The Impact of Shift-Work and Night Shift-Work on Thyroid: A Systematic Review
Abstract
1. Introduction
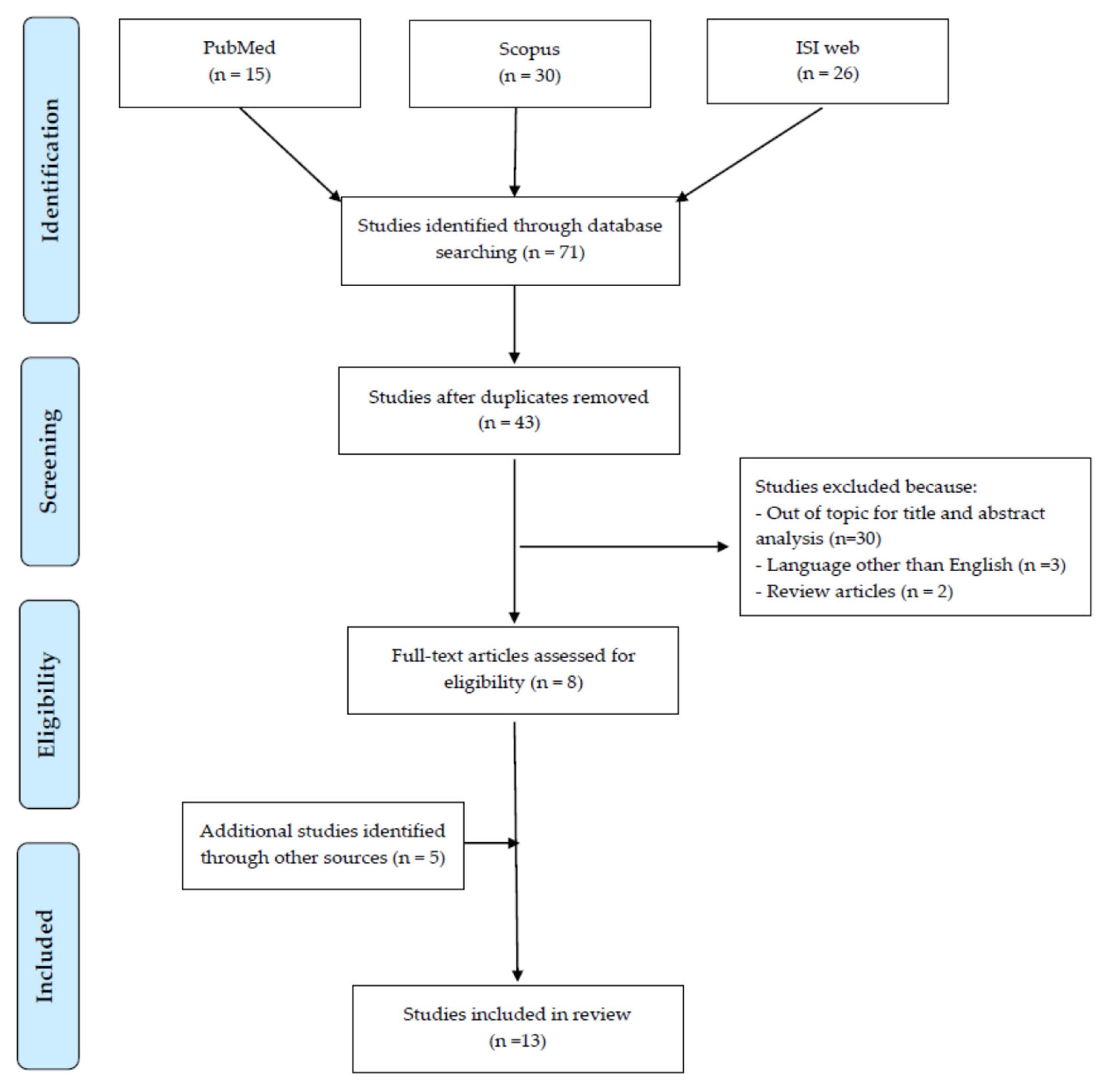
2. Materials and Methods
3. Results
3.1. Night Shift-Work and Thyroid Hormonal Changes
3.1.1. Healthcare Sector
3.1.2. Industrial Sector
3.2. Night on-Call-Work and Thyroid Hormonal Changes
3.3. Shift Work and Other Thyroid Effects
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ikegami, K.; Refetoff, S.; Van Cauter, E.; Yoshimura, T. Interconnection between circadian clocks and thyroid function. Nat. Rev. Endocrinol. 2019, 15, 590–600. [Google Scholar] [CrossRef] [PubMed]
- Ortiga-Carvalho, T.M.; Chiamolera, M.I.; Pazos-Moura, C.C.; Wondisford, F.E. Hypothalamus-pituitary-thyroid axis. Compr. Physiol. 2016, 6, 1387–1428. [Google Scholar] [CrossRef] [PubMed]
- Kalsbeek, A.; Fliers, E. Daily regulation of hormone profile. Handb. Exp. Pharmacol. 2013, 217, 185–226. [Google Scholar] [CrossRef]
- Morris, C.J.; Aeschbach, D.; Scheer, F.A. Circadian system, sleep and endocrinology. Mol. Cell. Endocrinol. 2012, 349, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, K.; Leproult, R.; Van Cauter, E. Impact of sleep debt on metabolic and endocrine function. Lancet 1999, 354, 1435–1439. [Google Scholar] [CrossRef]
- Brabant, G.; Prank, K.; Ranft, U.; Schuermeyer, T.; Wagner, T.O.F.; Hauser, H.; Kummer, B.; Feistner, H.; Hesch, R.D.; von sur Mühlen, A. Physiological regulation of circadian and pulsatile thyrotropin secretion in normal man and Woman. J. Clin. Endocr. Metab. 1990, 70, 403–409. [Google Scholar] [CrossRef]
- International Labour Office (ILO). Shift Work. Conditions of Work and Employment Programme. Information Sheet No. WT-8 2004. Available online: https://www.ilo.org/wcmsp5/groups/public/---ed_protect/---protrav/---travail/documents/publication/wcms_170713.pdf (accessed on 8 January 2020).
