The Repolarization Period during the Head-Up Tilt Test in Children with Vasovagal Syncope
Abstract
1. Introduction
2. Materials and Methods
2.1. The Studied Subjects
2.1.1. Head-Up Tilt Table Test
2.1.2. Electrography
2.2. Statistical Analysis
2.3. Ethical Approval
3. Results
3.1. Characteristics of the Study Groups
3.2. T-wave and Repolarization Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wieling, W.; Ganzeboom, K.S.; Saul, J.P. Reflex syncope in children and adolescents. Heart 2004, 90, 1094–1100. [Google Scholar] [CrossRef]
- Kanjwal, K.; Calkins, H. Syncope in children and adolescents. Card. Electrophysiol. Clin. 2015, 33, 397–409. [Google Scholar] [CrossRef]
- Nowakowska, E.; Morka, A.; Szydłowski, L. Syncope in children and adolescents as a sudden, transient, short-term and spontaneously reversible loss of consciousness caused by a decrease in cerebral perfusion. Pediatr. Med. Rodz. 2014, 10, 234–241. [Google Scholar] [CrossRef]
- Brignole, M.; Moya, A.; de Lange, F.J.; Deharo, J.C.; Elliott, P.M.; Fanciulli, A.; Fedorowski, A.; Furlan, R.; Kenny, R.A.; Martín, A.; et al. 2018 ESC Guidelines for the diagnosis and management of syncope. Eur. Heart J. 2018, 39, 1883–1948. [Google Scholar] [CrossRef]
- Brignole, M.; Menozzi, C.; Del Rosso, A.; Costa, S. New classification of haemodynamics of vasovagal syncope: Beyond the VASIS classification. Analysis of the pre-syncopal phase of the tilt test without and with nitroglycerin challenge. Vasovagal Syncope International Study. Europace 2000, 2, 66–76. [Google Scholar] [CrossRef]
