Daily Resting Heart Rate Variability in Adolescent Swimmers during 11 Weeks of Training
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Training Load Monitoring
2.4. HRV Measurement
2.5. Sleep Monitoring
2.6. Statistical Analyses
3. Results
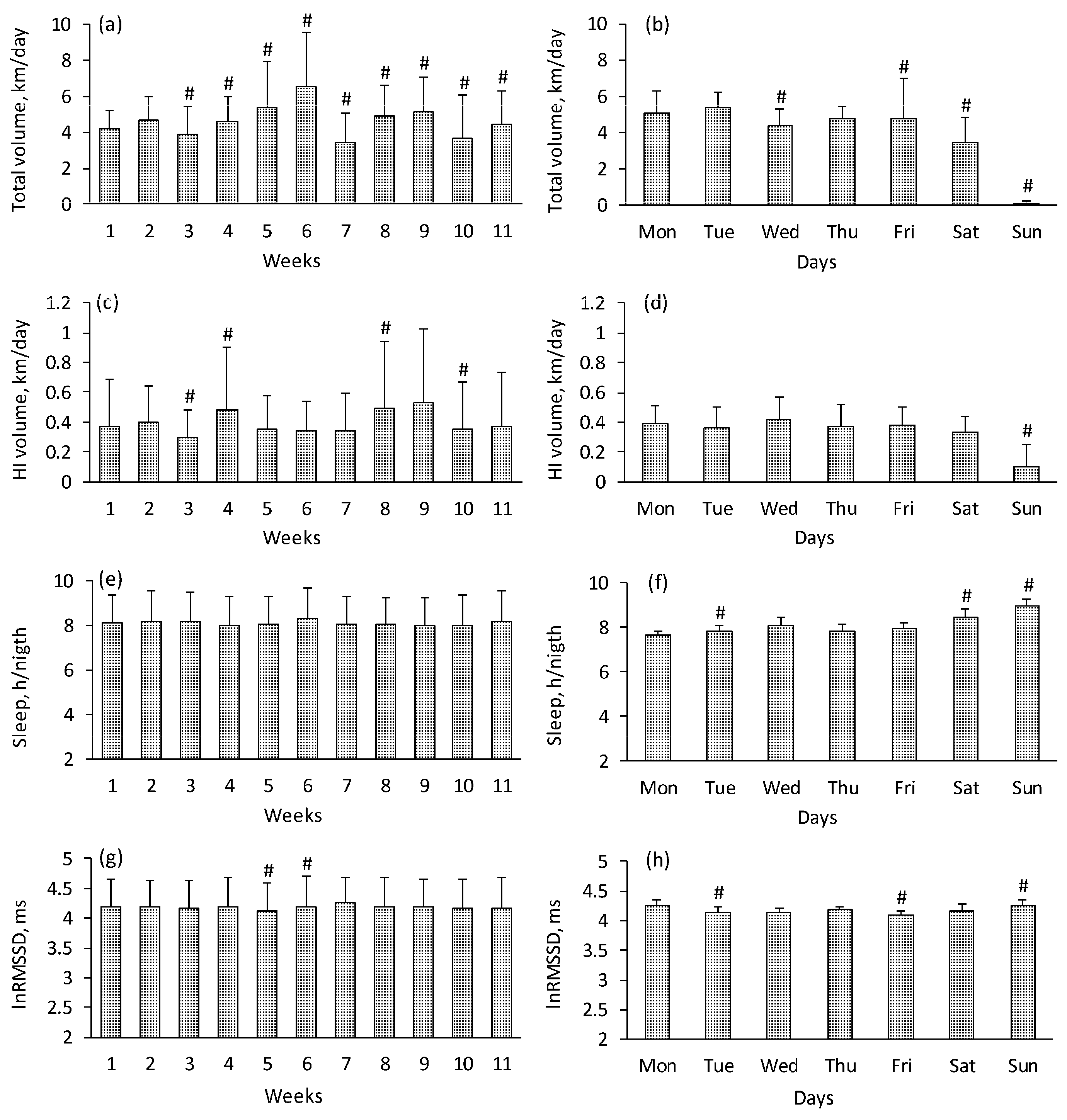
3.1. Training Volume
3.2. Sleep
3.3. Resting HRV (lnRMSSD)
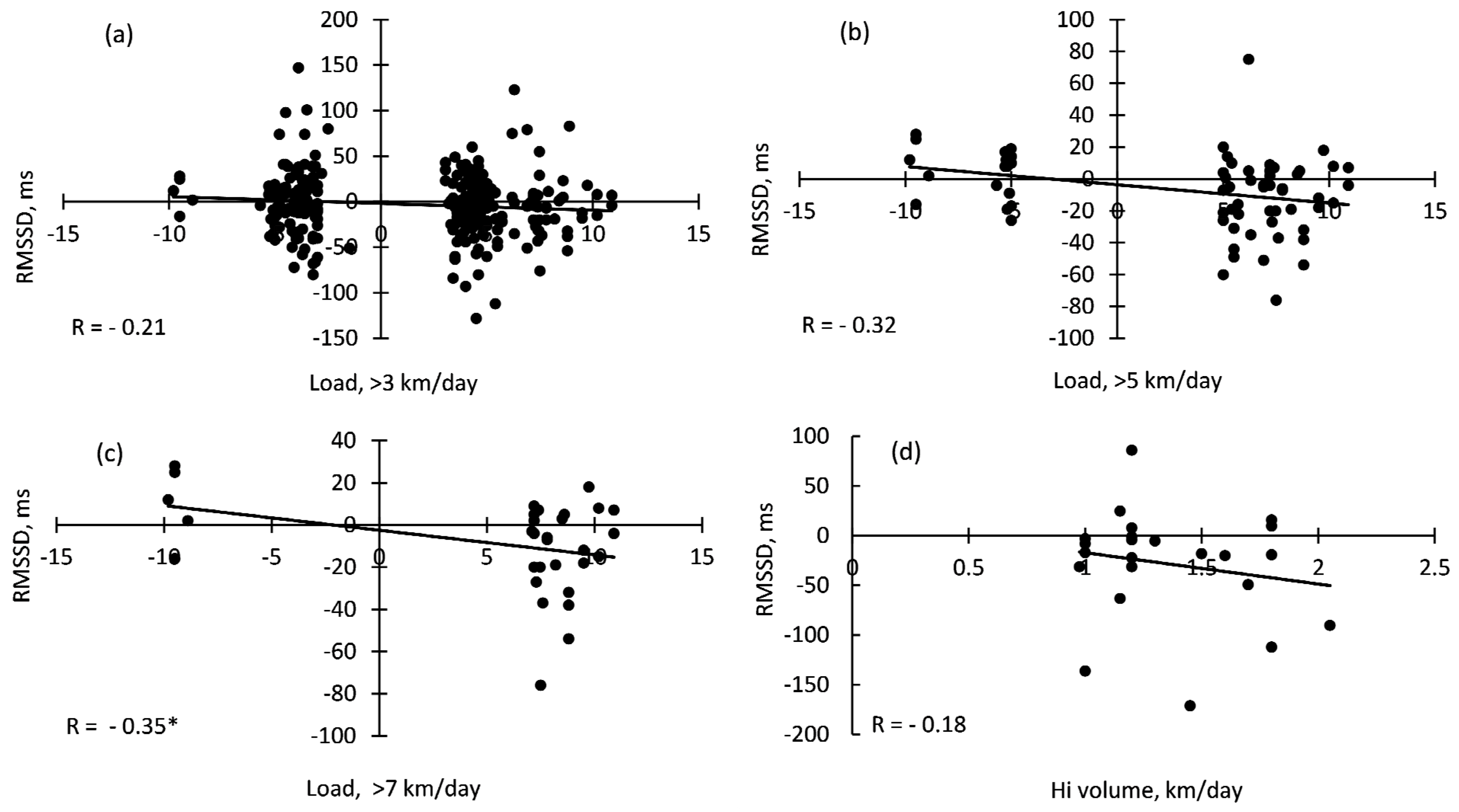
3.4. Correlations between Daily HRV, Training Volume, and Sleep Quantity
4. Discussion
4.1. Limitations
4.2. Practical Application and Recommendations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Halson, S.L. Monitoring training load to understand fatigue in athletes. Sports Med. 2014, 44, 139–147. [Google Scholar] [CrossRef]
- Buchheit, M. Monitoring training status with HR measures: Do all roads lead to Rome? Front. Physiol. 2014, 5, 73. [Google Scholar] [CrossRef]
- Bellenger, C.R.; Fuller, J.T.; Thomson, R.L.; Davison, K.; Robertson, E.Y.; Buckley, J.D. Monitoring athletic training status through autonomic heart rate regulation: A systematic review and meta-analysis. Sports Med. 2016, 46, 1461–1486. [Google Scholar] [CrossRef]
- Jiménez Morgan, S.; Molina Mora, J.A. Effect of heart rate variability biofeedback on sport performance, a systematic review. Appl. Psychophysiol. Biofeedback 2017, 42, 235–245. [Google Scholar] [CrossRef]
- Bhati, P.; Moiz, J.A.; Menon, G.R.; Hussain, M.E. Does resistance training modulate cardiac autonomic control? A systematic review and meta-analysis. Clin. Auton. Res. Off. J. Clin. Auton. Res. Soc. 2019, 29, 75–103. [Google Scholar] [CrossRef]
- Sekiguchi, Y.; Adams, W.M.; Benjamin, C.L.; Curtis, R.M.; Giersch, G.E.W.; Casa, D.J. Relationships between resting heart rate, heart rate variability and sleep characteristics among female collegiate cross-country athletes. J. Sleep Res. 2019, 28, e12836. [Google Scholar] [CrossRef]
- Dong, J.-G. The role of heart rate variability in sports physiology. Exp. Ther. Med. 2016, 11, 1531–1536. [Google Scholar] [CrossRef]
- Shaffer, F.; Ginsberg, J.P. An Overview of Heart Rate Variability Metrics and Norms. Front. Public Health 2017, 5, 258. [Google Scholar] [CrossRef]
- Gevirtz, R.N.; Lehrer, P.M.; Schwartz, M.S. Cardiorespiratory biofeedback. In Biofeedback: A Practitioner’s Guide, Schwartz, M.S., Andrasik, F., Eds.; 4th ed.; The Guilford Press: New York, NY, USA, 2016; pp. 196–213. [Google Scholar]