- Your Europe—European Union. Working Hours. Available online: https://europa.eu/youreurope/business/human-resources/working-hours-holiday-leave/working-hours/index_en.htm#shortcut-5 (accessed on 8 January 2020).
- Fischer, F.M.; Silva-Costa, A.; Griep, R.H.; Smolensky, M.H.; Bohle, P.; Rotenberg, L. Working Time Society consensus statements: Psychosocial stressors relevant to the health and wellbeing of night and shift workers. Ind. Health 2019, 57, 175–183. [Google Scholar] [CrossRef]
- Matheson, A.; O’Brien, L.; Reid, J.A. The impact of shiftwork on health: A literature review. J. Clin. Nurs. 2014, 23, 3309–3320. [Google Scholar] [CrossRef]
- IARC Monographs Vol 124 Group. Carcinogenicity of night shift work. Lancet Oncol. 2019, 20, 1058–1059. [Google Scholar] [CrossRef]
- Proper, K.I.; van de Langenberg, D.; Rodenburg, W.; Vermeulen, R.C.H.; van der Beek, A.J.; van Steeg, H.; van Kerkhof, L.W.M. The Relationship between Shift Work and Metabolic Risk Factors: A Systematic Review of Longitudinal Studies. J. Prev. Med. 2016, 50, e147–e157. [Google Scholar] [CrossRef]
- Kecklund, G.; Axelsson, J. Health consequences of shift work and insufficient sleep. BMJ 2016, 355, i5210. [Google Scholar] [CrossRef] [PubMed]
- American Thyroid Association. World Thyroid Day Aims to Raise Awareness of Disease. 2014. Available online: http://www.thyroid.org (accessed on 26 January 2020).
- Global Wellness Institute. Night Shift Work Is on Rise Globally—And It’s a New Wellness Problem. 2018. Available online: https://globalwellnessinstitute.org/global-wellness-institute-blog/2018/06/19/night-shift-work-is-on-the-rise-globally-and-its-a-new-wellness-problem/ (accessed on 28 January 2020).
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Int. J. Surg. 2010, 8, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomized Studies in Meta-Analysis. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 28 January 2020).
