Setting Priorities to Inform Assessment of Care Homes’ Readiness to Participate in Healthcare Innovation: A Systematic Mapping Review and Consensus Process
Abstract
1. Introduction
2. Methods
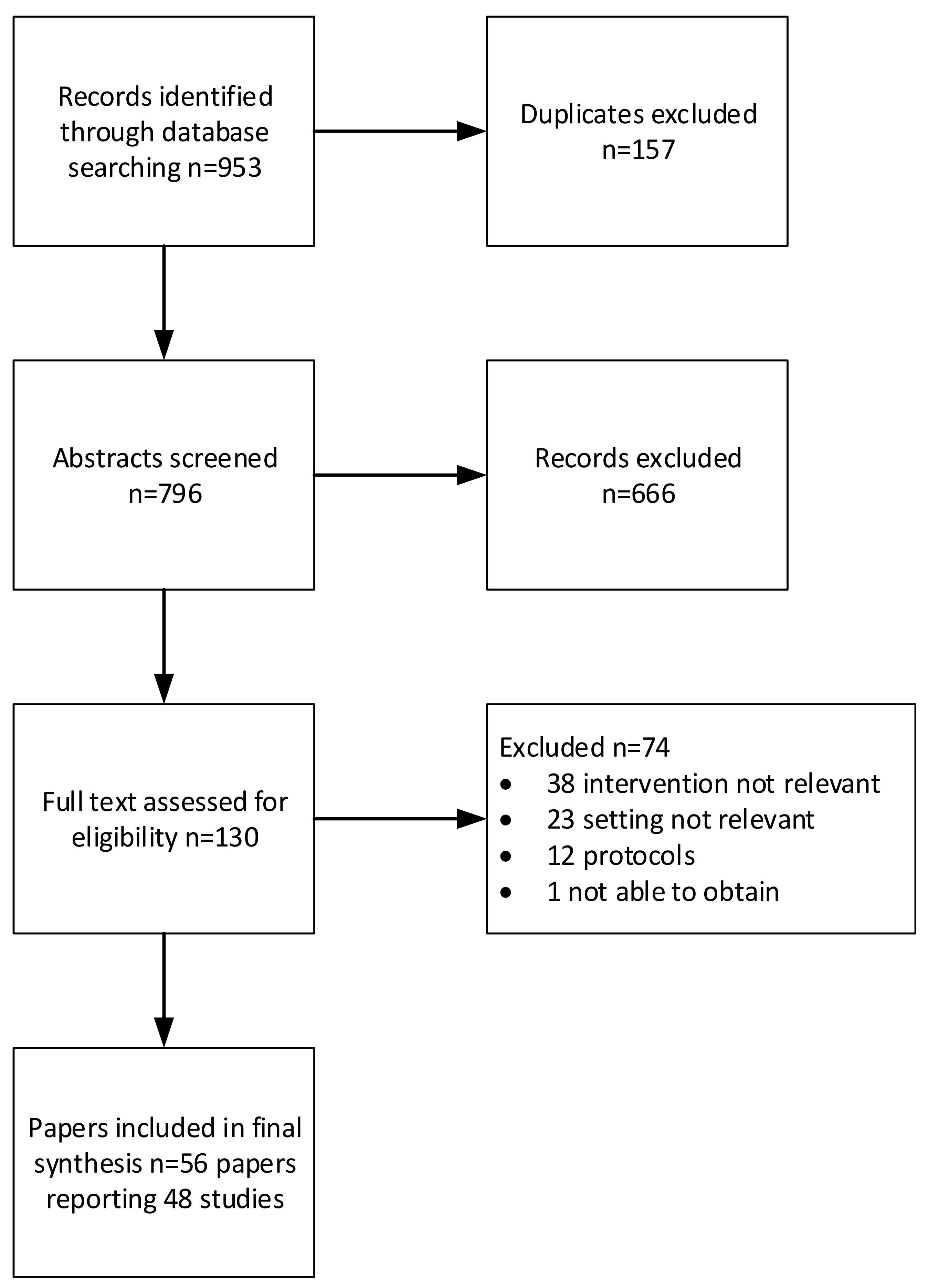
2.1. Phase 1 Systematic Mapping of The Literature
2.1.1. Inclusion Criteria
2.1.2. Search Strategy
2.1.3. Data Extraction and Analysis
2.2. Phase 2 Consensus Workshops
3. Results
3.1. Systematic Mapping
3.1.1. Leadership
3.1.2. Culture
3.1.3. Evaluation
3.1.4. Social Capital
3.1.5. Informal and Formal Interactions
3.1.6. Structural/Electronic Resources
3.1.7. Organisational Slack
3.2. Consensus Workshops
4. Discussion
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lee, S.K.; Posthauer, M.E.; Dorner, B.; Redovian, V.; Maloney, M.J. Pressure Ulcer Healing with a Concentrated, Fortified, Collagen Protein Hydrolysate Supplement: A randomized Controlled Trial. Adv Ski Wound Care. 2006, 19, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Iliffe, S.; Davies, S.L.; Gordon, A.L.; Schneider, J.; Dening, T.; Bowman, C.; Gage, H.; Martin, F.C.; Gladman, J.R.; Victor, C.; et al. Provision of NHS generalist and specialist services to care homes in England: Review of surveys. Prim. Health Care Res. Dev. 2016, 17, 122–137. [Google Scholar] [CrossRef] [PubMed]
- CQC. The State of Adult Social Care Services 2014 to Findings from CQC’s Initial Programme of Comprehensive Inspections in Adult Social Care. 2017. Available online: https://www.cqc.org.uk/sites/default/files/20170703_ASC_end_of_programme_FINAL2.pdf (accessed on 28 August 2019).
