solutions by two aquatic macrophytes, Ceratophyllum demersum and Lemna gibba L. Environ. Technol. 2012, 33, 1609–1614. [Google Scholar] [CrossRef]
- Ingole, N.W.; Bhole, A.G. Removal of heavy metals from aqueous solution by water hyacinth (Eichhornia crassipes). J. Water Supply Res. Technol. 2003, 52, 119–128. [Google Scholar] [CrossRef]
- Odjegba, V.J.; Fasidi, I.O. Phytoremediation of heavy metals by Eichhornia crassipes. Environmentalist 2007, 27, 349–355. [Google Scholar] [CrossRef]
- Sakakibara, M.; Ohmori, Y.; Ha, N.T.H.; Sano, S.; Sera, K. Phytoremediation of heavy metal-contaminated water and sediment by Eleocharis acicularis. Clean Soil Air Water 2011, 39, 735–741. [Google Scholar] [CrossRef]
- Mukhtar, S.A.I.M.A.; Bhatti, H.N.; Khalid, M.; Haq, M.A.U.; Shahzad, S.M. Potential of sunflower (Helianthus annuus L.) for phytoremediation of nickel (Ni) and lead (Pb) contaminated water. Pak. J. Bot. 2010, 42, 4017–4026. [Google Scholar]
- Dixit, S.; Dhote, S. Evaluation of uptake rate of heavy metals by Eichhornia crassipes and Hydrilla verticillata. Environ. Monit. Assess. 2009, 169, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Sinha, S.; Saxena, R.; Singh, S. Fluoride Removal from Water by Hydrilla verticillata (l.f.) Royle and Its Toxic Effects. Bull. Environ. Contam. Toxicol. 2000, 65, 683–690. [Google Scholar] [CrossRef]
- Xue, P.-Y.; Yan, C.-Z. Arsenic accumulation and translocation in the submerged macrophyte Hydrilla verticillata (L.f.) Royle. Chemosphere 2011, 85, 1176–1181. [Google Scholar] [CrossRef]
- Chanu, L.B.; Gupta, A. Phytoremediation of lead using Ipomoea aquatica Forsk. in hydroponic solution. Chemosphere 2016, 156, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Weerasinghe, A.; Ariyawnasa, S.; Weerasooriya, R. Phyto-remediation potential of Ipomoea aquatica for Cr(VI) mitigation. Chemosphere 2008, 70, 521–524. [Google Scholar] [CrossRef] [PubMed]
- Caldelas, C.; Araus, J.L.; Febrero, A.; Bort, J. Accumulation and toxic effects of chromium and zinc in Iris pseudacorus L. Acta Physiol. Plant. 2012, 34, 1217–1228. [Google Scholar] [CrossRef]
- Najeeb, U.; Ahmad, W.; Zia, M.H.; Zaffar, M.; Zhou, W. Enhancing the lead phytostabilization in wetland plant Juncus effusus L. through somaclonal manipulation and EDTA enrichment. Arab. J. Chem. 2017, 10, S3310–S3317. [Google Scholar] [CrossRef]
- Mkandawire, M.; Taubert, B.; Dudel, E.G. Capacity of Lemna gibba L. (Duckweed) for Uranium and Arsenic Phytoremediation in Mine Tailing Waters. Int. J. Phytoremediat. 2004, 6, 347–362. [Google Scholar] [CrossRef]
- Wang, Q.; Cui, Y.; Dong, Y. Phytoremediation of Polluted Waters Potentials and Prospects of Wetland Plants. Acta Biotechnol. 2002, 22, 199–208. [Google Scholar] [CrossRef]
- Vernay, P.; Gauthier-Moussard, C.; Hitmi, A. Interaction of bioaccumulation of heavy metal chromium with water relation, mineral nutrition and photosynthesis in developed leaves of Lolium perenne L. Chemosphere 2007, 68, 1563–1575. [Google Scholar] [CrossRef] [PubMed]
- Galal, T.M.; Al-Sodany, Y.M.; Al-Yasi, H.M. Phytostabilization as a phytoremediation strategy for mitigating water pollutants by the floating macrophyte Ludwigia stolonifera (Guill. & Perr.) P.H. Raven. Int. J. Phytoremediat. 2019, 22, 373–382. [Google Scholar] [CrossRef]
- Zurayk, R.; Sukkariyah, B.; Baalbaki, R.; Ghanem, D.A. Ni Phytoaccumulation in Mentha aquatica L. and Mentha sylvestris L. Water Air Soil Pollut. 2002, 139, 355–364. [Google Scholar] [CrossRef]
- Colzi, I.; Lastrucci, L.; Rangoni, M.; Coppi, A.; Gonnelli, C. Using Myriophyllum aquaticum (Vell.) Verdc. to remove heavy metals from contaminated water: Better dead or alive? J. Environ. Manag. 2018, 213, 320–328. [Google Scholar] [CrossRef]
- Sivaci, E.R.; Sivaci, A.; Sökmen, M. Biosorption of cadmium by Myriophyllum spicatum L. and Myriophyllum triphyllum orchard. Chemosphere 2004, 56, 1043–1048. [Google Scholar] [CrossRef]
- Xia, J.; Hua, T.; Xue, Y.; Zhao, L.; Sun, H.; Liu, C. Myriophyllum elatinoides: A potential candidate for the phytoremediation of water with low level boron contamination. J. Hazard. Mater. 2021, 401, 123333. [Google Scholar] [CrossRef]
- Kumar, J.N.; Soni, H.; Kumar, R.N.; Bhatt, I. Macrophytes in phytoremediation of heavy metal contaminated water and sediments in Pariyej Community Reserve, Gujarat, India. Turk. J. Fish. Aquat. Sci. 2008, 8, 193–200. [Google Scholar]
- Cheng, S. Heavy metals in plants and phytoremediation. Environ. Sci. Pollut. Res. 2003, 10, 335–340. [Google Scholar] [CrossRef]
- Bello, A.O.; Tawabini, B.S.; Khalil, A.B.; Boland, C.R.; Saleh, T.A. Phytoremediation of cadmium-, lead- and nickel-contaminated water by Phragmites australis in hydroponic systems. Ecol. Eng. 2018, 120, 126–133. [Google Scholar] [CrossRef]
