Analysis of Cumulative Cancer Risk Associated with Disinfection Byproducts in United States Drinking Water
Abstract
1. Introduction
2. Materials and Methods
2.1. Exposure Assessment for Trihalomethanes and Haloacetic Acids in Drinking Water
2.2. Calculation of Lifetime Cancer Risk Based on Toxicological Studies of Disinfection Byproducts
2.3. Calculation of Lifetime Cancer Risk Based on Epidemiological Studies of Disinfection Byproducts
2.4. Calculation of Economic Costs due to Estimated Bladder Cancer Cases Associated with Disinfection Byproducts
3. Results
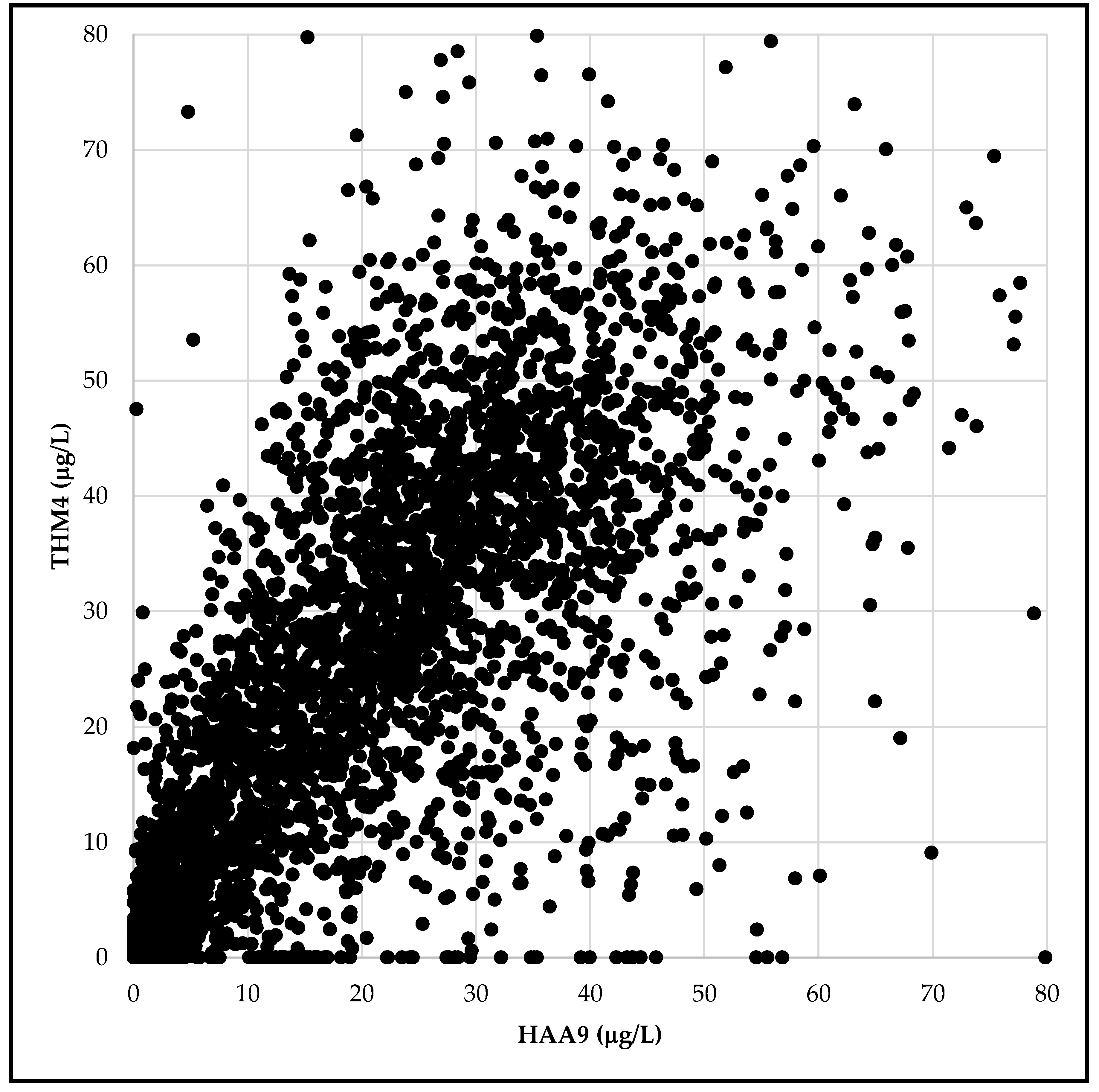
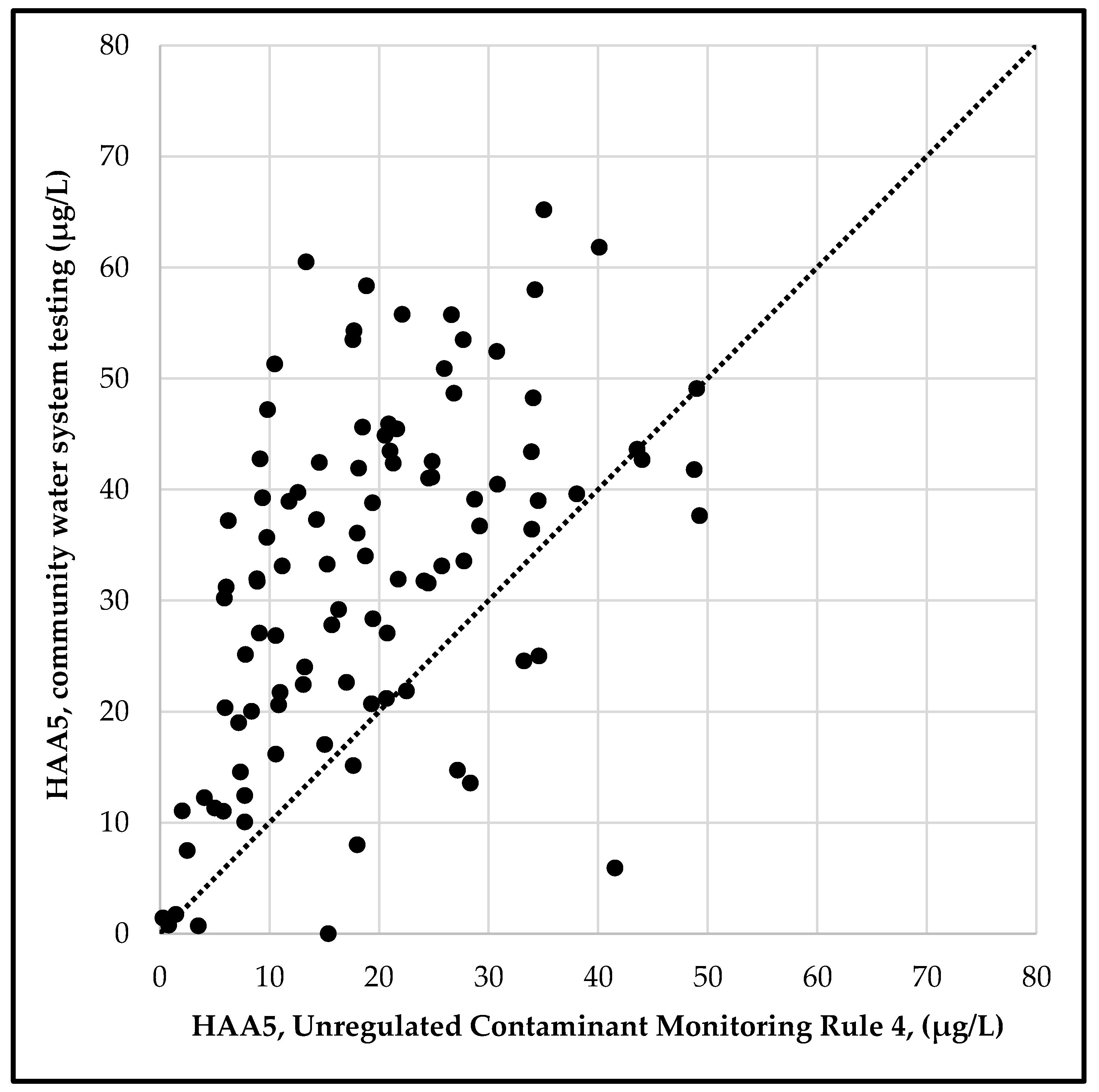
3.1. Exposure Assessment for Trihalomethanes and Haloacetic Acids in Drinking Water
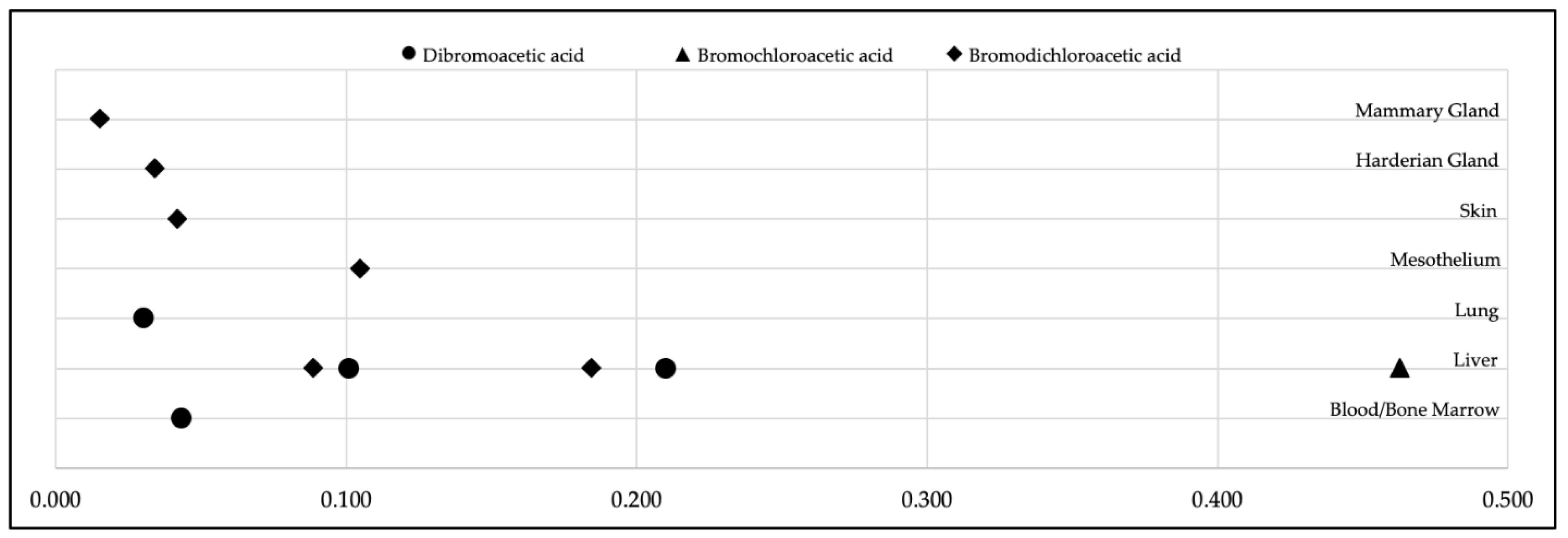
3.2. Calculation of Lifetime Cancer Risk Based on Toxicological Studies of Disinfection Byproducts
3.3. Calculation of Annual and Lifetime Cancer Risk Based on Epidemiological Studies of Disinfection Byproducts
3.4. Calculation of Economic Costs due to Estimated Bladder Cancer Cases Associated with Disinfection Byproducts
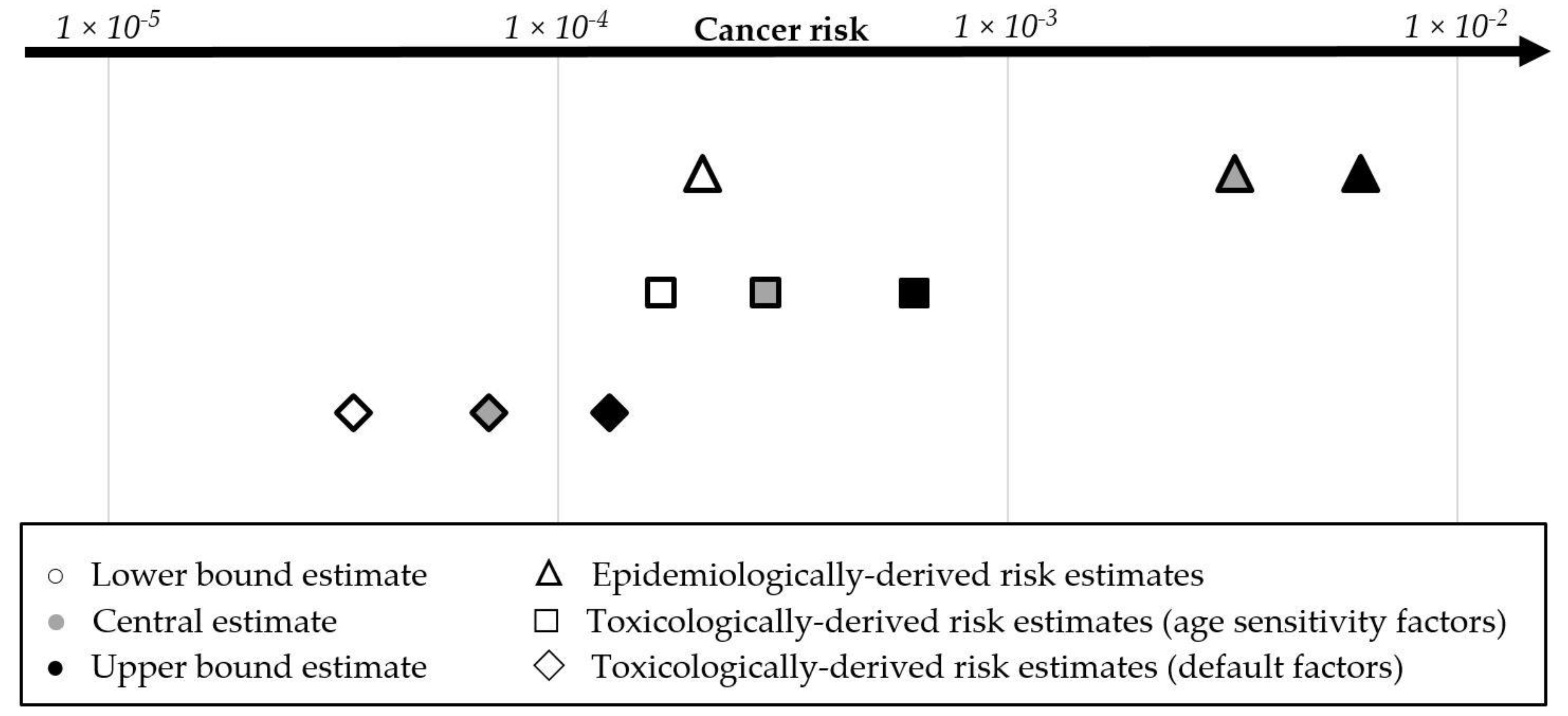
3.5. Comparison of Epidemiologically-Based and Toxicologically Based Cancer Risk Estimates
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Diana, M.; Felipe-Sotelo, M.; Bond, T. Disinfection byproducts potentially responsible for the association between chlorinated drinking water and bladder cancer: A review. Water Res. 2019, 162, 492–504. [Google Scholar] [CrossRef]
- Von Gunten, U. Oxidation Processes in Water Treatment: Are We on Track? Environ. Sci. Technol. 2018, 52, 5062–5075. [Google Scholar] [CrossRef]
- Villanueva, C.M.; Cantor, K.P.; Cordier, S.; Jaakkola, J.J.; King, W.D.; Lynch, C.F.; Porru, S.; Kogevinas, M. Disinfection byproducts and bladder cancer: A pooled analysis. Epidemiology 2004, 15, 357–367. [Google Scholar] [CrossRef]