- Gordon, A.; Logan, P.; Jones, R.; Forrester-Paton, C.; Mamo, J.; Gladman, J.; Medical Crises in Older People Study Group. A systematic mapping review of Randomized Controlled Trials (RCTs) in care homes. BMC Geriatr. 2012, 12, 31. [Google Scholar] [CrossRef] [PubMed]
- Morley, J.E.; Caplan, G.; Cesari, M.; Dong, B.; Flaherty, J.H.; Grossberg, G.T.; Holmerova, I.; Katz, P.R.; Koopmans, R.; Little, M.O.; et al. International Survey of Nursing Home Research Priorities. J. Am. Med. Dir Assoc. 2014, 15, 309–312. [Google Scholar] [CrossRef]
- Goodman, C.; Dening, T.; Gordon, A.L.; Davies, S.L.; Meyer, J.; Martin, F.C.; Gladman, J.R.; Bowman, C.; Victor, C.; Handley, M.; et al. Effective health care for older people living and dying in care homes: A realist review. BMC Health Serv Res. 2016, 16, 269. [Google Scholar] [CrossRef]
- Ko, M.; Wagner, L.; Spetz, J. Nursing Home Implementation of Health Information Technology: Review of the Literature Finds Inadequate Investment in Preparation, Infrastructure, and Training. Inquiry 2018, 55, 46958018778902. [Google Scholar] [CrossRef]
- Vlaeyen, E.; Stas, J.; Leysens, G.; Van der Elst, E.; Janssens, E.; Dejaeger, E.; Dobbels, F.; Milisen, K. Implementation of fall prevention in residential care facilities: A systematic review of barriers and facilitators. Int. J. Nurs. Stud. 2017, 70, 110–121. [Google Scholar] [CrossRef]
- Greenhalgh, T.; Robert, G.; Macfarlane, F.; Bate, P.K.O. Diffusion of innovations in service organizations: Systematic review and recommendationso Title. Milbank Q. 2004, 82, 581–629. [Google Scholar] [CrossRef]
- Benzer, J.K.; Charns, M.P.; Hamdan, S.; Afable, M. The role of organizational structure in readiness for change: A conceptual integration. Health Serv. Manag. Res. 2017, 30, 34–46. [Google Scholar] [CrossRef]
- Shortell, S.M. Increasing value: A research agenda for addressing the managerial and organizational challenges facing health care delivery in the United States. Med. Care Res. Rev. 2004, 61 (Suppl. 3), 12–30. [Google Scholar] [CrossRef]
- Cammer, A.; Morgan, D.; Stewart, N.; McGilton, K.; Rycroft-Malone, J.; Dopson, S.; Estabrooks, C. The Hidden Complexity of Long-Term Care: How Context Mediates Knowledge Translation and Use of Best Practices. Gerontologist 2014, 54, 1013–1023. [Google Scholar] [CrossRef] [PubMed]
- McConnell, E.S.; Corazzini, K.N.; Lekan, D.; Bailey, D.C.; Sloane, R.; Landerman, L.R.; Champagne, M.T. Diffusion of Innovation in Long-Term Care (DOI-LTC) Measurement Battery. Res. Gerontol. Nurs. 2012, 5, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Estabrooks, C.A.; Squires, J.E.; Cummings, G.G.; Birdsell, J.M.; Norton, P.G. Development and assessment of the Alberta Context Tool. BMC Health Serv. Res. 2009, 9, 234. [Google Scholar] [CrossRef] [PubMed]
- Estabrooks, C.A.; Squires, J.E.; Hayduk, L.A.; Cummings, G.G.; Norton, P.G. Advancing the argument for validity of the Alberta Context Tool with healthcare aides in residential long-term care. BMC Med. Res. Methodol. 2011, 11, 107. [Google Scholar] [CrossRef]
- Estabrooks, C.A.; Hoben, M.; Poss, J.W.; Chamberlain, S.A.; Thompson, G.N.; Silvius, J.L.; Norton, P.G. Dying in a nursing home: Treatable symptom burden and its link to modifiable features of work context. J. Am. Med. Dir. Assoc. 2015, 16, 515–520. [Google Scholar] [CrossRef]
- Hanson, L.C.; Carey, T.S.; Caprio, A.J.; Lee, T.J.; Ersek, M.; Garrett, J.; Jackman, A.; Gilliam, R.; Wessell, K.; Mitchell, S.L. Improving Decision-Making for Feeding Options in Advanced Dementia: A Randomized, Controlled Trial. J. Am. Geriatr. Soc. 2011, 59, 2009–2016. [Google Scholar] [CrossRef]
- Cooper, I.D. What is a “mapping study?”. J. Med. Libr. Assoc. 2016, 104, 76–78. [Google Scholar] [CrossRef]
- NHS England. Five Year Forward View. Available online: http://www.england.nhs.uk/wp-content/uploads/2014/10/5yfv-web.pdf (accessed on 28 August 2019).