- Rogers, G.; O’Flynn, N. NICE guideline: Transient loss of consciousness (blackouts) in adults and young people. Br. J. Gen. Pract. 2011, 61, 40–42. [Google Scholar] [CrossRef]
- Sutton, R.; Petersen, M.; Brignole, M.; Raviele, A.; Menozzi, C.; Giani, P. Proposed classification for vasovagal syncope. Eur. J. Card. Pacing Electrophysiol. 1992, 2, 180–186. [Google Scholar]
- Dovgalyuk, J.; Holstege, C.; Mattu, A.; Brady, W.J. The electrocardiogram in the patient with syncope. Am. J. Emerg. Med. 2007, 25, 688–701. [Google Scholar] [CrossRef]
- Yan, G.X.; Antzelevitch, C. Cellular basis for the normal T wave and the electrocardiographic manifestation of the long QT syndrome. Circulation 1998, 98, 1928–1936. [Google Scholar] [CrossRef]
- Watanabe, N.; Kobayashi, Y.; Tanno, K.; Miyoshi, F.; Asano, T.; Kawamura, M.; Mikami, Y.; Adachi, T.; Ryu, S.; Miyata, A.; et al. Transmural dispersion of repolarization and ventricular tachyarrhythmias. J. Electrocardiol. 2004, 37, 191–200. [Google Scholar] [CrossRef]
- Sucu, M.; Ozer, O.; Davutoglu, V.; Ercan, S.; Yuce, M.; Coscun, F.Y. Relationship between Neurocardiogenic Syncope and Ventricular Repolarization. Pacing Clin. Electrophysiol. 2015, 38, 625–629. [Google Scholar] [CrossRef]
- Rautaharju, P.M.; Surawicz, B.; Gettes, L.S.; Bailey, J.J.; Childers, R.; Deal, B.J.; Gorgels, A.; Hancock, E.W.; Josephson, M.; Kligfield, P.; et al. AHA/ACCF/HRS recommendations for the standardization and interpretation of the electrocardiogram: part IV: The ST segment, T and U waves, and the QT interval: A scientific statement from the American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; the American College of Cardiology Foundation; and the Heart Rhythm Society: Endorsed by the International Society for Computerized Electrocardiology. Circulation 2009, 119, e241–e250. [Google Scholar]