- Beane-Freeman, L.E.; Cantor, K.P.; Baris, D.; Nuckols, J.R.; Johnson, A.; Colt, J.S.; Schwenn, M.; Ward, M.H.; Lubin, J.H.; Waddell, R.; et al. Bladder Cancer and Water Disinfection By-product Exposures through Multiple Routes: A Population-Based Case Control Study (New England, USA). Environ. Health Perspect. 2017, 125, 067010. [Google Scholar] [CrossRef]
- Costet, N.; Villanueva, C.M.; Jaakkola, J.J.; Kogevinas, M.; Cantor, K.P.; King, W.D.; Lynch, C.F.; Nieuwenhuijsen, M.J.; Cordier, S. Water disinfection by-products and bladder cancer: Is there a European specificity? A pooled and meta-analysis of European case-control studies. Occup. Environ. Med. 2011, 68, 379–385. [Google Scholar] [CrossRef]
- Regli, S.; Chen, J.; Messner, M.; Elovitz, M.S.; Letkiewicz, F.J.; Pegram, R.A.; Pepping, T.J.; Richardson, S.D.; Wright, J.M. Estimating Potential Increased Bladder Cancer Risk Due to Increased Bromide Concentrations in Sources of Disinfected Drinking Waters. Environ. Sci. Technol. 2015, 49, 13094–13102. [Google Scholar] [CrossRef]
- Karagas, M.R.; Villanueva, C.M.; Nieuwenhuijsen, M.; Weisel, C.P.; Cantor, K.P.; Kogevinas, M. Disinfection byproducts in drinking water and skin cancer? A hypothesis. Cancer Causes Control 2008, 19, 547–548. [Google Scholar] [CrossRef][Green Version]
- Villanueva, C.M.; Gracia-Lavedan, E.; Bosetti, C.; Righi, E.; Molina, A.J.; Martin, V.; Boldo, E.; Aragones, N.; Perez-Gomez, B.; Pollan, M.; et al. Colorectal Cancer and Long-Term Exposure to Trihalomethanes in Drinking Water: A Multicenter Case-Control Study in Spain and Italy. Environ. Health Perspect. 2017, 125, 56–65. [Google Scholar] [CrossRef]
- Wright, J.M.; Evans, A.; Kaufman, J.A.; Rivera-Nunez, Z.; Narotsky, M.G. Disinfection By-Product Exposures and the Risk of Specific Cardiac Birth Defects. Environ. Health Perspect. 2017, 125, 269–277. [Google Scholar] [CrossRef]
- Waller, K.; Swan, S.H.; DeLorenze, G.; Hopkins, B. Trihalomethanes in drinking water and spontaneous abortion. Epidemiology 1998, 9, 134–140. [Google Scholar] [CrossRef]
- Chaves, R.S.; Guerreiro, C.S.; Cardoso, V.V.; Benoliel, M.J.; Santos, M.M. Hazard and mode of action of disinfection by-products (DBPs) in water for human consumption: Evidences and research priorities. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2019, 223, 53–61. [Google Scholar] [CrossRef]