- Borresen, J.; Lambert, M.I. Autonomic control of heart rate during and after exercise: Measurements and implications for monitoring training status. Sports Med. Auckl. N.Z. 2008, 38, 633–646. [Google Scholar] [CrossRef]
- Bell, D.R.; Post, E.G.; Trigsted, S.M.; Hetzel, S.; McGuine, T.A.; Brooks, M.A. Prevalence of sport specialization in high school athletics: A 1-year observational study. Am. J. Sports Med. 2016, 44, 1469–1474. [Google Scholar] [CrossRef]
- Koenig, J.; Jarczok, M.N.; Wasner, M.; Hillecke, T.K.; Thayer, J.F. Heart rate variability and swimming. Sports Med. 2014, 44, 1377–1391. [Google Scholar] [CrossRef] [PubMed]
- Chalencon, S.; Pichot, V.; Roche, F.; Lacour, J.-R.; Garet, M.; Connes, P.; Barthélémy, J.C.; Busso, T. Modeling of performance and ANS activity for predicting future responses to training. Eur. J. Appl. Physiol. 2015, 115, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Clemente-Suárez, V.J.; Arroyo-Toledo, J.J. Use of biotechnology devices to analyse fatigue process in swimming training. J. Med. Syst. 2017, 41, 94. [Google Scholar] [CrossRef] [PubMed]
- Lima-Borges, D.S.; Martinez, P.F.; Vanderlei, L.C.M.; Barbosa, F.S.S.; Oliveira-Junior, S.A. Autonomic modulations of heart rate variability are associated with sports injury incidence in sprint swimmers. Phys. Sports Med. 2018, 46, 374–384. [Google Scholar] [CrossRef]
- Pla, R.; Aubry, A.; Resseguier, N.; Merino, M.; Toussaint, J.-F.; Hellard, P. Training organization, physiological profile and heart rate variability changes in an open-water world champion. Int. J. Sports Med. 2019, 40, 519–527. [Google Scholar] [CrossRef]
- Schneider, C.; Wiewelhove, T.; Raeder, C.; Flatt, A.A.; Hoos, O.; Hottenrott, L.; Schumbera, O.; Kellmann, M.; Meyer, T.; Pfeiffer, M.; et al. Heart rate variability monitoring during strength and high-intensity interval training overload microcycles. Front. Physiol. 2019, 10, 582. [Google Scholar] [CrossRef]
- Adirim, T.A.; Cheng, T.L. Overview of injuries in the young athlete. Sports Med. Auckl. N.Z. 2003, 33, 75–81. [Google Scholar] [CrossRef]