- NHS England. New Care Model: Vanguards-Developing a Blueprint for the Future of NHS and Care Services. Available online: https://www.england.nhs.uk/wp-content/uploads/2015/11/new_care_models.pdf (accessed on 15 March 2016).
- Squires, J.; Hayduk, L.; Hutchinson, A.; Mallick, R.; Norton, P.; Cummings, G.; Carole, A. Reliability and validity of the Alberta context tool (ACT) with professional nurses: Findings from a multi-study analysis. PLoS ONE 2015, 10, E0127405. [Google Scholar] [CrossRef]
- Rycroft-Malone, J. The PARIHS framework-A framework for guiding the implementation of evidence-based practice. J. Nurs. Care Qual. 2004, 19, 297–304. [Google Scholar] [CrossRef]
- Bartunek, J.; Murninghan, J. The nominal group technique: Expanding the basic procedure and underlying assumptions. Group Organ. Manag. 1984, 9, 417–432. [Google Scholar] [CrossRef]
- Arendts, G.; Deans, P.; Brien, K.O.; Etherton-beer, C.; Howard, K.; Lewin, G.; Sim, M. A clinical trial of nurse practitioner care in residential aged care facilities. Arch. Gerontol. Geriatr. 2018, 77, 129–132. [Google Scholar] [CrossRef] [PubMed]
- Boorsma, M.; Frijters, D.H.; Knol, D.L.; Ribbe, M.E.; Nijpels, G.; van Hout, H.P. Effects of multidisciplinary integrated care on quality of care in residential care facilities for elderly people: A cluster randomized trial. Cmaj 2011, 183, E724–E732. [Google Scholar] [CrossRef] [PubMed]
- Colon-Emeric, C.S.; Mcconnell, E.; Pinheiro, S.O.; Corazzini, K.; Porter, K.; Earp, K.M.; Landerman, L.; Beales, J.; Lipscomb, J.; Hancock, K.; et al. CONNECT for Better Fall Prevention in Nursing Homes: Results from a Pilot Intervention Study. J. Am. Geriatr. Soc. 2014, 61, 2150–2159. [Google Scholar] [CrossRef] [PubMed]
- Connolly, M.J.; Boyd, M.; Broad, J.B.; Kerse, N.; Lumley, T.; Whitehead, N.; Foster, S. The Aged Residential Care Healthcare Utilization Study (ARCHUS): A Multidisciplinary, Cluster Randomized Controlled Trial Designed to Reduce Acute Avoidable Hospitalizations From Long-Term Care Facilities. J. Am. Med. Dir. Assoc. 2015, 16, 49–55. [Google Scholar] [CrossRef]
- Crotty, M.; Halbert, J.; Rowett, D.; Giles, L.; Birks, R.; Williams, H.; Whitehead, C. An Outreach Geriatric Medication Advisory Service in Residential Aged Care: A Randomised Controlled Trial of Case Conferencing. Age Ageing 2004, 33, 612–617. [Google Scholar] [CrossRef] [PubMed]
- Davison, T.E.; Karantzas, G.; Mellor, D.; McCabe, M.P.; Mrkic, D. Staff-focused interventions to increase referrals for depression in aged care facilities: A cluster randomized controlled trial. Aging Ment. Health 2013, 17, 449–455. [Google Scholar] [CrossRef]
- De Visschere, L.; Schols, J.; van der Putten, G.J.; de Baat, C.; Vanobbergen, J. Effect evaluation of a supervised versus non-supervised implementation of an oral health care guideline in nursing homes: A cluster randomised controlled clinical trial. Gerodontology 2012, 29, e96–e106. [Google Scholar] [CrossRef]
- Dorsey, E.R.; Deuel, L.M.; Voss, T.S.; Finnigan, K.; George, B.P.; Eason, S.; Miller, D.; Reminick, J.I.; Appler, A.; Polanowicz, J.; et al. Increasing access to specialty care: A pilot, randomized controlled trial of telemedicine for Parkinson’s disease. Mov. Disord. 2010, 25, 1652–1659. [Google Scholar] [CrossRef]
- Dozeman, E.; Marwijk, H.W.J.; Van Schaik, D.J.F.; Van Smit, F.; Stek, M.L.; Horst, E.V.D.; Bohlmeijer, E.T.; Beekman, A.T. Contradictory effects for prevention of depression and anxiety in residents in homes for the elderly: A pragmatic randomized controlled trial. Int. Psychogeriatr. 2012, 24, 1242–1251. [Google Scholar] [CrossRef]
- Field, T.S.; Tjia, J.; Mazor, K.M.; Donovan, J.L.; Kanaan, A.O.; Harrold, L.R.; Reed, G.; Doherty, P.; Spenard, A.; Gurwitz, J.H. Randomized trial of a warfarin communication protocol for nursing homes: An SBAR-based approach. Am. J. Med. 2011, 124, 179. [Google Scholar] [CrossRef]
- Kinley Stone, L.; Dewey, M.; Levy, J.; Stewart, R.; McCrone, P.; Sykes, N.; Hansford, P.; Begum, A.; Hockley, J. The effect of using high facilitation when implementing the Gold Standards Framework in Care Homes programme: A cluster randomised controlled trial. Palliat. Med. 2014, 28, 1099–1109. [Google Scholar] [CrossRef]
