Test-Retest Reliability of Vibration Perception Threshold Test in People with Type 2 Diabetes Mellitus
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Calculation
2.2. Participants
2.3. Procedure
2.4. Vibratron II ®
2.5. Sensitivity Recording
2.6. Setting the Threshold
2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kerner, W.; Brückel, J. Definition, Classification and Diagnosis of Diabetes Mellitus. Exp. Clin. Endocrinol. Diabetes 2014, 122, 384–386. [Google Scholar] [CrossRef] [PubMed]
- Ogurtsova, K.; da Rocha Fernandes, J.D.; Huang, Y.; Linnenkamp, U.; Guariguata, L.; Cho, N.H.; Cavan, D.; Shaw, J.E.; Makaroff, L.E. IDF Diabetes Atlas: Global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res. Clin. Pract. 2017, 128, 40–50. [Google Scholar] [CrossRef]
- Yang, W.; Dall, T.M.; Halder, P.; Gallo, P.; Kowal, S.L.; Hogan, P.F.; Petersen, M. Economic costs of diabetes in the U.S. in 2012. Diabetes Care 2013, 36, 1033–1046. [Google Scholar]
- Boulton, A.J.M.; Malik, R.A.; Arezzo, J.C.; Sosenko, J.M. Diabetic somatic neuropathies. Diabetes Care 2004, 27, 1458–1486. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.K. Classification, differential diagnosis, and staging of diabetic peripheral neuropathy. Diabetes 1997, 46, S54–S57. [Google Scholar] [CrossRef]
- Andersen, H.; Nielsen, S.; Mogensen, C.E.; Jakobsen, J. Muscle strength in type 2 diabetes. Diabetes 2004, 53, 1543–1548. [Google Scholar] [CrossRef]
- Mustapa, A.; Justine, M.; Mohd Mustafah, N.; Jamil, N.; Manaf, H. Postural Control and Gait Performance in the Diabetic Peripheral Neuropathy: A Systematic Review. Biomed Res. Int. 2016, 2016, 9305025. [Google Scholar] [CrossRef]
- Van Deursen, R.W.M.; Simoneau, G.G. Foot and ankle sensory neuropathy, proprioception, and postural stability. J. Orthop. Sports Phys. Ther. 1999, 29, 718–726. [Google Scholar] [CrossRef]
- Ducic, I.; Short, K.W.; Dellon, A.L.; Disa, J.J. Relationship between loss of pedal sensibility, balance, and falls in patients with peripheral neuropathy. Ann. Plast. Surg. 2004, 52, 535–540. [Google Scholar] [CrossRef]
- Boulton, A.J.M. Diabetic neuropathy and foot complications. Handbook Clin. Neurol. 2014, 126, 97–107. [Google Scholar]
- Young, M.J.; Breddy, J.L.; Veves, A.; Boulton, A.J.M. The prediction of diabetic neuropathic foot ulceration using vibration perception thresholds: A prospective study. Diabetes Care 1994, 17, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Siao, P.; Cros, D.P. Quantitative sensory testing. Phys. Med. Rehabil. Clin. N. Am. 2003, 14, 261–286. [Google Scholar] [CrossRef]