- Antzelevitch, C.; Di Diego, J.M. Tpeak-Tend interval as a marker of arrhythmic risk. Heart Rhythm. 2019, 16, 954–955. [Google Scholar] [CrossRef]
- Mayuga, K.A.; Fouad-Tarazi, F. Dynamic changes in T-wave amplitude during tilt table testing: Correlation with outcomes. Ann. Noninvas. Electr. 2007, 12, 246–250. [Google Scholar] [CrossRef]
- Zhang, L.; Timothy, K.W.; Vincent, G.M.; Zhang, L.; Timothy, K.W.; Vincent, G.M.; Lehmann, M.H.; Fox, J.; Giuli, L.C.; Shen, J.; et al. Spectrum of ST-T-wave patterns and repolarization parameters in congenital long-QT syndrome: ECG findings identify genotypes. Circulation 2000, 10, 2849–2855. [Google Scholar] [CrossRef]
- Takenaka, K.; Ai, T.; Shimizu, W.; Kobori, A.; Ninomiya, T.; Otani, H.; Kubota, T.; Kamakura, S.; Horie, M. Exercise stress test amplifies genotype-phenotype correlation in the LQT1 and LQT2 forms of the long-QT syndrome. Circulation 2003, 107, 838–844. [Google Scholar] [CrossRef]
- Walker, B.D.; Krahn, A.D.; Klein, G.J.; Skanes, A.C.; Yee, R.; Wang, J.; Hegele, R.A. Effect of change in posture and exercise on repolarization in patients with long QT syndrome with HERG channel mutations. Can. J. Cardiol. 2005, 21, 33–38. [Google Scholar]
- Hermosillo, A.G.; Falcon, J.C.; Marquez, M.F.; Arteaga, D.; Cárdenas, M. Positive head-up tilt table test in patients with the long QT syndrome. Europace 1999, 1, 213–217. [Google Scholar] [CrossRef]