- McSweeney, K.; Jeffreys, A.; Griffith, J.; Plakiotis, C.; Kharsas, R.; O’Connor, D.W. Specialist mental health consultation for depression in Australian aged care residents with dementia: A cluster randomized trial. Int. J. Geriatr. Psychiatry 2012, 27, 1163–1171. [Google Scholar] [CrossRef]
- Boyd, M.; Armstrong, D.; Parker, J.; Pilcher, C.; Zhou, L.; McKenzie-Green, B.; Connolly, M.J. Do gerontology nurse specialists make a difference in hospitalization of long-term care residents? Results of a randomized comparison trial. J. Am. Geriatr. Soc. 2014, 62, 1962–1967. [Google Scholar] [CrossRef]
- Meeks, S.; Van Haitsma, K.; Schoenbachler, B.; Looney, S.W. BE-ACTIV for depression in nursing homes: Primary outcomes of a randomized clinical trial. J. Gerontol. B Psychol. Sci. Soc. Sci. 2015, 70, 13–23. [Google Scholar] [CrossRef]
- Rantz, M.; Zwygart-Stauffacher, M.; Hicks, L.; Mehr, D.; Flesner, M.; Petrovski, G.F.; Madsen, R.W.; Scott-Cawiezell, J. Randomized Multilevel Intervention to Improve Outcomes of Residents in Nursing Homes in Need of Improvement. J. Am. Med. Dir. Assoc. 2012, 4, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Underwood, M.; Lamb, S.E.; Eldridge, S.; Sheehan, B.; Slowther, A.M.; Spencer, A.; Thorogood, M.; Atherton, N.; Bremner, S.A.; Devine, A.; et al. Exercise for depression in elderly residents of care homes: A cluster-randomised controlled trial. Lancet 2013, 382, 41–49. [Google Scholar] [CrossRef]
- Van der Kooij, C.H.; Droes, R.M.; de Lange, J.; Ettema, T.P.; Cools, H.J.; van Tilburg, W. The implementation of integrated emotion-oriented care: Did it actually change the attitude, skills and time spent of trained caregivers? Dement 2013, 12, 536–550. [Google Scholar] [CrossRef] [PubMed]
- Vowden, K.; Vowden, P. A pilot study on the potential of remote support to enhance wound care for nursing-home patients. J. Wound Care 2013, 22, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Wenborn, J.; Challis, D.; Head, J.; Miranda-Castillo, C.; Popham, C.; Thakur, R.; Illes, J.; Orrell, M. Providing activity for people with dementia in care homes: A cluster randomised controlled trial. Int. J. Geriatr. Psychiatry 2013, 28, 1296–1304. [Google Scholar] [CrossRef] [PubMed]
- Agar, M.; Luckett, T.; Luscombe, G.; Phillips, J.; Beattie, E.; Pond, D.; Mitchell, G.; Davidson, P.M.; Cook, J.; Brooks, D.; et al. Effects of facilitated family case conferencing for advanced dementia: A cluster randomised clinical trial. PLoS ONE 2017, 12, e0181020. [Google Scholar] [CrossRef] [PubMed]
- Ballard, C.; Orrell, M.; Sun, Y.; Moniz-Cook, E.; Stafford, J.; Whitaker, R.; Woods, B.; Corbett, A.; Banerjee, S.; Testad, I.; et al. Impact of antipsychotic review and non-pharmacological intervention on health-related quality of life in people with dementia living in care homes: WHELD—a factorial cluster randomised controlled trial. Int. J. Geriatr. Psychiatry 2017, 32, 1094–1103. [Google Scholar] [CrossRef] [PubMed]
- Livingston, G.; Barber, J.; Marston, L.; Stringer, A.; Panca, M.; Hunter, R.; Cooper, C.; Laybourne, A.; La Frenais, F.; Reeves, S.; et al. Clinical and cost-effectiveness of the Managing Agitation and Raising Quality of Life (MARQUE) intervention for agitation in people with dementia in care homes: A single-blind, cluster-randomised controlled trial. Lancet Psychiatry 2019, 6, 293–304. [Google Scholar] [CrossRef]
- Overbeek, A.; Korfage, I.J.; Jabbarian, L.J.; Billekens, P.; Hammes, B.J.; Polinder, S.; Severijnen, J.; Swart, S.J.; Witkamp, F.E.; van der Heide, A.; et al. Advance Care Planning in Frail Older Adults: A Cluster Randomized Controlled Trial. J. Am. Geriatr. Soc. 2018, 66, 1089–1095. [Google Scholar] [CrossRef] [PubMed]
- Beer, C.; Horner, B.; Flicker, L.; Scherer, S.; Lautenschlager, N.T.; Bretland, N.; Flett, P.; Schaper, F.; Almeida, O.P. A cluster-randomised trial of staff education to improve the quality of life of people with dementia living in residential care: The DIRECT study. PLoS ONE 2011, 6, e28155. [Google Scholar] [CrossRef] [PubMed]