- Das, S.; Goswami, S.; Das Talukdar, A. A Study on Cadmium Phytoremediation Potential of Water Lettuce, Pistia stratiotes L. Bull. Environ. Contam. Toxicol. 2013, 92, 169–174. [Google Scholar] [CrossRef]
- Kumar, V.; Singh, J.; Saini, A.; Kumar, P. Phytoremediation of copper, iron and mercury from aqueous solution by water lettuce (Pistia stratiotes L.). Environ. Sustain. 2019, 2, 55–65. [Google Scholar] [CrossRef]
- Romeh, A.A. Phytoremediation of cyanophos insecticide by Plantago major L. in water. J. Environ. Health Sci. Eng. 2014, 12, 38. [Google Scholar] [CrossRef] [PubMed]
- Fritioff, Å.; Greger, M. Uptake and distribution of Zn, Cu, Cd, and Pb in an aquatic plant Potamogeton natans. Chemosphere 2006, 63, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.C.; Chen, T.B.; Lei, M.; Hu, T.D. Direct determination of arsenic species in arsenic hyperaccumulator Pteris vittata by EXAFS. Acta Bot. 2004, 46, 46–50. [Google Scholar]
- Loría, K.C.; Emiliani, J.; Bergara, C.D.; Herrero, M.S.; Salvatierra, L.M.; Pérez, L.M. Effect of daily exposure to Pb-contaminated water on Salvinia biloba physiology and phytoremediation performance. Aquat. Toxicol. 2019, 210, 158–166. [Google Scholar] [CrossRef]
- Hoffmann, T.L.; Kutter, C.; Santamaria, J.M. Capacity of Salvinia minima Baker to Tolerate and Accumulate As and Pb. Eng. Life Sci. 2004, 4, 61–65. [Google Scholar] [CrossRef]
- Da Silva, A.A.; De Oliveira, J.A.; De Campos, F.V.; Ribeiro, C.; Farnese, F.D.S.; Costa, A.C. Phytoremediation potential of Salvinia molesta for arsenite contaminated water: Role of antioxidant enzymes. Theor. Exp. Plant Physiol. 2018, 30, 275–286. [Google Scholar] [CrossRef]
- Dhir, B.; Srivastava, S. Heavy metal removal from a multi-metal solution and wastewater by Salvinia natans. Ecol. Eng. 2011, 37, 893–896. [Google Scholar] [CrossRef]
- Rahman, M.A.; Hasegawa, H.; Ueda, K.; Maki, T.; Okumura, C. Arsenic accumulation in duckweed (Spirodela polyrhiza L.): A good option for phytoremediation. Chemosphere 2007, 69, 493–499. [Google Scholar] [CrossRef]
- Kumar, V.; Chopra, A.K. Phytoremediation potential of water caltrop (Trapa natans L.) using municipal wastewater of the activated sludge process-based municipal wastewater treatment plant. Environ. Technol. 2018, 39, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Eid, E.M.; Shaltout, K.H.; El-Sheikh, M.A.; Asaeda, T. Seasonal courses of nutrients and heavy metals in water, sediment and above- and below-ground Typha domingensis biomass in Lake Burullus (Egypt): Perspectives for phytoremediation. Flora 2012, 207, 783–794. [Google Scholar] [CrossRef]
- Gomes, M.V.T.; de Souza, R.R.; Teles, V.S.; Mendes, É.A. Phytoremediation of water contaminated with mercury using Typha domingensis in constructed wetland. Chemosphere 2014, 103, 228–233. [Google Scholar] [CrossRef]
- Hejna, M.; Moscatelli, A.; Stroppa, N.; Onelli, E.; Pilu, S.; Baldi, A.; Rossi, L. Bioaccumulation of heavy metals from wastewater through a Typha latifolia and Thelypteris palustris phytoremediation system. Chemosphere 2020, 241, 125018. [Google Scholar] [CrossRef]
- Li, B.; Gu, B.; Yang, Z.; Zhang, T. The role of submerged macrophytes in phytoremediation of arsenic from contaminated water: A case study on Vallisneria natans (Lour.) Hara. Ecotoxicol. Environ. Saf. 2018, 165, 224–231. [Google Scholar] [CrossRef]
- Boonyapookana, B.; Upatham, E.S.; Kruatrachue, M.; Pokethitiyook, P.; Singhakaew, S. Phytoaccumulation and Phytotoxicity of Cadmium and Chromium in Duckweed Wolffia globosa. Int. J. Phytoremediat. 2002, 4, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhao, F.; Huang, Q.; Williams, P.N.; Sun, G.; Zhu, Y. Arsenic uptake and speciation in the rootless duckweed Wolffia globosa. New Phytol. 2009, 182, 421–428. [Google Scholar] [CrossRef]
- Datta, S.; Rajnish, K.N.; Samuel, M.S.; Pugazlendhi, A.; Selvarajan, E. Metagenomic applications in microbial diversity, bioremediation, pollution monitoring, enzyme and drug discovery. A review. Environ. Chem. Lett. 2020, 18, 1229–1241. [Google Scholar] [CrossRef]
- Gałązka, A.; Grządziel, J.; Gałązka, R.; Gawryjołek, K.; Ukalska-Jaruga, A.; Smreczak, B. Fungal Community, Metabolic Diversity, and Glomalin-Related Soil Proteins (GRSP) Content in Soil Contaminated with Crude Oil after Long-Term Natural Bioremediation. Front. Microbiol. 2020, 11, 2247. [Google Scholar] [CrossRef]
- Camacho-Montealegre, C.M.; Rodrigues, E.M.; Tótola, M.R. Microbial diversity and bioremediation of rhizospheric soils from Trindade Island-Brazil. J. Environ. Manag. 2019, 236, 358–364. [Google Scholar] [CrossRef]
- Christenhusz, M.J.; Byng, J.W. The number of known plants species in the world and its annual increase. Phytotaxa 2016, 261, 201–217. [Google Scholar] [CrossRef]