- Findler, M.; Birger, A.; Diamant, S.; Viskin, S. Effects of head-up tilt-table test on the QT interval. Ann. Noninvas. Electr. 2010, 15, 245–249. [Google Scholar] [CrossRef]
- Huh, J.; Noh, C.I.; Choi, J.Y.; Yun, Y.S. The effect of beta adrenergic stimulation on QT and QTc interval in syncope children with or without coexisting ventricular arrhythmias. J. Korean Med. Sci. 2003, 18, 179–183. [Google Scholar] [CrossRef]
- Buttà, C.; Tuttolomondo, A.; Casuccio, A.; Di Raimondo, D.; Giarrusso, L.; Miceli, G.; Lo Vecchio, S.; Canino, B.; Licata, G.; Pinto, A. Use of QT intervals for a more accurate diagnose of syncope and evaluation of syncope severity. Int. J. Clin. Pract. 2014, 68, 864–870. [Google Scholar] [CrossRef][Green Version]
- Yamaguchi, M.; Shimizu, M.; Ino, H.; Terai, H.; Uchiyama, K.; Oe, K.; Mabuchi, H. T wave peak-to-end interval and QT dispersion in acquired long QT syndrome: A new index for arrhythmogenicity. Clin. Sci. 2003, 105, 671–676. [Google Scholar] [CrossRef]
- Topilski, I.; Rogowski, O.; Rosso, R.; Justo, D.; Copperman, Y.; Glikson, M.; Belhassen, B. The morphology of the QT interval predicts torsade de pointes during acquired bradyarrhythmias. J. Am. Coll. Cardiol. 2007, 49, 320–328. [Google Scholar] [CrossRef]
- Côté, J.M. Syncope in children and adolescents: Evaluation and treatment. Paediatr. Child Health 2001, 6, 549–551. [Google Scholar] [CrossRef]
- Macfarlane, P.W. Measurement of QT dispersion. Heart 1998, 80, 421–423. [Google Scholar] [CrossRef][Green Version]
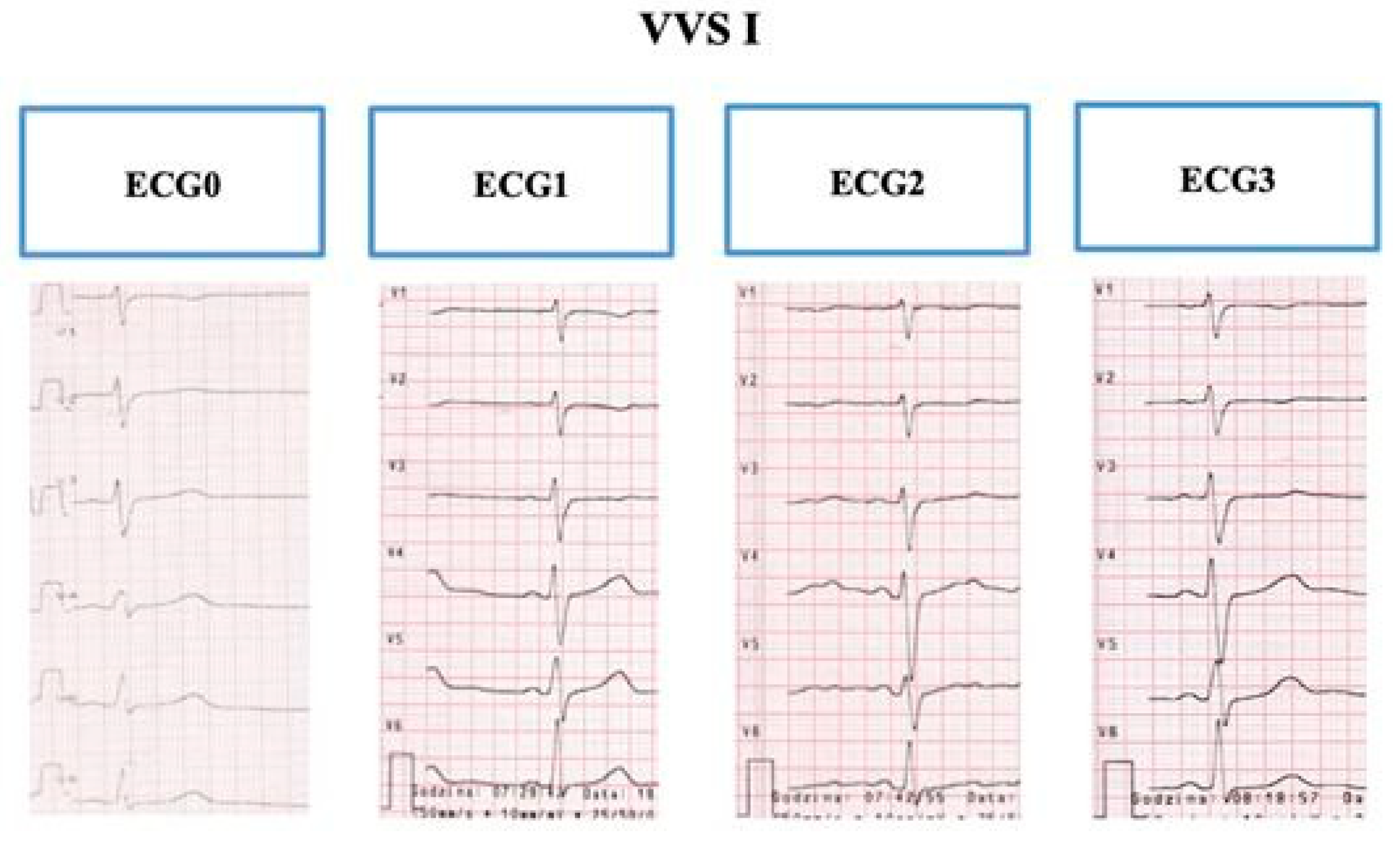
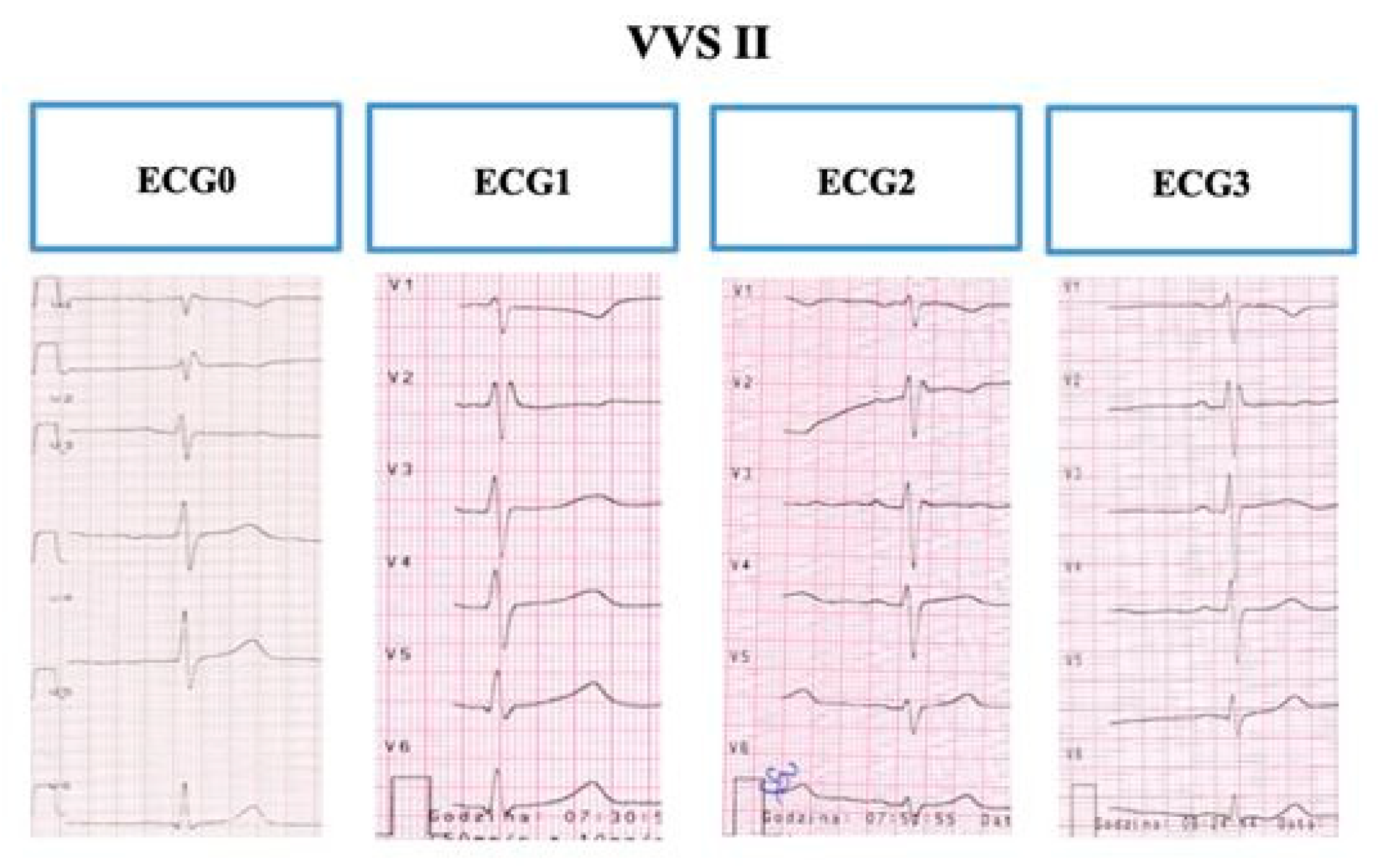
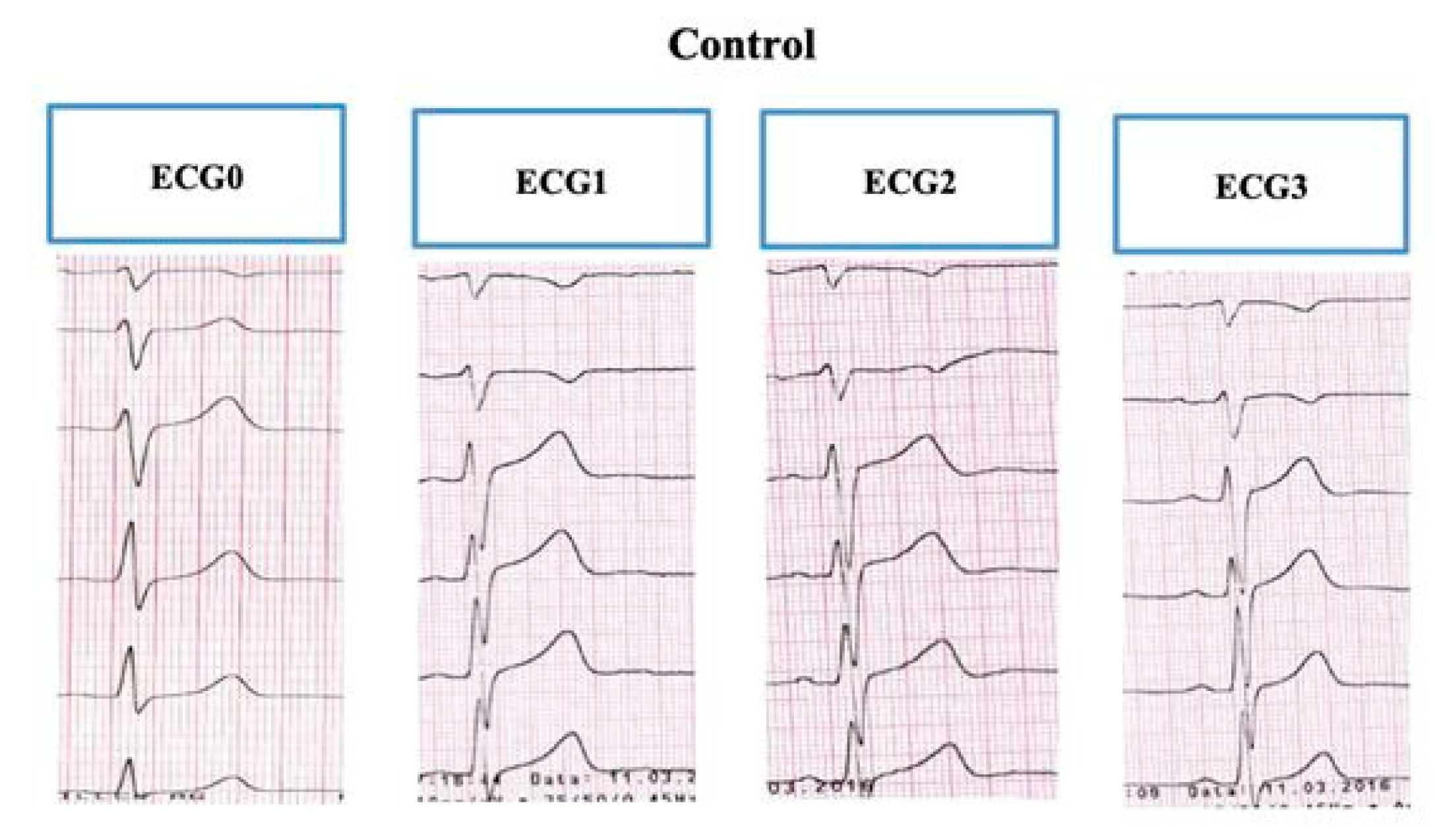
| Characteristics | VVS I 1 (n = 19) | VVS II 2 (n = 11) | Control 3 (n = 30) |