- Palmer, J.A.; Mor, V.; Volandes, A.E.; McCreedy, E.; Loomer, L.; Carter, P.; Dvorchak, F.; Mitchell, S.L. A dynamic application of PRECIS-2 to evaluate implementation in a pragmatic, cluster randomized clinical trial in two nursing home systems. Trials 2018, 19, 453. [Google Scholar] [CrossRef] [PubMed]
- Surr, C.A.; Holloway, I.; Walwyn, R.E.A.; Griffiths, A.W.; Meads, D.; Kelley, R.; Martin, A.; McLellan, V.; Ballard, C.; Fossey, J.; et al. Dementia Care Mapping TM to reduce agitation in care home residents with dementia: The DCM TM EPIC cluster randomised controlled trial. Health Technol. Assess. 2019, 2019, 1–239. [Google Scholar]
- Husebo, B.S.; Ballard, C.; Aarsland, D.; Selbaek, G.; Slettebo, D.D.; Gulla, C.; Aasmul, I.; Habiger, T.; Elvegaard, T.; Testad, I.; et al. The Effect of a Multicomponent Intervention on Quality of Life in Residents of Nursing Homes: A Randomized Controlled Trial (COSMOS). J. Am. Med. Dir. Assoc. 2019, 20, 330–339. [Google Scholar] [CrossRef]
- Brazil, K.; Carter, G.; Cardwell, C.; Clarke, M.; Hudson, P.; Froggatt, K.; McLaughlin, D.; Passmore, P.; Kernohan, W.G. Effectiveness of advance care planning with family carers in dementia nursing homes: A paired cluster randomized controlled trial. Palliat. Med. 2018, 32, 603–612. [Google Scholar] [CrossRef]
- Beeckman, D.; Clays, E.; Hecke, A.; Vanderwee, K.; Schoonhoven, L.; Verhaeghe, S. A multi-faceted tailored strategy to implement an electronic clinical decision support system for pressure ulcer prevention in nursing homes: A two-armed randomized controlled trial. Int. J. Nurs. Stud. 2013, 50, 475–486. [Google Scholar] [CrossRef]
- Blekken, L.E.; Nakrem, S.; Gjeilo, K.H.; Norton, C.; Morkved, S.; Vinsnes, A.G. Feasibility, acceptability, and adherence of two educational programs for care staff concerning nursing home patients’ fecal incontinence: A pilot study preceding a cluster-randomized controlled trial. Implement. Sci. 2015, 10, 72. [Google Scholar] [CrossRef]
- Brodaty, H.; Low, L.F.; Liu, Z.; Fletcher, J.; Roast, J.; Goodenough, B.; Chenoweth, L. Successful ingredients in the SMILE study: Resident, staff, and management factors influence the effects of humor therapy in residential aged care. Am. J. Geriatr. Psychiatry 2014, 22, 1427–1437. [Google Scholar] [CrossRef] [PubMed]
- Brooker, D.J.; Latham, I.; Evans, S.C.; Jacobson, N.; Perry, W.; Bray, J.; Ballard, C.; Fossey, J.; Pickett, J. FITS into practice: Translating research into practice in reducing the use of anti-psychotic medication for people with dementia living in care homes. Aging Ment. Health 2015, 7863, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Chami, K.; Gavazzi, G.; Bar-Hen, A.; Carrat, F.; de Wazières, B.; Lejeune, B.; Armand, N.; Rainfray, M.; Hajjar, J.; Piette, F.; et al. A Short-Term, Multicomponent Infection Control Program in Nursing Homes: A Cluster Randomized Controlled Trial. J. Am. Med. Dir. Assoc. 2012, 13, 569. [Google Scholar] [CrossRef] [PubMed]
- Chi, I.; Law, B.V.; Leung, A.C.; Liu, C.P.; Yeoh, C.S.; Cheng, Y.H.; Ma, A.M.; Kwan, C.K.; Lai, C. Residential Assessment Instrument 2.0 in care planning for residents in nursing homes. Hong Kong Med. J. 2010, 16 (Suppl. 3), 29–33. [Google Scholar] [PubMed]
- Stern, A.; Mitsakakis, N.; Paulden, M.; Alibhai, S.; Wong, J.; Tomlinson, G.; Brooker, A.S.; Krahn, M.; Zwarenstein, M. Pressure ulcer multidisciplinary teams via telemedicine: A pragmatic cluster randomized stepped wedge trial in long term care. BMC Health Serv. Res. 2014, 14, 83. [Google Scholar] [CrossRef] [PubMed]
- Anderson, K.; Bird, M.; Macpherson, S.; McDonough, V.; Davis, T. Findings from a pilot investigation of the effectiveness of a snoezelen room in residential care: Should we be engaging with our residents more? Geriatr. Nurs. 2011, 32, 166–177. [Google Scholar] [CrossRef]
- Close, H.; Hancock, H.; Mason, J.M.; Murphy, J.J.; Fuat, A.; de Belder, M.; Hungin, A.P. “It’s Somebody else’s responsibility”-Perceptions of general practitioners, heart failure nurses, care home staff, and residents towards heart failure diagnosis and management for older people in long-term care: A qualitative interview study. BMC Geriatr. 2013, 13, 69. [Google Scholar] [CrossRef]