- Schneller, J.J. Salviniaceae. In Pteridophytes and Gymnosperms; Springer: Berlin/Heidelberg, Germany, 1990; pp. 256–258. [Google Scholar]
- Arana, M.D. Familia SALVINIACEAE Martinov; Dykinson: Madrid, Spain, 2016; pp. 343–348. [Google Scholar]
- Cusimano, N.; Bogner, J.; Mayo, S.J.; Boyce, P.C.; Wong, S.Y.; Hesse, M.; Hetterscheid, W.L.A.; Keating, R.C.; French, J.C. Relationships within the Araceae: Comparison of morphological patterns with molecular phylogenies. Am. J. Bot. 2011, 98, 654–668. [Google Scholar] [CrossRef] [PubMed]
- Goetghebeur, P. Cyperaceae. In Flowering Plants Monocotyledons; Springer: Berlin/Heidelberg, Germany, 1998; pp. 141–190. [Google Scholar] [CrossRef]
- Saarela, J.M.; Burke, S.V.; Wysocki, W.P.; Barrett, M.D.; Clark, L.G.; Craine, J.M.; Peterson, P.M.; Soreng, R.J.; Vorontsova, M.S.; Duvall, M.R. A 250 plastome phylogeny of the grass family (Poaceae): Topological support under different data partitions. PeerJ 2018, 6, e4299. [Google Scholar] [CrossRef] [PubMed]
- Schweingruber, F.H.; Kučerová, A.; Adamec, L.; Doležal, J. Haloragaceae. In Anatomic Atlas of Aquatic and Wetland Plant Stems; Springer: Cham, Switzerland, 2020; pp. 139–142. [Google Scholar] [CrossRef]
- Semmouri, I.; Bauters, K.; Léveillé-Bourret, É.; Starr, J.R.; Goetghebeur, P.; Larridon, I. Phylogeny and Systematics of Cyperaceae, the Evolution and Importance of Embryo Morphology. Bot. Rev. 2018, 85, 1–39. [Google Scholar] [CrossRef]
- Chen, Z.; Cuervo, D.P.; Müller, J.A.; Wiessner, A.; Köser, H.; Vymazal, J.; Kästner, M.; Kuschk, P. Hydroponic root mats for wastewater treatment—A review. Environ. Sci. Pollut. Res. 2016, 23, 15911–15928. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, L.; Lobet, G.; Tocquin, P.; Périlleux, C. “Rhizoponics”: A novel hydroponic rhizotron for root system analyses on mature Arabidopsis thaliana plants. Plant Methods 2015, 11, 1–8. [Google Scholar] [CrossRef]
- Cheng, S.; Grosse, W.; Karrenbrock, F.; Thoennessen, M. Efficiency of constructed wetlands in decontamination of water polluted by heavy metals. Ecol. Eng. 2002, 18, 317–325. [Google Scholar] [CrossRef]
- Vymazal, J.; Březinová, T. Accumulation of heavy metals in aboveground biomass of Phragmites australis in horizontal flow constructed wetlands for wastewater treatment: A review. Chem. Eng. J. 2016, 290, 232–242. [Google Scholar] [CrossRef]
- Almasi, A.; Dargahi, A.; Ahagh, M.; Janjani, H.; Mohammadi, M.; Tabandeh, L. Efficiency of a constructed wetland in controlling organic pollutants, nitrogen, and heavy metals from sewage. J. Chem. Pharm. Sci. 2016, 9, 2924–2928. [Google Scholar]
- Horne, A.J. Phytoremediation by constructed wetlands. In Phytoremediation of Contaminated Soil and Water; CRC Press: Boca Raton, FL, USA, 2000. [Google Scholar]
- Guittonny-Philippe, A.; Petit, M.E.; Masotti, V.; Monnier, Y.; Malleret, L.; Coulomb, B.; Laffont-Schwob, I. Selection of wild macrophytes for use in constructed wetlands for phytoremediation of contaminant mixtures. J. Environ. Manag. 2015, 147, 108–123. [Google Scholar] [CrossRef]
- Salt, D.E.; Smith, R.D.; Raskin, I. Phytoremediation. Annu. Rev. Plant Biol. 1998, 49, 643–668. [Google Scholar] [CrossRef] [PubMed]
- Chaney, R.L.; Malik, M.; Li, Y.M.; Brown, S.L.; Brewer, E.P.; Angle, J.S.; Baker, A.J. Phytoremediation of soil metals. Curr. Opin. Biotechnol. 1997, 8, 279–284. [Google Scholar] [CrossRef]
- Flathman, P.E.; Lanza, G.R. Phytoremediation: Current Views on an Emerging Green Technology. Soil Sediment Contam. Int. J. 1998, 7, 415–432. [Google Scholar] [CrossRef]
- Peuke, A.D.; Rennenberg, H. Phytoremediation. EMBO Rep. 2005, 6, 497–501. [Google Scholar] [CrossRef] [PubMed]
- Pilon-Smits, E. Phytoremediation. Annu. Rev. Plant Biol. 2005, 56, 15–39. [Google Scholar] [CrossRef]
- Yang, X.; Feng, Y.; He, Z.; Stoffella, P.J. Molecular mechanisms of heavy metal hyperaccumulation and phytoremediation. J. Trace Elem. Med. Biol. 2005, 18, 339–353. [Google Scholar] [CrossRef] [PubMed]
- Jabeen, R.; Ahmad, A.; Iqbal, M.F. Phytoremediation of Heavy Metals: Physiological and Molecular Mechanisms. Bot. Rev. 2009, 75, 339–364. [Google Scholar] [CrossRef]
- Ali, H.; Khan, E.; Sajad, M.A. Phytoremediation of heavy metals—Concepts and applications. Chemosphere 2013, 91, 869–881. [Google Scholar] [CrossRef]
- Suman, J.; Uhlik, O.; Viktorova, J.; Macek, T. Phytoextraction of Heavy Metals: A Promising Tool for Clean-Up of Polluted Environment? Front. Plant Sci. 2018, 9, 1476. [Google Scholar] [CrossRef]
- Lombi, E.; Tearall, K.L.; Howarth, J.R.; Zhao, F.-J.; Hawkesford, M.J.; McGrath, S.P. Influence of Iron Status on Cadmium and Zinc Uptake by Different Ecotypes of the Hyperaccumulator Thlaspi caerulescens. Plant Physiol. 2002, 128, 1359–1367. [Google Scholar] [CrossRef]