- Modesti, P.A.; Reboldi, G.; Cappuccio, F.P.; Agyemang, C.; Remuzzi, G.; Rapi, S.; Perruolo, E.; Parati, G.; ESH Working Group on CV Risk in Low Resource Settings. Panethnic Differences in Blood Pressure in Europe: A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0147601. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.S.; Barker, A.R.; Wilkinson, K.M.; Abbott, R.A.; Williams, C.A. Is cardiac autonomic function associated with cardiorespiratory fitness and physical activity in children and adolescents? A systematic review of cross-sectional studies. Int. J. Cardio 2017, 236, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Attarchi, M.; Darkhi, H.; Khodarahmian, M.; Dolati, M.; Kashanian, M.; Ghaffari, M.; Mirzamohammadi, E.; Mohammadi, S. Characteristics of menstrual cycle in shift workers. Glob. J. Health Sci. 2013, 5, 163–172. [Google Scholar] [CrossRef]
- Chang, Y.S.; Chen, H.L.; Hsu, C.Y.; Su, S.F.; Liu, C.K.; Hsu, C. Nurses working on fast rotating shifts overestimate cognitive function and the capacity of maintaining wakefulness during the daytime after a rotating shift. SleepMed 2013, 14, 605–613. [Google Scholar] [CrossRef]
- Harbeck, B.; Suefke, S.; Haas, C.S.; Lehnert, H.; Kropp, P.; Moenig, H. No stress after 24-h on-call shifts? J. Occup. Health. 2015, 57, 438–447. [Google Scholar] [CrossRef]
- Korompeli, A.; Sourtzi, P.; Tzavara, C.; Velonakis, E. Rotating shift-related changes in hormone levels in intensive care unit nurses. J. Adv. Nurs. 2009, 65, 1274–1282. [Google Scholar] [CrossRef]
- Kuetting, D.L.R.; Feisst, A.; Sprinkart, A.M.; Homsi, R.; Luetkens, J.; Thomas, D.; Schild, H.H.; Dabir, D. Effects of a 24-hr-shift-related short-term sleep deprivation on cardiac function: A cardiac magnetic resonance-based study. J. Sleep Res. 2019, 28, e12665. [Google Scholar] [CrossRef]
- Malmberg, B.; Persson, R.; Jönsson, B.A.; Erfurth, E.M.; Flisberg, P.; Ranklev, E.; Orbaek, P. Physiological restitution after night-call duty in anaesthesiologists: Impact on metabolic factors. Acta Anaesthesiol. Scand. 2007, 51, 823–830. [Google Scholar] [CrossRef]
- Moon, S.H.; Lee, B.J.; Kim, S.J.; Kim, H.C. Relationship between thyroid stimulating hormone and night shift work. Ann. Occup. Environ. Med. 2016, 28, 53. [Google Scholar] [CrossRef] [PubMed]
- Sathyanarayana, S.M.; Gangadharaiah, S. Night shift working and its impact on sleep quality, calorie intake, and serum thyroid stimulating hormone level among health-care workers in a tertiary hospital, Bengaluru. Natl. J. Physiol. Pharm. Pharmacol. 2018, 8, 1330–1334. [Google Scholar] [CrossRef]
- Shaker, D.; Samir, A.; Zyada, F.; El-Sharkawy, M.; Ekladious, S.M. Impact of shift work on sleep problems, hormonal changes, and features of metabolic syndrome in a sample of Egyptian industrial workers: A cross-sectional study. Middle East Curr. Psychiatry 2018, 25, 91–97. [Google Scholar] [CrossRef]
- Weibel, L.; Brandenberger, G. Disturbances in hormonal profiles of night workers during their usual sleep and work times. J. Biol. Rhythms 1998, 13, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Burdelak, W.; Bukowska, A.; Krysicka, J.; Pepłońska, B. Night work and health status of nurses and midwives. Cross-sectional study. Med Pr. 2012, 63, 517–529. [Google Scholar]
- Magrini, A.; Pietroiusti, A.; Coppeta, L.; Babbucci, A.; Barnaba, E.; Papadia, C.; Iannaccone, U.; Boscolo, P.; Bergamaschi, E.; Bergamaschi, A. Shift work and autoimmune thyroid disorders. Int. J. Immunopathol. Pharmacol. 2006, 19 (Suppl. 4), 31–36. [Google Scholar]
- Rizza, S.; Neri, A.; Capanna, A.; Grecuccio, C.; Pietroiusti, A.; Magrini, A.; Federici, M.; Coppeta, L. Night Shift Working Is Associated with an Increased Risk of Thyroid Nodules. J. Occup. Environ. Med. 2020, 62, 1–3. [Google Scholar] [CrossRef]