- Cohen-Mansfield, J.; Thein, K.; Marx, M.S.; Dakheel-Ali, M. What are the barriers to performing nonpharmalogical interventions for Behavioral symptoms in the nursing home? J. Am. Med. Dir. Assoc. 2012, 100, 130–134. [Google Scholar]
- Stein-Parbury, J.; Chenoweth, L.; Jeon, Y.H.; Brodaty, H.; Haas, M.; Norman, R. Implementing Person-Centered Care in Residential Dementia Care. Clin. Gerontol. 2012, 35, 404–424. [Google Scholar] [CrossRef]
- Beer, C.; Lowry, R.; Horner, B.; Almeida, O.P.; Scherer, S.; Lautenschlager, N.T.; Bretland, N.; Flett, P.; Schaper, F.; Flicker, L. Development and evaluation of an educational intervention for general practitioners and staff caring for people with dementia living in residential facilities. Int. Psychogeriatr. 2011, 23, 221–229. [Google Scholar] [CrossRef]
- Fossey, J.; Garrod, L.; Tolbol Froiland, C.; Ballard, C.; Lawrence, V.; Testad, I. What influences the sustainability of an effective psychosocial intervention for people with dementia living in care homes? A 9 to 12-month follow-up of the perceptions of staff in care homes involved in the WHELD randomised controlled trail. Int. J. Geriatr. Psychiatry 2019, 34, 674–682. [Google Scholar] [CrossRef] [PubMed]
- Fossey, J.; Garrod, L.; Lawrence, V.; Testad, I.; Stafford, J.; Murray, J. “We should see her like part of the team”: An investigation into care home staff’s experiences of being part of an RCT of a complex psychosocial intervention. Aging Ment. Health 2018, 2018, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Fossey, J.; Garrod, L.; Guzman, A.; Testad, I. A qualitative analysis of trainer/coach experiences of changing care home practice in the Well-being and Health in Dementia randomised control trial. Dementia 2018, 19, 237–252. [Google Scholar] [CrossRef] [PubMed]
- Colon-Emeric, C.S.; Pinheiro, S.O.; Anderson, R.A.; Porter, K.; McConnell, E.; Corazzini, K.; Hancock, K.; Lipscomb, J.; Beales, J.; Simpson, K.M. Connecting the learners: Improving uptake of a nursing home educational program by focusing on staff interactions. Gerontologist 2014, 54, 446–459. [Google Scholar] [CrossRef] [PubMed]
- Ellard, D.R.; Thorogood, M.; Underwood, M.; Seale, C.; Taylor, S.J. Whole home exercise intervention for depression in older care home residents (the OPERA study): A process evaluation. BMC Med. 2014, 12, 1. [Google Scholar] [CrossRef] [PubMed]
- Gage, H.; Dickinson, A.; Victor, C.; Williams, P.; Cheynel, J.; Davies, S.L.; Iliffe, S.; Froggatt, K.; Martin, W.; Goodman, C. Integrated working between residential care homes and primary care: A survey of care homes in England. BMC Geriatr. 2012, 12, 71. [Google Scholar] [CrossRef] [PubMed]
- Greenspan, S.L.; Nace, M.D.; Perera, S.F.M.; Ferchak, M.; Fiorito, G.; Medich, D.; Zukowski, K.; Adams, D.; Lee, C.; Saul, M. Lessons Learned from an Osteoporosis Clinical Trial in Frail Long Term Care Residents. Clin. Trials 2012, 100, 130–134. [Google Scholar] [CrossRef]
- Hall, S.; Longhurst, S.; Higginson, I.J. Challenges to conducting research with older people living in nursing homes. BMC Geriatr. 2009, 9, 38. [Google Scholar] [CrossRef]
- Innis, J.; Berta, W. Routines for change: How managers can use absorptive capacity to adopt and implement evidence-based practice. J. Nurs. Manag. 2016. [Google Scholar] [CrossRef]
- Oye, C.; Mekki, T.E.; Jacobsen, F.F.; Førland, O. Facilitating change from a distance-A story of success? A discussion on leaders’ styles in facilitating change in four nursing homes in Norway. J. Nurs. Manag. 2016, 24, 745–754. [Google Scholar] [CrossRef]
- Simpson, K.M.; Porter, K.; McConnell, E.S.; Colon-Emeric, C.; Daily, K.A.; Stalzer, A.; Anderson, R.A. Tool for evaluating research implementation challenges: A sense-making protocol for addressing implementation challenges in complex research settings. Implement. Sci. 2013, 8, 2. [Google Scholar] [CrossRef] [PubMed]
- Van Ness, P.H.; Peduzzi, P.N.; Quagliarello, V.J. Efficacy and effectiveness as aspects of cluster randomized trials with nursing home residents: Methodological insights from a pneumonia prevention trial. Contemp. Clin. Trials 2012, 33, 1124–1131. [Google Scholar] [CrossRef]