- Hernández-Mocholi, M.A.; Adsuar, J.C.; Davila Romero, C.; Prieto Prieto, J.; Domínguez-Munoz, F.J.; Olivares, P.R. Fiabilidad test-retest del umbral de sensibilidad a la vibración periférica en los pacientes con dolor crónico de espalda baja. Rehabilitacion 2013, 47, 82–89. [Google Scholar] [CrossRef]
- Byl, N.N.; Holland, S.; Jurek, A.; Hu, S.S. Postural imbalance and vibratory sensitivity in patients with idiopathic scoliosis: Implications for treatment. J. Orthop. Sports Phys. Ther. 1997, 26, 60–68. [Google Scholar] [CrossRef]
- Dominguez-Muñoz, F.J.; Hernández-Mocholi, M.A.; Manso, L.J.; Collado-Mateo, D.; Villafaina, S.; Adsuar, J.C.; Gusi, N. Test-retest reliability of kinematic parameters of timed up and go in people with type 2 diabetes. Appl. Sci. 2019, 9, 4709. [Google Scholar] [CrossRef]
- Dixon, C.J.; Knight, T.; Binns, E.; Ihaka, B.; O’Brien, D. Clinical measures of balance in people with type two diabetes: A systematic literature review. Gait Posture 2017, 58, 325–332. [Google Scholar] [CrossRef]
- Munro, B. Statistical Methods for Health Care Research, 4th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2005. [Google Scholar]
- Deng, H.; He, F.; Zhang, S.; Calleman, C.J.; Costa, L.G. Quantitative measurements of vibration threshold in healthy adults and acrylamide workers. Int. Arch. Occup. Environ. Health 1993, 65, 53–56. [Google Scholar] [CrossRef]
- Weir, J.P. Quantifying Test-Retest Reliability Using the Intraclass Correlation Coefficient and the SEM. J. Strength Cond. Res. 2005, 19, 231. [Google Scholar]
- Flansbjer, U.B.; Holmbäck, A.M.; Downham, D.; Lexell, J. What change in isokinetic knee muscle strength can be detected in men and women with hemiparesis after stroke? Clin. Rehabil. 2005, 19, 514–522. [Google Scholar] [CrossRef]
- Martin Bland, J.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 327, 307–310. [Google Scholar] [CrossRef]
- Duke, J.; McEvoy, M.; Sibbritt, D.; Guest, M.; Smith, W.; Attia, J. Vibrotactile threshold measurement for detecting peripheral neuropathy: Defining variability and a normal range for clinical and research use. Diabetologia 2007, 50, 2305–2312. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Van Deursen, R.W.M.; Sanchez, M.M.; Derr, J.A.; Becker, M.B.; Ulbrecht, J.S.; Cavanagh, P.R. Vibration perception threshold testing in patients with diabetic neuropathy: Ceiling effects and reliability. Diabet. Med. 2001, 18, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Riandini, T.; Wee, H.L.; Khoo, E.Y.H.; Tai, B.C.; Wang, W.; Koh, G.C.H.; Tai, E.S.; Tavintharan, S.; Chandran, K.; Hwang, S.W.; et al. Functional status mediates the association between peripheral neuropathy and health-related quality of life in individuals with diabetes. Acta Diabetol. 