- Parker, D.C.; Rossman, L.G.; Pekary, A.E.; Hershman, J.M. Effect of 64-h sleep deprivation on the circadian waveform of TSH: Further evidence of sleep related inhibition of TSH release. J. Clin. Endocrinol. Metab. 1987, 64, 157–161. [Google Scholar] [CrossRef]
- Allan, J.S.; Czeisler, C.A. Persistence of the circadian thyrotropin rhythm under constant conditions and after light-induced shifts of circadian phase. J. Clin. Endocrinol. Metab. 1994, 79, 508–512. [Google Scholar] [CrossRef]
- Boscolo, P.; Youinou, P.; Theoharides, T.C.; Cerulli, G.; Conti, P. Environmental and occupational stress and autoimmunity. Autoimmun. Rev. 2008, 7, 340–343. [Google Scholar] [CrossRef]
- Holmbäck, U.; Forslund, A.; Lowden, A.; Forslund, J.; Akerstedt, T.; Lennernäs, M.; Hambraeus, L.; Stridsberg, M. Endocrine responses to nocturnal eating–possible implications for night work. Eur J Nutr. 2003, 42, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Hollowell, J.G.; Staehling, N.W.; Flanders, W.D.; Hannon, W.H.; Gunter, E.W.; Spencer, C.A.; Braverman, L.E. Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J. Clin. Endocrinol. Metab. 2002, 87, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Boucai, L.; Hollowell, J.G.; Surks, M.I. An approach for development of age-, gender-, and ethnicity-specific thyrotropin reference limits. Thyroid 2011, 21, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Surks, M.I.; Hollowell, J.G. Age-specific distribution of serum thyrotropin and antithyroid antibodies in the US population: Implications for the prevalence of subclinical hypothyroidism. J. Clin. Endocrinol. Metab. 2007, 92, 4575–4582. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Kim, H.I.; Oh, H.K.; Kim, T.H.; Jang, H.W.; Chung, J.H.; Shin, M.H.; Kim, S.W. Age- and gender-specific reference intervals of TSH and free T4 in an iodine-replete area: Data from Korean National Health and Nutrition Examination Survey IV (2013–2015). PLoS ONE 2018, 13, e0190738. [Google Scholar] [CrossRef] [PubMed]
- Roelfsema, F.; Pijl, H.; Kok, P.; Endert, E.; Fliers, E.; Biermasz, N.R.; Pereira, A.M.; Veldhuis, J.D. Thyrotropin secretion in healthy subjects is robust and independent of age and gender, and only weakly dependent on body mass index. J. Clin. Endocrinol. Metab. 2014, 99, 570–578. [Google Scholar] [CrossRef]
- Goichot, B.; Weibel, L.; Chapotot, F.; Gronfier, C.; Piquard, F.; Brandenberger, G. Effect of the shift of the sleep-wake cycle on three robust endocrine markers of the circadian clock. Am. J. Physiol. 1998, 275 Pt 1, E243–E248. [Google Scholar] [CrossRef]
- Yilmaz, B.; Terekeci, H.; Sandal, S.; Kelestimur, F. Endocrine disrupting chemicals: Exposure, effects on human health, mechanism of action, models for testing and strategies for prevention. Rev. Endocr. Metab. Disord. 2019, in press. [Google Scholar] [CrossRef]
- Özütürk, B.; Ersoy, A.; Altan, A.; Uygur, L.M. Comparison of the Effects of Remifentanil and Dexmedetomidine Infusions on Hemodynamic Parameters and Thyroid Hormones. Turk. J. Anaesthesiol. Reanim. 2013, 41, 206–210. [Google Scholar] [CrossRef]
- Şentürk, B.; Umuroğlu, T.; Saraçoğlu, A.; Eti, Z.; Göğüş, F.Y. The Effects of Desflurane and Propofol on the Release of Thyroid Hormones in Euthyroid Patients Undergoing Elective Lumbar Discectomy. Turk. J. Anaesthesiol. Reanim. 2015, 43, 68–72. [Google Scholar] [CrossRef]
- Calsolaro, V.; Pasqualetti, G.; Niccolai, F.; Caraccio, N.; Monzani, F. Thyroid Disrupting Chemicals. Int. J. Mol. Sci. 2017, 18, 2583. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Kim, S.S.; Chung, E.; Dietrich, K.N. Thyroid hormones in relation to lead, mercury, and cadmium exposure in the National Health and Nutrition Examination Survey, 2007–2008. Environ. Health Perspect. 2013, 121, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Iavicoli, I.; Fontana, L.; Bergamaschi, A. The effects of metals as endocrine disruptors. J. Toxicol. Environ. Health B Crit. Rev. 2009, 12, 206–223. [Google Scholar] [CrossRef] [PubMed]