- Shepherd, V.; Nuttall, J.; Hood, K.; Butler, C.C. Setting up a clinical trial in care homes: Challenges encountered and recommendations for future research practice. BMC Res. Notes 2015, 8, 306. [Google Scholar] [CrossRef] [PubMed]
- Aasmul, I.; Husebo, B.S.; Flo, E. Description of an advance care planning intervention in nursing homes: Outcomes of the process evaluation. BMC Geriatr. 2018, 18, 26. [Google Scholar] [CrossRef] [PubMed]
- Luckett, T.; Chenoweth, L.; Phillips, J.; Brooks, D.; Cook, J.; Mitchell, G.; Pond, D.; Davidson, P.M.; Beattie, E.; Luscombe, G.; et al. A facilitated approach to family case conferencing for people with advanced dementia living in nursing homes: Perceptions of palliative care planning coordinators and other health professionals in the IDEAL study. Int. Psychogeriatr. 2017, 29, 1713–1722. [Google Scholar] [CrossRef] [PubMed]
- Poot, A.J.; de Waard, C.S.; Wind, A.W.; Caljouw, M.A.A.; Gussekloo, J. A Structured Process Description of a Pragmatic Implementation Project: Improving Integrated Care for Older Persons in Residential Care Homes. Inquiry 2017, 54, 0046958017737906. [Google Scholar] [CrossRef] [PubMed]
- Loudon, K.; Treweek, S.; Sullivan, F.; Donnan, P.; Thorpe, K.E.; Zwarenstein, M. The PRECIS-2 tool: Designing trials that are fit for purpose. BMJ 2015, 350, h2147. [Google Scholar] [CrossRef] [PubMed]
- Poot, A.J.; Caljouw, M.A.A.; Waard, C.S.; de Wind, A.W.; Gussekloo, J. Satisfaction in Older Persons and General Practitioners during the Implementation of Integrated Care. PLoS ONE 2016, 11, e0164536. [Google Scholar] [CrossRef]
- Chaudoir, S.R.; Dugan, A.G.; Barr, C.H.I. Measuring factors affecting implementation of health innovations: A systematic review of structural, organizational, provider, patient, and innovation level measures. Implement. Sci. 2013, 8, 22. [Google Scholar] [CrossRef]
- Marshall, M.; de Silva, D.; Cruickshank, L.; Shand, J.; Wei, L.; Anderson, J. What we know about designing an effective improvement intervention (but too often fail to put into practice). BMJ Qual. Saf. 2017, 26, 578–582. [Google Scholar] [CrossRef]
- Goodman, C.; Davies, S.L.; Gordon, A.L.; Dening, T.; Gage, H.; Meyer, J.; Schneider, J.; Bell, B.; Jordan, J.; Martin, F.; et al. Optimal NHS service delivery to care homes: A realist evaluation of the features and mechanisms that support effective working for the continuing care of older people in residential settings. Health Serv. Deliv. Res. 2017, 5, 1–204. [Google Scholar] [CrossRef] [PubMed]
- Marshall, M.; Pagel, C.; French, C.; Utley, M.; Allwood, D.; Fulop, N.; Pope, C.; Banks, V.; Goldmann, A. Moving improvement research closer to practice: The Researcher-in-Residence model. BMJ Qual. Saf. 2014, 23, 801–805. [Google Scholar] [CrossRef] [PubMed]
| PubMed. |
|---|
| “nursing home” OR “residential facilities” OR “homes for the aged” (MESH) OR nursing homes (TI/AB], care home [TI/AB] OR residential care [TI/AB] |
| AND “randomised controlled trial” OR “randomized controlled trial” (MESH) |
| CINAHL |
| “nursing homes” OR “residential facilities” OR “skilled nursing facilities” |
| Workshop | Participants | |
|---|---|---|
| London workshop n=21 + 3 facilitators | Care home manager Care home representative organisation/charity NHS physician/nurse/therapist working with care homes NHS manager/commissioner Care home researchers | 3 4 6 3 5 |
| Nottingham n=14 + 2 facilitators | Care home manager Care home representative organisation/charity NHS physician/nurse/therapist working with care homes NHS commissioner/manager Care home researchers | 1 3 4 4 2 |
| Total participants | 35 |
| Type of Intervention | N of Papers | Citations |
|---|---|---|
| Integrated working between care home staff and visiting health professionals | 24 | [24,25,27,28,32,35,36,38,39,42,43,44,45,47,50,58,60,63,64,65,66,69,78,79] |
| Comprehensive assessment and care planning | 8 | [44,45,46,48,49,50,51,64,65,66,77] |
| Telehealth | 3 | [31,33,41] |
| Other type of intervention (but provides detail on organisational context) | 27 | [26,28,32,33,34,35,38,39,44,47,48,49,54,58,60,61,62,67,68,70,71,72,73,75] |
| Domain of ACT | Considered in paper (n=) | Citations |