- Whiting, S.N.; De Souza, A.M.P.; Terry, N. Rhizosphere Bacteria Mobilize Zn for Hyperaccumulation by Thlaspicaerulescens. Environ. Sci. Technol. 2001, 35, 3144–3150. [Google Scholar] [CrossRef] [PubMed]
- Flores, H. ‘Radicle’ biochemistry: The biology of root-specific metabolism. Trends Plant Sci. 1999, 4, 220–226. [Google Scholar] [CrossRef]
- Lin, Y.P.; Lin, C.M.; Mukhtar, H.; Lo, H.F.; Ko, M.C.; Wang, S.J. Temporal Variability in the Rhizosphere Bacterial and Fungal Community Structure in the Melon Crop Grown in a Closed Hydroponic System. Agronomy 2021, 11, 719. [Google Scholar] [CrossRef]
- Sheridan, C.; Depuydt, P.; De Ro, M.; Petit, C.; Van Gysegem, E.; Delaere, P.; Dixon, M.; Stasiak, M.; Aciksöz, S.B.; Frossard, E.; et al. Microbial Community Dynamics and Response to Plant Growth-Promoting Microorganisms in the Rhizosphere of Four Common Food Crops Cultivated in Hydroponics. Microb. Ecol. 2016, 73, 378–393. [Google Scholar] [CrossRef]
- Clarkson, D.T. Roots and the delivery of solutes to the xylem. Philos. Trans. R. Soc. B Biol. Sci. 1993, 341, 5–17. [Google Scholar] [CrossRef]
- Tester, M.; Leigh, R.A. Partitioning of nutrient transport processes in roots. J. Exp. Bot. 2001, 52, 445–457. [Google Scholar] [CrossRef]
- Cobbett, C.; Goldsbrough, P. Phytochelatins and metallothioneins: Roles in Heavy Metal Detoxification and Homeostasis. Annu. Rev. Plant Biol. 2002, 53, 159–182. [Google Scholar] [CrossRef] [PubMed]
- Dushenkov, V.; Kumar, P.B.A.N.; Motto, H.; Raskin, I. Rhizofiltration: The Use of Plants to Remove Heavy Metals from Aqueous Streams. Environ. Sci. Technol. 1995, 29, 1239–1245. [Google Scholar] [CrossRef]
- Burken, J.G. Uptake and Metabolism of Organic Compounds: Green-Liver Model. Phytoremediat. Transform. Control Contam. 2004, 59, 59–84. [Google Scholar] [CrossRef]
- Garrison, A.W.; Nzengung, V.A.; Avants, J.K.; Ellington, J.J.; Jones, W.J.; Rennels, D.; Wolfe, N.L. Phytodegradation of p,p′-DDT and the Enantiomers of o,p′-DDT. Environ. Sci. Technol. 2000, 34, 1663–1670. [Google Scholar] [CrossRef]
- Newman, L.A.; Reynolds, C.M. Phytodegradation of organic compounds. Curr. Opin. Biotechnol. 2004, 15, 225–230. [Google Scholar] [CrossRef] [PubMed]
- McCutcheon, S.C.; Schnoor, J.L. Overview of phytotransformation and control of wastes. In Phytoremediation: Transformation and Control of Contaminants; Wiley: New York, NY, USA, 2003; pp. 1–58. [Google Scholar] [CrossRef]
- Kuiper, I.; Lagendijk, E.L.; Bloemberg, G.V.; Lugtenberg, B.J.J. Rhizoremediation: A Beneficial Plant-Microbe Interaction. Mol. Plant Microbe Interact. 2004, 17, 6–15. [Google Scholar] [CrossRef]
- Nichols, T.D.; Wolf, D.C.; Rogers, H.B.; Beyrouty, C.A.; Reynolds, C.M. Rhizosphere microbial populations in contaminated soils. Water Air Soil Pollut. 1997, 95, 165–178. [Google Scholar] [CrossRef]
- Banks, M.K.; Kulakow, P.; Schwab, A.P.; Chen, Z.; Rathbone, K. Degradation of Crude Oil in the Rhizosphere of Sorghum bicolor. Int. J. Phytoremediat. 2003, 5, 225–234. [Google Scholar] [CrossRef]
- Muratova, A.; Hübner, T.; Tischer, S.; Turkovskaya, O.; Möder, M.; Kuschk, P. Plant—Rhizosphere-Microflora Association During Phytoremediation of PAH-Contaminated Soil. Int. J. Phytoremediat. 2003, 5, 137–151. [Google Scholar] [CrossRef]
- Khataee, A.; Movafeghi, A.; Torbati, S.; Lisar, S.S.; Zarei, M. Phytoremediation potential of duckweed (Lemna minor L.) in degradation of C.I. Acid Blue 92: Artificial neural network modeling. Ecotoxicol. Environ. Saf. 2012, 80, 291–298. [Google Scholar] [CrossRef]
- Mendez, M.O.; Maier, R.M. Phytostabilization of Mine Tailings in Arid and Semiarid Environments—An Emerging Remediation Technology. Environ. Health Perspect. 2008, 116, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Bolan, N.S.; Park, J.H.; Robinson, B.; Naidu, R.; Huh, K.Y. Phytostabilization: A green approach to contaminant containment. In Advances in Agronomy; Academic Press: Cambridge, MA, USA, 2011; Volume 112, pp. 145–204. [Google Scholar] [CrossRef]
- Zeng, P.; Guo, Z.; Cao, X.; Xiao, X.; Liu, Y.; Shi, L. Phytostabilization potential of ornamental plants grown in soil contaminated with cadmium. Int. J. Phytoremediat. 2018, 20, 311–320. [Google Scholar] [CrossRef]
- Cunningham, S.D.; Berti, W.R.; Huang, J.W. Phytoremediation of contaminated soils. Trends Biotechnol. 1995, 13, 393–397. [Google Scholar] [CrossRef]
- Limmer, M.A.; Burken, J.G. Phytovolatilization of Organic Contaminants. Environ. Sci. Technol. 2016, 50, 6632–6643. [Google Scholar] [CrossRef]
- Terry, N.; Zayed, A.M.; De Souza, M.P.; Tarun, A.S. Selenium in higher plants. Annu. Rev. Plant Biol. 2000, 51, 401–432. [Google Scholar] [CrossRef]
- Gordon, M.; Choe, N.; Duffy, J.; Ekuan, G.; Heilman, P.; Muiznieks, I.; Newman, L.; Ruszaj, M.; Shurtleff, B.B.; Strand, S.; et al. Phytoremediation of Trichloroethylene with Hybrid Poplars. ACS Symp. Ser. 1997, 106, 177–185. [Google Scholar] [CrossRef]