- Frisch, A.; Croisier, J.-L.; Urhausen, A.; Seil, R.; Theisen, D. Injuries, risk factors and prevention initiatives in youth sport. Br. Med. Bull. 2009, 92, 95–121. [Google Scholar] [CrossRef][Green Version]
- Marshall, W.A.; Tanner, J.M. Variations in the pattern of pubertal changes in boys. Arch. Dis. Child. 1970, 45, 13–23. [Google Scholar] [CrossRef]
- Williams, S.; West, S.; Howells, D.; Kemp, S.P.T.; Flatt, A.A.; Stokes, K. Modelling the HRV response to training loads in elite Rugby Sevens players. J. Sports Sci. Med. 2018, 17, 402–408. [Google Scholar]
- Nakamura, F.Y.; Flatt, A.A.; Pereira, L.A.; Ramirez-Campillo, R.; Loturco, I.; Esco, M.R. Ultra-short-term heart rate variability is sensitive to training effects in team sports players. J. Sports Sci. Med. 2015, 14, 602–605. [Google Scholar]
- Ameen, M.S.; Cheung, L.M.; Hauser, T.; Hahn, M.A.; Schabus, M. About the accuracy and problems of consumer devices in the assessment of sleep. Sensors (Basel) 2019, 19, 4160. [Google Scholar] [CrossRef]
- Kubala, A.G.; Barone, G.B.; Buysse, D.J.; Patel, S.R.; Hall, M.H.; Kline, C.E. Field-based Measurement of Sleep: Agreement between Six Commercial Activity Monitors and a Validated Accelerometer. Behav. Sleep Med. 2019, 27, 1–16. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; L. Erlbaum Associates: Hillsdale, MI, USA, 1988; pp. 77–81. [Google Scholar]
- Lehrer, P.M.; Vaschillo, E.; Vaschillo, B.; Lu, S.-E.; Eckberg, D.L.; Edelberg, R.; Shih, W.J.; Lin, Y.; Kuusela, T.A.; Tahvanainen, K.U.O.; et al. Heart rate variability biofeedback increases baroreflex gain and peak expiratory flow. Psychosom. Med. 2003, 65, 796–805. [Google Scholar] [CrossRef]
- Schmitt, L.; Regnard, J.; Millet, G.P. Monitoring fatigue status with HRV measures in elite athletes: An Avenue Beyond RMSSD? Front. Physiol. 2015, 6, 343. [Google Scholar] [CrossRef]
- Pichot, V.; Roche, F.; Gaspoz, J.M.; Enjolras, F.; Antoniadis, A.; Minini, P.; Costes, F.; Busso, T.; Lacour, J.R.; Barthélémy, J.C. Relation between heart rate variability and training load in middle-distance runners. Med. Sci. Sports Exerc. 2000, 32, 1729–1736. [Google Scholar] [CrossRef]
- Iellamo, F.; Legramante, J.M.; Pigozzi, F.; Spataro, A.; Norbiato, G.; Lucini, D.; Pagani, M. Conversion from vagal to sympathetic predominance with strenuous training in high-performance world class athletes. Circulation 2002, 105, 2719–2724. [Google Scholar] [CrossRef]