- Roberson, J.A.; Cromwell, J.E.I.; Krasner, S.W.; McGuire, M.J.; Owen, D.M.; Regli, S.; Summers, R.S. The D/DBP Rule: Where did the numbers come from? J. Am. Water Works Assoc. 1995, 87, 46–57. [Google Scholar] [CrossRef]
- Ding, S.; Deng, Y.; Bond, T.; Fang, C.; Cao, Z.; Chu, W. Disinfection byproduct formation during drinking water treatment and distribution: A review of unintended effects of engineering agents and materials. Water Res. 2019, 160, 313–329. [Google Scholar] [CrossRef]
- Li, X.-F.; Mitch, W.A. Drinking Water Disinfection Byproducts (DBPs) and Human Health Effects: Multidisciplinary Challenges and Opportunities. Environ. Sci. Technol. 2018, 52, 1681–1689. [Google Scholar] [CrossRef]
- Environmental Working Group. EWG’s Tap Water Database 2019 Update. Available online: https://www.ewg.org/tapwater/ (accessed on 3 March 2020).
- Evans, S.; Campbell, C.; Naidenko, O.V. Cumulative risk analysis of carcinogenic contaminants in United States drinking water. Heliyon 2019. [Google Scholar] [CrossRef]
- Stoiber, T.; Temkin, A.; Andrews, D.; Campbell, C.; Naidenko, O.V. Applying a cumulative risk framework to drinking water assessment: A commentary. Environ. Health 2019, 18, 37. [Google Scholar] [CrossRef]
- Schaider, L.A.; Swetschinski, L.; Campbell, C.; Rudel, R.A. Environmental justice and drinking water quality: Are there socioeconomic disparities in nitrate levels in U.S. drinking water? Environ. Health 2019, 18, 3. [Google Scholar] [CrossRef]
- Temkin, A.; Evans, S.; Manidis, T.; Campbell, C.; Naidenko, O.V. Exposure-based assessment and economic valuation of adverse birth outcomes and cancer risk due to nitrate in United States drinking water. Environ. Res. 2019, 108442. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. Occurrence Data for the Unregulated Contaminant Monitoring Rule; 2020. Available online: https://www.epa.gov/dwucmr/occurrence-data-unregulated-contaminant-monitoring-rule#4 (accessed on 3 March 2020).