- Wan, W.; Tan, J.; Wang, Y.; Qin, Y.; He, H.; Wu, H.; Zuo, W.; He, D. Responses of the rhizosphere bacterial community in acidic crop soil to pH: Changes in diversity, composition, interaction, and function. Sci. Total Environ. 2020, 700, 134418. [Google Scholar] [CrossRef] [PubMed]
- Ju, W.; Liu, L.; Jin, X.; Duan, C.; Cui, Y.; Wang, J.; Ma, D.; Zhao, W.; Wang, Y.; Fang, L. Co-inoculation effect of plant-growth-promoting rhizobacteria and rhizobium on EDDS assisted phytoremediation of Cu contaminated soils. Chemosphere 2020, 254, 126724. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A. Role of PGPR in the Phytoremediation of Heavy Metals and Crop Growth under Municipal Wastewater Irrigation. In Phytoremediation (Management of Environmental Contaminants, Volume 6); Ansari, A.A., Gill, S.S., Gill, R., Lanza, G.R., Newman, L., Eds.; Springer: Cham, Switzerland, 2018; pp. 135–149. [Google Scholar] [CrossRef]
- Guarino, F.; Miranda, A.; Castiglione, S.; Cicatelli, A. Arsenic phytovolatilization and epigenetic modifications in Arundo donax L. assisted by a PGPR consortium. Chemosphere 2020, 251, 126310. [Google Scholar] [CrossRef]
- Nadeem, N.; Asif, R.; Ayyub, S.; Salman, S.; Shafique, F.; Ali, Q.; Malik, A. Role of rhizobacteria in phytoremediation of heavy metals. Biol. Clin. Sci. Res. J. 2020, 2020, e035. [Google Scholar]
- Khan, N.; Zandi, P.; Ali, S.; Mehmood, A.; Shahid, M.A.; Yang, J. Impact of Salicylic Acid and PGPR on the Drought Tolerance and Phytoremediation Potential of Helianthus annus. Front. Microbiol. 2018, 9, 2507. [Google Scholar] [CrossRef] [PubMed]
- Mesa-Marín, J.; Del-Saz, N.F.; Rodríguez-Llorente, I.D.; Redondo-Gómez, S.; Pajuelo, E.; Ribas-Carbó, M.; Mateos-Naranjo, E. PGPR Reduce Root Respiration and Oxidative Stress Enhancing Spartina maritima Root Growth and Heavy Metal Rhizoaccumulation. Front. Plant Sci. 2018, 9, 1500. [Google Scholar] [CrossRef]
- Sang, M.; Jeong, J.-J.; Kim, J.; Kim, K. Growth promotion and root colonisation in pepper plants by phosphate-solubilising Chryseobacterium sp. strain ISE14 that suppresses Phytophthora blight. Ann. Appl. Biol. 2018, 172, 208–223. [Google Scholar] [CrossRef]
- Hansda, A.; Kumar, V.; Anshumali, A.; Usmani, Z. Phytoremediation of heavy metals contaminated soil using plant growth promoting rhizobacteria (PGPR): A current perspective. Recent Res. Sci. Technol. 2014, 6, 131–134. [Google Scholar]
- Patel, P.R.; Shaikh, S.S.; Sayyed, R.Z. Dynamism of PGPR in bioremediation and plant growth promotion in heavy metal contaminated soil. Indian J. Exp. Boil. 2016, 54, 286–290. [Google Scholar]
- Javed, M.T.; Tanwir, K.; Akram, M.S.; Shahid, M.; Niazi, N.K.; Lindberg, S. Phytoremediation of Cadmium-Polluted Water/Sediment by Aquatic Macrophytes: Role of Plant-Induced pH Changes. Cadmium Toxic. Toler. Plants 2019, 495–529. [Google Scholar] [CrossRef]
- Aisien, F.A.; Faleye, O.; Aisien, E.T. Phytoremediation of heavy metals in aqueous solutions. Leonardo J. Sci. 2010, 17, 37–46. [Google Scholar]
- Lu, Q.; He, Z.L.; Graetz, D.A.; Stoffella, P.J.; Yang, X. Phytoremediation to remove nutrients and improve eutrophic stormwaters using water lettuce (Pistia stratiotes L.). Environ. Sci. Pollut. Res. 2010, 17, 84–96. [Google Scholar] [CrossRef]
- Devi, S.; Nandwal, A.; Angrish, R.; Arya, S.; Kumar, N.; Sharma, S. Phytoremediation potential of some halophytic species for soil salinity. Int. J. Phytoremediat. 2015, 18, 693–696. [Google Scholar] [CrossRef] [PubMed]
- Rehman, S.; Abbas, G.; Shahid, M.; Saqib, M.; Farooq, A.B.U.; Hussain, M.; Murtaza, B.; Amjad, M.; Naeem, M.A.; Farooq, A. Effect of salinity on cadmium tolerance, ionic homeostasis and oxidative stress responses in conocarpus exposed to cadmium stress: Implications for phytoremediation. Ecotoxicol. Environ. Saf. 2019, 171, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.S.; Kadier, A.; Malyan, S.K.; Ahmad, A.; Bishnoi, N.R. Phytoremediation and rhizoremediation: Uptake, mobilization and sequestration of heavy metals by plants. In Plant-Microbe Interactions in Agro-Ecological Perspectives; Singh, D., Singh, H., Prabha, R., Eds.; Springer: Singapore, 2017; pp. 367–394. [Google Scholar]
- Yavari, S.; Malakahmad, A.; Sapari, N.B.; Yavari, S. Nutrients balance for improvement of phytoremediation ability of teak seedlings (Tectona grandis). J. Plant Nutr. 2018, 41, 545–551. [Google Scholar] [CrossRef]
- Al-Baldawi, I.A.; Abdullah, S.R.S.; Anuar, N.; Abu Hasan, H. Phytotransformation of methylene blue from water using aquatic plant (Azolla pinnata). Environ. Technol. Innov. 2018, 11, 15–22. [Google Scholar] [CrossRef]
- Kumar, V.; Kumar, P.; Singh, J.; Kumar, P. Potential of water fern (Azolla pinnata R.Br.) in phytoremediation of integrated industrial effluent of SIIDCUL, Haridwar, India: Removal of physicochemical and heavy metal pollutants. Int. J. Phytoremediat. 2019, 22, 392–403. [Google Scholar] [CrossRef]