- Gratze, G.; Rudnicki, R.; Urban, W.; Mayer, H.; Schlögl, A.; Skrabal, F. Hemodynamic and autonomic changes induced by Ironman: Prediction of competition time by blood pressure variability. J. Appl. Physiol. (1985) 2005, 99, 1728–1735. [Google Scholar] [CrossRef]
- Clemente-Suárez, V.J.; Fernandes, R.J.; de Jesus, K.; Pelarigo, J.G.; Arroyo-Toledo, J.J.; Vilas-Boas, J.P. Do traditional and reverse swimming training periodizations lead to similar aerobic performance improvements? J. Sports Med. Phys. Fitness 2018, 58, 761–767. [Google Scholar]
- Stanley, J.; D’Auria, S.; Buchheit, M. Cardiac parasympathetic activity and race performance: An Elite Triathlete Case Study. Int. J. Sports Physiol. Perform. 2015, 10, 528–534. [Google Scholar] [CrossRef]
- Le Meur, Y.; Pichon, A.; Schaal, K.; Schmitt, L.; Louis, J.; Gueneron, J.; Vidal, P.P.; Hausswirth, C. Evidence of parasympathetic hyperactivity in functionally overreached athletes. Med. Sci. Sports Exerc. 2013, 45, 2061–2071. [Google Scholar] [CrossRef]
- Manzi, V.; Castagna, C.; Padua, E.; Lombardo, M.; D’Ottavio, S.; Massaro, M.; Volterrani, M.; Iellamo, F. Dose-response relationship of autonomic nervous system responses to individualized training impulse in marathon runners. Am. J. Physiol. Heart Circ. Physiol. 2009, 296, H1733–H1740. [Google Scholar] [CrossRef]
- Iellamo, F.; Lucini, D.; Volterrani, M.; Casasco, M.; Salvati, A.; Gianfelici, A.; Di Gianfrancesco, A.; Urso, A.; Manzi, V. Autonomic nervous system responses to strength training in top-level weight lifters. Physiol. Rep. 2019, 7, e14233. [Google Scholar] [CrossRef]
- Fullagar, H.H.K.; Skorski, S.; Duffield, R.; Hammes, D.; Coutts, A.J.; Meyer, T. Sleep and athletic performance: The effects of sleep loss on exercise performance, and physiological and cognitive responses to exercise. Sports Med. Auckl. N.Z. 2015, 45, 161–186. [Google Scholar] [CrossRef]
- Knufinke, M.; Nieuwenhuys, A.; Geurts, S.A.E.; Coenen, A.M.L.; Kompier, M.A.J. Self-reported sleep quantity, quality and sleep hygiene in elite athletes. J. Sleep Res. 2018, 27, 78–85. [Google Scholar] [CrossRef]
- Tobaldini, E.; Nobili, L.; Strada, S.; Casali, K.R.; Braghiroli, A.; Montano, N. Heart rate variability in normal and pathological sleep. Front. Physiol. 2013, 4, 294. [Google Scholar] [CrossRef]