- United States Environmental Protection Agency. Envirofacts; US EPA: Washington, DC, USA, 2017.
- United States Census Bureau. American Fact Finder. Available online: https://factfinder.census.gov/faces/nav/jsf/pages/index.xhtml (accessed on 3 March 2020).
- United States Geological Survey. Estimated Use of Water in the United States in 2005; 2005. Available online: https://pubs.usgs.gov/circ/1344/pdf/c1344.pdf (accessed on 3 March 2020).
- United States Environmental Protection Agency. Benchmark Dose Software Version 2.7. Available online: https://www.epa.gov/bmds (accessed on 3 March 2020).
- National Toxicology Program. NTP Technical Report on the Toxicology and Carcinogenesis Studies of Dibromoacetic Acid in F344/N Rats and B6C3F1 Mice; United States Department of Health and Human Services: Washington, DC, USA, 2007.
- National Toxicology Program. NTP Technical Report on the Toxicology and Carcinogenesis Studies of Bromochloroacetic Acid in F344/N Rats and B6C3F1 Mice; United States Department of Health and Human Services: Washington, DC, USA, 2009.
- National Toxicology Program. NTP Technical Report on the Toxicology Studies of Bromodichloroacetic Acid in F344/N Rats and B6C3F1/N Mice and Toxicology and Carcinogenesis Studies of Bromodichloroacetic Acid in F344/NTac Rats and B6C3F1/N Mice; United States Department of Health and Human Services: Washington, DC, USA, 2015.
- United States Environmental Protection Agency. Benchmark Dose Technical Guidance; United States Environmental Protection Agency: Washington, DC, USA, 2012. Available online: https://www.epa.gov/risk/benchmark-dose-technical-guidance (accessed on 3 March 2020).
- United States Environmental Protection Agency. Benchmark Dose Software; US EPA: Washington, DC, USA, 2009.
- California Office of Environmental Health Hazard Assessment. First Public Review Draft, Haloacetic Acids in Drinking Water; 2020. Available online: https://oehha.ca.gov/water/crnr/announcement-availability-draft-technical-support-document-and-public-workshop-proposed (accessed on 31 January 2020).