- Odjegba, V.J.; Fasidi, I.O. Accumulation of Trace Elements by Pistia stratiotes: Implications for phytoremediation. Ecotoxicology 2004, 13, 637–646. [Google Scholar] [CrossRef]
- Su, C.; Jiang, Y.; Li, F.; Yang, Y.; Lu, Q.; Zhang, T.; Hu, D.; Xu, Q. Investigation of subcellular distribution, physiological, and biochemical changes in Spirodela polyrhiza as a function of cadmium exposure. Environ. Exp. Bot. 2017, 142, 24–33. [Google Scholar] [CrossRef]
- Singh, V.; Pandey, B.; Suthar, S. Phytotoxicity and degradation of antibiotic ofloxacin in duckweed (Spirodela polyrhiza) system. Ecotoxicol. Environ. Saf. 2019, 179, 88–95. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Langenhoff, A.A.M.; Sutton, N.B.; Rijnaarts, H.H.M.; Blokland, M.H.; Chen, F.; Huber, C.; Schröder, P. Metabolism of Ibuprofen by Phragmites australis: Uptake and Phytodegradation. Environ. Sci. Technol. 2017, 51, 4576–4584. [Google Scholar] [CrossRef]
- Rezania, S.; Park, J.; Rupani, P.F.; Darajeh, N.; Xu, X.; Shahrokhishahraki, R. Phytoremediation potential and control of Phragmites australis as a green phytomass: An overview. Environ. Sci. Pollut. Res. 2019, 26, 7428–7441. [Google Scholar] [CrossRef]
- Ravi, R.; Rajendran, D.; Oh, W.-D.; Rasat, M.S.M.; Hamzah, Z.; Ishak, I.H.; Amin, M.F.M. The potential use of Azolla pinnata as an alternative bio-insecticide. Sci. Rep. 2020, 10, 19245. [Google Scholar] [CrossRef] [PubMed]
- Das, M.; Ibn Rahim, F.; Hossain, A. Evaluation of Fresh Azolla pinnata as a Low-Cost Supplemental Feed for Thai Silver Barb Barbonymus gonionotus. Fishes 2018, 3, 15. [Google Scholar] [CrossRef]
- Elrasoul, A.S.A.; Mousa, A.A.; Orabi, S.H.; Mohamed, M.A.E.-G.; Gad-Allah, S.M.; Almeer, R.; Abdel-Daim, M.M.; Khalifa, S.A.M.; El-Seedi, H.R.; Eldaim, M.A.A. Antioxidant, Anti-Inflammatory, and Anti-Apoptotic Effects of Azolla pinnata Ethanolic Extract against Lead-Induced Hepatotoxicity in Rats. Antioxidants 2020, 9, 1014. [Google Scholar] [CrossRef]
- Tyagi, T.; Agarwal, M. Phytochemical screening and GC-MS analysis of bioactive constituents in the ethanolic extract of Pistia stratiotes L. and Eichhornia crassipes (Mart.) solms. J. Pharmacogn. Phytochem. 2017, 6, 195–206. [Google Scholar]
- Kadir, A.A.; Abdullah, S.R.S.; Othman, B.A.; Abu Hasan, H.; Othman, A.R.; Imron, M.F.; Izzati Ismail, N.; Kurniawan, S.B. Dual function of Lemna minor and Azolla pinnata as phytoremediator for Palm Oil Mill Effluent and as feedstock. Chemosphere 2020, 259, 127468. [Google Scholar] [CrossRef]
- Casebier, K. Water Purification System for Storm Drain Channels. U.S. Patent US7686956B1, 30 March 2010. [Google Scholar]
- Sakakibara, Y.; Rodrigues dos, R.A.; Inagaki, Y.; Komori, M. Water Treatment Method Using Aquatic Plant. Japanese Patent WO2012029736A1, 8 March 2012. Available online: https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2012029736 (accessed on 13 January 2021).
- Na, Z.; Yang, H.; Na, Z. Eichhornia Crassipes with Purple Root and Technology for Purifying Algae-Type Eutrophic Contaminated Water Bodies at Source. Chinese Patent CN102524084A, 4 July 2012. [Google Scholar]
- Ma, Y.; Li, J.; Pan, B.; Zhou, H.; Ye, Z.; Shang, Y. Method for Removing Nitrogen and Phosphorus in Water Body by Hydrilla Verticillate. Chinese Patent CN102311173A, 11 January 2012. [Google Scholar]
- Bauer, H.; Kozianka, F.; Endisch, S. Wastewater Purification Plant by Means of Plants. U.S. Patent US7718062B2, 18 May 2010. [Google Scholar]
- Grieco, W.J. Marine Biomass Reactor and Methods Related Thereto. U.S. Patent US20180206423A1, 26 July 2018. [Google Scholar]
- Ogden, M.H. Waste Treatment Systems. U.S. Patent US7361268B2, 22 April 2008. [Google Scholar]
- Wallace, S.D.; Lambrecht, P.E. System and Method for Removing Pollutants from Water. U.S. Patent US6652743B2, 25 November 2003. [Google Scholar]
- Gong, S. Method for Removing Soil Arsenic Pollution by Interplanting Pteris Vittate. Chinese Patent CN105945042A, 21 September 2016. [Google Scholar]
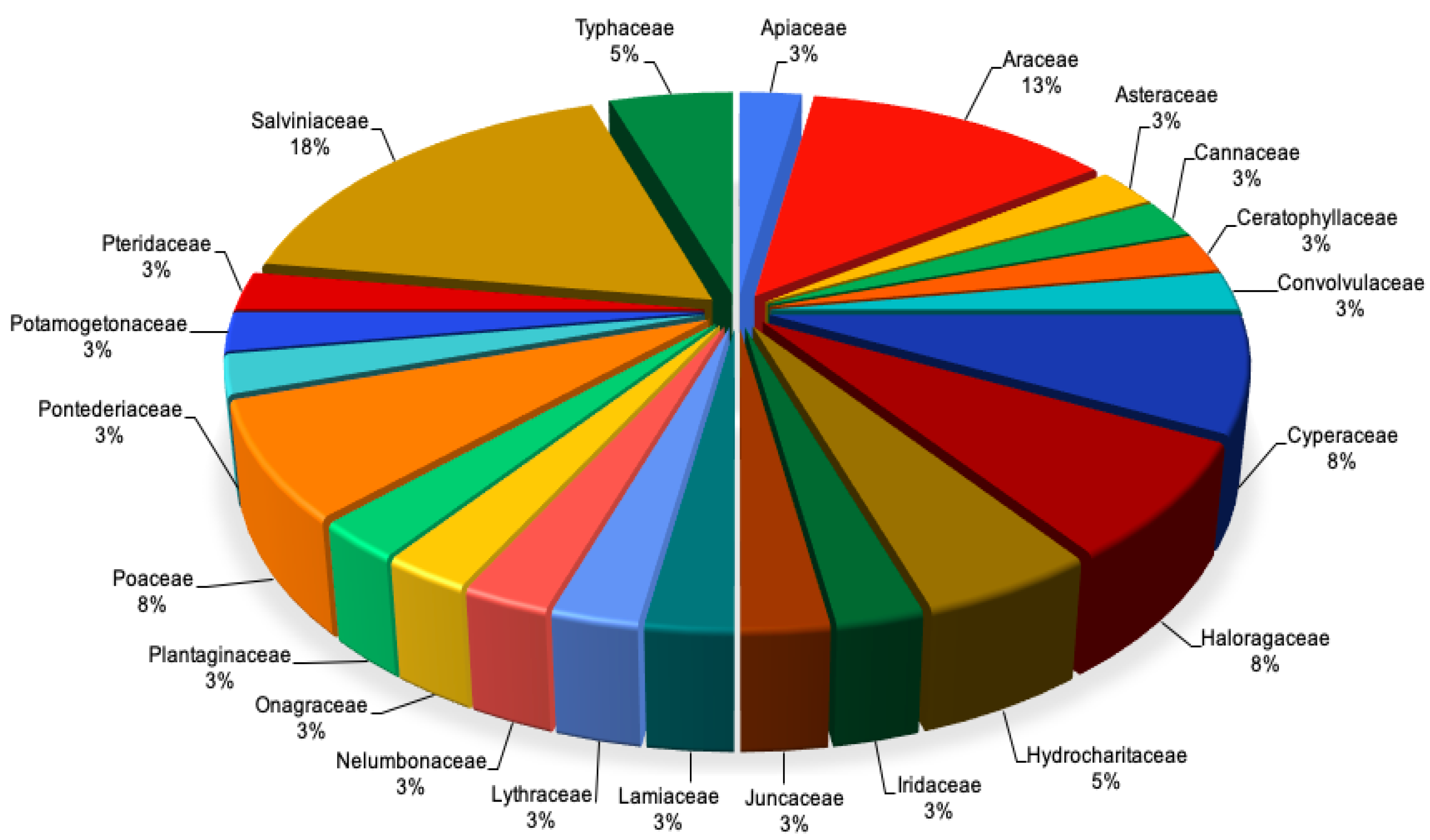
| Plant Species | Family | Country | Contaminants Treated | Phytoremediation Process | Results | References |
|---|---|---|---|---|---|---|
| Arundo donax | Poaceae | Pakistan | As | Phytoextraction | Removal of at least 15% of the pollutant in the treatment of 600 μg L−1 | [35] |
| Azolla caroliniana | Salvi-niaceae | India | Heavy metals in metal enriched fly ash pond (Cr, Pb, Cu and Ni) | Phytoaccumula-tion | High sequestration of metals (175–538 and 86–753 mg kg−1 plant tissue) BCF 1.7–18.6 and 1.8–11.0. | [36] |
| Azolla filiculoides | Salvi-niaceae | Chile, Israel | Cd, Cu, Pb | Phytoextraction | High concentration in plant tissues, more than 1000 micrograms per kg−1, | [37,38] |