- Lehrer, P.M.; Gevirtz, R. Heart rate variability biofeedback: How and why does it work? Front. Psychol. 2014, 5, 756. [Google Scholar] [CrossRef]
- McCraty, R.; Atkinson, M.; Tiller, W.A.; Rein, G.; Watkins, A.D. The effects of emotions on short-term power spectrum analysis of heart rate variability. Am. J. Cardiol. 1995, 76, 1089–1093. [Google Scholar] [CrossRef]
- McCole, S.D.; Brown, M.D.; Moore, G.E.; Zmuda, J.M.; Cwynar, J.D.; Hagberg, J.M. Enhanced cardiovascular hemodynamics in endurance-trained postmenopausal women athletes. Med. Sci. Sports Exerc. 2000, 32, 1073–1079. [Google Scholar] [CrossRef][Green Version]
- Stanley, J.; Peake, J.M.; Buchheit, M. Cardiac parasympathetic reactivation following exercise: Implications for training prescription. Sports Med. 2013, 43, 1259–1277. [Google Scholar] [CrossRef]
- Boudreau, P.; Yeh, W.H.; Dumont, G.A.; Boivin, D.B. A circadian rhythm in heart rate variability contributes to the increased cardiac sympathovagal response to awakening in the morning. Chronobiol. Int. 2012, 29, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Riganello, F.; Prada, V.; Soddu, A.; di Perri, C.; Sannita, W.G. Circadian rhythms and measures of CNS/autonomic interaction. Int. J. Environ. Res. Public Health 2019, 16, 2336. [Google Scholar] [CrossRef] [PubMed]
- Edmonds, R.; Leicht, A.; Burkett, B.; McKean, M. Cardiac autonomic and salivary responses to a repeated training bout in elite swimmers. Sports 2016, 4, 13. [Google Scholar] [CrossRef]
- Rabbani, A.; Clemente, F.M.; Kargarfard, M.; Chamari, K. Match Fatigue Time-Course Assessment Over Four Days: Usefulness of the Hooper Index and Heart Rate Variability in Professional Soccer Players. Front. Physiol. 2019, 10, 109. [Google Scholar] [CrossRef]
- Moreira, A.; Aoki, M.S.; Franchini, E.; da Silva Machado, D.G.; Paludo, A.C.; Okano, A.H. Mental fatigue impairs technical performance and alters neuroendocrine and autonomic responses in elite young basketball players. Physiol. Behav. 2018, 196, 112–118. [Google Scholar] [CrossRef]
- Sansone, P.; Tessitore, A.; Paulauskas, H.; Lukonaitiene, I.; Tschan, H.; Pliauga, V.; Conte, D. Physical and physiological demands and hormonal responses in basketball small-sided games with different tactical tasks and training regimes. J. Sci. Med. Sport 2019, 22, 602–606. [Google Scholar] [CrossRef]