- United States Environmental Protection Agency. Human Health Benchmarks for Pesticides: Updated 2017 Technical Document; 2017. Available online: https://www.epa.gov/sdwa/human-health-benchmarks-pesticides-drinking-water (accessed on 3 March 2020).
- Kahn, H.D.; Stralka, K. Estimated daily average per capita water ingestion by child and adult age categories based on USDA’s 1994-1996 and 1998 continuing survey of food intakes by individuals. J. Expo. Sci. Environ. Epidemiol 2009, 19, 396–404. [Google Scholar] [CrossRef]
- Fryar, C.D.; Kruszon-Moran, D.; Gu, Q.; Ogden, C.L. Mean Body Weight, Height, Waist Circumference, and Body Mass Index Among Adults: United States, 1999–2000 through 2015–2016. 2018. Available online: https://stacks.cdc.gov/view/cdc/61430 (accessed on 3 March 2020).
- United States Environmental Protection Agency. Estimated Per Capita Water Ingestion and Body Weight in the United States—An Update; EPA-822-R-00-001; 2004. Available online: https://hero.epa.gov/hero/index.cfm/reference/download/reference_id/730449 (accessed on 3 March 2020).
- California Office of Environmental Health Hazard Assessment. Public Health Goals for Chemicals in Drinking Water: Bromate; 2009. Available online: https://oehha.ca.gov/water/public-health-goal/final-public-health-goal-bromate-drinking-water (accessed on 3 March 2020).
- Rosinger, A.Y.; Herrick, K.A.; Wutich, A.Y.; Yoder, J.S.; Ogden, C.L. Disparities in plain, tap and bottled water consumption among US adults: National Health and Nutrition Examination Survey (NHANES) 2007–2014. Public Health Nutr. 2018, 21, 1455–1464. [Google Scholar] [CrossRef]
- California Office of Environmental Health Hazard Assessment. Draft Public Health Goal for Trihalomethanes in Drinking Water. 2010. Available online: https://oehha.ca.gov/media/downloads/water/document/thmphg090910.pdf (accessed on 3 March 2020).
- Minnesota Department of Health. Risk Assessment Advice for Incorporating Early-Life Sensitivity into Cancer Risk Assessments for Linear Carcinogens. 2010. Available online: https://www.health.state.mn.us/communities/environment/risk/docs/guidance/adafrecmd.pdf (accessed on 3 March 2020).
- United States Environmental Protection Agency. Regulatory Impact Analysis of the Final Revision to the National Ambient Air Quality Standards for Ground-Level Ozone; EPA-452/R-15-007; 2015. Available online: https://www3.epa.gov/ttnecas1/docs/20151001ria.pdf (accessed on 3 March 2020).
- United States Environmental Protection Agency. Technical Support Document: EPA’s 2014 National Air Toxics Assessment; United States Environmental Protection Agency: Research Triangle Park, NC, USA, 2018. Available online: https://www.epa.gov/sites/production/files/2018-09/documents/2014_nata_technical_support_document.pdf (accessed on 3 March 2020).
- California Office of Environmental Health Hazard Assessment. Appendix J: In Utero and Early Life Susceptibility to Carcinogens: The Derivation of Age-at-Exposure Sensitivity Measures; 2009. Available online: https://oehha.ca.gov/air/crnr/technical-support-document-cancer-potency-factors-2009 (accessed on 3 March 2020).
- California Office of Environmental Health Hazard Assessment. First Public Review Draft, Trihalomethanes in Drinking Water; 2018. Available online: https://oehha.ca.gov/media/downloads/crnr/thmsdraft100518.pdf (accessed on 3 March 2020).
- Higgins, J.P.T.; Green, S.; Cochrane Collaboration. Cochrane Handbook for Systematic Reviews of Interventions; Wiley-Blackwell: Chichester, England; Hoboken, NJ, USA, 2008; 649p. [Google Scholar]
- National Cancer Institute. DevCan—Probability of Developing or Dying of Cancer; DevCan 6.7.7 (Released April 2019); National Cancer Institute: Bethesda, MD, USA, 2019.
- National Cancer Institute. Cancer Stat Facts: Bladder Cancer. Available online: https://seer.cancer.gov/statfacts/html/urinb.html (accessed on 3 March 2020).
- Mariotto, A.B.; Yabroff, K.R.; Shao, Y.; Feuer, E.J.; Brown, M.L. Projections of the cost of cancer care in the United States: 2010–2020. J. Natl. Cancer Inst. 2011, 103, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Bureau of Economic Analysis. Health Care Satellite Account. Available online: https://www.bea.gov/data/special-topics/health-care (accessed on 3 March 2020).
- United States Environmental Protection Agency. Guidelines for Carcinogen Risk Assessment; 630-P-03-001F; 2005. Available online: https://www.epa.gov/risk/guidelines-carcinogen-risk-assessment (accessed on 3 March 2020).
- Barton, H.A.; Cogliano, V.J.; Flowers, L.; Valcovic, L.; Setzer, R.W.; Woodruff, T.J. Assessing susceptibility from early-life exposure to carcinogens. Environ. Health Perspect. 2005, 113, 1125–1133. [Google Scholar] [CrossRef]
- EFSA Scientific Committee; Hardy, A.; Benford, D.; Halldorsson, T.; Jeger, M.J.; Knutsen, K.H.; More, S.; Mortensen, A.; Naegeli, H.; Noteborn, H.; et al. Update: Use of the benchmark dose approach in risk assessment. EFSA J. 2017, 15, e04658. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. Bromodichloromethane; CASRN 75-27-4; 1993. Available online: https://cfpub.epa.gov/ncea/iris/iris_documents/documents/subst/0213_summary.pdf (accessed on 3 March 2020).
- United States Environmental Protection Agency. Trichloroacetic Acid (TCA); CASRN 76-03-9; 2011. Available online: https://cfpub.epa.gov/ncea/iris/iris_documents/documents/subst/0655_summary.pdf (accessed on 3 March 2020).