2018, 55, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Martinelli, A.R.; Mantovani, A.M.; Nozabieli, A.J.L.; Ferreira, D.M.A.; Barela, J.A.; de Camargo, M.R.; Fregonesi, C.E.P.T. Muscle strength and ankle mobility for the gait parameters in diabetic neuropathies. Foot 2013, 23, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Manor, B.; Li, L. Characteristics of functional gait among people with and without peripheral neuropathy. Gait Posture 2009, 30, 253–256. [Google Scholar] [CrossRef]
- Richardson, J.K.; Hurvitz, E.A. Peripheral neuropathy: A true risk factor for falls. J. Gerontol. A. Biol. Sci. Med. Sci. 1995, 50, M211–M215. [Google Scholar] [CrossRef]
- Wongsurawat, N.; Armbrecht, H.J. Insulin modulates the stimulation of renal 1,25-dihydroxyvitamin D3 production by parathyroid hormone. Acta Endocrinol. 1985, 109, 243–248. [Google Scholar] [CrossRef]
- Pietschmann, P.; Schernthaner, G.; Woloszczuk, W. Serum osteocalcin levels in diabetes mellitus: Analysis of the type of diabetes and microvascular complications. Diabetologia 1988, 31, 892–895. [Google Scholar] [CrossRef]
- Schwartz, A.V.; Sellmeyer, D.E.; Ensrud, K.E.; Cauley, J.A.; Tabor, H.K.; Schreiner, P.J.; Jamal, S.A.; Black, D.M.; Cummings, S.R. Older women with diabetes have an increased risk of fracture: A prospective study. J. Clin. Endocrinol. Metab. 2001, 86, 32–38. [Google Scholar] [CrossRef]
- Aaserud, O.; Juntunen, J.; Matikainen, E. Vibration sensitivity thresholds: Methodological considerations. Acta Neurol. Scand. 1990, 82, 277–283. [Google Scholar] [CrossRef]
- Goldberg, J.M.; Lindblom, U. Standardised method of determining vibratory perception thresholds for diagnosis and screening in neurological investigation. J. Neurol. Neurosurg. Psychiatry 1979, 42, 793–803. [Google Scholar] [CrossRef] [PubMed]
- Dyck, P.J.; Zimmerman, I.R.; O’ Brien, P.C.; Ness, A.; Caskey, P.E.; Karnes, J.; Bushek, W. Introduction of automated systems to evaluate touch-pressure, vibration, and thermal cutaneous sensation in man. Ann. Neurol. 1978, 4, 502–510. [Google Scholar] [CrossRef] [PubMed]
- Gregg, E.C. Absolute measurement of the vibratory threshold. Arch. Neurol. Psychiatry 1951, 66, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Peters, E.W.; Bienfait, H.M.E.; De Visser, M.; De Haan, R.J. The reliability of assessment of vibration sense. Acta Neurol. Scand. 2003, 107, 293–298. [Google Scholar] [CrossRef]