| Azolla pinnata | Salvi-niaceae | India, Nigeria | Hg, Cd, Zi, Fe | Phytoextraction | Metal content decreased to 70–94%, there is no significant removal of Fe, but Zi decreased more than 30% | [39] |
| Canna indica | Cannaceae | India | F | Phytoaccumula-tion | 95% fluoride removal | [40] |
| Ceratophyllum demersum | Ceratophyllaceae | Egypt | Cr, Pb | Phytoaccumula-tion | 95% removal of lead and 84% of chromium | [41] |
| Cyperus alternifolius | Cyperaceae | India | F | Phytoaccumula-tion | 65% fluoride removal | [40] |
| Eichhornia crassipes | Pontederiaceae | India, Nigeria | As, Hg, Ni, Pb, Zn, Cu, Ag | Phytoaccumula-tion | Acummulation from 26 mg/kg to 327 mg/kg in dry weight | [42,43] |
| Eleocharis acicularis | Cyperaceae | Japan | Cu, Zn, As, Cd, Pb | Phytoextraction | Remotion higher than 90% of the heavy metals | [44] |
| Helianthus annuus | Asteraceae | Pakistan | Ni, Pb | Phytoextraction | More than 50% of removal, 17 mg Kg−1 in plant tissue | [45] |
| Hydrilla verticillata | Hydro-charitaceae | India, China | F, As, and other heavy metals | Phytoaccumula-tion, Phyto-degradation | Maximum removal 24.4% at 2.5 ppm without dramatically affecting associated physiological parameters, and the resultant degradation products are non-toxic | [46,47,48] |
| Ipomoea aquatica | Convolvulaceae | Iran, Sri Lanka | Pb, Cr | Rhizofiltration | The highest BCF (4179.07) value was registered in root tissue (0.63 mg L−1 Pb) More than 90% Cr(VI) sequestrated in leaves and steams. In none of the Cr(VI) dosing experiments did the I. aquatica show toxicity symptoms. | [49,50] |
| Iris pseuda-corus | Iridaceae | Spain | Cr, Zn | Rhizofiltration | 59.97 mg Cr and 25.64 mg Zn in roots | [51] |
| Juncus effusus | Juncaceae | China | Pb | Phytodegradation | Concentrations higher than 2000 mg kg−1 in roots | [52] |
| Lemna gibba | Araceae | Germany | U, As | Phytoextraction | Accumulation in plant tissue, around 500 mg kg−1 | [53] |
| Lemna minor | Araceae | Pakistan, Iran | Heavy metals in contaminated effluents | Phytoaccumula-tion | Considerable reduction in every metal in municipal effluent | [27] |
| Lepironia articulata | Cyperaceae | USA | Pb | Rhizofiltration | More than 500 mg/kg in its plant tissue (roots) and 217 of BCF value | [54] |
| Lolium perenne | Poaceae | France | Cr | Phytostabilization | High accumulation in roots, higuer than 2000 μg−1 DW | [55] |
| Ludwigia stolonifera | Onagra-ceae | Egypt | Cd, Ni, Zn, Pb | Phytostabilization | Bioaccumulation and translocation factor showed positive interaction for the uptake of metals highlighted | [56] |
| Mentha aquatica | Lamiaceae | Lebanon | Ni | Rhizofiltration | 8327 mg kg−1 accumulated mainly in root tissue | [57] |
| Myrio-phyllum aquaticum | Haloragaceae | Italy | Cd, Cr, Ni, Zn | Phytoaccumulation | High accumulation in plant tisssue at high concentrations, more than 500 μg g−1 DW | [58] |
| Myrio-phyllum triphyllum | Haloragaceae | Turkey | Cd | Phytoaccumu-lation | 17.03 μg Cd accumulation was found in a gram in dried sample | [59] |
| Myrio-phyllum elatinoides | Haloragaceae | China | B | Phytoaccumulation | Maximal tissue accumulation in shoot tissue and root section (1296.5 and 350.7 mg/kg, each one) | [60] |
| Nelumbo nucifera | Nelum-bona-ceae | India | Cd, Co, Cu, Ni, Pb and Zn | Phytoextraction | Accumulation in tissue more than 340 ppm of metals | [61] |
| Oenanthe javanica | Apiaceae | USA | Hg | Phytoaccumulation | More than 1 mg/kg remediated and 807 of BCF value | [62] |
| Phragmites australis | Poaceae | Saudi Arabia, Denmark | Cd, Pb, Ni | Rhizofiltration | High concentration in roots, more than 3 mg kg−1 | [63] |
| Pistia stratiotes | Araceae | USA, India | Cd, Cu, Fe, Hg | Phytoextraction and rhizofiltration | Accumulation of Cd in roots (more than 10 mg kg−1), Cu, Fe and Hg concentrations from 1 to 15 mg kg−1 DW. | [64,65] |
| Plantago major | Plantaginaceae | Switzerland | Pb | Rhizofiltration | High uptake, more than 20 mg/kg of Pb in root tissue | [66] |
| Potamo-geton natans | Potamogetonaceae | Sweden | Zn, Cu, Cd, Pb | Rhizofiltration | Highest accumulation found in the roots | [67] |
| Pteris vittata | Pteridaceae | USA | As | Phytoaccumulation | Reduced arsenic concentration by 98.6% | [68] |
| Salvinia biloba | Salviniaceae | Brazil | Pb | Phytoextraction | Almost 90% of Pb remotion | [69] |
| Salvinia minima | Salviniaceae | Mexico | Pb, As | Phytoaccumu-lation | More than 34 mg/g Pb in dry weight tissue and high As uptake, with 0.5 mg/g DW). | [70] |
| Salvinia molesta | Salviniaceae | Brazil | As | Phytoaccumu-lation | Accumulation in leaves, highest accumulation 148.63 μg g−1 DW | [71] |
| Salvinia natans | Salviniaceae | India | Zn, Cu, Ni, Cr | Phytoaccumu-lation | High removal, more than 50% average for each metal | [72] |