- Uzma, N.; Salar, B.M.; Kumar, B.S.; Aziz, N.; David, M.A.; Reddy, V.D. Impact of organic solvents and environmental pollutants on the physiological function in petrol filling workers. Int. J. Environ. Res. Public Health 2008, 5, 139–146. [Google Scholar] [CrossRef]
- Chen, T.Y.; Hsu, C.C.; Feng, I.J.; Wang, J.J.; Su, S.B.; Guo, H.R.; Huang, C.C.; Lin, H.J. Higher risk for thyroid diseases in physicians than in the general population: A Taiwan nationwide population-based secondary analysis study. QJM 2017, 110, 163–168. [Google Scholar] [CrossRef]
| Reference | Study Location | Study Design | Occupational Sector; Investigated Population | Outcome | Occupational Risk Factors | Additional Information | Results | Quality Rating According to NOS |
|---|---|---|---|---|---|---|---|---|
| Chang et al. [21] | Southern Taiwan | Cross sectional | Healthcare sector Female nurses involved in 2 continuous night shifts (n = 20; mean age ± SD: 26.0 ± 2.0 years); Off duty nurses who had been free of duty for at least 3 days before entering the study (n = 23; mean age 26.1 ± 1.9 years) | Hormonal changes | Night shift-work | Working schedule All subjects had worked daytime shifts, evening-shifts, or had been free of duty for at least 3 days before entering the study day. All subjects had been working in the hospital for at least one year. Biological sampling Blood samples were collected and tested for TSH at the end of a night shift or on the off-duty day. The test was repeated 4 times every 2 h starting at 9:20 am. | Significant increase in TSH level in night shift workers (1.3 ± 0.7; 1.5 ± 0.7, 1.9 ± 0.9, 2.0 ± 0.7 mIU/L at morning time 1 and 2, afternoon time 1 and 2, respectively) compared to off-duty workers (1.0 ± 06, 1.0 ± 06, 1.0 ± 06, 1.1 ± 0.6 mIU/L, at morning time 1, and 2, afternoon time 1 and 2, respectively). | Unsatisfactory |
| Moon et al. [26] | Incheon, Korea | Cross sectional | Healthcare sector Total female adult workers (n = 967; age range: ≤29–≥ 50 years); night shift workers (n = 546); non-night shift workers (n = 421). | Hormonal changes | Night shift-work | Working schedule Night shift workers: ≥4 night shifts per month. Departmental status Nurses (n = 654): general ward, outpatient unit, emergency room, operating room, and/or intensive care unit; workers in other departments (n = 313): support workers or administrative workers. | No significant differences in TSH annual mean levels between night shift workers (3.27 mIU/L) and non-night shift workers (2.98 mIU/L). Significant increase in TSH levels of night shift workers (0.303 mIU/L) compared to non-night shift workers at the GEE analysis adjusted for age and department. Risk of subclinical hypothyroidism (TSH levels of 4.5 ≥ mIU/L): 1.399-fold higher risk in night shift workers compared to their non-night shift workers. | Satisfactory |
| Sathyanarayana et al. [27] | Bangalore, India | Cross sectional | Healthcare sector Doctors, nurses, technicians and support staff (n. 40). Day-shift workers (n = 20; mean age 34.25 ± 8.16 years); Night shift workers (n = 20; mean age 27.50 ± 5.14 years) | Hormonal changes | Night shift-work | Working schedule Day-shift workers: schedule timings between 8:00 and 17:00 Night shift workers: schedule timings between 20:00 and 8:00 Night shift workers had ≥3 night shifts per week and monthly night shift working hours of 60–90 h. Participants had a minimum of 1 year working experience in these schedules. Biological sampling Blood sample to determine the serum TSH was collected after 10–12 h of fasting between 7:30 and 8:30 am. | Significant increase in TSH levels in night shift workers (3.11 ± 1.81 mIU/L) compared to day-shift workers (2.04 ± 0.8 mIU/L). | Unsatisfactory |