- de Neeling, J.N.D.; Beks, P.J.; Bertelsmann, F.W.; Heine, R.J.; Bouter, L.M. Sensory thresholds in older adults: Reproducibility and reference values. Muscle Nerve 1994, 17, 454–461. [Google Scholar] [CrossRef]
- Gerr, F.E.; Letz, R. Reliability of a widely used test of peripheral cutaneous vibration sensitivity and a comparison of two testing protocols. Br. J. Ind. Med. 1988, 45, 635–639. [Google Scholar] [CrossRef]
- Louraki, M.; Tsentidis, C.; Kallinikou, D.; Katsalouli, M.; Kanaka-Gantenbein, C.; Kafassi, N.; Papathanasiou, A.; Karavanaki, K. Reproducibility of vibration perception threshold values in children and adolescents with type 1 diabetes mellitus and associated factors. Prim. Care Diabetes 2014, 8, 147–157. [Google Scholar] [CrossRef]
- Bloom, S.; Till, S.; Sonksen, P.; Smith, S. Use of a biothesiometer to measure individual vibration thresholds and their variation in 519 non-diabetic subjects. Br. Med. J. 1984, 288, 1793–1795. [Google Scholar] [CrossRef]
- Halonen, P. Quantitative vibration perception thresholds in healthy subjects of working age. Eur. J. Appl. Physiol. Occup. Physiol. 1986, 54, 647–655. [Google Scholar] [CrossRef]
- Sosenko, J.M.; Gadia, M.T.; Fournier, A.M.; O’Connell, M.T.; Aguiar, M.C.; Skyler, J.S. Body stature as a risk factor for diabetic sensory neuropathy. Am. J. Med. 1986, 80, 1031–1034. [Google Scholar] [CrossRef]
- Iandolo, R.; Carè, M.; Shah, V.A.; Schiavi, S.; Bommarito, G.; Boffa, G.; Giannoni, P.; Inglese, M.; Mrotek, L.A.; Scheidt, R.A.; et al. A two alternative forced choice method for assessing vibrotactile discrimination thresholds in the lower limb. Somatosens. Mot. Res. 2019, 36, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Campbell, W.W.; Ward, L.C.; Swift, T.R. Nerve conduction velocity varies inversely with height. Muscle Nerve 1981, 4, 520–523. [Google Scholar] [CrossRef] [PubMed]
- Soudmand, R.; Ward, L.C.; Swift, T.R.; Hawes, A. Effect of height on nerve conduction velocity. Neurology 1982, 32, 407–410. [Google Scholar] [CrossRef] [PubMed]
- Hyllienmark, L.; Brismar, T.; Ludvigsson, J. Subclinical nerve dysfunction in children and adolescents with IDDM. Diabetologia 1995, 38, 685–692. [Google Scholar] [CrossRef]
- Louraki, M.; Karayianni, C.; Kanaka-Gantenbein, C.; Katsalouli, M.; Karavanaki, K. Peripheral neuropathy in children with type 1 diabetes. Diabetes Metab. 2012, 38, 281–289. [Google Scholar] [CrossRef]
- Cinar, N.; Sahin, S.; Sahin, M.; Okluoglu, T.; Karsidag, S. Effects of anthropometric factors on nerve conduction an electrophysiologic study of feet. J. Am. Podiatr. Med. Assoc. 2013, 103, 43–49. [Google Scholar] [CrossRef]