|---|---|---|---|
| Age (at time of hospitalization), years | 16 ± 1.3 * | 16.4 ± 1.3 * | 14.7 ± 2.3 |
| Male, n (%) | 5 (26.3%) | 3 (27.3%) | 8 (26.7%) |
| Female, n (%) | 14 (73.7%) | 8 (72.7%) | 22 (73.3%) |
| Age at which the child had their first episode of syncope | 15.5 ± 1.4 * | 14.5 ± 2.8 | 13.8 ± 2.5 |
| Circumstances of syncope | |||
| After effort, n (%) | 8 (42.1%) | 7 (63.6%) | 13 (43.3%) |
| Emotions/stress, n (%) | 8 (42.1%) * | 3 (27.3%) * | 1 (3.3%) |
| Change of body position, n (%) | 13 (68.4%) * | 6 (54.5%) * | 3 (10%) |
| Long-term verticalization, n (%) | 10 (52.6%) | 6 (54.5%) | 7 (23.3%) |
| Lying and sitting position, n (%) | 0 (0%) | 0 (0%) | 2 (6.7%) |
| Prodromal symptoms | |||
| Headache, n (%) | 8 (42.1%) * | 5 (45.5%) * | 4 (13.3%) |
| Dizziness, n (%) | 7 (36.8%) | 4 (36.4%) | 7 (23.3%) |
| Weakness, n (%) | 4 (21.1%) | 8 (72.7%) * | 3 (10%) |
| Hands tremor, n (%) | 2 (10.5%) | 2 (18.2%) | 1 (3.3%) |
| Pale skin, n (%) | 3 (15.8%) * | 0 (0%) | 0 (0%) |
| Sweating, n (%) | 2 (10.5%) * | 0 (0%) | 1 (3.3%) |
| Tinnitus, n (%) | 2 (10.5%) | 2 (18.2%) | 1 (3.3%) |
| Blurred vision, n (%) | 5 (26.3%) | 5 (45.5%) | 6 (20%) |
| Dyspnea, n (%) | 5 (26.3%) | 3 (27.3%) | 7 (23.3%) |
| Palpitations, n (%) | 3 (15.8%) | 2 (18.2%) | 8 (27.7%) |
| Chest pain, n (%) | 3 (15.8%) | 1 (9.1%) | 8 (26.7%) |
| Numbness in the limbs, n (%) | 0 (0%) | 0 (0%) | 3 (10%) |
| Hot feeling, n (%) | 0 (0%) * | 0 (0%) * | 1 (3.3%) |
| Nausea, n (%) | 0 (0%) * | 0 (0%) * | 3 (10%) |
| Fatigue, n (%) | 0 (0%) * | 0 (0%) * | 3 (10%) |
| QT Variables | VVS I 1 (n = 19) | VVS II 2 (n = 11) | Control 3 (n = 30) | p | p | p | |