- United States Environmental Protection Agency. Dibromochloromethane; CASRN 124-48-1; 1990. Available online: http://cfpub.epa.gov/ncea/iris/iris_documents/documents/subst/0222_summary.pdf (accessed on 3 March 2020).
- United States Environmental Protection Agency. Dichloroacetic Acid; CASRN 79-43-6; 2003. Available online: https://cfpub.epa.gov/ncea/iris/iris_documents/documents/subst/0654_summary.pdf (accessed on 3 March 2020).
- United States Environmental Protection Agency. Bromoform; CASNR 75-25-2; 1990. Available online: http://cfpub.epa.gov/ncea/iris/iris_documents/documents/subst/0214_summary.pdf (accessed on 3 March 2020).
- National Cancer Institute. Report on Carcinogenesis Bioassay of Chloroform; Report on Carcinogenesis Bioassay of Chloroform; National Institute of Health: Bethesda, MD, USA, 1976.
- National Toxicology Program. Report on Carcinogens: Monograph on Haloacetic Acids Found as Water Disinfection by-Products; 2018. Available online: https://ntp.niehs.nih.gov/ntp/roc/monographs/haafinal_508.pdf (accessed on 3 March 2020).
- Atwood, S.T.; Lunn, R.M.; Garner, S.C.; Jahnke, G.D. New Perspectives for Cancer Hazard Evaluation by the Report on Carcinogens: A Case Study Using Read-Across Methods in the Evaluation of Haloacetic Acids Found as Water Disinfection By-Products. Environ. Health Perspect. 2019, 127, 125003. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer. IARC Monographs on the Evaluation fo Carcinogenic Risks to Humans; 1999; Volume 73: Some Chemicals that Cause Tumours of the Kidney or Urinary Bladder in Rodents and Some Other Substances. [Google Scholar]
- United States Environmental Protection Agency. Drinking Water Criteria Document for Brominated Trihalomethanes; EPA-822-R-05-011; 2005. Available online: https://www.epa.gov/dwstandardsregulations/drinking-water-criteria-document-brominated-trihalomethanes (accessed on 3 March 2020).
- Melnick, R.L.; Boorman, G.A.; Dellarco, V. Water chlorination, 3-chloro-4-(dichloromethyl)-5-hydroxy-2(5H)-furanone (MX), and potential cancer risk. J. Natl. Cancer Inst. 1997, 89, 832–833. [Google Scholar] [CrossRef][Green Version]
- International Agency for Research on Cancer. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; International Agency for Research on Cancer: Lyon, France, 1991; Volume 52: Chlorinated Drinking Water; Chlorination Byproducts; Some Other Halogenated Compounds; Cobalt and Cobalt Compounds. [Google Scholar]
- United States Environmental Protection Agency. Drinking Water Criteria Document for Brominated Trihalomethanes; 2005. Available online: https://www.epa.gov/sites/production/files/2019-03/documents/dw-brominated-trihalomethanes.pdf (accessed on 3 March 2020).
- National Toxicology Program. Report on Carcinogens, Fourteenth Edition: Bromodichloromethane CAS No. 75-27-4; 2016. Available online: https://ntp.niehs.nih.gov/ntp/roc/content/profiles/bromodichloromethane.pdf (accessed on 3 March 2020).
- United States Environmental Protection Agency. Chloroform; CASRN 67-66-3; 2001. Available online: https://cfpub.epa.gov/ncea/iris2/chemicalLanding.cfm?substance_nmbr=25 (accessed on 3 March 2020).
- National Toxicology Program. Report on Carcinogens, Fourteenth Edition: Chloroform CAS No. 67-66-3; 2016. Available online: https://ntp.niehs.nih.gov/ntp/roc/content/profiles/chloroform.pdf (accessed on 3 March 2020).
- International Agency for Research on Cancer. IARC Monographs on the Evaluation fo Carcinogenic Risks to Humans; International Agency for Research on Cancer: Lyon, France, 2013; Volume 101: Some Chemicals Present in Industrial and Consumer Products, Food, and Drinking Water. [Google Scholar]
- International Agency for Research on Cancer. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; International Agency for Research on Cancer: Lyon, France, 2014; Volume 106: Trichloroethylene, Tetrachloroethylene, and Some Other Chlorinated Agents. [Google Scholar]
- Cantor, K.P. Water chlorination, mutagenicity, and cancer epidemiology. Am. J. Public Health 1994, 84, 1211–1213. [Google Scholar] [CrossRef]
- Bull, R.J.; Birnbaum, L.S.; Cantor, K.P.; Rose, J.B.; Butterworth, B.E.; Pegram, R.; Tuomisto, J. Water chlorination: Essential process or cancer hazard? Fundam Appl. Toxicol. 1995, 28, 155–166. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. Economic Analysis for the Final Stage 2 Disinfectants and Disinfection Byproducts Rule; EPA 815-R-05-101; 2005. Available online: https://nepis.epa.gov/Exe/ZyPDF.cgi?Dockey=P1005OOX.txt (accessed on 3 March 2020).