- Venckunas, T.; Krusnauskas, R.; Snieckus, A.; Eimantas, N.; Baranauskiene, N.; Skurvydas, A.; Brazaitis, M.; Kamandulis, S. Acute effects of very low-volume high-intensity interval training on muscular fatigue and serum testosterone level vary according to age and training status. Eur. J. Appl. Physiol. 2019, 119, 1725–1733. [Google Scholar] [CrossRef]
| Variable | Male (n = 7) | Female (n = 15) | Total (n = 22) |
|---|---|---|---|
| Age, years | 15.4 (0.7) | 13.8 (0.6) ^ | 14.3 (1.0) |
| Height, cm | 179.5 (6.0) | 165.1 (6.7) ^ | 169.7 (9.3) |
| Weight, kg | 65.5 (6.7) | 56.5 (6.1) ^ | 59.4 (7.5) |
| Body fat, % | 10.8 (3.9) | 21.5 (3.8) ^ | 18.1 (6.3) |
| Knee extension peak torque, Nm/s | 197.3 (20.7) | 141.4 (27.5) ^ | 159.2 (36.6) |
| Vertical jump height, cm | 40.2 (2.1) | 31.2 (3.3) ^ | 33.8 (5.1) |
| VO2peak, mL/min/kg | 47.8 (5.0) | 38.8 (6.8) ^ | 41.6 (7.5) |
| Maturity | |||
| Tanner II, n (%) | 1 (14.3 %) | 2 (13.3 %) | 3 (13.6 %) |
| Tanner III, n (%) | 5 (71.4 %) | 11 (73.4 %) | 16 (72.8 %) |
| Tanner IV, n (%) | 1 (14.3 %) | 2 (13.3 %) | 3 (13.6 %) |
| LnRMSDD, ms | Correl LnRMSDD | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| No | Gender | Average | Max | Min | SD | CV% | Training Volume | HI Volume | Sleep |
| 1 | M | 4.30 | 4.91 | 3.61 | 0.29 | 6.7 | −0.05 | −0.02 | −0.01 |
| 2 | M | 4.25 | 4.88 | 2.20 | 0.36 | 8.5 | −0.22 | −0.24 | −0.13 |
| 3 | M | 4.53 | 4.99 | 4.04 | 0.21 | 4.6 | −0.07 | −0.03 | −0.13 |
| 4 | M | 3.90 | 5.19 | 3.18 | 0.44 | 11.3 | −0.05 | 0.28 | −0.03 |
| 5 | M | 4.61 | 5.08 | 4.13 | 0.20 | 4.3 | −0.31 # | 0.11 | −0.02 |
| 6 | M | 4.47 | 5.24 | 3.71 | 0.35 | 7.8 | −0.18 | 0.05 | −0.08 |
| 7 | M | 4.18 | 4.92 | 3.78 | 0.25 | 6.0 | −0.09 | 0.11 | −0.22 |
| 8 | F | 4.62 | 5.05 | 3.91 | 0.24 | 5.2 | −0.08 | 0.30 | −0.17 |
| 9 | F | 4.12 | 4.85 | 3.00 | 0.37 | 9.0 | −0.28 # | 0.04 | −0.06 |
| 10 | F | 3.60 | 4.43 | 2.30 | 0.44 | 12.2 | −0.20 | −0.01 | 0.00 |
| 11 | F | 3.38 | 4.29 | 2.40 | 0.41 | 12.1 | −0.22 | −0.12 | 0.06 |
| 12 | F | 3.65 | 3.99 | 3.22 | 0.18 | 4.9 | −0.20 | −0.06 | −0.08 |
| 13 | F | 4.26 | 5.00 | 3.26 | 0.31 | 7.3 | −0.05 | 0.25 | −0.27 |