| Korompeli et al. [23] | Athens, Greece | Cross sectional | Healthcare sector Total intensive care unit nurses (n = 32; mean age: 36.7 ± 1.2 years); Rotating shift group (n = 25; 13 females, 12 males); Exclusively morning-shift group (n = 7, 2 males, 5 females). | Hormonal changes | Irregular rotating shifts, including night shifts | Working schedule Rotating shift group (morning: 7 am–3 pm; evening: 3–11 pm; night shifts 11 pm–7 am); Exclusively morning shift group (7 am–3 pm); All nurses had been working for more than 3 years in an intensive care unit. Biological sampling Blood samples were collected from each participant at the beginning and at the end of the shift. | Significant decrease in TSH concentrations from the beginning and the end of the shift for rotating workers (2.34 ± 0.32 vs 1.78 ± 0.27 mIU/L). No significant differences in morning group (1.67 ± 0.44 vs. 1.36 ± 0.30 mIU/L) Significant reduction of T3 values from the beginning and the end of the shift for morning workers (1.17 ± 0.04 vs. 1.05 ± 0.05 ng/mL), non-significant differences in rotating workers (1.06 ± 0.04 vs 1.01 ± 0.04 ng/mL). Significant increase in the mean T4 concentrations from the beginning and the end of the shift in rotating workers (7.10 ± 0.25 vs. 7.35 ± 0.22 μg/dL, respectively); non-significant differences in morning workers (7.40 ± 0.25 vs. 7.26 ± 0.26 μg/dL, respectively). | Unsatisfactory |
| Weibel and Brandenberger [29] | Strasbourg, France | Cross sectional | Night workers (n = 11 males; age range: 24–25 years); Day-active workers (n = 8; age range 23–32 years) | Hormonal changes | Night shift-work | Working schedule Workers had been working on night shift (4–5 consecutive night shifts per week) for at least 2 years; No naps were allowed during the night work. Biological sampling Workers were studied during their usual 24-h sleep-wake cycle following day sleep from 7 am–15 pm in night shift workers and the nocturnal sleep 23 pm–7 am sleep period in daytime workers. | Hormonal profile during usual sleep time: TSH concentrations plateaued during night sleep (23:00–6:00 h) among day-active subjects and declined progressively during day sleep (07:00–15:00 h) among night shift workers, with a significant different slope between the 2 groups (Spearman’s R: −0.07 ± 0.17 vs. −0.57 ± 0.09, respectively). Hormonal profiles during usual work time: TSH were low among day-active subjects (09:00–17:00 h), but increased among night workers (22:00–6:00 h), with significant different slope between the 2 groups (Spearman’s R: −0.59 ± 0.11 vs. 0.74 ± 0.05, respectively) | Unsatisfactory |
| Harbeck et al. [22] | Luebeck, Germany | Prospective crossover study | Healthcare sector Physicians working in department of internal medicine (n = 20; 11 females, 9 males; median age: 32 years) | Hormonal changes | Twenty-four hours on-call-occupational shift | Working schedule Each physician completed a 24-h control period including a regular 8 h non-on-call-shift and a 24-h on-call-shift 2–4 weeks later. Working experience ranged from 1–2 to >than 4 years. Biological sampling Biochemical parameters were assessed at 8:00 am prior to a normal working day and, 2–4 weeks later, at 8:00 am of the morning following a 24-h on-call shift in internal medicine. | Significant increase in TSH levels were detected the morning after the 24-h on-call-duty (2.9 ± 1.7 mIU/L) compared to the TSH levels determined in the same group before a normal working day (2.0 ± 0.8 mIU/L). | Unsatisfactory |