| Leadership | 40 | [24,25,27,28,29,30,31,34,36,38,40,42,43,44,45,46,47,48,49,50,52,53,54,55,56,58,59,60,62,63,64,66,67,68,69,72,73,75,77,78] |
| Culture | 32 | [24,27,34,36,37,42,45,46,48,49,52,53,54,55,58,59,62,63,64,66,67,68,69,70,72,73,74,75,76,78] |
| Evaluation | 12 | [25,39,40,44,45,46,48,49,50,51,64,66] |
| Social capital | 13 | [25,31,49,59,64,65,66,68,69,70,72,76,77] |
| Informal interactions | 11 | [30,49,55,57,59,60,64,65,67,72,77] |
| Formal interactions | 30 | [27,28,29,30,31,34,38,42,45,46,48,49,50,51,52,55,61,63,64,65,66,68,69,70,72,73,76,77,78,79] |
| Structural/electronic resources | 22 | [25,30,31,41,42,44,45,46,48,49,50,51,52,53,55,57,61,64,65,66,72,77] |
| Organisational slack—staff | 36 | [24,26,27,29,30,34,36,38,42,45,47,48,49,50,52,53,54,56,57,58,59,60,61,63,64,65,66,67,68,73,74,75,76,77,78] |
| Organisational slack—space | 6 | [33,51,66,68,70,71] |
| Organisational slack—time | 30 | [24,27,28,29,30,34,37,38,39,45,47,49,50,51,52,53,55,56,58,59,61,65,66,68,70,71,72,73,77,78] |
| Set of Questions Combining Review Findings with the Workshop Priorities | |
|---|---|
| 1 | Does this intervention align with care home priorities? |
| 2 | What evidence is there of senior management interest and enthusiasm for this intervention at the organisational and unit level? Are they willing and able to take an ongoing leadership role in supporting the proposed change? |
| 3 | Do care home staff have enough ‘slack and flexibility’ to accommodate the change into their current workload? Will it be recognised as core to their work? |
| 4 | How is change discussed (formally and informally) in the care home setting? Who needs to be involved in decision-making about what is being proposed and how it is implemented? |
| 5 | What are the recent changes or health-related projects this care home has been involved with? |
| 6 | Is there a champion in both the care home and in the linked NHS service with protected time to facilitate change? |
| 7 | What are the pre-existing relationships between NHS services and care home staff and networks of care and support around the care home (e.g., general practitioners (GPs), visiting specialists, links with local hospital)? |
| 8 | Could the intervention appear judgmental by signalling in a negative way that the care home needs to change? |
| 9 | How well do existing care home training programmes and work schedules fit with what is proposed? |
| 10 | Will care home staff have to collect and enter new data or are they held in existing systems? |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bunn, F.; Goodman, C.; Corazzini, K.; Sharpe, R.; Handley, M.; Lynch, J.; Meyer, J.; Dening, T.; Gordon, A.L. Setting Priorities to Inform Assessment of Care Homes’ Readiness to Participate in Healthcare Innovation: A Systematic Mapping Review and Consensus Process. Int. J. Environ. Res. Public Health 2020, 17, 987. https://doi.org/10.3390/ijerph17030987
Bunn F, Goodman C, Corazzini K, Sharpe R, Handley M, Lynch J, Meyer J, Dening T, Gordon AL. Setting Priorities to Inform Assessment of Care Homes’ Readiness to Participate in Healthcare Innovation: A Systematic Mapping Review and Consensus Process. International Journal of Environmental Research and Public Health. 2020; 17(3):987. https://doi.org/10.3390/ijerph17030987
Chicago/Turabian StyleBunn, Frances, Claire Goodman, Kirsten Corazzini, Rachel Sharpe, Melanie Handley, Jennifer Lynch, Julienne Meyer, Tom Dening, and Adam L Gordon. 2020. "Setting Priorities to Inform Assessment of Care Homes’ Readiness to Participate in Healthcare Innovation: A Systematic Mapping Review and Consensus Process" International Journal of Environmental Research and Public Health 17, no. 3: 987. https://doi.org/10.3390/ijerph17030987
APA StyleBunn, F., Goodman, C., Corazzini, K., Sharpe, R., Handley, M., Lynch, J., Meyer, J., Dening, T., & Gordon, A. L. (2020). Setting Priorities to Inform Assessment of Care Homes’ Readiness to Participate in Healthcare Innovation: A Systematic Mapping Review and Consensus Process. International Journal of Environmental Research and Public Health, 17(3), 987. https://doi.org/10.3390/ijerph17030987