| 14 | F | 3.80 | 5.06 | 2.48 | 0.59 | 15.5 | −0.17 | 0.23 | 0.41 # |
| 15 | F | 4.84 | 5.32 | 4.20 | 0.23 | 4.8 | −0.03 | −0.21 | −0.04 |
| 16 | F | 4.21 | 4.69 | 3.56 | 0.23 | 5.5 | −0.06 | −0.10 | −0.16 |
| 17 | F | 4.59 | 5.13 | 3.71 | 0.36 | 7.8 | −0.16 | 0.15 | −0.05 |
| 18 | F | 4.03 | 5.00 | 3.26 | 0.38 | 9.4 | −0.27 # | 0.02 | −0.08 |
| 19 | F | 4.19 | 4.68 | 3.09 | 0.29 | 6.9 | −0.29 # | 0.26 | 0.30 # |
| 20 | F | 3.95 | 4.89 | 2.83 | 0.55 | 13.9 | −0.45 # | 0.03 | −0.26 |
| 21 | F | 4.23 | 4.90 | 3.4 | 0.35 | 8.3 | −0.16 | 0.21 | −0.04 |
| 22 | F | 4.01 | 5.32 | 3.18 | 0.37 | 9.2 | −0.17 | −0.03 | 0.21 |
| Mean | 4.17 | 4.90 | 3.29 | 0.34 | 8.1 | −0.17 | 0.06 | −0.04 | |
| SD | 0.37 | 0.33 | 0.59 | 0.11 | 3.1 | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamandulis, S.; Juodsnukis, A.; Stanislovaitiene, J.; Zuoziene, I.J.; Bogdelis, A.; Mickevicius, M.; Eimantas, N.; Snieckus, A.; Olstad, B.H.; Venckunas, T. Daily Resting Heart Rate Variability in Adolescent Swimmers during 11 Weeks of Training. Int. J. Environ. Res. Public Health 2020, 17, 2097. https://doi.org/10.3390/ijerph17062097
Kamandulis S, Juodsnukis A, Stanislovaitiene J, Zuoziene IJ, Bogdelis A, Mickevicius M, Eimantas N, Snieckus A, Olstad BH, Venckunas T. Daily Resting Heart Rate Variability in Adolescent Swimmers during 11 Weeks of Training. International Journal of Environmental Research and Public Health. 2020; 17(6):2097. https://doi.org/10.3390/ijerph17062097
Chicago/Turabian StyleKamandulis, Sigitas, Antanas Juodsnukis, Jurate Stanislovaitiene, Ilona Judita Zuoziene, Andrius Bogdelis, Mantas Mickevicius, Nerijus Eimantas, Audrius Snieckus, Bjørn Harald Olstad, and Tomas Venckunas. 2020. "Daily Resting Heart Rate Variability in Adolescent Swimmers during 11 Weeks of Training" International Journal of Environmental Research and Public Health 17, no. 6: 2097. https://doi.org/10.3390/ijerph17062097
APA StyleKamandulis, S., Juodsnukis, A., Stanislovaitiene, J., Zuoziene, I. J., Bogdelis, A., Mickevicius, M., Eimantas, N., Snieckus, A., Olstad, B. H., & Venckunas, T. (2020). Daily Resting Heart Rate Variability in Adolescent Swimmers during 11 Weeks of Training. International Journal of Environmental Research and Public Health, 17(6), 2097. https://doi.org/10.3390/ijerph17062097