| Kuetting et al. [24] | Bonn, Germany | Prospective cohort study | Healthcare sector Radiology residents (n = 20; 19 males, 1 female; mean age: 31.6 ± 2.1 years); Controls from the same group (n = 10) | Hormonal changes | Twenty-four hours on-call-occupational shift | Working schedule Twenty-four-hour shift with an average of 3 h of sleep. Biological sampling Examinations were performed before and after the 24-h on-call-duty and before and after a normal day of 9 h of work in controls (at least 7 days from the 24-h on-call-duties). | Significant increase in TSH (2.44 ± 0.9 mIU/L), FT3 (3.4 ± 0.4 pg/mL) and FT4 (1.0 ± 0.1 ng/dL) values following 24-h on-call-duty, in comparison to concentrations detected before the on-call-shift (TSH: 1.54 ± 0.5 mIU/L; FT3: 3.1 ± 0.20 pg/mL; FT4: 0.92 ± 0.1 ng/dL). No significant changes in TSH (1.53 ± 0.3 vs. 1.516 ± 0.4 mIU/L), FT3 (3.03 ± 0.2 vs. 3.12 ± 0.19 pg/mL) and FT4 (0.97 ± 0.01 vs. 0.95 ± 0.05 ng/dL) before and after a regular day of work (9 h) and a regular sleep (at least 6 h). | Unsatisfactory |
| Malmberg et al. [25] | Lund, Sweden | Cross sectional | Healthcare sector Anesthesiologists (n = 19; females: 39%; median age 42 years; median years’ experience on night call: 9 years); Pediatricians and ear-nose-throat surgeons (n = 18; females: 59%; median age 39 years; median years’ experience on night call: 10 years) | Hormonal changes | Night-call duty | Working schedule Night-call duty started at approximately 16:00 h and lasted for about 16 h. Anesthesiologists and pediatricians: separate night call weeks (2–3 nights) every fourth to sixth week. Ear-nose-throat surgeons: single nights on call every second or third week. Biological sampling Blood samples were collected on an ordinary workday, 1 and 3 days after work on a night call. | Significant reduction in mean TSH levels (26%–28%) one day after night-call duty (anesthesiologists: 1.37–95% CI 1.04–1.70; pediatricians and ENT surgeons: 1.43–95% CI 1.06–1.79 mIU/L) compared to ordinary days of work (anesthesiologists: 1.83-CI 95% 1.51–2.14; pediatricians and ENT surgeons: 1.92-CI95% 1.56–2.29 mIU/L). Three day after the night-call duty: anesthesiologists: 1.52-CI 95% 1.19–1.86 mIU/L; pediatricians and ear-nose-throat surgeons: 2.03-CI95% 1.67–2.40 mIU/L. | Satisfactory |
| Shaker et al. [28] | Cairo, Egypt | Cross sectional | Industrial sector Total male investigated workers (n = 99; mean age: 46.8 ± 10.0 years); Fixed morning-shift (n = 36); Fixed afternoon-shift (n = 19); Fixed night- shift (n = 44) (median age range 25–60) | Hormonal changes | Shift-work | Working schedule The factory was engaged in the manufactures of railway sleepers, rubber, pottery, and cement bricks. Workers were employed in fixed morning, afternoon or night shifts for at least 2 years. Biological sampling Blood samples were collected in the morning at the workplace. | Significant increase in median TSH values in night shift workers (2.8 mIU/L; IQR 2.2–3.6 mIU/L) than in morning (0.7 mIU/L; IQR 0.5–0.9 mIU/L) and afternoon-shift workers (1.4 mIU/L; IQR 1.2–1.7 mIU/L). | Satisfactory |
| Attarchi et al. [20] | Tehran, Iran | Cross sectional | Industrial sector Total female workers in packaging units of a pharmaceutical industry (n = 406; mean age 31.3 years); Shift workers (n = 113 years); Non-shift-workers (n = 293). | Hormonal changes | Shift-work | Working schedule No data are available on shift-work organization; All women working in the packaging units of the factory had at least one year of work experience. Biological sampling Blood samples for hormone analysis were collected on the third day of menstruation. | No significant differences in mean TSH levels were detected between shift workers (2.98 ± 0.70 mIU/L) and non-shift workers (2.89 ± 0.98 mIU/L). | Satisfactory |