|---|---|---|---|---|---|---|---|
| Mean (SD), ms | Mean (SD), ms | Mean (SD), ms | (VVS I vs. VVS II) | (VVS I vs. Control) | (VVS II vs. Control) | ||
| RR | 843.2 (136.6) | 864.6 (131.6) | 860.7 (208.7) | 0.553 | 0.748 | 0.955 | |
| ECG0 | QT | 380.0 (22.9) | 375.5 (16.9) | 370.0 (20.7) | 0.582 | 0.120 | 0.439 |
| QTc | 416.0 (14.8) | 406.7 (29.6) | 403.4 (29.3) | 0.350 | 0.09 | 0.752 | |
| QTp | 289.5 (21.5) | 285.5 (16.9) | 290.7 (20.8) | 0.445 | 0.848 | 0.462 | |
| QTpc | 316.6 (10.9) | 309.1 (23.0) | 316.9 (26.7) | 0.326 | 0.956 | 0.395 | |
| TpTe | 90.5 (5.2) | 90.0 (0.0) | 79.3 (5.2) | 0.832 | <0.001 * | <0.001 * | |
| RR | 827.4 (145.7) | 790.9 (85.4) | 861.3 (212.4) | 0.457 | 0.544 | 0.295 | |
| ECG1 | QT | 385.8 (21.2) | 377.3 (14.9) | 384.7 (31.5) | 0.251 | 0.892 | 0.461 |
| QTc | 427.4 (25.5) | 425.6 (22.4) | 419.0 (27.0) | 0.849 | 0.285 | 0.474 | |
| QTp | 294.7 (21.2) | 288.2 (15.4) | 306.0 (29.2) | 0.378 | 0.153 | 0.062 | |
| QTpc | 326.2 (20.2) | 325.0 (18.5) | 333.1 (23.7) | 0.870 | 0.301 | 0.313 | |
| TpTe | 91.1 (4.6) | 89.1 (3.0) | 78.7 (5.7) | 0.420 | <0.001 * | <0.001 * | |
| RR | 595.3 (72.5) | 650.9 (100.1) | 635.3 (206.5) | 0.089 | 0.421 | 0.813 | |
| ECG2 | QT | 347.4 (19.4) | 349.1 (22.6) | 348.0 (34.8) | 0.827 | 0.943 | 0.924 |
| QTc | 451.3 (13.4) | 434.4 (15.4) | 442.3 (20.3) | 0.004* | 0.092 | 0.253 | |
| QTp | 247.4 (19.1) | 260.9 (22.1) | 283.0 (31.4) | 0.085 | <0.001 * | 0.039 * | |
| QTpc | 321.0 (11.3) | 324.3 (12.2) | 359.4 (19.8) | 0.470 | <0.001 * | <0.001 * | |
| TpTe | 100.0 (3.3) | 88.2 (4.0) | 65.0 (6.8) | <0.001* | <0.001 * | <0.001 * | |