- Burger, M.; Catto, J.W.; Dalbagni, G.; Grossman, H.B.; Herr, H.; Karakiewicz, P.; Kassouf, W.; Kiemeney, L.A.; La Vecchia, C.; Shariat, S.; et al. Epidemiology and risk factors of urothelial bladder cancer. Eur. Urol. 2013, 63, 234–241. [Google Scholar] [CrossRef]
- Cantor, K.P.; Villanueva, C.M.; Silverman, D.T.; Figueroa, J.D.; Real, F.X.; Garcia-Closas, M.; Malats, N.; Chanock, S.; Yeager, M.; Tardon, A.; et al. Polymorphisms in GSTT1, GSTZ1, and CYP2E1, disinfection by-products, and risk of bladder cancer in Spain. Environ. Health Perspect. 2010, 118, 1545–1550. [Google Scholar] [CrossRef]
- Yin, X.; Xiong, W.; Wang, Y.; Tang, W.; Xi, W.; Qian, S.; Guo, Y. Association of CYP2E1 gene polymorphisms with bladder cancer risk: A systematic review and meta-analysis. Medicine 2018, 97, e11910. [Google Scholar] [CrossRef]
| Disinfection Byproduct | Number of Community Water Systems Included in the Analysis | Population Weighted-Average Concentration (µg/L) | HAA Group | |
|---|---|---|---|---|
| Monochloroacetic acid | 29,672 | 0.48 | HAA5 | |
| Dichloroacetic acid | 29,673 | 7.8 | HAA5 | |
| Trichloroacetic acid | 29,669 | 6.4 | HAA5 | |
| Monobromoacetic acid | 29,669 | 0.24 | HAA5 | HAA6Br |
| Dibromoacetic acid | 29,669 | 1.2 | HAA5 | HAA6Br |
| Tribromoacetic acid | 986 | 0.21 | HAA6Br | |
| Bromochloroacetic acid | 8024 | 2.9 | HAA6Br | |
| Bromodichloroacetic acid | 992 | 1.1 | HAA6Br | |
| Chlorodibromoacetic acid | 986 | 0.28 | HAA6Br | |
| Disinfection Byproduct | Number of Community Water Systems Included in the Analysis a | Population Weighted-Average Concentration (µg/L) |
|---|---|---|
| HAA5 | 3576 | 19.1 |
| HAA6Br | 3579 | 7.0 |
| HAA9 | 3576 | 29.4 |
| Disinfection Byproduct and Source of Animal Toxicology Data | Benchmark Dose Calculated from Animal Bioassay (mg/kg-day) a | Human-Equivalent Cancer Slope Factor (lower 95% Confidence Limit) b (mg/kg-day)−1 | Benchmark Dose Calculated from Animal Bioassay (mg/kg-day) a | Human-Equivalent Cancer Slope Factor (lower 95% Confidence Limit) b (mg/kg-day)−1 |
|---|---|---|---|---|
| Benchmark response | 5% excess risk | 10% excess risk | ||
| Bromochloroacetic acid [26] | 1.0 (0.7–1.6) | 0.463 | 2.0 (1.3–3.2) | 0.451 |
| Bromodichloroacetic acid [27] | 2.8 (1.6–9.1) | 0.185 | 5.7 (3.2–18.7) | 0.180 |
| Dibromoacetic acid [25] | 2.2 (1.4–3.8) | 0.210 | 4.5 (3.0–7.7) | 0.205 |
| Disinfection Byproduct | Cancer Slope Factor Reported by OEHHA a (mg/kg/day−1) | OEHHA One-in-a-Million Cancer Risk Benchmark Concentration (µg/L) | Cancer Slope Factor Reported by the U.S. EPA a (mg/kg-day−1) | U.S. EPA One-in-a-Million Cancer Risk Benchmark Concentration (µg/L) |
|---|---|---|---|---|
| Dibromoacetic acid | 0.250 | 0.03, 2020 [30] | N/A | N/A |
| Bromodichloromethane | 0.087 | 0.06, 2018 [42] | 0.062 | 0.6, 1993 [51] |
| Trichloroacetic acid | 0.071 | 0.1, 2020 [30] | 0.067 | 0.5, 2011 [52] |
| Dibromochloromethane | 0.045 | 0.1, 2018 [42] | 0.084 | 0.4, 1990 [53] |
| Dichloroacetic acid | 0.041 | 0.2, 2020 [30] | 0.048 | 0.7, 2003 [54] |
| Chloroform | 0.014 | 0.4, 2018 [42] | N/A | N/A |
| Bromoform | 0.011 | 0.5, 2018 [42] | 0.008 | 4.0, 1990 [55] |
| Disinfection Byproduct a | Benchmark Doses from Animal Bioassay, 5% Excess Cancer Risk (mg/kg-day) | Concentrations Corresponding to One-in-a-Million Cancer Risk c (µg/L) | Source of the Risk Benchmark |
|---|---|---|---|
| Bromochloroacetic acid | 1.0 (0.7–1.6) b | 0.02 | Calculated in this study |
| Chlorodibromoacetic acid | Not Available | 0.02 | Applied by read-across from bromochloroacetic acid |
| Bromodichloroacetic acid | 2.8 (1.6–9.1) b | 0.04 | Calculated in this study |
| Dibromoacetic acid | 2.2 (1.4–3.8) b | 0.04 | Calculated in this study |
| Tribromoacetic acid | Not Available | 0.04 | Applied by read-across from dibromoacetic acid |
| Trichloroacetic acid | 8.1 (4.4–28.8) | 0.1 | OEHHA 2020 [30] |
| Dichloroacetic acid | 32.7 (7.9–40.2) | 0.2 | OEHHA 2020 [30] |
| HAA5 group | Not applicable | 0.1 | Calculated in this study |
| HAA6Br group | Not applicable | 0.03 | Calculated in this study |
| HAA9 group | Not applicable | 0.06 | Calculated in this study |
| Bromodichloromethane | 11.1 (3.9–21.9) | 0.06 | OEHHA 2018 [42] |
| Dibromochloromethane | 26.2 (7.5–39.1) | 0.1 | OEHHA 2018 [42] |
| Chloroform | 33.4 (14.1–51.6) d | 0.4 e | OEHHA 2018 [42] |
| Bromoform | 31.0 (18.7–82.7) | 0.5 | [42] |
| THM4 group | Not applicable | 0.15 | Evans et al. [16] |
| Chemical Group | Cancer Estimates Using Default Parameters a,b | Cancer Estimates Using Age Sensitivity Factors a,c |
|---|---|---|
| Lifetime cancer cases calculated using the national tap water dataset (thousands) | ||
| THM4 d | 3.1 (1.7–10.2) | 19.1 (10.2–57.2) |