| Spirodela polyrhiza | Araceae | Japan | As | Phytoaccumu-lation | Accumulations on DW tissue higher than 0.35 μmol/g for arsenate and around 7.6 nmol/g DW for DMAA | [73] |
| Trapa natans | Lythraceae | India | Heavy metals in wastewater | Phytoaccumu-lation | Metal contents translocated in leaves, whereas most contents of Cr and Pb were accumulated in the root. | [74] |
| Typha domin-gensis | Typhaceae | Egypt, Brazil | P, Na, K, Zn, Hg | Phytoextraction | Reduced P, Na, K almost in 80%, reduced Zn in 10% with respect to initial values, Reduces 99.6 ± 0.4% of the mercury in contaminated water | [75,76] |
| Typha latifolia | Typhaceae | Italy | Cu, Zn | Phytoextraction | Higher accumulation of Zinc, more than 55 mg Kg DW in root tissue | [77] |
| Vallisneria natans | Hydrocharitaceae | China | As | Rhizo-filtration | High accumulation in roots (more than 200 mg/kg−1 DW of As (IV)) | [78] |
| Wolffia globosa | Araceae | China, Thailand | As, Cd, Cr | Phyto-accumu-lation | Accumulate more than 1000 mg As kg−1 in DW tissue, Max accumulation Cd 5931 µg/g DW. 3500 µg/g DW Cr | [79,80] |
| Microorganism | Process | Reference |
|---|---|---|
| PGPR (Paenibacillus mucilaginosus, Sinorhizobium meliloti) | Increase the bioavailability of metals | [134] |
| PGPR (Pseudomonas spp.) | Increase water uptake in roots, increasing HM mobilization | [135] |
| PGPR (Stenotrophomonas maltophilia) | Reduce toxicity of HMs, increasing bioaccumulation factor (BF) | [136] |
| PGPR (non specified) | Transformation of HMs into less toxic compounds for faster uptake | [137] |
| PGPR (Planomicrobium chinense, Bacillus cereus) | Increase biomass gain and root growth during HM stress | [138] |
| PGPR (Bacillus spp.) | Reduction in oxidative stress, increasing metabolite production | [139] |
| Chryseobacterium sp. | Creation of antagonistic metabolites to improve resistance to pathogens | [140] |
| PGPR (Pseudomonas fluorescence, Bacillus subtilis) | Increase HM uptake, especially Pb and Ni | [141] |
| Plant Species | Patent | Patent Number | Reference |
|---|---|---|---|
| Azolla pinnata | Water purification system | EP0333218B1 | [162] |
| Spirodela polyrhiza | Purification method of wastewater | WO2012029736A1 | [163] |
| Eichhornia crassipes | Purifying algae-type eutrophic contaminated water bodies at a source | CN102524084A | [164] |
| Hydrilla verticillata | The invention discloses a method for removing nitrogen and phosphorus in a water body | CN102311173A | [165] |
| Iris pseudacorus | Waste-water purification plant | US7718062B2 | [166] |
| Myriophyllum triphyllum | Marine biomass reactor | WO2018140449A1 | [167] |
| Phragmites australis | Waste treatment systems, biological restoration of water body, system and method for removal of pollutants from water | US7361268B2 | [168] |
| Potamogeton natans | Method for repairing water ecology, purifying method, waste treatment process | US6652743B2 | [169] |
| Pteris vittata | Method for removing arsenic from soil and water | CN105945042A | [170] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Delgado-González, C.R.; Madariaga-Navarrete, A.; Fernández-Cortés, J.M.; Islas-Pelcastre, M.; Oza, G.; Iqbal, H.M.N.; Sharma, A. Advances and Applications of Water Phytoremediation: A Potential Biotechnological Approach for the Treatment of Heavy Metals from Contaminated Water. Int. J. Environ. Res. Public Health 2021, 18, 5215. https://doi.org/10.3390/ijerph18105215
Delgado-González CR, Madariaga-Navarrete A, Fernández-Cortés JM, Islas-Pelcastre M, Oza G, Iqbal HMN, Sharma A. Advances and Applications of Water Phytoremediation: A Potential Biotechnological Approach for the Treatment of Heavy Metals from Contaminated Water. International Journal of Environmental Research and Public Health. 2021; 18(10):5215. https://doi.org/10.3390/ijerph18105215
Chicago/Turabian StyleDelgado-González, Cristián Raziel, Alfredo Madariaga-Navarrete, José Miguel Fernández-Cortés, Margarita Islas-Pelcastre, Goldie Oza, Hafiz M. N. Iqbal, and Ashutosh Sharma. 2021. "Advances and Applications of Water Phytoremediation: A Potential Biotechnological Approach for the Treatment of Heavy Metals from Contaminated Water" International Journal of Environmental Research and Public Health 18, no. 10: 5215. https://doi.org/10.3390/ijerph18105215
APA StyleDelgado-González, C. R., Madariaga-Navarrete, A., Fernández-Cortés, J. M., Islas-Pelcastre, M., Oza, G., Iqbal, H. M. N., & Sharma, A. (2021). Advances and Applications of Water Phytoremediation: A Potential Biotechnological Approach for the Treatment of Heavy Metals from Contaminated Water. International Journal of Environmental Research and Public Health, 18(10), 5215. https://doi.org/10.3390/ijerph18105215