| RR | 743.7 (170.5) | 787.3 (185.9) | 788.3 (191.9) | 0.519 | 0.412 | 0.987 | |
| ECG3 | QT | 368.4 (29.9) | 365.5 (21.6) | 368.0 (29.8) | 0.776 | 0.962 | 0.797 |
| QTc | 431.8 (25.5) | 417.4 (31.1) | 419.6 (28.2) | 0.180 | 0.132 | 0.831 | |
| QTp | 275.8 (28.5) | 275.5 (21.6) | 290.3 (27.4) | 0.973 | 0.081 | 0.113 | |
| QTpc | 322.5 (16.8) | 314.1 (21.1) | 330.8 (24.0) | 0.239 | 0.162 | 0.049 * | |
| TpTe | 92.6 (4.5) | 90.0 (0.0) | 77.7 (5.7) | 0.250 | <0.001 * | <0.001 * | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kolarczyk, E.; Markiewicz-Łoskot, G.; Szydłowski, L. The Repolarization Period during the Head-Up Tilt Test in Children with Vasovagal Syncope. Int. J. Environ. Res. Public Health 2020, 17, 1908. https://doi.org/10.3390/ijerph17061908
Kolarczyk E, Markiewicz-Łoskot G, Szydłowski L. The Repolarization Period during the Head-Up Tilt Test in Children with Vasovagal Syncope. International Journal of Environmental Research and Public Health. 2020; 17(6):1908. https://doi.org/10.3390/ijerph17061908
Chicago/Turabian StyleKolarczyk, Ewelina, Grażyna Markiewicz-Łoskot, and Lesław Szydłowski. 2020. "The Repolarization Period during the Head-Up Tilt Test in Children with Vasovagal Syncope" International Journal of Environmental Research and Public Health 17, no. 6: 1908. https://doi.org/10.3390/ijerph17061908
APA StyleKolarczyk, E., Markiewicz-Łoskot, G., & Szydłowski, L. (2020). The Repolarization Period during the Head-Up Tilt Test in Children with Vasovagal Syncope. International Journal of Environmental Research and Public Health, 17(6), 1908. https://doi.org/10.3390/ijerph17061908