| HAA5 | 4.0 (2.0–8.4) | 18.5 (8.6–40.1) |
| Lifetime cancer cases calculated for systems in the UCMR4 program (thousands) | ||
| HAA5 | 2.9 (1.5–6.1) | 13.4 (6.2–29.1) |
| HAA6Br | 8.6 (4.4–12.8) | 32.1 (21.4–42.8) |
| HAA9 | 10.9 (5.4–17.5) | 41.4 (25.3–76.0) |
| THM4 d and HAA9 | 12.8 (6.5–24.1) | 53.7 (31.9–112.8) |
| Lifetime cancer risk calculated for systems in the UCMR4 program | ||
| Cumulative cancer risk estimates for THM4 d and HAA9 | 7.0 × 10−5 (3.5 × 10−5–1.3 × 10−4) | 2.9 × 10−4 (1.7 × 10−4–6.2 × 10−4) |
| Water Source | Population Served | Up to 20 ug/L | 20–40 ug/L | 40–60 ug/L | 60–80 ug/L | Above 80 ug/L | All Concentrations | ||
|---|---|---|---|---|---|---|---|---|---|
| Groundwater | 10,000 or less | 13.2 (0–29.4) | 13.7 (0–27.4) | 6.7 (1–11.9) | 3.2 (1.2–4.9) | 2.3 (0.3–5.1) | 39.2 (2.5–78.6) | 141.5 (10.2–276.5) | 828.1 (57.2–1590.2) |
| 10,001 to 100,000 | 17.1 (0–37.8) | 28.4 (0–56.8) | 16.9 (2.7–29.6) | 3.6 (1.3–5.6) | 0.5 (0.1–1) | 66.4 (4–130.8) | |||
| More than 100,000 | 4.4 (0–9.7) | 14.6 (0–28.5) | 11.6 (1.8–20.4) | 5.3 (1.8–8.4) | 0 (0–0) | 35.9 (3.6–67) | |||
| Surface Water | 10,000 or less | 5.2 (0–11.4) | 22.8 (0–44.9) | 26.5 (4.2–46.5) | 10.6 (3.9–16.4) | 5.1 (1–10.6) | 70.1 (9.1–129.8) | 686.6 (47.1–1313.6) | |
| 10,001 to 100,000 | 27.5 (0–60.9) | 100.1 (0–197.8) | 74.4 (10.9–131.3) | 9.7 (3.5–15.2) | 4.9 (0.6–11.2) | 216.7 (15–416.4) | |||
| More than 100,000 | 35.4 (0–78.4) | 217.6 (0–427.9) | 131.1 (19.2–231.4) | 10.8 (3.8–17.1) | 4.9 (0–12.6) | 399.9 (23–767.4) | |||
| Disinfection Byproduct | International Agency for Research on Cancer (IARC) | U.S. Environmental Protection Agency | National Toxicology Program (U.S.) |
|---|---|---|---|
| Trihalomethanes | |||
| Bromodichloromethane | Possibly carcinogenic to humans (Group 2B) [62]; | Probable human carcinogen (Group B2) [51]; Likely to be carcinogenic to humans [63]; | Reasonably anticipated to be a human carcinogen [64]; |
| Bromoform | Unclassifiable as to carcinogenicity in humans (Group 3) [62]; | Probable human carcinogen (Group B2) [55]; Likely to be carcinogenic to humans [63]; | |
| Chloroform | Possibly carcinogenic to humans (Group 2B) [59]; | Probable human carcinogen (Group B2) [65]; Likely to be carcinogenic to humans by all routes of exposure under exposure conditions that lead to cytotoxicity and regenerative hyperplasia in susceptible tissues; not likely to be carcinogenic to humans by any route of exposure under exposure conditions that do not cause cytotoxicity and cell regeneration [65]; | Reasonably anticipated to be a human carcinogen [66]; |
| Dibromochloromethane | Not classifiable as to its carcinogenicity to humans (Group 3) [67]; | Possible human carcinogen (Group C) [53]; | |
| Haloacetic acids | |||
| Dichloroacetic Acid | Possibly carcinogenic to humans (Group 2B) [68]; | Likely to be carcinogenic to humans [54]; | Reasonably anticipated to be a human carcinogen [57] |
| Dibromoacetic acid | Possibly carcinogenic to humans (Group 2B) [67]; | Reasonably anticipated to be a human carcinogen [57] | |
| Bromochloroacetic acid | Possibly carcinogenic to humans (Group 2B) [67]; | Reasonably anticipated to be a human carcinogen [57] | |
| Bromodichloroacetic acid | Reasonably anticipated to be a human carcinogen [57] | ||
| Trichloroacetic acid | Possibly carcinogenic to humans (Group 2B) [68]; | Suggestive evidence of carcinogenic potential [52]; | |
| Chlorodibromoacetic acid | Reasonably anticipated to be a human carcinogen [57] | ||
| Tribromoacetic acid | Reasonably anticipated to be a human carcinogen [57] | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Evans, S.; Campbell, C.; Naidenko, O.V. Analysis of Cumulative Cancer Risk Associated with Disinfection Byproducts in United States Drinking Water. Int. J. Environ. Res. Public Health 2020, 17, 2149. https://doi.org/10.3390/ijerph17062149
Evans S, Campbell C, Naidenko OV. Analysis of Cumulative Cancer Risk Associated with Disinfection Byproducts in United States Drinking Water. International Journal of Environmental Research and Public Health. 2020; 17(6):2149. https://doi.org/10.3390/ijerph17062149
Chicago/Turabian StyleEvans, Sydney, Chris Campbell, and Olga V. Naidenko. 2020. "Analysis of Cumulative Cancer Risk Associated with Disinfection Byproducts in United States Drinking Water" International Journal of Environmental Research and Public Health 17, no. 6: 2149. https://doi.org/10.3390/ijerph17062149
APA StyleEvans, S., Campbell, C., & Naidenko, O. V. (2020). Analysis of Cumulative Cancer Risk Associated with Disinfection Byproducts in United States Drinking Water. International Journal of Environmental Research and Public Health, 17(6), 2149. https://doi.org/10.3390/ijerph17062149