| Burdelak et al. [30] | Lodz, Poland | Cross sectional | Female nurses and midwives (n = 725; age range: 40–60 years); Rotating night shift workers (n = 354; median age: 48 years); Day workers (n = 371; median age: 50 years) | Thyroid diseases | Shift-work and night shift-work | Working schedule Women working rotating on night shifts had been working at night for an average of 25.4 years; Women working days only, but who had had a history of night shift-work, had, on average, worked for 12 years on night shifts. Out of daily workers, the majority (83%) had changed to day work more than 5 years before their recruitment into the study. | No significant difference in the prevalence of thyroid diseases between night shift and daily workers. Thyroid disease prevalence in the total investigated population: 21.2%; in shift workers: 22.6%; in day workers. 20%. The risk of thyroid disease was higher in ≥15 year night shift-workers compared to <15 year night shift workers (OR = 1.97; 95% CI: 1.21–3.20) | Satisfactory |
| Magrini et al. [31] | Rome, Italy | Cross sectional | Workers in a large teaching hospital (n = 642); Shift workers (n. 220; 81 females, 129 males, radiation exposed n = 81; mean age: 32.67 ± 7.29 years); Day-time workers (n = 422; 222 females, 200 males; radiation exposed 95; mean age: 40.17 ± 11.32) | Thyroid diseases | Shift work | Working schedule Day time workers: shift from 6 AM to 6 PM; Shift workers: involved in rotating shifts, including night shifts. | Significant increase in subclinical autoimmune hypothyroidism prevalence in shift workers (7.7%) and in day-time workers (3.8%). OR for shift workers: 2.26 (CI 95% 1.02–4.98) Significant higher prevalence of abnormal anti-TPO antibodies level in shift workers (13.6%) than in day-time workers (8.6%). No significant differences in hypothyroidism and hyperthyroidism unrelated to altered anti-TPO antibodies in shift- and day-time workers (5.45% vs 2.10%; 2.84% vs 1.36%, for hypo and hyperthyroidism, respectively). | Good |
| Rizza et al. [32] | Rome, Italy | Retrospective cohort study | Healthcare sector Workers employed in a teaching hospital (n = 299); Rotating night shift workers (n = 160; 51 males; mean age 38.7 ± 9.4 years); Day-workers (n = 139; 39 males; mean age 36.5 ± 7.9 years). | Thyroid diseases | Shift-work | Working schedule Rotating night shift workers: shift schedule of four up to seven 12 h nights per month, followed by 2 days off; mean length of employment in night shifts: 9 ± 4 years; Day workers: had never worked night shifts. | No significant differences in TSH, FT3, FT4 and anti-TPO levels between rotating night and day- workers. Night shift work was statistically related to thyroid nodules (odds ratio: 1.78; 95%CI 1.09–3.23). Nodule size and the percentage of multiple nodules were similar in both groups. | Satisfactory |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leso, V.; Vetrani, I.; Sicignano, A.; Romano, R.; Iavicoli, I. The Impact of Shift-Work and Night Shift-Work on Thyroid: A Systematic Review. Int. J. Environ. Res. Public Health 2020, 17, 1527. https://doi.org/10.3390/ijerph17051527
Leso V, Vetrani I, Sicignano A, Romano R, Iavicoli I. The Impact of Shift-Work and Night Shift-Work on Thyroid: A Systematic Review. International Journal of Environmental Research and Public Health. 2020; 17(5):1527. https://doi.org/10.3390/ijerph17051527
Chicago/Turabian StyleLeso, Veruscka, Ilaria Vetrani, Alessandra Sicignano, Rosaria Romano, and Ivo Iavicoli. 2020. "The Impact of Shift-Work and Night Shift-Work on Thyroid: A Systematic Review" International Journal of Environmental Research and Public Health 17, no. 5: 1527. https://doi.org/10.3390/ijerph17051527
APA StyleLeso, V., Vetrani, I., Sicignano, A., Romano, R., & Iavicoli, I. (2020). The Impact of Shift-Work and Night Shift-Work on Thyroid: A Systematic Review. International Journal of Environmental Research and Public Health, 17(5), 1527. https://doi.org/10.3390/ijerph17051527