- Hilz, M.J.; Axelrod, F.B.; Hermann, K.; Haertl, U.; Duetsch, M.; Neundörfer, B. Normative values of vibratory perception in 530 children, juveniles and adults aged 3–79 years. J. Neurol. Sci. 1998, 159, 219–225. [Google Scholar] [CrossRef]
- Hwu, C.M.; Chang, H.Y.; Chen, J.Y.; Wang, S.L.; Ho, L.T.; Pan, W.H. Quantitative vibration perception thresholds in normal and diabetic Chinese: Influence of age, height and body mass index. Neuroepidemiology 2002, 21, 271–278. [Google Scholar] [CrossRef]
- Maiya, A.G.; Parameshwar, A.; Hande, M.; Nandalike, V. Relationship Between Glycated Hemoglobin and Vibration Perception Threshold in Diabetic Peripheral Neuropathy. Int. J. Low. Extrem. Wounds 2019. [Google Scholar] [CrossRef]
- Kullberg, C.E.; Arnqvist, H.J. Impaired Vibration Perception Threshold and Long-term Mean HbA1c in Patients with Type 1 Diabetes Mellitus. Diabet. Med. 1996, 13, 1027–1032. [Google Scholar] [CrossRef]
- Hillson, R.M.; Hockaday, T.D.R.; Newton, D.J. Hyperglycaemia is one correlate of deterioration in vibration sense during the 5 years after diagnosis of Type 2 (non-insulin-dependent) diabetes. Diabetologia 1984, 26, 122–126. [Google Scholar] [CrossRef] [PubMed]
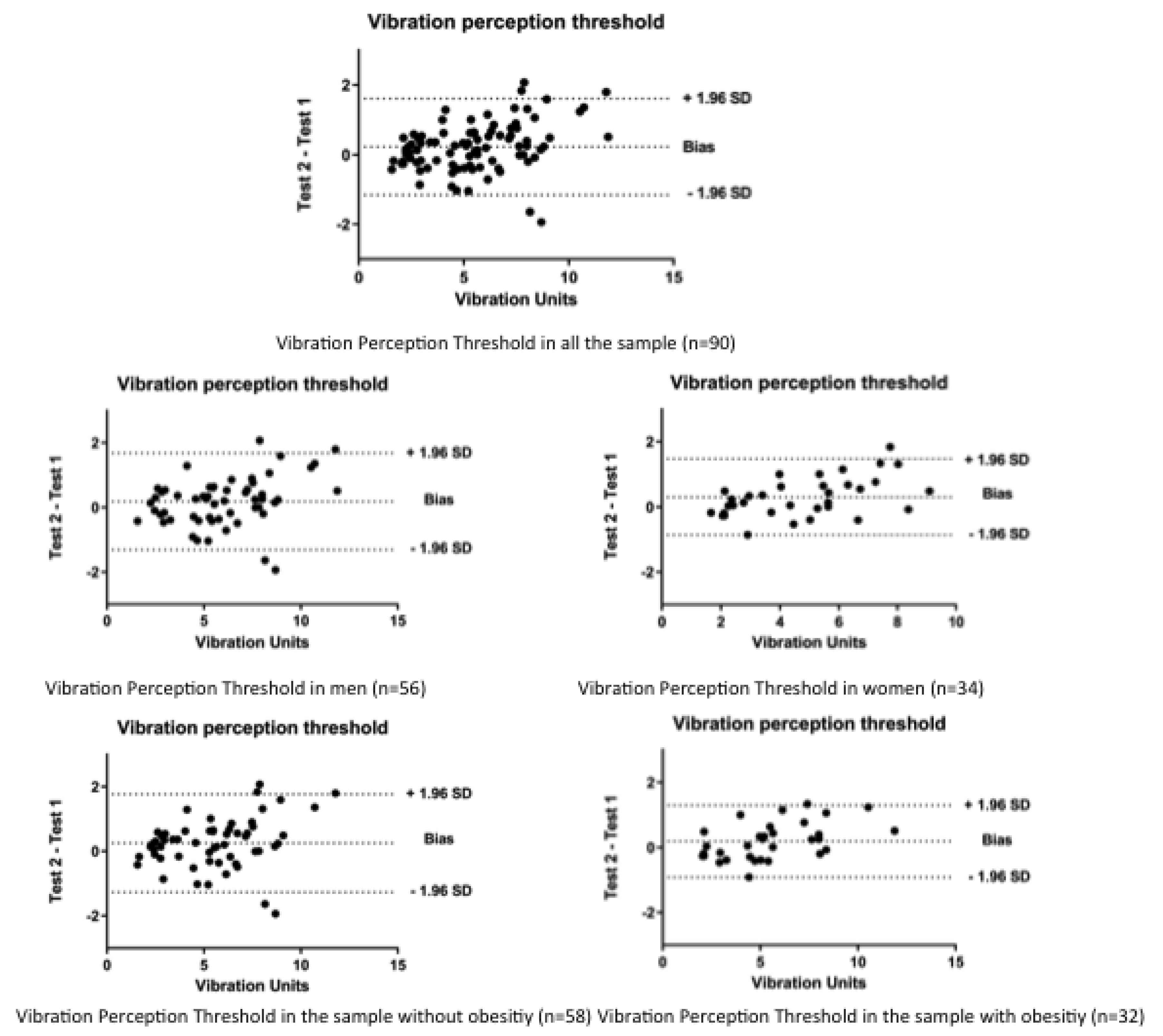
| All (n = 90) | Men (n = 56) | Women (n = 34) | Without Obesity (n = 58) | With Obesity (n = 32) | |
|---|---|---|---|---|---|
| Age (years) | 65.64 ± 8.65 | 65.51 ± 8.16 | 65.85 ± 9.52 | 66.01 ± 8.16 | 64.96 ± 9.57 |
| Height (cm) | 164.89 ± 10.00 | 169.93 ± 7.80 | 156.59 ± 7.37 | 165.76 ± 9.50 | 163.31 ± 10.82 |
| Weight (kg) * | 80.63 ± 16.19 | 85.41 ± 17.28 | 72.74 ± 10.29 | 74.74 ± 10.47 | 91.29 ± 19.20 |
| Fat Mass (%) * | 32.97 ± 7.49 | 28.89 ± 5.20 | 39.68 ± 5.62 | 30.67 ± 6.50 | 37.12 ± 7.47 |
| Fat Free Mass (%) * | 66.84 ± 8.08 | 71.11 ± 5.21 | 59.80 ± 7.00 | 69.33 ± 6.49 | 62.32 ± 8.79 |
| Body Water (%) * | 49.08 ± 5.47 | 52.05 ± 3.81 | 44.20 ± 4.11 | 50.77 ± 4.73 | 46.03 ± 5.46 |
| Basal Metabolism (kcal) * | 161.61 ± 482.97 | 1818.70 ± 524.72 | 1323.47 ± 113.90 | 1528.02 ± 260.39 | 1819.37 ± 698.95 |
| BMI (kg/m2) * | 29.65 ± 4.39 | 29.63 ± 4.77 | 29.67 ± 3.74 | 27.10 ± 1.94 | 34.26 ± 3.80 |
| Glycosylated Hemoglobin (%) | 6.78 ± 1.02 | 6.85 ± 0.98 | 6.67 ± 1.09 | 6.79 ± 1.13 | 6.77 ± 0.80 |
| Years of diagnosis | 9.96 ± 8.83 | 9.55 ± 7.49 | 10.62 ± 10.78 | 9.26 ± 7.64 | 11.22 ± 10.68 |
| Falls in 6 months | 0.25 ± 0.71 | 0.12 ± 0.50 | 0.47 ± 0.92 | 0.31 ± 0.84 | 0.15 ± 0.36 |
| Falls in 1 year | 0.46 ± 1.21 | 0.17 ± 0.74 | 0.94 ± 1.65 | 0.53 ± 1.42 | 0.34 ± 0.70 |
| Day 1 Mean ± TD | Day 2 Mean ± TD | p * | ||
|---|---|---|---|---|
| All (n = 90) | Vibration Threshold (vu) | 5.43 ± 2.32 | 5.66 ± 2.55 | 0.003 |
| Men (n = 56) | Vibration Threshold (vu) | 5.93 ± 2.37 | 6.11 ± 2.61 | 0.076 |
| Women (n = 34) | Vibration Threshold (vu) | 4.59 ± 2.00 | 4.90 ± 2.30 | 0.006 |
| Without obesity (n = 58) | Vibration Threshold (vu) | 5.44 ± 2.31 | 5.69 ± 2.51 | 0.017 |
| With obesity (n = 32) | Vibration Threshold (vu) | 5.40 ± 2.38 | 5.59 ± 2.66 | 0.064 |
| ICC (95% CI) | SEM (Nvu) | SEM (%) | SRD (Nvu) | SRD (%) | ||
|---|---|---|---|---|---|---|
| All (n = 90) | Vibration threshold (vu) | 0.958 (0.938, 0.972) | 0.49 | 8.99 | 1.38 | 24.94 |
| Men (n = 56) | Vibration threshold (vu) | 0.953 (0.922, 0.972) | 0.53 | 8.96 | 1.49 | 24.85 |
| Women (n = 34) | Vibration threshold (vu) | 0.962 (0.924, 0.981) | 0.41 | 8.83 | 1.16 | 24.48 |
| Without obesity (n = 58) | Vibration threshold (vu) | 0.949 (0.915, 0.969) | 0.54 | 9.77 | 1.50 | 27.10 |
| With obesity (n = 32) | Vibration threshold (vu) | 0.975 (0.949, 0.988) | 0.39 | 7.25 | 1.10 | 20.09 |
| Variables | Vibration Perception Threshold (R2 = 0.362) | ||
|---|---|---|---|
| β | SE | p | |
| Age | 0.163 | 0.026 | <0.001 |
| Height | 0.108 | 0.023 | <0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domínguez-Muñoz, F.J.; Adsuar, J.C.; Villafaina, S.; García-Gordillo, M.A.; Hernández-Mocholí, M.Á.; Collado-Mateo, D.; Gusi, N. Test-Retest Reliability of Vibration Perception Threshold Test in People with Type 2 Diabetes Mellitus. Int. J. Environ. Res. Public Health 2020, 17, 1773. https://doi.org/10.3390/ijerph17051773
Domínguez-Muñoz FJ, Adsuar JC, Villafaina S, García-Gordillo MA, Hernández-Mocholí MÁ, Collado-Mateo D, Gusi N. Test-Retest Reliability of Vibration Perception Threshold Test in People with Type 2 Diabetes Mellitus. International Journal of Environmental Research and Public Health. 2020; 17(5):1773. https://doi.org/10.3390/ijerph17051773
Chicago/Turabian StyleDomínguez-Muñoz, Francisco Javier, José Carmelo Adsuar, Santos Villafaina, Miguel Angel García-Gordillo, Miguel Ángel Hernández-Mocholí, Daniel Collado-Mateo, and Narcís Gusi. 2020. "Test-Retest Reliability of Vibration Perception Threshold Test in People with Type 2 Diabetes Mellitus" International Journal of Environmental Research and Public Health 17, no. 5: 1773. https://doi.org/10.3390/ijerph17051773
APA StyleDomínguez-Muñoz, F. J., Adsuar, J. C., Villafaina, S., García-Gordillo, M. A., Hernández-Mocholí, M. Á., Collado-Mateo, D., & Gusi, N. (2020). Test-Retest Reliability of Vibration Perception Threshold Test in People with Type 2 Diabetes Mellitus. International Journal of Environmental Research and Public Health, 17(5), 1773. https://doi.org/10.3390/ijerph17051773











